Introduction
Atrial fibrillation (AF), which is characterized by rapid and irregular beating of the atria, and known as the most common type of cardiac arrhythmia. According to the epidemiologic data, the prevalence of AF ranged from 0.7% to 1% in the general population, and up to 8% in elders greater than 80 years [1, 2]. Meanwhile, AF increases the risk of stroke, congestive heart failure, sudden cardiac death, and increase the rate of substantial morbidity and mortality for about 2 folds [3].
AF is often associated with complications such as hypertension, valvular heart disease, coronary artery disease, heart failure, hyperthyroidism, structural heart diseases, and is also clearly heritable [4, 5]. The important role of genetic factors in the pathogenesis of AF has shown by the identification of AF-causing mutations or rare variants in some families with lone AF, which occurs in structurally normal hearts and without known risk factors [6–8]. Meanwhile, in general AF, the non-hypothesis-driven genome-wide association studies (GWAS) and the candidate gene based association studies have identified a panel of common variants confer risk to AF [9–11]. These studies have set up a key role for the genetic background in generating for AF.
GWAS investigate associations between genomic variants and a disease or trait at the whole genome level without priori assumptions of genomic locations or potential functions of candidate genes. In this case, most of the identified single nucleotide polymorphisms (SNPs) associated with disease were found located within intergenic or intronic genomic regions, and whether the positive SNPs have a real biological function is unknown, and the real target gene need to be further studied [12, 13]. For example, SNP rs2200733 on chromosome 4p25 is the first risk variant for AF identified by GWAS and is the most robustly replicated AF locus to date. The gene that closest proximity to rs2200733 and other AF susceptibility variants in 4q25 is the PITX2. Studies in mice showed that pitx2 haplo-insufficiency promotes an atrial arrhythmia [14]. However, functional evidence about the mechanisms linking these non-coding variants with PITX2 or the incidence of AF is limited, until a recent study found that these non-coding variants in 4q25 possessing a long-range enhancer–promoter interactions and exert as a transcriptional regulatory directed function at PITX2 [15]. Understanding the biological nature of non-coding variants associated with AF can enable us to point the real causal genes causing AF and provide insight into the mechanism of AF.
Considering one of the most important challenges of AF genetic study is to elucidate functional mechanisms that how the susceptibility loci modulate AF risk, in the current study, we summarized the results of the studies including variants identified by GWAS and candidate gene based association studies, and give a comprehensive functional annotation of all these AF susceptibility loci. The non-synonymous SNPs were first identified and classified as functional SNPs, and for SNPs in the non-coding region, we try to predict their potential functions including microRNA binding, promoter activity, enhancer activity, transcription factors binding activity, expression quantitative trait loci (eQTL), and long-range transcriptional regulatory function. Our results may provide an encyclopedia of AF susceptibility SNPs and shed light on the functional mechanisms of AF variants identified through genetic studies.
Results
AF susceptibility loci
Through searching the public databases including GWAS catalog (https://www.ebi.ac.uk/gwas/), GWAS central (https://www.gwascentral.org), and literatures in Pubmed, Embase and Medline, we included 18 AF GWAS and exome-wide association study (EWAS) in our study, which published from 2007 to 2019 (Table 1). The workflow of the current study is shown in Figure 1. Participants of these studies were mainly European ancestry (15 of 18 studies), and the rest were East Asian (Korean ancestry and Japanese) (Table 1). A total of 338 SNPs (refer as index SNPs) passed the multiple corrections (P<5×10-8 or corresponding multiple correction threshold) and showed a significant association with AF in GWAS and EWAS (Figure 2 and Supplementary Table 1). We also include 40 common SNPs which showed significant associated with AF in case control or population prospective study in candidate gene based analysis, or replication study of GWAS loci (Figure 2 and Supplementary Table 1). Totally, we included 378 AF susceptibility SNPs in our further functional annotation.
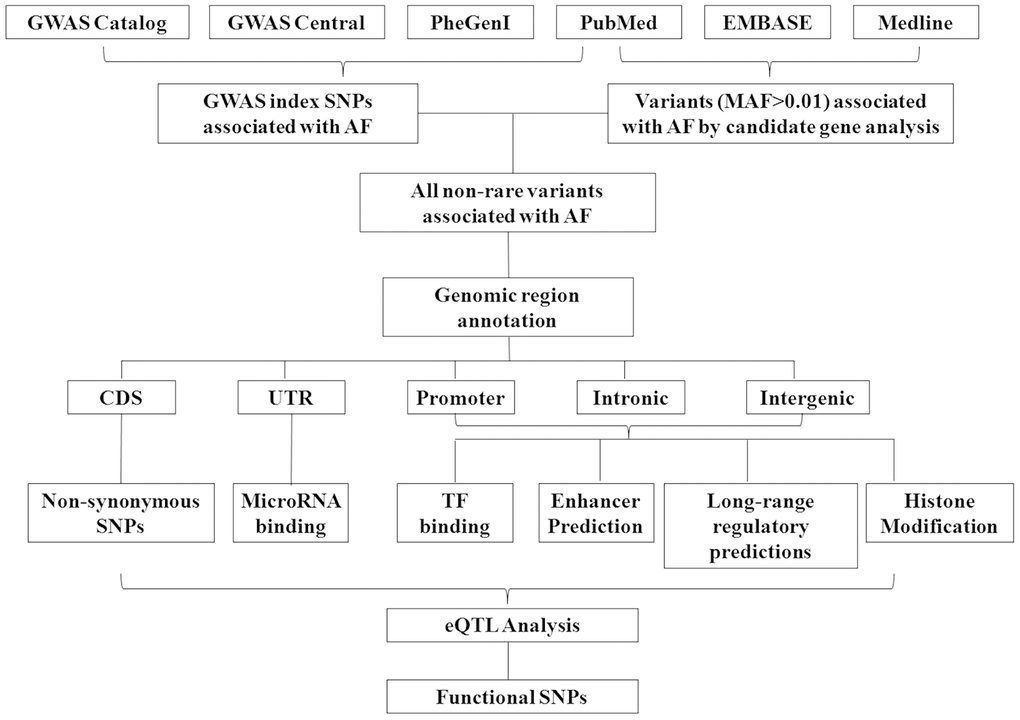
Figure 1. Workflow of the annotation of susceptibility SNPs associated with atrial fibrillation.
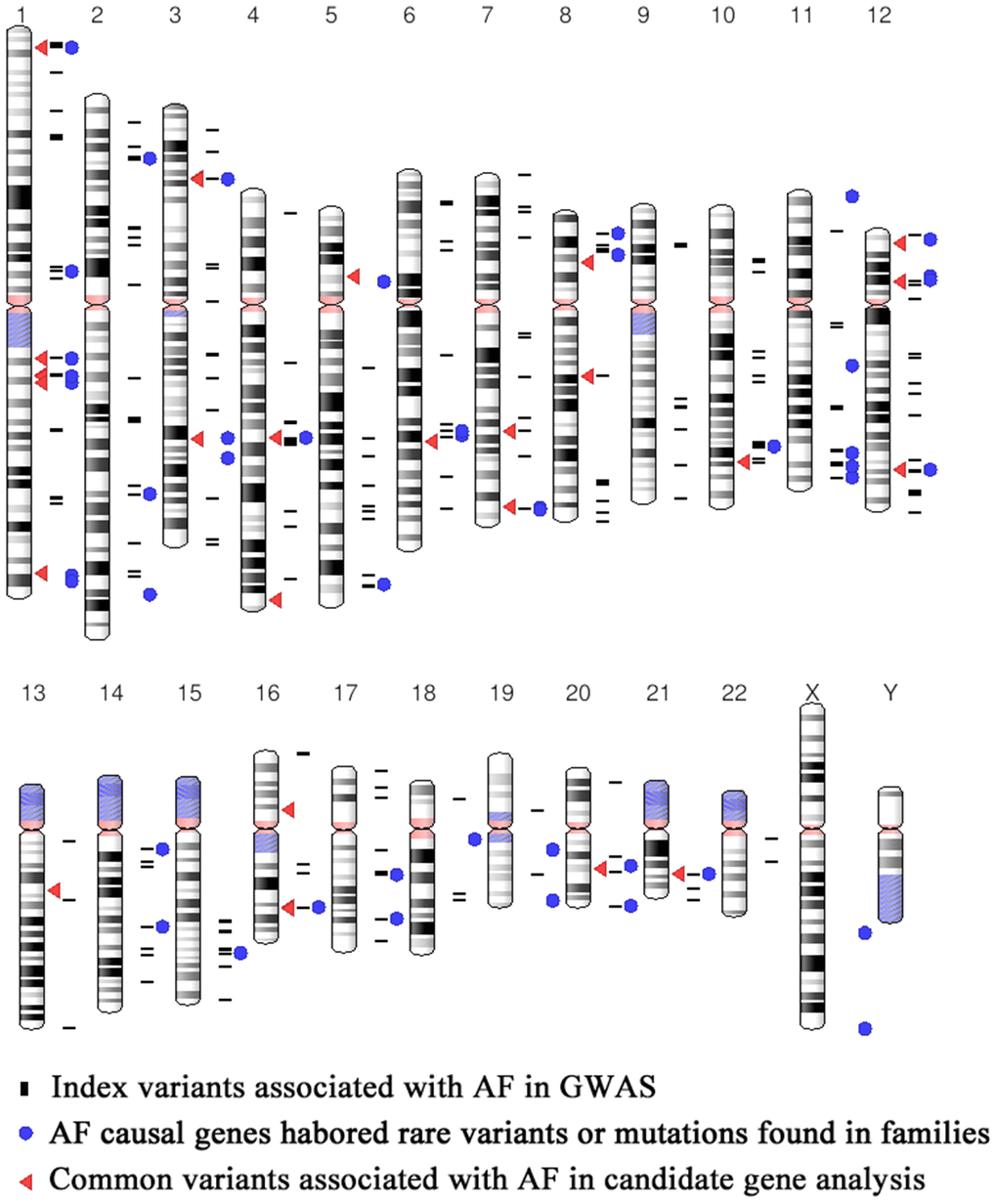
Figure 2. Distribution of the 378AF susceptibility SNPs and AF causal genes.
Table 1. Included studies with SNPs associated with AF from 2007 to 2019.
| Number | Studies | Year | Discovery population | Replication population |
| enome-wide association study | ||||
| 1 | Nielsen JB. et al [16] | 2018 | 60,620 European ancestry cases and 970,216 European ancestry controls | NA |
| 2 | Roselli C. et al [17] | 2018 | 55,114 European ancestry cases and 482,295 European ancestry controls, 8,180 Japanese ancestry cases and 28,612 Japanese ancestry controls, 1,307 African American ancestry cases and 7,660 African American ancestry controls, 845 Hispanic cases and 4,177 Hispanic controls | NA |
| 3 | Nielsen JB. et al [18] | 2018 | 6,337 European ancestry cases and 61,607 European ancestry controls | 30,679 European ancestry cases and 278,895 European ancestry controls |
| 4 | Thorolfsdottir RB. et al [19] | 2018 | 14,710 cases and 373,897 controls from Iceland, 14,792 cases and 393,863 controls from the UK Biobank | 9,204 cases and 76,161 controls, European ancestry |
| 5 | Lee JY. et al [20] | 2017 | 672 Korean ancestry cases and 3,700 Korean ancestry controls | 200 Korean ancestry cases and 1,812 Korean ancestry controls |
| 6 | Christophersen IE. et al [21] | 2017 | GWAS: 18,398 individuals with atrial fibrillation and 91,536 referents, EWAS: 22,806 AF cases and 132,612 referents. | NA |
| 7 | Low SK. et al [22] | 2017 | 8,180 Japanese ancestry cases and 28,612 Japanese ancestry controls | 3,120 Japanese ancestry cases and 125,064 Japanese ancestry controls, 15,993 European ancestry cases and113,719 European ancestry controls |
| 8 | Thorolfsdottir RB. et al [23] | 2017 | 14,255 AF cases and 374,939 controls, Iceland | 2,002 non-Icelandic cases and 12,324 controls |
| 9 | Yamada Y. et al [24] | 2017 | 884 patients with atrial fibrillation and 12,282 controls, Japanese | NA |
| 10 | Lubitz SA. et al [25] | 2016 | 1,734 individuals with and 9,423 without AF, European ancestry | NA |
| 11 | Kertai MD. et al [26] | 2015 | 620 European ancestry cases, 257 European ancestry controls | 220 cases and 84 controls |
| 12 | Sinner MF. et al [27] | 2014 | 6,707 AF cases and 52,426 controls in Europeans, 843 AF and 3,350 controls in Japanese | 6,691 AF cases and 17,144 controls in Europeans, 1,618 AF cases and 17,190 controls |
| 13 | Ellinor PT. et al [28] | 2012 | 6,707 European ancestry cases and 52,426 European ancestry controls | 5,381 European ancestry cases and 10,030 European ancestry controls |
| 14 | Ellinor PT. et al [29] | 2010 | 1,335 European ancestry lone AF cases and 12,844 European ancestry controls | 1,164 European ancestry AF cases, 3,607 European ancestry controls |
| 15 | Gudbjartsson DF. et al [30] | 2009 | 2,385 European ancestry cases and 33,752 European ancestry controls | 2,427 European ancestry cases and 3,379 European ancestry controls, 286 Han Chinese ancestry cases and 2,763 Han Chinese ancestry controls |
| 16 | Benjamin EJ. et al [31] | 2009 | 3,413 cases and 37,105 referents, European ancestry | 2,145 cases and 4,073 controls, European ancestry |
| 17 | Larson MG. et al [32] | 2007 | 151 cases and 1,190 controls from 310 families | NA |
| 18 | Gudbjartsson DF. et al [33] | 2007 | 550 European ancestry cases and 4,476 European ancestry controls | 3,030 European ancestry cases and 14,780 European ancestry controls, 333 Han Chinese ancestry cases and 2,836 Han Chinese ancestry controls |
| Candidate gene based association study | ||||
| 19 | Cao H. et al [34] | 2019 | 828 patients and 834 controls in Chinese population | NA |
| 20 | Xiong H. et al [35] | 2019 | 944 AF patients and 981 non-AF controls in Chinese population | 732 cases and 1,291 controls in Chinese population |
| 21 | Wang P. et al [36] | 2018 | 1,164 AF patients and 1,460 controls | NA |
| 21 | Zaw KTT. et al [37] | 2017 | 452 cases and 1,981 controls in Japanese | NA |
| 22 | Feng W. et al [38] | 2017 | 300 AF cases and 300 controls | NA |
| 23 | Nakano Y. et al [39] | 2016 | 577 cases and 1935 controls in Japanese | NA |
| 24 | Seppälä I. et al [40] | 2016 | 1,834 individuals with AF and 7,159 unaffected individuals | NA |
| 25 | Fang Z. et al [41] | 2016 | 1,150 AF cases and 1,150 AF-free controls in Chinese | NA |
| 26 | Wang C. et al [42] | 2016 | 1,127 unrelated AF patients and 1,583 non-AF subjects | NA |
| 27 | Fang Z. et al [41] | 2016 | 597 AF cases and 996 AF-free controls in Chinese | NA |
| 28 | Zhang R. et al [43] | 2016 | 1,132AF patients and 1,206 controls | NA |
| 29 | Roberts JD. et al [44] | 2016 | 2,601 incident of AF in a total of 17,529 participant | NA |
| 30 | Luo Z. et al [45] | 2016 | 889 AF patients and 1015 controls, Chinese | NA |
| 31 | Chen S. et al [46] | 2015 | 941 cases and 562 controls, Chinese | 2,113 cases and 3,381 controls |
| 32 | Liu Y. et al [47] | 2015 | 597 AF patients and 996 non-AF controls in Chinese | NA |
| 33 | Rosenberg MA. et al [48] | 2014 | 879 incident AF in a total 3,309 participants | NA |
| 34 | Andreasen L. et al [49] | 2014 | 657 patients diagnosed with AF and a control group comprising 741 individuals | NA |
| 35 | Luo Z. et al [50] | 2014 | 889 AF patients and 1,015 controls in Chinese | NA |
| 36 | Voudris KV. et al [51] | 2014 | 509 patients of whom 203 with AF | NA |
| 37 | Liu Y. et al [52] | 2014 | 597 AF patients and 996 non-AF controls in Chinese | NA |
| 37 | Andreasen L. et al [49] | 2014 | 657 AF cases and 741 controls, European ancestry | NA |
| 38 | Lin H. et al [53] | 2014 | 948 cases and 3,330 controls, European ancestry | NA |
| 39 | Adamsson S. et al [54] | 2014 | 520 incident AF in a total 3,900 subjects, European ancestry | 2,247 cases, 2,208 controls |
| 40 | Cao H. et al [55] | 2014 | 840 AF patients and 845 controls in Chinese | NA |
| 41 | Marott SC. et al [56] | 2013 | 358 patients with lone AF, 299 non-lone AF, and 741 controls, European ancestry | NA |
| 42 | Jeff JM. et al [57] | 2014 | 1,288 patients with cardiac surgery, European ancestry | NA |
| 43 | VoudrisKv. et al [51] | 2014 | 509 patients with cardiac surgery, European ancestry | NA |
| 44 | Ilkhanoff L. et al [58] | 2014 | 241 cases and 3,144 controls, African Americans | NA |
| 45 | Marott SC. et al [56] | 2013 | 2,570 AF events in 69,611 participants, European ancestry | NA |
| 46 | Andreasen L. et al [59] | 2013 | 358 patients with lone AF and a control of 751 individuals, European ancestry | NA |
| 47 | Olesen MS. et al [60] | 2012 | 209 patients with early-onset lone AF, and a control group consisting of 534 individuals free of AF | NA |
| 48 | Schnabel RB. et al [61] | 2011 | European (n=18,524; 2260 AF cases in a total 18,524individuals cohort in European ancestry), 263 AF cases in a total of 3,662 African American descent. | 468 AF cases and 438 controls |
| 49 | Wirka RC. et al [62] | 2011 | 384 early onset lone AF cases and 3,010 population control | NA |
| 50 | Li C. et al [63] | 2011 | 650 AF patients and 1,447 non-AF controls | NA |
| 51 | Lubitz SA. et al [64] | 2010 | 790 case and 1,177 control subjects, European ancestry | 5,066 case and 30,661 referent subjects, European ancestry |
| 52 | Roberts JD. et al [65] | 2010 | 620 AF cases and 2,446 healthy controls | NA |
| 53 | Ren X. et al [66] | 2010 | 384 sporadic AF patients and 844 controls | NA |
| 54 | Shi L. et al [67] | 2009 | 383 AF patients versus 851 non-AF controls | NA |
| 55 | Kääb S. et al [68] | 2009 | 3,508 AF cases and 12,173 controls, European ancestry | NA |
| 56 | Sinner MF. et al [69] | 2008 | 1,207 AF-cases and 2,475 controls | NA |
| 57 | Giusti B. et al [70] | 2007 | 456 AF patients and 912 matched controls | NA |
Genomic region annotation using Variant Effect Predictor (http://asia.ensembl.org/Homo_sapiens/Tools/VEP, GRCh38) showed that only a small portion of GWAS index AF SNPs was located in exon of known genes (21/338, 6.21%), 63.31% were found in introns (214/338), and 29.29% locate in intergenic regions (99/338) (Figure 3). In candidate gene based analysis, 50% identified AF related variants locate in intron, and the proportion of non-synonymous variants associated with AF (32.50%, 13/40) was higher than in GWAS index SNPs (2.936%, 10/338) (Figure 3).
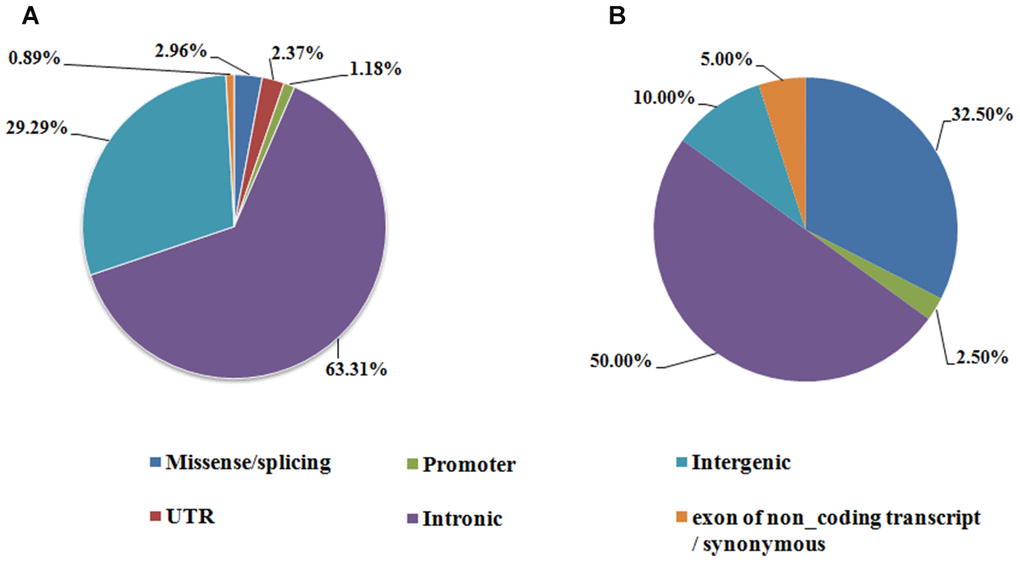
Figure 3. Genomic region annotation distribution of the AF susceptibility SNPs. (A) Index SNPs associated with AF identified in GWAS, (B) AF susceptibility SNPs identified by candidate gene based analysis.
Functional annotation of AF susceptibility SNPs in UTR
8 AF susceptibility SNPs were found in the UTR of protein-coding genes, and 4 of them were predicted to alter the microRNA binding ability predicted by MirSNP (http://bioinfo.bjmu.edu.cn/mirsnp/search) and miRNASNP (http://bioinfo.life.hust.edu.cn/). Rs1049334 in the 3’UTR of CAV1 was predicted change the binding with hsa-miR-125a-3p, hsa-miR-3620, hsa-miR-4299, hsa-miR-4726-3p, hsa-miR-4783-3p and hsa-miR-497-3p. Rs13385 in the 3’UTR of HBEGF was expected to alter the binding with hsa-miR-1207-5p and hsa-miR-4763-3p. Rs7508 in 3’UTR of ASAH1 may change the binding of hsa-miR-134, hsa-miR-3118, hsa-miR-5190 and hsa-miR-628-5p. Rs951366 in NUCKS1 was found in the binding region of hsa-miR-3929, hsa-miR-4419b, hsa-miR-4478, hsa-miR-4649-3p and hsa-miR-485-5p.
Functional annotation of AF susceptibility SNPs in non-coding regions
According to the data of the chromatin state and modification of histone binding, a total of 250 SNPs in non-coding regions were identified as located in enhancer regions or might affect the histone mark of promoters and enhancers (Supplementary Table 1), and further analysis found that 65 of them may change the situation of interaction with transcription factors (Supplementary Table 1). 40 transcription factors were found interact with these SNPs. After corrected by genome-wide expected binding ability, these SNPs were significantly enriched for disruption of 3 TFs including STAT6 (P=0.02), REST (P=0.05) and NFIC (P=3.86x10-3) (Figure 4).
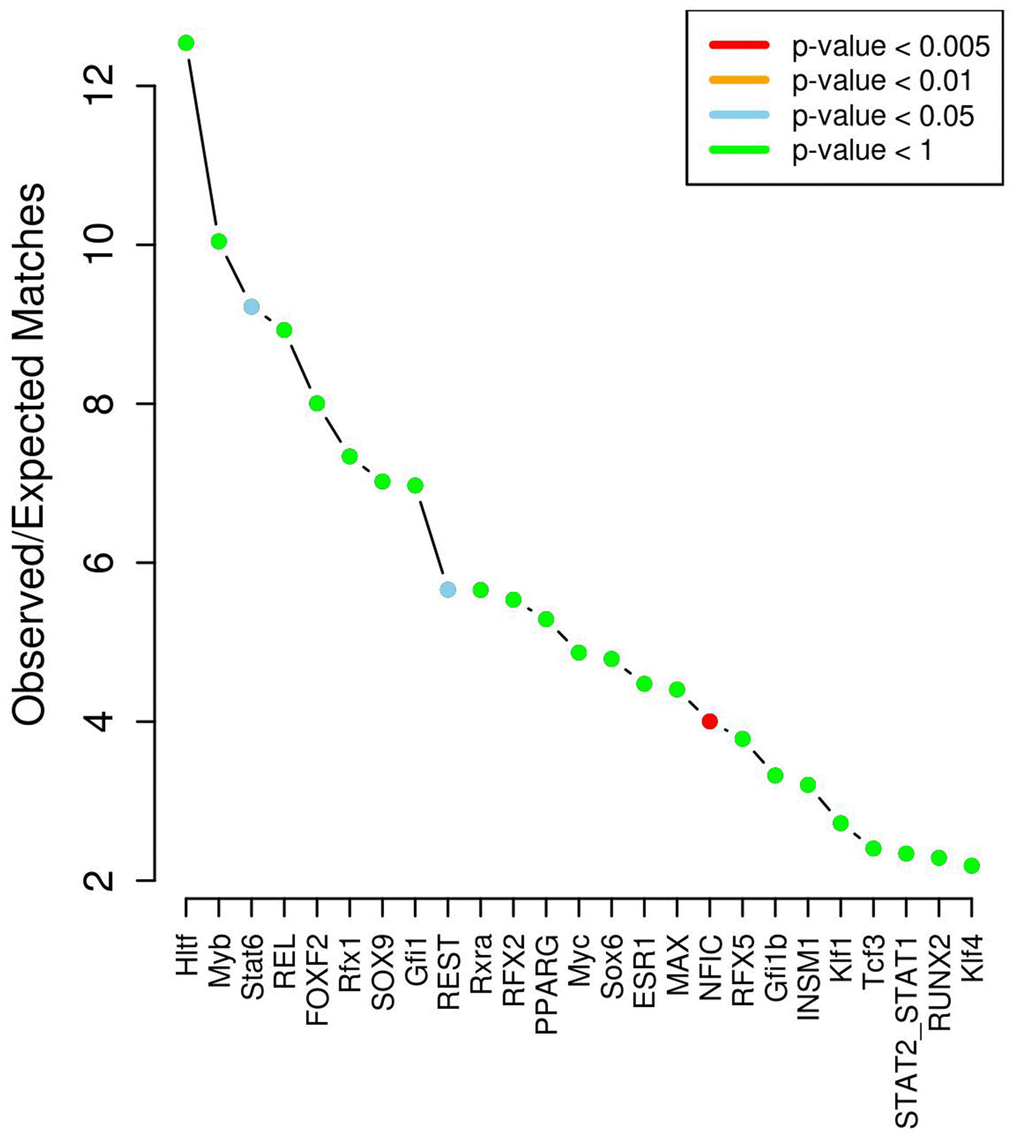
Figure 4. Transcription factor enrichment analysis results.
eQTL analyses
SNPs in the non-coding region may associate with the expression levels and act as eQTL. We assessed the data from GTEx database (https://gtexportal.org/home/) and evaluate whether AF susceptibility SNPs affect the target gene expression levels. The results showed that 151 SNPs can affect the expression levels of a total of 328 target genes, and 81 of them associated with the expression levels of the closest gene (Supplementary Table 1). Combined with the TF binding data, 39 eQTL effect SNPs were found may alter the binding with transcription factors (Supplementary Table 1).
Long-range transcriptional regulatory function predictions
We used 3dSNP database (http://cbportal.org/3dsnp/) to analyze whether AF susceptibility SNPs affect distal target genes through topological interactions and function as long-range transcriptional regulatory elements. Results indicated that a total of 211 SNPs interact with distal target genes, and 104 of them exert as an eQTL effect (Supplementary Table 1).
Gene ontology and pathway enrichment analyses of eQTL targeted genes
eQTL targeted genes of AF were mapped onto Gene ontology (GO) database using three primary categories including molecular function, protein class and biological process via PANTHER (http://www.pantherdb.org). The results showed that AF related genes were mainly enriched in binding, cellular process, metabolic process, protein modifying enzyme, gene-specific transcriptional regulator and membrane traffic protein (Figure 5).
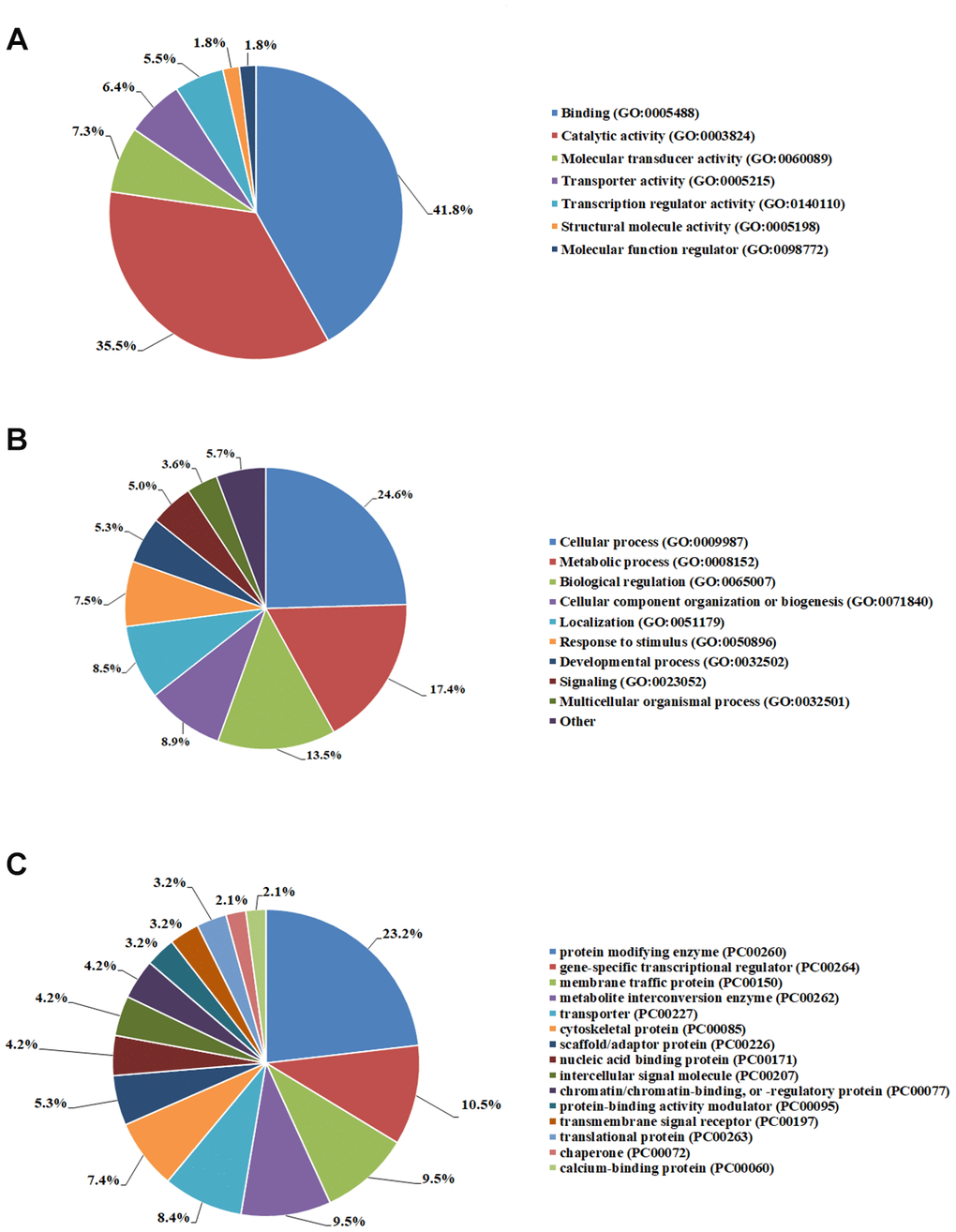
Figure 5. Gene ontology analyses of AF eQTL targeted genes. (A) Molecular function. (B) Biological process. (C) Protein class.
All eQTL targeted genes of AF were subjected to pathway enrichment analysis in the Search Tool for the Retrieval of Interacting Genes (STRING, v11.0, http://string-db.org). Statistical enrichment tests were executed on gene lists within the STRING by Gene Ontology and pathway annotations. The results uncovered some signaling pathway may play roles in AF including “organelle organization”, “striated muscle cell development”, “nuclear migration”, “endomembrane system organization” and “striated muscle cell differentiation” (Table 2).
Table 2. Significantly enriched pathways of AF eQTL targeted genes.
| #term ID | term description | observed gene count | false discovery rate | matching proteins in your network (labels) |
| GO:0006996 | organelle organization | 56 | 2.0x10-4 | BAZ2A,BRD8,CASQ2,CAV1,CAV2,CDC23,CEP68,CFL2,CHRAC1,CISD2,CTC1,DEK,DNM1L,GATAD1,GORAB,HDAC5,HEATR2,HIP1R,HPS6,IMMT,KANSL1,KDM1B,KIF3C,MAPT,MTHFR,MTSS1,MYH10,MYOZ1,NDUFB10,NEK6,NEURL1,NKX2-5,NR3C1,NSF,NUCKS1,PCID2,PCM1,PEX26,PFDN1,PTK2,RAB29,RAB3IP,REEP1,REEP2, REEP4, RPL3L, SCMH1, SEC24C, SYNE2, TMEM70, UBE2D3, USP36, VPS37B, WIPF1, ZNF462, ZPBP2 |
| GO:0055002 | striated muscle cell development | 8 | 0.03 | CASQ2, CAV2, CFL2, CHRNB1, FLNC, MYH10, MYOZ1, NKX2-5 |
| GO:0007097 | nuclear migration | 4 | 0.04 | MYH10, PCM1, PTK2, SYNE2 |
| GO:0010256 | endomembrane system organization | 12 | 0.04 | CAV1, CAV2, MTSS1, MYH10, NEK6, RAB29, REEP1, REEP2, REEP4, SYNE2, VPS37B, ZPBP2 |
| GO:0051146 | striated muscle cell differentiation | 9 | 0.04 | BMP4, CASQ2, CAV2, CFL2, CHRNB1, FLNC, MYH10, MYOZ1, NKX2-5 |
Discussion
Population-based genetic analysis including GWAS and candidate gene based analysis has identified several SNPs associated with the risk of atrial fibrillation, here, we summarized the current results of the common variants conferred risk to AF and totally including 378 SNPs. Considering most of the AF susceptibility SNPs were located in the non-coding genomic regions, we give a comprehensive functional annotation of all these AF susceptibility SNPs through microRNA binding prediction, promoter and enhancer activity prediction, transcription factors binding activity prediction, eQTL analysis, and long-range transcriptional regulatory function predictions.
Our functional annotation found that 151 AF susceptibility SNPs showed an eQTL effect, and 238 SNPs in non-coding regions were identified as located in enhancer regions or might affect the histone mark of promoters and enhancers, Previous studies also showed that 50-60% of the traits associated non-coding variants identified by GWAS were found located in DNase I hypersensitivity regions [71, 72], and these results also suggest that most of the SNPs identified by the GWAS as predisposing to atrial fibrillation may have biological functions and exert regulatory effects. Our results also showed that only 81 of the 151 eQTL SNPs associated with the expression levels of the closest genes, and a total of 328 target genes were identified affected by AF susceptibility SNPs. Our results identify novel genes that may be associated with the occurrence or development of AF. For example, rs35006907 located in 139bp upstream of a non-coding RNA gene LINC00964, was found associated with the expression level of MTSS1 gene (P=2.02×10-18) in the left ventricle, which 119 kb downstream of rs35006907. Rs35006907 was predicted within an enhancer in several types of tissues including the right ventricle and right atrium, and long-range transcriptional regulatory function predictions also showed that rs35006907 and its located enhancer can interact with MTSS1 through long-range 3D chromatin loops. MTSS1 can promote actin assembly at intercellular junctions and a recent functional study indicated that rs35006907 showed a cardioprotective effect [73].
Another interesting finding is about AF susceptibility loci in 10q22, which was reported as the first genetic locus for familial atrial fibrillation by Brugada R et al in 1997, and SNPs including rs10824026 [28, 44], rs7394190 [21], rs6480708 [17] and rs60212594 [17] in 10q22 and upstream of SYNPO2L gene were found robustly associated with AF in several GWAS project. What is more, a missense variant in SYNPO2L, rs3812629 (p.Pro707Leu) was found to confer risk to AF in the Framingham population by Whole Exome Sequencing in Atrial Fibrillation [25] (Figure 6A). However, our eQTL analysis using GTEx data showed that all these GWAS positive AF SNPs including rs10824026, rs7394190, rs6480708, and rs60212594 were strong associated with the expression level of MYOZ1 in human atrial appendage tissue with a P value from 1.3x10-28 to 1.4x10-45 (Figure 6B). Furthermore, the missense variant in SYNPO2L, rs3812629 (p.Pro707Leu), which confer risk to AF, was also found associated with MYOZ1 expression level in human atrial appendage tissue, and the median normalized expression level of MYOZ1 in homozygous risk allele GG carriers was -0.28 and extremely lower than in heterozygous GA carriers (0.94) (Figure 6B). MYOZ1 encode myozenin 1, which is an intracellular binding protein involved in linking Z-disk proteins, and was known as a calcineurin-interacting protein, and help tether calcineurin to the sarcomere of skeletal and cardiac muscle [74–76]. Mutations in MYOZ1 were found in the patient with dilated cardiomyopathy [77–78]. These results suggested that MYOZ1, but not SYNPO2L is the causal gene of AF.
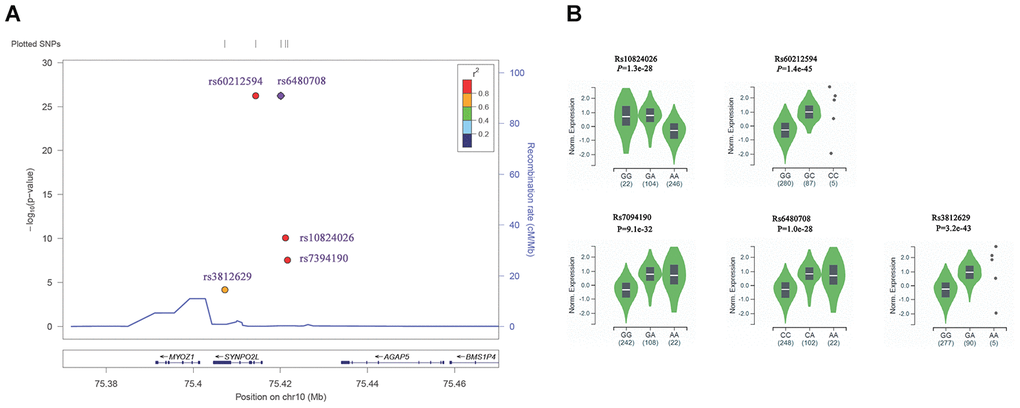
Figure 6. Association of SNPs in 10q22 with AF and eQTL analysis. (A) Regional plots for significant association with AF in 10q22. The P value was obtained from GWAS catalog database. SNPs plotted by their positions (UCSC hg19) on the corresponding chromosome against –log10 (P). Estimated recombination rates from 1000 genomes EUR populations were plotted in blue to reflect the local linkage disequilibrium (LD) structure on a secondary y axis. The most significant lead SNP (diamond) is denoted with the SNP identification number. Flanking SNPs (circles) are color-coded to represent the pairwise r2 measure of LD with the lead SNP: red, r2 ≥ 0.8; orange, 0.6 ≤ r2< 0.8; green, 0.4 ≤ r2<0.6; light blue, 0.2 ≤ r2<0.4; blue, r2< 0.2. These plots were generated by Locuszoom (https://statgen.sph.umich.edu/locuszoom/). (B) eQTL analysis showed that the association between AF susceptibility SNPs in 10q22 with the expression level of MYOZ1 in human atrial appendage tissues (n=372). eQTL analysis were performed using GTEx data.
Previously genetic studies in familial or sporadic AF have identified numerous mutations or rare variants that putatively cause AF [5, 79–83], and to recently, 44 genes that putatively cause AF were mapped to pathway of ion channels/ion channels related (ABCC9, HCN4, KCNA5, KCNE1, KCND3, JPH2, KCNE2, KCNE3, KCNE4, KCNE5, KCNH2, KCNJ2, KCNJ5, KCNJ8, KCNK3,KCNN3, KCNQ1, RYR2, SCN1B, SCN2B, SCN3B, SCN4B, SCN5A, SCN10A), transcription factors (GATA4, GATA5, GATA6, NKX2-5, NKX2-6, PITX2, SHOX2, SOX5, TBX5, ZFHX3), myocardial structural components (GJA1, GJA5, LMNA, MYH6, MYL4, SYNE2), signaling, protein turnover and others (GREM2, NPPA,SH3PXD2A, PLN). Compared to the list AF susceptibility genes including the 328 eQTL target genes what we have identified and combined with the closest gene of GWAS index SNPs, only 10 genes including HCN4, KCND3, KCNJ5, KCNN3, PITX2, TBX5, ZFHX3, GJA1, SYNE2, SH3PXD2A, and PLN were found have bath rare variants and common variants related with AF. These may result from most mutation screening were carried out in familial AF, early-onset AF or lone AF, and AF patients in GWAS were more complex.
In conclusion, we summarized the current results of the genetic studies including common variants identified by GWAS (338 index SNPs) and candidate gene based association studies (40 SNPs), and performed a comprehensive annotation of all these AF susceptibility loci found by GWAS and candidate gene based association. We identified 4 AF susceptibility SNPs in UTRs may change the microRNA binding ability, and 250 AF susceptibility SNPs in non-coding regions were identified as located in enhancer regions or might affect the histone mark of promoters and enhancers, 65 SNPs may change the situation of interaction with transcription factors and totally 40 transcription factors were found interact with these SNPs. Our results also showed that 151 SNPs can affect the expression levels of a total of 328 target genes and 81 of them associated with the expression levels of the closest gene. Long-range transcriptional regulatory function predictions showed that 211 SNPs interact with distal target genes, and 104 of them exert as an eQTL effect. We also performed a GO and pathway enrichment of the AF eQTL genes. Taken together, our study suggested the relationship between genetic variants and possible targeted genes, and provides insight into potential genetic pathways underlying AF incidence and development.
Materials and Methods
Acquisition of AF susceptibility variants and search strategy
The workflow of the current study is presented in Figure 1. First, results of the current GWAS of AF were extracted from three public databases, including GWAS catalog (https://www.ebi.ac.uk/gwas/), GWAS central (https://www.gwascentral.org) and phenotype–genotype integrator (https://www.ncbi.nlm.nih.gov/gap/phegeni). We also searched the literature in Pumbed (https://pubmed.ncbi.nlm.nih.gov) to include all studies of AF GWAS. The keywords include atrial fibrillation, genome wide association or GWAS.
Besides the GWAS, several candidate gene based association studies have also identified a panel of genetic variants confer risk to AF. Results of these associated genetic variants were obtained by searching from the PubMed, EMBASE (https://www.embase.com) and Medline (https://www.nlm.nih.gov/bsd/medline.html) (Figure 1), and the searching keywords of medical subject headings (MeSH) including “atrial fibrillation” combined with “polymorphism, polymorphisms, variant, variants, single nucleotide polymorphism, single nucleotide polymorphisms, SNP, SNPs”. The results of literature searching were eligibility screened by two reviewers based on titles and abstracts. Studies published between 1 January 2007 and 1 November 2019 were included. Only case control association studies or cohort-based prospective studies were included. Functional researches, animal model studies or studies not performed in a population were excluded.
Information of AF GWAS index SNPs was extracted from the database of GWAS catalog, and the threshold of significant level for the association was set as P value lower than 5×10-8. For the SNPs from candidate gene based association studies, publications were reviewed by two reviewers independently and extracted the information about the variant(s) and the details of the population. Discrepancies in data extraction were resolved by discussion or submitted to a third reviewer if required. We divided the variants analyzed in candidate gene based studies into three groups, (i) replication study of the GWAS identified susceptibility loci of AF, (ii) novel variants with minor allele frequency (MAF) ≥0.1% (according to 1000 genome phase III global data), (iii) rare variants with a low frequency (MAF<0.1%) associated with AF by candidate gene association study or mutation screening. In our study, we excluded rare variants and mutations in (iii) from our further annotations, for the causal genes harbored mutations or rare variants of AF which were found in families or cohort were well summarized in previously reviews [8, 84] The significant level for SNPs in candidate gene based association studies was set as satisfying the Bonferroni correction. To reduce the probability of false positives, we exclude case controls studies if the statistical power <70%. The power was extracted from publications or calculated by PS: Power and Simple Size Calculation software [85].
Genomic region annotations
All AF susceptibility SNPs including index SNPs identified by GWAS and SNPs identified by candidate gene based association studies were first annotated using Variant Effect Predictor (http://asia.ensembl.org/Homo_sapiens/Tools/VEP, GRCh38) in Ensembl to obtain their genomic region information.
Functional annotation of AF susceptibility SNPs in exon
According to the genomic region information obtained from Variant Effect Predictor, non-synonymous SNPs were directly recognized as functional variants. SNPs in untranslated region (UTR) were analyzed the microRNA binding ability using MirSNP tool (http://bioinfo.bjmu.edu.cn/mirsnp/search) [86] and miRNASNP v2.0 (http://bioinfo.life.hust.edu.cn/miRNASNP2/) [87].
Enhancer prediction and transcription factor (TF) binding sites prediction of AF susceptibility SNPs in non-coding regions
Splicing variants identified by Variant Effect Predictor were classified as functional SNPs directly. Next, for intronic or intergenic SNPs, the chromatin states data from the Roadmap and ENCODE to analyze whether they are overlapping any enhancers in possible AF related tissues and cell types.
For the AF susceptibility SNPs in the non-coding genomic regions, including in UTR, promoter, intron, and intergenic regions, SNP2TFBS database (http://ccg.vital-it.ch/snp2tfbs/) was used to predict potential binding ability between SNPs and transcription factors [88].
Histone modification analysis
SNPs in promoter, intron, and intergenic regions may modify the histone binding ability, and here, using HaploReg (version 4.1) (https://pubs.broadinstitute.org/mammals/haploreg/haploreg.php) [89], we analyzed whether the identified non-coding AF associated SNPs overlap the major histone modifications (H3K9ac and H3K4me3 for promoter regions, H3K27ac and H3K4me1for enhancer regions) in AF related tissues and cell types.
Long-range transcriptional regulatory function predictions
SNPs in the noncoding region may reside within or near regulatory elements controlling the expression of distal target genes through topological interactions, and using 3DSNP [90], we annotated the possible regulatory effect of identified AF associated SNPs by examining their 3D interactions with distal genes mediated by chromatin loops.
Expression quantitative trait loci analyses
Genotype-Tissue Expression (GTEx) data were used in determining whether identified AF associated SNPs affect gene expression levels. eQTL analysis were performed bases on raw RNA-Seq data (RPKM) by genes from the GTEx V6 analysis freeze (dbGaP Accession phs000424.v6.p1) and included 25 tissues.
Gene ontology (GO) and pathway enrichment analysis of eQTL targeted genes
Gene Ontology including biological process, molecular function, and protein class were annotated using PANTHER (http://www.pantherdb.org). KEGG pathway enrichment analysis were used in the Search Tool for the Retrieval of Interacting Genes (STRING, v11.0, http://string-db.org).
Supplementary Materials
Author Contributions
Study concept and design: Chengqi Xu, data collection: Chengqi Xu, Pengyun Wang and Rongfeng Zhang, Critical revision of the manuscript for important intellectual content: Yunlong Xia. Statistical analysis: Chengqi Xu and Rongfeng Zhang. Obtained funding: Wang PY and Wei Yang. Study supervision: Wei Yang.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflicts of interest.
Funding
This study was supported by the National Natural Science Foundation of China Program (Grant No. 81600263, 31671302 and 81971276), National Key R&D Program of China (Grant #2018YFC1311600), Young and Middle-aged Talents of Wuhan Municipal Health Commission (to Wang PY), Hubei health and family planning scientific research project (Grant No.WJ2019Q037).
References
- 1. Bai Y, Wang YL, Shantsila A, Lip GY. The global burden of atrial fibrillation and stroke: a systematic review of the clinical epidemiology of atrial fibrillation in Asia. Chest. 2017; 152:810–20. https://doi.org/10.1016/j.chest.2017.03.048 [PubMed]
- 2. Béjot Y, Ben Salem D, Osseby GV, Couvreur G, Durier J, Marie C, Cottin Y, Moreau T, Giroud M. Epidemiology of ischemic stroke from atrial fibrillation in dijon, France, from 1985 to 2006. Neurology. 2009; 72:346–53. https://doi.org/10.1212/01.wnl.0000341280.31919.bd [PubMed]
- 3. Ceornodolea AD, Bal R, Severens JL. Epidemiology and management of atrial fibrillation and stroke: review of data from four european countries. Stroke Res Treat. 2017; 2017:8593207. https://doi.org/10.1155/2017/8593207 [PubMed]
- 4. Kokubo Y, Matsumoto C. Traditional cardiovascular risk factors for incident atrial fibrillation. Circ J. 2016; 80:2415–22. https://doi.org/10.1253/circj.CJ-16-0919 [PubMed]
- 5. Wang P, Yang Q, Wu X, Yang Y, Shi L, Wang C, Wu G, Xia Y, Yang B, Zhang R, Xu C, Cheng X, Li S, et al. Functional dominant-negative mutation of sodium channel subunit gene SCN3B associated with atrial fibrillation in a chinese GeneID population. Biochem Biophys Res Commun. 2010; 398:98–104. https://doi.org/10.1016/j.bbrc.2010.06.042 [PubMed]
- 6. Feghaly J, Zakka P, London B, MacRae CA, Refaat MM. Genetics of atrial fibrillation. J Am Heart Assoc. 2018; 7:e009884. https://doi.org/10.1161/JAHA.118.009884 [PubMed]
- 7. Hayashi K, Tada H, Yamagishi M. The genetics of atrial fibrillation. Curr Opin Cardiol. 2017; 32:10–16. https://doi.org/10.1097/HCO.0000000000000356 [PubMed]
- 8. Fatkin D, Santiago CF, Huttner IG, Lubitz SA, Ellinor PT. Genetics of atrial fibrillation: state of the art in 2017. Heart Lung Circ. 2017; 26:894–901. https://doi.org/10.1016/j.hlc.2017.04.008 [PubMed]
- 9. Kalstø SM, Siland JE, Rienstra M, Christophersen IE. Atrial fibrillation genetics update: toward clinical implementation. Front Cardiovasc Med. 2019; 6:127. https://doi.org/10.3389/fcvm.2019.00127 [PubMed]
- 10. Campbell HM, Wehrens XH. Genetics of atrial fibrillation: an update. Curr Opin Cardiol. 2018; 33:304–10. https://doi.org/10.1097/HCO.0000000000000505 [PubMed]
- 11. Pérez-Serra A, Campuzano O, Brugada R. Update about atrial fibrillation genetics. Curr Opin Cardiol. 2017. [Epub ahead of print]. https://doi.org/10.1097/HCO.0000000000000387 [PubMed]
- 12. Zhu Y, Tazearslan C, Suh Y. Challenges and progress in interpretation of non-coding genetic variants associated with human disease. Exp Biol Med (Maywood). 2017; 242:1325–34. https://doi.org/10.1177/1535370217713750 [PubMed]
- 13. Zhang F, Lupski JR. Non-coding genetic variants in human disease. Hum Mol Genet. 2015; 24:R102–10. https://doi.org/10.1093/hmg/ddv259 [PubMed]
- 14. Chinchilla A, Daimi H, Lozano-Velasco E, Dominguez JN, Caballero R, Delpón E, Tamargo J, Cinca J, Hove-Madsen L, Aranega AE, Franco D. PITX2 insufficiency leads to atrial electrical and structural remodeling linked to arrhythmogenesis. Circ Cardiovasc Genet. 2011; 4:269–79. https://doi.org/10.1161/CIRCGENETICS.110.958116 [PubMed]
- 15. Zhang M, Hill MC, Kadow ZA, Suh JH, Tucker NR, Hall AW, Tran TT, Swinton PS, Leach JP, Margulies KB, Ellinor PT, Li N, Martin JF. Long-range Pitx2c enhancer-promoter interactions prevent predisposition to atrial fibrillation. Proc Natl Acad Sci USA. 2019; 116:22692–98. https://doi.org/10.1073/pnas.1907418116 [PubMed]
- 16. Nielsen JB, Thorolfsdottir RB, Fritsche LG, Zhou W, Skov MW, Graham SE, Herron TJ, McCarthy S, Schmidt EM, Sveinbjornsson G, Surakka I, Mathis MR, Yamazaki M, et al. Biobank-driven genomic discovery yields new insight into atrial fibrillation biology. Nat Genet. 2018; 50:1234–39. https://doi.org/10.1038/s41588-018-0171-3 [PubMed]
- 17. Roselli C, Chaffin MD, Weng LC, Aeschbacher S, Ahlberg G, Albert CM, Almgren P, Alonso A, Anderson CD, Aragam KG, Arking DE, Barnard J, Bartz TM, et al. Multi-ethnic genome-wide association study for atrial fibrillation. Nat Genet. 2018; 50:1225–33. https://doi.org/10.1038/s41588-018-0133-9 [PubMed]
- 18. Nielsen JB, Fritsche LG, Zhou W, Teslovich TM, Holmen OL, Gustafsson S, Gabrielsen ME, Schmidt EM, Beaumont R, Wolford BN, Lin M, Brummett CM, Preuss MH, et al. Genome-wide study of atrial fibrillation identifies seven risk loci and highlights biological pathways and regulatory elements involved in cardiac development. Am J Hum Genet. 2018; 102:103–15. https://doi.org/10.1016/j.ajhg.2017.12.003 [PubMed]
- 19. Thorolfsdottir RB, Sveinbjornsson G, Sulem P, Nielsen JB, Jonsson S, Halldorsson GH, Melsted P, Ivarsdottir EV, Davidsson OB, Kristjansson RP, Thorleifsson G, Helgadottir A, Gretarsdottir S, et al. Coding variants in RPL3L and MYZAP increase risk of atrial fibrillation. Commun Biol. 2018; 1:68. https://doi.org/10.1038/s42003-018-0068-9 [PubMed]
- 20. Lee JY, Kim TH, Yang PS, Lim HE, Choi EK, Shim J, Shin E, Uhm JS, Kim JS, Joung B, Oh S, Lee MH, Kim YH, Pak HN. Korean atrial fibrillation network genome-wide association study for early-onset atrial fibrillation identifies novel susceptibility loci. Eur Heart J. 2017; 38:2586–94. https://doi.org/10.1093/eurheartj/ehx213 [PubMed]
- 21. Christophersen IE, Rienstra M, Roselli C, Yin X, Geelhoed B, Barnard J, Lin H, Arking DE, Smith AV, Albert CM, Chaffin M, Tucker NR, Li M, et al, METASTROKE Consortium of the ISGC, Neurology Working Group of the CHARGE Consortium, and AFGen Consortium. Large-scale analyses of common and rare variants identify 12 new loci associated with atrial fibrillation. Nat Genet. 2017; 49:946–52. https://doi.org/10.1038/ng.3843 [PubMed]
- 22. Low SK, Takahashi A, Ebana Y, Ozaki K, Christophersen IE, Ellinor PT, Ogishima S, Yamamoto M, Satoh M, Sasaki M, Yamaji T, Iwasaki M, Tsugane S, et al, and AFGen Consortium. Identification of six new genetic loci associated with atrial fibrillation in the Japanese population. Nat Genet. 2017; 49:953–58. https://doi.org/10.1038/ng.3842 [PubMed]
- 23. Thorolfsdottir RB, Sveinbjornsson G, Sulem P, Helgadottir A, Gretarsdottir S, Benonisdottir S, Magnusdottir A, Davidsson OB, Rajamani S, Roden DM, Darbar D, Pedersen TR, Sabatine MS, et al. A missense variant in PLEC increases risk of atrial fibrillation. J Am Coll Cardiol. 2017; 70:2157–68. https://doi.org/10.1016/j.jacc.2017.09.005 [PubMed]
- 24. Yamada Y, Sakuma J, Takeuchi I, Yasukochi Y, Kato K, Oguri M, Fujimaki T, Horibe H, Muramatsu M, Sawabe M, Fujiwara Y, Taniguchi Y, Obuchi S, et al. Identification of TNFSF13, SPATC1L, SLC22A25 and SALL4 as novel susceptibility loci for atrial fibrillation by an exome-wide association study. Mol Med Rep. 2017; 16:5823–32. https://doi.org/10.3892/mmr.2017.7334 [PubMed]
- 25. Lubitz SA, Brody JA, Bihlmeyer NA, Roselli C, Weng LC, Christophersen IE, Alonso A, Boerwinkle E, Gibbs RA, Bis JC, Cupples LA, Mohler PJ, Nickerson DA, et al, and NHLBI GO Exome Sequencing Project. Whole exome sequencing in atrial fibrillation. PLoS Genet. 2016; 12:e1006284. https://doi.org/10.1371/journal.pgen.1006284 [PubMed]
- 26. Kertai MD, Li YJ, Ji Y, Qi W, Lombard FW, Shah SH, Kraus WE, Stafford-Smith M, Newman MF, Milano CA, Waldron N, Podgoreanu MV, Mathew JP, and Duke Perioperative Genetics and Safety Outcomes (PEGASUS) Investigative Team. Genome-wide association study of new-onset atrial fibrillation after coronary artery bypass grafting surgery. Am Heart J. 2015; 170:580–90.e28. https://doi.org/10.1016/j.ahj.2015.06.009 [PubMed]
- 27. Sinner MF, Tucker NR, Lunetta KL, Ozaki K, Smith JG, Trompet S, Bis JC, Lin H, Chung MK, Nielsen JB, Lubitz SA, Krijthe BP, Magnani JW, et al, METASTROKE Consortium, and AFGen Consortium. Integrating genetic, transcriptional, and functional analyses to identify 5 novel genes for atrial fibrillation. Circulation. 2014; 130:1225–35. https://doi.org/10.1161/CIRCULATIONAHA.114.009892 [PubMed]
- 28. Ellinor PT, Lunetta KL, Albert CM, Glazer NL, Ritchie MD, Smith AV, Arking DE, Müller-Nurasyid M, Krijthe BP, Lubitz SA, Bis JC, Chung MK, Dörr M, et al. Meta-analysis identifies six new susceptibility loci for atrial fibrillation. Nat Genet. 2012; 44:670–75. https://doi.org/10.1038/ng.2261 [PubMed]
- 29. Ellinor PT, Lunetta KL, Glazer NL, Pfeufer A, Alonso A, Chung MK, Sinner MF, de Bakker PI, Mueller M, Lubitz SA, Fox E, Darbar D, Smith NL, et al. Common variants in KCNN3 are associated with lone atrial fibrillation. Nat Genet. 2010; 42:240–44. https://doi.org/10.1038/ng.537 [PubMed]
- 30. Gudbjartsson DF, Holm H, Gretarsdottir S, Thorleifsson G, Walters GB, Thorgeirsson G, Gulcher J, Mathiesen EB, Njølstad I, Nyrnes A, Wilsgaard T, Hald EM, Hveem K, et al. A sequence variant in ZFHX3 on 16q22 associates with atrial fibrillation and ischemic stroke. Nat Genet. 2009; 41:876–78. https://doi.org/10.1038/ng.417 [PubMed]
- 31. Benjamin EJ, Rice KM, Arking DE, Pfeufer A, van Noord C, Smith AV, Schnabel RB, Bis JC, Boerwinkle E, Sinner MF, Dehghan A, Lubitz SA, D’Agostino RB
Sr , et al. Variants in ZFHX3 are associated with atrial fibrillation in individuals of european ancestry. Nat Genet. 2009; 41:879–81. https://doi.org/10.1038/ng.416 [PubMed] - 32. Larson MG, Atwood LD, Benjamin EJ, Cupples LA, D’Agostino RB
Sr , Fox CS, Govindaraju DR, Guo CY, Heard-Costa NL, Hwang SJ, Murabito JM, Newton-Cheh C, O’Donnell CJ, et al. Framingham heart study 100K project: genome-wide associations for cardiovascular disease outcomes. BMC Med Genet. 2007 (Suppl 1); 8:S5. https://doi.org/10.1186/1471-2350-8-S1-S5 [PubMed] - 33. Gudbjartsson DF, Arnar DO, Helgadottir A, Gretarsdottir S, Holm H, Sigurdsson A, Jonasdottir A, Baker A, Thorleifsson G, Kristjansson K, Palsson A, Blondal T, Sulem P, et al. Variants conferring risk of atrial fibrillation on chromosome 4q25. Nature. 2007; 448:353–57. https://doi.org/10.1038/nature06007 [PubMed]
- 34. Cao H, Xu W, Chen X, Zhou Q, Lan R, Chen Y, Wang D. Functional promoter -1816C>G variant of RANKL predicts risk and prognosis of lone atrial fibrillation. Heart Vessels. 2019; 34:151–58. https://doi.org/10.1007/s00380-018-1222-5 [PubMed]
- 35. Xiong H, Yang Q, Zhang X, Wang P, Chen F, Liu Y, Wang P, Zhao Y, Li S, Huang Y, Chen S, Wang X, Zhang H, et al. Significant association of rare variant p.Gly8Ser in cardiac sodium channel β4-subunit SCN4B with atrial fibrillation. Ann Hum Genet. 2019; 83:239–48. https://doi.org/10.1111/ahg.12305 [PubMed]
- 36. Wang P, Qin W, Wang P, Huang Y, Liu Y, Zhang R, Li S, Yang Q, Wang X, Chen F, Liu J, Yang B, Cheng X, et al. Genomic variants in NEURL, GJA1 and CUX2 significantly increase genetic susceptibility to atrial fibrillation. Sci Rep. 2018; 8:3297. https://doi.org/10.1038/s41598-018-21611-7 [PubMed]
- 37. Zaw KT, Sato N, Ikeda S, Thu KS, Mieno MN, Arai T, Mori S, Furukawa T, Sasano T, Sawabe M, Tanaka M, Muramatsu M. Association of ZFHX3 gene variation with atrial fibrillation, cerebral infarction, and lung thromboembolism: an autopsy study. J Cardiol. 2017; 70:180–84. https://doi.org/10.1016/j.jjcc.2016.11.005 [PubMed]
- 38. Feng W, Sun L, Qu XF. Association of AGTR1 and ACE2 gene polymorphisms with structural atrial fibrillation in a chinese han population. Pharmazie. 2017; 72:17–21. https://doi.org/10.1691/ph.2017.6752 [PubMed]
- 39. Nakano Y, Ochi H, Onohara Y, Sairaku A, Tokuyama T, Matsumura H, Tomomori S, Amioka M, Hironomobe N, Motoda C, Oda N, Chayama K, Chen CH, et al. Genetic variations of aldehyde dehydrogenase 2 and alcohol dehydrogenase 1B are associated with the etiology of atrial fibrillation in Japanese. J Biomed Sci. 2016; 23:89. https://doi.org/10.1186/s12929-016-0304-x [PubMed]
- 40. Seppälä I, Kleber ME, Bevan S, Lyytikäinen LP, Oksala N, Hernesniemi JA, Mäkelä KM, Rothwell PM, Sudlow C, Dichgans M, Mononen N, Vlachopoulou E, Sinisalo J, et al. Associations of functional alanine-glyoxylate aminotransferase 2 gene variants with atrial fibrillation and ischemic stroke. Sci Rep. 2016; 6:23207. https://doi.org/10.1038/srep23207 [PubMed]
- 41. Fang Z, Jiang Y, Wang Y, Lin Y, Liu Y, Zhao L, Xu Y, Toorabally MB, He S, Zhang F. The rs6771157 C/G polymorphism in SCN10A is associated with the risk of atrial fibrillation in a chinese han population. Sci Rep. 2016; 6:35212. https://doi.org/10.1038/srep35212 [PubMed]
- 42. Wang C, Wu M, Qian J, Li B, Tu X, Xu C, Li S, Chen S, Zhao Y, Huang Y, Shi L, Cheng X, Liao Y, et al. Identification of rare variants in TNNI3 with atrial fibrillation in a chinese GeneID population. Mol Genet Genomics. 2016; 291:79–92. https://doi.org/10.1007/s00438-015-1090-y [PubMed]
- 43. Zhang R, Tian X, Gao L, Li H, Yin X, Dong Y, Yang Y, Xia Y. Common variants in the TBX5 gene associated with atrial fibrillation in a chinese han population. PLoS One. 2016; 11:e0160467. https://doi.org/10.1371/journal.pone.0160467 [PubMed]
- 44. Roberts JD, Hu D, Heckbert SR, Alonso A, Dewland TA, Vittinghoff E, Liu Y, Psaty BM, Olgin JE, Magnani JW, Huntsman S, Burchard EG, Arking DE, et al. Genetic investigation into the differential risk of atrial fibrillation among black and white individuals. JAMA Cardiol. 2016; 1:442–50. https://doi.org/10.1001/jamacardio.2016.1185 [PubMed]
- 45. Luo Z, Yan C, Yu P, Bao W, Shen X, Zheng W, Lin X, Wang Z, Chen H, Chen F, Liu D, Huang M. CASP3 genetic variants and susceptibility to atrial fibrillation in chinese han population. Int J Cardiol. 2015; 183:1–5. https://doi.org/10.1016/j.ijcard.2015.01.048 [PubMed]
- 46. Chen S, Wang C, Wang X, Xu C, Wu M, Wang P, Tu X, Wang QK. Significant association between CAV1 variant rs3807989 on 7p31 and atrial fibrillation in a chinese han population. J Am Heart Assoc. 2015; 4:e001980. https://doi.org/10.1161/JAHA.115.001980 [PubMed]
- 47. Liu Y, Ni B, Lin Y, Chen XG, Chen M, Hu Z, Zhang F. The rs3807989 g/a polymorphism in CAV1 is associated with the risk of atrial fibrillation in chinese han populations. Pacing Clin Electrophysiol. 2015; 38:164–70. https://doi.org/10.1111/pace.12494 [PubMed]
- 48. Rosenberg MA, Kaplan RC, Siscovick DS, Psaty BM, Heckbert SR, Newton-Cheh C, Mukamal KJ. Genetic variants related to height and risk of atrial fibrillation: the cardiovascular health study. Am J Epidemiol. 2014; 180:215–22. https://doi.org/10.1093/aje/kwu126 [PubMed]
- 49. Andreasen L, Nielsen JB, Darkner S, Christophersen IE, Jabbari J, Refsgaard L, Thiis JJ, Sajadieh A, Tveit A, Haunsø S, Svendsen JH, Schmitt N, Olesen MS. Brugada syndrome risk loci seem protective against atrial fibrillation. Eur J Hum Genet. 2014; 22:1357–61. https://doi.org/10.1038/ejhg.2014.46 [PubMed]
- 50. Luo Z, Yan C, Zhang W, Shen X, Zheng W, Chen F, Cao X, Yang Y, Lin X, Wang Z, Huang M. Association between SNP rs13376333 and rs1131820 in the KCNN3 gene and atrial fibrillation in the chinese han population. Clin Chem Lab Med. 2014; 52:1867–73. https://doi.org/10.1515/cclm-2014-0491 [PubMed]
- 51. Voudris KV, Apostolakis S, Karyofillis P, Doukas K, Zaravinos A, Androutsopoulos VP, Michalis A, Voudris V, Spandidos DA. Genetic diversity of the KCNE1 gene and susceptibility to postoperative atrial fibrillation. Am Heart J. 2014; 167:274–80.e1. https://doi.org/10.1016/j.ahj.2013.09.020 [PubMed]
- 52. Liu Y, Ni B, Lin Y, Chen XG, Fang Z, Zhao L, Hu Z, Zhang F. Genetic polymorphisms in ZFHX3 are associated with atrial fibrillation in a chinese han population. PLoS One. 2014; 9:e101318. https://doi.org/10.1371/journal.pone.0101318 [PubMed]
- 53. Lin H, Sinner MF, Brody JA, Arking DE, Lunetta KL, Rienstra M, Lubitz SA, Magnani JW, Sotoodehnia N, McKnight B, McManus DD, Boerwinkle E, Psaty BM, et al, and CHARGE Atrial Fibrillation Working Group. Targeted sequencing in candidate genes for atrial fibrillation: the cohorts for heart and aging research in genomic epidemiology (CHARGE) targeted sequencing study. Heart Rhythm. 2014; 11:452–57. https://doi.org/10.1016/j.hrthm.2013.11.012 [PubMed]
- 54. Adamsson Eryd S, Sjögren M, Smith JG, Nilsson PM, Melander O, Hedblad B, Engström G. Ceruloplasmin and atrial fibrillation: evidence of causality from a population-based mendelian randomization study. J Intern Med. 2014; 275:164–71. https://doi.org/10.1111/joim.12144 [PubMed]
- 55. Cao H, Zhou Q, Lan R, Røe OD, Chen X, Chen Y, Wang D. A functional polymorphism C-509T in TGFβ-1 promoter contributes to susceptibility and prognosis of lone atrial fibrillation in chinese population. PLoS One. 2014; 9:e112912. https://doi.org/10.1371/journal.pone.0112912 [PubMed]
- 56. Marott SC, Nordestgaard BG, Jensen GB, Tybjaerg-Hansen A, Benn M. AT1 mutations and risk of atrial fibrillation based on genotypes from 71,000 individuals from the general population. Br J Clin Pharmacol. 2013; 76:114–24. https://doi.org/10.1111/bcp.12050 [PubMed]
- 57. Jeff JM, Donahue BS, Brown-Gentry K, Roden DM, Crawford DC, Stein CM, Kurnik D. Genetic variation in the β1-adrenergic receptor is associated with the risk of atrial fibrillation after cardiac surgery. Am Heart J. 2014; 167:101–08.e1. https://doi.org/10.1016/j.ahj.2013.09.016 [PubMed]
- 58. Ilkhanoff L, Arking DE, Lemaitre RN, Alonso A, Chen LY, Durda P, Hesselson SE, Kerr KF, Magnani JW, Marcus GM, Schnabel RB, Smith JG, Soliman EZ, et al, Candidate-Gene Association Resource (CARE) Consortium and the Cardiac Arrest Blood Study (CABS) Investigators. A common SCN5A variant is associated with PR interval and atrial fibrillation among african americans. J Cardiovasc Electrophysiol. 2014; 25:1150–57. https://doi.org/10.1111/jce.12483 [PubMed]
- 59. Andreasen L, Nielsen JB, Christophersen IE, Holst AG, Sajadieh A, Tveit A, Haunsø S, Svendsen JH, Schmitt N, Olesen MS. Genetic modifier of the QTc interval associated with early-onset atrial fibrillation. Can J Cardiol. 2013; 29:1234–40. https://doi.org/10.1016/j.cjca.2013.06.009 [PubMed]
- 60. Olesen MS, Holst AG, Jabbari J, Nielsen JB, Christophersen IE, Sajadieh A, Haunsø S, Svendsen JH. Genetic loci on chromosomes 4q25, 7p31, and 12p12 are associated with onset of lone atrial fibrillation before the age of 40 years. Can J Cardiol. 2012; 28:191–95. https://doi.org/10.1016/j.cjca.2011.11.016 [PubMed]
- 61. Schnabel RB, Kerr KF, Lubitz SA, Alkylbekova EL, Marcus GM, Sinner MF, Magnani JW, Wolf PA, Deo R, Lloyd-Jones DM, Lunetta KL, Mehra R, Levy D, et al, and Candidate Gene Association Resource (CARe) Atrial Fibrillation/Electrocardiography Working Group. Large-scale candidate gene analysis in whites and african americans identifies IL6R polymorphism in relation to atrial fibrillation: the national heart, lung, and blood institute’s candidate gene association resource (CARe) project. Circ Cardiovasc Genet. 2011; 4:557–64. https://doi.org/10.1161/CIRCGENETICS.110.959197 [PubMed]
- 62. Wirka RC, Gore S, Van Wagoner DR, Arking DE, Lubitz SA, Lunetta KL, Benjamin EJ, Alonso A, Ellinor PT, Barnard J, Chung MK, Smith JD. A common connexin-40 gene promoter variant affects connexin-40 expression in human atria and is associated with atrial fibrillation. Circ Arrhythm Electrophysiol. 2011; 4:87–93. https://doi.org/10.1161/CIRCEP.110.959726 [PubMed]
- 63. Li C, Wang F, Yang Y, Fu F, Xu C, Shi L, Li S, Xia Y, Wu G, Cheng X, Liu H, Wang C, Wang P, et al. Significant association of SNP rs2106261 in the ZFHX3 gene with atrial fibrillation in a chinese han GeneID population. Hum Genet. 2011; 129:239–46. https://doi.org/10.1007/s00439-010-0912-6 [PubMed]
- 64. Lubitz SA, Sinner MF, Lunetta KL, Makino S, Pfeufer A, Rahman R, Veltman CE, Barnard J, Bis JC, Danik SP, Sonni A, Shea MA, Del Monte F, et al. Independent susceptibility markers for atrial fibrillation on chromosome 4q25. Circulation. 2010; 122:976–84. https://doi.org/10.1161/CIRCULATIONAHA.109.886440 [PubMed]
- 65. Roberts JD, Davies RW, Lubitz SA, Thibodeau IL, Nery PB, Birnie DH, Benjamin EJ, Lemery R, Ellinor PT, Gollob MH. Evaluation of non-synonymous NPPA single nucleotide polymorphisms in atrial fibrillation. Europace. 2010; 12:1078–83. https://doi.org/10.1093/europace/euq161 [PubMed]
- 66. Ren X, Xu C, Zhan C, Yang Y, Shi L, Wang F, Wang C, Xia Y, Yang B, Wu G, Wang P, Li X, Wang D, et al. Identification of NPPA variants associated with atrial fibrillation in a chinese GeneID population. Clin Chim Acta. 2010; 411:481–85. https://doi.org/10.1016/j.cca.2009.12.019 [PubMed]
- 67. Shi L, Li C, Wang C, Xia Y, Wu G, Wang F, Xu C, Wang P, Li X, Wang D, Xiong X, Bai Y, Liu M, et al. Assessment of association of rs2200733 on chromosome 4q25 with atrial fibrillation and ischemic stroke in a chinese han population. Hum Genet. 2009; 126:843–49. https://doi.org/10.1007/s00439-009-0737-3 [PubMed]
- 68. Kääb S, Darbar D, van Noord C, Dupuis J, Pfeufer A, Newton-Cheh C, Schnabel R, Makino S, Sinner MF, Kannankeril PJ, Beckmann BM, Choudry S, Donahue BS, et al. Large scale replication and meta-analysis of variants on chromosome 4q25 associated with atrial fibrillation. Eur Heart J. 2009; 30:813–19. https://doi.org/10.1093/eurheartj/ehn578 [PubMed]
- 69. Sinner MF, Pfeufer A, Akyol M, Beckmann BM, Hinterseer M, Wacker A, Perz S, Sauter W, Illig T, Näbauer M, Schmitt C, Wichmann HE, Schömig A, et al. The non-synonymous coding IKr-channel variant KCNH2-K897T is associated with atrial fibrillation: results from a systematic candidate gene-based analysis of KCNH2 (HERG). Eur Heart J. 2008; 29:907–14. https://doi.org/10.1093/eurheartj/ehm619 [PubMed]
- 70. Giusti B, Gori AM, Marcucci R, Sestini I, Saracini C, Sticchi E, Gensini F, Fatini C, Abbate R, Gensini GF. Role of C677T and A1298C MTHFR, A2756G MTR and -786 C/T eNOS gene polymorphisms in atrial fibrillation susceptibility. PLoS One. 2007; 2:e495. https://doi.org/10.1371/journal.pone.0000495 [PubMed]
- 71. Maurano MT, Humbert R, Rynes E, Thurman RE, Haugen E, Wang H, Reynolds AP, Sandstrom R, Qu H, Brody J, Shafer A, Neri F, Lee K, et al. Systematic localization of common disease-associated variation in regulatory DNA. Science. 2012; 337:1190–95. https://doi.org/10.1126/science.1222794 [PubMed]
- 72. Dong SS, Zhang YJ, Chen YX, Yao S, Hao RH, Rong Y, Niu HM, Chen JB, Guo Y, Yang TL. Comprehensive review and annotation of susceptibility SNPs associated with obesity-related traits. Obes Rev. 2018; 19:917–30. https://doi.org/10.1111/obr.12677 [PubMed]
- 73. Morley MP, Wang X, Hu R, Brandimarto J, Tucker NR, Felix JF, Smith NL, van der Harst P, Ellinor PT, Margulies KB, Musunuru K, Cappola TP. Cardioprotective effects of MTSS1 enhancer variants. Circulation. 2019; 139:2073–76. https://doi.org/10.1161/CIRCULATIONAHA.118.037939 [PubMed]
- 74. Gontier Y, Taivainen A, Fontao L, Sonnenberg A, van der Flier A, Carpen O, Faulkner G, Borradori L. The z-disc proteins myotilin and FATZ-1 interact with each other and are connected to the sarcolemma via muscle-specific filamins. J Cell Sci. 2005; 118:3739–49. https://doi.org/10.1242/jcs.02484 [PubMed]
- 75. Takada F, Vander Woude DL, Tong HQ, Thompson TG, Watkins SC, Kunkel LM, Beggs AH. Myozenin: an alpha-actinin- and gamma-filamin-binding protein of skeletal muscle Z lines. Proc Natl Acad Sci USA. 2001; 98:1595–600. https://doi.org/10.1073/pnas.041609698 [PubMed]
- 76. Roberts MD, Romero MA, Mobley CB, Mumford PW, Roberson PA, Haun CT, Vann CG, Osburn SC, Holmes HH, Greer RA, Lockwood CM, Parry HA, Kavazis AN. Skeletal muscle mitochondrial volume and myozenin-1 protein differences exist between high versus low anabolic responders to resistance training. PeerJ. 2018; 6:e5338. https://doi.org/10.7717/peerj.5338 [PubMed]
- 77. Posch MG, Perrot A, Dietz R, Ozcelik C, Pankuweit S, Ruppert V, Richter A, Maisch B. Mutations in MYOZ1 as well as MYOZ2 encoding the calsarcins are not associated with idiopathic and familial dilated cardiomyopathy. Mol Genet Metab. 2007; 91:207–08. https://doi.org/10.1016/j.ymgme.2007.02.014 [PubMed]
- 78. Arola AM, Sanchez X, Murphy RT, Hasle E, Li H, Elliott PM, McKenna WJ, Towbin JA, Bowles NE. Mutations in PDLIM3 and MYOZ1 encoding myocyte Z line proteins are infrequently found in idiopathic dilated cardiomyopathy. Mol Genet Metab. 2007; 90:435–40. https://doi.org/10.1016/j.ymgme.2006.12.008 [PubMed]
- 79. Chen YH, Xu SJ, Bendahhou S, Wang XL, Wang Y, Xu WY, Jin HW, Sun H, Su XY, Zhuang QN, Yang YQ, Li YB, Liu Y, et al. KCNQ1 gain-of-function mutation in familial atrial fibrillation. Science. 2003; 299:251–54. https://doi.org/10.1126/science.1077771 [PubMed]
- 80. Yang Y, Xia M, Jin Q, Bendahhou S, Shi J, Chen Y, Liang B, Lin J, Liu Y, Liu B, Zhou Q, Zhang D, Wang R, et al. Identification of a KCNE2 gain-of-function mutation in patients with familial atrial fibrillation. Am J Hum Genet. 2004; 75:899–905. https://doi.org/10.1086/425342 [PubMed]
- 81. Ravn LS, Aizawa Y, Pollevick GD, Hofman-Bang J, Cordeiro JM, Dixen U, Jensen G, Wu Y, Burashnikov E, Haunso S, Guerchicoff A, Hu D, Svendsen JH, et al. Gain of function in IKs secondary to a mutation in KCNE5 associated with atrial fibrillation. Heart Rhythm. 2008; 5:427–35. https://doi.org/10.1016/j.hrthm.2007.12.019 [PubMed]
- 82. Tsai CT, Lai LP, Hwang JJ, Lin JL, Chiang FT. Molecular genetics of atrial fibrillation. J Am Coll Cardiol. 2008; 52:241–50. https://doi.org/10.1016/j.jacc.2008.02.072 [PubMed]
- 83. Bartos DC, Duchatelet S, Burgess DE, Klug D, Denjoy I, Peat R, Lupoglazoff JM, Fressart V, Berthet M, Ackerman MJ, January CT, Guicheney P, Delisle BP. R231C mutation in KCNQ1 causes long QT syndrome type 1 and familial atrial fibrillation. Heart Rhythm. 2011; 8:48–55. https://doi.org/10.1016/j.hrthm.2010.09.010 [PubMed]
- 84. Xiao J, Liang D, Chen YH. The genetics of atrial fibrillation: from the bench to the bedside. Annu Rev Genomics Hum Genet. 2011; 12:73–96. https://doi.org/10.1146/annurev-genom-082410-101515 [PubMed]
- 85. Dupont WD, Plummer WD
Jr . Power and sample size calculations for studies involving linear regression. Control Clin Trials. 1998; 19:589–601. https://doi.org/10.1016/s0197-2456(98)00037-3 [PubMed] - 86. Liu C, Zhang F, Li T, Lu M, Wang L, Yue W, Zhang D. MirSNP, a database of polymorphisms altering miRNA target sites, identifies miRNA-related SNPs in GWAS SNPs and eQTLs. BMC Genomics. 2012; 13:661. https://doi.org/10.1186/1471-2164-13-661 [PubMed]
- 87. Gong J, Liu C, Liu W, Wu Y, Ma Z, Chen H, Guo AY. An update of miRNASNP database for better SNP selection by GWAS data, miRNA expression and online tools. Database (Oxford). 2015; 2015:bav029. https://doi.org/10.1093/database/bav029 [PubMed]
- 88. Kumar S, Ambrosini G, Bucher P. SNP2TFBS - a database of regulatory SNPs affecting predicted transcription factor binding site affinity. Nucleic Acids Res. 2017; 45:D139–44. https://doi.org/10.1093/nar/gkw1064 [PubMed]
- 89. Ward LD, Kellis M. HaploReg v4: systematic mining of putative causal variants, cell types, regulators and target genes for human complex traits and disease. Nucleic Acids Res. 2016; 44:D877–81. https://doi.org/10.1093/nar/gkv1340 [PubMed]
- 90. Lu Y, Quan C, Chen H, Bo X, Zhang C. 3DSNP: a database for linking human noncoding SNPs to their three-dimensional interacting genes. Nucleic Acids Res. 2017; 45:D643–49. https://doi.org/10.1093/nar/gkw1022 [PubMed]


