Introduction
Breast cancer is the most common cancer in women worldwide [1], as well as in China [2]. The incidence of breast cancer in China continues to rise rapidly at a rate of 3% to 4% per year, which is double the global average [3]. Even though the breast cancer treatment in China has greatly improved, the overall survival rates are still lower than in other countries [4]. The increasing incidence of breast cancer, the huge population base, and the lower survival prognosis, represent a severe challenge for the prevention and control of breast cancer in China. Therefore, identifying the environmental and genetic risk factors of breast cancer in China is very important to reduce the incidence of breast cancer.
Occurrence and development of breast cancer are affected by many genetic and environmental factors and their interactions [5]. Many potential environmental risk factors of breast cancer have been identified, including smoking [6], drinking [7], diet [8], and hormone exposure [9]. In addition, genetic factors play a very important role in the development and progression of breast cancer [10]. Previous studies have suggested that the increased risk of breast cancer is associated with breast cancer susceptibility gene 1 [11], leptin G-2548A gene [12], interleukin 4 (IL-4) [13], and insulin-like growth factor 1 [14].
Tissue inhibitor of metalloproteinase-2 (TIMP-2) is an endogenous inhibitor of matrix metalloproteinase-2 (MMP-2)[15], an important enzyme in the regulation of cancer cell proliferation and metastasis [16]. MMP-2 is over-expressed in cancer tissues, and its increased expression correlates with advanced cancer stage and poor prognosis. TIMP-2 forms a complex with MMP-2, thus regulating MMP-2 proteolytic activity. TIMP-2 mutations on chromosome 17q25 can affect its binding to MMP-2, resulting in cancer development [17]. Previous studies have indicated that TIMP-2 gene polymorphism is associated with gastric cancer [18], lung cancer [19], and prostate cancer [20]. In addition, TIMP-2 gene rs4789936/CT genotype is associated with an increased risk of colorectal cancer compared with CC genotype [21]. A recent study from Northern Chinese population has suggested that the mutation of TIMP-2 gene rs4789936 polymorphism is not associated with an increased risk of breast cancer [22]. However, the results remain controversial due to the different eating habits, environmental factors, and genetic mutation frequency. In this study, we evaluated the relationship between TIMP-2 gene rs4789936 polymorphism and the risk of breast cancer in Southern Chinese population. We also performed a follow-up study to determine the effect of rs4789936 polymorphism on breast cancer prognosis.
Results
Characteristics of study population
The study included 480 breast cancer patients and 530 healthy controls; the main characteristics of the patient case group and the healthy control group are presented in Table 1. There were no significant differences in age (P=0.110), BMI (P=0.405), and drinking (P=0.283). Notably, the distribution of drinking population was significantly different between the case group and the control group (19.4% vs 13.6%, P=0.013). Among the case population, 52.1% of breast cancer patients had tumors with diameter ≥ 2cm, 54.2% of patients belonged to stage I/II, 42.7% of patients were ER positive, 48.5% were PR positive, and 67.5% were her-2 positive. Follow-up revealed that 33.8% of patients received radiation alone, and 66.3% received chemotherapy and radiotherapy.
Table 1. General characteristics of case group and control group.
| Parameters | Case group | Control group | χ2/t | P | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age(y) | 49.6±7.0 | 50.3±6.9 | 1.598 | 0.110 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI (kg/m2) | 23.6±3.0 | 23.7±3.2 | 0.832 | 0.405 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking (n, %) | 93(19.4%) | 72(13.6%) | 6.178 | 0.013 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Drinking (n, %) | 115(24.0%0 | 112(21.1%) | 1.1555 | 0.283 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor size, cm | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| <2cm | 230(47.9%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≥2cm | 250(52.1%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lymph node metastases | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 231(48.1%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No | 249(51.9%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I/II | 260(54.2%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| III/IV | 220(45.8%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ER | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive | 205(42.7%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Negative | 275(57.3%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PR | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive | 233(48.5%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Negative | 247(51.5%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Her-2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive | 324(67.5%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Negative | 156(32.5%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | 162(33.8%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemoradiotherapy | 318(66.3%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ER, estrogen receptor; PR, progesterone receptor. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
TIMP-2 gene rs4789936 polymorphism and breast cancer risk
The genotype of control group did not exhibit a departure from Hardy-Weinberg equilibrium (P>0.05). The genotype distributions of case and control groups are presented in Table 2. Compared with CC genotype, the patients with CT/TT genotype had an elevated risk of breast cancer (CT: OR=1.46, 95%CI:1.11-1.92, P=0.007; TT: OR=2.57, 95%CI: 1.76-3.77, P<0.001). Significant differences were also found using dominant model (CT+TT: OR=1.71, 95%CI: 1.33-2.20, P<0.001), recessive model (TT vs CT+CC: OR=2.19, 95%CI: 1.52-3.15, P<0.001), and allele model (T vs C: OR=1.69, 95%CI: 1.40-2.04, P<0.001). After adjusting for age, smoking, drinking, and BMI, significant differences were still observed. Detailed results are presented in Table 2. Stratified analysis (Table 3) indicated a significantly increased breast cancer risk in the homozygote model (TT vs CC) that was associated with age, smoking, drinking, and BMI. The heterozygote model (CT vs CC) was significant among population of <60 years, non-smokers and non-drinkers. For BMI>24, the dominant model showed an increased risk of breast cancer.
Table 2. Genotype of frequencies of TIMP-2 gene rs4789936 in cases and controls.
| Modes | Genotype | Case (n, %) | Control (n, %) | OR (95%CI) | P | OR (95%CI)* | P* | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs4789936 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Co-dominant | CC | 204(42.5%) | 296(55.8%) | 1.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heterozygote | CT | 182(37.9%) | 181(34.2%) | 1.46(1.11-1.92) | 0.007 | 1.45(1.10-1.91) | 0.007 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Homozygote | TT | 94(19.6%) | 53(10.0%) | 2.57(1.76-3.77) | <0.001 | 2.41(1.63-3.55) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dominant | CC | 204(42.5%) | 296(55.8%) | 1.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CT+TT | 276(57.5%) | 234(44.2%) | 1.71(1.33-2.20) | <0.001 | 1.68(1.30-2.15) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Recessive | CT+CC | 386(80.4%) | 477(90.0%) | 1.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | 94(19.6%) | 53(10.0%) | 2.19(1.52-3.15) | <0.001 | 2.05(1.42-2.97) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Allele | C | 590(61.4%) | 773(72.9%) | 1.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T | 370(38.5%) | 287(27.1%) | 1.69(1.40-2.04) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| *Adjust age, smoking, drinking, and BMI. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 3. Stratified analyses between rs4789936 polymorphism and the risk of breast cancer.
| Variable | Case/Control | CT vs CC | P | TT vs CC | P | TT vs CT+CC | P | TT+CT vs CC | P | ||
| CC | CT | TT | |||||||||
| Age | |||||||||||
| <60 | 133/188 | 104/103 | 67/36 | 1.43(1.00-2.03) | 0.047 | 2.63(1.66-4.18) | <0.001 | 2.29(1.47-3.55) | <0.001 | 1.74(1.27-2.38) | 0.001 |
| ≥60 | 71/108 | 78/78 | 27/17 | 1.52(0.99-2.35) | 0.058 | 2.42(1.23-4.75) | 0.011 | 1.98(1.04-3.78) | 0.037 | 1.68(1.12-2.53) | 0.013 |
| Smoking | |||||||||||
| Yes | 27/33 | 28/29 | 38/10 | 1.18(0.57-2.44) | 0.655 | 4.64(1.96-11.00) | <0.001 | 4.28(1.95-9.40) | <0.001 | 2.07(1.09-3.94) | 0.027 |
| No | 177/263 | 154/152 | 56/43 | 1.51(1.12-2.02) | 0.006 | 1.94(1.25-3.01) | 0.003 | 1.63(1.07-2.49) | 0.023 | 1.60(1.22-2.10) | 0.001 |
| Drinking | |||||||||||
| Yes | 48/64 | 36/39 | 31/9 | 1.23(0.68-2.22) | 0.489 | 4.59(2.00-10.55) | <0.001 | 1.86(1.10-3.15) | 0.021 | 4.22(1.91-9.36) | <0.001 |
| No | 156/232 | 146/142 | 63/44 | 1.53(1.12-2.08) | 0.007 | 2.13(1.38-3.29) | 0.001 | 1.77(1.17-2.68) | 0.007 | 1.67(1.62-2.22) | <0.001 |
| BMI | |||||||||||
| >24 | 84/126 | 69/53 | 43/10 | 1.95(1.24-3.07) | 0.004 | 6.45(3.07-13.54) | <0.001 | 1.78(0.99-3.20) | 0.054 | 2.67(1.76-4.04) | <0.001 |
| ≤24 | 120/170 | 113/128 | 51/43 | 1.25(0.89-1.77) | 0.203 | 1.68(1.05-2.68) | 0.030 | 2.50(1.58-3.97) | 0.000 | 1.36(0.99-1.87) | 0.058 |
Gene-environment interactions and breast cancer risk
Using cross-over analysis, we evaluated the gene-environment interactions (smoking and drinking) and the risk of breast cancer (Table 4). For non-smokers and non-drinkers, the TC genotype was not associated with an increased risk of breast cancer compared to the CC genotype. For smokers, the TC genotype increased the risk of breast cancer (OR=1.51, 95%CI: 1.12-2.02, P=0.006) compared to non-smokers carrying the CC genotype. For drinkers, an elevated risk of breast cancer was found in the TC genotype group compared to non-drinkers carrying the CC genotype (OR=1.53, 95%CI: 1.12-2.08, P=0.007). For non-smokers and non-drinkers, the TT genotype was associated with an increased breast cancer risk compared to the CC genotype, but smoking or drinking alone was not associated with an increased risk of breast cancer. However, drinkers and smokers carrying the TT genotype had a significantly increased risk of breast cancer compared to non-smokers and non-drinkers carrying the CC genotype (OR=5.56, 95%CI: 2.74-11.63, P<0.001; OR=5.12, 95%C: 2.37-11.06, P<0.001). These results suggest that the interactions between the genotype and smoking or drinking are associated with increased breast cancer risk in Southern Chinese women.
Table 4. Genetic environment factors 2*4 fork analysis.
| G | E | Case/control | OR (95%CI) | P |
| TT/CC | Smoking | |||
| + | + | 38/10 | 5.56(2.74-11.63) | <0.001 |
| + | - | 56/43 | 1.94(1.25-3.01) | 0.003 |
| - | + | 27/33 | 1.22(0.71-2.09) | 0.480 |
| - | - | 177/263 | 1.00 | |
| TC/CC | Smoking | |||
| + | + | 28/29 | 1.51(1.12-2.02) | 0.006 |
| + | - | 154/152 | 1.43(0.83-2.49) | 0.199 |
| - | + | 27/33 | 1.22(071-2.09) | 0.480 |
| - | - | 177/263 | 1.00 | |
| TT/CC | Drinking | |||
| + | + | 31/9 | 5.12(2.37-11.06) | <0.001 |
| + | - | 63/44 | 2.13(1.38-3.29) | 0.001 |
| - | + | 48/64 | 1.55(0.99-2.44) | 0.055 |
| - | - | 156/232 | 1.00 | |
| TC/CC | Drinking | |||
| + | + | 36/39 | 1.53(1.12-2.08) | 0.007 |
| + | - | 146/142 | 1.37(0.84-2.26) | 0.209 |
| - | + | 48/64 | 1.55(0.99-2.44) | 0.055 |
| - | - | 156/232 | 1.00 |
Association between rs4789936 polymorphism and clinical parameters of breast cancer patients
We further explored the association between clinical parameters and rs4789936 polymorphism among breast cancer patients (Table 5). Compared to CC genotype, the TT and CT genotypes were associated with lymph node metastases (TT: OR=2.54, 95%CI: 1.54-4.18, P<0.001; CT: OR=2.10, 95%CI: 1.40-3.15, P<0.001), advanced stage (TT: OR=1.91, 95%CI: 1.17-3.14, P=0.010; CT: OR=1.80, 95%CI: 1.20-2.70, P=0.005), and PR positive status (TT: OR=2.02, 95%CI: 1.23-3.32, P=0.005). The T genotype frequency was not related to tumor size, ER, Her-2, or treatment.
Table 5. Association between rs4789936 polymorphism and clinical parameters in patients with breast cancer.
| Parameters | CC | CT | P | TT | P | CT/TT | P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor size | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≥2cm vs<2cm | 107/97 | 92/90 | 51/43 | 143/133 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OR (95%CI) | 1.00 | 0.93(0.62-1.38) | 0.709 | 1.08(0.66-1.76) | 0.772 | 0.98(0.68-1.40) | 0.890 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lymph node metastases | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes vs No | 75/129 | 100/82 | 56/38 | 156/120 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OR (95%CI) | 1.00 | 2.10(1.40-3.15) | <0.001 | 2.54(1.54-4.18) | <0.001 | 2.24(1.54-3.24) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| III+IV/I+II | 76/128 | 94/88 | 50/44 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OR (95%CI) | 1.00 | 1.80(1.20-2.70) | 0.005 | 1.91(1.17-3.14) | 0.010 | 1.84(1.27-2.66) | 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ER | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive vs Negative | 92/112 | 75/107 | 38/56 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OR (95%CI) | 1.00 | 0.85(0.57-1.28) | 0.441 | 0.83(0.50-1.36) | 0.450 | 0.84(0.59-1.22) | 0.363 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PR | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive vs Negative | 86/118 | 91/91 | 56/38 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OR (95%CI) | 1.00 | 1.37(0.92-2.05) | 0.123 | 2.02(1.23-3.32) | 0.005 | 1.56(1.08-2.52) | 0.016 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Her-2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive vs Negative | 138/66 | 132/50 | 62/32 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OR (95%CI) | 1.00 | 1.26(0.82-1.95) | 0.297 | 0.93(0.55-1.56) | 0.773 | 1.13(0.77-1.67) | 0.535 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation vs Chemoradiotherapy | 74/130 | 65/117 | 27/67 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OR (95%CI) | 1.00 | 1.03(0.68-1.55) | 0.909 | 1.41(0.83-2.40) | 0.202 | 1.14(0.78-1.66) | 0.503 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ER, estrogen receptor; PR, progesterone receptor | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
TIMP-2 gene rs4789936 polymorphism and prognosis of breast cancer patients
Follow-up analysis of breast cancer patients showed a median survival time of 17.6 months. The Kaplan-Meier analysis indicated that the TT/CT genotypes had a poor survival prognosis compared to the CC genotype (CT vs CC: P=0.005; TT vs CC: P<0.001, Figure 1A). Patients with CT and TT genotypes also had poor survival rates (P<0.001, Figure 1B) compared to CC patients. The univariate cox regression indicated that CT and TT genotypes were associated with a poor overall survival prognosis (HR=5.97, 95%CI: 1.99-17.95, P=0.001; HR=5.28, 95%CI: 1.39-20.03, P=0.014). After adjusting the clinical parameters, the CT and TT genotypes (HR=4.65, 95%CI: 1.26-17.18, P=0.021; HR=6.62, 95%CI: 1.51-29.11, P=0.012) were still associated with poor prognosis of breast cancer patients (Table 6). Advanced stage, lymph node metastases, and Her-2 positive status were also predictors of a poor prognosis.
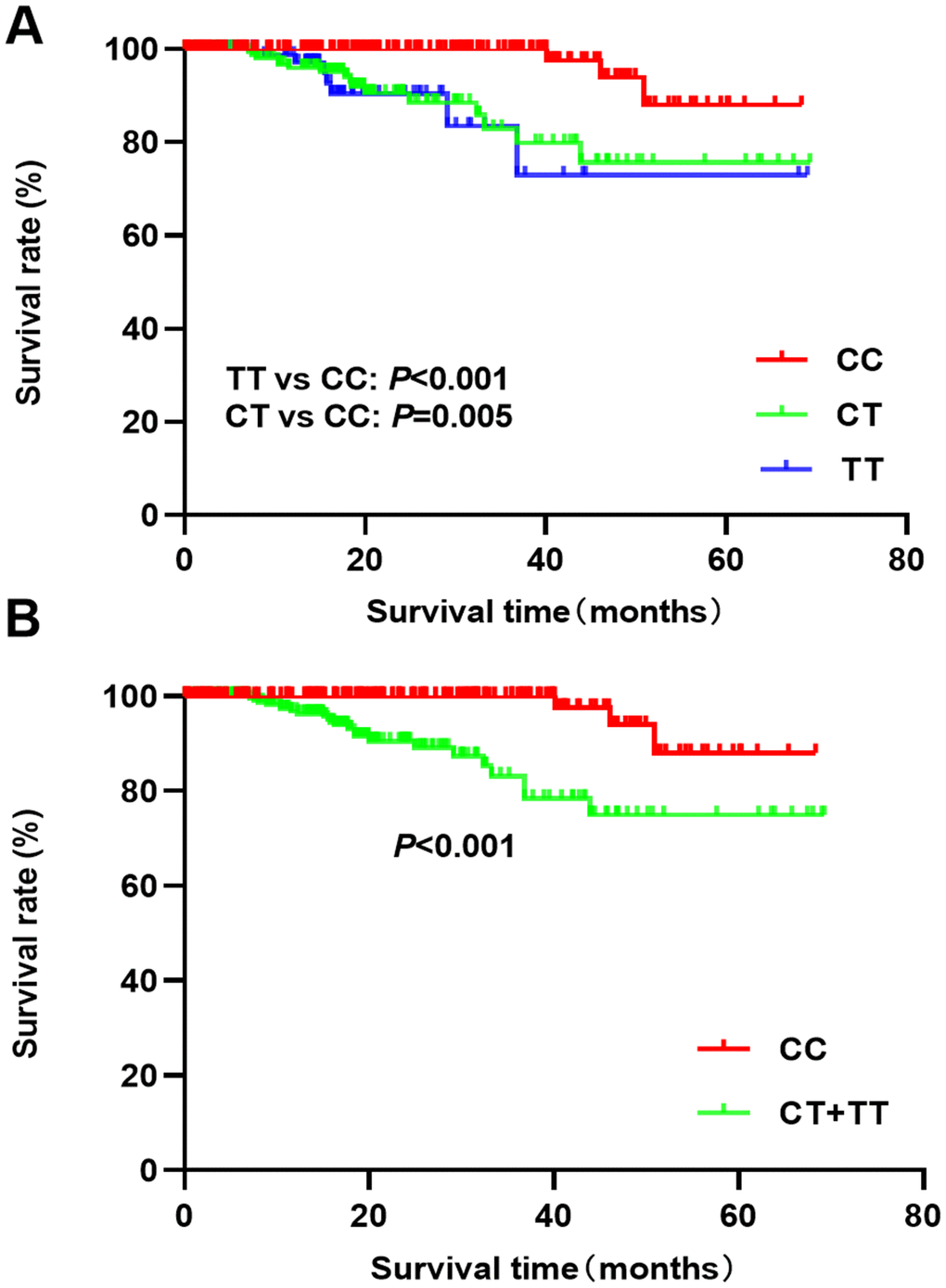
Figure 1. Kaplan-Meier analysis of breast cancer prognosis and rs4789936 polymorphism ((A) TT vs CT vs CC; (B) CT+TT vs CC).
Table 6. Prognostic factors in cox regression for breast cancer.
| Factors | Univariate analysis | Multivariate analysis | ||
| HR (95%CI) | P | HR (95%CI) | P | |
| Age,year | ||||
| ≥60 vs <60 | 1.43(0.64-3.19) | 0.386 | 1.24(0.54-2.83) | 0.617 |
| Smoking | ||||
| Yes vs No | 0.85(0.29-2.49) | 0.770 | 0.76(0.26-2.24) | 0.613 |
| Drinking | ||||
| Yes vs No | 0.36(0.11-1.19) | 0.094 | 0.55(0.15-2.00) | 0.365 |
| BMI | ||||
| >24 vs ≤24 | 1.06(0.48-2.36) | 0.890 | 0.87(0.38-2.00) | 0.747 |
| Tumor size | ||||
| ≥2cm vs <2cm | 0.82(0.37-1.81) | 0.617 | 0.46(0.20-1.09) | 0.077 |
| Lymph node metastases | ||||
| Yes vs No | 2.35(1.05-5.28) | 0.038 | 2.85(1.03-7.83) | 0.043 |
| Stage | ||||
| III/IV vs I/II | 3.81(1.52-9.51) | 0.004 | 1.91(1.67-4.76) | 0.016 |
| ER | ||||
| Positive vs negative | 0.76(0.33-1.77) | 0.524 | 0.61(0.24-1.58) | 0.306 |
| PR | ||||
| Positive vs negative | 1.40(0.64-3.09) | 0.403 | 0.62(0.26-1.47) | 0.273 |
| Her-2 | ||||
| Positive vs negative | 3.79(1.14-12.63) | 0.030 | 2.37(1.73-7.56) | 0.041 |
| Treatment | ||||
| Radiation vs chemoradiotherapy | 2.27(1.05-4.92) | 0.037 | 1.86(0.81-4.29) | 0.146 |
| rs4789936 | ||||
| CT vs CC | 5.97(1.99-17.95) | 0.001 | 4.65(1.26-17.18) | 0.021 |
| TT vs CC | 5.28(1.39-20.03) | 0.014 | 6.62(1.51-29.11) | 0.012 |
Discussion
The main findings of this study are the following: (1) The TIMP-2 gene rs4789936 polymorphism is associated with an increased risk of breast cancer in Southern Chinese women; (2) There are interactions between SNP rs4789936 polymorphism, smoking, drinking, and breast cancer risk; (3) The rs4789936 TT and CT genotypes are more frequent in breast cancer patients with lymph node metastases, advanced stage, and PR positive status; (4) The rs4789936 TT and CT genotypes are associated with a poor prognosis in breast cancer patients.
Tissue inhibitors of metalloproteinases (TIMPs) are natural inhibitors of MMPs that are overexpressed in many tumors and stromal tissues [23]. The TIMPs have two functional domains: the N-terminal region containing the MMP binding site, and the C-terminal region [24]. TIMP-2 inhibits the MMP-mediated proteolysis, and TIMP-2 decreased expression has been associated with cancer progression. TIMP-2 forms a stable complex with MMP-1 precursor to prevent MMP-1 activation. In addition, TIMP-2 can bind to MMP and directly inhibit its catalytic activity [25]. A previous study has indicated that the dynamic balance between TIMP-2 and MMP-2 can affect tumor invasion and metastasis [26]. A decreased expression of TIMP in tumor tissues results in increased activity of MMP and its increased degradation of the extracellular matrix, thus promoting tumor cell invasion and metastasis [27, 28].
The rs4789936 polymorphism is the transition from C to T that causes a synonymous amino acid change, which can result in dysregulated transcription of the TIMP-2 gene. TIMP-2 gene mutations have been associated with many diseases, including acne vulgaris [29], intracerebral hemorrhage [30], and cancer [31]. A previous study has indicated that TIMP-2 gene rs817990 and rs2277698 polymorphisms affect the risk of primary ovarian insufficiency in women [32]. In addition, two SNPs (rs7501477 and rs8136830) have been associated with an increased risk of breast cancer [33].
A recent case-control study to assess the association between TIMP-2 polymorphism and breast cancer in Northern Chinese population has suggested that the rs2277698 gene polymorphism is associated with a decreased risk of breast cancer [22]. This result is different from our study. Our results show that the T allele of rs4789936 is a risk factor for breast cancer in Southern Chinese women. There are several factors that may explain these differences. First, the eating habits are different between the two populations. In Northern China, people eat cooked wheat food, while in Southern China people prefer rice. Second, the environmental factors are different. It is wet in the South, while the North is dry. Third, there might be a clinical heterogeneity of breast cancer patients from different areas. Finally, some differences in sample size may be a potential factor.
Our cross-over analysis has demonstrated that compared to non-smokers and non-drinkers, women with TT/CT genotype, and drinking or smoking, have a significantly elevated risk for breast cancer, indicating a gene-environment interaction. In addition, we have found that rs4789936 polymorphism is associated with an advanced stage, lymph node metastases, and a PR positive status, indicating that the rs4789936 polymorphism may affect the prognosis of breast cancer patients. A previous study has also suggested that TIMP-2 gene rsrs8136803 TT genotype is associated with a poor disease-free survival [33]. Our data show that the rs4789936 TT/CT genotype is associated with a poor overall survival prognosis compared to the CC genotype, and suggest that the TT/CT genotype might serve as an independent prognosis factor of breast cancer patients. The GTEx data indicate that the rs4789936 TT genotype is quite common in blood samples, and has higher expression levels than the CC genotype. These findings suggest that TIMP-2 regulates breast cancer progression, and that TIMP-2 mutation might result in a poor prognosis in breast cancer patients.
Some of the limitations of this study include the fact that the selected population was recruited from a hospital, and a selection bias may thus exist. Although we identified an association between TIMP-2 gene polymorphism and breast cancer, the potential molecular mechanisms are unclear. There are chances that this SNP has an impact on another gene in which it resides, CEP295NL, because it is position within its 5'UT (HapMap data). Or, probably the truth is that this SNP is in large linkage disequilibrium with a nearby, functional SNP, and further research are required. Besides, since this study only explored gene-environment interactions, future studies should analyze gene-gene interactions that might affect the susceptibility to breast cancer.
In conclusion, our data demonstrate that the TIMP-2 gene rs4789936 polymorphism is associated with an elevated risk of breast cancer in Southern Chinese women, and indicate that it might serve as an independent prognosis factor for breast cancer patients.
Materials and Methods
Patient population
The study consisted of a case-control population and a follow-up population of breast cancer patients. For case-control population, we enrolled 480 breast cancer patients and 530 healthy controls from the Department of Women’s Cancer, Hunan Cancer Hospital (The Affiliated Cancer Hospital of Xiangya School of Medicine, Central South University). Criteria for enrollment were the following: All cases were newly diagnosed by pathology diagnosis; they were Han ethnic, and aged from 25 to 70 years old. Cases with other types of tumors, cardiovascular diseases, severe infectious disease, and immune disease were excluded. The control population was recruited from the physical examination center of the hospital during the same period. The follow-up population was from the case group.
General data of all human subjects were collected including age, height, and weight for calculating the body mass index (BMI), and the history of smoking and drinking. For the case group, we also collected the tumor size, lymph node metastases, stage, the status of estrogen receptor (ER), progesterone receptor (PR), Her-2, and treatment. A telephone follow-up was performed, and the primary outcome was overall survival (defined as the time from surgery to death). This study was approved by the ethics committee of Hunan Cancer Hospital. Written informed consent was obtained from all study subjects.
Blood sampling and genotyping
rs4789936 with minor allele frequency> 5% in East Asian population was selected for this study. 5 ml of fasting peripheral blood was collected from each participant, and was stored in anti-coagulative tubes with EDTA-disodium salt. The genomic DNA was extracted by TaKaRa Genome DNA Extraction Kit (Dalian Biological Engineering CO., LTD, China), and stored at -20°C.
The single nucleotide polymorphism (SNP) was genotyped by polymerase chain reaction restriction fragment length polymorphism (PCR-RFLP) method. The primer sequences of TIMP-2 gene rs4789936 were designed by Primer Premier 5.0, and synthesized in Shanghai Sangon biotech Co., Ltd (ACGTTGGATGGCGTC, TCACTACCTACAAAG). The PCR reaction was performed in a total volume of 25 μl, including 5.0 μl of DNA template, 2.5μl of 10×Buffer, 2.0μl of MgC12, 0.5μl of dNTPs, each 0.5μl of forward and reverse primers, 0.2μl of Taq enzyme, and 13.8 μl of ddH2O. The amplification conditions of PCR were as follows, starting with pre-denaturation at 94°C for 2 min; followed by 36 cycles of 94°C degeneration for 20s, 53°C annealing for 30s, 72°C extension for 30s; and finally, 72°C extension for 10 min. Purity of the PCR products was examined by 2% agarose gel electrophoresis. The PCR products of rs4789936 were digested, and the digested products were analyzed by 3% agarose gel electrophoresis and visualized by UV light. Random samples of the PCR products were selected for direct sequencing to verify the accuracy and reliability of the genotyping results.
Statistical analysis
SPSS 20.0 statistical package was used to perform the statistical analyses. The quantitative variables were expressed using mean±standard deviation, and t test was used for the comparison between case group and control group. The genotype frequency of cases and controls was compared using the Chi-square test. Hardy-Weinberg equilibrium test was performed in the control group. For rs4789936 polymorphism, we used five models for compassion: homozygote model (TT vs CC), heterozygote model (CT vs CC), dominant model (CT+TT vs CC), recessive model (TT vs CT+CC), and allele model (T vs C). Univariate and multivariate (adjusting for age, BMI, smoking, and drinking) logistic regression analyses were used to calculate the odds ratios (ORs) of breast cancer, and the 95% confidence intervals (CIs). Stratified analysis was performed according to age (≥60 vs <60), smoking (Yes vs No), drinking (Yes vs No), and BMI (>24 vs ≤24). The cross-over analysis was used to estimate the gene-environment interactions (gene-smoking, gene-drinking). We also explored the relationship between clinical parameters and genotype distribution (tumor size, lymph node metastases, stage, ER, PR, Her-2 and treatment). The Kaplan-Meier analysis was used to compare overall survival (OS) among different genotypes. Univariate and multivariate (age, smoking, drinking, tumor size, lymph node metastases, stage, ER, PR, Her-2 and treatment) cox regression analyses were used to determine the relationship between genotype and breast cancer prognosis. Hazard ratio (HR) and 95% CI were also calculated. P<0.05 was considered significant.
Author Contributions
LJY designed the study. LGM analyzed and interpreted the data, and wrote the manuscript. LJY proofread the final version. All authors read and approved the final manuscript.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This study is supported by the Natural Science Foundation of Hunan Province (2020JJ8063).
References
- 1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018; 68:394–424. https://doi.org/10.3322/caac.21492 [PubMed]
- 2. Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J. Cancer statistics in China, 2015. CA Cancer J Clin. 2016; 66:115–32. https://doi.org/10.3322/caac.21338 [PubMed]
- 3. Feng RM, Zong YN, Cao SM, Xu RH. Current cancer situation in China: good or bad news from the 2018 global cancer statistics? Cancer Commun (Lond). 2019; 39:22. https://doi.org/10.1186/s40880-019-0368-6 [PubMed]
- 4. Chen W. Cancer statistics: updated cancer burden in China. Chin J Cancer Res. 2015; 27:1. https://doi.org/10.3978/j.issn.1000-9604.2015.02.07 [PubMed]
- 5. Ma HM, Wang L, Shi JF, Ying JM, Zhu J, Chen LL, Yue XP, Gong JY, Li X, Wang JL, Dai M. [A systematic review of international simulation models on the natural history of breast cancer: current understanding and challenges for Chinese-population-specific model development]. Zhonghua Liu Xing Bing Xue Za Zhi. 2017; 38:1419–25. https://doi.org/10.3760/cma.j.issn.0254-6450.2017.10.025 [PubMed]
- 6. Abdel-Rahman O, Cheung WY. Impact of smoking history on the outcomes of women with early-stage breast cancer: a secondary analysis of a randomized study. Med Oncol. 2018; 35:68. https://doi.org/10.1007/s12032-018-1129-0 [PubMed]
- 7. Ellingjord-Dale M, Vos L, Hjerkind KV, Hjartåker A, Russnes HG, Tretli S, Hofvind S, Dos-Santos-Silva I, Ursin G. Alcohol, physical activity, smoking, and breast cancer subtypes in a large, nested case-control study from the norwegian breast cancer screening program. Cancer Epidemiol Biomarkers Prev. 2017; 26:1736–44. https://doi.org/10.1158/1055-9965.EPI-17-0611 [PubMed]
- 8. Laudisio D, Barrea L, Muscogiuri G, Annunziata G, Colao A, Savastano S. Breast cancer prevention in premenopausal women: role of the mediterranean diet and its components. Nutr Res Rev. 2019. [Epub ahead of print]. https://doi.org/10.1017/S0954422419000167 [PubMed]
- 9. Lovett JL, Chima MA, Wexler JK, Arslanian KJ, Friedman AB, Yousif CB, Strassmann BI. Oral contraceptives cause evolutionarily novel increases in hormone exposure: a risk factor for breast cancer. Evol Med Public Health. 2017; 2017:97–108. https://doi.org/10.1093/emph/eox009 [PubMed]
- 10. Chandler MR, Bilgili EP, Merner ND. A review of whole-exome sequencing efforts toward hereditary breast cancer susceptibility gene discovery. Hum Mutat. 2016; 37:835–46. https://doi.org/10.1002/humu.23017 [PubMed]
- 11. Irminger-Finger I, Siegel BD, Leung WC. The functions of breast cancer susceptibility gene 1 (BRCA1) product and its associated proteins. Biol Chem. 1999; 380:117–28. https://doi.org/10.1515/BC.1999.019 [PubMed]
- 12. Hao JQ, Zhang QK, Zhou YX, Chen LH, Wu PF. Association between circulating leptin concentration and G-2548A gene polymorphism in patients with breast cancer: a meta-analysis. Arch Med Sci. 2019; 15:275–83. https://doi.org/10.5114/aoms.2018.75638 [PubMed]
- 13. Ibrahimi M, Jamalzei B, Akbari ME, Ibrahimi R, Alaei M, Moossavi M, Mohammadoo-Khorasani M. Association between interleukin 4 (IL-4) VNTR, gene polymorphism, and breast cancer susceptibility in Iranian population: experimental and web base analysis. Bratisl Lek Listy. 2018; 119:651–54. https://doi.org/10.4149/BLL_2018_116 [PubMed]
- 14. Costa-Silva DR, Barros-Oliveira MD, Borges RS, Tavares CB, Borges US, Alves-Ribeiro FA, Silva VC, Silva BB. Insulin-like growth factor 1 gene polymorphism and breast cancer risk. An Acad Bras Cienc. 2016; 88:2349–56. https://doi.org/10.1590/0001-3765201620160169 [PubMed]
- 15. Verstappen J, Von den Hoff JW. Tissue inhibitors of metalloproteinases (TIMPs): their biological functions and involvement in oral disease. J Dent Res. 2006; 85:1074–84. https://doi.org/10.1177/154405910608501202 [PubMed]
- 16. Li X, Deng D, Xue J, Qu L, Achilefu S, Gu Y. Quantum dots based molecular beacons for in vitro and in vivo detection of MMP-2 on tumor. Biosens Bioelectron. 2014; 61:512–18. https://doi.org/10.1016/j.bios.2014.05.035 [PubMed]
- 17. Ara T, Kusafuka T, Inoue M, Kuroda S, Fukuzawa M, Okada A. Determination of imbalance between MMP-2 and TIMP-2 in human neuroblastoma by reverse-transcription polymerase chain reaction and its correlation with tumor progression. J Pediatr Surg. 2000; 35:432–37. https://doi.org/10.1016/s0022-3468(00)90208-2 [PubMed]
- 18. Zhang DY, Wang J, Zhang GQ, Chu XQ, Zhang JL, Zhou Y. Correlations of MMP-2 and TIMP-2 gene polymorphisms with the risk and prognosis of gastric cancer. Int J Clin Exp Med. 2015; 8:20391–401. [PubMed]
- 19. Bayramoglu A, Gunes HV, Metintas M, Değirmenci I, Mutlu F, Alataş F. The association of MMP-9 enzyme activity, MMP-9 C1562T polymorphism, and MMP-2 and -9 and TIMP-1, -2, -3, and -4 gene expression in lung cancer. Genet Test Mol Biomarkers. 2009; 13:671–78. https://doi.org/10.1089/gtmb.2009.0053 [PubMed]
- 20. Yaykaşli KO, Kayikçi MA, Yamak N, Soğuktaş H, Düzenli S, Arslan AO, Metın A, Kaya E, Hatıpoğlu ÖF. Polymorphisms in MMP-2 and TIMP-2 in turkish patients with prostate cancer. Turk J Med Sci. 2014; 44:839–43. [PubMed]
- 21. Wang N, Zhou S, Fang XC, Gao P, Qiao Q, Wu T, He XL. MMP-2, -3 and TIMP-2, -3 polymorphisms in colorectal cancer in a Chinese han population: a case-control study. Gene. 2020; 730:144320. https://doi.org/10.1016/j.gene.2019.144320 [PubMed]
- 22. Wang K, Wang G, Huang S, Luo A, Jing X, Li G, Zhou Y, Zhao X. Association between TIMP-2 gene polymorphism and breast cancer in han Chinese women. BMC Cancer. 2019; 19:446. https://doi.org/10.1186/s12885-019-5655-8 [PubMed]
- 23. Chowdhury A, Brinson R, Wei B, Stetler-Stevenson WG. Tissue inhibitor of metalloprotease-2 (TIMP-2): bioprocess development, physicochemical, biochemical, and biological characterization of highly expressed recombinant protein. Biochemistry. 2017; 56:6423–33. https://doi.org/10.1021/acs.biochem.7b00700 [PubMed]
- 24. Vincent ZL, Mitchell MD, Ponnampalam AP. Regulation of MT1-MMP/MMP-2/TIMP-2 axis in human placenta. J Inflamm Res. 2015; 8:193–200. https://doi.org/10.2147/JIR.S88039 [PubMed]
- 25. Niu LN, Zhang L, Jiao K, Li F, Ding YX, Wang DY, Wang MQ, Tay FR, Chen JH. Localization of MMP-2, MMP-9, TIMP-1, and TIMP-2 in human coronal dentine. J Dent. 2011; 39:536–42. https://doi.org/10.1016/j.jdent.2011.05.004 [PubMed]
- 26. van Waveren C, Sun Y, Cheung HS, Moraes CT. Oxidative phosphorylation dysfunction modulates expression of extracellular matrix—remodeling genes and invasion. Carcinogenesis. 2006; 27:409–18. https://doi.org/10.1093/carcin/bgi242 [PubMed]
- 27. Bekes EM, Schweighofer B, Kupriyanova TA, Zajac E, Ardi VC, Quigley JP, Deryugina EI. Tumor-recruited neutrophils and neutrophil TIMP-free MMP-9 regulate coordinately the levels of tumor angiogenesis and efficiency of Malignant cell intravasation. Am J Pathol. 2011; 179:1455–70. https://doi.org/10.1016/j.ajpath.2011.05.031 [PubMed]
- 28. Culhaci N, Metin K, Copcu E, Dikicioglu E. Elevated expression of MMP-13 and TIMP-1 in head and neck squamous cell carcinomas may reflect increased tumor invasiveness. BMC Cancer. 2004; 4:42. https://doi.org/10.1186/1471-2407-4-42 [PubMed]
- 29. Gao R, Yu H, Zhao Q, Wang S, Bai B. Role of MMP-2(-1306 C/T) and TIMP-2(-418G/C) polymorphism in Chinese han patients with acne vulgaris. Biomed Res Int. 2019; 2019:2364581. https://doi.org/10.1155/2019/2364581 [PubMed]
- 30. Reuter B, Bugert P, Stroick M, Bukow S, Griebe M, Hennerici MG, Fatar M. TIMP-2 gene polymorphism is associated with intracerebral hemorrhage. Cerebrovasc Dis. 2009; 28:558–63. https://doi.org/10.1159/000247599 [PubMed]
- 31. Zhang S, Gao X, Yang J, Ji Z. TIMP-2 G-418C polymorphism and cancer risk: a meta-analysis. J Cancer Res Ther. 2015; 11:308–12. https://doi.org/10.4103/0973-1482.140961 [PubMed]
- 32. An HJ, Ahn EH, Kim JO, Park HS, Ryu CS, Cho SH, Kim JH, Lee WS, Kim NK. Association between tissue inhibitor of metalloproteinase (TIMP) genetic polymorphisms and primary ovarian insufficiency (POI). Maturitas. 2019; 120:77–82. https://doi.org/10.1016/j.maturitas.2018.11.018 [PubMed]
- 33. Peterson NB, Beeghly-Fadiel A, Gao YT, Long J, Cai Q, Shu XO, Zheng W. Polymorphisms in tissue inhibitors of metalloproteinases-2 and -3 and breast cancer susceptibility and survival. Int J Cancer. 2009; 125:844–50. https://doi.org/10.1002/ijc.24405 [PubMed]


