Introduction
Breast cancer (BC) is an age-related disease that mainly affects elderly women (aged ≥65 years), and accounting for approximately 50% of women with newly diagnosed BC [1, 2]. The increasing cancer incidence among elderly women has influenced healthcare planning and delivery [3, 4]. Standard treatment modalities for elderly patients with BC, including surgery, chemotherapy, endocrine therapy, target therapy, and radiotherapy (RT) [5]. Prospective randomized controlled trials have reported similar survival outcomes in patients treated with breast-conservation surgery (BCS) and mastectomy (MAST) [6, 7]. However, elderly patients were largely excluded from these trials. A recent population-based study showed that a larger proportion of BCS-eligible women were treated with MAST [2]. An increased risk of comorbidities, less aggressive biological behavior, and shorter life expectancy might influence the treatment decision making for this population. Therefore, an evidence-based guide to treatment of elderly BC patients is scarce.
The anatomic American Joint Committee on Cancer (AJCC) TNM system (T, tumor; N, nodes; M, metastasis) has been widely used to assess survival outcomes and guide treatment decisions for BC worldwide [8]. Several biologic factors, including tumor grade, estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2) have been integrated into the 8th AJCC pathological prognostic staging system, which provides a better predictive performance for prognosis compared to the 7th AJCC anatomic TNM stages [9–14]. The 7th AJCC anatomic TNM stages does not predict the survival outcome accurately while incorporating biologic factors may refine the risk stratification and have more important role on decision-making of locoregional or systemic treatments. However, whether the new AJCC staging system would provide critical implications to clinicians in making well-informed decisions throughout the course of treatment not yet being validated. Therefore, the role of the pathological prognostic staging system in treatment decision-making for elderly patients with BC should be investigated.
Elderly BC patients have a higher probability of comorbidities, which increase their risk for competing non-cancer events [15]. Failure to explain competing risks in the elderly might lead to misleading conclusions in epidemiological studies or clinical trials [16]. The Kaplan-Meier method or Cox’s proportional-hazards model might not be appropriate for prognostic analyses because they consider competing events as independent censorings and overestimate the proportion of deaths due to cancer. However, the validated studies of the new AJCC pathological prognostic staging system were based on Kaplan-Meier analysis, which considered competing events as independent censorings [11–14]. The competing events are known to be very frequent in the elderly. It is crucial to analyze competitive risks when assessing the prognosis of elderly patients. Therefore, competitive risks analysis may be a more appropriate method because it takes into account the nature of the censoring and corresponds to the probability of a particular event occurring, without having to assume independence between event types [17, 18]. Research on the benefits of treatments for elderly BC patients will help identify effective treatment modalities and ultimately guide optimal treatment decisions. In light of this, we conducted a competing risks analysis to validate the prognostic effect and assess the impact of treatment decision making of the 8th AJCC pathological prognostic stages for elderly BC patients from a large contemporary population-based dataset.
Results
Patient characteristics
We identified 67699 patients in this study. Overall, patients with stage IA (78.6%) and IIA (21.4%) diseases in the 7th AJCC edition criteria were restaged in to stage IA (84.9%), IB (8.9%), and IIA (6.2%) diseases using the 8th AJCC pathological staging criteria. The patients’ characteristics are presented in Table 1. Overall, 89.2% of patients resided in the metropolitan area. Most patients in this series were ER-positive (88.4%) and HER2 negative (91.2%). In addition, 78.6% were Non-Hispanic White (NHW) (78.6%), 72.6% were infiltrating ductal carcinoma, 78.6% were T1 stage, 79.2% were well-moderately differentiated, and 77.5% were PR-positive (77.5%). Moreover, 87.3% of them were not received chemotherapy.
Table 1. Patients’ baseline characteristics.
| Variables | n | BCS alone (%) | BCS+RT (%) | MAST (%) | Pa | Pb | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 65-69 | 21683 | 2363 (16.6) | 12840 (39.2) | 6480 (31.3) | <0.001 | 0.054 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 70-74 | 17435 | 2942 (20.6) | 9139 (27.9) | 5354 (25.9) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 75-79 | 13079 | 2837 (19.9) | 6167 (18.8) | 4075 (19.7) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≥80 | 15502 | 6113 (42.9) | 4607 (14.1) | 4782 (23.1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Race/ethnicity | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-Hispanic White | 53221 | 11347 (79.6) | 26281 (80.2) | 15593 (75.4) | <0.001 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-Hispanic Black | 5038 | 1027 (7.2) | 2288 (7.0) | 1723 (8.3) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hispanic (All Races) | 4742 | 1085 (7.6) | 2136 (6.5) | 1524 (7.4) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other | 4698 | 799 (5.6) | 2048 (6.3) | 1851 (8.9) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Histological subtype | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IDC | 49182 | 10339 (72.5) | 24444 (74.6) | 14399 (69.6) | <0.001 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ILC | 6590 | 1120 (7.9) | 2999 (9.2) | 2471 (11.9) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other | 11927 | 2796 (19.6) | 5310 (16.2) | 3821 (18.5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T1 | 53210 | 11596 (81.3) | 27353 (83.5) | 14261 (68.9) | <0.001 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T2 | 14489 | 2659 (18.7) | 5400 (16.5) | 6430 (31.1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| G1 | 21812 | 5337 (37.4) | 10975 (33.5) | 5500 (26.6) | <0.001 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| G2 | 31816 | 6383 (44.8) | 15432 (47.1) | 10001 (48.3) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| G3 | 14071 | 2535 (17.8) | 6346 (19.4) | 5190 (25.1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ER status | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Negative | 7866 | 1305 (9.2) | 3476 (10.6) | 3085 (14.9) | <0.001 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive | 59833 | 12950 (90.8) | 29277 (89.4) | 17606 (85.1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PR status | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Negative | 15224 | 2825 (19.8) | 6827 (20.8) | 5572 (26.9) | <0.001 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive | 52475 | 11430 (80.2) | 25926 (79.2) | 15119 (73.1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HER2 status | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Negative | 61720 | 13127 (92.1) | 30259 (92.4) | 18334 (88.6) | <0.001 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive | 5979 | 1128 (7.9) | 2494 (7.6) | 2357 (11.4) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathological stages | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IA | 57483 | 12464 (87.4) | 28577 (87.3) | 16442 (79.5) | <0.001 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IB | 6022 | 1030 (7.2) | 2776 (8.5) | 2216 (10.7) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IIA | 4194 | 761 (5.3) | 1400 (4.3) | 2033 (9.8) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marital status | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Married | 33874 | 5949 (41.7) | 17989 (54.9) | 9936 (48.0) | <0.001 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Divorced | 7771 | 1505 (10.6) | 3870 (11.8) | 2396 (11.6) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Single | 6457 | 1370 (9.6) | 3004 (9.2) | 2083 (10.1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Widowed | 19597 | 5431 (38.1) | 7890 (24.1) | 6276 (30.3) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemotherapy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No/unknown | 59115 | 13206 (92.6) | 28303 (86.4) | 17606 (85.1) | <0.001 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 8584 | 1049 (7.4) | 4450 (13.6) | 3085 (14.9) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: BCS, breast-conservation surgery; ER, estrogen receptor; G1, well differentiated; G2, moderately differentiated; G3, poorly/undifferentiated; HER2, human epidermal growth factor receptor 2; IDC, infiltrating ductal carcinoma; ILC, infiltrating lobular carcinoma; PR, progesterone receptor; MAST, mastectomy; RT, radiotherapy; T, tumor. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| a BCS alone vs. BCS+RT vs. MAST; b BCS vs. MAST. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
In the entire cohort, 47008 (69.4%) and 20691 (30.6%) patients underwent BCS and MAST, respectively. In patients who received BCS, 30.3% (n=14255) of them treated with postoperative RT. Significant differences were found in age at diagnosis, race/ethnicity, histology, tumor size, tumor grade, ER status, PR status, HER2 status, pathological prognostic stages, marital status, and chemotherapy (Table 1). In comparison to the elderly women who underwent BCS with or without RT, the patients that underwent MAST included a higher proportion of other races (8.9% vs. 6.3-5.6%), infiltrating lobular cancers (11.9% vs. 7.9%-9.2%), T2 stage (31.1% vs. 16.5-18.7%), poorly/undifferentiated disease (25.1% vs. 17.8-19.4%), ER-negative (14.9% vs. 9.2-10.6%), PR-negative (26.9% vs. 19.8-20.8%), HER2-positive (11.4% vs. 7.6-7.9%), and chemotherapy recipients (14.9% vs. 7.4-13.6%). Patients with a higher pathological prognostic stage (stage IB and IIA) were more likely to be treated with MAST compared to those with stage IA disease. The percentages of treatments received by pathological prognostic stages are presented in Figure 1. Moreover, married patients were more likely to receive BCS and RT, and widowed patients comprised a higher proportion of recipients of BCS alone. No significant difference was found in age between BCS and MAST recipients (P=0.054).
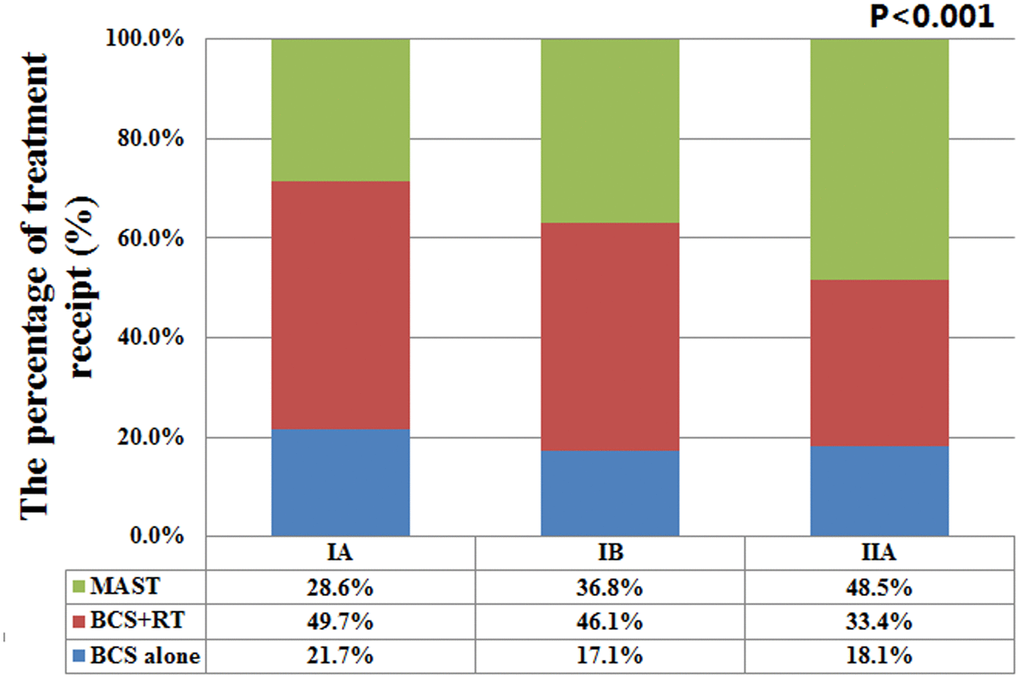
Figure 1. The percentages of three different local treatments receipt by pathological prognostic stages.
Stage migration
According to the 7th AJCC staging system, 20.7% (n=13988) of patients were restaged according to the 8th AJCC pathological staging criteria. Overall, 5.5% (n=3693) were upstaged and 15.2% (n=10295) were downstaged (Table 2). Among patients with 7th edition stage IA disease, 6.9% were upstaged to stage IB using the 8th edition criteria. Similarly, 55.0% and 16.1% of the 7th edition stage IIA patients were downstaged to stage IA and IB diseases, respectively.
Table 2. Comparison of the 7th and 8th editions of the AJCC breast cancer staging systems.
| 8th ed. stage IA | 8th ed. stage IB | 8th ed. stage IIA | Total | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 7th ed. stage IA | 49517 (93.1%) | 3693 (6.9%) | 0 | 53210 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 7th ed. stage IIA | 7966 (55.0%) | 2329 (16.1%) | 4194 (28.9%) | 14489 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total | 57483 (84.9%) | 6022 (8.9%) | 4194 (6.2%) | 67699 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviation: AJCC, American Joint Committee on Cancer. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yellow, blue, and grey boxes represent patients whose 7th edition stages were upstaged, down staged, or unchanged, respectively. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Predictors of the treatments received
We assessed the independent predictors of treatments received using binomial regression with the following variables: histology, tumor stage, pathological stage, race/ethnicity, marital status, and chemotherapy received (Table 3). Non-infiltrating ductal carcinoma, T2 stage, higher pathological stage, non-NHW race, and being unmarried were independent predictors of receipt of MAST.
Table 3. Predictors of the surgical procedure received (MAST vs. BCS).
| Variables | OR | 95%CI | P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Race/ethnicity | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-Hispanic White | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-Hispanic Black | 1.167 | 1.096-1.243 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hispanic (All Races) | 1.119 | 1.049-1.194 | 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other | 1.579 | 1.484-1.681 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Histological subtype | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IDC | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ILC | 1.400 | 1.325-1.479 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other | 1.126 | 1.077-1.176 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathological stages | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IA | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IB | 1.234 | 1.163-1.309 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IIA | 1.329 | 1.231-1.433 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marital status | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Married | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Divorced | 1.058 | 1.002-1.117 | 0.044 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Single | 1.098 | 1.036-1.165 | 0.002 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Widowed | 1.089 | 1.047-1.132 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemotherapy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No/unknown | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 1.096 | 1.040-1.154 | 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: BCS, breast-conservation surgery; CI, confidence interval; IDC, infiltrating ductal carcinoma; ILC, infiltrating lobular carcinoma; MAST, mastectomy; OR, odds ratio. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Survival and prognostic analysis
In the entire cohort, 9177 deaths occurred, but only 20.4% (n=1868) were breast-cancer related. The top five mortality rates were heart disease (25.6%), chronic obstructive pulmonary disease and related conditions (7.6%), cerebrovascular diseases (7.0%), lung and bronchus carcinomas (5.5%), and Alzheimer's disease (4.6%).
The cumulative incidence estimates of breast cancer-specific mortality (BCSM) by the 7th AJCC staging and the 8th AJCC pathological prognostic staging are presented in Figure 2. Regarding the 7th AJCC staging, the 5-year BCSM rates were 2.2% and 7.4% in patients with stages IA and IIA disease, respectively (P<0.001) (Figure 2A). Patients with a higher pathological stage had a higher cumulative incidence of BCSM. The 5-year BCSM rates were 2.2%, 6.5%, and 13.7% in patients with stages IA, IB, and IIA using the 8th edition criteria, respectively (P<0.001) (Figure 2B). The 8th AJCC staging was examined against the 7th AJCC staging using the receiver operating characteristics (ROC) curve. The area under the curve (AUC) under the ROC curve in 8th AJCC staging (AUC=0.655, 95% confidence interval [CI] 0.643-0.667) was significantly higher than that of the 7th AJCC staging (AUC=0.638, 95%CI 0.627-0.649) (P=0.026) (Figure 3). The results indicated that the 8th AJCC staging had a better predictive performance for BCSM compared to the 7th AJCC staging.
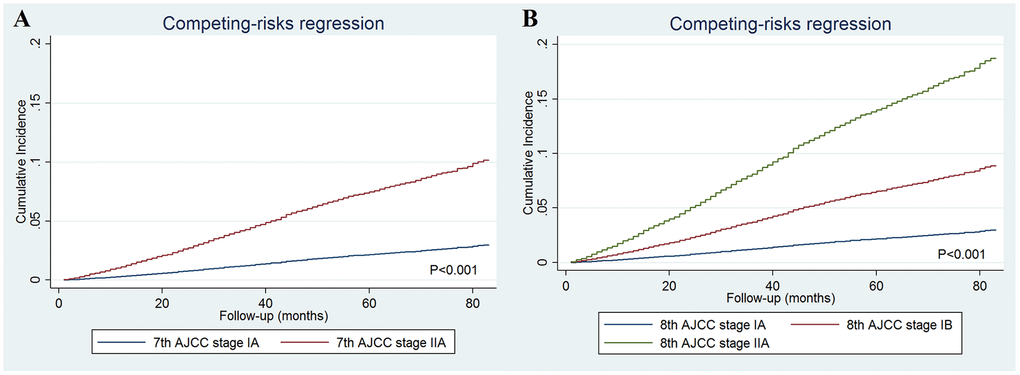
Figure 2. The cumulative incidence estimates of breast cancer-specific mortality rates by the 7th AJCC anatomic staging (A) and the 8th AJCC pathological prognostic staging (B).
We developed two Gray’s proportional sub-distribution hazards models to investigate the prognostic factors associated with BCSM. The first multivariate prognostic model included biologic factors such as tumor grade, ER, PR, and HER2 status for predicting the BCSM (Table 4). The results showed that higher tumor grade, ER-negative, and PR-negative were the independent adverse prognostic factors related to BCSM. However, HER2 status was not associated with BCSM in the multivariate prognostic analysis. The second multivariate prognostic model included the pathological prognostic staging for predicting BCSM (Table 5). The competing risks model using the Cox model framework showed that pathological prognostic staging was a significant predictor of BCSM. Using stage IA as a reference, the sub-distribution hazard ratios (sdHR) for stages IB and IIA were 2.946 (95%CI 2.598-3.341, P<0.001) and 5.908 (95%CI 5.272-6.620, P<0.001), respectively, compared to stage IA. Age at diagnosis, race/ethnicity, histology, and marital status were also the independent predictors of BCSM (Table 5).
Table 4. The multivariate prognostic analysis included biologic factors for predicting the breast cancer-specific mortality using the competing risks model.
| Variables | sdHR | 95% CI | P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 65-69 | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 70-74 | 1.287 | 1.117-1.482 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 75-79 | 1.594 | 1.379-1.843 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≥80 | 2.224 | 1.946-2.542 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Race/ethnicity | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-Hispanic White | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-Hispanic Black | 1.114 | 0.955-1.299 | 0.170 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hispanic (All Races) | 1.007 | 0.837-1.211 | 0.940 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other | 0.740 | 0.599-0.914 | 0.005 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Histological subtype | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Infiltrating ductal carcinoma | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lobular carcinoma | 0.874 | 0.731-1.046 | 0.143 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other | 0.908 | 0.797-1.034 | 0.143 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T1 | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T2 | 2.469 | 2.242-2.719 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| G1 | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| G2 | 1.613 | 1.393-1.868 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| G3 | 2.912 | 2.478-3.422 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ER status | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Negative | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive | 0.647 | 0.559-0.750 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PR status | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Negative | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive | 0.647 | 0.567-0.738 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HER2 status | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Negative | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive | 0.902 | 0.783-1.038 | 0.149 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marital status | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Married | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Divorce | 1.159 | 0.990-1.356 | 0.066 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Single | 1.265 | 1.076-1.489 | 0.005 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Widowed | 1.284 | 1.150-1.433 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: CI, confidence interval; ER, estrogen receptor; G1, well differentiated; G2, moderately differentiated; G3, poorly/undifferentiated; HER2, human epidermal growth factor receptor 2; IDC, infiltrating ductal carcinoma; ILC, infiltrating lobular carcinoma; PR, progesterone receptor; sdHR, sub-distribution hazard ratios; T, tumor. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 5. The multivariate prognostic analysis included 8th AJCC pathological prognostic stages for predicting the breast cancer-specific mortality using the competing risks model.
| Variables | sdHR | 95% CI | P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 65-69 | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 70-74 | 1.228 | 1.067-1.414 | 0.004 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 75-79 | 1.467 | 1.269-1.696 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≥80 | 1.921 | 1.676-2.203 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Race/ethnicity | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-Hispanic White | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-Hispanic Black | 1.171 | 1.005-1.366 | 0.044 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hispanic (All Races) | 1.006 | 0.837-1.209 | 0.951 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other | 0.728 | 0.589-0.900 | 0.003 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Histological subtype | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Infiltrating ductal carcinoma | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lobular carcinoma | 0.761 | 0.639-0.907 | 0.002 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other | 0.830 | 0.729-0.943 | 0.004 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pathological stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IA | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IB | 2.946 | 2.598-3.341 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IIA | 5.908 | 5.272-6.620 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marital status | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Married | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Divorce | 1.156 | 0.987-1.352 | 0.071 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Single | 1.230 | 1.045-1.447 | 0.013 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Widowed | 1.274 | 1.142-1.423 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: CI, confidence interval; sdHR, sub-distribution hazard ratios. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The effects of the treatment receipt on BCSM
After adjustment of age at diagnosis, race/ethnicity, histology, pathological stages, chemotherapy, and marital status, the results of the competing risks model indicated that local treatment procedure was an independent predictor of BCSM (Table 6). Using MAST as the reference, patients who received BCS alone (sdHR 1.003, P=0.948) had comparable BCSM compared to those treated with MAST, while patients treated with BCS and RT (sdHR 0.520, P<0.001) had significantly lower risk of BCSM than those treated with MAST (Table 6). The cumulative incidence estimates of BCSM by local treatment received are presented in Figure 4A. The rates of the 5-year BCSM were 4.9%, 4.2%, and 1.9% in patients treated with MAST, BCS alone, and BCS with RT, respectively (P<0.001).
Table 6. The multivariate prognostic analysis of predictors of breast cancer-specific mortality by local treatment receipt according to pathological prognostic stages.
| Variables | sdHR | 95% CI | P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Entire cohort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MAST | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BCS | 1.003 | 0.894-1.126 | 0.948 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BCS+RT | 0.520 | 0.464-0.583 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stage IA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MAST | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BCS | 0.858 | 0.738-0.998 | 0.047 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BCS+RT | 0.420 | 0.362-0.489 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stage IB | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MAST | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BCS | 1.269 | 0.970-1.660 | 0.083 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BCS+RT | 0.632 | 0.491-0.814 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stage IIA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MAST | 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BCS | 1.212 | 0.969-1.517 | 0.092 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BCS+RT | 0.793 | 0.638-0.986 | 0.037 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: BCS, breast-conservation surgery; CI, confidence interval; MAST, mastectomy; RT, radiotherapy; sdHR, sub-distribution hazard ratios. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
After stratification by pathological prognostic stages, similar survival difference by local treatment received was found in the subgroups of women with stages IB and IIA, adjusting for age at diagnosis, race/ethnicity, histology, chemotherapy, and marital status (Table 6). Regarding the stage IA disease, patients treated with BCS alone (sdHR 0.858, 95%CI 0.738-0.998, P=0.047) and BCS with RT (sdHR 0.420, 95%CI 0.362-0.489, P<0.001) had significantly lower risk of BCSM compared to those treated with MAST (Table 6). The cumulative incidence estimates of BCSM by local treatment receipt after stratification by pathological prognostic stages are presented in Figure 4B–3D.
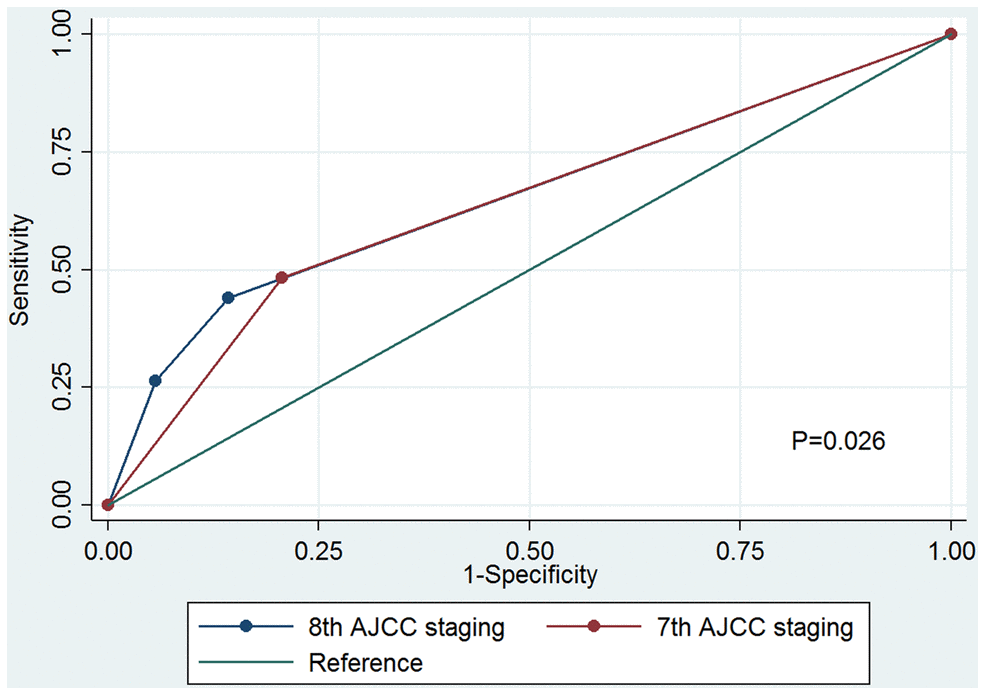
Figure 3. Receiver operating characteristics analyses for prediction of breast cancer-specific mortality between the 7th AJCC anatomic staging and the 8th AJCC pathological prognostic staging.
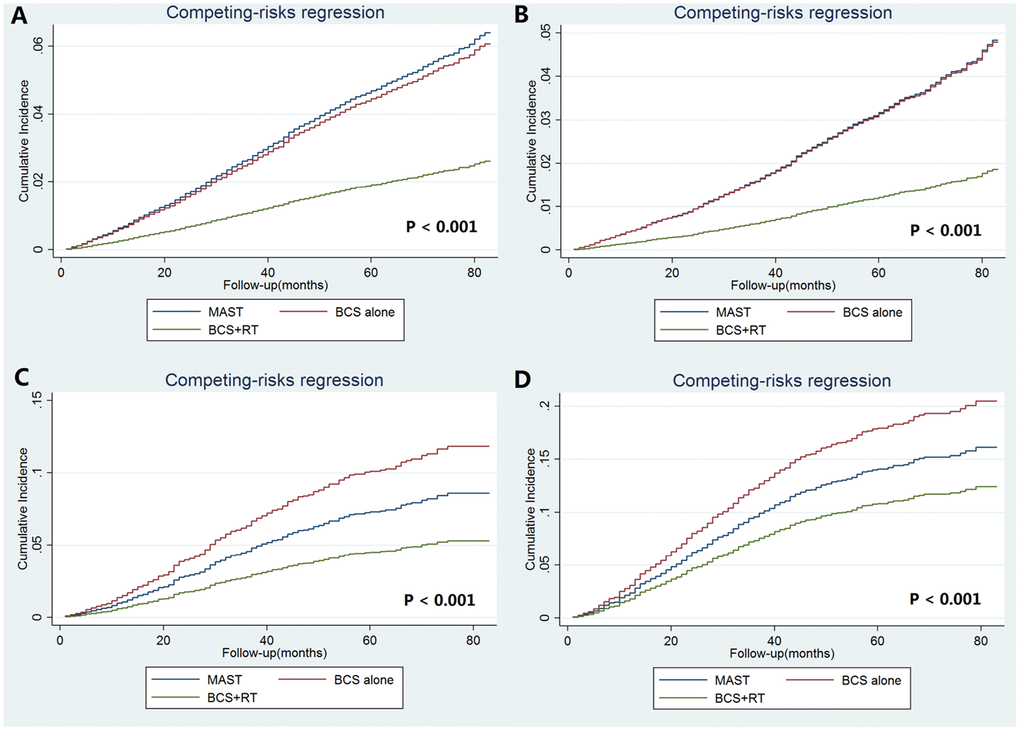
Figure 4. The cumulative incidence estimates of breast cancer-specific mortality rates by local treatment receipt according to the pathological prognostic stages (A: entire cohort; B: stage IA; C stage IB; D: stage IIA).
Finally, the results of the Fine and Gray’s proportional sub-distribution hazard model for BCSM indicated that patients treated with BCS and RT (sdHR 0.521, 95%CI 0.458-0.592, P<0.001) had significantly lower risk of BCSM compared to those treated with BCS alone. Stratified analysis replicated similar findings after stratification according to the pathological prognostic stages (stage IA: sdHR 0.490, 95%CI 0.414-0.579, P<0.001; IB: sdHR 0.497, 95%CI 0.375-0.660, P<0.001; IIA: sdHR 0.654, 95%CI 0.503-0.851, P=0.002).
Discussion
In this study, we used a population-based database to validate the effect of 8th AJCC pathological staging system on BCSM and treatment decision making in elderly women (aged ≥65 years) with T1-2N0M0 BC. Our results indicated that patients with a higher pathological stage was more likely to have a higher risk of BCSM and more likely to be treated with MAST. Patients treated with BCS and RT had significantly lower risk of BCSM compared to those treated with MAST or with BCS alone regardless of the pathological prognostic stages. The present study has two main merits: the using of the 8th edition of AJCC, and using of a competing risk model instead of Kaplan-Meier and Cox regression analyses of estimating cumulative survival in the elderly.
Given continuous innovations in diagnostic techniques and standard-of-care treatments for BC, recommendations have been made to include tumor grade, ER, PR, and HER2 status in the assessment of the disease’s prognosis and treatment decision-making [19]. The 8th edition of the AJCC Cancer Staging Manual has incorporated these biologic biomarkers into the prognostic staging groups for the first time [9, 10]. Patients aged ≥65 years presented with a higher percentage of ER-positive tumors and a lower percentage of HER2-positive tumors [20, 21]. Therefore, only 20.7% of the patients were restaged in our study, while approximately 50% of patients in all age groups were restaged in other studies [11, 22]. In this study, 71.1% of the patients in stage IIA of the 7th AJCC staging system were downstaged to IA (55.0%) and IB (16.1%) using the 8th edition criteria. We also found patients with a higher pathological stage had a higher cumulative incidence of BCSM using the competing risks model to reduce the potential competitive risk bias in the elderly. Currently, there are no published studies on the assessment of the prognostic effect of the newly pathological prognostic stages for elderly BC patients. Our study indicated that the new pathological prognostic staging system was well suited for the prognostic classification for this population, which could improve the accuracy of predicting outcomes and serve as a guide for optimal adjuvant treatment.
The new edition of the pathological prognostic staging system was developed using data retrieved from the National Cancer Data Base (NCDB) on patients who were mostly treated with appropriate multidisciplinary therapies [9, 10]. A recent study from nine European countries suggested that elderly patients should receive standard medical treatment whenever possible to maximize the benefits of modern evidence-based treatments [23]. Given the increased burden of comorbidities [24], frailty among elderly patients could lead to therapy delays or refusals, which might lead to lower survival rates [25, 26]. In this study, only 20% of deaths were from BC, while cardiovascular, cerebrovascular, and lung diseases were common causes of death in elderly patients. However, life expectancy may be underestimated in elderly BC patients due to better management of their comorbidities [27]. Therefore, although age and comorbidities should be considered in treatment decision making for elderly patients, they should never be considered as obstacles to standard treatment [27, 28].
In the current clinical practice, the 8th pathological prognostic staging alone does not guide treatment decisions that remain based on T stage, N stage, hormone receptor status, HER2 status, and multigene assays, and pathological prognostic staging is valuable in prognostic counseling of patients [29]. However, only limited studies were available regarding the locoregional treatment-decision making of the new staging. A recent study from ours included patients with stage T1-2 and one to three lymph node metastasis (restaged as IA, IB, IIA, IIB, and IIIA according to the 8th AJCC edition criteria), the results showed that postmastectomy radiotherapy was only associated with better BC-specific survival in stage IIIA disease [30]. In addition, we also investigated the treatment decision-making of the new AJCC pathological prognostic staging in T3N0 BC patients (restaged as IA, IB, IIA, IIB, and IIIA according to the 8th AJCC edition criteria), the results showed that postmastectomy radiotherapy was correlated with better BC-specific survival in stage IIB disease [31]. Therefore, the new AJCC staging may also have implications on decisions about locoregional treatment.
Although adjuvant RT may affect decision making about surgical procedures among elderly patients [32], BCS remains the main surgical procedure for this patient group (56-63% with BCS and 37-43% with MAST) [33, 34]. Prospective clinical trials have reported lower local recurrence rates but this finding does not translate into the benefit of distant recurrence-free survival or overall survival rates among elderly women with ER-positive BC [35, 36]. However, the results from the NCDB and a German clinical cohort showed improved survival with additional adjuvant RT following BCS among elderly women [37, 38]. The present study therefore, analyzed the potential role of biologic factors in treatment decision making for this patient group. Our results showed that patients with a higher pathological prognostic stage were more likely to receive MAST, (i.e., 28.6%, 36.8%, and 48.5% of the patients with stage IA, IB, and IIA BC, respectively, were treated with MAST). We also found that MAST was associated with a higher risk of BCSM compared to BCS + RT, and similar findings were replicated after stratification by the pathological prognostic stages. However, among the patients with stages IB and IIA BC, a comparable risk of BCSM was observed in recipients of MAST and BCS alone, indicating that BCS may be an alternative treatment strategy for patients with RT contraindications or those who refuse to receive RT.
Approximately one-third of the patients in this study were not treated with postoperative RT following BCS, which was similar to a previous study [39]. We further analyzed the effect of postoperative RT on BCSM according to pathological prognostic stages. Our results showed that additional postoperative RT was associated with a lower risk of BCSM compared to BCS alone. Concerns about the toxicity of RT might lead practitioners to choose MAST for elderly patients. However, recent studies showed that postoperative RT had no more toxic in elderly women than younger women [28, 40]. A previous population-based cohort study also showed the benefits of postoperative RT for elderly patients did not appear to be influenced by the presence of comorbidities [41]. According to our results, postoperative RT might be considered for elderly patients when their comorbidities are well managed [27, 40, 42].
We used a competing risks model for predicting BCSM in an elderly cohort with a high frequency of competing events. Nevertheless, several inherent limitations should be acknowledged. First, information on selection bias in the choice of treatments by providers and patients was unavailable in the Surveillance, Epidemiology, and End Results (SEER) database. Second, data on comorbidities and functional status were not recorded in the SEER database, which might have an influence on treatment decision making. Moreover, chemotherapy regime, anti-HER2 therapy, and endocrine therapy were also unavailable in the SEER database. Finally, a high rate of under-reporting of RT administration was found in the SEER database.
In conclusion, our study suggests that the 8th AJCC pathological prognostic staging system provides accurate risk stratification and impacts the treatment decision making for elderly women with early-stage BC. Elderly patients with early-stage BC undergoing BCS and RT have lower risk of BCSM than those undergoing MAST or with BCS alone regardless of the pathological prognostic stages. More studies are needed to guide treatment decision making by the new pathological prognostic stages for elderly patients with BC.
Materials and Methods
Patients
We performed this retrospective analysis, including elderly women (aged ≥65 years) with T1-2N0M0 BC who underwent BCS or MAST between 2010 and 2014. Data on the patients were retrieved from the SEER database. The SEER program is an open-access population-based cancer registry, which including data on tumor incidence, demographic features, tumor characteristics, the first course of treatment, and survival outcomes for approximately 28% of the United States population [43]. We excluded patients with a non-positive pathological diagnosis or insufficient data on race/ethnicity, tumor grade, ER status, PR status, HER2 status, or marital status. This study was exempted from approval by the Institutional Review Board because patients’ information in the SEER database is de-identified.
Variables
We retrieved the following information for each patient: age, race/ethnicity, tumor stage, tumor grade, histology, ER status, PR status, HER2 status, and marital status. We also collected data on the receipt of surgical procedures, chemotherapy, or postoperative RT for statistical analyses. Patients with stage T1-2N0M0 were reassigned to stages IA, IB, and IIA using the 8th edition of the AJCC pathological prognostic staging manual (9,10).
Statistical analyses
Patients’ characteristics were compared using the chi-square test, and predictors of receiving local treatments were assessed using binomial logistic regression. Univariate and multivariable competing risks models were used to assess the cumulative incidence of BCSM. The BCSM was defined as the interval from the diagnosis of BC to the date of death from BC. ROC curves were used to assess the discriminatory ability of the 7th AJCC staging system and the 8th AJCC staging system. Competing risks models with the Cox model framework, as proposed by Fine and Gray were used to assess the combined effects of the variables to determine the predictors of BCSM. All statistical analyses were performed using IBM SPSS 22.0 (IBM Corp., Armonk, NY), R statistical software (version 3.5.0; https://www.r-project.org/), and Stata/SE version 14 (StataCorp, TX, USA). All statistical tests were two-tailed, with P values <0.05 considered statistically significant.
Author Contributions
SGW, JS, WWZ, JZ, and ZYH are lead authors who participated in manuscript drafting, table/figure creation, and manuscript revision. SGW and ZHY aided in data collection. SGW, JS, WWZ, JW, CLL, LH, and JW, and LH are senior authors who aided in drafting the manuscript and manuscript revision. ZYH and JZ are the corresponding authors who initially developed the concept and drafted and revised the manuscript. All authors read and approved the final manuscript.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This work was partly supported by the National Natural Science Foundation of China (No. 81802600, 81872459, 81803050), the Commission Young and Middle-aged Talents Training Project of Fujian Health Commission (No. 2019-ZQNB-25), and Medical Scientific Research Foundation of Health Commission of Guangdong Province of China (A2018194).
References
- 1. Miller KD, Siegel RL, Lin CC, Mariotto AB, Kramer JL, Rowland JH, Stein KD, Alteri R, Jemal A. Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin. 2016; 66:271–89. https://doi.org/10.3322/caac.21349 [PubMed]
- 2. Kummerow KL, Du L, Penson DF, Shyr Y, Hooks MA. Nationwide trends in mastectomy for early-stage breast cancer. JAMA Surg. 2015; 150:9–16. https://doi.org/10.1001/jamasurg.2014.2895 [PubMed]
- 3. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018; 68:394–424. https://doi.org/10.3322/caac.21492 [PubMed]
- 4. Lodi M, Scheer L, Reix N, Heitz D, Carin AJ, Thiébaut N, Neuberger K, Tomasetto C, Mathelin C. Breast cancer in elderly women and altered clinico-pathological characteristics: a systematic review. Breast Cancer Res Treat. 2017; 166:657–68. https://doi.org/10.1007/s10549-017-4448-5 [PubMed]
- 5. Bastiaannet E, Liefers GJ, de Craen AJ, Kuppen PJ, van de Water W, Portielje JE, van der Geest LG, Janssen-Heijnen ML, Dekkers OM, van de Velde CJ, Westendorp RG. Breast cancer in elderly compared to younger patients in the Netherlands: stage at diagnosis, treatment and survival in 127,805 unselected patients. Breast Cancer Res Treat. 2010; 124:801–07. https://doi.org/10.1007/s10549-010-0898-8 [PubMed]
- 6. Fisher B, Anderson S, Bryant J, Margolese RG, Deutsch M, Fisher ER, Jeong JH, Wolmark N. Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer. N Engl J Med. 2002; 347:1233–41. https://doi.org/10.1056/NEJMoa022152 [PubMed]
- 7. Veronesi U, Cascinelli N, Mariani L, Greco M, Saccozzi R, Luini A, Aguilar M, Marubini E. Twenty-year follow-up of a randomized study comparing breast-conserving surgery with radical mastectomy for early breast cancer. N Engl J Med. 2002; 347:1227–32. https://doi.org/10.1056/NEJMoa020989 [PubMed]
- 8. Cserni G, Chmielik E, Cserni B, Tot T. The new TNM-based staging of breast cancer. Virchows Arch. 2018; 472:697–703. https://doi.org/10.1007/s00428-018-2301-9 [PubMed]
- 9. AJCC cancer staging manual. New York: Springer International Publishing, 2018.
- 10. Giuliano AE, Connolly JL, Edge SB, Mittendorf EA, Rugo HS, Solin LJ, Weaver DL, Winchester DJ, Hortobagyi GN. Breast cancer-major changes in the american joint committee on cancer eighth edition cancer staging manual. CA Cancer J Clin. 2017; 67:290–303. https://doi.org/10.3322/caac.21393 [PubMed]
- 11. Kim I, Choi HJ, Ryu JM, Lee SK, Yu JH, Kim SW, Nam SJ, Lee JE. Prognostic validation of the american joint committee on cancer 8th staging system in 24,014 korean patients with breast cancer. J Breast Cancer. 2018; 21:173–81. https://doi.org/10.4048/jbc.2018.21.2.173 [PubMed]
- 12. Weiss A, Chavez-MacGregor M, Lichtensztajn DY, Yi M, Tadros A, Hortobagyi GN, Giordano SH, Hunt KK, Mittendorf EA. Validation study of the American joint committee on cancer eighth edition prognostic stage compared with the anatomic stage in breast cancer. JAMA Oncol. 2018; 4:203–9. https://doi.org/10.1001/jamaoncol.2017.4298 [PubMed]
- 13. Lee SB, Sohn G, Kim J, Chung IY, Lee JW, Kim HJ, Ko BS, Son BH, Ahn SH. A retrospective prognostic evaluation analysis using the 8th edition of the american joint committee on cancer staging system for breast cancer. Breast Cancer Res Treat. 2018; 169:257–66. https://doi.org/10.1007/s10549-018-4682-5 [PubMed]
- 14. Shao N, Xie C, Shi Y, Ye R, Long J, Shi H, Shan Z, Thompson AM, Lin Y. Comparison of the 7th and 8th edition of american joint committee on cancer (AJCC) staging systems for breast cancer patients: a surveillance, epidemiology and end results (SEER) analysis. Cancer Manag Res. 2019; 11:1433–42. https://doi.org/10.2147/CMAR.S185212 [PubMed]
- 15. Janssen-Heijnen ML, Houterman S, Lemmens VE, Louwman MW, Maas HA, Coebergh JW. Prognostic impact of increasing age and co-morbidity in cancer patients: a population-based approach. Crit Rev Oncol Hematol. 2005; 55:231–40. https://doi.org/10.1016/j.critrevonc.2005.04.008 [PubMed]
- 16. Noordzij M, Leffondré K, van Stralen KJ, Zoccali C, Dekker FW, Jager KJ. When do we need competing risks methods for survival analysis in nephrology? Nephrol Dial Transplant. 2013; 28:2670–77. https://doi.org/10.1093/ndt/gft355 [PubMed]
- 17. Gooley TA, Leisenring W, Crowley J, Storer BE. Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med. 1999; 18:695–706. https://doi.org/10.1002/(sici)1097-0258(19990330)18:6<695::aid-sim60>3.0.co;2-o [PubMed]
- 18. Putter H, Fiocco M, Geskus RB. Tutorial in biostatistics: competing risks and multi-state models. Stat Med. 2007; 26:2389–430. https://doi.org/10.1002/sim.2712 [PubMed]
- 19. Weiss A, King TA, Hunt KK, Mittendorf EA. Incorporating biologic factors into the american joint committee on cancer breast cancer staging system: review of the supporting evidence. Surg Clin North Am. 2018; 98:687–702. https://doi.org/10.1016/j.suc.2018.03.005 [PubMed]
- 20. Cappellani A, Di Vita M, Zanghì A, Cavallaro A, Piccolo G, Majorana M, Barbera G, Berretta M. Prognostic factors in elderly patients with breast cancer. BMC Surg. 2013 (Suppl 2); 13:S2. https://doi.org/10.1186/1471-2482-13-S2-S2 [PubMed]
- 21. Blows FM, Driver KE, Schmidt MK, Broeks A, van Leeuwen FE, Wesseling J, Cheang MC, Gelmon K, Nielsen TO, Blomqvist C, Heikkilä P, Heikkinen T, Nevanlinna H, et al. Subtyping of breast cancer by immunohistochemistry to investigate a relationship between subtype and short and long term survival: a collaborative analysis of data for 10,159 cases from 12 studies. PLoS Med. 2010; 7:e1000279. https://doi.org/10.1371/journal.pmed.1000279 [PubMed]
- 22. Kim JY, Lim JE, Jung HH, Cho SY, Cho EY, Lee SK, Yu JH, Lee JE, Kim SW, Nam SJ, Park YH, Ahn JS, Im YH. Validation of the new AJCC eighth edition of the TNM classification for breast cancer with a single-center breast cancer cohort. Breast Cancer Res Treat. 2018; 171:737–45. https://doi.org/10.1007/s10549-018-4858-z [PubMed]
- 23. Minicozzi P, Van Eycken L, Molinie F, Innos K, Guevara M, Marcos-Gragera R, Castro C, Rapiti E, Katalinic A, Torrella A, Žagar T, Bielska-Lasota M, Giorgi Rossi P, et al, and European HR Working Group on breast cancer. Comorbidities, age and period of diagnosis influence treatment and outcomes in early breast cancer. Int J Cancer. 2019; 144:2118–27. https://doi.org/10.1002/ijc.31974 [PubMed]
- 24. Efird JT, Hunter S, Chan S, Jeong S, Thomas SL, Jindal C, Biswas T. The association between age, comorbidities and use of radiotherapy in women with breast cancer: implications for survival. Medicines (Basel). 2018; 5:62. https://doi.org/10.3390/medicines5030062 [PubMed]
- 25. Restrepo DJ, Sisti A, Boczar D, Huayllani MT, Fishe J, Gabriel E, McLaughlin SA, Bagaria S, Spaulding A, Rinker BD, Forte AJ. Characteristics of breast cancer patients who refuse surgery. Anticancer Res. 2019; 39:4941–45. https://doi.org/10.21873/anticanres.13682 [PubMed]
- 26. Chen SJ, Kung PT, Huang KH, Wang YH, Tsai WC. Characteristics of the delayed or refusal therapy in breast cancer patients: a longitudinal population-based study in taiwan. PLoS One. 2015; 10:e0131305. https://doi.org/10.1371/journal.pone.0131305 [PubMed]
- 27. Biganzoli L, Wildiers H, Oakman C, Marotti L, Loibl S, Kunkler I, Reed M, Ciatto S, Voogd AC, Brain E, Cutuli B, Terret C, Gosney M, et al. Management of elderly patients with breast cancer: updated recommendations of the International Society of Geriatric Oncology (SIOG) and European Society of Breast Cancer Specialists (EUSOMA). Lancet Oncol. 2012; 13:e148–60. https://doi.org/10.1016/S1470-2045(11)70383-7 [PubMed]
- 28. Thavarajah N, Menjak I, Trudeau M, Mehta R, Wright F, Leahey A, Ellis J, Gallagher D, Moore J, Bristow B, Kay N, Szumacher E. Towards an optimal multidisciplinary approach to breast cancer treatment for older women. Can Oncol Nurs J. 2015; 25:384–408. https://doi.org/10.5737/23688076254384395 [PubMed]
- 29. NCCN. NCCN clinical Practice guidelines in oncology V.2.2019. Breast Cancer. (2019) Available online at: https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf.
- 30. Wu SG, Wang J, Lian CL, Lei J, Hua L, Lin Q, Chen YX, He ZY. Evaluation of the 8th edition of the American joint committee on cancer’s pathological staging system in prognosis assessment and treatment decision making for stage T1-2N1 breast cancer after mastectomy. Breast. 2020; 51:2–10. https://doi.org/10.1016/j.breast.2020.02.012 [PubMed]
- 31. Wu SG, Wang J, Lei J, Lian CL, Hua L, Zhou J, He ZY. Prognostic validation and therapeutic decision-making of the AJCC eighth pathological prognostic staging for T3N0 breast cancer after mastectomy. Clin Transl Med. 2020; 10:125–36. https://doi.org/10.1002/ctm2.3 [PubMed]
- 32. Yamada A, Narui K, Sugae S, Shimizu D, Takabe K, Ichikawa Y, Ishikawa T, Endo I. Operation with less adjuvant therapy for elderly breast cancer. J Surg Res. 2016; 204:410–17. https://doi.org/10.1016/j.jss.2016.05.031 [PubMed]
- 33. Marmor S, Altman AM, Mayleben WT, Hui JY, Denbo JW, Jensen EH, Tuttle TM. The use of contralateral prophylactic mastectomy among elderly patients in the United States. Breast Cancer Res Treat. 2019; 177:175–83. https://doi.org/10.1007/s10549-019-05288-8 [PubMed]
- 34. Angarita FA, Acuna SA, Cordeiro E, Elnahas A, Sutradhar S, Jackson T, Cil TD. Thirty-day postoperative morbidity and mortality in elderly women with breast cancer: an analysis of the NSQIP database. Breast Cancer Res Treat. 2018; 170:373–79. https://doi.org/10.1007/s10549-018-4747-5 [PubMed]
- 35. Hughes KS, Schnaper LA, Bellon JR, Cirrincione CT, Berry DA, McCormick B, Muss HB, Smith BL, Hudis CA, Winer EP, Wood WC. Lumpectomy plus tamoxifen with or without irradiation in women age 70 years or older with early breast cancer: long-term follow-up of CALGB 9343. J Clin Oncol. 2013; 31:2382–87. 10.1200/JCO.2012.45.2615 [PubMed]
- 36. Kunkler IH, Williams LJ, Jack WJ, Cameron DA, Dixon JM, and PRIME II investigators. Breast-conserving surgery with or without irradiation in women aged 65 years or older with early breast cancer (PRIME II): a randomised controlled trial. Lancet Oncol. 2015; 16:266–73. https://doi.org/10.1016/S1470-2045(14)71221-5 [PubMed]
- 37. Herskovic AC, Wu X, Christos PJ, Nagar H. Omission of adjuvant radiotherapy in the elderly breast cancer patient: missed opportunity? Clin Breast Cancer. 2018; 18:418–31. https://doi.org/10.1016/j.clbc.2018.02.006 [PubMed]
- 38. Hancke K, Denkinger MD, König J, Kurzeder C, Wöckel A, Herr D, Blettner M, Kreienberg R. Standard treatment of female patients with breast cancer decreases substantially for women aged 70 years and older: a german clinical cohort study. Ann Oncol. 2010; 21:748–53. https://doi.org/10.1093/annonc/mdp364 [PubMed]
- 39. Luu C, Goldstein L, Goldner B, Schoellhammer HF, Chen SL. Trends in radiotherapy after breast-conserving surgery in elderly patients with early-stage breast cancer. Ann Surg Oncol. 2013; 20:3266–73. https://doi.org/10.1245/s10434-013-3150-z [PubMed]
- 40. Kunkler IH, Williams LJ, King CC, Jack W. Breast radiotherapy: considerations in older patients. Clin Oncol (R Coll Radiol). 2009; 21:111–17. https://doi.org/10.1016/j.clon.2008.11.012 [PubMed]
- 41. Joslyn SA. Radiation therapy and patient age in the survival from early-stage breast cancer. Int J Radiat Oncol Biol Phys. 1999; 44:821–26. https://doi.org/10.1016/s0360-3016(99)00071-1 [PubMed]
- 42. Algan O, Zhao YD, Herman T. Radiotherapy in patients 70 years and older with triple-negative breast cancer. Clin Breast Cancer. 2016; 16:e99–106. https://doi.org/10.1016/j.clbc.2016.05.011 [PubMed]
- 43. Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database: Incidence - SEER 18 Regs Custom Data (with additional treatment fields), Nov 2018 Sub (1975-2016 varying) - Linked To County Attributes - Total U.S., 1969-2017 Counties, National Cancer Institute, DCCPS, Surveillance Research Program, released April 2019, based on the November 2018 submission.



