Introduction
Sarcopenia result from a progressive loss of skeletal muscle and is associated with reduced physical function, falls, fractures, disability, hospitalization, and a poor quality of life [1]. Moreover, sarcopenia increases the mortality rate of the elderly by a factor of up to 2.34 fold [2]. The 2018 European Working Group on Sarcopenia in Older People 2 (EWGSOP2) reported that these adverse muscle changes would accrue throughout a lifetime, not only in elderly adults, but also in younger adults [3]. It is estimated that 30% of people > 60 years old, and that half of people > 80 years old might have sarcopenia and a higher-than-average risk for falling [4]. Of all accidental deaths in the USA, 36.8/100,000 population are attributable to falls [5]. Therefore, the EWGSOP2 encouraged more research on sarcopenia to prevent or delay adverse health outcomes that incur heavy burdens on either individuals or healthcare systems [3].
Exercise positively affects muscle mass and strength [6], but the elderly, and especially those that are physically frail, often find it difficult to exercise. Dietary and nutritional interventions may also increase muscle mass and help maintain physical performance and function [7]. Recent studies have claimed that protein supplementation can help prevent sarcopenia [8–10]. Protein is composed of amino acids that can induce a muscle protein anabolic response conditioned by the availability of branched-chain amino acids (BCAAs) such as leucine, isoleucine, and valine [11]. Delayed amino acid absorption and anabolic resistance are common in the elderly [12]. Low levels of BCAAs are associated with low muscle mass, poor muscle function, and low strength in the community-dwelling elderly [13]. Studies report that supplementary BCAAs, leucine, essential amino acids, whey protein, or vitamin D plus low-intensity resistance training, ranging from 3 days to 12 weeks, stimulate muscle protein synthesis and attenuate sarcopenia, even in the elderly confined to bedrest [13–20]. Compliance with supplementation treatment is influenced by the form of nutrients, personal digestion tolerance to oral supplementation, and dosing frequency, among others [21, 22]. Furthermore, few studies have investigated the changes after those above supplementations were discontinued..
The aim of the present study was to evaluate the short-term effects of enriched supplementation with BCAAs in subjects with pre-sarcopenia or sarcopenia. Here, we hypothesized that the physical performance, muscle strength, and muscle mass of elderly patients would improve after 5 weeks of treatment with enriched BCAAs but would decline after a 12-week discontinuation of treatment.
Results
Five- week BCAA intervention
The mean short-form MNA score of 33 participants (6 M/27 F; mean age = 66.6 ± 10.3 years) who underwent enriched BCAA treatment was 12.0 ± 1.5, which indicated normal nutritional status (Table 1). After a 5-week enriched BCAA intervention, skeletal mass index (SMI), gait speed, and grip strength were all improved (Table 1). The changes were consistent in both the sarcopenic subgroup (Supplementary Table 1) and the pre-sarcopenic subgroup (Supplementary Table 2), except for SMI.
Table 1. Demographic characteristics, physical status, and performance before and after 5 weeks of enriched BCAA treatment.
| Variable | Baseline | 5 weeks | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Case no. (presarcopenia/sarcopenia) | 33 (12/21) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex (M/F) | 6 M/27 F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years) | 66.6 ± 10.3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Education (< 6 years) | 21 (63.6%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking habit | 4 (12.1%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alcohol drinking habit | 2 (6.1%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Medical history | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension | 10 (30.3%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes | 6 (18.2%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dyslipidemia | 6 (30.3%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cardiovascular disease | 3 (9.1%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thyroid dysfunction | 3 (9.1%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Height (cm) | 154.0 ± 5.6 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mini Nutrition Assessment Short Form score | 12.0 ± 1.5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| International physical assessment activity (kcal/week) | 5549.6 ± 4963.1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Body Weight (kg) | 49.7 ± 8.7 | 50.0 ± 9.2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Body Mass Index (kg/m2) | 20.9 ± 3.0 | 21.0 ± 3.2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Body fat (%) | 27.8 ± 6.1 | 27.5 ± 6.6 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Waist Circumference (cm) | 77.5 ± 12.0 | 77.9 ± 11.9 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Skeletal Muscle Index (kg/m2) | 5.84 ± 0.91 | 5.99 ± 1.01* | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gait Speed (m/sec) | 0.82 ± 0.17 | 0.94 ± 0.19** | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grip Strength (kg) | 18.0 ± 4.8 | 21.4 ± 5.1** | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-parametric Wilcoxon signed rank test: *p < 0.01, **p < 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Twelve week BCAA discontinuation
No participant dropped out between 0-5 weeks but during the next 12-week discontinuation of BCAAs, there were 7 dropouts. There was no statistical difference in basic characteristics and sarcopenic parameters between the dropouts and the other 26 participants (Supplementary Table 3). The 26 completers in the 5-week intervention group had higher SMI, gait speed, and grip strength than at baseline, but those in the 12-week discontinuation group had lower SMI, gait speed, and grip strength (Table 2).
Table 2. Physical status and performance at baseline, after 5 weeks of BCAAs, and after 17 weeks (12 weeks without BCAAs) in 26 completers.
| Variable | Baseline | 5 weeks | 17 weeks | P# | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Case no. (presarcopenia/sarcopenia) | 26 (7/19) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex | 5 M/21 F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years) | 66.4 ± 10.2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Height (cm) | 154.3 ± 6.0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Body Weight (kg) | 50.4 ± 9.2 | 50.7 ± 9.9 | 49.0 ± 9.2 | 0.832 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Body Fat (%) | 28.1 ± 6.0 | 27.9 ± 6.5 | 28.8 ± 6.2§** | 0.004 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Skeletal Muscle Index (kg/m2) | 5.86 ± 0.94 | 5.99 ± 1.06† | 5.80 ± 0.92** | 0.006 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gait Speed (m/sec) | 0.84 ± 0.16 | 0.97 ± 0.17* | 0.91 ± 0.15§ | 0.007 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grip Strength (kg) | 18.2 ± 4.9 | 21.7 ± 5.2* | 20.9 ± 4.9‡¶ | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| #Non-parametric Friedman and Wilcoxon Signed Rank test comparing among baseline, week 5 and week 17: *p < 0.001, †p < 0.05 baseline vs. week 5; ‡p < 0.01, §p < 0.05 baseline vs. week 17; ¶p < 0.05, **p < 0.01 week 5 vs. week 17. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Comparison between BCAA and placebo treatments at five weeks
Before we acquired 33 BCAA interventional samples, our study had 39 subjects at the beginning. However, six of them who agreed to join the intervention study refused to take any BCAA supplements while initiating the intervention. Compared with those six subjects (similar to the open-labeled placebo group), the 12 BCAA interventional subjects (1:2 age and sex matched) showed improved changes in sarcopenic parameters at five weeks (Supplementary Table 4).
Effect of BCAA on different age groups
To determine the interventional effects on participants in different age groups, participants were divided into two subgroups: those who were < 65 years old and those ≥ 65 years old (Supplementary Figure 1). Grip strength was higher in both subgroups after 5 weeks of BCAA therapy. After 12 weeks of discontinuation, SMI and grip strength were lower than the week 5 test in the < 65-year-old group (1 M/11 F), while grip strength and gait speed were lower than at the week 5 test in the ≥ 65-year-old group (4 M/10 F).
Discussion
We found that five weeks of supplementation with BCAAs led to increased physical performance, muscle strength, and muscle mass in middle-aged and elderly patients, but the effect subsided after participants discontinued BCAAs for 12 weeks. In the sarcopenic subgroup (n = 21) (Supplementary Table 1), SMI increased, as did grip strength and gait speed (Supplementary Table 1), similar to the results of Takeuchi et al [19]. In the pre-sarcopenic subgroup (n = 12) (Supplementary Table 2), grip strength and gait-speed were higher after BCAA treatment; however, SMI did not change, perhaps owing to our relative small sample size. It is plausible to suggest that a better response to BCAAs can be expected in sarcopenic than in pre-sarcopenic participants.
The duration of BCAA treatment relative to the duration of discontinuation is a critical factor for physical function, grip strength, and muscle mass changes, as demonstrated by results from studies using a wide range of interventional durations, from three days to 12 weeks [13–20]. One study found that muscle protein synthesis (MPS) increased after supplementation with leucine-enriched essential amino acids (containing 3 g of leucine) for just two hours in old women [23]. A recent study using 4.2 g leucine supplementation for six days may also be helpful to increase MPS in the rest leg group of healthy individuals [24]. In agreement with these results, our study here showed an improvement in sarcopenic parameters after five weeks of 7.2 gm BCAAs. On the contrary, several studies have reported that muscle disuse for 10 to 42 days leads to a daily loss of between 0.6% and 5% of muscle mass and to a daily decline in grip strength between 0.3% and 4.2% [17, 25, 26]. We also confirmed lower muscle parameters after a 12-week discontinuation of enriched BCAAs. An interesting finding was the decline of muscle mass followed by a decline in other physical functions and performance. Whether there is also a cause-effect relationship is unclear and warrants further investigation.
Researchers have attempted to determine nutritional strategies to reduce the higher anabolic turnover rate of muscle as humans get older [27]. Leucine is unique because it activates mammalian target of rapamycin complex 1 (mTORC1) and the downstream phosphorylation of p70S6 kinase (p70S6k) and 4E (eIF4E)-binding protein 1 (4E-BP1) and related signaling pathways [28–30]. A 55-mg dose of leucine/kg/d was claimed to be optimal and recommended for younger adults [31] because it activates mTOR and leads to the increased synthesis of muscle protein [32, 33]. Other studies [34, 35] have also reported that leucine supplements helped slow age-related muscle mass reductions in the elderly. Moreover, MPS is stimulated in older mice by leucine-enriched whey protein but not by leucine only [36]. However, one meta-analysis [37] of randomized controlled nutritional interventions for treating sarcopenia reported no effects on appendicular skeletal muscle mass, grip strength, or gait speed. Nevertheless, beta-hydroxy beta-methylbutyrate (HMB), leucine, or BCAAs may strongly promote increases in muscle mass [38, 39]. Compatible with our study here, a randomized controlled trial [40] of eight-week interventions consisting of a leucine-enriched amino acid supplementation (Amino L40; Ajinomoto Co., Inc., Tokyo, Japan) and low-intensity resistance training increased muscle mass, strength, and physical function in post-stroke subjects with sarcopenia. Supplementation with BCAAs shifted the net protein balance from catabolism to anabolism, the result of protein synthesis exceeding protein breakdown [34] and, therefore, an increase in SMI. However, there’s a potential relationship between BCAA supplementation and the development of insulin resistance or type-2 diabetes mellitus [41–43]. Lowering dietary BCAAs increased energy expenditure and improved insulin sensitivity in animal models [44]. On the contrary, one study showed higher amounts of dietary BCAAs to be associated with lower T2DM risk. [45] Nevertheless, subjects with diabetic concerns were suggested to closely follow up the variation of blood glucose level while taking BCAAs.
Although different changes of sarcopenic parameters were noted between the groups aged < 65 years and ≥ 65years, the interference of the relatively small sample size and diverse distribution of gender cannot be overlooked. Moreover, the decline of sarcopenic parameters after discontinuation was partially compatible in both age groups. These preliminary findings might reflect a potential role of aging in response to BCAA intervention.
Our study had limitations. First, we analyzed only 33 samples with seven dropouts during the discontinuation period and had no randomized placebo group. The results indicate that no statistical difference of basic characteristics was found between the seven dropouts and the other 26 per protocol patients (Supplementary Table 3). Nonetheless, several studies have enrolled less than 30 subjects. For example, the HMB supplementation trial recruiting 16 subjects showed changes in muscle mass [46]. Furthermore, our study met the calculated minimum sample size. In addition to 33 samples, our study had six participants in the open-labeled placebo group. Using 1:2 age and sex matched with the six placebo subjects, the 12 BCAA subjects have showed improved changes in sarcopenic parameters at five weeks (Supplementary Table 4). Both the six subjects without BCAA (Supplementary Table 4) and the 12-week discontinuation evaluation (Table 2) can reflect the placebo effect. Therefore, our findings provided useful evidence of the nutritional intervention for sarcopenia, but whether our findings could be extrapolated to younger or non-sarcopenic groups was uncertain. Second, although we asked the participants to maintain their daily diets and activities as usual during intervention and discontinuation, our study was observational and some may not have compiled. The influence of daily physical activity and dietary food cannot be overlooked in further studies. However, with unchanged lifestyle intervention, our study results would be much closer to real-world interventions and applied in community dwelling populations. Third, we used only two sachets of enriched BCAAs daily. Future studies need more groups with different treatment doses and durations. Fourth, this study was initiated before the announcement of 2019 AWGS [47]. Compared with the new definitions (handgrip, gait speed, SMI) of 2019 AWGS, the enrolled case number remains unchanged. Finally, we did not examine biochemical parameters; thus, we did not examine changes in endocrine and hormone biomarkers.
Supplementation with enriched BCAAs for five weeks may help improve parameters with low values (SMI, gait speed, and grip strength) in pre-sarcopenic and sarcopenic subjects. After 12 weeks discontinuation, these improvements will be obscured, especially SMI and grip strength. However, further investigation is warranted to confirm the optimal types, doses, and durations of treatment and discontinuation effects in clinical practice.
Materials and Methods
Participants
A quasi-experimental single-arm intervention followed by discontinuation study was designed (Figure 1). We surveyed 630 community-dwelling middle-aged and elderly Taiwanese who lived close to Jia-Li Chi Mei Hospital. Based on the power calculation from the Wilcoxon signed-rank test (effect size 0.5, power 0.95, pre- and post-intervention matched pairs; or Takeuchi study [19]), we estimated that at least 29 to 33 participants would satisfy the requirement of statistical analysis. All eligible pre-sarcopenic and sarcopenic recruits were enrolled. Exclusion criteria were uncontrolled hypertension or diabetes, stroke; severe liver or renal disease, gastrointestinal disease, neuromuscular disease, infectious disease, pulmonary disease, endocrine system disease, neurological or acute/advanced psychiatric disease; cancer, a history of seizures, and sensitivity to any study ingredients.
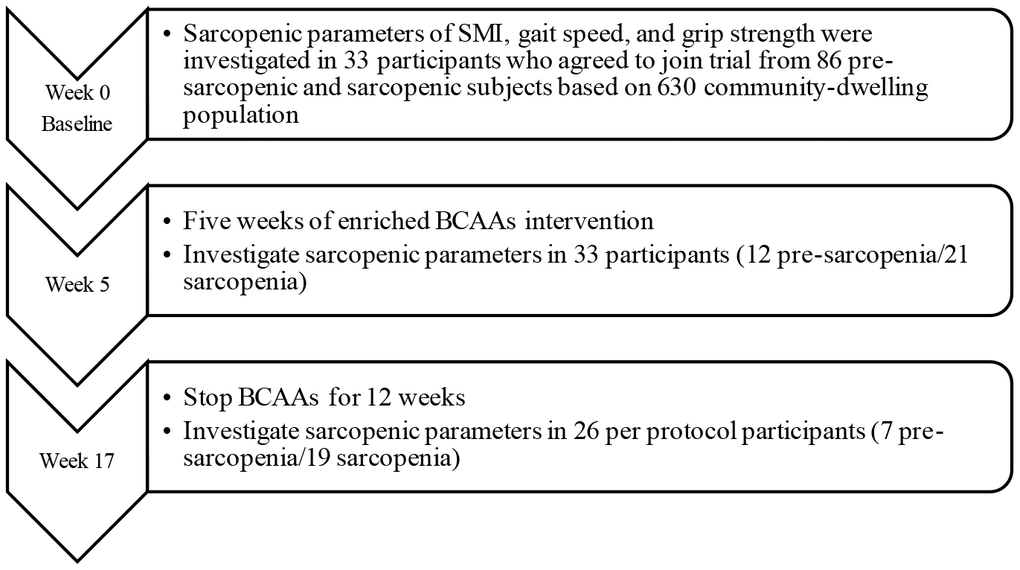
Figure 1. Flowchart of study participants enrolled from community survey.
Assessments
An overview of the interventional study design is shown in Figure 1. At the initial screening, recruits underwent a review of the inclusion and exclusion criteria, medical history, concomitant therapies, daily activity evaluation, and anthropometric and body composition measurements. The short form Mini-Nutritional Assessment (MNA) was used to evaluate nutritional status [48, 49].
Bodyweight and standing height were measured using a medical weight- and height-analyzing scale (Detecto, Webb City, MO, USA); participants were barefoot and dressed in light clothing. Body mass index (BMI) was calculated using this formula: BMI = bodyweight in kg/height in m2. Waist circumference (WC) was measured to the nearest mm using a tape measure (Gulick II; Gays Mills, WI, USA) midway between the lateral lower rib margin and the superior anterior iliac crest after a gentle expiration [50]. A single frequency 8-electrode bioelectrical impedance analysis (BIA) device (BC-418; Tanita Corp., Itabashi-ku, Tokyo, Japan) was used to measure body composition, including body fat and skeletal muscle mass (SMM) [51] (estimated using Janssen’s equation (SMI = kg/m2) [1, 52–54]. Gait speed was measured by the walking test modified to a 6-meter distance [53]. Low physical performance is predicted when gait speed is < 0.8 m/s [55]. Grip strength (Grip-D [TKK 5401]; Japan) was obtained from three separate 30-s fast-twist tests for one hand for each participant. The maximum value of grip strength was used to assess low-level muscle function based on the corresponding cut points of < 26 kg/m2 for men and < 18 kg/m2 for women [54, 55]. An SMI < the cut points (7.6 kg/m2 for men and 5.67 kg/m2for women) was classified as pre-sarcopenia. Sarcopenia was defined as a low SMI and either a low grip strength or a low walking speed by the modified 2014 consensus of the Asian Working Group on Sarcopenia [55].
BCAA interventions
Eighty-six (13.7%) of 630 participants were pre-sarcopenic or sarcopenic. Twelve pre-sarcopenic and 21 sarcopenic participants were purposively sampled for one sachet enriched BCAAs (Amino VITAL® PRO 3600; Ajinomoto Co., Inc., Tokyo, Japan, 3.6g/sachet) interventions [56] twice daily for 5 weeks. One sachet included leucine 0.54g, isoleucine 0.43g, valine 0.36g, glutamine 0.65g, arginine 0.61g and other amino acids 1.01g. A total of 1.08g leucine per day was supplied for each participant. All participants were asked to habituate themselves to their diet and activity during the study. Participants were contacted by phone every week to discuss compliance, adverse events, and study requirements. The compliance of 5-week BCAAs supplement usage was 97%. After 5 weeks of BCAAs, all BCAA-treated participants discontinued them for the next 12 weeks. Serial sarcopenia-associated tests were done at week 0, week 5, and week 17 for each participant.
Statistics
All statistical analyses were done using the Statistical Package for the Social Sciences 22 for Windows (IBM Corp., Armonk, NY, USA). Categorical and continuous variables are expressed as percentages or as means ± standard deviation (SD), as indicated. Continuous variables were analyzed using the Wilcoxon Signed Rank non-parametric test. The Mann-Whitney non-parametric test was used to compare dropouts and study completers. The non-parametric Friedman and Wilcoxon Signed Rank test was used to compare the means of variance within three groups and subgroups under the age of 65. Significance was set at p < 0.05 for two-tailed analysis.
Ethical approval
The study (ClinicalTrials.gov Identifier: NCT03891134) was approved and monitored by the Institutional Review Board of the Chi Mei Medical Center (CMMC10504-J01). Each participant was informed of the purpose of the study, experimental procedures, and potential risks after providing signed written consent.
Author Contributions
Conceptualization, Chun-Hung Ko; Data creation, Chun-Hung Ko, Shin-Jiuan Wu, Shan-Tair Wang, Yin-Fan Chang, Chin-Sung Chang and Chih-Hsing Wu; Formal analysis, Shan-Tair Wang; Funding acquisition, Chih-Hsing Wu; Investigation, Chun-Hung Ko, Shin-Jiuan Wu, Yin-Fan Chang, Chin-Sung Chang and Chih-Hsing Wu; Methodology, Ta-Shen Kuan, Chia-Ming Chang and Chih-Hsing Wu; Resources, Shin-Jiuan Wu, Hua-Ying Chuang and Chih-Hsing Wu; Supervision, Shan-Tair Wang, Willy Chou and Chih-Hsing Wu; Validation, Chia-Ming Chang; Writing – original draft, Chun-Hung Ko; Writing – review and editing, Chih-Hsing Wu. All authors have approved the final manuscript.
Acknowledgments
The authors would like to thank the Taiwanese Osteoporosis Association, the Taiwan Ministry of Science and Technology (MOST No. 106-2314-B-006-064-MY2), Chi Mei Hospital (Holistic Health Care No. JCHCR 10510), and National Cheng Kung University Hospital (No. NCKUH-10709012) for the listed grants.
Funding
The funding sources had no role in the study. We also wish to thank Ms. Xu-Shan Huang for her administrative assistance and Bill Franke for the English editing. Finally, we want to express our gratitude to students from the Department of Food and Nutrition, Chung Hwa University of Medical Technology, for their generous support and assistance.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- 1. Janssen I, Heymsfield SB, Ross R. Low relative skeletal muscle mass (sarcopenia) in older persons is associated with functional impairment and physical disability. J Am Geriatr Soc. 2002; 50:889–96. https://doi.org/10.1046/j.1532-5415.2002.50216.x [PubMed]
- 2. Landi F, Liperoti R, Fusco D, Mastropaolo S, Quattrociocchi D, Proia A, Tosato M, Bernabei R, Onder G. Sarcopenia and mortality among older nursing home residents. J Am Med Dir Assoc. 2012; 13:121–26. https://doi.org/10.1016/j.jamda.2011.07.004 [PubMed]
- 3. Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T, Cooper C, Landi F, Rolland Y, Sayer AA, Schneider SM, Sieber CC, Topinkova E, et al, Writing Group for the European Working Group on Sarcopenia in Older People 2 (EWGSOP2), and the Extended Group for EWGSOP2. Sarcopenia: revised european consensus on definition and diagnosis. Age Ageing. 2019; 48:16–31. https://doi.org/10.1093/ageing/afy169 [PubMed]
- 4. Baumgartner RN, Koehler KM, Gallagher D, Romero L, Heymsfield SB, Ross RR, Garry PJ, Lindeman RD. Epidemiology of sarcopenia among the elderly in new Mexico. Am J Epidemiol. 1998; 147:755–63. https://doi.org/10.1093/oxfordjournals.aje.a009520 [PubMed]
- 5. Centers for Disease Control and Prevention (CDC). Fatalities and injuries from falls among older adults—United States, 1993-2003 and 2001-2005. MMWR Morb Mortal Wkly Rep. 2006; 55:1221–24. [PubMed]
- 6. Yu SC, Khow KS, Jadczak AD, Visvanathan R. Clinical screening tools for sarcopenia and its management. Curr Gerontol Geriatr Res. 2016; 2016:5978523. https://doi.org/10.1155/2016/5978523 [PubMed]
- 7. Cruz-Jentoft AJ, Kiesswetter E, Drey M, Sieber CC. Nutrition, frailty, and sarcopenia. Aging Clin Exp Res. 2017; 29:43–48. https://doi.org/10.1007/s40520-016-0709-0 [PubMed]
- 8. Naseeb MA, Volpe SL. Protein and exercise in the prevention of sarcopenia and aging. Nutr Res. 2017; 40:1–20. https://doi.org/10.1016/j.nutres.2017.01.001 [PubMed]
- 9. Yoshimura Y, Uchida K, Jeong S, Yamaga M. Effects of Nutritional Supplements on Muscle Mass and Activities of Daily Living in Elderly Rehabilitation Patients with Decreased Muscle Mass: A Randomized Controlled Trial. J Nutr Health Aging. 2016; 20:185–91. https://doi.org/10.1007/s12603-015-0570-4 [PubMed]
- 10. Bauer JM, Verlaan S, Bautmans I, Brandt K, Donini LM, Maggio M, McMurdo ME, Mets T, Seal C, Wijers SL, Ceda GP, De Vito G, Donders G, et al. Effects of a vitamin D and leucine-enriched whey protein nutritional supplement on measures of sarcopenia in older adults, the PROVIDE study: a randomized, double-blind, placebo-controlled trial. J Am Med Dir Assoc. 2015; 16:740–7. https://doi.org/10.1016/j.jamda.2015.05.021 [PubMed]
- 11. Walker DK, Dickinson JM, Timmerman KL, Drummond MJ, Reidy PT, Fry CS, Gundermann DM, Rasmussen BB. Exercise, amino acids, and aging in the control of human muscle protein synthesis. Med Sci Sports Exerc. 2011; 43:2249–58. https://doi.org/10.1249/MSS.0b013e318223b037 [PubMed]
- 12. Burd NA, Gorissen SH, van Loon LJ. Anabolic resistance of muscle protein synthesis with aging. Exerc Sport Sci Rev. 2013; 41:169–73. https://doi.org/10.1097/JES.0b013e318292f3d5 [PubMed]
- 13. Ter Borg S, Luiking YC, van Helvoort A, Boirie Y, Schols JM, de Groot CP. Low levels of branched chain amino acids, eicosapentaenoic acid and micronutrients are associated with low muscle mass, strength and function in community-dwelling older adults. J Nutr Health Aging. 2019; 23:27–34. https://doi.org/10.1007/s12603-018-1108-3 [PubMed]
- 14. Komar B, Schwingshackl L, Hoffmann G. Effects of leucine-rich protein supplements on anthropometric parameter and muscle strength in the elderly: a systematic review and meta-analysis. J Nutr Health Aging. 2015; 19:437–46. https://doi.org/10.1007/s12603-014-0559-4 [PubMed]
- 15. Shad BJ, Thompson JL, Breen L. Does the muscle protein synthetic response to exercise and amino acid-based nutrition diminish with advancing age? a systematic review. Am J Physiol Endocrinol Metab. 2016; 311:E803–17. https://doi.org/10.1152/ajpendo.00213.2016 [PubMed]
- 16. Murphy CH, Saddler NI, Devries MC, McGlory C, Baker SK, Phillips SM. Leucine supplementation enhances integrative myofibrillar protein synthesis in free-living older men consuming lower- and higher-protein diets: a parallel-group crossover study. Am J Clin Nutr. 2016; 104:1594–1606. https://doi.org/10.3945/ajcn.116.136424 [PubMed]
- 17. Paddon-Jones D, Sheffield-Moore M, Urban RJ, Sanford AP, Aarsland A, Wolfe RR, Ferrando AA. Essential amino acid and carbohydrate supplementation ameliorates muscle protein loss in humans during 28 days bedrest. J Clin Endocrinol Metab. 2004; 89:4351–58. https://doi.org/10.1210/jc.2003-032159 [PubMed]
- 18. Evans M, Guthrie N, Pezzullo J, Sanli T, Fielding RA, Bellamine A. Efficacy of a novel formulation of l-carnitine, creatine, and leucine on lean body mass and functional muscle strength in healthy older adults: a randomized, double-blind placebo-controlled study. Nutr Metab (Lond). 2017; 14:7. https://doi.org/10.1186/s12986-016-0158-y [PubMed]
- 19. Takeuchi I, Yoshimura Y, Shimazu S, Jeong S, Yamaga M, Koga H. Effects of branched-chain amino acids and vitamin D supplementation on physical function, muscle mass and strength, and nutritional status in sarcopenic older adults undergoing hospital-based rehabilitation: A multicenter randomized controlled trial. Geriatr Gerontol Int. 2019; 19:12–17. https://doi.org/10.1111/ggi.13547 [PubMed]
- 20. Arnarson A, Gudny Geirsdottir O, Ramel A, Briem K, Jonsson PV, Thorsdottir I. Effects of whey proteins and carbohydrates on the efficacy of resistance training in elderly people: double blind, randomised controlled trial. Eur J Clin Nutr. 2013; 67:821–26. https://doi.org/10.1038/ejcn.2013.40 [PubMed]
- 21. Watson RR, Gerald JK, Preedy VR, eds. Nutrients, dietary supplements, and nutriceuticals: cost analysis versus clinical benefits. New York: Humana Press: 2011. https://doi.org/10.1007/978-1-60761-308-4
- 22. Villagra A, Merkel MC, Rodriguez Bugueiro J, Lacquaniti N, Remoli R. [Adherence to oral nutrition supplements in hospitalized patients with clinical pathology-surgical]. Nutr Hosp. 2014; 31:1376–80. https://doi.org/10.3305/nh.2015.31.3.8044 [PubMed]
- 23. Bukhari SS, Phillips BE, Wilkinson DJ, Limb MC, Rankin D, Mitchell WK, Kobayashi H, Greenhaff PL, Smith K, Atherton PJ. Intake of low-dose leucine-rich essential amino acids stimulates muscle anabolism equivalently to bolus whey protein in older women at rest and after exercise. Am J Physiol Endocrinol Metab. 2015; 308:E1056–65. https://doi.org/10.1152/ajpendo.00481.2014 [PubMed]
- 24. Devries MC, McGlory C, Bolster DR, et al: Protein leucine content is a determinant of shorter-and longer-term muscle protein synthetic responses at rest and following resistance exercise in healthy older women: a randomized, controlled trial. Am J Clin Nutr. 2018; 107:217–226. https://doi.org/10.1093/ajcn/nqx028 [PubMed]
- 25. LeBlanc AD, Schneider VS, Evans HJ, Pientok C, Rowe R, Spector E. Regional changes in muscle mass following 17 weeks of bed rest. J Appl Physiol (1985). 1992; 73:2172–78. https://doi.org/10.1152/jappl.1992.73.5.2172 [PubMed]
- 26. Thom JM, Thompson MW, Ruell PA, Bryant GJ, Fonda JS, Harmer AR, Janse de Jonge XA, Hunter SK. Effect of 10-day cast immobilization on sarcoplasmic reticulum calcium regulation in humans. Acta Physiol Scand. 2001; 172:141–47. https://doi.org/10.1046/j.1365-201X.2001.00853.x [PubMed]
- 27. Dardevet D, Rémond D, Peyron MA, Papet I, Savary-Auzeloux I, Mosoni L. Muscle wasting and resistance of muscle anabolism: the “anabolic threshold concept” for adapted nutritional strategies during sarcopenia. ScientificWorldJournal. 2012; 2012:269531. https://doi.org/10.1100/2012/269531 [PubMed]
- 28. Anthony JC, Yoshizawa F, Anthony TG, Vary TC, Jefferson LS, Kimball SR. Leucine stimulates translation initiation in skeletal muscle of postabsorptive rats via a rapamycin-sensitive pathway. J Nutr. 2000; 130:2413–19. https://doi.org/10.1093/jn/130.10.2413 [PubMed]
- 29. Atherton PJ, Smith K, Etheridge T, Rankin D, Rennie MJ. Distinct anabolic signalling responses to amino acids in C2C12 skeletal muscle cells. Amino Acids. 2010; 38:1533–39. https://doi.org/10.1007/s00726-009-0377-x [PubMed]
- 30. Kimball SR, Jefferson LS. Signaling pathways and molecular mechanisms through which branched-chain amino acids mediate translational control of protein synthesis. J Nutr. 2006; 136:227S–31S. https://doi.org/10.1093/jn/136.1.227S [PubMed]
- 31. Riazi R, Wykes LJ, Ball RO, Pencharz PB. The total branched-chain amino acid requirement in young healthy adult men determined by indicator amino acid oxidation by use of L-[1-13C]phenylalanine. J Nutr. 2003; 133:1383–89. https://doi.org/10.1093/jn/133.5.1383 [PubMed]
- 32. Kim JS, Ro SH, Kim M, Park HW, Semple IA, Park H, Cho US, Wang W, Guan KL, Karin M, Lee JH. Sestrin2 inhibits mTORC1 through modulation of GATOR complexes. Sci Rep. 2015; 5:9502. https://doi.org/10.1038/srep09502 [PubMed]
- 33. Wang X, Proud CG. The mTOR pathway in the control of protein synthesis. Physiology (Bethesda). 2006; 21:362–69. https://doi.org/10.1152/physiol.00024.2006 [PubMed]
- 34. Blomstrand E, Eliasson J, Karlsson HK, Köhnke R. Branched-chain amino acids activate key enzymes in protein synthesis after physical exercise. J Nutr. 2006; 136:269S–73S. https://doi.org/10.1093/jn/136.1.269S [PubMed]
- 35. Xu ZR, Tan ZJ, Zhang Q, Gui QF, Yang YM. The effectiveness of leucine on muscle protein synthesis, lean body mass and leg lean mass accretion in older people: a systematic review and meta-analysis. Br J Nutr. 2015; 113:25–34. https://doi.org/10.1017/S0007114514002475 [PubMed]
- 36. Dijk FJ, van Dijk M, Walrand S, van Loon LJ, van Norren K, Luiking YC. Differential effects of leucine and leucine-enriched whey protein on skeletal muscle protein synthesis in aged mice. Clin Nutr ESPEN. 2018; 24:127–33. https://doi.org/10.1016/j.clnesp.2017.12.013 [PubMed]
- 37. Yoshimura Y, Wakabayashi H, Yamada M, Kim H, Harada A, Arai H. Interventions for treating sarcopenia: a systematic review and meta-analysis of randomized controlled studies. J Am Med Dir Assoc. 2017; 18:553.e1–16. https://doi.org/10.1016/j.jamda.2017.03.019 [PubMed]
- 38. Cruz-Jentoft AJ, Landi F, Schneider SM, Zúñiga C, Arai H, Boirie Y, Chen LK, Fielding RA, Martin FC, Michel JP, Sieber C, Stout JR, Studenski SA, et al. Prevalence of and interventions for sarcopenia in ageing adults: a systematic review. Report of the international sarcopenia initiative (EWGSOP and IWGS). Age Ageing. 2014; 43:748–59. https://doi.org/10.1093/ageing/afu115 [PubMed]
- 39. Malafarina V, Uriz-Otano F, Iniesta R, Gil-Guerrero L. Effectiveness of nutritional supplementation on muscle mass in treatment of sarcopenia in old age: a systematic review. J Am Med Dir Assoc. 2013; 14:10–17. https://doi.org/10.1016/j.jamda.2012.08.001 [PubMed]
- 40. Yoshimura Y, Bise T, Shimazu S, Tanoue M, Tomioka Y, Araki M, Nishino T, Kuzuhara A, Takatsuki F. Effects of a leucine-enriched amino acid supplement on muscle mass, muscle strength, and physical function in post-stroke patients with sarcopenia: a randomized controlled trial. Nutrition. 2019; 58:1–6. https://doi.org/10.1016/j.nut.2018.05.028 [PubMed]
- 41. Bloomgarden Z. Diabetes and branched-chain amino acids: what is the link? J Diabetes. 2018; 10:350–52. https://doi.org/10.1111/1753-0407.12645 [PubMed]
- 42. Zheng Y, Li Y, Qi Q, Hruby A, Manson JE, Willett WC, Wolpin BM, Hu FB, Qi L. Cumulative consumption of branched-chain amino acids and incidence of type 2 diabetes. Int J Epidemiol. 2016; 45:1482–92. https://doi.org/10.1093/ije/dyw143 [PubMed]
- 43. Isanejad M, LaCroix AZ, Thomson CA, Tinker L, Larson JC, Qi Q, Qi L, Cooper-DeHoff RM, Phillips LS, Prentice RL, Beasley JM. Branched-chain amino acid, meat intake and risk of type 2 diabetes in the women’s health initiative. Br J Nutr. 2017; 117:1523–30. https://doi.org/10.1017/S0007114517001568 [PubMed]
- 44. Cummings NE, Williams EM, Kasza I, Konon EN, Schaid MD, Schmidt BA, Poudel C, Sherman DS, Yu D, Arriola Apelo SI, Cottrell SE, Geiger G, Barnes ME, et al. Restoration of metabolic health by decreased consumption of branched-chain amino acids. J Physiol. 2018; 596:623–45. https://doi.org/10.1113/JP275075 [PubMed]
- 45. Nagata C, Nakamura K, Wada K, Tsuji M, Tamai Y, Kawachi T. Branched-chain amino acid intake and the risk of diabetes in a Japanese community: the takayama study. Am J Epidemiol. 2013; 178:1226–32. https://doi.org/10.1093/aje/kwt112 [PubMed]
- 46. Din US, Brook MS, Selby A, Quinlan J, Boereboom C, Abdullah H, Franchi M, Narici MV, Phillips BE, Williams JW, Rathmacher JA, Wilkinson DJ, Atherton PJ, Smith K. A double-blind placebo controlled trial into the impacts of HMB supplementation and exercise on free-living muscle protein synthesis, muscle mass and function, in older adults. Clin Nutr. 2019; 38:2071–78. https://doi.org/10.1016/j.clnu.2018.09.025 [PubMed]
- 47. Chen LK, Woo J, Assantachai P, Auyeung TW, Chou MY, Iijima K, Jang HC, Kang L, Kim M, Kim S, Kojima T, Kuzuya M, Lee JS, et al. Asian working group for sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J Am Med Dir Assoc. 2020; 21:300–07.e2. https://doi.org/10.1016/j.jamda.2019.12.012 [PubMed]
- 48. Young AM, Kidston S, Banks MD, Mudge AM, Isenring EA. Malnutrition screening tools: comparison against two validated nutrition assessment methods in older medical inpatients. Nutrition. 2013; 29:101–06. https://doi.org/10.1016/j.nut.2012.04.007 [PubMed]
- 49. Wyka J, Biernat J, Mikołajczak J, Piotrowska E. Assessment of dietary intake and nutritional status (MNA) in polish free-living elderly people from rural environments. Arch Gerontol Geriatr. 2012; 54:44–49. https://doi.org/10.1016/j.archger.2011.02.001 [PubMed]
- 50. Chang CS, Chang YF, Wang MW, Chen CY, Chao YJ, Chang HJ, Kuo PH, Yang YC, Wu CH. Inverse relationship between central obesity and osteoporosis in osteoporotic drug naive elderly females: the tianliao old people (TOP) study. J Clin Densitom. 2013; 16:204–11. https://doi.org/10.1016/j.jocd.2012.03.008 [PubMed]
- 51. Pietrobelli A, Rubiano F, St-Onge MP, Heymsfield SB. New bioimpedance analysis system: improved phenotyping with whole-body analysis. Eur J Clin Nutr. 2004; 58:1479–84. https://doi.org/10.1038/sj.ejcn.1601993 [PubMed]
- 52. Janssen I, Heymsfield SB, Baumgartner RN, Ross R. Estimation of skeletal muscle mass by bioelectrical impedance analysis. J Appl Physiol (1985). 2000; 89:465–71. https://doi.org/10.1152/jappl.2000.89.2.465 [PubMed]
- 53. Wu CH, Chen KT, Hou MT, Chang YF, Chang CS, Liu PY, Wu SJ, Chiu CJ, Jou IM, Chen CY. Prevalence and associated factors of sarcopenia and severe sarcopenia in older taiwanese living in rural community: the tianliao old people study 04. Geriatr Gerontol Int. 2014 (Suppl 1); 14:69–75. https://doi.org/10.1111/ggi.12233 [PubMed]
- 54. Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, Martin FC, Michel JP, Rolland Y, Schneider SM, Topinková E, Vandewoude M, Zamboni M, European Working Group on Sarcopenia in Older People. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010; 39:412–23. https://doi.org/10.1093/ageing/afq034 [PubMed]
- 55. Chen LK, Liu LK, Woo J, Assantachai P, Auyeung TW, Bahyah KS, Chou MY, Chen LY, Hsu PS, Krairit O, Lee JS, Lee WJ, Lee Y, et al. Sarcopenia in Asia: consensus report of the Asian working group for sarcopenia. J Am Med Dir Assoc. 2014; 15:95–101. https://doi.org/10.1016/j.jamda.2013.11.025 [PubMed]
- 56. Murakami S, Sawaki K, Shiraishi Y, Ohtani M. Effects of oral supplementation with a mixture of amino acids on immune function in athletes at a summer training camp for track and field and long-distance runners. J Jap Soc Clin Sports Med. 2011; 19:280–289.


