Introduction
Telomeres are specialized structures that, through the formation of a loop, protect chromosome ends from DNA damage response activation [1]. Telomeres progressively shorten with age, leading to the loss of chromosome end protection and the activation of a p53-dependent DNA damage response that triggers senescence or apoptosis [2, 3]. Short telomeres are associated with a higher risk of aplasia and lung fibrosis, probably linked to early progenitor cell exhaustion [4, 5]. More recently, several studies have demonstrated the impact of shorter telomeres on immune cell function, a phenomenon referred to as immunosenescence. Immunosenescence is an age-dependent process associated with the progressive depletion of naïve T cells and the reduced proliferation ability of T cell that likely impact immune surveillance against persistent viral infections like Cytomegalovirus (CMV) in the elderly [6, 7]. A recent report established that abnormally short telomeres in patients with telomere-related gene mutations are sufficient to drive T cell aging, although additional and still undefined telomere length-independent molecular programs further contribute to immunosenescence in the elderly [8].
Other recent data support a role for telomeres in defense against pathogens: adults with shorter telomeres are more sensitive to experimentally-induced respiratory viral infection [9]. Along the same lines, short leukocyte telomere length (TL) was associated with more severe acute respiratory distress syndrome and worse survival in patients with sepsis [10].
Outcome of the current coronavirus disease 19 (COVID-19) pandemic is highly heterogeneous, ranging from asymptomatic people to patients hospitalized in intensive care units (ICU) with need of mechanical ventilation and eventual fatal outcome due to respiratory failure. To date, the strongest risk factor associated with severe disease and death in COVID-19 is older age [11], with infection fatality rate ranging from eight to 36% in people aged ≥80 years [12]. Yet, studies performed exclusively in younger hospitalized patients –representing the most severely ill patients- reported similar fatality ratios of 8-28%, suggesting that age is not the only factor modulating COVID-19 outcome [12]. Lymphopenia is another risk factor for poor outcome, pointing towards a potential role for telomere modulation in COVID-19 [13, 14].
Based on the above, we hypothesized that shorter TL might be linked to poorer outcome in COVID-19 and addressed this hypothesis in a prospective cohort of hospitalized patients.
Results
Study population
We prospectively recruited 70 patients hospitalized in COVID-19 dedicated units between April 7th and May 27th, 2020, during the main wave of COVID-19 in Belgium. The clinical characteristics of our patients are provided in Table 1. Our cohort included 48 men (68.6%). Median age was 63 years-old (range 27-96). Fifty-three (75.7%) patients had at least one chronic disease including hypertension (22, 31.4%), previously documented hypercholesterolemia (20, 28.6%), diabetes (13, 18.6%), obesity defined as a BMI>30 kg/m² (9, 12.9%) or ischemic cardiovascular disease (8, 11.4%). Twenty-five patients (35.7%) were current or ex-smokers. At the day of admission in the hospital (baseline), no patient had received any COVID-19-related treatment. During their hospitalization, a majority of subjects received hydroxychloroquine (59, 84.3%), as this drug was at that time recommended for hospitalized patients in Belgium. Eleven patients (15.7%) experienced a thrombotic event requiring therapeutic anticoagulation (five deep venous thrombosis, three arterial thrombosis, two ischemic strokes and one pulmonary embolism). During hospitalization, median peak oxygen flow was 10 liter/minute administered through a mask, which corresponds to a fraction of inspired oxygen of about 0.9. Fourteen patients (20%) beneficiated from continuous positive airway pressure (CPAP).
Table 1. Clinical characteristics of the TELECOVID cohort (all values are median and range, unless specified).
| Baseline clinical features | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years) | 63 (27-96) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex ratio (M/F) | 48/22 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ethnicity (N) | 49 Caucasians, 11 Northern Africans, 5 Asians, 4 Africans, 1 Southern American | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Current or ex-smokers (N, %) | 25 (35.7%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension | 22 (31.4%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Known hypercholesterolaemia (N, %) | 20 (28.6%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes | 13 (18.6%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Obesity (BMI>30 kg/m²) (N, %) | 9 (12.9%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ischemic cardiovascular disease | 8 (11.4%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symptoms duration prior to admission (days) | 7 (1-15) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Disease extend on HRCT (%)* | 20.61 (0.47-68.78) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline biological features | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CRP (mg/L) | 95.95 (1.3-353.2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDH (IU/L) | 372 (162-1855) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ASAT/GOT (U/L) | 41 (8-242) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PaO2with fraction of inspired oxygen 0.21 (ambient air, mmHg) | 64.5 (26-134) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Blood lymphocytes (x10³/μL) | 0.68 (0.17-2.13) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Blood neutrophils (x10³/μL) | 4.99 (0.48-15.91) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Neutrophils/lymphocytes ratio | 6.03 (0.86-50.57) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Eosinophils (x10³/μL) | 0 (0-0.18) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| COVID-19 management characteristics | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hospitalisation duration (days)† | 19.5 (4-102) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Specific treatment (N, %), including
| 59 (84.3%)
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Admission in ICU (N, %) | 33 (47.1%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Days in ICU‡ | 23 (2-70) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Death (N, %) | 18 (25.7%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| *: absence of baseline HRCT for 8 patients. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| †: 2 missing data (patients still hospitalised). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ‡: 1 patients received an anti-IL-6 monoclonal antibody, 2 patients received azithromycin. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Thirty-three patients (47.1%) were admitted in intensive care, of whom 30 patients required mechanical ventilation (42.8%). Nine patients (12.9%) benefited from extra-corporeal membrane oxygenation (ECMO). Eighteen patients (25.7%) died from COVID-19.
Increased frequency of short telomere individuals in hospitalized COVID-19 patients
We measured TL in leucocytes using the Flow-FISH technique [15]. Figure 1A shows the distribution of individuals within the indicated percentile ranges of TL. When compared to the reference cohort, we found a clear enrichment of patients with telomeres
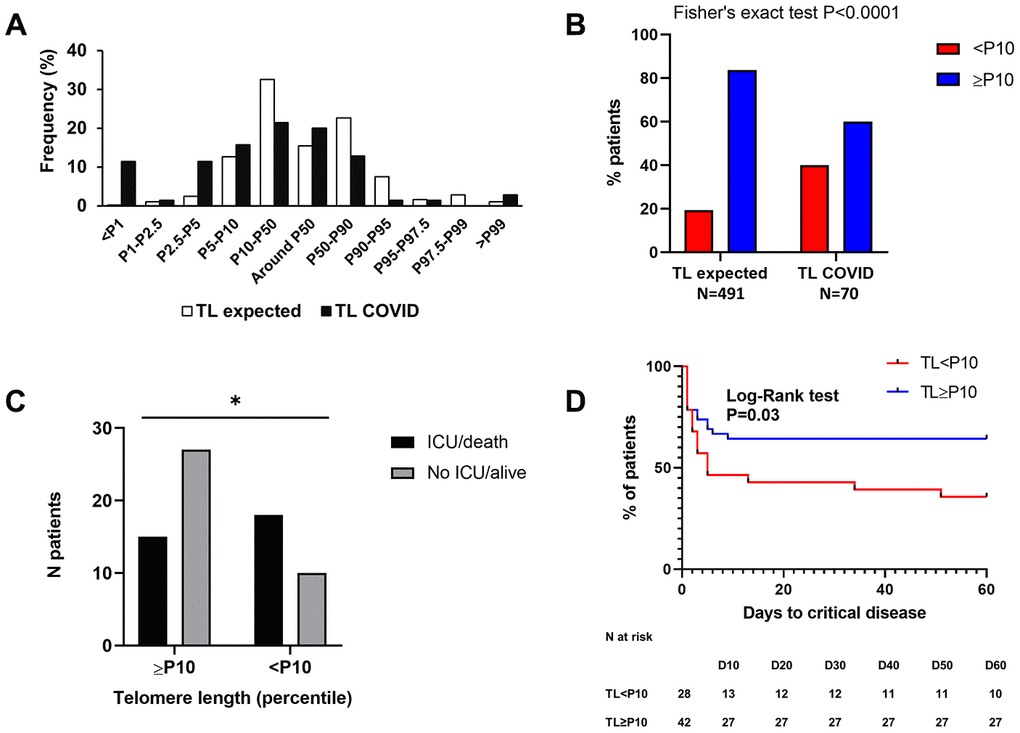
Figure 1. High proportion of COVID-19 patients with short telomeres and link with outcome. (A) As compared to the reference population (white bars), we found an enrichment of COVID-19 patients with short telomeres (black bars). (B) There is a statistically significant association between short telomeres (
Short telomeres are associated with a higher risk of unfavorable COVID-19 outcome and correlate with prognostic biological factors
We segregated our population based on short (<10th percentile, P10) or normal (≥P10) telomere length (Table 2). Twenty-eight (40%) patients had a TL We found that TL We then studied the correlation between TL (expressed in percentile) and biological factors previously associated with poorer COVID-19 outcome, namely C-reactive protein level (CRP), lymphocyte count, NLR and eosinophils. We found a moderate but statistically significant correlation between TL and CRP (Spearman r = -0.259, P=0.03) and NLR (Spearman r = -0.268, P=0.025), meaning that the lower the TL, the higher the CRP and NLR. There was a trend for a significant correlation between TL and lymphocyte count (Spearman r = 0.197, P=0.11), suggesting that the longer the telomeres, the higher the blood lymphocytes at admission.Table 2. Comparison of key clinical and biological features between patients with telomere length below and equal or above the 10th percentile.
TL < 10th percentile (N=28) TL ≥ 10th percentile (N=42) P-value (Mann-Whitney U test) Clinical data and outcome Age (years, range) 64 (47-85) 62.5 (27-96) 0.21μ Sex ratio (M/F) 23/5 25/17 0.07* HRCT extend (%, range) 23.80 (2.28-68.78) 18.57 (0.47-68.39) 0.41 Hospitalization duration (days, range) † 24 (6-96) 16 (4-74) 0.10 Death 10 (35.7%) 8 (19.0%) 0.16 Admission in ICU and/or death 18 (64.3%) 15 (35.7%) 0.02* Biological features NLR (median, range) 7.65 (3.08-50.57) 5.36 (1.23-25.6) 0.01 Blood lymphocytes at admission (x10³/μL, median, range) 0.68 (0.17-2.01) 0.74 (0.19-2.13) 0.69 Blood neutrophils at admission (x10³/μL, median, range) 5.17 (1.98-12.15) 4.36 (0.48-15.91) 0.20 CRP at admission (mg/L, median, range) 120 (8.6-353.2) 71.15 (1.3-330.8) 0.11 μ: Student T-test *: Fisher’s exact test $: Chi-Square test †: 2 missing data (patients still hospitalised)
Increased cellular senescence in lungs from COVID-19 patients with very short telomeres
To evaluate whether short telomeres of COVID-19 patients may be associated with immune cell senescence, we evaluated SA-β-gal activity in lungs from non-survivor patients. Our results revealed high levels of SA-β-gal activity in both immune and structural lung cells from two COVID-19 patients of 55 and 74 years old with TL below the first percentile (P1) (Figure 2A). We did not detect any SA-β-gal activity in the lung of another 55 years old deceased COVID-19 patient with telomeres lying in the P50-P90 range (Figure 2A). In the remaining lungs from deceased patients with TL >P1, low to moderate levels of SA-β-gal activity were detected (Figure 2A, 2B). As previously demonstrated [16], negative controls obtained from healthy lung tissues of donors aged 38-82 years old did not reveal any SA-β-gal activity (Figure 2A, 2B).
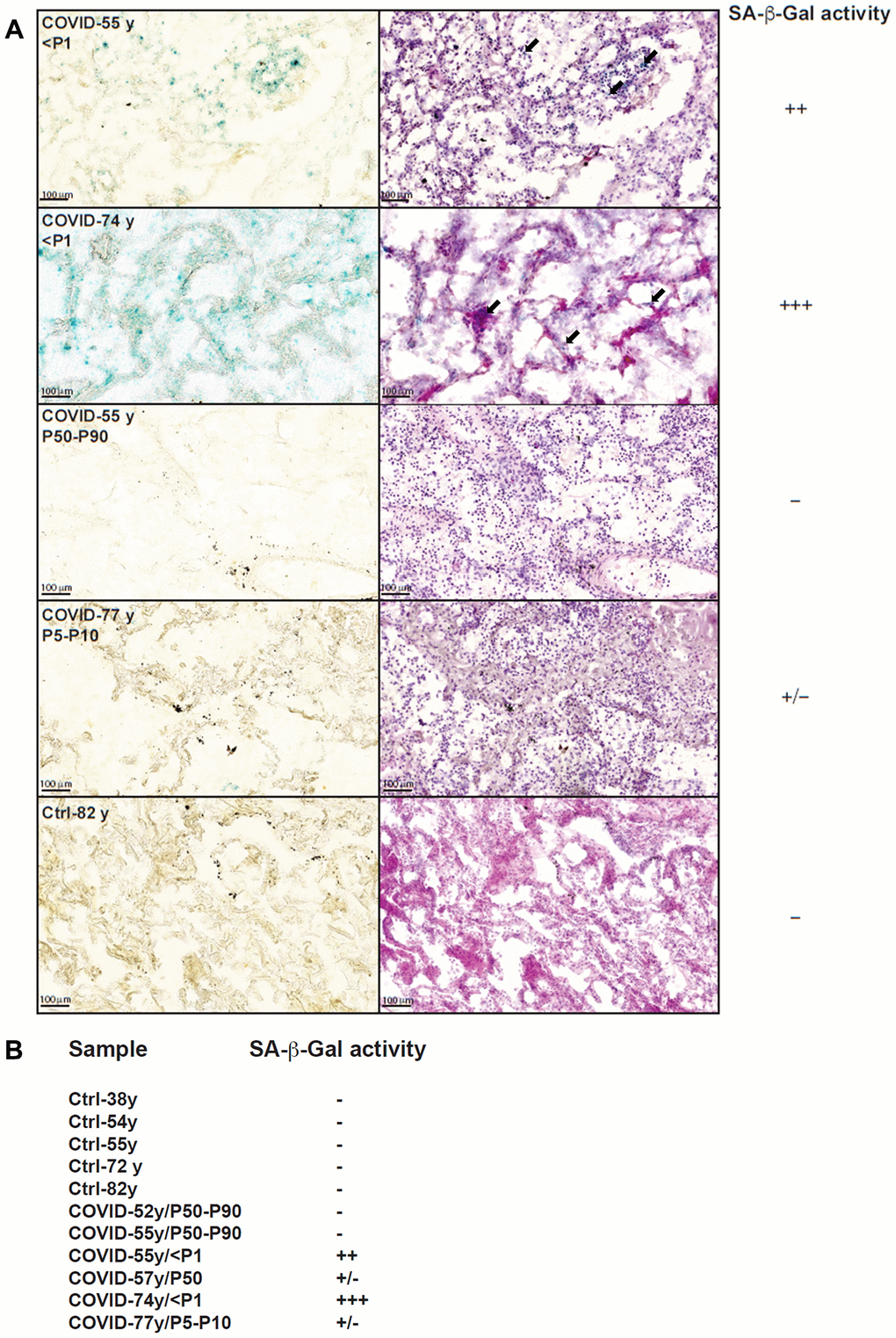
Figure 2. SA-β-Gal staining of lung slices from COVID-19 and control patients. (A) SA-β-Gal staining (left) and HE staining (right) of four COVID-19 lungs and one control lung. We only found signs of senescence (blue staining) in lungs from individuals with very short telomeres (
Discussion
Our study in a cohort of 70 hospitalized COVID-19 patients revealed an enrichment for individuals with short telomeres in comparison with the reference population. Furthermore, short telomeres (
The progressive shortening of lymphocyte telomeres with aging has been proposed to contribute to the age-associated defects of immunity and the higher risk of long-term infection with CMV [6] and mortality [19]. Conversely, healthy elderly exhibit longer telomeres than their peers do [20, 21]. Beyond COVID-19 and other viral diseases, differences in TL and telomere homeostasis may underlie impaired immune response towards various pathogens, as illustrated by the recent demonstration that sepsis outcome was also linked to TL [10].
We [22] and others previously showed that prematurely aged skin fibroblasts from patients with abnormally short telomeres display increased SA-β-gal activity, a marker first described in senescent skin fibroblasts and keratinocytes. Here, we only found signs of senescence in the lungs of non-survivors with very short telomeres ( Short telomeres also possibly impact COVID-19 outcome through the modulation of cytokine production, as illustrated by our finding that TL correlated with NLR, which constitutes a prognostic factor in multiple diseases [23], and CRP. Along this line, the increased transcription of short telomeres into TERRA non-coding RNAs was reported to promote IL-6 and TNF-α secretion [24, 25]. Short telomeres have also been associated with an increased transcription of ISG15 [26], a gene related to Type 1 interferon signaling. Hence, short telomeres may also possibly contribute to the harmful cytokine storm of COVID-19 patients. As a limitation, our cohort was composed of severely to critically ill patients, with about a half of them being admitted to ICU and a fatality rate of 25.6%. Therefore, whether our result may apply to the overall population of COVID-19 patients still needs to be confirmed. In conclusion, we uncovered a link between telomere length and COVID-19 outcome, with a potential impact of TL on biological parameters. Altogether, our study paves the way for further investigations on a potential interest of TL as a prognostic factor of COVID-19. Whether TL may influence long-term outcome like lung fibrosis is also unknown at this point.
Materials and Methods
Study design
The TElomere LEngth in COronaVIrus Disease 2019 (TELECOVID) trial is a single-center prospective trial. We prospectively included patients admitted in our dedicated COVID-19 units that fulfilled the following criteria: evidence of COVID-19 defined as the combination of a positive PCR on nasopharyngeal swab and lung infiltrates on high resolution computed tomography (HRCT) or chest X-ray at admission. All patients underwent blood sampling for TL measurement on peripheral blood granulocytes. We collected clinical, biological and radiological data on the day of admission in the hospital. We quantified the extension of lesions on admission HRCT with the CT Pneumonia Analysis software (Siemens Helthineers, Forchheim, Germany) and obtained a percentage opacity score, defined as the percentage of predicted volume of abnormalities compared to the total lung volume.
TL measurements obtained in COVID-19 patients were compared to those of a reference cohort of 491 healthy volunteers (from 0 to 99 years) obtained in the Cliniques Universitaires Saint-Luc (ISO15189).
We have conducted this cohort study in accordance with STROBE statement (https://www.strobe-statement.org/index.php?id=strobe-home).
Study objectives
Our primary objectives were to measure telomere length in hospitalized COVID-19 patients and compare it to nomograms from our reference population and to study the potential association between short telomeres (below tenth percentile -P10) and unfavorable COVID-19 outcome, defined as admission in ICU and/or death without ICU (critical disease, according to the definition of NIH, covid19treatmentguidelines.nih.gov/overview/management-of-covid-19/). Our secondary objectives were to correlate TL to baseline (at the day of admission in hospital) biological and radiological features. Finally, we sought to detect signs of senescence in lung samples from deceased COVID-19 patients.
Blood sample processing and telomere length (TL) measurement
TL was measured by Flow-FISH, currently considered as the gold standard technique [27]. The severe lymphopenia of most COVID-19 patients, together with the possible acute infection-driven shortening of telomeres in T lymphocytes, support the measurement of TL in the granulocytes rather than the lymphocytes. To confirm that granulocyte TL properly reflects lymphocyte TL, we compared their respective percentile ranges for individuals of the cohort. We found a good consistency between the two cell populations as, for nearly 60% of the individuals of the cohort, the percentile range was the same while, for 33% of them, only one category difference was observed (Supplementary Figure 1). Hence, for the large majority of individuals, granulocyte TL properly reflects lymphocyte TL.
All TL measurements were performed in duplicate in the Cliniques Universitaires Saint-Luc through Flow-FISH as described previously [15]. After red blood cell lysis with ammonium chloride as described in [15], white blood cells were frozen at -80° C until analysis. Aliquots of calf thymus were mixed to each sample for internal control. Telomeres were stained with FITC-labelled (CCCTAA)3 PNA probe (Panagene) and fluorescence was measured with a Navios EX flow cytometer (Beckman Coulter).
Senescence-associated-β-galactosidase activity in lung tissues
We had access to autopsy lung tissue from COVID-19 deceased patients and control lung tissue from our local biobank (IREC-PNEU). We slightly adapted the protocol previously described to perform senescence-associated β-galactosidase (SA-β-Gal) [28]. Briefly, after fixation with 0.2% formaldehyde for 10 min, 16 μm-thick sections of snap-frozen lung tissues were rinsed with PBS before overnight incubation at 37° C in staining solution at pH 5.6. After scanning, slides were incubated overnight in PBS 1x at RT, rinsed with ddH20 and stained first with hematoxylin and then with eosin. After a new rinsing with water, tissues were dehydrated through a series of ethanol baths, from 30% to 100%, before incubation in isopropanol and then xylene and, finally, mounting.
Statistics
We used Chi-Square and Fisher’s exact test for associations and Spearman R test for correlation analysis. We used Student t test and Mann-Whitney U test for single comparisons when appropriate. We performed a Kaplan-Meyer curve and used Log-Rank test for comparing populations. All statistics were performed on SPSS 25 software (IBM, Armonk, NY, USA) and GraphPad Prism 8.4.1 (Graphpad Software, San Diego, CA, USA). A P-value under 0.05 was considered significant.
Ethical considerations
All patients provided informed consent prior to inclusion. This study was approved by our internal review board (Comité d’éthique hospitalo-facultaire), approval number 2020/06AVR/200.
Author Contributions
A. Froidure designed the study, supervised the recruitment of patients, conducted analysis and wrote the manuscript. J-P Defour and A. Decottignies supervised Flow-FISH and lung staining, conducted analysis and wrote the manuscript. M. Mahieu performed Flow-FISH and lung staining. D. Hoton analyzed lung slices. PF Laterre and JC Yombi reviewed the study design and supervised recruitment within their respective departments. S. Koenig collected clinical data and coordinated the recruitment of patients. B. Ghaye reviewed all high-resolution computed tomography (HCT) and quantified lesion extend. All authors revised the manuscript.
Acknowledgments
We are indebted to all the COVID-19 patients who volunteered for this study. We sincerely acknowledge the help from Nicolas Dauguet (de Duve Institute, Brussels) in setting up the FACS analyses for Flow-FISH. We thank Mrs Leslie Gielens, Caroline Berghe, Marie-France Dujardin and Suzanne Renard for their help in the recruitment of patients admitted in ICU. We also wish to thank Mrs Carole Dekelver (Clinical Trial Center, Cliniques Universitaires Saint-Luc) for her support in setting up the study.
Conflicts of Interest
None of the authors has any conflicts of interest related to the present work.
Funding
This work is supported by a grant from the Fondation Louvain (grant number M1.21221.004), Université catholique de Louvain, Belgium.
References
- 1. de Lange T. Shelterin-mediated telomere protection. Annu Rev Genet. 2018; 52:223–47. https://doi.org/10.1146/annurev-genet-032918-021921 [PubMed]
- 2. d’Adda di Fagagna F, Reaper PM, Clay-Farrace L, Fiegler H, Carr P, Von Zglinicki T, Saretzki G, Carter NP, Jackson SP. A DNA damage checkpoint response in telomere-initiated senescence. Nature. 2003; 426:194–98. https://doi.org/10.1038/nature02118 [PubMed]
- 3. Hemann MT, Strong MA, Hao LY, Greider CW. The shortest telomere, not average telomere length, is critical for cell viability and chromosome stability. Cell. 2001; 107:67–77. https://doi.org/10.1016/s0092-8674(01)00504-9 [PubMed]
- 4. Armanios M, Blackburn EH. The telomere syndromes. Nat Rev Genet. 2012; 13:693–704. https://doi.org/10.1038/nrg3246 [PubMed]
- 5. Alder JK, Barkauskas CE, Limjunyawong N, Stanley SE, Kembou F, Tuder RM, Hogan BL, Mitzner W, Armanios M. Telomere dysfunction causes alveolar stem cell failure. Proc Natl Acad Sci USA. 2015; 112:5099–104. https://doi.org/10.1073/pnas.1504780112 [PubMed]
- 6. Wherry EJ, Kurachi M. Molecular and cellular insights into T cell exhaustion. Nat Rev Immunol. 2015; 15:486–99. https://doi.org/10.1038/nri3862 [PubMed]
- 7. Fülöp T, Larbi A, Pawelec G. Human T cell aging and the impact of persistent viral infections. Front Immunol. 2013; 4:271. https://doi.org/10.3389/fimmu.2013.00271 [PubMed]
- 8. Wagner CL, Hanumanthu VS, Talbot CC
Jr , Abraham RS, Hamm D, Gable DL, Kanakry CG, Applegate CD, Siliciano J, Jackson JB, Desiderio S, Alder JK, Luznik L, Armanios M. Short telomere syndromes cause a primary T cell immunodeficiency. J Clin Invest. 2018; 128:5222–34. https://doi.org/10.1172/JCI120216 [PubMed] - 9. Cohen S, Janicki-Deverts D, Turner RB, Casselbrant ML, Li-Korotky HS, Epel ES, Doyle WJ. Association between telomere length and experimentally induced upper respiratory viral infection in healthy adults. JAMA. 2013; 309:699–705. https://doi.org/10.1001/jama.2013.613 [PubMed]
- 10. Liu S, Wang C, Green G, Zhuo H, Liu KD, Kangelaris KN, Gomez A, Jauregui A, Vessel K, Ke S, Hendrickson C, Matthay MA, Calfee CS, et al. Peripheral blood leukocyte telomere length is associated with survival of sepsis patients. Eur Respir J. 2020; 55:1901044. https://doi.org/10.1183/13993003.01044-2019 [PubMed]
- 11. Docherty AB, Harrison EM, Green CA, Hardwick HE, Pius R, Norman L, Holden KA, Read JM, Dondelinger F, Carson G, Merson L, Lee J, Plotkin D, et al, and ISARIC4C investigators. Features of 20 133 UK patients in hospital with covid-19 using the ISARIC WHO clinical characterisation protocol: prospective observational cohort study. BMJ. 2020; 369:m1985. https://doi.org/10.1136/bmj.m1985 [PubMed]
- 12. Verity R, Okell LC, Dorigatti I, Winskill P, Whittaker C, Imai N, Cuomo-Dannenburg G, Thompson H, Walker PG, Fu H, Dighe A, Griffin JT, Baguelin M, et al. Estimates of the severity of coronavirus disease 2019: a model-based analysis. Lancet Infect Dis. 2020; 20:669–77. https://doi.org/10.1016/S1473-3099(20)30243-7 [PubMed]
- 13. Jordan RE, Adab P, Cheng KK. Covid-19: risk factors for severe disease and death. BMJ. 2020; 368:m1198. https://doi.org/10.1136/bmj.m1198 [PubMed]
- 14. Aviv A. Telomeres and COVID-19. FASEB J. 2020; 34:7247–52. https://doi.org/10.1096/fj.202001025 [PubMed]
- 15. Baerlocher GM, Vulto I, de Jong G, Lansdorp PM. Flow cytometry and FISH to measure the average length of telomeres (flow FISH). Nat Protoc. 2006; 1:2365–76. https://doi.org/10.1038/nprot.2006.263 [PubMed]
- 16. Minagawa S, Araya J, Numata T, Nojiri S, Hara H, Yumino Y, Kawaishi M, Odaka M, Morikawa T, Nishimura SL, Nakayama K, Kuwano K. Accelerated epithelial cell senescence in IPF and the inhibitory role of SIRT6 in TGF-β-induced senescence of human bronchial epithelial cells. Am J Physiol Lung Cell Mol Physiol. 2011; 300:L391–401. https://doi.org/10.1152/ajplung.00097.2010 [PubMed]
- 17. Cao Y, Li L, Feng Z, Wan S, Huang P, Sun X, Wen F, Huang X, Ning G, Wang W. Comparative genetic analysis of the novel coronavirus (2019-nCoV/SARS-CoV-2) receptor ACE2 in different populations. Cell Discov. 2020; 6:11. https://doi.org/10.1038/s41421-020-0147-1 [PubMed]
- 18. Hu Q, Guan H, Sun Z, Huang L, Chen C, Ai T, Pan Y, Xia L. Early CT features and temporal lung changes in COVID-19 pneumonia in Wuhan, China. Eur J Radiol. 2020; 128:109017. https://doi.org/10.1016/j.ejrad.2020.109017 [PubMed]
- 19. Cawthon RM, Smith KR, O’Brien E, Sivatchenko A, Kerber RA. Association between telomere length in blood and mortality in people aged 60 years or older. Lancet. 2003; 361:393–95. https://doi.org/10.1016/S0140-6736(03)12384-7 [PubMed]
- 20. Tedone E, Huang E, O’Hara R, Batten K, Ludlow AT, Lai TP, Arosio B, Mari D, Wright WE, Shay JW. Telomere length and telomerase activity in T cells are biomarkers of high-performing centenarians. Aging Cell. 2019; 18:e12859. https://doi.org/10.1111/acel.12859 [PubMed]
- 21. Steenstrup T, Kark JD, Verhulst S, Thinggaard M, Hjelmborg JV, Dalgård C, Kyvik KO, Christiansen L, Mangino M, Spector TD, Petersen I, Kimura M, Benetos A, et al. Telomeres and the natural lifespan limit in humans. Aging (Albany NY). 2017; 9:1130–42. https://doi.org/10.18632/aging.101216 [PubMed]
- 22. Benyelles M, Episkopou H, O’Donohue MF, Kermasson L, Frange P, Poulain F, Burcu Belen F, Polat M, Bole-Feysot C, Langa-Vives F, Gleizes PE, de Villartay JP, Callebaut I, et al. Impaired telomere integrity and rRNA biogenesis in PARN-deficient patients and knock-out models. EMBO Mol Med. 2019; 11:e10201. https://doi.org/10.15252/emmm.201810201 [PubMed]
- 23. Forget P, Khalifa C, Defour JP, Latinne D, Van Pel MC, De Kock M. What is the normal value of the neutrophil-to-lymphocyte ratio? BMC Res Notes. 2017; 10:12. https://doi.org/10.1186/s13104-016-2335-5 [PubMed]
- 24. Wang Z, Deng Z, Dahmane N, Tsai K, Wang P, Williams DR, Kossenkov AV, Showe LC, Zhang R, Huang Q, Conejo-Garcia JR, Lieberman PM. Telomeric repeat-containing RNA (TERRA) constitutes a nucleoprotein component of extracellular inflammatory exosomes. Proc Natl Acad Sci USA. 2015; 112:E6293–300. https://doi.org/10.1073/pnas.1505962112 [PubMed]
- 25. Arnoult N, Van Beneden A, Decottignies A. Telomere length regulates TERRA levels through increased trimethylation of telomeric H3K9 and HP1α. Nat Struct Mol Biol. 2012; 19:948–56. https://doi.org/10.1038/nsmb.2364 [PubMed]
- 26. Lou Z, Wei J, Riethman H, Baur JA, Voglauer R, Shay JW, Wright WE. Telomere length regulates ISG15 expression in human cells. Aging (Albany NY). 2009; 1:608–21. https://doi.org/10.18632/aging.100066 [PubMed]
- 27. Aubert G, Hills M, Lansdorp PM. Telomere length measurement-caveats and a critical assessment of the available technologies and tools. Mutat Res. 2012; 730:59–67. https://doi.org/10.1016/j.mrfmmm.2011.04.003 [PubMed]
- 28. Debacq-Chainiaux F, Erusalimsky JD, Campisi J, Toussaint O. Protocols to detect senescence-associated beta-galactosidase (SA-betagal) activity, a biomarker of senescent cells in culture and in vivo. Nat Protoc. 2009; 4:1798–806. https://doi.org/10.1038/nprot.2009.191 [PubMed]



