Introduction
Metabolic syndrome (MetS) is composed of a constellation of metabolic disorders, including hypertension, dyslipidemia, abdominal obesity and insulin resistance or glucose intolerance [1, 2]. In spite of the association between MetS and health problems, its influence on human reproductivity has yet to be discussed [3]. First, as the typical characteristics of MetS, obesity, dyslipidemia and insulin resistance are thought to have an adverse impact on female reproductivity for impaired endometrial receptivity and compromised embryo development [4]. Second, MetS has a relatively high coincidence of the endocrine syndrome of polycystic ovary syndrome (PCOS). Infertility and cardio-metabolic disorders are more common in PCOS, which has an incidence of 5~15% in female [5]. However, the study by Mulder et al. demonstrated that infertile women had a higher level of cholesterol, low density lipoprotein cholesterol, triglycerides and body mass index, but blood pressure, fasting insulin, insulin resistance and fasting glucose showed no significant difference between fertile and infertile women [5]. Considering MetS as a set of these clinical conditions, the results further confused us regarding the association between MetS and female reproductivity.
On the other hand, as the constituent part of MetS, obesity could dysregulate the sex hormones, and cause oxidative stress damage of the semen microenvironment, sperms and interstitial glands [6, 7]. However, the MacDonald et al. study suggested no significant impact of BMI on the semen parameters, while the Sermondade et al. study found a higher incidence of azoospermia or oligozoospermia in the overweight and obese cohort [8, 9]. Moreover, diabetes, hypertension and dyslipidemia could down-regulate the secretion of testosterone, damage the testiculus and erectile function, and finally affect male fertility [10–12].
As a composite syndrome, MetS is deduced in association with the development of human reproductivity in view of its constituent disorders. Previous studies demonstrated a negative correlation between MetS and blood testosterone levels, while it was positively associated with oestrogen levels [13, 14]. However, the impact of MetS on reproductive function and other sex hormones is still controversial, and has not been clearly illuminated. Thus, this meta-analysis aimed to clarify the impact of MetS on reproductive function and sex hormones.
Results
Characteristics of included studies
The literature search retrieved 12998 records: 7822 from PubMed, 5147 from Embase, and 29 from other sources (Figure 1). After removing the duplication and unrelated records, 18 records (21 studies) were included into the meta-analysis [15–32] (Supplementary Table 1). Finally, we included a total of 2923 MetS cases and 14062 controls. The Ehala-Aleksejev et al. study investigated the male partners of fertile and infertile couples respectively, while the Natah et al. study and the Olszanecka et al. study focused on the premenopausal and postmenopausal women respectively. Thus, these studies were internally divided into two individual studies. Moreover, 13 studies focused on male cohort, 8 on female cohort, 5 on infertile cohort, 4 on postmenopausal female cohort, and 2 on PCOS cohort. None of the studies were prospective designed. Ten studies were conducted in Europe, 8 in Asia and 3 in Africa. In the assessment of methodological quality, the included studies reached a mean NOS score of 6.58.
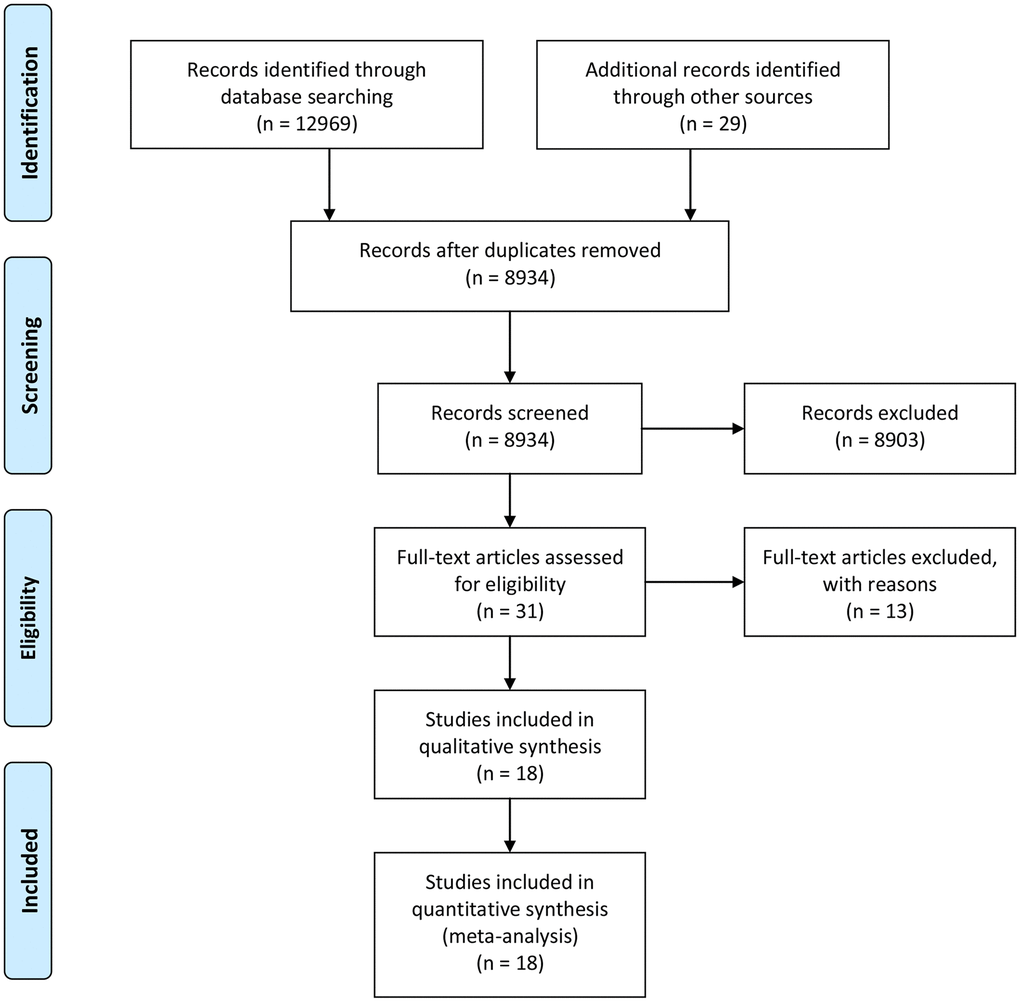
Figure 1. Flowchart of literature search.
Impact of MetS on sex hormones in males
Comparatively, MetS cases had a lower level of testosterone (T) (n=8; SMD (95% CI): -5.00 (-8.48 to -1.52)) and inhibin B (InhB) (n=4; SMD (95% CI): -2.95 (-5.19 to -0.71)). No significant difference was found in follicle-stimulating hormone (FSH) (n=8; SMD (95% CI): -0.37 (-0.89 to 0.15)), luteinizing hormone (LH) (n=7; SMD (95% CI): -0.16 (-2.23 to 1.92)), oestradiol (E2) (n=5; SMD (95% CI): 0.62 (-2.09 to 3.33)), prolactin (PRL) (n=3; SMD (95% CI): 0.04 (-0.11 to 0.20)), and anti-Müllerian hormone (AMH) (n=2; SMD (95% CI): -0.92 (-2.06 to 0.22)) (Table 1). No obvious publication bias was detected.
Table 1. Meta-analyses of the impact of metabolic syndrome on peripheral sex hormones and reproductive function.
| Variables | No. of included studies | No. of cases | SMD (95% CI) | I2 (%) | P in Egger's test | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MetS | Control | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Follicle-stimulating hormone (FSH) | 8 | 1064 | 4339 | -0.37 (-0.89 to 0.15) | 97 | 0.432 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Testosterone (T) | 8 | 1064 | 4339 | -5.00 (-8.48 to -1.52) | 100 | 0.102 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Luteinizing hormone (LH) | 7 | 1014 | 4309 | -0.16 (-2.23 to 1.92) | 100 | 0.192 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oestradiol (E2) | 5 | 695 | 3848 | 0.62 (-2.09 to 3.33) | 100 | 0.308 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Prolactin (PRL) | 3 | 175 | 1680 | 0.04 (-0.11 to 0.20) | 0 | 0.760 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Inhibin B (InhB) | 4 | 245 | 1498 | -2.95 (-5.19 to -0.71) | 99 | 0.091 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Anti-Müllerian hormone (AMH) | 2 | 148 | 1356 | -0.92 (-2.06 to 0.22) | 95 | - | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Semen volume | 11 | 891 | 4339 | -0.51 (-2.18 to 1.16) | 100 | 0.097 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total sperm count | 6 | 729 | 3766 | -0.66 (-1.26 to -0.05) | 97 | 0.461 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sperm concentration | 12 | 1776 | 11849 | -0.85 (-1.55 to -0.16) | 99 | 0.298 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sperm normal morphology | 10 | 1717 | 11778 | -0.56 (-0.93 to -0.19) | 97 | 0.418 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sperm total motility | 7 | 1536 | 10097 | -0.68 (-1.30 to -0.05) | 99 | 0.618 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sperm progressive motility | 10 | 1276 | 9469 | -0.54 (-0.91 to -0.17) | 94 | 0.089 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sperm vitality | 4 | 138 | 217 | -0.78 (-1.00 to -0.55) | 0 | 0.123 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DNA fragmentation | 4 | 128 | 197 | 0.69 (0.46 to 0.93) | 0 | 0.304 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mitochondrial membrane potential (MMP) | 2 | 54 | 55 | 0.89 (0.49 to 1.28) | 0 | - | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oestradiol (E2) | 8 | 853 | 2071 | 0.04 (-0.19 to 0.28) | 80 | 0.399 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Follicle-stimulating hormone (FSH) | 8 | 853 | 2071 | -0.20 (-0.48 to 0.07) | 86 | 0.300 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Luteinizing hormone (LH) | 5 | 631 | 1820 | 0.03 (-0.28 to 0.34) | 82 | 0.482 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Progesterone (P) | 1 | 159 | 620 | -0.13 (-0.30 to 0.05) | - | - | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thyroid-stimulating hormone (TSH) | 2 | 558 | 1270 | 0.22 (-0.03 to 0.48) | 76 | - | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Testosterone (T) | 4 | 642 | 1799 | 0.14 (0.02 to 0.26) | 23 | 0.412 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MetS, metabolic syndrome; No., number; SMD, standardized mean difference; CI, confidence interval. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Impact of MetS on semen quality in males
Comparatively, MetS cases had a lower level of sperm total count (n=6; SMD (95% CI): -0.66 (-1.26 to -0.05)), sperm concentration (n=12; SMD (95% CI): -0.85 (-1.55 to -0.16)), normal sperm morphology (n=10; SMD (95% CI): -0.56 (-0.93 to -0.19)), total sperm motility (n=7; SMD (95% CI): -0.68 (-1.30 to -0.05)), progressive sperm motility (n=10; SMD (95% CI): -0.54 (-0.91 to -0.17)), and sperm vitality (n=4; SMD (95% CI): -0.78 (-1.00 to -0.55)) (Table 1). MetS cases had a higher level of deoxyribonucleic acid (DNA) fragmentation (n=4; SMD (95% CI): 0.69 (0.46 to 0.93)), and mitochondrial membrane potential (MMP) (n=2; SMD (95% CI): 0.89 (0.49 to 1.28)). There found no obvious difference in semen volume (n=11; SMD (95% CI): -0.51 (-2.18 to 1.16)). No obvious publication bias was detected.
Impact of MetS on sex hormones in females
Comparatively, MetS cases had a higher level of T (n=4; SMD (95% CI): 0.14 (0.02 to 0.26)) (Table 1). There found no obvious difference in E2 (n=8; SMD (95% CI) 0.04 (-0.19 to 0.28)), FSH (n=8; SMD (95% CI): -0.20 (-0.48 to 0.07)), LH (n=5; SMD (95% CI): 0.03 (-0.28 to 0.34)), progesterone (P) (n=1; SMD (95% CI): -0.13 (-0.30 to 0.05)), and thyroid-stimulating hormone (TSH) (n=2; SMD (95% CI): 0.22 (-0.03 to 0.48)). No obvious publication bias was detected.
Discussion
Infertility is characterized by the failure to conceive after more than one year’s unprotected intercourse [33]. Approximately, one in six couples suffers from infertility across the world, which also leads to a decrease in life quality [34]. Among the infertile couples, 50% was caused by the female-related factors, while 20% by male-related factors and 30% by unexplained factors [35].
Recently, the pathogenesis of female infertility is considered not limited to the endocrine or reproductive systems. Furthermore, female infertility have been found to coincide with several metabolic disorders [36]. Infertile females have a higher incidence of hyperlipemia, and are at high risk of pregnant hypertension after medically assisted reproduction [37]. The mechanism is still not illuminated. The Banuls et al. study found an increasing level of lipolysis in the follicular fluid of MetS patients, as well as an elevated level of lower high-density lipoprotein, triglyceride and cholesterol. This might lead to the alteration in embryo development [38]. Growing evidence suggests an association between MetS and female infertility. However, it is confused by the limitation in sample size and the inconsistency in measuring methods. In this meta-analysis, we only found a higher level of testosterone in MetS women. High testosterone level has been reported in association with female infertility [39]. Due to the lack of relevant studies, we failed to investigate the impact of MetS on female reproductive cells.
Male hypogonadism usually accompanies with testosterone deficiency, spermatogenesis impairment, and metabolic disorders like diabetes and obesity [40]. In recent years, it is classified into “organic hypogonadism” and “functional hypogonadism” [41]. Organic hypogonadism is characterized by an irreversible pathological impairment, and testosterone replacement therapy is considered low to the benefits. By far, functional hypogonadism is the most common form of hypogonadism in adulthood, characterized by no recognizable pathological alteration in the reproductive system. Metabolic disorders are common among the individuals with hypogonadotropic hypogonadism or late-onset hypogonadism, and thus obesity, diabetes and MetS are hypothesized in association with sexual dysfunction [40]. Both the reproductive parameters and metabolic profiles could benefit from the testosterone supplementation treatment for hypogonadism or insulin sensitizer administration for the concomitant metabolic disorders [41]. These indicate a potential association between hypogonadism and MetS, but previous studies reached inconsistent results, probably caused by the limitation in sample size. Thus, this meta-analysis aimed to investigate the impact of MetS on male reproduction. Finally, we found that MetS patients had a significant lower level of several semen parameters, circulating FSH, T and inhibin B, while they had a higher level of MMP and DNA fragmentation in sperms.
The mechanism is complicated, and might attribute to the superimposed effects of the component factors of MetS, especially overweight and diabetes. Several studies have reported the association between overweight and the abnormalities in sex hormone levels or semen quality [9]. The DNA fragmentation in sperms was more common in obese males than normal-weight males [42]. Furthermore, overweight and obese individuals had an increasing level of oxidative stress and inflammation, which disturbed the spermatogenesis [43]. On the other hand, diabetes also demonstrated an association with the abnormalities in sex hormone levels and semen quality [44]. Metformin treatment could significantly improve the sperm concentration, the percentage of motile cells, the percentage of normal cells, LH and testosterone levels in the men suffering from type 2 diabetes mellitus [45, 46]. Thus, as the syndrome of these metabolic disorders, MetS was deduced in association with the development of infertility.
Despite of the first meta-analysis in this field, the limitations should not be ignored. First, partial included studies failed to include a large study cohort. Second, none of the included studies were prospective designed. Third, there found significant heterogeneity among studies, which probably attributed to the study-specific heterogeneity caused by sampling errors. Nevertheless, these limitations could not prevent us from arousing people’s attention to reproductive health especially among those with MetS.
Conclusively, this study indicated the impact of MetS on sex hormones and reproductive function, and MetS cases had a potential risk of infertility.
Materials and Methods
Literature search
Public databases of Embase and PubMed were used to retrieve related studies from inception to July 16th, 2020. The combined key words were used: (‘metabolic cardiovascular syndrome’ OR ‘dysmetabolic syndrome’ OR ‘metabolic X syndrome’ OR ‘reaven syndrome’ OR ‘insulin resistance syndrome’ OR ‘syndrome X’ OR ‘metabolic syndrome’) AND (’follicle-stimulating hormone‘ OR ’testosterone‘ OR ’luteinizing hormone‘ OR ’oestradiol‘ OR ’prolactin‘ OR ’inhibin B‘ OR ’anti-Müllerian hormone‘ OR ’progesterone‘ OR ’thyroid-stimulating hormone‘ OR ’semen‘ OR ’sperm‘ OR ‘azoospermia’ OR ‘oligozoospermia’ OR ’ovum‘ OR ’germ cell‘ OR ’follicle’). We only included the studies published in English. Moreover, the references of relative publications were also reviewed for potential studies. Our meta-analysis has been authorized by the ethics committee of The First Affiliated Hospital of Harbin Medical University.
Study inclusion
Lihong Zhou and Liou Han selected the studies seperatively. Studies were included if fulfilling the following criteria: (i) contained both MetS individuals and the controls; (ii) focused on any indicators of sex hormones, semen or ovum parameters; (iii) the indicator levels were provided. We excluded animal studies, case reports, reviews and abstracts without full texts.
Data extraction
Lihong Zhou and Liou Han designed a standardized collection form to extract the data, and the authors resolved the differences by discussion. The following data were extracted from each included study: publication year, author, study location, study population, MetS diagnosis criteria, sample size, and measurement parameters. If the studies originated from the same location, the study area and duration were reviewed to remove duplication. In quality assessment of the included studies, we adopted the Newcastle-Ottawa Scale (NOS, 0~9).
Statistical analysis
Some studies presented the parameters as the median or average with standard error (SD), inter-quartile range (IQR), range or 95% confidence interval (95% CI). Uniformly, we converted the parameters to average ± SD according to the previous method [47]. In the meta-analysis of the impact of MetS on selected parameters, the inverse variance method and the random-effects model were adopted to pool the standardized mean differences (SMD) with 95% CIs. Q test and I2 statistic were used to estimate the heterogeneity among studies, and the I2 of more than 50% was considered significant heterogeneity [48]. Egger’s test was conducted to evaluate potential publication bias [49]. Most statistical analyses were realized with the software of Review Manager 5.4, only Egger’s test was conducted using the software of STATA version 11.0. A two-tailed P value of less than 0.05 was considered statistically significant.
Supplementary Materials
Author Contributions
LZ and LH conceived and designed the study, which were proofed by LZ, LH and ML. LZ, JL and SP analyzed the data. LZ and LH wrote the manuscript. All authors read and approved the manuscript.
Conflicts of Interest
All authors declare that they have no conflicts of interest.
Funding
Granted by Scientific Research Project of the Health Commission of Heilongjiang Province (2019-025), and Research and Innovation Fund of The First Affiliated Hospital of Harbin Medical University (2017 Y012).
References
- 1. Kurl S, Laaksonen DE, Jae SY, Mäkikallio TH, Zaccardi F, Kauhanen J, Ronkainen K, Laukkanen JA. Metabolic syndrome and the risk of sudden cardiac death in middle-aged men. Int J Cardiol. 2016; 203:792–97. https://doi.org/10.1016/j.ijcard.2015.10.218 [PubMed]
- 2. Kaneko K, Yatsuya H, Li Y, Uemura M, Chiang C, Hirakawa Y, Ota A, Tamakoshi K, Aoyama A. Risk and population attributable fraction of metabolic syndrome and impaired fasting glucose for the incidence of type 2 diabetes mellitus among middle-aged Japanese individuals: Aichi Worker's Cohort Study. J Diabetes Investig. 2020; 11:1163–69. https://doi.org/10.1111/jdi.13230 [PubMed]
- 3. Greenfield DM, Snowden JA. Cardiovascular Diseases and Metabolic Syndrome. In: Carreras E, Dufour C, Mohty M, Kröger N, editors. The EBMT Handbook: Hematopoietic Stem Cell Transplantation and Cellular Therapies. ed. Cham (CH): Springer. 2019. Chapter 55. https://doi.org/10.1007/978-3-030-02278-5_55 [PubMed]
- 4. Cardozo E, Pavone ME, Hirshfeld-Cytron JE. Metabolic syndrome and oocyte quality. Trends Endocrinol Metab. 2011; 22:103–9. https://doi.org/10.1016/j.tem.2010.12.002 [PubMed]
- 5. Mulder CL, Lassi ZS, Grieger JA, Ali A, Jankovic-Karasoulos T, Roberts CT, Andraweera PH. Cardio-metabolic risk factors among young infertile women: a systematic review and meta-analysis. BJOG. 2020; 127:930–39. https://doi.org/10.1111/1471-0528.16171 [PubMed]
- 6. Brand JS, Rovers MM, Yeap BB, Schneider HJ, Tuomainen TP, Haring R, Corona G, Onat A, Maggio M, Bouchard C, Tong PC, Chen RY, Akishita M, et al. Testosterone, sex hormone-binding globulin and the metabolic syndrome in men: an individual participant data meta-analysis of observational studies. PLoS One. 2014; 9:e100409. https://doi.org/10.1371/journal.pone.0100409 [PubMed]
- 7. Benedetti S, Tagliamonte MC, Catalani S, Primiterra M, Canestrari F, De Stefani S, Palini S, Bulletti C. Differences in blood and semen oxidative status in fertile and infertile men, and their relationship with sperm quality. Reprod Biomed Online. 2012; 25:300–06. https://doi.org/10.1016/j.rbmo.2012.05.011 [PubMed]
- 8. MacDonald AA, Herbison GP, Showell M, Farquhar CM. The impact of body mass index on semen parameters and reproductive hormones in human males: a systematic review with meta-analysis. Hum Reprod Update. 2010; 16:293–311. https://doi.org/10.1093/humupd/dmp047 [PubMed]
- 9. Sermondade N, Faure C, Fezeu L, Shayeb AG, Bonde JP, Jensen TK, Van Wely M, Cao J, Martini AC, Eskandar M, Chavarro JE, Koloszar S, Twigt JM, et al. BMI in relation to sperm count: an updated systematic review and collaborative meta-analysis. Hum Reprod Update. 2013; 19:221–31. https://doi.org/10.1093/humupd/dms050 [PubMed]
- 10. Colli LG, Belardin LB, Echem C, Akamine EH, Antoniassi MP, Andretta RR, Mathias LS, Rodrigues SF, Bertolla RP, de Carvalho MH. Systemic arterial hypertension leads to decreased semen quality and alterations in the testicular microcirculation in rats. Sci Rep. 2019; 9:11047. https://doi.org/10.1038/s41598-019-47157-w [PubMed]
- 11. Hagiuda J, Ishikawa H, Furuuchi T, Hanawa Y, Marumo K. Relationship between dyslipidaemia and semen quality and serum sex hormone levels: an infertility study of 167 Japanese patients. Andrologia. 2014; 46:131–35. https://doi.org/10.1111/and.12057 [PubMed]
- 12. Lu X, Huang Y, Zhang H, Zhao J. Effect of diabetes mellitus on the quality and cytokine content of human semen. J Reprod Immunol. 2017; 123:1–2. https://doi.org/10.1016/j.jri.2017.08.007 [PubMed]
- 13. Maggio M, Lauretani F, Ceda GP, Bandinelli S, Basaria S, Paolisso G, Giumelli C, Luci M, Najjar SS, Metter EJ, Valenti G, Guralnik J, Ferrucci L. Estradiol and metabolic syndrome in older Italian men: the InCHIANTI study. J Androl. 2010; 31:155–62. https://doi.org/10.2164/jandrol.108.006098 [PubMed]
- 14. Corona G, Mannucci E, Schulman C, Petrone L, Mansani R, Cilotti A, Balercia G, Chiarini V, Forti G, Maggi M. Psychobiologic correlates of the metabolic syndrome and associated sexual dysfunction. Eur Urol. 2006; 50:595–604. https://doi.org/10.1016/j.eururo.2006.02.053 [PubMed]
- 15. Leisegang K, Udodong A, Bouic PJ, Henkel RR. Effect of the metabolic syndrome on male reproductive function: a case-controlled pilot study. Andrologia. 2014; 46:167–76. https://doi.org/10.1111/and.12060 [PubMed]
- 16. Chen YY, Kao TW, Peng TC, Yang HF, Wu CJ, Chen WL. Metabolic syndrome and semen quality in adult population. J Diabetes. 2020; 12:294–304. https://doi.org/10.1111/1753-0407.12995 [PubMed]
- 17. Pilatz A, Hudemann C, Wolf J, Halefeld I, Paradowska-Dogan A, Schuppe HC, Hossain H, Jiang Q, Schultheiss D, Renz H, Weidner W, Wagenlehner F, Linn T. Metabolic syndrome and the seminal cytokine network in morbidly obese males. Andrology. 2017; 5:23–30. https://doi.org/10.1111/andr.12296 [PubMed]
- 18. Ventimiglia E, Capogrosso P, Colicchia M, Boeri L, Serino A, Castagna G, Clementi MC, La Croce G, Regina C, Bianchi M, Mirone V, Damiano R, Montorsi F, Salonia A. Metabolic syndrome in white European men presenting for primary couple's infertility: investigation of the clinical and reproductive burden. Andrology. 2016; 4:944–51. https://doi.org/10.1111/andr.12232 [PubMed]
- 19. Ventimiglia E, Capogrosso P, Serino A, Boeri L, Colicchia M, La Croce G, Scano R, Papaleo E, Damiano R, Montorsi F, Salonia A. Metabolic syndrome in White-European men presenting for secondary couple’s infertility: an investigation of the clinical and reproductive burden. Asian J Androl. 2017; 19:368–73. https://doi.org/10.4103/1008-682X.175783 [PubMed]
- 20. Leisegang K, Bouic PJ, Henkel RR. Metabolic syndrome is associated with increased seminal inflammatory cytokines and reproductive dysfunction in a case-controlled male cohort. Am J Reprod Immunol. 2016; 76:155–63. https://doi.org/10.1111/aji.12529 [PubMed]
- 21. Lotti F, Corona G, Degli Innocenti S, Filimberti E, Scognamiglio V, Vignozzi L, Forti G, Maggi M. Seminal, ultrasound and psychobiological parameters correlate with metabolic syndrome in male members of infertile couples. Andrology. 2013; 1:229–39. https://doi.org/10.1111/j.2047-2927.2012.00031.x [PubMed]
- 22. Saikia UK, Saikia K, Sarma D, Appaiah S. Sertoli cell function in young males with metabolic syndrome. Indian J Endocrinol Metab. 2019; 23:251–56. https://doi.org/10.4103/ijem.IJEM_574_18 [PubMed]
- 23. Elsamanoudy AZ, Abdalla HA, Hassanien M, Gaballah MA. Spermatozoal cell death-inducing DNA fragmentation factor-α-like effector A (CIDEA) gene expression and DNA fragmentation in infertile men with metabolic syndrome and normal seminogram. Diabetol Metab Syndr. 2016; 8:76. https://doi.org/10.1186/s13098-016-0192-y [PubMed]
- 24. Ehala-Aleksejev K, Punab M. The effect of metabolic syndrome on male reproductive health: a cross-sectional study in a group of fertile men and male partners of infertile couples. PLoS One. 2018; 13:e0194395. https://doi.org/10.1371/journal.pone.0194395 [PubMed]
- 25. Siddiqui K, Al-Rubeaan K, Nawaz SS, Aburisheh KH, Alaabdin AM, Tolba IA. Serum sex hormone binding globulin (SHBG) relation with different components of metabolic syndrome in men with type 2 diabetes. Horm Metab Res. 2018; 50:138–44. https://doi.org/10.1055/s-0043-123348 [PubMed]
- 26. Elfassy Y, Bongrani A, Levy P, Foissac F, Fellahi S, Faure C, McAvoy C, Capeau J, Dupont J, Fève B, Levy R, Bastard JP, and Metasperme group. Relationships between metabolic status, seminal adipokines, and reproductive functions in men from infertile couples. Eur J Endocrinol. 2020; 182:67–77. https://doi.org/10.1530/EJE-19-0615 [PubMed]
- 27. Stefanska A, Sypniewska G, Ponikowska I, Cwiklinska-Jurkowska M. Association of follicle-stimulating hormone and sex hormone binding globulin with the metabolic syndrome in postmenopausal women. Clin Biochem. 2012; 45:703–06. https://doi.org/10.1016/j.clinbiochem.2012.03.011 [PubMed]
- 28. Güdücü N, Görmüş U, Kutay SS, Kavak ZN, Telatar B. Endogenous sex hormones and their associations with cardiovascular risk factors in post-menopausal women. J Endocrinol Invest. 2013; 36:588–92. https://doi.org/10.3275/8881 [PubMed]
- 29. Olszanecka A, Kawecka-Jaszcz K, Czarnecka D. Association of free testosterone and sex hormone binding globulin with metabolic syndrome and subclinical atherosclerosis but not blood pressure in hypertensive perimenopausal women. Arch Med Sci. 2016; 12:521–28. https://doi.org/10.5114/aoms.2016.59925 [PubMed]
- 30. He Y, Lu Y, Zhu Q, Wang Y, Lindheim SR, Qi J, Li X, Ding Y, Shi Y, Wei D, Chen ZJ, Sun Y. Influence of metabolic syndrome on female fertility and in vitro fertilization outcomes in PCOS women. Am J Obstet Gynecol. 2019; 221:138.e1–e12. https://doi.org/10.1016/j.ajog.2019.03.011 [PubMed]
- 31. Chang H, Xie L, Ge H, Wu Q, Wen Y, Zhang D, Zhang Y, Ma H, Gao J, Wang CC, Stener-Victorin E, Ng EH, Wu X. Effects of hyperhomocysteinaemia and metabolic syndrome on reproduction in women with polycystic ovary syndrome: a secondary analysis. Reprod Biomed Online. 2019; 38:990–98. https://doi.org/10.1016/j.rbmo.2018.12.046 [PubMed]
- 32. Natah TM. Serum Adiponectin Levels in Pre-Postmenopausal Metabolic Syndrome Women and the Correlation with Some Physio-Biochemical Parameters. Australian Journal of Basic and Applied Sciences. 2014; 8:360–66.
- 33. Zegers-Hochschild F, Adamson GD, Dyer S, Racowsky C, de Mouzon J, Sokol R, Rienzi L, Sunde A, Schmidt L, Cooke ID, Simpson JL, van der Poel S. The International Glossary on Infertility and Fertility Care, 2017. Fertil Steril. 2017; 108:393–406. https://doi.org/10.1016/j.fertnstert.2017.06.005 [PubMed]
- 34. Baldur-Felskov B, Kjaer SK, Albieri V, Steding-Jessen M, Kjaer T, Johansen C, Dalton SO, Jensen A. Psychiatric disorders in women with fertility problems: results from a large Danish register-based cohort study. Hum Reprod. 2013; 28:683–90. https://doi.org/10.1093/humrep/des422 [PubMed]
- 35. Evers JL. Female subfertility. Lancet. 2002; 360:151–59. https://doi.org/10.1016/S0140-6736(02)09417-5 [PubMed]
- 36. Hanson B, Johnstone E, Dorais J, Silver B, Peterson CM, Hotaling J. Female infertility, infertility-associated diagnoses, and comorbidities: a review. J Assist Reprod Genet. 2017; 34:167–77. https://doi.org/10.1007/s10815-016-0836-8 [PubMed]
- 37. Chen XK, Wen SW, Bottomley J, Smith GN, Leader A, Walker MC. In vitro fertilization is associated with an increased risk for preeclampsia. Hypertens Pregnancy. 2009; 28:1–12. https://doi.org/10.1080/10641950802001859 [PubMed]
- 38. Bañuls C, Rovira-Llopis S, Martinez de Marañon A, Veses S, Jover A, Gomez M, Rocha M, Hernandez-Mijares A, Victor VM. Metabolic syndrome enhances endoplasmic reticulum, oxidative stress and leukocyte-endothelium interactions in PCOS. Metabolism. 2017; 71:153–162. https://doi.org/10.1016/j.metabol.2017.02.012 [PubMed]
- 39. Almalki HH, Alshibani TM, Alhifany AA, Almohammed OA. Comparative efficacy of statins, metformin, spironolactone and combined oral contraceptives in reducing testosterone levels in women with polycystic ovary syndrome: a network meta-analysis of randomized clinical trials. BMC Womens Health. 2020; 20:68. https://doi.org/10.1186/s12905-020-00919-5 [PubMed]
- 40. Pivonello R, Menafra D, Riccio E, Garifalos F, Mazzella M, de Angelis C, Colao A. Metabolic Disorders and Male Hypogonadotropic Hypogonadism. Front Endocrinol (Lausanne). 2019; 10:345. https://doi.org/10.3389/fendo.2019.00345 [PubMed]
- 41. Corona G, Rastrelli G, Reisman Y, Sforza A, Maggi M. The safety of available treatments of male hypogonadism in organic and functional hypogonadism. Expert Opin Drug Saf. 2018; 17:277–92. https://doi.org/10.1080/14740338.2018.1424831 [PubMed]
- 42. Sepidarkish M, Maleki-Hajiagha A, Maroufizadeh S, Rezaeinejad M, Almasi-Hashiani A, Razavi M. The effect of body mass index on sperm DNA fragmentation: a systematic review and meta-analysis. Int J Obes (Lond). 2020; 44:549–58. https://doi.org/10.1038/s41366-020-0524-8 [PubMed]
- 43. Pini T, Parks J, Russ J, Dzieciatkowska M, Hansen KC, Schoolcraft WB, Katz-Jaffe M. Obesity significantly alters the human sperm proteome, with potential implications for fertility. J Assist Reprod Genet. 2020; 37:777–787. https://doi.org/10.1007/s10815-020-01707-8 [PubMed]
- 44. Pergialiotis V, Prodromidou A, Frountzas M, Korou LM, Vlachos GD, Perrea D. Diabetes mellitus and functional sperm characteristics: A meta-analysis of observational studies. J Diabetes Complications. 2016; 30:1167–76. https://doi.org/10.1016/j.jdiacomp.2016.04.002 [PubMed]
- 45. Morgante G, Tosti C, Orvieto R, Musacchio MC, Piomboni P, De Leo V. Metformin improves semen characteristics of oligo-terato-asthenozoospermic men with metabolic syndrome. Fertil Steril. 2011; 95:2150–52. https://doi.org/10.1016/j.fertnstert.2010.12.009 [PubMed]
- 46. Bosman E, Esterhuizen AD, Rodrigues FA, Becker PJ, Hoffmann WA. Effect of metformin therapy and dietary supplements on semen parameters in hyperinsulinaemic males. Andrologia. 2015; 47:974–79. https://doi.org/10.1111/and.12366 [PubMed]
- 47. Wan X, Wang W, Liu J, Tong T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol. 2014; 14:135. https://doi.org/10.1186/1471-2288-14-135 [PubMed]
- 48. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003; 327:557–60. https://doi.org/10.1136/bmj.327.7414.557 [PubMed]
- 49. Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997; 315:629–34. https://doi.org/10.1136/bmj.315.7109.629 [PubMed]


