Introduction
Evidence has suggested that the normal aging process involves a wide range of physiological changes, among which impairment in the initiation and maintenance of sleep in older age is one of the most pervasive [1]. For example, a seminal meta-analysis based on objective measures reported that, in healthy individuals aged 60 years or older, sleep efficiency continued to decrease with aging while changes in sleep latency and total sleep time were not significant [2]. These age-associated changes in sleep (AACS) in healthy older adults, based on both subjective and objective measures, have been investigated by a host of researchers over the past several decades (Supplementary Table 1) providing evidence for clinical care guidelines.
As for the methods used to obtain measurements of sleep, previous literature suggested that subjective reports could be biased by personality [3], mood, or memory [4]. However, it has also been proposed that subjective measures might reflect physiological characteristics or internal factors that are fundamentally distinct from objective findings [5, 6] and have their own clinical significance. Additionally, the self-perception of sleep habits differs by sex, with women reporting more frequent sleep disturbances [7], and a much-increased sleep latency [8], compared with men.
However, most studies on subjective AACS were cross-sectional. Since rapidly changing sociocultural factors, such as gender roles, influence sleep considerably [9, 10], cross-sectional comparisons of sleep between different age groups may be biased by cohort effects [11] and may not reliably capture intraindividual AACS. Furthermore, AACS has barely been prospectively investigated in older populations. Although there have been several prospective studies on AACS, they examined adolescents or individuals under 70 years of age [12–14], were limited to the assessment of sleep duration, efficiency, or the frequency of sleep disturbances [12–15], and showed a high number of missing data with non-random dropouts [15]. Moreover, no study has thus far focused on sex differences in subjective AACS in the elderly using a longitudinal design.
In this study, we prospectively investigated a large, nationwide, randomly-sampled, community-dwelling elderly population without major psychiatric or neurological disorders to examine the sex difference in subjective AACS.
Results
Supplementary Figure 1 shows the flow of study participants. We had 4,686 individuals at baseline after excluding those with significant psychiatric or neurological disorders, of whom 2,248 completed the 6-year follow-up. Participant characteristics at baseline are presented by sex (Table 1) and at eave assessment wave (Supplementary Table 2). Men were younger, more educated, more likely to be employed, less likely to be socioeconomically disadvantaged and to live alone, consumed more alcohol, cigarettes, and coffee, were less depressive, more physically active, more likely to be ill, and less likely to be diagnosed with mild cognitive impairment (MCI) than women at baseline. The mean (SD) follow-up duration of participants was 3.87 (2.35) years. During this period, 600 (12.8 %) participants reported having taken sleeping pills at least once. Compared with those who were lost at any follow-up assessment, participants who completed all four waves were younger (mean age [SD]; 68.52 [5.68] vs. 70.98 [7.02], p < 0.001), more educated (mean years of education [SD]; 9.16 [5.19] vs. 7.94 [5.31], p < 0.001), less likely to live in rural areas (22.8 % vs. 28.4 %, p < 0.001), less likely to live alone (11.6 % vs. 14.3 %, p = 0.005), less depressive (mean Geriatric Depression Scale [GDS] score [SD]; 7.14 [4.16] vs. 7.39 [4.02], p = 0.044), more physically active (total energy expenditure in kilocalories per week over the last year [SD]; 82.73 [156.59] vs. 68.70 [142.14], p = 0.001), and less likely to be diagnosed with MCI (20.9 % vs. 29.9 %, p < 0.001). There were no observable differences between the groups in terms of sex ratio, employment status, socioeconomic status, the average amount of alcohol, cigarettes, and coffee consumed, Pittsburgh Sleep Quality Index (PSQI) score, and Cumulative Illness Rating Scale (CIRS) total score.
Table 1. Baseline characteristics of the study participants.
| Men (N = 2,148) | Women (N = 2,538) | pa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age, year | 69.39 (6.31) | 69.99 (6.61) | 0.002 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Education, year | 10.76 (4.89) | 6.71 (4.88) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Employed (%) | 1,011 (47.1) | 512 (20.2) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Low SES (%)a | 44 (2.1) | 99 (3.9) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Living in a rural area (%) | 546 (25.5) | 643 (25.5) | 0.985 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Living alone (%) | 105 (4.9) | 496 (19.6) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alcohol, SU/weekb | 7.88 (16.04) | 0.60 (6.50) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking, packs/dayb | 0.18 (0.78) | 0.01 (0.11) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Coffee, cups/dayb | 1.66 (1.98) | 0.95 (1.12) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GDS, score | 6.75 (4.03) | 7.70 (4.10) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical activity, kcal/weekb | 108.62 (174.65) | 48.24 (118.36) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CIRS total score | 4.44 (2.84) | 4.11 (2.60) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diagnosed with MCI (%) | 486 (22.6) | 700 (27.6) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Values are mean (standard deviation) unless specified otherwise. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| a Student t-test for continuous variables and χ2 test for categorical variables. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| b Amount averaged over the past one year. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: CIRS, Cumulative Illness Rating Scale; GDS, Geriatric Depression Scale; MCI, mild cognitive impairment; SU, standard unit. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Linear mixed-effects models for sleep measures obtained from the PSQI showed that, overall, participants’ sleep latency increased, and daytime dysfunction and sleep quality worsened over 6 years in both the unadjusted and adjusted models. In the adjusted model, women showed shorter sleep duration and more severe daytime dysfunction than men (Table 2, Figure 1).
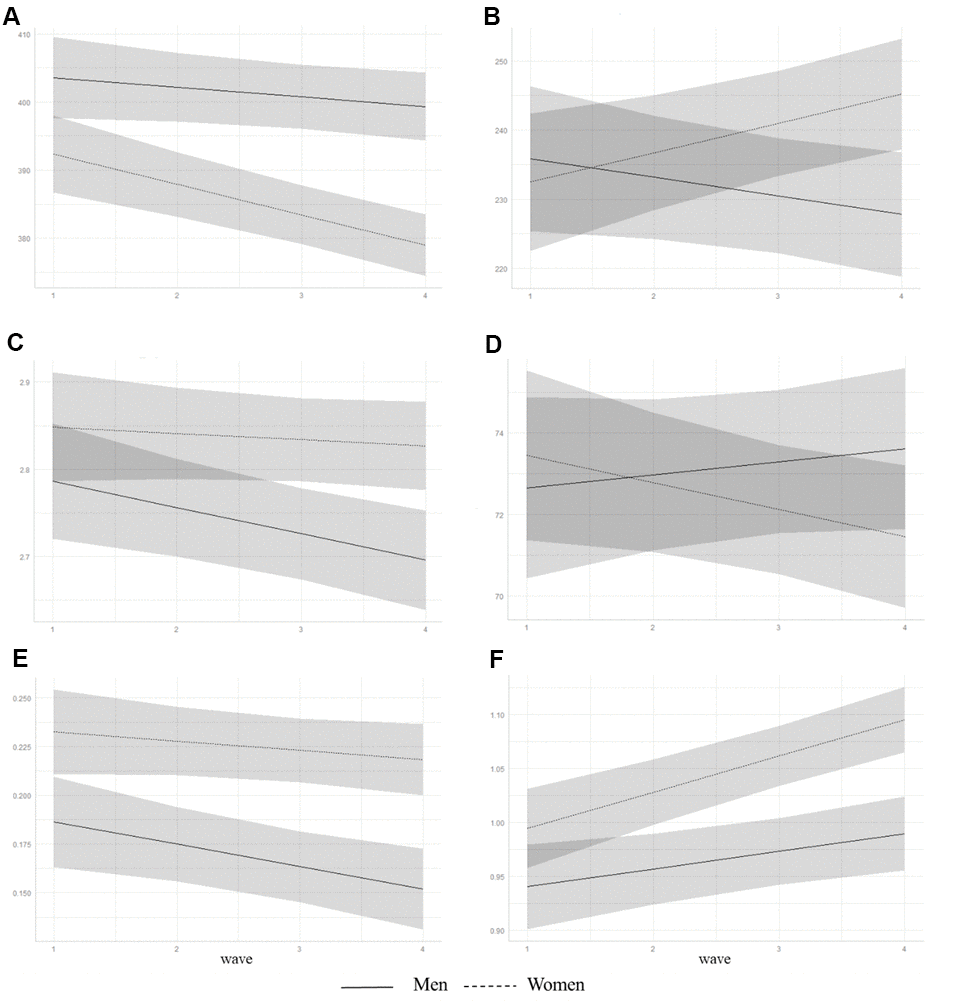
Figure 1. Trajectories of predicted subjective sleep measures from adjusted linear mixed-effects models for men and women. Predicted values of (A) sleep duration, min; (B) mid-sleep time, min; (C) loge transformed sleep latency, min; (D) sleep efficiency, %; (E) loge transformed daytime dysfunction, points; and (F) sleep quality, points. Shaded area represents 95% confidence intervals.
Table 2. Unadjusted and adjusted coefficients for sleep measures using linear mixed-effects models.
| Variable | Unadjusted | Adjusteda | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Coefficient (95% CI) | p | Coefficient (95% CI) | p | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sleep duration | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intercept | 396.70 (392.67 to 400.72) | <0.001 | 375.32 (340.40 to 410.25) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time | -0.55 (-1.96 to 0.86) | 0.442 | -1.42 (-3.21 to 0.37) | 0.121 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex | -5.13 (-10.59 to 0.33) | 0.066 | -8.20 (-14.65 to -1.74) | 0.013 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time * Sex | -3.20 (-5.11 to -1.30) | 0.001 | -3.04 (-5.00 to -1.08) | 0.002 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mid-sleep time | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intercept | 236.66 (229.41 to 243.90) | <0.001 | 301.83 (240.27 to 363.33) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time | -4.14 (-6.72 to -1.56) | 0.002 | -2.67 (-5.93 to 0.59) | 0.109 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex | -21.14 (-30.96 to -11.31) | <0.001 | -10.23 (-21.89 to 1.44) | 0.086 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time * Sex | 6.67 (3.18 to 10.15) | <0.001 | 6.90 (3.26 to 10.54) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sleep latencyb | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intercept | 2.79 (2.74 to 2.83) | <0.001 | 2.42 (2.04 to 2.81) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time | -0.03c (-0.05 to -0.02) | <0.001 | -0.03c (-0.05 to -0.01) | 0.005 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex | 0.15d (0.08 to 0.21) | <0.001 | 0.04 (-0.04 to 0.11) | 0.303 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time * Sex | 0.02 (-0.002 to 0.04) | 0.077 | 0.02 (-0.001 to 0.05) | 0.057 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sleep efficiency | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intercept | 71.33 (69.68 to 72.97) | <0.001 | 66.36 (53.47 to 79.25) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time | 0.55 (-0.08 to 1.18) | 0.088 | 0.32 (-0.44 to 1.08) | 0.410 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex | 2.64 (0.41 to 4.88) | 0.021 | 1.77 (-0.88 to 4.42) | 0.191 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time * Sex | -1.03 (-1.88 to -0.17) | 0.018 | -0.98 (-1.87 to -0.09) | 0.031 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Daytime dysfunctionb | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intercept | 0.22 (0.20 to 0.24) | <0.001 | -0.11 (-0.24 to 0.02) | 0.111 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time | -0.01e (-0.02 to -0.004) | 0.003 | -0.01e (-0.02 to -0.003) | 0.006 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex | 0.05f (0.03 to 0.08) | <0.001 | 0.04g (0.01 to 0.07) | 0.007 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time * Sex | 0.003 (-0.007 to 0.012) | 0.557 | 0.01 (-0.003 to 0.02) | 0.182 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sleep quality | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intercept | 0.95 (0.92 to 0.98) | <0.001 | 0.79 (0.56 to 1.02) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time | 0.02 (0.01 to 0.03) | 0.003 | 0.02 (0.003 to 0.03) | 0.013 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex | 0.08 (0.04 to 0.12) | <0.001 | 0.04 (-0.01 to 0.08) | 0.113 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time * Sex | 0.01 (-0.001 to 0.028) | 0.070 | 0.02 (0.002 to 0.03) | 0.024 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| a Adjusted for age, years of education, employment status, socioeconomic status (whether covered by National Medicaid Program), place of residence (urban vs. rural), presence of cohabitants, physical activity, Geriatric Depression Scale score, amount of alcohol, smoking, and coffee in the past one year, total score of Cumulative Illness Rating Scale, whether diagnosed with mild cognitive impairment, whether being at high risk of obstructive sleep apnea or REM sleep behavior disorder, birth cohort (age < 69 vs. ≥ 69 at baseline), and usage of sleeping pills in the past one month | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| b Loge transformed; c +0.97 in minutes; d +1.16 in minutes; e +0.99 in points; f +1.05 in points; g +1.04 in points | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: CI, confidence interval | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
We also found a significant sex difference in AACS for sleep duration, mid-sleep time, sleep efficiency, and sleep quality under the adjusted model. Post hoc analyses revealed that only women showed decreased sleep duration, delayed mid-sleep time, and decreased sleep efficiency over a period of 6 years (Table 3). Sleep quality worsened in both groups but a more pronounced change was observed in women. The AACS of daytime dysfunction was found only in men with a worsening trend.
Table 3. Adjusted coefficients for sleep measures of men and women using linear mixed-effects models.
| Variable | Mena | Womena | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Coefficient (95% CI) | p | Coefficient (95% CI) | p | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sleep duration | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intercept | 350.25 (299.18 to 401.31) | <0.001 | 381.65 (335.33 to 428.01) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time | -1.91 (-3.98 to 0.16) | 0.072 | -4.22 (-6.16 to -2.28) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mid-sleep time | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intercept | 345.66 (252.47 to 438.73) | <0.001 | 259.80 (180.52 to 339.06) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time | -1.92 (-5.79 to 1.96) | 0.333 | 3.87 (0.46 to 7.28) | 0.026 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sleep latencyb | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intercept | 2.67 (2.09 to 3.24) | <0.001 | 2.29 (1.79 to 2.80) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time | -0.02c (-0.05 to 0.002) | 0.076 | -0.01d (-0.03 to 0.01) | 0.263 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sleep efficiency | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intercept | 70.65 (51.21 to 90.10) | <0.001 | 6.40 (47.43 to 80.62) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time | 0.45 (-0.42 to 1.32) | 0.309 | -0.85 (-1.62 to -0.07) | 0.033 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Daytime dysfunctionb | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intercept | -0.38 (-0.68 to -0.08) | 0.012 | 0.07 (-0.21 to 0.35) | 0.626 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time | -0.02e (-0.03 to -0.01) | 0.007 | -0.004f (-0.02 to 0.01) | 0.537 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sleep quality | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intercept | 0.86 (0.52 to 1.19) | <0.001 | 0.81 (0.51 to 1.10) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time | 0.02 (0.003 to 0.03) | 0.020 | 0.03 (0.02 to 0.05) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| a Adjusted for age, years of education, employment status, socioeconomic status (whether covered by National Medicaid Program), place of residence (urban vs. rural), presence of cohabitants, physical activity, Geriatric Depression Scale score, amount of alcohol, smoking, and coffee in the past one year, total score of Cumulative Illness Rating Scale, whether diagnosed with mild cognitive impairment, whether being at high risk of obstructive sleep apnea or REM sleep behavior disorder, birth cohort (age < 69 vs. ≥ 69 at baseline), and usage of sleeping pills in the past one month | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| b Loge transformed; c +0.98 in minutes; d +0.99 in minutes; e +0.98 in points; f +1.00 in points | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: CI, confidence interval | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Discussion
This study found that community-dwelling healthy elderly Koreans did report changes in subjective sleep habits over time, such that sleep latency increased, and daytime dysfunction and sleep quality worsened over 6 years, while sleep duration, mid-sleep time, and sleep efficiency were largely unchanged. However, we observed significant sex differences in AACS: for every two-year increase in age, women showed a shortening sleep duration by 4.22 minutes, delayed mid-sleep time by 3.87 min, and worsening sleep efficiency by 0.85%. Sleep quality worsened in both men and women by 0.02 and 0.03 points, respectively, with women showing a more statistically pronounced change. In addition, every two years, daytime dysfunction worsened by 0.98 points in men, while no substantial changes were observed in women.
To the best of our knowledge, there have been only a few prospective studies on subjective AACS that included a sizable elderly population. In one study, for every 2 years, weekday sleep duration increased by approximately 15 min whereas weekend sleep duration decreased by approximately 1.5 min compensatorily over 8 years in 8,159 participants aged 57 – 68 years after adjusting for sex and occupation [14]. The researchers suggested that the increase in weekday sleep duration may have been attributed to the retirement of the elderly participants during the follow-up period. However, that study included fairly young elderly adults and the analyses did not adjust for important confounders such as usage of sleeping pills. A recent cohort study with an initial sample size of 6,375 adults aged 42 – 94 years who were followed up to 27 years, reported that sleep efficiency decreased by 3.1% per decade [15]. Though that study accounted for numerous variables such as social class, subjective health rating, marital and working status, and usage of sleeping pills in their linear mixed-effects model, the analysis was not adjusted for cognitive function, as people with MCI could distort the subjective sleep measures [6, 16]. Highly irregular drop-out rates between assessment waves of that study was another limitation that could be a source of bias.
A seminal meta-analysis based on cross-sectional studies of sleep measures by polysomnography or actigraphy suggested that, after 60 years of age, total sleep time decreased non-significantly, sleep latency increased non-significantly, and sleep efficiency decreased significantly, with women having a larger effect size than men [2]. These results were largely in accordance with ours notwithstanding the apparent discrepancy between using self-reported and objective sleep measures.
We also found that, in case of mid-sleep time, men showed a nonsignificant advance while women exhibited a significant delay. These results could be contradictory to the common knowledge that aging is generally characterized by the advance of bedtime and wake-up time to earlier hours [17]. However, a cross-sectional telephone survey conducted in a metropolitan area of France involving 1,026 participants aged 60 and older indicated that the advancement of bedtime and wake-up time was not evident, and even a delaying tendency was observed between women aged 60–64 years and 65–69 years [18]. This phenomenon could be partly explained by the homeostatic effect of sleep need. An increase in sleep need, as shown by pronounced worsening of sleep quality in women, might advance bedtime or delay wake-up time [19] which in turn, coupled with a nonsignificant increase in sleep latency in women as shown in our findings, could lead to a delay in mid-sleep time. It is also possible that the relatively short follow-up period of 6 years could not capture the secular trend of mid-sleep time.
In regard to the self-reported overall sleep quality, which should be distinguished from the global PSQI score that reflects both qualitative and quantitative aspects, previous studies have shown conflicting results in the elderly population. There was a report of a worsening trend of the sleep quality component score from the PSQI in 824 randomly-sampled Japanese elderly participants aged older than 60 years in a cross-sectional study, with women having a more marked change [20], which is in line with our findings. On the other hand, a cross-sectional study from the HypnoLaus Cohort reported that the sleep quality component score from the PSQI improved steadily with age in the 2,966 participants aged between 40 and 80 years old [8], indicating that a spontaneous adaptive adjustment of sleep disturbances might occur in the elderly. However, the latter study excluded approximately 40% of the initial sample of participants who had sleep complaints or any documented sleep disorders, which could have led to a bias toward a super-healthy population.
The underlying mechanisms of the sex differences in AACS or of the individual sleep measure itself are yet to be elucidated. Zhang et al. suggested that this disparity may be attributable to the higher prevalence of depressive mood or anxiety in women compared with men [21]. Though we adjusted our models for depression by including GDS score, it still remains possible that the observed sex difference in AACS is influenced by the presence of affective disorders. Another possible explanation for this phenomenon is the difference between the sexes in the age-associated changes in sex hormones. In older men, sleep fragmentation due to age-associated decrease in testosterone levels could be attenuated by the loss of diurnal fluctuation of the hormone [22]. In contrast, in women, a progressive decrease of estradiol level after menopause may disturb sleep, prolong sleep latency [23], and lead to sleep-disordered breathing through its detrimental effect on the upper respiratory tract [24]. Additionally, women have heightened bodily vigilance and tend to express more somatic symptoms or emotional distress than men [25]. We suggest that it might be the case that subjective AACS concerning sleep duration, mid-sleep time, sleep efficiency, and overall sleep quality might be particularly vulnerable to these effects, though further research is warranted to ascertain these hypotheses.
This study has several limitations. First, the self-reported sleep measures used in our study may lead to a reporting bias related to, as mentioned above, personality, mood, and memory [3, 4]. Nevertheless, there have been reports regarding decent correlations between PSQI and polysomnographic findings in terms of sleep efficiency and latency [26], and between a questionnaire assessing mid-sleep time and sleep duration and corresponding actigraphy findings [27]. Moreover, because self-reported measures are inexpensive and easy to apply, they are highly efficient, and probably the only practical way to collect data over a long-term period with a large sample size. Second, it is possible that 6 years of follow-up was not long enough to capture AACS, leading to false-negative study results. Third, the difference in sleep habits between weekdays and weekends was not taken into account. However, by adjusting for employment status in our analysis models, we believe that we partially compensated for this drawback. Fourth, we did not quantify the duration of naps, which preclude the estimation of sleep duration over a 24-hour period, though instead, we did measure the degree of daytime dysfunction. Fifth, the concept of the “normality” in regard to sleep is difficult to define to date. According to Mowbray et al. [28], the word “normal” can imply several meanings. For practical purposes, we use it in terms of the “statistical” norm where the abnormal is perceived to be that which lies outside the population average range, rather than the “value” norm which takes the ideal, healthier state as its concept. Therefore, we included elderly participants with common sleep problems but excluded those with severe psychiatric or neurologic disorders and with cognitive impairment that could significantly compromise the reliability of the self-reported sleep measures. Sixth, because the participants who completed all follow-ups had substantially different characteristics compared with those who dropped out, with approximately 20% attrition rate per two years, it raised the possibility of bias in our assessment.
In conclusion, for the healthy individuals aged 60 years or older, normative age-associated changes in subjective sleep measures do occur in latency, daytime dysfunction, and sleep quality. As for sex differences, decreased sleep duration, delayed mid-sleep time, and decreased sleep efficiency were found in women, and the worsening of sleep quality was more pronounced in women than in men. It would be imperative for clinicians to understand these changes in sleep habits when determining the necessity to treat declared sleep disturbances of the elderly population.
Materials and Methods
Participants
This study was conducted as a part of the Korean Longitudinal Study on Cognitive Aging and Dementia (KLOSCAD) [29]. The KLOSCAD is an ongoing nationwide, population-based, prospective elderly cohort study on cognitive aging and dementia. In this study, 6,818 community-dwelling elderly Koreans were randomly sampled from 30 villages and towns across South Korea using residential rosters of the individuals aged 60 years or older. A baseline assessment of the study participants was conducted from November 2010 to October 2012, with follow-ups occurring every two years until the period of November 2017 to October 2018.
To examine the effect of normative human aging, we excluded participants at baseline if they (1) were positive on the Cambridge-Hopkins questionnaire for restless legs syndrome (CHRLSq) [30]; (2) scored 20 or more on Alcohol Use Disorder Identification Test-Korean version (AUDIT-K) [31]; (3) were diagnosed with dementia according to the fourth edition of the Diagnostic and Statistical Manual of Mental Disorders, Text Revision (DSM-IV-TR) [32]; (4) scored 16 or more on the Korean version of Geriatric Depression Scale (GDS-K) [33]; (5) scored 3 or more on the psychiatry category of the CIRS [34]; and (6) scored 3 or more on the neurology category of CIRS. In addition, once an individual was diagnosed with dementia during the study period, we terminated their follow-up and excluded them from that time point, as dementia involves progressive and irreversible neurodegeneration that significantly affects sleep habits [35]. This study was approved by the institutional ethics review board of the Seoul National University Bundang Hospital.
Assessment of sleep measures
We used the Korean version of the PSQI [36] to obtain subjective sleep measures regarding its duration, mid-sleep time, latency, efficiency, daytime dysfunction, and quality over the past one month at each assessment. We defined mid-sleep time as the midpoint between self-reported sleep onset and wake-up time where sleep onset is the time after sleep latency has elapsed from bedtime [37]. The mid-sleep time reportedly showed excellent agreement with self-awareness chronotype [37] and superior correlation with dim light melatonin onset, the most reliable circadian phase marker in humans, compared with sleep onset or wake-up time [38]. We defined sleep efficiency as the ratio of the self-reported duration of sleep to the time spent in bed and rated daytime dysfunction and sleep quality on a 4-point Likert-type scale with higher scores indicating worsening of symptoms. The “sleep quality” variable, one of the component scores of the PSQI, used in our study denotes the subjective assessment of the overall sleep in a purely qualitative way and was evaluated by asking “How would you rate your sleep quality overall?” This variable needs to be differentiated from the global PSQI score which reflects both the qualitative and quantitative aspects of sleep [39].
Demographic information and assessment of confounders
Using a study-specific standard interview, trained research nurses collected data on demographic information, physical activity, the amount of alcohol, cigarettes, and coffee consumed over the last one year, and questionnaires including the PSQI, REM Sleep Behavior Disorder Screening Questionnaire (RBDSQ) [40], STOP-Bang [41], CHRLSq, AUDIT-K, GDS-K, and CIRS. We calculated the physical activity over the last one year in terms of total energy expenditure in kilocalories per week, using a formula with relative metabolic rate and metabolic equivalent task as its variables [42]. We quantified the amount of smoking and of alcohol and coffee consumption as packs per day, standard units per week [43], and cups per day, respectively. A score of 5 or more on the RBDSQ indicates a high risk of REM sleep behavior disorder (RBD) [40]. STOP-Bang assesses snoring (S), tiredness during daytime (T), observed apnea (O), high blood pressure (P), body mass index (B), age (A), neck circumference (N), and gender (G), with a score of 5 or more indicating a high risk of obstructive sleep apnea (OSA) [41]. CIRS comprehensively measures the extent and severity of comorbid illnesses on a 5-point scale in regard to the organ-specific categories including cardiovascular, hematopoietic, respiratory, otorhinolaryngologic, gastrointestinal, hepato-renal, genito-urinary, musculoskeletal, neurological, endocrinologic, and psychiatric domains [34].
To assess the cognitive function of study participants, geriatric psychiatrists performed a face-to-face standardized diagnostic test, including physical and neurological examinations, using the Korean version of the Consortium to Establish a Registry for Alzheimer’s Disease Assessment Packet Clinical Assessment Battery (CERAD-K-C) [44] and the Korean version of the Mini International Neuropsychiatric Interview [45]. Trained research neuropsychologists or nurses also performed the CERAD-K Neuropsychological Assessment Battery [44, 46], Digit Span Test [47], and Frontal Assessment Battery [48] on all participants. Results from laboratory tests, such as complete blood cell counts, chemistry panels, apolipoprotein E genotyping, and a serologic test for syphilis, were obtained. A consensus conference attended by four geriatric psychiatrists (KWK, JWH, JHP, and THK) confirmed the final cognitive diagnosis of the participants. Dementia and MCI were diagnosed using the DSM-IV-TR [32] and criteria set by the International Working Group on MCI [49], respectively.
Statistical analysis
We compared baseline characteristics of study participants between men and women, and between those who completed all four waves of assessment and those who did not using Student t-test for continuous variables and χ2 test for categorical variables. To analyze the effects of time and sex on subjective sleep measures, six separate linear mixed-effects models were employed, with sleep duration, mid-sleep time, sleep latency, sleep efficiency, daytime dysfunction, and sleep quality as the dependent variables. The effects of time, sex, and their interaction were considered as fixed effects. Intercepts and slopes of individual participants were permitted to vary as random effects.
These models were adjusted for age, years of education, employment status, socioeconomic status (whether covered by National Medicaid Program), place of residence (urban vs. rural), presence of cohabitants, physical activity, GDS-K score, amount of smoking, and alcohol and coffee consumptions in the past one year, CIRS total score, whether diagnosed with MCI, whether at high risk of OSA or RBD, birth cohort (age < 69 vs. ≥ 69 at baseline), and usage of sleeping pills, as these variables have been reported to be associated with age or sex, and related to sleep measures [11, 50–53]. We assumed the missing data over the follow-up to be missing at random. Due to positively skewed distributions, we loge transformed sleep latency, the degree of daytime dysfunction, physical activity, and the amount of smoking, and alcohol and coffee consumptions thereby enhancing the fit of our models. We did not find any apparent heteroscedasticity from the visual inspection of residual plots.
A post hoc analysis for a sleep measure was conducted with men and women separately. The level of significance was set at α = 0.05. Analyses were performed using R Statistical Software (version 3.5.1; R Foundation for Statistical Computing, Vienna, Austria) and the lme4 [54] package.
Author Contributions
Conception and design: SWS and KWK; Acquisition of the data: All authors; Analysis and interpretation of data: All authors; Drafting of the manuscript and figures: SWS and KWK; Critical revision of the manuscript for important intellectual content: All authors.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This work was supported by a grant from the Korean Health Technology Research and Development Project of the Ministry of Health, Welfare, and Family Affairs, Republic of Korea (grant number A092077).
References
- 1. Mander BA, Winer JR, Walker MP. Sleep and human aging. Neuron. 2017; 94:19–36. https://doi.org/10.1016/j.neuron.2017.02.004 [PubMed]
- 2. Ohayon MM, Carskadon MA, Guilleminault C, Vitiello MV. Meta-analysis of quantitative sleep parameters from childhood to old age in healthy individuals: developing normative sleep values across the human lifespan. Sleep. 2004; 27:1255–73. https://doi.org/10.1093/sleep/27.7.1255 [PubMed]
- 3. Blagrove M, Akehurst L. Effects of sleep loss on confidence-accuracy relationships for reasoning and eyewitness memory. J Exp Psychol Appl. 2000; 6:59–73. https://doi.org/10.1037//1076-898x.6.1.59 [PubMed]
- 4. Krystal AD, Edinger JD. Measuring sleep quality. Sleep Med. 2008 (Suppl 1); 9:S10–17. https://doi.org/10.1016/S1389-9457(08)70011-X [PubMed]
- 5. Argyropoulos SV, Hicks JA, Nash JR, Bell CJ, Rich AS, Nutt DJ, Wilson SJ. Correlation of subjective and objective sleep measurements at different stages of the treatment of depression. Psychiatry Res. 2003; 120:179–90. https://doi.org/10.1016/s0165-1781(03)00187-2 [PubMed]
- 6. Westerberg CE, Lundgren EM, Florczak SM, Mesulam MM, Weintraub S, Zee PC, Paller KA. Sleep influences the severity of memory disruption in amnestic mild cognitive impairment: results from sleep self-assessment and continuous activity monitoring. Alzheimer Dis Assoc Disord. 2010; 24:325–33. https://doi.org/10.1097/WAD.0b013e3181e30846 [PubMed]
- 7. Vitiello MV, Larsen LH, Moe KE. Age-related sleep change: gender and estrogen effects on the subjective-objective sleep quality relationships of healthy, noncomplaining older men and women. J Psychosom Res. 2004; 56:503–10. https://doi.org/10.1016/S0022-3999(04)00023-6 [PubMed]
- 8. Luca G, Haba Rubio J, Andries D, Tobback N, Vollenweider P, Waeber G, Marques Vidal P, Preisig M, Heinzer R, Tafti M. Age and gender variations of sleep in subjects without sleep disorders. Ann Med. 2015; 47:482–91. https://doi.org/10.3109/07853890.2015.1074271 [PubMed]
- 9. Ayres RU, Warr B. Accounting for growth: the role of physical work. Struct Change Econ Dyn. 2005; 16:181–209. https://doi.org/10.1016/j.strueco.2003.10.003
- 10. Ohayon MM. Interactions between sleep normative data and sociocultural characteristics in the elderly. J Psychosom Res. 2004; 56:479–86. https://doi.org/10.1016/j.psychores.2004.04.365 [PubMed]
- 11. Keyes KM, Maslowsky J, Hamilton A, Schulenberg J. The great sleep recession: changes in sleep duration among US adolescents, 1991-2012. Pediatrics. 2015; 135:460–68. https://doi.org/10.1542/peds.2014-2707 [PubMed]
- 12. Kalak N, Lemola S, Brand S, Holsboer-Trachsler E, Grob A. Sleep duration and subjective psychological well-being in adolescence: a longitudinal study in Switzerland and Norway. Neuropsychiatr Dis Treat. 2014; 10:1199–207. https://doi.org/10.2147/NDT.S62533 [PubMed]
- 13. Lytle LA, Murray DM, Laska MN, Pasch KE, Anderson SE, Farbakhsh K. Examining the longitudinal relationship between change in sleep and obesity risk in adolescents. Health Educ Behav. 2013; 40:362–70. https://doi.org/10.1177/1090198112451446 [PubMed]
- 14. Åkerstedt T, Discacciati A, Miley-Åkerstedt A, Westerlund H. Aging and the change in fatigue and sleep - a longitudinal study across 8 years in three age groups. Front Psychol. 2018; 9:234. https://doi.org/10.3389/fpsyg.2018.00234 [PubMed]
- 15. Didikoglu A, Maharani A, Tampubolon G, Canal MM, Payton A, Pendleton N. Longitudinal sleep efficiency in the elderly and its association with health. J Sleep Res. 2020; 29:e12898. https://doi.org/10.1111/jsr.12898 [PubMed]
- 16. Hita-Yañez E, Atienza M, Cantero JL. Polysomnographic and subjective sleep markers of mild cognitive impairment. Sleep. 2013; 36:1327–34. https://doi.org/10.5665/sleep.2956 [PubMed]
- 17. Duffy JF, Zitting KM, Chinoy ED. Aging and circadian rhythms. Sleep Med Clin. 2015; 10:423–34. https://doi.org/10.1016/j.jsmc.2015.08.002 [PubMed]
- 18. Ohayon MM, Vecchierini MF. Normative sleep data, cognitive function and daily living activities in older adults in the community. Sleep. 2005; 28:981–89. [PubMed]
- 19. Brown SA, Schmitt K, Eckert A. Aging and circadian disruption: causes and effects. Aging (Albany NY). 2011; 3:813–17. https://doi.org/10.18632/aging.100366 [PubMed]
- 20. Doi Y, Minowa M, Uchiyama M, Okawa M. Subjective sleep quality and sleep problems in the general Japanese adult population. Psychiatry Clin Neurosci. 2001; 55:213–15. https://doi.org/10.1046/j.1440-1819.2001.00830.x [PubMed]
- 21. Zhang B, Wing YK. Sex differences in insomnia: a meta-analysis. Sleep. 2006; 29:85–93. https://doi.org/10.1093/sleep/29.1.85 [PubMed]
- 22. Bremner WJ, Vitiello MV, Prinz PN. Loss of circadian rhythmicity in blood testosterone levels with aging in normal men. J Clin Endocrinol Metab. 1983; 56:1278–81. https://doi.org/10.1210/jcem-56-6-1278 [PubMed]
- 23. Pandi-Perumal S, Monti JM, Monjan AA. Principles and practice of geriatric sleep medicine: Cambridge University Press. 2009. https://doi.org/10.1017/CBO9780511770661
- 24. Lin CM, Davidson TM, Ancoli-Israel S. Gender differences in obstructive sleep apnea and treatment implications. Sleep Med Rev. 2008; 12:481–96. https://doi.org/10.1016/j.smrv.2007.11.003 [PubMed]
- 25. Barsky AJ, Peekna HM, Borus JF. Somatic symptom reporting in women and men. J Gen Intern Med. 2001; 16:266–75. [PubMed]
- 26. Gooneratne NS, Bellamy SL, Pack F, Staley B, Schutte-Rodin S, Dinges DF, Pack AI. Case-control study of subjective and objective differences in sleep patterns in older adults with insomnia symptoms. J Sleep Res. 2011; 20:434–44. https://doi.org/10.1111/j.1365-2869.2010.00889.x [PubMed]
- 27. Santisteban JA, Brown TG, Gruber R. Association between the munich chronotype questionnaire and wrist actigraphy. Sleep Disord. 2018; 2018:5646848. https://doi.org/10.1155/2018/5646848 [PubMed]
- 28. Mowbray RM, Rodger TF. Psychology in relation to medicine: Churchill Livingstone. 1970.
- 29. Han JW, Kim TH, Kwak KP, Kim K, Kim BJ, Kim SG, Kim JL, Kim TH, Moon SW, Park JY, Park JH, Byun S, Suh SW, et al. Overview of the Korean longitudinal study on cognitive aging and dementia. Psychiatry Investig. 2018; 15:767–74. https://doi.org/10.30773/pi.2018.06.02 [PubMed]
- 30. Allen RP, Burchell BJ, MacDonald B, Hening WA, Earley CJ. Validation of the self-completed Cambridge-Hopkins questionnaire (CH-RLSq) for ascertainment of restless legs syndrome (RLS) in a population survey. Sleep Med. 2009; 10:1097–100. https://doi.org/10.1016/j.sleep.2008.10.007 [PubMed]
- 31. Kim KW, Choi EA, Lee SB, Park JH, Lee JJ, Huh Y, Youn JC, Jhoo JH, Choo IH, Kim MH, Lee DY, Woo JI. Prevalence and neuropsychiatric comorbidities of alcohol use disorders in an elderly Korean population. Int J Geriatr Psychiatry. 2009; 24:1420–28. https://doi.org/10.1002/gps.2280 [PubMed]
- 32. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. edition, Text Revision. American Psychiatric Pub. 2000.
- 33. Kim JY, Park JH, Lee JJ, Huh Y, Lee SB, Han SK, Choi SW, Lee DY, Kim KW, Woo JI. Standardization of the Korean version of the geriatric depression scale: reliability, validity, and factor structure. Psychiatry Investig. 2008; 5:232–38. https://doi.org/10.4306/pi.2008.5.4.232 [PubMed]
- 34. Miller MD, Paradis CF, Houck PR, Mazumdar S, Stack JA, Rifai AH, Mulsant B, Reynolds CF
3rd . Rating chronic medical illness burden in geropsychiatric practice and research: application of the cumulative illness rating scale. Psychiatry Res. 1992; 41:237–48. https://doi.org/10.1016/0165-1781(92)90005-n [PubMed] - 35. Boeve BF, Silber MH, Ferman TJ. Current management of sleep disturbances in dementia. Curr Neurol Neurosci Rep. 2002; 2:169–77. https://doi.org/10.1007/s11910-002-0027-0 [PubMed]
- 36. Sohn SI, Kim DH, Lee MY, Cho YW. The reliability and validity of the Korean version of the Pittsburgh sleep quality index. Sleep Breath. 2012; 16:803–12. https://doi.org/10.1007/s11325-011-0579-9 [PubMed]
- 37. Roenneberg T, Wirz-Justice A, Merrow M. Life between clocks: daily temporal patterns of human chronotypes. J Biol Rhythms. 2003; 18:80–90. https://doi.org/10.1177/0748730402239679 [PubMed]
- 38. Terman JS, Terman M, Lo ES, Cooper TB. Circadian time of morning light administration and therapeutic response in winter depression. Arch Gen Psychiatry. 2001; 58:69–75. https://doi.org/10.1001/archpsyc.58.1.69 [PubMed]
- 39. Buysse DJ, Reynolds CF
3rd , Monk TH, Berman SR, Kupfer DJ. Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989; 28:193–213. https://doi.org/10.1016/0165-1781(89)90047-4 [PubMed] - 40. Stiasny-Kolster K, Mayer G, Schäfer S, Möller JC, Heinzel-Gutenbrunner M, Oertel WH. The REM sleep behavior disorder screening questionnaire—a new diagnostic instrument. Mov Disord. 2007; 22:2386–93. https://doi.org/10.1002/mds.21740 [PubMed]
- 41. Chung F, Elsaid H. Screening for obstructive sleep apnea before surgery: why is it important? Curr Opin Anaesthesiol. 2009; 22:405–11. https://doi.org/10.1097/ACO.0b013e32832a96e2 [PubMed]
- 42. Nishihara T, Katsuki F, Hori M, Kagawa C, Okuda S, Utsu T, Yoneda I, Nasu M, Yoneda K. Estimation of energy expenditure and daily activity index on 185 subjects by a new personal computer system. Jpn J Nutr Dieta. 1988; 46:73–84. https://doi.org/10.5264/eiyogakuzashi.46.73
- 43. World Health Organization. Global status report on alcohol and health, 2014: World Health Organization. 2014.
- 44. Lee JH, Lee KU, Lee DY, Kim KW, Jhoo JH, Kim JH, Lee KH, Kim SY, Han SH, Woo JI. Development of the Korean version of the consortium to establish a registry for Alzheimer’s disease assessment packet (CERAD-K): clinical and neuropsychological assessment batteries. J Gerontol B Psychol Sci Soc Sci. 2002; 57:P47–53. https://doi.org/10.1093/geronb/57.1.p47 [PubMed]
- 45. Yoo SW, Kim YS, Noh JS, Oh KS, Kim CH, NamKoong K, Chae JH, Lee GC, Jeon SI, Min KJ. Validity of Korean version of the mini-international neuropsychiatric interview. Anxiety Mood. 2006; 2:50–55.
- 46. Lee DY, Lee KU, Lee JH, Kim KW, Jhoo JH, Kim SY, Yoon JC, Woo SI, Ha J, Woo JI. A normative study of the CERAD neuropsychological assessment battery in the Korean elderly. J Int Neuropsychol Soc. 2004; 10:72–81. https://doi.org/10.1017/S1355617704101094 [PubMed]
- 47. Wechsler D. Instruction manual for the Wechsler Memory Scale - Revised. Psychological Corporation. 1987.
- 48. Kim TH, Huh Y, Choe JY, Jeong JW, Park JH, Lee SB, Lee JJ, Jhoo JH, Lee DY, Woo JI, Kim KW. Korean version of frontal assessment battery: psychometric properties and normative data. Dement Geriatr Cogn Disord. 2010; 29:363–70. https://doi.org/10.1159/000297523 [PubMed]
- 49. Winblad B, Palmer K, Kivipelto M, Jelic V, Fratiglioni L, Wahlund LO, Nordberg A, Bäckman L, Albert M, Almkvist O, Arai H, Basun H, Blennow K, et al. Mild cognitive impairment—beyond controversies, towards a consensus: report of the international working group on mild cognitive impairment. J Intern Med. 2004; 256:240–46. https://doi.org/10.1111/j.1365-2796.2004.01380.x [PubMed]
- 50. Kabir ZN, Tishelman C, Agüero-Torres H, Chowdhury AM, Winblad B, Höjer B. Gender and rural-urban differences in reported health status by older people in Bangladesh. Arch Gerontol Geriatr. 2003; 37:77–91. https://doi.org/10.1016/s0167-4943(03)00019-0 [PubMed]
- 51. Hays JC, Blazer DG, Foley DJ. Risk of napping: excessive daytime sleepiness and mortality in an older community population. J Am Geriatr Soc. 1996; 44:693–98. https://doi.org/10.1111/j.1532-5415.1996.tb01834.x [PubMed]
- 52. Ursin R, Bjorvatn B, Holsten F. Sleep duration, subjective sleep need, and sleep habits of 40- to 45-year-olds in the hordaland health study. Sleep. 2005; 28:1260–69. https://doi.org/10.1093/sleep/28.10.1260 [PubMed]
- 53. Sherrill DL, Kotchou K, Quan SF. Association of physical activity and human sleep disorders. Arch Intern Med. 1998; 158:1894–98. https://doi.org/10.1001/archinte.158.17.1894 [PubMed]
- 54. Bates D, Mächler M, Bolker B, Walker S. Fitting linear mixed-effects models using lme4. J Stat Softw. 2015; 67:1–48. https://doi.org/10.18637/jss.v067.i01


