Introduction
Testicular cancer (TC) is a rare malignant tumor in the genitourinary system, accounting for about 5% of genitourinary tumors [1]. In 2018, 71,105 new cases of TC (1.7% of male incidence) and 9,507 deaths (0.2% of male mortality) were diagnosed worldwide [2]. Despite the lower overall incidence, TC is the most common malignant tumor in men aged 15-34 years [3].
Germ cell TC (GCTC) is the most common type of TC. GCTC mostly occurs on one side and only about 1% on both sides [4]. The main risk factor for GCTC is cryptorchidism, which occurs in 2-5% of boys born at term [5]. The other risk factors include gonadal dysgenesis and genetic diseases such as Down’s syndrome [6, 7]. Although the overall 10-year cancer-specific mortality (CSM)-free survival rate in patients with GCTC is approaching 95%, the incidence has increased significantly over the past 30 years [8, 9]. Therefore, further research is important to determine the predictors that may affect the long-term survival of GCTC patients.
The American Joint Committee on Cancer (AJCC) tumor node metastasis (TNM) staging system is widely used to evaluate the prognosis of patients with GCTC. However, some other factors such as age, race, SEER stage, surgery and radiotherapy can also affect the outcome of GCTC patients. The nomogram based on the equations derived from the regression coefficients of each variable integrates many prognostic factors, which can more accurately predict the individual survival [10]. The nomogram can incorporate important clinicopathological and demographic variables in clinical practice to create a more comprehensive prognostic evaluation system.
In this study, we analyzed the clinicopathological features and prognostic factors of GCTC patients using the Surveillance, Epidemiology, and End Results (SEER) database. Based on the results of survival analysis, we further developed and validated the prognostic nomogram for patients with GCTC to better predict the patient's prognosis.
Results
Demographic and clinicopathologic characteristics
From 2004 to 2015, our study cohort included 22,165 eligible GCTC patients, including 15,515 patients in the training cohort and 6,650 patients in the validation cohort. Table 1 shows the demographic and clinical characteristics of patients with GCTC. In the entire cohort, the majority of GCTC patients were white (90.3%), and the age of onset was concentrated between 21-40 years (65.8%). The most common types were AJCC I stage (77.2%), T1 stage (66.9%), N0 stage (80.3%), M0 stage (90.8%) and localized stage (72.2%). In addition, nearly 99.9% of patients received surgery while only 20.2% received radiotherapy.
Table 1. Baseline demographic and clinical characteristics with testicular germ cell tumor (TGCT) patients in our study.
| Characteristic | Total No. (%) | The training cohort | The validation cohort | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No. (%) | No. (%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total | 22165 | 15515 (70.0) | 6650 (30.0) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age at diagnosis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0-20 | 1809 (8.2) | 1238 (8.0) | 571 (8.6) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 21-40 | 14569 (65.7) | 10211 (65.8) | 4358 (65.5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 41-60 | 5334 (24.1) | 3739 (24.1) | 1595 (24.0) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| > 60 | 453 (2.0) | 327 (2.1) | 126 (1.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Race | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| White | 20026 (90.3) | 14029 (90.4) | 5997 (90.2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Black | 586 (2.6) | 402 (2.6) | 184 (2.8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Others | 1553 (7.0) | 1084 (7.0) | 469 (7.1) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AJCC stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I | 17121 (77.2) | 11997 (77.3) | 5124 (77.1) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| II | 2519 (11.4) | 1753 (11.3) | 766 (11.5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| III | 2525 (11.4) | 1765 (11.4) | 760 (11.4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T1 | 14829 (66.9) | 10367 (66.8) | 4462 (67.1) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T2 | 6132 (27.7) | 4302 (27.7) | 1830 (27.5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T3 | 1071 (4.8) | 761 (4.9) | 310 (4.7) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T4 | 133 (0.6) | 85 (0.5) | 48 (0.7) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N0 | 17804 (80.3) | 12457 (80.3) | 5347 (80.4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N1 | 2145 (9.7) | 1482 (9.6) | 663 (10.0) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N2 | 1250 (5.6) | 898 (5.8) | 352 (5.3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N3 | 966 (4.4) | 678 (4.4) | 288 (4.3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M0 | 20121 (90.8) | 14087 (90.8) | 6034 (90.7) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M1 | 2044 (9.2) | 1428 (9.2) | 616 (9.3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SEER stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Localized | 16001 (72.2) | 11193 (72.1) | 4808 (72.3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Regional | 4085 (18.4) | 2881 (18.6) | 1204 (18.1) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Distant | 2079 (9.4) | 1441 (9.3) | 638 (9.6) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Surgery | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No | 19 (0.1) | 13 (0.1) | 6 (0.1) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 22146 (99.9) | 15502 (99.9) | 6644 (99.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiotherapy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 4472 (20.2) | 3155 (20.3) | 1317 (19.8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No | 17693 (79.8) | 12360 (79.7) | 5333 (80.2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: AJCC, American Joint Committee on Cancer; SEER, Surveillance, Epidemiology, and End Results. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Percentages may not total 100 because of rounding. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Survival of patients with GCTC
By analyzing the Kaplan-Meier curve with a log-rank test we found that age at diagnosis, race, AJCC stage, T stage, N stage, M stage, SEER stage, surgery and radiotherapy (All p<0.05) were associated with OS and CSS of GCTC patients (Table 2). In the whole cohort, the 3 -, 5- and 10-year OS of GCTC patients were 96.4%, 95.6% and 93.6%, respectively, and the 3 -, 5- and 10-year CSS were 97.7%, 97.4% and 97.2%, respectively. We found that patients aged 21-40 and received chemotherapy had a higher survival rate.
Table 2. Kaplan–Meier analysis overall survival (OS) and cancer-specific survival (CSS) for testicular germ cell tumor (TGCT) patients.
| Characteristic | Overall Survival (OS) | Cancer-Specific Survival (CSS) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3-year OS % | 5-year OS % | 10-year OS % | Kaplan-Meier | 3-year CSS % | 5-year CSS % | 10-year OS % | Kaplan-Meier | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Log Rank χ2 test | P value | Log Rank χ2 test | P value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All | 96.4 | 95.6 | 93.6 | 97.7 | 97.4 | 97.2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age at diagnosis | 260.021 | <.001 | 29.805 | <.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0-20 | 95.9 | 95.6 | 94.2 | 97.1 | 96.7 | 96.5 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 21-40 | 97.0 | 96.2 | 95.0 | 97.9 | 97.6 | 97.4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 41-60 | 95.7 | 94.9 | 91.7 | 97.6 | 97.4 | 97.0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| > 60 | 89.3 | 83.3 | 67.4 | 95.1 | 93.5 | 93.0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Race | 17.099 | <.001 | 9.473 | .009 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| White | 96.5 | 95.7 | 93.7 | 97.8 | 97.5 | 97.2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Black | 93.1 | 92.6 | 89.4 | 96.4 | 95.9 | 95.1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Others | 96.6 | 95.6 | 94.5 | 97.6 | 97.1 | 96.9 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AJCC stage | 1594.930 | <.001 | 1865.778 | <.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I | 98.6 | 97.9 | 96.1 | 99.4 | 99.2 | 99.1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| II | 97.4 | 96.4 | 94.9 | 98.6 | 98.3 | 98.1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| III | 81.3 | 79.9 | 76.3 | 85.6 | 84.8 | 83.8 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T stage | 410.212 | <.001 | 427.797 | <.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T1 | 97.5 | 96.6 | 94.8 | 98.4 | 98.2 | 97.9 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T2 | 96.2 | 95.7 | 93.2 | 97.5 | 97.3 | 97.1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T3 | 89.5 | 87.2 | 85.3 | 93.0 | 91.9 | 91.5 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T4 | 71.0 | 68.7 | 68.7 | 76.8 | 75.5 | 75.5 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N stage | 455.331 | <.001 | 505.318 | <.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N0 | 97.8 | 97.0 | 95.2 | 98.8 | 98.5 | 98.4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N1 | 90.7 | 89.4 | 87.2 | 93.5 | 92.7 | 92.2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N2 | 94.8 | 93.7 | 90.6 | 95.6 | 95.4 | 94.9 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N3 | 87.2 | 86.2 | 82.7 | 91.0 | 90.4 | 88.6 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M stage | 1866.861 | <.001 | 2192.811 | <.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M0 | 98.4 | 97.6 | 95.8 | 99.2 | 99.0 | 98.8 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M1 | 78.4 | 76.8 | 73.0 | 83.1 | 82.0 | 81.1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SEER stage | 1874.705 | <.001 | 2171.302 | <.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Localized | 98.8 | 98.1 | 96.3 | 99.5 | 99.3 | 99.2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Regional | 96.8 | 95.9 | 94.0 | 98.2 | 97.8 | 97.5 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Distant | 78.8 | 77.0 | 73.3 | 83.4 | 82.3 | 81.4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Surgery | 166.335 | <.001 | 155.316 | <.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No | 62.1 | 60.8 | 58.1 | 74.2 | 74.2 | 74.2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 96.6 | 95.8 | 93.8 | 97.8 | 97.5 | 97.3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiotherapy | 26.456 | <.001 | 19.854 | <.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 97.5 | 96.8 | 95.4 | 98.4 | 98.3 | 98.1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No | 96.1 | 95.2 | 93.0 | 97.5 | 97.2 | 96.9 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Note: P-value<0.05 are shown in bold. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: AJCC, American Joint Committee on Cancer; SEER, Surveillance, Epidemiology, and End Results. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Identification of prognostic factors of OS and CSS in GCTC patients
Univariate and multivariate Cox regression were used to analyze the related factors of OS and CSS in patients with GCTC. In the training cohort, univariate Cox regression analysis showed that age at diagnosis, race, AJCC stage, T stage, N stage, M stage, SEER stage, surgery and radiotherapy were related factors of OS and CSS in GCTC patients. After all the above factors were included in the multivariate Cox regression analysis, we found that T stage, N stage, M stage and radiotherapy were not independent risk factors for OS, while N stage and surgery were not independent risk factors for CSS (Table 3).
Table 3. Univariate and multivariate analysis of overall survival (OS) and cancer-specific survival (CSS) rates in training cohort.
| Characteristic | OS | CSS | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Univariate analysis | Multivariate analysisa | Univariate analysis | Multivariate analysisb | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Ratio (95% CI) | P value | Hazard Ratio (95% CI) | P value | Hazard Ratio (95% CI) | P value | Hazard Ratio (95% CI) | P value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age at diagnosis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0-20 | Reference | Reference | Reference | Reference | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 21-40 | 0.94 (0.70-1.27) | .677 | 1.25 (0.92-1.69) | .151 | 0.80 (0.55-1.17) | .246 | 1.19 (0.82-1.75) | .361 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 41-60 | 1.52 (1.11-2.08) | .008 | 2.27 (1.66-3.11) | <.001 | 1.11 (0.74-1.65) | .627 | 1.89 (1.26-2.83) | .002 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| > 60 | 5.11 (3.48-7.49) | <.001 | 7.60 (5.17-11.16) | <.001 | 2.34 (1.31-4.18) | .004 | 4.07 (2.27-7.29) | <.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Race | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| White | Reference | Reference | Reference | Reference | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Black | 1.64 (1.12-2.39) | .011 | 1.49 (1.02-2.18) | .040 | 1.26 (0.69-2.31) | .445 | 1.16 (0.64-2.12) | .626 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Others | 1.11 (0.83-1.48) | .481 | 1.26 (0.94-1.68) | .121 | 1.46 (1.02-2.08) | .036 | 1.71 (1.19-2.44) | .003 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AJCC stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I | Reference | Reference | Reference | Reference | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| II | 1.50 (1.15-1.96) | .003 | 0.90 (0.60-1.35) | .606 | 2.38 (1.57-3.60) | <.001 | 0.96 (0.52-1.75) | .882 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| III | 8.21 (7.03-9.58) | <.001 | 1.81 (1.14-2.89) | .012 | 20.54 (16.07-26.25) | <.001 | 2.22 (1.08-4.55) | .029 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T1 | Reference | Reference | Reference | Reference | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T2 | 1.32 (1.12-1.57) | .001 | - | .918 | 1.41 (1.10-1.79) | .006 | 0.97 (0.76-1.24) | .811 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T3 | 3.46 (2.75-4.34) | <.001 | - | .365 | 4.45 (3.29-6.00) | <.001 | 1.03 (0.75-1.40) | .879 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T4 | 10.11 (6.75-15.16) | <.001 | - | .016 | 16.23 (10.11-26.04) | <.001 | 2.33 (1.43-3.80) | .001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N0 | Reference | Reference | Reference | Reference | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N1 | 3.16 (2.62-3.80) | <.001 | - | .407 | 5.06 (3.93-6.52) | <.001 | - | .831 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N2 | 2.00 (1.52-2.64) | <.001 | - | .034 | 3.41 (2.40-4.83) | <.001 | - | .204 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N3 | 4.13 (3.26-5.22) | <.001 | - | .721 | 7.45 (5.56-9.98) | <.001 | - | .442 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M0 | Reference | Reference | Reference | Reference | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M1 | 8.95 (7.71-10.38) | <.001 | - | .109 | 19.13 (15.43-23.72) | <.001 | 2.14 (0.90-5.08) | .084 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SEER stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Localized | Reference | Reference | Reference | Reference | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Regional | 1.81 (1.46-2.24) | <.001 | 1.81 (1.28-2.55) | .001 | 3.40 (2.41-4.79) | <.001 | 3.10 (1.80-5.34) | <.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Distant | 10.38 (8.82-12.23) | <.001 | 6.19 (3.80-10.08) | <.001 | 28.26 (21.61-36.94) | <.001 | 6.86 (2.69-17.49) | <.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Surgery | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No | Reference | Reference | Reference | Reference | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 0.06 (0.03-0.12) | <.001 | 0.28 (0.13-0.61) | .001 | 0.05 (0.02-0.14) | <.001 | - | .829 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiotherapy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | Reference | Reference | Reference | Reference | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No | 1.54 (1.27-1.87) | <.001 | - | .930 | 1.53 (1.15-2.03) | .003 | 0.67 (0.50-0.91) | .010 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: OS, Overall survival; CSS, Cancer-specific survival; AJCC, American Joint Committee on Cancer; SEER, Surveillance, Epidemiology, and End Results. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| aModel was adjusted by age at diagnosis, race, AJCC stage, TNM stage, SEER stage, surgery and radiotherapy. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| bModel was adjusted by age at diagnosis, race, AJCC stage, TNM stage, SEER stage, surgery and radiotherapy. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Prognostic nomograms for OS and CSS
In the training cohort, we developed and established two nomograms for OS and CSS: one nomogram of independent risk factors associated with prognosis based on the multivariate Cox regression analysis, and the other one nomogram based on TNM stage. Figure 1 and Supplementary Figure 1 (TNM stage) show the nomogram of the prognosis of 3-, 5- and 10-year OS and CSS. Each subtype of the variables on the nomogram corresponds to a point on the "Point" scale. By adding the scores associated with each variable and projecting the "Total point" to the lowest number, the probabilities of OS and CSS for 3-, 5-, and 10- years can be estimated.
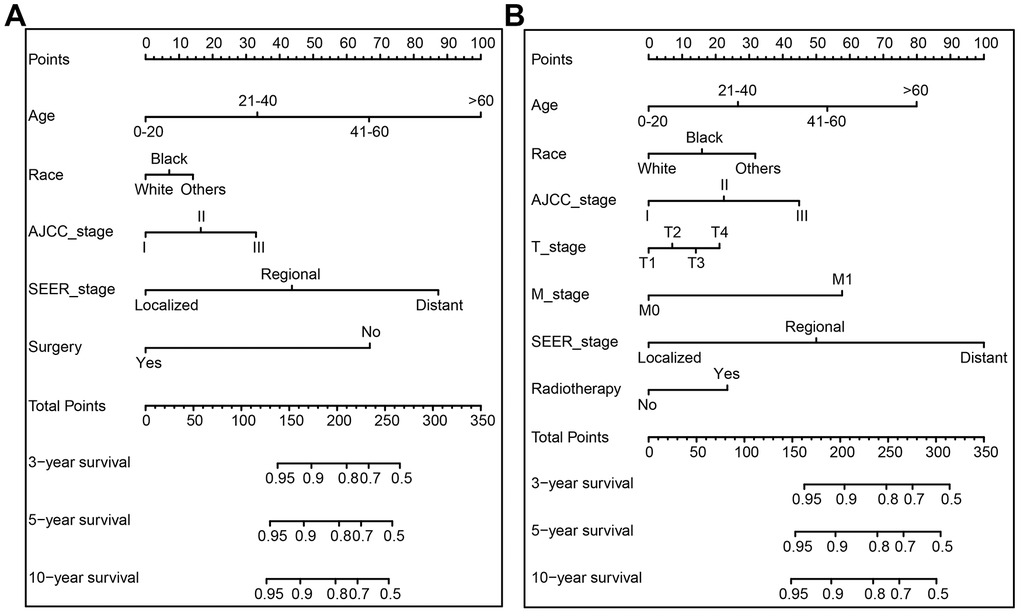
Figure 1. The nomogram predicting 3-, 5-, and 10-year overall survival (OS) and cancer-specific survival (CSS) rate of GCTC patients the training cohort. (A) OS nomogram; (B) CSS nomogram.
The length of the line corresponding to each variable in the nomogram represents the influence of the predictive variable on the survival outcome. We found that for nomogram generated by multivariate Cox regression analysis, age contributed the least to survival outcome in the OS nomogram, and SEER stage has the greatest contribution to the survival outcome in the nomogram of CSS, followed by age (Figure 1). Regardless of the OS or CSS nomogram generated by T stage, N stage and M stage, M stage made the greatest contribution to survival outcome.
Validation and calibration of the nomograms
Analysis of the time-dependent ROC curves for OS shows that the AUC for the ROC curve of the nomogram (training cohort: AUC=0.763; validation cohort: AUC=0.765) was significantly larger than that of TNM stage (training cohort: AUC=0.717; validation cohort: AUC=0.734), but the ROC curve of CSS was similar (Figure 2). Moreover, we evaluated the predictive performance of the nomogram for 3-, 5- and 10-year OS and CSS in the training and validation cohorts and found that the nomograms provided a good assessment of OS and CSS at 3-, 5- and 10-year in GCTC patients (Figure 3 and Supplementary Figure 2).
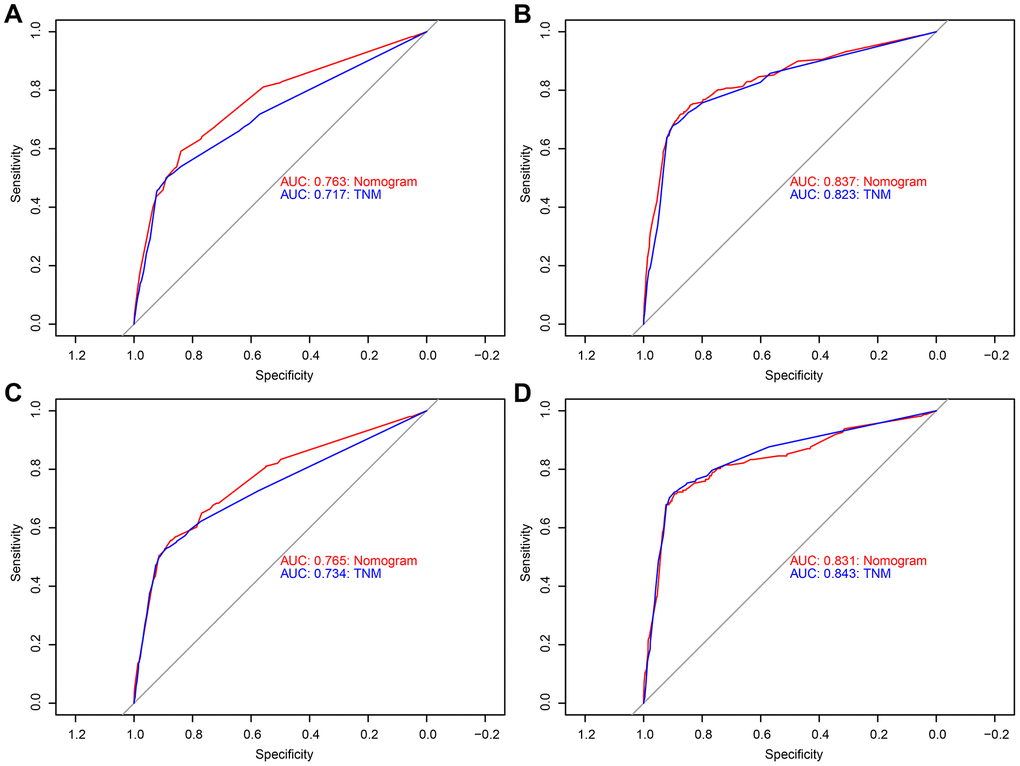
Figure 2. Receiver operating characteristic (ROC) curves detects the predictive value of two nomograms in GCTC prognosis. (A) Overall survival (OS) the training cohort. (B) Cancer-specific survival (CSS) the training cohort. (C) OS the validation cohort. (D) CSS the validation cohort.
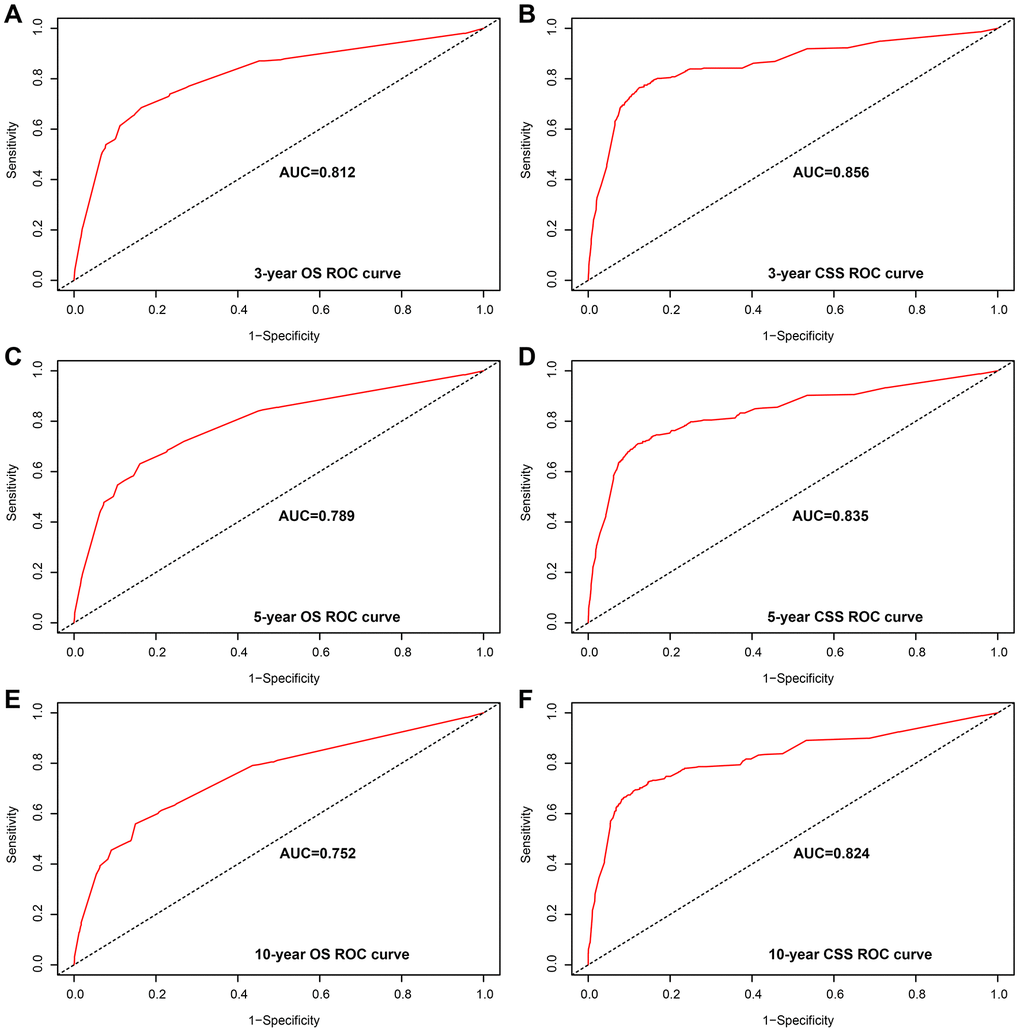
Figure 3. Area under the curve (AUC) value of the receiver operating characteristic (ROC) predicting in the training cohort. (A) 3-year overall survival (OS) rates. (B) 3-year cancer-specific survival (CSS) rates. (C) 5-year OS rates. (D) 5-year CSS rates. (E) 10-year OS rates. (F) 10-year CSS rates.
In order to compare the predicted survival time with the actual survival time, the C-index and calibration curves were used to verify the nomogram in the training and validation cohorts. We found that the C-index of the nomogram OS was larger than that of the TNM stage nomogram (training cohort: nomogram=0.784, 95% CI: 0.765-0.803; TNM stage=0.746, 95% CI: 0.725-0.767, p<0.001; validation cohort: nomogram=0.785, 95% CI: 0.756-0.814; TNM stage=0.765, 95% CI: 0.735-0.795; p=0.003), while the C-index of CSS was basically same in both training and validation cohorts (training cohort: nomogram=0.844, 95% CI: 0.820-0.869; TNM stage=0.833, 95% CI: 0.809-0.857, p=0.053; validation cohort: nomogram=0.841, 95% CI: 0.804-0.878; TNM stage=0.854, 95% CI: 0.822-0.886; p=0.186). Moreover, we calibrated the 3-, 5- and 10-year OS and CSS nomogram of the training cohort and the validation cohort. The calibration curves in Figure 4 and Supplementary Figure 3 were very close to the perfect curves, showing a good consistency between the prediction of the nomogram and the actual observation in the training and validation cohort.
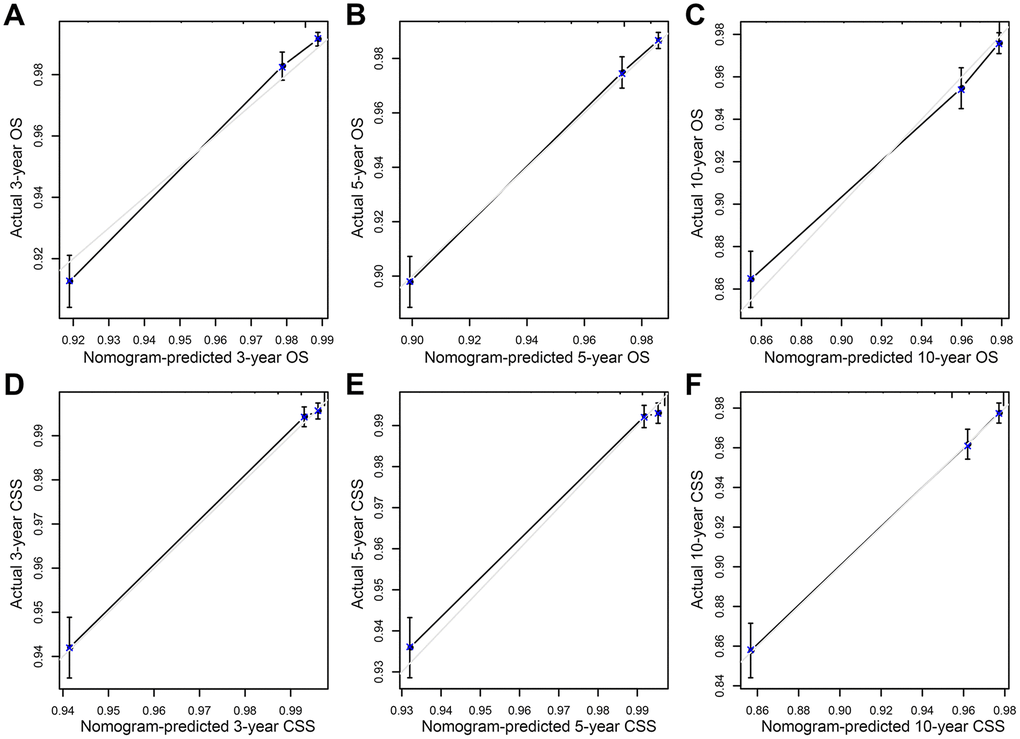
Figure 4. Calibration plot of the nomogram for predicting 3-, 5-, and 10-year overall survival (OS) and cancer-specific survival (CSS) in training cohort. (A) 3-year OS; (B) 5-year OS; (C) 10-year OS; (D) 3-year CSS; (E) 5-year CSS; (F) 10-year CSS.
In addition, DCA calculated the net benefit to evaluate the clinical utility of the nomograms. The results showed that in a wide range of OS thresholds the clinical net benefit of the nomograms generated by multivariate Cox regression analysis was greater than the nomograms produced by TNM stage (Figure 5) in both training and validation cohorts. Both in training cohort and validation cohort, the CIC results show that the multivariate Cox regression analysis produced nomograms that were classified as positive among the broad thresholds for OS, and the number of true positives was greater than the TNM stage nomograms (Figure 6 and Supplementary Figure 4).
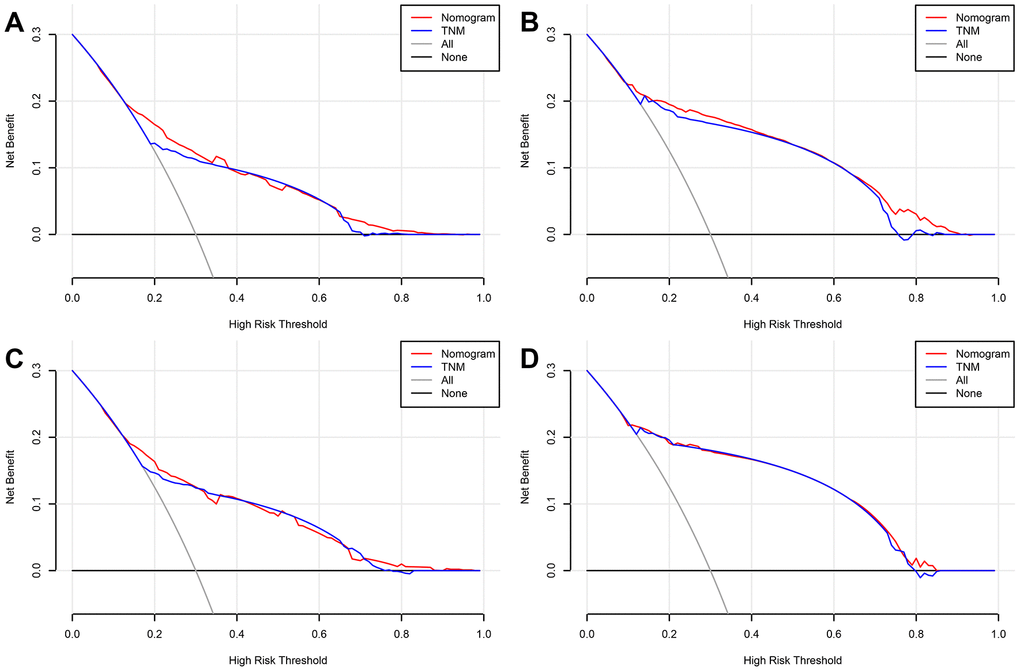
Figure 5. Decision curve analysis (DCA) curves detects the predictive value of two nomograms in GCTC prognosis. (A) Overall survival (OS) in the training cohort. (B) Cancer-specific survival (CSS) in the training cohort. (C) OS in the validation cohort. (D) CSS in the validation cohort.
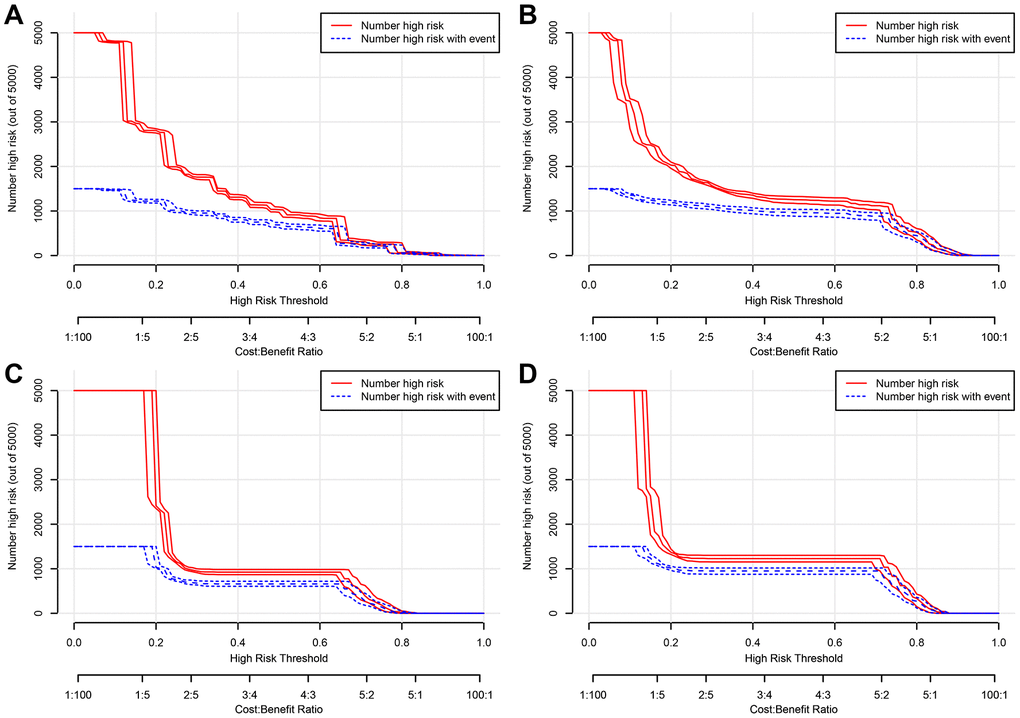
Figure 6. Clinical impact curve (CIC) detects the predictive value of two nomograms in GCTC prognosis in the training cohort. (A) The overall survival (OS) of the nomogram. (B) The OS of the TNM stage. (C) The cancer-specific survival (CSS) of the nomogram. (D) The CSS of the TNM stage.
Discussion
In this study, we first established prognostic nomograms of OS and CSS in patients with GCTC. We performed Cox regression analysis on a large number of GCTC patients using the SEER database and found that age at diagnosis, race, AJCC stage and SEER stage were independent risk factors for OS and CSS. We constructed two prognostic nomograms: one based on multivariate Cox regression analysis and the other one TNM stage. By examining the C-index, ROC curve, DCA curve and CIC we found that the nomogram based on multivariate Cox regression analysis has better OS prognosis than the TNM stage nomogram and has similar prognostic ability in CSS. In addition, we have verified and calibrated the established nomograms and have evaluated the accuracy of the OS and CSS alignment charts for 3-, 5- and 10-year. The results show that there was a good consistency between the prediction of the nomograms and the actual observation and also good reliability in both internal and external verification.
TNM staging classification system was the most versatile tumor staging system in the world and also the foundation of GCTC prognosis [11]. The TNM stage was determined based on the results of laboratory tests and postoperative pathological examinations [12]. In this classification system, clinicians determine TNM stage based on the depth of tumor invasion (T), number of lymph node metastasis (N) and distant metastasis (M). For cancer patients with different TNM stage high stage means complex drug treatment and short survival time. How to better combine the patients’ tumor characteristics and their own clinical factors to make a tailored assessment of the risk of patients has been a challenge for clinicians [13].
The nomogram was a predictive tool, which was a graphical representation based on multivariate prognostic regression analysis, making the prognostic factors more visual [14, 15]. The model integrates variety of prognostic factors and is well prepared to evaluate the survival probability of individual patients [16]. At present, many cancer nomograms have been developed and shown more accurate predictions of the cancer prognosis than traditional TNM systems [17, 18]. In addition, nomograms allow clinicians to incorporate more prognostic factors and assess the patient's physical condition more intuitively in order to evaluate the personalized prediction for clinical trial participation. Therefore, it was of great significance to establish an effective and reliable nomogram for the prognosis of patients with GCTC and to provide them with individualized treatment.
The nomogram has been widely used in various urinary malignancies, which was of great significance for individualized and accurate prediction of prognosis [19–21]. Karakiewicz et al. [22] performed preoperative prediction of 726 patients treated with radical cystectomy and bilateral pelvic lymphadenectomy, and found that the multivariate nomogram was more accurate than the TUR T stage alone prediction. Similarly, kattan et al. [23] constructed a nomogram that included pre-treatment serum prostate-specific antigen levels, biopsy Gleason scores and clinical stages, and found that it could predict the 5-year treatment failure probability with clinically localized prostate cancer who underwent radical prostatectomy. Zhou et al. [24] found that nomogram and Aggtrmmns scoring system can effectively predict kidney cancer patient's OS and CSS. In our study, we developed a nomogram based on age, race, AJCC stage, TNM stage, SEER stage, surgery and radiotherapy variables, and the nomogram showed better ability to predict the prognosis than the TNM stage nomogram. Using this nomogram, urologists can evaluate the prognostic survival of patients with GCTC, enabling personalized treatment and monitoring possible. There are limitations to be recognized in this study. First, this study was a retrospective study with limitations, sample and ethnic selection bias and more cases needed for prospective studies. Second, the SEER database has certain limitations regarding type/duration of treatment and recurrence of disease and we cannot obtain detailed specific information (dose, beam energy and fractionation) of radiotherapy. Moreover, the information on the patient's physical condition and complications is lacking, both of which are prognostic factors for patients with GCTC.
Based on a large number of population data, we developed prognostic nomogram for GCTC patients, which can accurately and reliably predict the 3-, 5- and 10-year OS and CSS in individual GCTC patients. The proposed GCTC survival model can help clinicians make personalize therapies and adjust follow-up strategies.
Materials and Methods
Patients selection
The data presented in our study were retrieved from the Surveillance Epidemiology and End Results (SEER) database, which funded by the National Cancer Institute. The SEER database covers approximately 28% of the US population and includes demographic information and cancer characteristics, such as diagnosis age, insurance status, race, marital status, income status, primary tumor site, tumor grade and stage, histological type, Tumor-Node-Metastasis (TNM) stage, treatment modality and survival time [25]. The National Cancer Institute's SEER*Stat software version 8.3.5 (https://seer.cancer.gov/seerstat/)(SEER 18 Regs Custom Data (with additional treatment fields), Nov 2018 Sub (1975-2016 varying) database) was used in this study.
The International Classification of Diseases for Oncology (ICD-O) site codes C62.1 and C62.9 were used to identify patients diagnosed with TC between 2004 and 2015. We collected 26,780 GCTC patients according to the ninth edition of the International Classification of Diseases (codes: 9061 to 9064, 9070 to 9071, 9080 to 9085 and 9100 to 9102). Exclusion criteria in our study were as follows: (a) unknown T stage and T0 (n=1,808); (b) unknown N stage (n=831); (c) unknown M stage (n=118); (d) not one primary tumor only (n=1,948). All patients were followed up until December 2016 to ensure that all cases were observed for more than one year at the last follow-up. Finally, we left 22,165 eligible patients diagnosed with GCTC (Supplementary GCTC data).
Study variables
Variable definition information about age at diagnosis, race, AJCC stage, T stage, N stage, M stage, SEER stage, surgery, radiotherapy, cause of death and survival time can be found in the SEER database. The starting point of the follow-up was the date of diagnosis of GCTC, and the end point was cancer-specific death or the last follow-up in December 2016. The overall survival (OS) time corresponded to the length of time from the date of diagnosis to the death from any cause or the date on which data were censored. During analyzing cancer-specific survival (CSS), mortality cases associated with other causes were excluded.
Statistical analysis
We randomly assigned 70% of patients to the training cohort (n=15,515) and the remaining 30% to the validation cohort (n=6,650). Kaplan-Meier curve was used to estimate the OS and CSS of GCTC, and the difference between the curves was analyzed by log-rank test. Univariate and multivariate Cox regression models were performed to estimate the hazard ratios (HR) and 95% confidence intervals (CI) to analyze independent prognostic factors of GCTC.
Using R software, we constructed two nomograms: one nomogram the multivariate Cox regression analysis, and the other one nomogram based on TNM stage, to predict the OS and CSS probabilities of individual patients. We first used the R software to generate the receiver operating characteristic (ROC) curve for the two nomograms and determined the area under the curve (AUC). In addition, by comparing the predicted survival time with the observed survival time, the predictive performance of the nomogram was evaluated using the consistency index (C-index) and calibration curve, and the nomogram was calibrated for 3-, 5- and 10-years OS and CSS. The C index was similar to the AUC, but seems to be more suitable for censored data. The value of the C-index statistic was between 0.5 (non-discrimination) and 1 (perfect discrimination), and a higher C-index value indicates a better prognostic model. These evaluations were performed using a bootstrap with 1000 resamples.
There was no direct clinical interpretation for C-index. Therefore, we also analyzed the decision curve analysis (DCA), which is a novel method to evaluate the predictive model for evaluating net benefits from the perspective of clinical outcome, and plotted the clinical impact curve (CIC) based on the results of DCA.
The above statistical analysis uses SPSS version 20.0 (SPSS, Chicago, USA) and statistical software package R version 3.5.3 (http://www.r-project.org/) and R software packages rms, survival, formula, ggplot2 and rmda. P-value ≤ 0.05 (two-sided) was considered statistically significant.
Author Contributions
WM, BX and MC studied the concept and design. WM, JW, QK and JL collected the data. WM, JW and QK analyzed and interpreted the data. WM and JW drafted the manuscript. BX and MC critically revised the manuscript for important intellectual content. WM and JW performed the statistical analysis.
Conflicts of Interest
We declare that there are no conflicts of interest between authors.
Funding
This work was supported by the National Natural Science Foundation of China (No. 81572517 and 81872089), Natural Science Foundation of Jiangsu Province (BK20161434), Jiangsu Provincial Medical Innovation Team (CXTDA2017025), and Jiangsu Provincial Medical Talent (ZDRCA2016080).
Research involving Human Participants and/or Animals: This article does not contain any studies with human participants or animals performed by any of the authors.
References
- 1. Pearce SM, Golan S, Gorin MA, Luckenbaugh AN, Williams SB, Ward JF, Montgomery JS, Hafez KS, Weizer AZ, Pierorazio PM, Allaf ME, Eggener SE. Safety and early oncologic effectiveness of primary robotic retroperitoneal lymph node dissection for nonseminomatous germ cell testicular cancer. Eur Urol. 2017; 71:476–82. https://doi.org/10.1016/j.eururo.2016.05.017 [PubMed]
- 2. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018; 68:394–424. https://doi.org/10.3322/caac.21492 [PubMed]
- 3. McGlynn KA, Trabert B. Adolescent and adult risk factors for testicular cancer. Nat Rev Urol. 2012; 9:339–49. https://doi.org/10.1038/nrurol.2012.61 [PubMed]
- 4. Horwich A, Shipley J, Huddart R. Testicular germ-cell cancer. Lancet. 2006; 367:754–65. https://doi.org/10.1016/S0140-6736(06)68305-0 [PubMed]
- 5. Hanna NH, Einhorn LH. Testicular cancer—discoveries and updates. N Engl J Med. 2014; 371:2005–16. https://doi.org/10.1056/NEJMra1407550 [PubMed]
- 6. Cook MB, Graubard BI, Rubertone MV, Erickson RL, McGlynn KA. Perinatal factors and the risk of testicular germ cell tumors. Int J Cancer. 2008; 122:2600–06. https://doi.org/10.1002/ijc.23424 [PubMed]
- 7. Dalgaard MD, Weinhold N, Edsgärd D, Silver JD, Pers TH, Nielsen JE, Jørgensen N, Juul A, Gerds TA, Giwercman A, Giwercman YL, Cohn-Cedermark G, Virtanen HE, et al. A genome-wide association study of men with symptoms of testicular dysgenesis syndrome and its network biology interpretation. J Med Genet. 2012; 49:58–65. https://doi.org/10.1136/jmedgenet-2011-100174 [PubMed]
- 8. Verdecchia A, Francisci S, Brenner H, Gatta G, Micheli A, Mangone L, Kunkler I, and EUROCARE-4 Working Group. Recent cancer survival in Europe: a 2000-02 period analysis of EUROCARE-4 data. Lancet Oncol. 2007; 8:784–96. https://doi.org/10.1016/S1470-2045(07)70246-2 [PubMed]
- 9. Shanmugalingam T, Soultati A, Chowdhury S, Rudman S, Van Hemelrijck M. Global incidence and outcome of testicular cancer. Clin Epidemiol. 2013; 5:417–27. https://doi.org/10.2147/CLEP.S34430 [PubMed]
- 10. Zhang G, Wu Y, Zhang J, Fang Z, Liu Z, Xu Z, Fan Y. Nomograms for predicting long-term overall survival and disease-specific survival of patients with clear cell renal cell carcinoma. Onco Targets Ther. 2018; 11:5535–44. https://doi.org/10.2147/OTT.S171881 [PubMed]
- 11. Mead GM, Stenning SP. The international germ cell consensus classification: a new prognostic factor-based staging classification for metastatic germ cell tumours. Clin Oncol (R Coll Radiol). 1997; 9:207–09. https://doi.org/10.1016/s0936-6555(97)80001-5 [PubMed]
- 12. Amin MB, Greene FL, Edge SB, Compton CC, Gershenwald JE, Brookland RK, Meyer L, Gress DM, Byrd DR, Winchester DP. The eighth edition AJCC cancer staging manual: continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin. 2017; 67:93–99. https://doi.org/10.3322/caac.21388 [PubMed]
- 13. Ge H, Yan Y, Xie M, Guo L, Tang D. Construction of a nomogram to predict overall survival for patients with M1 stage of colorectal cancer: a retrospective cohort study. Int J Surg. 2019; 72:96–101. https://doi.org/10.1016/j.ijsu.2019.10.021 [PubMed]
- 14. Iasonos A, Schrag D, Raj GV, Panageas KS. How to build and interpret a nomogram for cancer prognosis. J Clin Oncol. 2008; 26:1364–70. https://doi.org/10.1200/JCO.2007.12.9791 [PubMed]
- 15. Eastham JA, Kattan MW, Scardino PT. Nomograms as predictive models. Semin Urol Oncol. 2002; 20:108–15. https://doi.org/10.1053/suro.2002.32936 [PubMed]
- 16. Hou G, Zheng Y, Wei D, Li X, Wang F, Tian J, Zhang G, Yan F, Zhu Z, Meng P, Yuan J, Gao M, Li Z, et al. Development and validation of a SEER-based prognostic nomogram for patients with bone metastatic prostate cancer. Medicine (Baltimore). 2019; 98:e17197. https://doi.org/10.1097/MD.0000000000017197 [PubMed]
- 17. Wu S, Chen JN, Zhang QW, Tang CT, Zhang XT, Tang MY, Li XB, Ge ZZ. A new metastatic lymph node classification-based survival predicting model in patients with small bowel adenocarcinoma: a derivation and validation study. EBioMedicine. 2018; 32:134–41. https://doi.org/10.1016/j.ebiom.2018.05.022 [PubMed]
- 18. Touijer K, Scardino PT. Nomograms for staging, prognosis, and predicting treatment outcomes. Cancer. 2009; 115:3107–11. https://doi.org/10.1002/cncr.24352 [PubMed]
- 19. Shariat SF, Karakiewicz PI, Suardi N, Kattan MW. Comparison of nomograms with other methods for predicting outcomes in prostate cancer: a critical analysis of the literature. Clin Cancer Res. 2008; 14:4400–07. https://doi.org/10.1158/1078-0432.CCR-07-4713 [PubMed]
- 20. Bochner BH, Kattan MW, Vora KC, and International Bladder Cancer Nomogram Consortium. Postoperative nomogram predicting risk of recurrence after radical cystectomy for bladder cancer. J Clin Oncol. 2006; 24:3967–72. https://doi.org/10.1200/JCO.2005.05.3884 [PubMed]
- 21. Ljungberg B. Kidney cancer: a new nomogram predicting survival in renal cell carcinoma. Nat Rev Urol. 2010; 7:423–24. https://doi.org/10.1038/nrurol.2010.117 [PubMed]
- 22. Karakiewicz PI, Shariat SF, Palapattu GS, Perrotte P, Lotan Y, Rogers CG, Amiel GE, Vazina A, Gupta A, Bastian PJ, Sagalowsky AI, Schoenberg M, Lerner SP. Precystectomy nomogram for prediction of advanced bladder cancer stage. Eur Urol. 2006; 50:1254–60. https://doi.org/10.1016/j.eururo.2006.06.010 [PubMed]
- 23. Kattan MW, Eastham JA, Stapleton AM, Wheeler TM, Scardino PT. A preoperative nomogram for disease recurrence following radical prostatectomy for prostate cancer. J Natl Cancer Inst. 1998; 90:766–71. https://doi.org/10.1093/jnci/90.10.766 [PubMed]
- 24. Zhou Y, Zhang R, Ding Y, Wang Z, Yang C, Tao S, Liang C. Prognostic nomograms and aggtrmmns scoring system for predicting overall survival and cancer-specific survival of patients with kidney cancer. Cancer Med. 2020; 9:2710–22. https://doi.org/10.1002/cam4.2916 [PubMed]
- 25. Mao W, Zhang Z, Huang X, Fan J, Geng J. Marital status and survival in patients with penile cancer. J Cancer. 2019; 10:2661–69. https://doi.org/10.7150/jca.32037 [PubMed]



