Introduction
Traditionally, in most common cancers, including breast cancer (BC), clinicopathological features (e.g., tumor size, lymph node status, TNM stage, histological grade, hormone receptor status, human epidermal growth factor receptor 2 [HER-2] amplification) are used to predict patient outcome [1]. Biomarkers such as tumor-associated macrophages (TAMs), microRNAs, matrix metalloproteinases (MMPs), retinoic acid receptor a (RARA), and Ki-67 are also useful in predicting prognosis of certain cancers (e.g., colon cancer, gastric cancer, acute promyelocytic leukemia) [2–6]. There has been increasing research focus on protein glycosylation and related glycosyltransferases as prognostic biomarkers in various human cancers [7–9].
Glycosylation (attachment of glycans to proteins or other organic molecules) is a common posttranslational modification in all organisms. Aberrant glycosylation is a characteristic phenomenon in carcinogenesis, plays essential roles in specific steps of tumor development [10], and directly promotes tumor progression and metastasis [11–17]. Glycosylation is mediated by enzymatic activities of glycosyltransferases and glycosidases in glycoproteins and/or lipids. Alterations of glycosyltransferase expression are associated with both pro-metastatic and metastasis-suppressing functions [12].
Fucosyltransferase 8 (FUT8) is an α(1,6)-fucosyltransferase responsible for addition of fucose to asparagine-linked N-acetylglucosamine (GlcNAc) moieties, a common feature of N-linked glycan core structures [18]. Aberrant fucosylated glycan structures and associated glycosyltransferases have been observed in development of various cancers [19]; e.g., increased core fucosylation in BC [20], non-small cell lung cancer (NSCLC) [21], ovarian cancer [22], colon cancer [23], prostate cancer [24], and melanoma [25]. A positive feedback mechanism of FUT8-mediated receptor core fucosylation was recently shown to enhance TGF-β signaling and epithelial-mesenchymal transition (EMT), thus promoting BC cell invasion and metastasis; such fucosylation in BC patients is a potential diagnostic/ prognostic biomarker, or therapeutic target [26]. In BC patients, high FUT8 protein expression is correlated with lymphatic metastasis and stage status, whereas reduced FUT8 expression is correlated with disease-free survival and overall survival [20].
FUT8 expression level has been linked to tumor clinical features and outcomes in numerous studies. However, few attempts have been made to systematically evaluate such associations. We performed a systematic review and meta-analysis based on collection of references and Gene Expression Omnibus (GEO) microarray data, to clarify the correlations between FUT8 expression and clinical pathology and patient survival in various common types of cancer.
Results
Characteristics of studies included in the meta-analysis
A total of 366 articles were found through database mining and manual searches (see M&M); these comprised 152 articles directly related to FUT8, and 214 articles that included microarray data and were indirectly related to FUT8. After screening out duplicate titles and abstracts, 27 full-text articles and 78 datasets remained; of these, 20 articles and 43 datasets were excluded on the basis of criteria described in M&M, finally leaving 7 articles and 35 microarray datasets (involving 6124 patients) for inclusion in the meta-analysis. Characteristics and quality scores of the included studies are summarized in Table 1. Among these, the 35 datasets involved 7 types of malignant tumors and descriptions of 29 types of clinicopathological features related to FUT8, and the 7 articles involved 5 types of malignant tumors and described correlations between FUT8 expression and patient survival.
Table 1. Characteristics of studies included in the meta-analysis.
| Tumor source | First author and year | Country | Ethnicity | Number of patients | Sample Type | Method | GEO ID | Cutoff | Survival | Follow-up (Months) | Quality Score | Reference | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Breast Cancer | Lasham 2012 | New Zealand | Caucasian | 107 | tissue | Microarray | gse36771 | average | NR | NR | 7 | [49] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chin 2006 | America | Caucasian | 130 | tissue | Microarray | gse69031 | average | NR | NR | 7 | [50] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Desmedt 2009 | Belgium | Caucasian | 55 | tissue | Microarray | gse16391 | average | NR | NR | 6 | [51] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| EXPO 2005 | America | Caucasian | 351 | tissue | Microarray | gse2109 | average | NR | NR | 8 | R2 platform | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lu 2008 | America | Caucasian | 123 | tissue | Microarray | gse5460 | average | NR | NR | 6 | [52] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Concha 2011 | Spain | Caucasian | 66 | tissue | Microarray | gse29431 | average | NR | NR | 7 | R2 platform | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Iwamoto 2011 | America | Caucasian | 103 | tissue | Microarray | gse22093 | average | NR | NR | 7 | [53] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yue 2016 | China | Asian | 189 | tissue | IHC | median | OS, DFS | 72 | 7 | [20] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Colorectal Cancer | EXPO 2005 | America | Caucasian | 315 | tissue | Microarray | gse2109 | average | NR | NR | 7 | R2 platform | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Laibe 2012 | France | Caucasian | 130 | tissue | Microarray | gse37892 | average | NR | NR | 7 | [54] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Jorissen 2009 | Australia | Caucasian | 290 | tissue | Microarray | gse14333 | average | NR | NR | 8 | [55] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smith 2010 | America | Caucasian | 232 | tissue | Microarray | gse17538 | average | NR | NR | 7 | [56] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Watanabe 2006 | Japan | Asian | 84 | tissue | Microarray | gse4554 | average | NR | NR | 5 | [57] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tsukamoto 2011 | Japan | Asian | 148 | tissue | Microarray | gse21510 | average | NR | NR | 7 | [58] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Jorissen 2008 | Denmark | Caucasian | 155 | tissue | Microarray | gse13294 | average | NR | NR | 6 | [59] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Schlicker 2012 | United Kingdom | Caucasian | 62 | tissue | Microarray | gse35896 | average | NR | NR | 5 | [60] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Barras 2017 | Australia | Caucasian | 59 | tissue | Microarray | gse75316 | average | NR | NR | 5 | [61] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ependymoma | Donson 2009 | America | Caucasian | 19 | tissue | Microarray | gse16155 | average | NR | NR | 7 | [62] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Johnson 2010 | America | Caucasian | 83 | tissue | Microarray | NR | average | NR | NR | 7 | [63] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hoffman 2014 | America | Caucasian | 65 | tissue | Microarray | gse50385 | average | NR | NR | 7 | [64] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vladoiu 2019 | Germany | Caucasian | 209 | tissue | Microarray | gse64415 | average | NR | NR | 8 | [65] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Glioma | Freije 2004 | America | Caucasian | 85 | tissue | Microarray | gse4412 | average | NR | NR | 6 | [66] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gravendeel 2009 | Netherlands | Caucasian | 276 | tissue | Microarray | gse16011 | average | OS | 240 | 7 | [67] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kawaguchi 2013 | Japan | Asian | 50 | tissue | Microarray | gse43378 | average | NR | NR | 6 | [68] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Zhang 2014 | America | Caucasian | 21 | tissue | Microarray | gse50774 | average | NR | NR | 6 | [69] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-Small Cell Cancer | Tarca 2013 | Switzerland | Caucasian | 150 | tissue | Microarray | gse43580 | average | NR | NR | 7 | [70] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Muley 2014 | Germany | Caucasian | 100 | tissue | Microarray | gse33532 | average | NR | NR | 8 | R2 platform | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Honma 2015 | Japan | Asian | 129 | tissue | IHC | median | OS | 168 | 7 | [71] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chen 2013 | China | Asian | 140 | tissue | IHC | median | OS, DFS | 120 | 7 | [21] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Wu 2019 | China | Asian | 135 | tissue | IHC | median | OS, DFS | 60 | 7 | [72] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Park 2020 | Korea | Asian | 217 | tissue | Microarray | gse31210 | median | DFS | 120 | 8 | [73] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Medulloblastoma | Robinson 2012 | America | Caucasian | 76 | tissue | Microarray | gse37418 | average | NR | NR | 6 | [74] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Northcott 2017 | Germany | Caucasian | 223 | tissue | Microarray | NR | average | NR | NR | 7 | [75] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kool 2008 | Netherlands | Caucasian | 62 | tissue | Microarray | gse10327 | average | NR | NR | 7 | [76] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Delattre 2012 | NR | NR | 57 | tissue | Microarray | NR | average | NR | NR | 6 | R2 platform | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Neuroblastoma | Delattre 2009 | France | Caucasian | 34 | tissue | Microarray | gse14880 | average | NR | NR | 5 | R2 platform | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ohtaki 2010 | Japan | Asian | 51 | tissue | Microarray | gse16237 | average | NR | NR | 6 | [77] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lastowska 2007 | United Kingdom | Caucasian | 30 | tissue | Microarray | gse13136 | average | NR | NR | 5 | [78] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molenaar 2012 | Netherlands | Caucasian | 88 | tissue | Microarray | gse16476 | average | NR | NR | 7 | [79] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pancreatic Ductal Adenocarcinoma | Tada 2019 | Japan | Asian | 62 | tissue | IHC | median | RFS | 120 | 7 | [80] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diffuse Large B Cell Lymphoma | Xiao 2008 | America | Caucasian | 420 | tissue | Microarray | gse10846 | average | OS | 240 | 8 | R2 platform | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gastric Cancer | Tan 2018 | Switzerland | Caucasian | 192 | tissue | Microarray | gse15459 | average | OS | 60 | 7 | R2 platform | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OS, overall survival; DFS, disease-free survival; RFS, relapse-free survival; IHC, immunohistochemistry; NR, not reported. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Association of FUT8 expression with clinicopathological features of various types of malignant tumors
Seven malignant tumors (ependymoma, glioma, BC, colorectal cancer (CRC), medulloblastoma (MBL), neuroblastoma (NB), NSCLC) were included in meta-analyses for clinicopathological features. Pooled results are presented in Supplementary Table 1. For BC, high FUT8 expression level was related to positive PR and positive ER status (odds ratio [OR]= 3.34, 95% confidence interval [CI]: 1.60-6.96, p= 0.001 and odds ratio [OR]= 7.42, 95% confidence interval [CI]: 2.94-18.7, p< 0.0001). FUT8 expression level was also correlated with tumor histological grade (OR= 2.55, 95% CI: 1.10-5.95, p= 0.03) (Figure 1A). For CRC, elevated FUT8 expression level was associated with TNM stage I-III (OR= 1.79, 95% CI: 1.09–2.95, p= 0.02), and was also related to microsatellite instability (MSI) (OR= 4.37, 95% CI: 2.65-7.20, p< 0.00001) and female gender (OR= 0.65, 95% CI: 0.50-0.83, p= 0.0007) (Figure 1B). For ependymoma, high FUT8 expression level was related to patient age (≤10 years) (OR= 3.69, 95% CI: 2.28-5.99, p< 0.00001) and tumor recurrence status (OR= 2.29, 95% CI: 1.07-4.93, p= 0.03) (Supplementary Figure 1A). For glioma, increased FUT8 level was associated with glioblastoma multiforme (GBM) (OR= 1.69, 95% CI: 1.07–2.66, p= 0.02), and FUT8 level was inversely correlated with patient age (≤40 years) (OR= 0.58, 95% CI: 0.35-0.95, p= 0.03) (Supplementary Figure 1B). For MBL, high FUT8 expression level was related to Wingless (WNT) (OR= 4.72, 95% CI: 1.99-11.22, p= 0.0004) and Sonic Hedgehog (SHH) molecular subgroups (OR= 12.11, 95% CI: 6.44-22.79, p< 0.00001). FUT8 level was inversely correlated with metastasis status (OR= 0.25, 95% CI: 0.10-0.60, p= 0.002) and male gender (OR= 0.60, 95% CI: 0.39-0.92, p= 0.02) (Figure 2A). For NSCLC, FUT8 expression level was inversely correlated with N0 status of TNM stage (OR= 0.58, 95% CI: 0.38-0.88, p= 0.01) and male gender (OR= 0.34, 95% CI: 0.18-0.68, p= 0.002) (Figure 2B). Clinicopathological features of malignant tumors related to FUT8 are summarized in Table 2. High FUT8 expression level in NB showed no association with any clinicopathological feature, including INSS stage, gender, or mycn amplified status (Supplementary Figure 2).
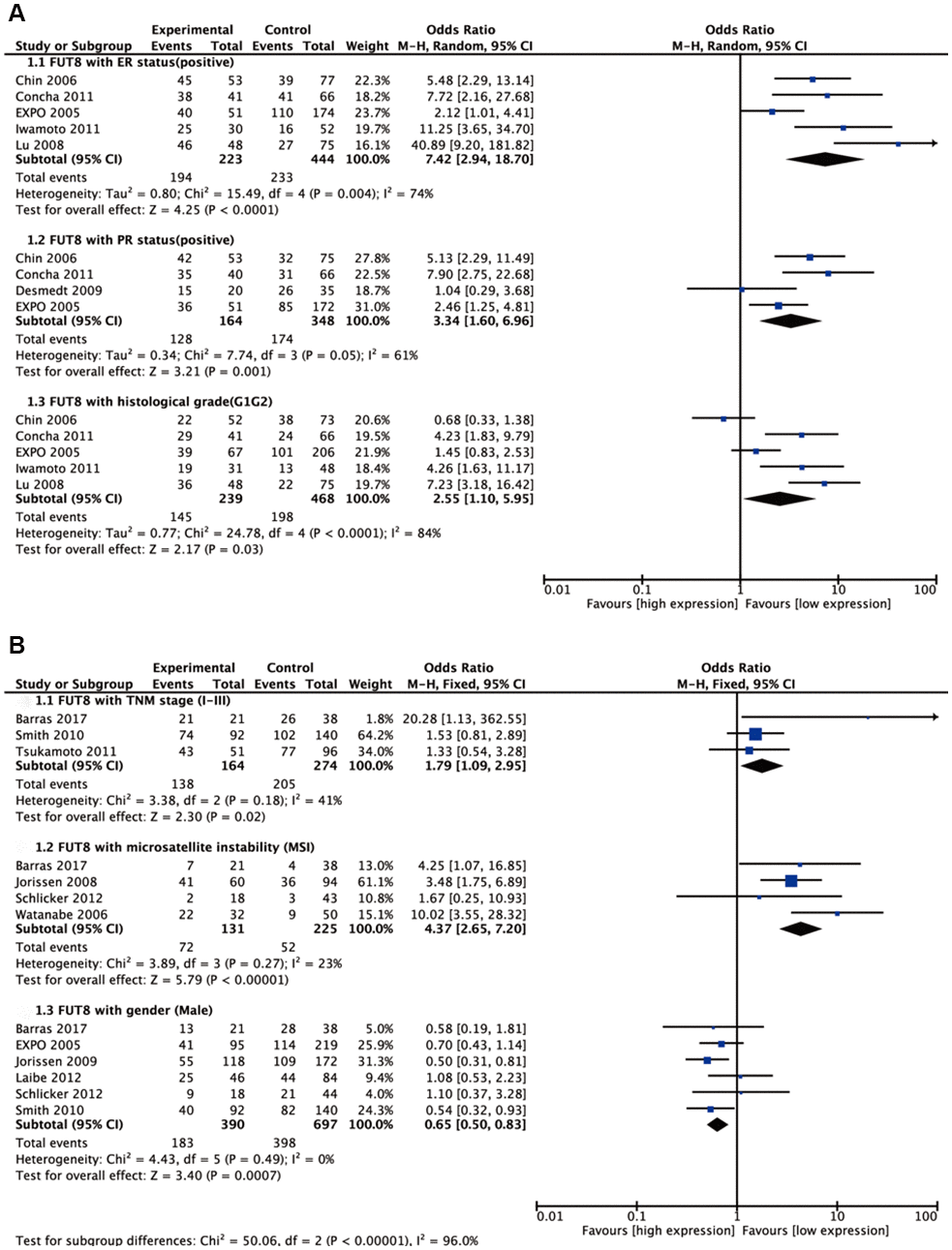
Figure 1. Forest plots of the significant associations between FUT8 expression and clinical features in two tumor types. (A) breast cancer; (B) colorectal cancer.
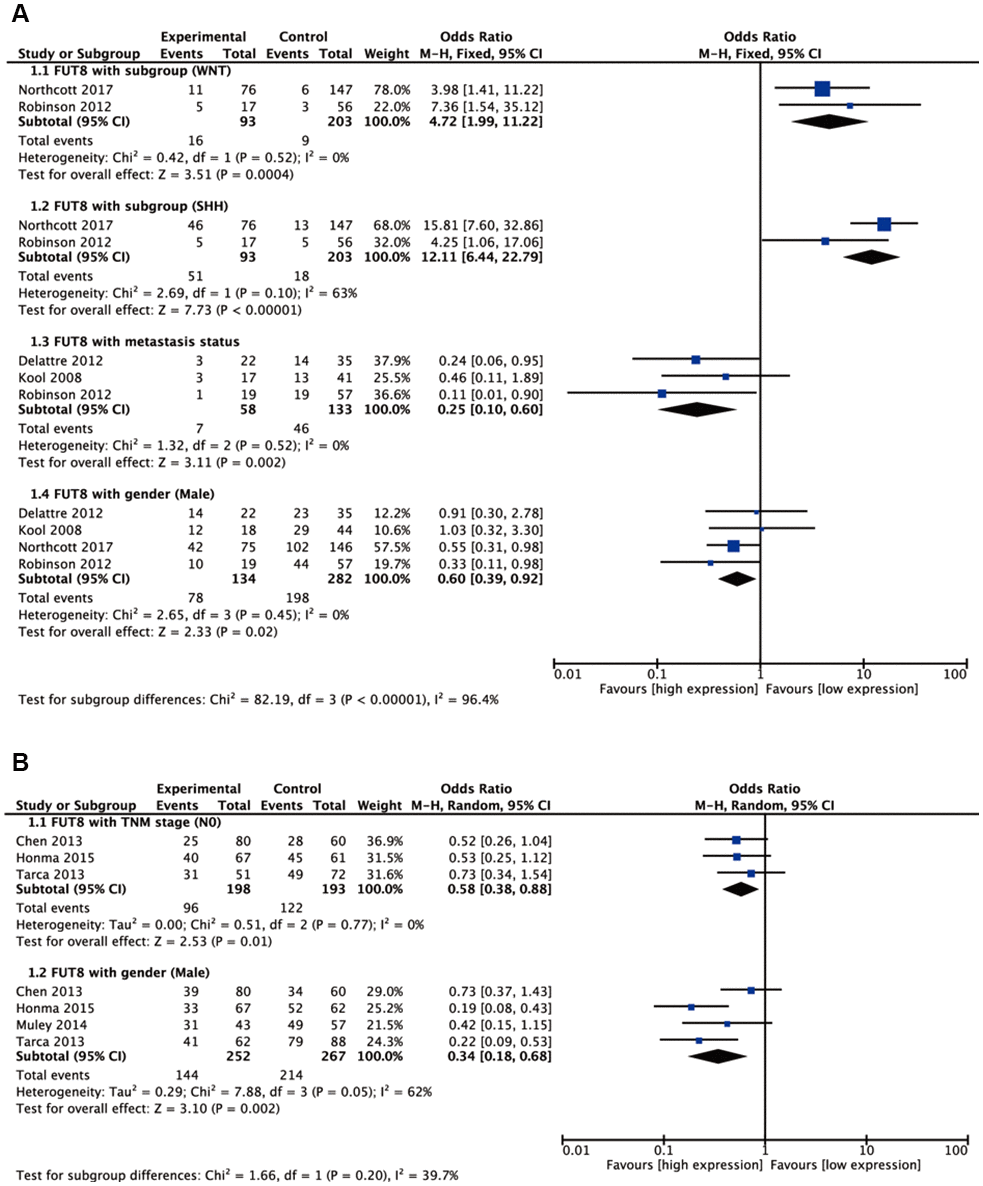
Figure 2. Forest plots of significant associations between FUT8 expression and clinical features in two tumor types. (A) medulloblastoma; (B) non-small cell lung cancer.
Table 2. Clinicopathological features related to enhanced FUT8 expression level in various malignant tumors.
| Tumor source | Clinicopathological features | Reference | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Breast cancer | ER status positive, PR status positive, histological grade (G1G2) histological grade (G1) | [50], [51], [52], [53], R2 platform | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Colorectal cancer | Duke stage A, Duke stage AB, location (Left), gender (male) microsatellite instability (MSI) | [54], [55], [56], [57], [58], [59], [60], [61], R2 platform | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Glioma | Age (≤40 years), glioblastoma multiforme (GBM) | [66], [67], [68], [69] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Medulloblastoma | Molecular subgroups (G3), molecular subgroups (G4), molecular subgroups (SHH), molecular subgroups (WNT) Gender (male), metastasis | [74], [75], [76], R2 platform | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-small cell lung cancer | Gender (male), TNM stage N0, TNM stage M1 | [21], [70], [71], R2 platform | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ependymoma | Age (≤10 years), recurrence | [63], [64], [65] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ER, estrogen receptor; PR, progesterone receptor. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Prognostic value of FUT8 expression in survival of various types of malignant tumors
Five types of malignant tumors (NSCLC, glioma, diffuse large B cell lymphoma (DLBCL), gastric cancer (GC), BC) were included in meta-analyses for survival. For NSCLC, elevated FUT8 expression level was associated with shorter disease-free survival (DFS) (hazard ratio [HR]= 2.32, 95% CI: 1.65-3.27, p< 0.00001), and with lower overall survival (OS) (HR= 2.24, 95% CI: 1.62-3.08, p< 0.00001) (Figure 3).
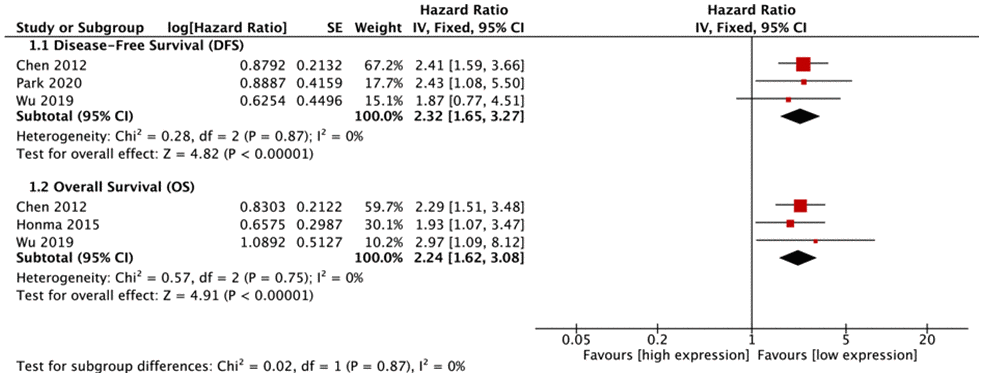
Figure 3. Forest plots of associations between FUT8 expression and non-small cell lung cancer overall survival.
FUT8 expression levels in the other 4 tumor types (glioma, DLBCL, GC, BC) were also correlated with OS (Figure 4). Among these, high FUT8 expression was associated with shorter OS in glioma (RR= 1.49, 95% CI: 1.13-1.96), DLBCL (RR= 1.76, 95% CI: 1.30-2.38), and BC (RR= 2.49, 95% CI: 1.15-5.38), but with longer OS in GC (RR= 0.60, 95% CI: 0.40-0.91). For glioma, upregulated FUT8 expression was associated with shorter OS in female patients (RR= 1.99, 95% CI: 1.15-3.44). For DLBCL, high FUT8 expression was associated with shorter OS in Ann Arbor stages I-III (RR= 1.87, 95% CI: 1.24-2.81) and in patients aged >50 years (RR= 1.42, 95% CI: 1.02-1.98). For GC, high FUT8 expression was associated with better OS in intestinal-type Lauren classification (RR= 0.50, 95% CI: 0.28-0.90), males (RR= 0.59, 95% CI: 0.35-0.99), and TNM stage I-III patients (RR= 0.53, 95% CI: 0.31-0.90). Pooled survival results are presented in Supplementary Table 2.
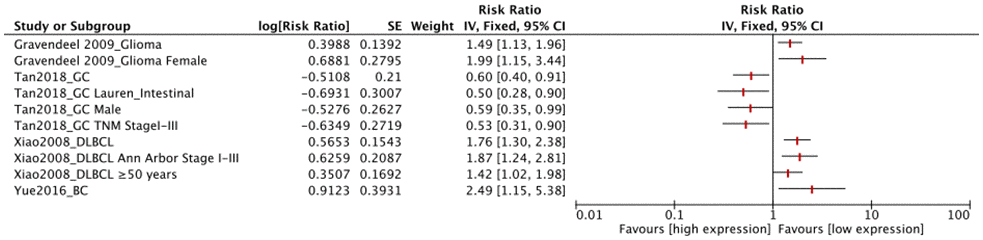
Figure 4. Forest plots of associations between FUT8 expression and tumor overall survival in single clinical features studied.
Discussion
FUT8 is clearly involved in tumor initiation and progression, and in various biological behaviors of cancer, including cell proliferation, apoptosis, migration, and metastasis [27]. Prognostic values of glycosylation or fucosylated antigens in many types of cancer have been documented in several previous reviews and meta-analyses [14, 15, 28–30]. However, very little meta-analysis of FUT8 has been performed, because of lack of sufficient studies and references. Many reports are based on use of microarrays to study cancer genomes; accordingly, we performed a systematic review after collecting GEO microarray data, and data from a large number of studies, focused on the prognostic value of FUT8 in cancer. This review / meta-analysis is, to our knowledge, the first to comprehensively clarify the association of FUT8 expression with specific clinicopathological features and survival data for various types of cancer.
Our analyses demonstrated that FUT8 expression levels were most often associated with tumor stage (n=4: BC, CRC, MBL, NSCLC), molecular classification (n=2: BC, MBL), age (n=2: glioma, ependymoma), and gender (n=2: MBL, NSCLC), but less associated with histological grade (n=1: BC) and pathological typing (n=1: glioma). FUT8 expression showed no association with disease induction, location, or family history for any of the above cancer types. These findings suggest that FUT8 plays an intrinsic role mainly in tumor development, and is therefore a potentially important biomarker for malignant tumors. Previous studies have focused mainly on the relationship between FUT8 expression and OS for survival. The majority of such studies (n=5: NSCLC, BC, DLBCL, GC, glioma) found significant correlations of FUT8 expression with OS of tumor, and also associations between survival and clinicopathological features, particularly tumor stage. A few studies focused on associations of FUT8 expression with other types of survival (DFS, RFS). For the research on pathways related to FUT8 and tumor survival, many references indicate that FUT8 activates several related signaling pathways, including Ras/MAPKK signaling, c-Met signaling, Akt/mTOR signaling and Wnt/β-catenin signaling, which ultimately lead to hepatocellular carcinoma or colorectal cancer affects the overall survival of patients [31–33]. Höti et al demonstrated that overexpression of FUT8 resulted in upregulation of epidermal growth factor receptor (EGFR) and corresponding downstream signaling, leading to increased prostate cancer cells survival [34]. In short, findings of these studies, taken together, demonstrate the strong prognostic value of FUT8 expression in malignant tumors.
NSCLC is the tumor type most frequently studied in regard to FUT8 expression status. High FUT8 expression was inversely correlated with N0 status of TNM stage and with male gender. High FUT8 expression was associated with shorter DFS, and with lower OS. Thus, FUT8 should be a useful survival prognostic predictor for female NSCLC patients with high FUT8 expression. Our analyses revealed an association of high FUT8 expression level with positive ER and PR status, and with shorter OS, in BC patients. FUT8 expression is therefore presumably enhanced in luminal A and B BC patients, and may be a useful survival prognostic predictor in such patients. A 2019 study indicated that FUT8 is highly expressed in ER+ BC patients, is associated with metastasis, and is a potential therapeutic target in these patients [35].
Microsatellite instability (MSI) is a molecular "fingerprint" of defective mismatch repair systems, and methods to detect MSI are well established and routinely incorporated into clinical practice. Prognosis for MSI tumors is better than that for microsatellite stable CRC [36]. In regard to CRC, we found that high FUT8 expression was associated with MSI, female gender, and TNM stages I-III. E-cadherin, a Ca2+-dependent cell adhesion molecule, was found to be significantly enhanced in dense culture of FUT8-transfected colorectal adenocarcinoma cells, resulting in increased cell-cell adhesion [23]. In addition, E-cadherin truncation was significantly higher in low MSI as compared with high MSI tumors [37]. The inferred mechanism is that the low expression of FUT8 in TNM stages IV or microsatellite stable colorectal cancer may cause E-cadherin to decrease or be truncated, resulting in decreased tumor cell-cell adhesion and increased metastasis.
Medulloblastoma, a common type of malignant brain cancer that accounts for 8-10% of childhood brain tumors [38], comprises four molecular subgroups (WNT, SHH, group 3, group 4) that have differing prognoses [39]. In general, subgroup WNT has very good prognosis and rare metastasis; subgroup SHH has good prognosis in infants and intermediate prognosis and uncommon metastasis in older children; subgroup G4 has intermediate prognosis and frequent metastasis; subgroup G3 has poor prognosis and very frequent metastasis [40–43]. Our analysis revealed association of high FUT8 expression with subgroups WNT and SHH, and inverse correlation with metastasis and male gender. High FUT8 expression in medulloblastoma therefore appears to be a positive prognostic indicator, particularly for female patients. In regard to NB, we did not find any notable relationship of high FUT8 expression with clinicopathological features. On the other hand, another group reported aberrant expression of N-glycans and short O-glycans in NB cells, and regulation of their expression levels by associated glycosyltransferases [44]. GnT-V expression in NB patients was correlated with favorable prognosis and treatment outcome [45]. GALNT9 was expressed in neuroblasts derived from primary tumor [46] but not in those derived from metastatic bone marrow, and may be a useful prognostic marker for positive clinical outcome in NB patients [47].
Overall, our results are comprehensive and seemingly reliable in view of the high quality of included articles and microarray data. On the other hand, there are inherent limitations in our analysis. (i) Heterogeneity is present among studies of a given tumor type, and is difficult to address because of methodological differences (e.g., sample selection, detection method, determination of cutoff value, statistical analysis). (ii) Nearly all our included studies report a statistically significant result. Although a Begg’s funnel plot indicates absence of publication bias (Supplementary Figure 3), our experience suggests that selective reporting bias is common in the literature regarding FUT8 and tumor prognosis. (iii) Roughly half of our included studies had small sample size (<100), which is often associated with inflated estimates of effect size and with high heterogeneity.
Results of our analyses – with due consideration of the above caveats – highlight the prognostic value of FUT8 expression in a variety of malignant tumors, and the important biological function of FUT8 in tumor progression. FUT8 may exert its effects in such tumors by regulating functional protein core fucosylations involved in tumor development and metastasis (e.g., L1CAM, P53, TGF-β, EGFR) [25, 34, 48]. Thus, dysregulation of FUT8 would have effects on tumor development, and consequently on clinical prognosis of patients. The molecular mechanisms remain to be clarified, and are being addressed in ongoing studies.
In conclusion, our systematic review and meta-analysis confirmed the association of FUT8 with clinicopathological features and patient survival rates for certain malignant tumor types. FUT8 expression is a significant predictor of prognosis for these tumors. The relative weight of FUT8 correlations with particular clinical features is currently difficult to evaluate because of the presence of many uncontrollable factors. Reliable verification of prognostic value of FUT8 in these tumor types will require a large-scale study using standardized methods of detection, analysis, and reporting.
Materials and Methods
We performed this review based on PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) criteria.
Search strategy
The PubMed, Embase, Web of Science, Springer, Chinese National Knowledge Infrastructure (CNKI), and Wan Fang databases were searched systematically to identify and retrieve all pertinent publications through June 24, 2020. Keywords and search terms used were: FUT8 or Fut8 or Fucosyltransferase 8 or α1-6 Fucosyltransferase or Core fucosylation or Fucosyltransferase; Malignant tumor or Cancer or Carcinoma or Neoplasm; and clinical or clinicopathological or clinicopathology or prognosis predictor or survival or Odds Ratio (OR) or Hazard Ratio (HR). References in retrieved articles were screened manually. Languages of retrieved articles were restricted to English and Chinese.
Data extraction and analysis of GEO datasets
Our first step was to read and screen articles related to tumor clinicopathological features and prognosis, and search for microarray datasets through the references. In this way, 96 datasets from 214 articles were extracted for preliminary screening. The second step was to re-screen the 96 datasets in regard to presence of FUT8 data, an appropriate clinical pathology track, and sufficient number of samples. In this way, 35 malignant tumor microarray datasets were selected and downloaded from the Gene Expression Omnibus (GEO) database (https://www.ncbi.nlm.nih.gov/gds) and R2 platform (http://r2.amc.nl). These datasets included a total of 4918 samples, from 7 types of malignant tumor: breast cancer (BC), colorectal cancer (CRC), ependymoma, glioma, non-small cell lung cancer (NSCLC), medulloblastoma (MBL), and neuroblastoma (NB). The datasets all used the Affymetrix Human Genome U133a or U133 Plus 2.0 expression arrays to detect expression value signals. Data were normalized using Microarray Suite (MAS) V. 5.0. In many cases, more than one probeset has been reported for FUT8 gene. We selected a probeset with highest average presence signal (APS) by default. mRNA expression of FUT8 was assigned to "low" or "high" category based on average expression value of each dataset.
Inclusion and exclusion criteria
Studies were included if they met the following criteria: (1) original study focused on human subjects; (2) presented FUT8 expression data and malignant tumor clinicopathological features and/or survival data; (3) reported an OR or HR with 95% confidence interval (CI), or sufficient data were presented so that we could calculate them; (4) full text was available. Exclusion criteria were as follows: (1) study lacked key information (e.g., clinical parameters, survival curves), or usable data; (2) APS of probeset was too low, or too many data were lost; (3) HRs applied to a combination of multiple FUTs; (4) article was a review, letter, single case report, or conference abstracts. In cases of multiple articles from the same group, reporting overlapping data, only the most complete one was included. The article selection process is shown in flow diagram form in Figure 5.
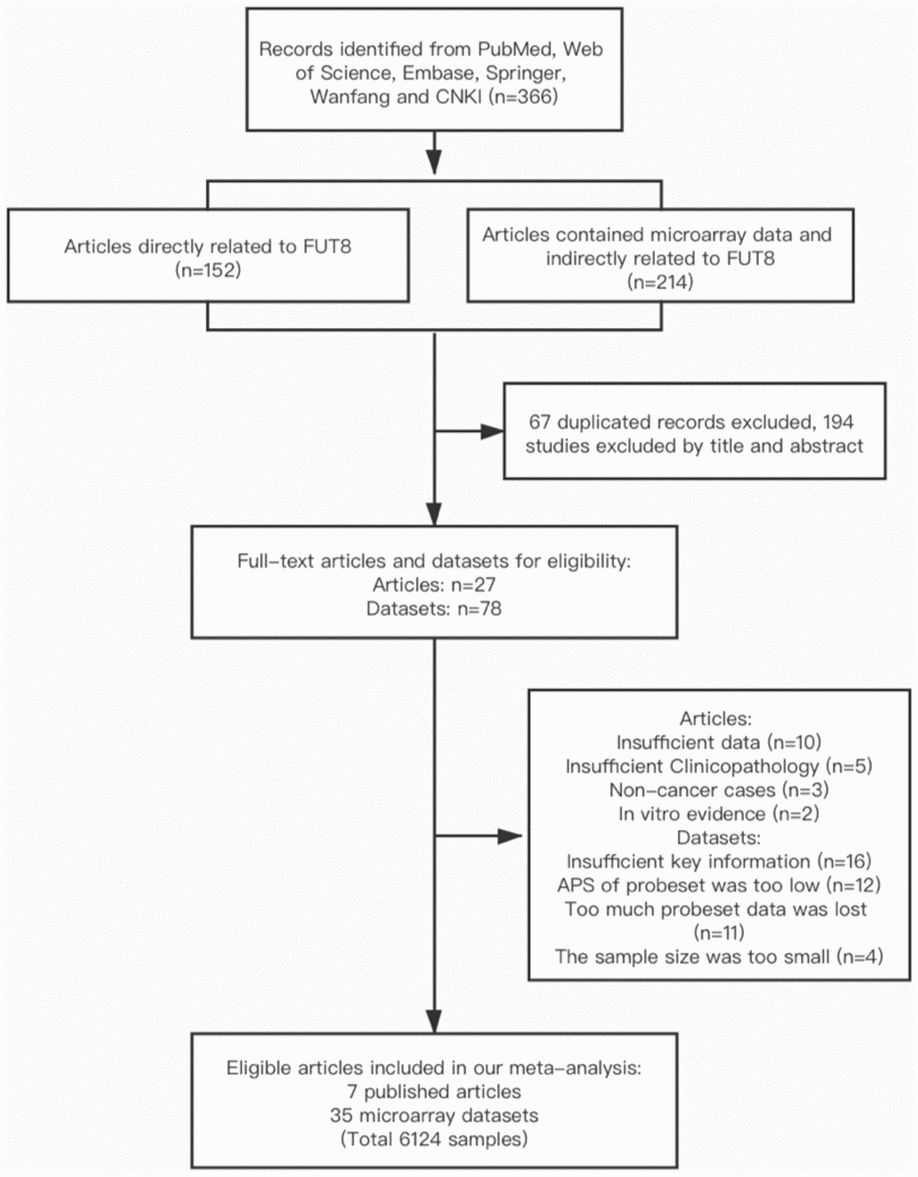
Figure 5. Flow diagram of article selection process.
Quality assessment and data extraction
Two authors (MXM and GXH) reviewed potentially eligible articles independently. Quality of each study was assessed using the Newcastle-Ottawa Scale. The following information was extracted from each included study:
(1) basic information: first author’s or uploaded dataset author’s name, publication year, country of origin, names of malignant tumors, sample size, FUT8 expression levels, detection methods, sample type, outcome measurements, follow-up duration, cutoff value, survival analysis method; (2) p values for correlation between FUT8 expression and clinicopathological features of malignant tumors, and original data used for calculation of ORs and their 95% CIs; (3) HRs and their 95% CIs for survival analysis. If HRs were not directly accessible in the text, Kaplan-Meier survival curves were read using Engauge Digitizer (V. 4.1) to obtain data. Different datasets for a particular malignant tumor were considered as separate studies, and respective HRs were extracted. In cases of possible discrepancy, a consensus was reached by discussion among all authors.
Statistical analysis
ORs and their 95% CIs were used to estimate associations of FUT8 with clinical features of malignant tumors. For purposes of comparison, patients were divided into paired categories (e.g., male vs. female; TNM stages I, II and III vs. IV; ER/PR status positive vs. negative). For survival rates, HRs with corresponding 95% CIs were used. All ORs and HRs were calculated for high FUT8 expression. When a given FUT8 was investigated in two or more different studies, a meta-analysis was performed to combine the effect size. Z test was used to determine significance of ORs or HRs. Heterogeneity between studies was tested using Q statistic and I2 test. When I2 value was >50% (indicating significant heterogeneity) a random-effects model was used. For I2 value ≤50%, a fixed-effects model was used. Statistical analyses were performed with software program Review Manager V. 5.3 (Cochrane Collaboration; London, UK). Differences with p< 0.05 were considered statistically significant.
Author Contributions
MXM and XL conceived and designed the study. MXM and GXH searched and reviewed literature. MXM, GXH, ZYZ and YW contributed to data collection, analysis, interpretation and prepared tables and figures. MXM drafted the manuscript. XL and FG revised the manuscript. All authors approved the final manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
This study was supported by the Youth Science Fund of the Fifth People’s Hospital of Qinghai Province (No. 2017-Y-01). The present study was supported by the National Natural Science Foundation of China (No. 81770123, 32071274), and Hundred-Talent Program of Shaanxi Province, and Youth Innovation Team of Shaanxi Universities.
References
- 1. Hayes DF. Prognostic and predictive factors revisited. Breast. 2005; 14:493–99. https://doi.org/10.1016/j.breast.2005.08.023 [PubMed]
- 2. Pan JH, Zhou H, Cooper L, Huang JL, Zhu SB, Zhao XX, Ding H, Pan YL, Rong L. LAYN is a prognostic biomarker and correlated with immune infiltrates in gastric and colon cancers. Front Immunol. 2019; 10:6. https://doi.org/10.3389/fimmu.2019.00006 [PubMed]
- 3. Bahrami A, Aledavood A, Anvari K, Hassanian SM, Maftouh M, Yaghobzade A, Salarzaee O, ShahidSales S, Avan A. The prognostic and therapeutic application of microRNAs in breast cancer: tissue and circulating microRNAs. J Cell Physiol. 2018; 233:774–86. https://doi.org/10.1002/jcp.25813 [PubMed]
- 4. Deng W, Peng W, Wang T, Chen J, Zhu S. Overexpression of MMPs functions as a prognostic biomarker for oral cancer patients: a systematic review and meta-analysis. Oral Health Prev Dent. 2019; 17:505–14. https://doi.org/10.3290/j.ohpd.a43636 [PubMed]
- 5. Testa U, Lo-Coco F. Prognostic factors in acute promyelocytic leukemia: strategies to define high-risk patients. Ann Hematol. 2016; 95:673–80. https://doi.org/10.1007/s00277-016-2622-1 [PubMed]
- 6. Menon SS, Guruvayoorappan C, Sakthivel KM, Rasmi RR. Ki-67 protein as a tumour proliferation marker. Clin Chim Acta. 2019; 491:39–45. https://doi.org/10.1016/j.cca.2019.01.011 [PubMed]
- 7. Hu H, Li Z, Zhou Y, Zhang Y, Zhao L, Zhao W, Huang Y, Song X. GLT8D1 overexpression as a novel prognostic biomarker in human cutaneous melanoma. Melanoma Res. 2019; 29:612–20. https://doi.org/10.1097/CMR.0000000000000631 [PubMed]
- 8. Gomes C, Almeida A, Barreira A, Calheiros J, Pinto F, Abrantes R, Costa A, Polonia A, Campos D, Osório H, Sousa H, Pinto-de-Sousa J, Kolarich D, Reis CA. Carcinoembryonic antigen carrying SLeX as a new biomarker of more aggressive gastric carcinomas. Theranostics. 2019; 9:7431–46. https://doi.org/10.7150/thno.33858 [PubMed]
- 9. Luo G, Fan Z, Cheng H, Jin K, Guo M, Lu Y, Yang C, Fan K, Huang Q, Long J, Liu L, Xu J, Lu R, et al. New observations on the utility of CA19-9 as a biomarker in lewis negative patients with pancreatic cancer. Pancreatology. 2018; 18:971–76. https://doi.org/10.1016/j.pan.2018.08.003 [PubMed]
- 10. Taniguchi N, Kizuka Y. Glycans and cancer: role of N-glycans in cancer biomarker, progression and metastasis, and therapeutics. Adv Cancer Res. 2015; 126:11–51. https://doi.org/10.1016/bs.acr.2014.11.001 [PubMed]
- 11. Bird-Lieberman EL, Neves AA, Lao-Sirieix P, O’Donovan M, Novelli M, Lovat LB, Eng WS, Mahal LK, Brindle KM, Fitzgerald RC. Molecular imaging using fluorescent lectins permits rapid endoscopic identification of dysplasia in barrett’s esophagus. Nat Med. 2012; 18:315–21. https://doi.org/10.1038/nm.2616 [PubMed]
- 12. Christiansen MN, Chik J, Lee L, Anugraham M, Abrahams JL, Packer NH. Cell surface protein glycosylation in cancer. Proteomics. 2014; 14:525–46. https://doi.org/10.1002/pmic.201300387 [PubMed]
- 13. Meany DL, Chan DW. Aberrant glycosylation associated with enzymes as cancer biomarkers. Clin Proteomics. 2011; 8:7. https://doi.org/10.1186/1559-0275-8-7 [PubMed]
- 14. Pinho SS, Reis CA. Glycosylation in cancer: mechanisms and clinical implications. Nat Rev Cancer. 2015; 15:540–55. https://doi.org/10.1038/nrc3982 [PubMed]
- 15. Stowell SR, Ju T, Cummings RD. Protein glycosylation in cancer. Annu Rev Pathol. 2015; 10:473–510. https://doi.org/10.1146/annurev-pathol-012414-040438 [PubMed]
- 16. Ciborowski P, Finn OJ. Non-glycosylated tandem repeats of MUC1 facilitate attachment of breast tumor cells to normal human lung tissue and immobilized extracellular matrix proteins (ECM) in vitro: potential role in metastasis. Clin Exp Metastasis. 2002; 19:339–45. https://doi.org/10.1023/a:1015590515957 [PubMed]
- 17. Gaziel-Sovran A, Segura MF, Di Micco R, Collins MK, Hanniford D, Vega-Saenz de Miera E, Rakus JF, Dankert JF, Shang S, Kerbel RS, Bhardwaj N, Shao Y, Darvishian F, et al. miR-30b/30d regulation of GalNAc transferases enhances invasion and immunosuppression during metastasis. Cancer Cell. 2011; 20:104–18. https://doi.org/10.1016/j.ccr.2011.05.027 [PubMed]
- 18. Ma M, Fu Y, Zhou X, Guan F, Wang Y, Li X. Functional roles of fucosylated and O-glycosylated cadherins during carcinogenesis and metastasis. Cell Signal. 2019; 63:109365. https://doi.org/10.1016/j.cellsig.2019.109365 [PubMed]
- 19. Cheng L, Gao S, Song X, Dong W, Zhou H, Zhao L, Jia L. Comprehensive N-glycan profiles of hepatocellular carcinoma reveal association of fucosylation with tumor progression and regulation of FUT8 by microRNAs. Oncotarget. 2016; 7:61199–214. https://doi.org/10.18632/oncotarget.11284 [PubMed]
- 20. Yue L, Han C, Li Z, Li X, Liu D, Liu S, Yu H. Fucosyltransferase 8 expression in breast cancer patients: a high throughput tissue microarray analysis. Histol Histopathol. 2016; 31:547–55. https://doi.org/10.14670/HH-11-693 [PubMed]
- 21. Chen CY, Jan YH, Juan YH, Yang CJ, Huang MS, Yu CJ, Yang PC, Hsiao M, Hsu TL, Wong CH. Fucosyltransferase 8 as a functional regulator of nonsmall cell lung cancer. Proc Natl Acad Sci USA. 2013; 110:630–35. https://doi.org/10.1073/pnas.1220425110 [PubMed]
- 22. Takahashi T, Ikeda Y, Miyoshi E, Yaginuma Y, Ishikawa M, Taniguchi N. Alpha1,6fucosyltransferase is highly and specifically expressed in human ovarian serous adenocarcinomas. Int J Cancer. 2000; 88:914–19. https://doi.org/10.1002/1097-0215(20001215)88:6<914::aid-ijc12>3.0.co;2-1 [PubMed]
- 23. Osumi D, Takahashi M, Miyoshi E, Yokoe S, Lee SH, Noda K, Nakamori S, Gu J, Ikeda Y, Kuroki Y, Sengoku K, Ishikawa M, Taniguchi N. Core fucosylation of E-cadherin enhances cell-cell adhesion in human colon carcinoma WiDr cells. Cancer Sci. 2009; 100:888–95. https://doi.org/10.1111/j.1349-7006.2009.01125.x [PubMed]
- 24. Höti N, Yang S, Hu Y, Shah P, Haffner MC, Zhang H. Overexpression of α (1,6) fucosyltransferase in the development of castration-resistant prostate cancer cells. Prostate Cancer Prostatic Dis. 2018; 21:137–46. https://doi.org/10.1038/s41391-017-0016-7 [PubMed]
- 25. Agrawal P, Fontanals-Cirera B, Sokolova E, Jacob S, Vaiana CA, Argibay D, Davalos V, McDermott M, Nayak S, Darvishian F, Castillo M, Ueberheide B, Osman I, et al. A systems biology approach identifies FUT8 as a driver of melanoma metastasis. Cancer Cell. 2017; 31:804–19.e7. https://doi.org/10.1016/j.ccell.2017.05.007 [PubMed]
- 26. Tu CF, Wu MY, Lin YC, Kannagi R, Yang RB. FUT8 promotes breast cancer cell invasiveness by remodeling TGF-β receptor core fucosylation. Breast Cancer Res. 2017; 19:111. https://doi.org/10.1186/s13058-017-0904-8 [PubMed]
- 27. Keeley TS, Yang S, Lau E. The diverse contributions of fucose linkages in cancer. Cancers (Basel). 2019; 11:1241. https://doi.org/10.3390/cancers11091241 [PubMed]
- 28. Wang M, Zhu J, Lubman DM, Gao C. Aberrant glycosylation and cancer biomarker discovery: a promising and thorny journey. Clin Chem Lab Med. 2019; 57:407–16. https://doi.org/10.1515/cclm-2018-0379 [PubMed]
- 29. Liang JX, Liang Y, Gao W. Clinicopathological and prognostic significance of sialyl lewis X overexpression in patients with cancer: a meta-analysis. Onco Targets Ther. 2016; 9:3113–25. https://doi.org/10.2147/OTT.S102389 [PubMed]
- 30. do Nascimento JC, Beltrão EI, Rocha CR. High FUT3 expression is a marker of lower overall survival of breast cancer patients. Glycoconj J. 2020; 37:263–75. https://doi.org/10.1007/s10719-020-09914-2 [PubMed]
- 31. Wang Y, Fukuda T, Isaji T, Lu J, Im S, Hang Q, Gu W, Hou S, Ohtsubo K, Gu J. Loss of α1,6-fucosyltransferase inhibits chemical-induced hepatocellular carcinoma and tumorigenesis by down-regulating several cell signaling pathways. FASEB J. 2015; 29:3217–27. https://doi.org/10.1096/fj.15-270710 [PubMed]
- 32. Zhang C, Wu Q, Huang H, Chen X, Huang T, Li W, Zhang J, Liu Y. Caveolin-1 upregulates Fut8 expression by activating the wnt/β-catenin pathway to enhance HCC cell proliferative and invasive ability. Cell Biol Int. 2020; 44:2202–12. https://doi.org/10.1002/cbin.11426 [PubMed]
- 33. Qi Y, Shan Y, Li S, Huang Y, Guo Y, Huang T, Jia L. LncRNA LEF1-AS1/LEF1/FUT8 axis mediates colorectal cancer progression by regulating α1, 6-fucosylation via Wnt/β-catenin pathway. Research Square. 2020. [Epub ahead of print]. https://doi.org/10.21203/rs.3.rs-49278/v1
- 34. Höti N, Lih TS, Pan J, Zhou Y, Yang G, Deng A, Chen L, Dong M, Yang RB, Tu CF, Haffner MC, Kay Li Q, Zhang H. A comprehensive analysis of FUT8 overexpressing prostate cancer cells reveals the role of EGFR in castration resistance. Cancers (Basel). 2020; 12:468. https://doi.org/10.3390/cancers12020468 [PubMed]
- 35. Shaabanpour aghamaleki F, Farivar S. Bioinformatics-Based Prediction of FUT8 as a Therapeutic Target in Estrogen Receptor-Positive Breast Cancer. Multidisciplinary Cancer Investigation. 2019; 03:25–31.
- 36. Vilar E, Gruber SB. Microsatellite instability in colorectal cancer-the stable evidence. Nat Rev Clin Oncol. 2010; 7:153–62. https://doi.org/10.1038/nrclinonc.2009.237 [PubMed]
- 37. Ortega P, Morán A, de Juan C, Frías C, Hernández S, López-Asenjo JA, Sánchez-Pernaute A, Torres A, Iniesta P, Benito M. Differential wnt pathway gene expression and E-cadherin truncation in sporadic colorectal cancers with and without microsatellite instability. Clin Cancer Res. 2008; 14:995–1001. https://doi.org/10.1158/1078-0432.CCR-07-1588 [PubMed]
- 38. Millard NE, De Braganca KC. Medulloblastoma. J Child Neurol. 2016; 31:1341–53. https://doi.org/10.1177/0883073815600866 [PubMed]
- 39. Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW. The 2016 world health organization classification of tumors of the central nervous system: a summary. Acta Neuropathol. 2016; 131:803–20. https://doi.org/10.1007/s00401-016-1545-1 [PubMed]
- 40. Cho YJ, Tsherniak A, Tamayo P, Santagata S, Ligon A, Greulich H, Berhoukim R, Amani V, Goumnerova L, Eberhart CG, Lau CC, Olson JM, Gilbertson RJ, et al. Integrative genomic analysis of medulloblastoma identifies a molecular subgroup that drives poor clinical outcome. J Clin Oncol. 2011; 29:1424–30. https://doi.org/10.1200/JCO.2010.28.5148 [PubMed]
- 41. Tamayo P, Cho YJ, Tsherniak A, Greulich H, Ambrogio L, Schouten-van Meeteren N, Zhou T, Buxton A, Kool M, Meyerson M, Pomeroy SL, Mesirov JP. Predicting relapse in patients with medulloblastoma by integrating evidence from clinical and genomic features. J Clin Oncol. 2011; 29:1415–23. https://doi.org/10.1200/JCO.2010.28.1675 [PubMed]
- 42. Fernandez-Teijeiro A, Betensky RA, Sturla LM, Kim JY, Tamayo P, Pomeroy SL. Combining gene expression profiles and clinical parameters for risk stratification in medulloblastomas. J Clin Oncol. 2004; 22:994–98. https://doi.org/10.1200/JCO.2004.03.036 [PubMed]
- 43. Pomeroy SL, Tamayo P, Gaasenbeek M, Sturla LM, Angelo M, McLaughlin ME, Kim JY, Goumnerova LC, Black PM, Lau C, Allen JC, Zagzag D, Olson JM, et al. Prediction of central nervous system embryonal tumour outcome based on gene expression. Nature. 2002; 415:436–42. https://doi.org/10.1038/415436a [PubMed]
- 44. Zhang Y, Iwasaki H, Wang H, Kudo T, Kalka TB, Hennet T, Kubota T, Cheng L, Inaba N, Gotoh M, Togayachi A, Guo J, Hisatomi H, et al. Cloning and characterization of a new human UDP-N-acetyl-alpha-D-galactosamine:polypeptide N-acetylgalactosaminyltransferase, designated pp-GalNAc-T13, that is specifically expressed in neurons and synthesizes GalNAc alpha-serine/threonine antigen. J Biol Chem. 2003; 278:573–84. https://doi.org/10.1074/jbc.M203094200 [PubMed]
- 45. Inamori K, Gu J, Ohira M, Kawasaki A, Nakamura Y, Nakagawa T, Kondo A, Miyoshi E, Nakagawara A, Taniguchi N. High expression of n-acetylglucosaminyltransferase V in favorable neuroblastomas: involvement of its effect on apoptosis. FEBS Lett. 2006; 580:627–32. https://doi.org/10.1016/j.febslet.2005.12.089 [PubMed]
- 46. Ferrandis E, Da Silva J, Riou G, Bénard I. Coactivation of the MDR1 and MYCN genes in human neuroblastoma cells during the metastatic process in the nude mouse. Cancer Res. 1994; 54:2256–61. [PubMed]
- 47. Berois N, Gattolliat CH, Barrios E, Capandeguy L, Douc-Rasy S, Valteau-Couanet D, Bénard J, Osinaga E. GALNT9 gene expression is a prognostic marker in neuroblastoma patients. Clin Chem. 2013; 59:225–33. https://doi.org/10.1373/clinchem.2012.192328 [PubMed]
- 48. Noda M, Okayama H, Kofunato Y, Chida S, Saito K, Tada T, Ashizawa M, Nakajima T, Aoto K, Kikuchi T, Sakamoto W, Endo H, Fujita S, et al. Prognostic role of FUT8 expression in relation to p53 status in stage II and III colorectal cancer. PLoS One. 2018; 13:e0200315. https://doi.org/10.1371/journal.pone.0200315 [PubMed]
- 49. Lasham A, Samuel W, Cao H, Patel R, Mehta R, Stern JL, Reid G, Woolley AG, Miller LD, Black MA, Shelling AN, Print CG, Braithwaite AW. YB-1, the E2F pathway, and regulation of tumor cell growth. J Natl Cancer Inst. 2012; 104:133–46. https://doi.org/10.1093/jnci/djr512 [PubMed]
- 50. Chin K, DeVries S, Fridlyand J, Spellman PT, Roydasgupta R, Kuo WL, Lapuk A, Neve RM, Qian Z, Ryder T, Chen F, Feiler H, Tokuyasu T, et al. Genomic and transcriptional aberrations linked to breast cancer pathophysiologies. Cancer Cell. 2006; 10:529–41. https://doi.org/10.1016/j.ccr.2006.10.009 [PubMed]
- 51. Desmedt C, Giobbie-Hurder A, Neven P, Paridaens R, Christiaens MR, Smeets A, Lallemand F, Haibe-Kains B, Viale G, Gelber RD, Piccart M, Sotiriou C. The gene expression grade index: a potential predictor of relapse for endocrine-treated breast cancer patients in the BIG 1-98 trial. BMC Med Genomics. 2009; 2:40. https://doi.org/10.1186/1755-8794-2-40 [PubMed]
- 52. Lu X, Lu X, Wang ZC, Iglehart JD, Zhang X, Richardson AL. Predicting features of breast cancer with gene expression patterns. Breast Cancer Res Treat. 2008; 108:191–201. https://doi.org/10.1007/s10549-007-9596-6 [PubMed]
- 53. Iwamoto T, Bianchini G, Booser D, Qi Y, Coutant C, Shiang CY, Santarpia L, Matsuoka J, Hortobagyi GN, Symmans WF, Holmes FA, O’Shaughnessy J, Hellerstedt B, et al. Gene pathways associated with prognosis and chemotherapy sensitivity in molecular subtypes of breast cancer. J Natl Cancer Inst. 2011; 103:264–72. https://doi.org/10.1093/jnci/djq524 [PubMed]
- 54. Laibe S, Lagarde A, Ferrari A, Monges G, Birnbaum D, Olschwang S, and COL2 Project. A seven-gene signature aggregates a subgroup of stage II colon cancers with stage III. OMICS. 2012; 16:560–65. https://doi.org/10.1089/omi.2012.0039 [PubMed]
- 55. Jorissen RN, Gibbs P, Christie M, Prakash S, Lipton L, Desai J, Kerr D, Aaltonen LA, Arango D, Kruhøffer M, Orntoft TF, Andersen CL, Gruidl M, et al. Metastasis-associated gene expression changes predict poor outcomes in patients with dukes stage B and C colorectal cancer. Clin Cancer Res. 2009; 15:7642–51. https://doi.org/10.1158/1078-0432.CCR-09-1431 [PubMed]
- 56. Smith JJ, Deane NG, Wu F, Merchant NB, Zhang B, Jiang A, Lu P, Johnson JC, Schmidt C, Bailey CE, Eschrich S, Kis C, Levy S, et al. Experimentally derived metastasis gene expression profile predicts recurrence and death in patients with colon cancer. Gastroenterology. 2010; 138:958–68. https://doi.org/10.1053/j.gastro.2009.11.005 [PubMed]
- 57. Watanabe T, Kobunai T, Toda E, Yamamoto Y, Kanazawa T, Kazama Y, Tanaka J, Tanaka T, Konishi T, Okayama Y, Sugimoto Y, Oka T, Sasaki S, et al. Distal colorectal cancers with microsatellite instability (MSI) display distinct gene expression profiles that are different from proximal MSI cancers. Cancer Res. 2006; 66:9804–08. https://doi.org/10.1158/0008-5472.CAN-06-1163 [PubMed]
- 58. Tsukamoto S, Ishikawa T, Iida S, Ishiguro M, Mogushi K, Mizushima H, Uetake H, Tanaka H, Sugihara K. Clinical significance of osteoprotegerin expression in human colorectal cancer. Clin Cancer Res. 2011; 17:2444–50. https://doi.org/10.1158/1078-0432.CCR-10-2884 [PubMed]
- 59. Jorissen RN, Lipton L, Gibbs P, Chapman M, Desai J, Jones IT, Yeatman TJ, East P, Tomlinson IP, Verspaget HW, Aaltonen LA, Kruhøffer M, Orntoft TF, et al. DNA copy-number alterations underlie gene expression differences between microsatellite stable and unstable colorectal cancers. Clin Cancer Res. 2008; 14:8061–69. https://doi.org/10.1158/1078-0432.CCR-08-1431 [PubMed]
- 60. Schlicker A, Beran G, Chresta CM, McWalter G, Pritchard A, Weston S, Runswick S, Davenport S, Heathcote K, Castro DA, Orphanides G, French T, Wessels LF. Subtypes of primary colorectal tumors correlate with response to targeted treatment in colorectal cell lines. BMC Med Genomics. 2012; 5:66. https://doi.org/10.1186/1755-8794-5-66 [PubMed]
- 61. Barras D, Missiaglia E, Wirapati P, Sieber OM, Jorissen RN, Love C, Molloy PL, Jones IT, McLaughlin S, Gibbs P, Guinney J, Simon IM, Roth AD, et al. BRAF V600E mutant colorectal cancer subtypes based on gene expression. Clin Cancer Res. 2017; 23:104–15. https://doi.org/10.1158/1078-0432.CCR-16-0140 [PubMed]
- 62. Donson AM, Birks DK, Barton VN, Wei Q, Kleinschmidt-Demasters BK, Handler MH, Waziri AE, Wang M, Foreman NK. Immune gene and cell enrichment is associated with a good prognosis in ependymoma. J Immunol. 2009; 183:7428–40. https://doi.org/10.4049/jimmunol.0902811 [PubMed]
- 63. Johnson RA, Wright KD, Poppleton H, Mohankumar KM, Finkelstein D, Pounds SB, Rand V, Leary SE, White E, Eden C, Hogg T, Northcott P, Mack S, et al. Cross-species genomics matches driver mutations and cell compartments to model ependymoma. Nature. 2010; 466:632–36. https://doi.org/10.1038/nature09173 [PubMed]
- 64. Hoffman LM, Donson AM, Nakachi I, Griesinger AM, Birks DK, Amani V, Hemenway MS, Liu AK, Wang M, Hankinson TC, Handler MH, Foreman NK. Molecular sub-group-specific immunophenotypic changes are associated with outcome in recurrent posterior fossa ependymoma. Acta Neuropathol. 2014; 127:731–45. https://doi.org/10.1007/s00401-013-1212-8 [PubMed]
- 65. Vladoiu MC, El-Hamamy I, Donovan LK, Farooq H, Holgado BL, Sundaravadanam Y, Ramaswamy V, Hendrikse LD, Kumar S, Mack SC, Lee JJ, Fong V, Juraschka K, et al. Childhood cerebellar tumours mirror conserved fetal transcriptional programs. Nature. 2019; 572:67–73. https://doi.org/10.1038/s41586-019-1158-7 [PubMed]
- 66. Freije WA, Castro-Vargas FE, Fang Z, Horvath S, Cloughesy T, Liau LM, Mischel PS, Nelson SF. Gene expression profiling of gliomas strongly predicts survival. Cancer Res. 2004; 64:6503–10. https://doi.org/10.1158/0008-5472.CAN-04-0452 [PubMed]
- 67. Gravendeel LA, Kouwenhoven MC, Gevaert O, de Rooi JJ, Stubbs AP, Duijm JE, Daemen A, Bleeker FE, Bralten LB, Kloosterhof NK, De Moor B, Eilers PH, van der Spek PJ, et al. Intrinsic gene expression profiles of gliomas are a better predictor of survival than histology. Cancer Res. 2009; 69:9065–72. https://doi.org/10.1158/0008-5472.CAN-09-2307 [PubMed]
- 68. Kawaguchi A, Yajima N, Tsuchiya N, Homma J, Sano M, Natsumeda M, Takahashi H, Fujii Y, Kakuma T, Yamanaka R. Gene expression signature-based prognostic risk score in patients with glioblastoma. Cancer Sci. 2013; 104:1205–10. https://doi.org/10.1111/cas.12214 [PubMed]
- 69. Zhang L, Chen LH, Wan H, Yang R, Wang Z, Feng J, Yang S, Jones S, Wang S, Zhou W, Zhu H, Killela PJ, Zhang J, et al. Exome sequencing identifies somatic gain-of-function PPM1D mutations in brainstem gliomas. Nat Genet. 2014; 46:726–30. https://doi.org/10.1038/ng.2995 [PubMed]
- 70. Tarca AL, Lauria M, Unger M, Bilal E, Boue S, Kumar Dey K, Hoeng J, Koeppl H, Martin F, Meyer P, Nandy P, Norel R, Peitsch M, et al, and IMPROVER DSC Collaborators. Strengths and limitations of microarray-based phenotype prediction: lessons learned from the IMPROVER diagnostic signature challenge. Bioinformatics. 2013; 29:2892–99. https://doi.org/10.1093/bioinformatics/btt492 [PubMed]
- 71. Honma R, Kinoshita I, Miyoshi E, Tomaru U, Matsuno Y, Shimizu Y, Takeuchi S, Kobayashi Y, Kaga K, Taniguchi N, Dosaka-Akita H. Expression of fucosyltransferase 8 is associated with an unfavorable clinical outcome in non-small cell lung cancers. Oncology. 2015; 88:298–308. https://doi.org/10.1159/000369495 [PubMed]
- 72. Li F, Zhao S, Cui Y, Guo T, Qiang J, Xie Q, Yu W, Guo W, Deng W, Gu C, Wu T. α1,6-Fucosyltransferase (FUT8) regulates the cancer-promoting capacity of cancer-associated fibroblasts (CAFs) by modifying EGFR core fucosylation (CF) in non-small cell lung cancer (NSCLC). Am J Cancer Res. 2020; 10:816–37. [PubMed]
- 73. Park S, Lim JM, Chun JN, Lee S, Kim TM, Kim DW, Kim SY, Bae DJ, Bae SM, So I, Kim HG, Choi JY, Jeon JH. Altered expression of fucosylation pathway genes is associated with poor prognosis and tumor metastasis in non-small cell lung cancer. Int J Oncol. 2020; 56:559–67. https://doi.org/10.3892/ijo.2019.4953 [PubMed]
- 74. Robinson G, Parker M, Kranenburg TA, Lu C, Chen X, Ding L, Phoenix TN, Hedlund E, Wei L, Zhu X, Chalhoub N, Baker SJ, Huether R, et al. Novel mutations target distinct subgroups of medulloblastoma. Nature. 2012; 488:43–48. https://doi.org/10.1038/nature11213 [PubMed]
- 75. Northcott PA, Buchhalter I, Morrissy AS, Hovestadt V, Weischenfeldt J, Ehrenberger T, Gröbner S, Segura-Wang M, Zichner T, Rudneva VA, Warnatz HJ, Sidiropoulos N, Phillips AH, et al. The whole-genome landscape of medulloblastoma subtypes. Nature. 2017; 547:311–17. https://doi.org/10.1038/nature22973 [PubMed]
- 76. Kool M, Koster J, Bunt J, Hasselt NE, Lakeman A, van Sluis P, Troost D, Meeteren NS, Caron HN, Cloos J, Mrsić A, Ylstra B, Grajkowska W, et al. Integrated genomics identifies five medulloblastoma subtypes with distinct genetic profiles, pathway signatures and clinicopathological features. PLoS One. 2008; 3:e3088. https://doi.org/10.1371/journal.pone.0003088 [PubMed]
- 77. Ohtaki M, Otani K, Hiyama K, Kamei N, Satoh K, Hiyama E. A robust method for estimating gene expression states using affymetrix microarray probe level data. BMC Bioinformatics. 2010; 11:183. https://doi.org/10.1186/1471-2105-11-183 [PubMed]
- 78. Łastowska M, Viprey V, Santibanez-Koref M, Wappler I, Peters H, Cullinane C, Roberts P, Hall AG, Tweddle DA, Pearson AD, Lewis I, Burchill SA, Jackson MS. Identification of candidate genes involved in neuroblastoma progression by combining genomic and expression microarrays with survival data. Oncogene. 2007; 26:7432–44. https://doi.org/10.1038/sj.onc.1210552 [PubMed]
- 79. Molenaar JJ, Koster J, Zwijnenburg DA, van Sluis P, Valentijn LJ, van der Ploeg I, Hamdi M, van Nes J, Westerman BA, van Arkel J, Ebus ME, Haneveld F, Lakeman A, et al. Sequencing of neuroblastoma identifies chromothripsis and defects in neuritogenesis genes. Nature. 2012; 483:589–93. https://doi.org/10.1038/nature10910 [PubMed]
- 80. Tada K, Ohta M, Hidano S, Watanabe K, Hirashita T, Oshima Y, Fujnaga A, Nakanuma H, Masuda T, Endo Y, Takeuchi Y, Iwashita Y, Kobayashi T, Inomata M. Fucosyltransferase 8 plays a crucial role in the invasion and metastasis of pancreatic ductal adenocarcinoma. Surg Today. 2020; 50:767–77. https://doi.org/10.1007/s00595-019-01953-z [PubMed]


