Introduction
Sudden cardiac death (SCD) is a major health issue worldwide and accounts for 180,000 to 250,000 deaths annually in the United States [1]. The age-adjusted incidence of SCD in the United States is 60 per 100,000 population [2]. Ventricular tachyarrhythmia is the major cause of SCD, and is not associated with any structural heart disease in 6 to 14% of cases [3, 4]. In some cases, SCD is associated with electrocardiographic abnormalities that affect ventricular repolarization, such as long or short QT syndrome [5]. Early repolarization pattern (ERP) is characterized by elevation of the QRS-ST junction or the J-point in a surface 12-lead electrocardiogram (ECG). Although ERP was considered benign, recent studies have suggested its potential association with cardiac arrhythmogenicity [6]. In case-control studies, the presence of ERP in the inferior or lateral leads is associated with susceptibility to ventricular fibrillation and SCD in patients without structural heart disease [7–9].
ERP is a common electrocardiographic finding that affects 1% to 13% of adults and is more common in young athletic males [7, 9–12]. The age of individuals enrolled in previous studies regarding ERP ranged widely from 25 to 95 years [13, 14]. Sinner et al conducted a large, prospective, population-based case-cohort study of individuals of Central-European descent (MONICA/KORA) and reported a high prevalence of ERP (13.1%) in individuals aged between 35-74 years and a 2- to 4-fold increased risk of cardiovascular mortality in individuals with ERP and aged between 35-54 years [15]. Haruta et al reviewed ECG records of 5976 atomic bomb survivors in Nagasaki, Japan with a mean age of 47.2 years and reported that ERP was associated with elevated risk of unexpected death and decreased risk of cardiac and all-cause death [16]. However, the long-term prognostic significance of ERP is poorly characterized in older middle-aged and elderly population.
In this study, we investigated the prevalence and prognostic value of ERP regarding cardiac and all-cause mortality in a large, multi-site, Healthy Aging Longitudinal Study (HALST) cohort consisting of older middle-aged and elderly adults belonging to the Han Chinese population in Taiwan.
Results
Study participants
The flowchart of enrollment and inclusion criteria of the study subjects is shown in Figure 1. We initially recruited 5,380 relatively healthy and ambulatory individuals from 7 communities across Taiwan. We then excluded 755 individuals with cancer and underlying severe cardiovascular diseases (e.g., myocardial infarction or pacemaker implantation), as well as those with missing follow-up information. Finally, we included 4,615 healthy individuals in this study. The prevalence of ERP in the study cohort was 19.3% (n=889/4,615). In the ERP (+) group, 122 out of 889 individuals died during follow-up (65.51±27.12 months). In the ERP (-) group, 561 out of 3726 individuals died during follow-up (62.63±28.44 months).
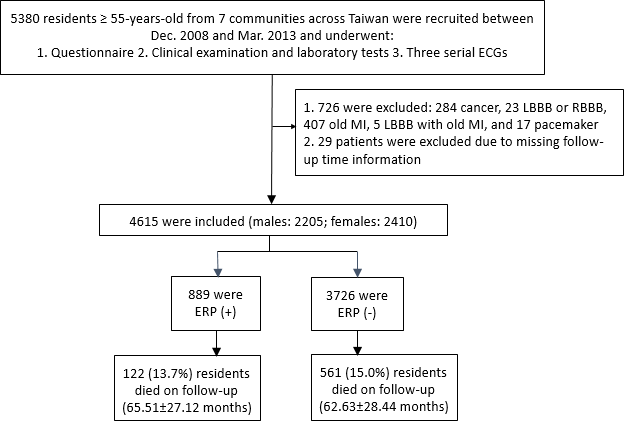
Figure 1. Schematic representation of the enrollment and inclusion criteria for the study subjects. ECG, electrocardiogram; LBBB, left bundle branch block; RBBB, right bundle branch block; MI, myocardial infarction; ERP, early repolarization pattern
The baseline clinical and demographic data of the study population is shown in Table 1. The mean age was 69.1±8.2 years and 47.8% of the study subjects were males. The average age of study participants was lower and the proportion of males and current smokers was higher in the ERP (+) group compared to the ERP (-) group. The mean values for the body mass index (BMI), systolic blood pressure (SBP), diastolic blood pressure (DBP), and hypertension were significantly lower (all P<0.005) and the length of the corrected QT interval (QTc) was shorter (P<0.001) in the ERP (+) group compared to the ERP (-) group.
Table 1. Demographic and clinical characteristics of the study population*.
| Parameters | All (N=4615) | ERP(+) (n=889) | ERP(-) (n=3,726) | P-value | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male, n (%) | 2205 (47.8%) | 470 (52.9%) | 1735 (46.6%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age at enrollment (years) | 69.1±8.2 | 67.7±7.8 | 69.5±8.2 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI (kg/m2) | 24.5±3.5 | 24.1±3.3 | 24.6±3.5 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Systolic BP (mmHg) | 128.4±18.7 | 125.89±18.7 | 128.9±18.7 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diastolic BP (mmHg) | 70.5±10.7 | 69.4±10.5 | 70.7±10.7 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Current | 608 (13.2%) | 168 (18.9%) | 440 (11.8%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Quit | 716 (15.5%) | 132 (14.8%) | 584 (15.8%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Never | 3291 (71.3%) | 589 (66.3%) | 2702 (72.5%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Essential hypertension, n (%) | 2011 (43.6%) | 349 (39.3%) | 1662 (44.6%) | 0.004 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes mellitus, n (%) | 823 (17.8%) | 159 (17.9%) | 664 (17.8%) | 0.99 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stroke, n (%) | 231 (5%) | 36 (4%) | 195 (5%) | 0.171 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hyperlipidemia, n (%) | 1456 (31.5%) | 269 (30.2%) | 1187 (31.8%) | 0.378 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CKD, n (%) | 667 (14.6%) | 126 (14.1%) | 541 (14.5%) | 0.833 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PR interval, ms | 171.1±34.1 | 170.7±32.5 | 171.2±34.4 | 0.677 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| QTc, ms | 437.0±22.6 | 431.4±20.7 | 438.3±22.9 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ERP: early repolarization pattern by the criteria adopted in 2015 [35]; BMI: body mass index; BP: blood pressure; CKD: chronic kidney disease; *Values are presented as n (%) or mean ± standard deviation; QTc was calculated by Bazett’s equations. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Figure 2 shows the representative ECGs of few selected individuals belonging to ERP (+) and ERP (-) groups. ERP was observed in the inferior and lateral leads, both separately and simultaneously. Among the 4,615 healthy individuals, 574 (12.4%) individuals were ERP-positive in the inferior leads (ERP-inf+), 214 (4.6%) individuals were ERP-positive in the lateral leads (ERP-lat+), and 101(2.2%) individuals were ERP-positive in both lateral and inferior leads (ERP-lat+ inf+). The agreement between the two initial interpreters was moderate to high (k=0.97, agreement proportion= 0.98).
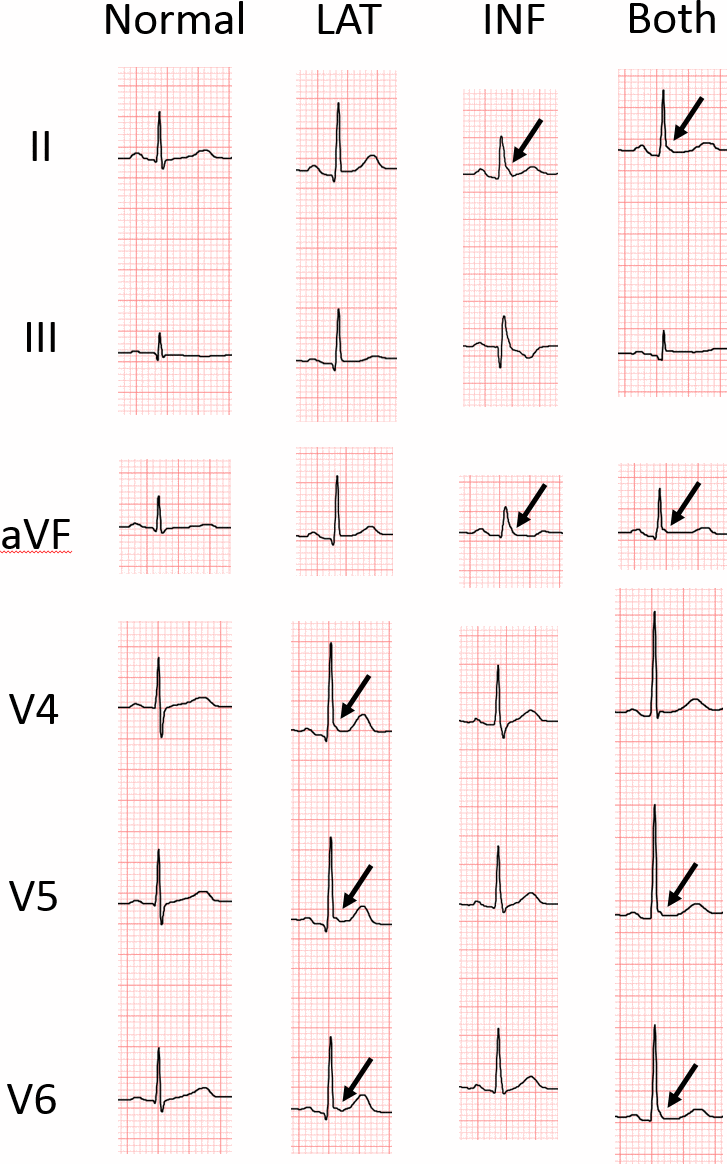
Figure 2. Representative ECGs of individuals with and without early repolarization pattern (ERP). LAT, ERP in lateral leads; INF, ERP in inferior leads; Both, ERP in both inferior and lateral leads. The arrow indicates junction (J)-point elevation greater than 0.1 mV.
Association between positive ERP and all-cause and cardiovascular mortality
We observed that all-cause and cardiovascular mortality rates were similar in ERP (+) and ERP (-) groups during a mean follow-up time of 95.1±21.9 months (log-rank test, P=0.13 and P=0.84, respectively; Figure 3A, 3B). Since previous studies showed that ERP in the inferior leads was a risk factor for all-cause and cardiovascular mortality [17], we performed Kaplan-Meier survival curve analysis to investigate the association between the two types of mortality and positive ERP in the inferior leads (ERP-inf+). The results showed that all-cause and cardiovascular mortality rates were similar for individuals with and without ERP in the inferior leads (log-rank test, P=0.58, 0.66, Figure 3C, 3D).
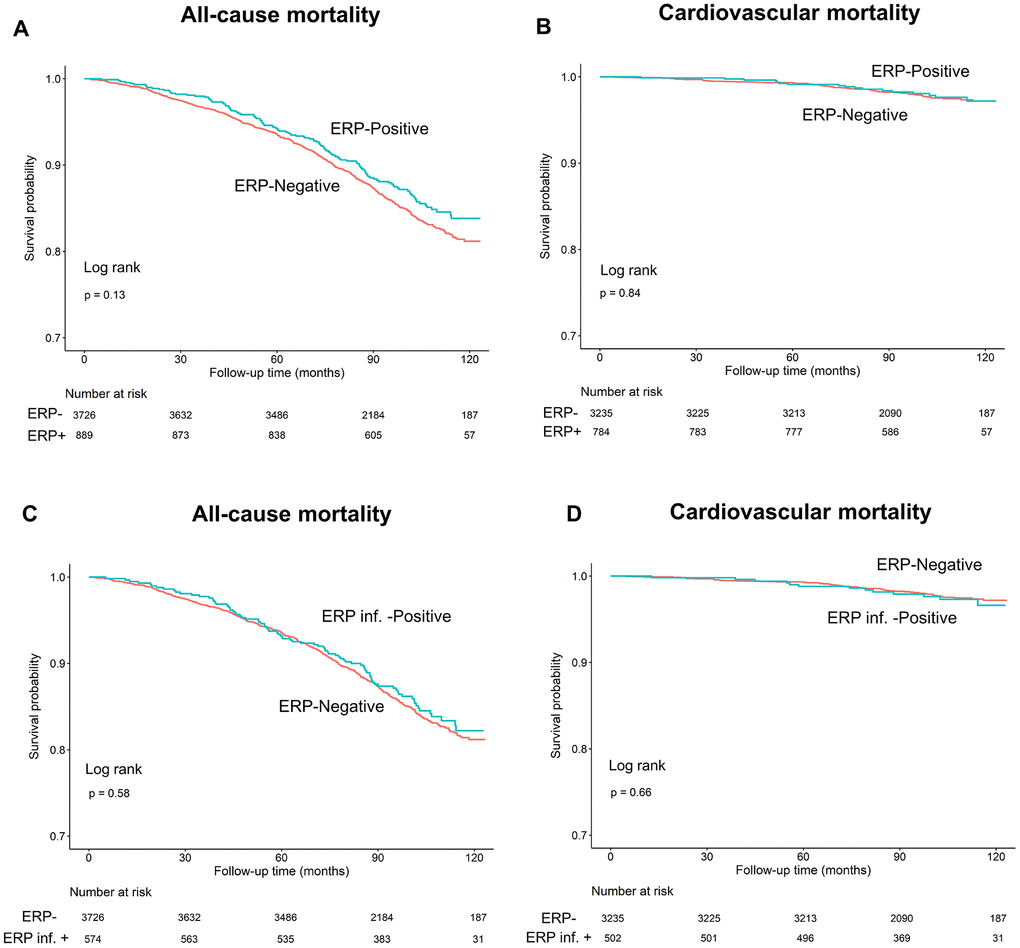
Figure 3. Kaplan–Meier survival curves show (A, C) all-cause and (B, D) cardiovascular mortality rates in individuals with and without early repolarization pattern, and individuals with and without early repolarization pattern in inferior leads.
Long-term outcomes of all-cause mortality and cardiovascular mortality and ERP stratified by age
Age is a known risk factor for both all-cause and cardiovascular mortality [18]. Therefore, we performed subgroup analyses in 3 age groups (55-64, 65-74, and ≥75 years) and observed no differences in all-cause or cardiovascular mortality rates in individuals with and without ERP in each subgroup (log-rank test, all P>0.05; Figure 4A, 4B). Moreover, the survival rates of individuals with and without ERP in the inferior leads among the 3 age subgroups were also similar (all P>0.05; Figure 5A, 5B).
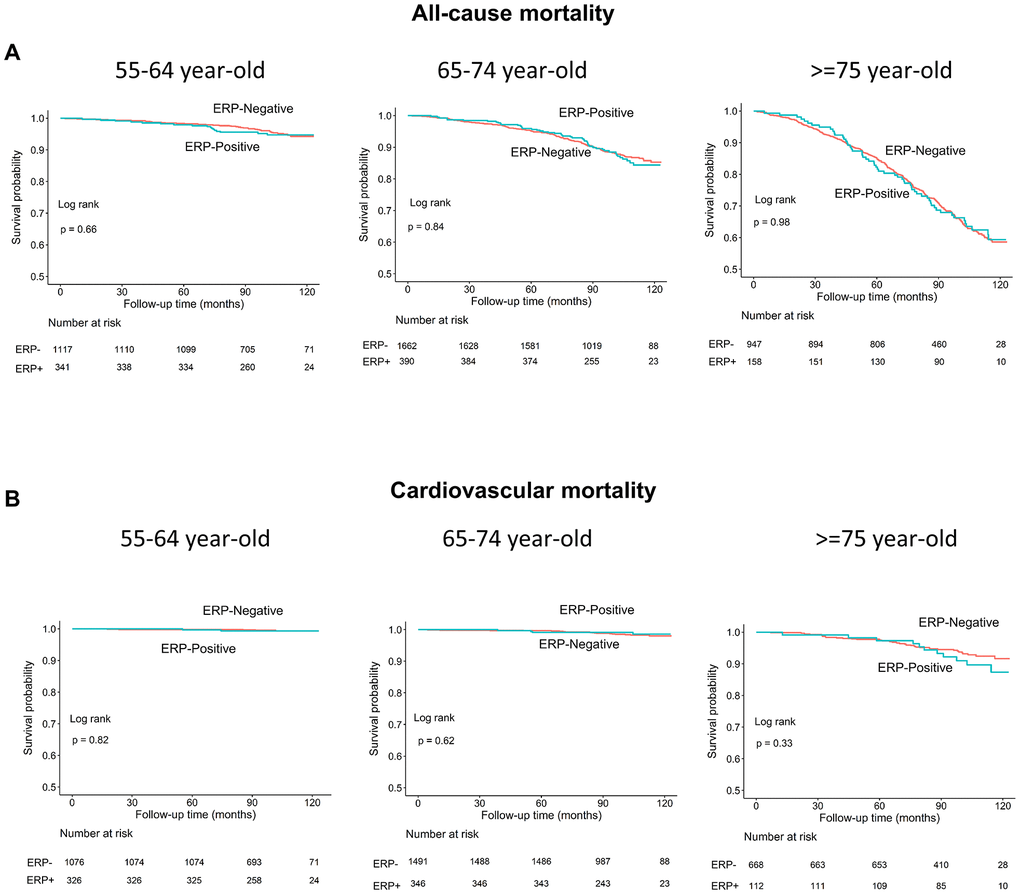
Figure 4. Kaplan–Meier survival curves show (A) all-cause and (B) cardiovascular mortality rates of individuals with and without early repolarization pattern stratified by age.
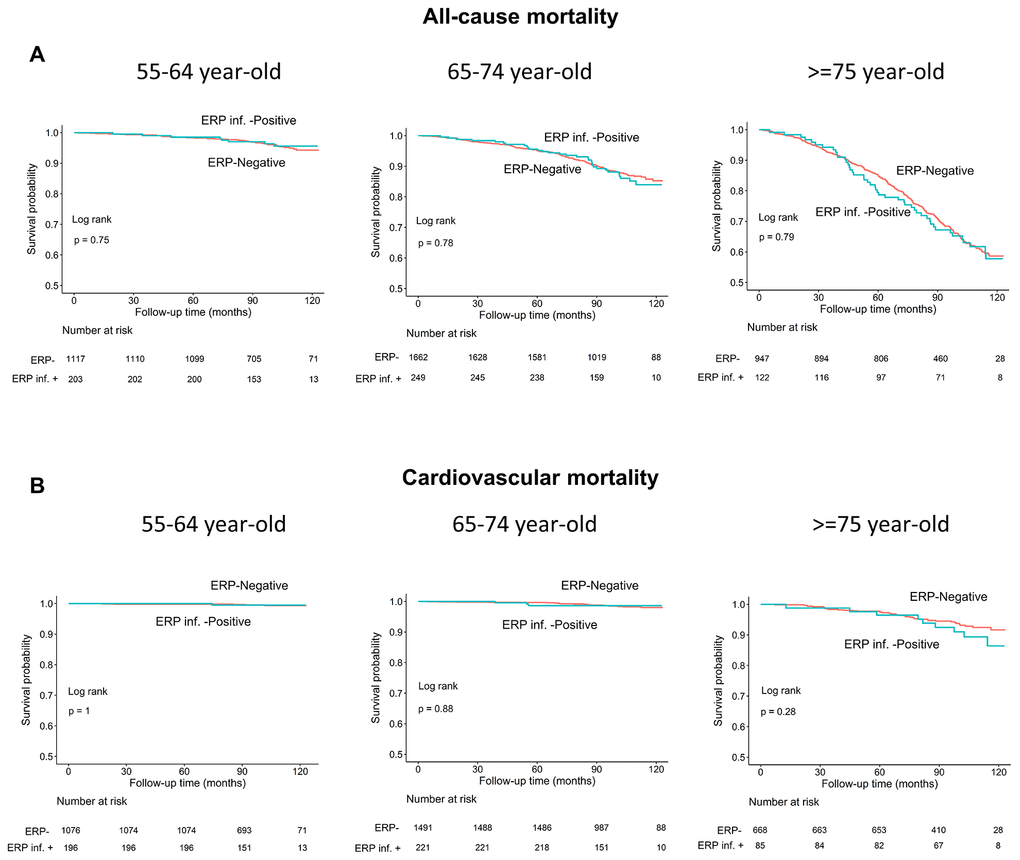
Figure 5. Kaplan–Meier survival curves show (A) all-cause and (B) cardiovascular mortality rates of individuals with and without early repolarization pattern in inferior leads stratified by age.
Correlation between ERP and other risk factors with all-cause and cardiovascular mortality
As shown in Tables 2, 3 and Supplementary Figures 1, 2, multivariate Cox proportional hazards analyses after adjusting for covariables showed that ERP was not a risk factor for both all-cause and cardiovascular disease mortality (P=0.12 and 0.7, respectively). Furthermore, our study showed that age, gender, BMI, SBP, DBP, smoking, diabetes mellitus, stroke, chronic kidney disease, and QTc were risk factors for all-cause mortality (all P values <0.05) and age and stroke were risk factors for cardiovascular mortality (all P values <0.05, Supplementary Figure 2). Similarly, after adjusting for covariables, multivariate analysis showed that individuals with ERP in inferior leads alone (ERP-inf +) were not associated with increased risk of all-cause and cardiovascular mortality (both P values >0.05; Supplementary Figures 3, 4). Moreover, multivariate analysis showed that patients with ERP in lateral leads alone (ERP-lat +) and ERP in both inferior leads and lateral leads (ERP-inf +/lat +) were associated with risk of all-cause mortality (P=0.029, 0.048, respectively; Supplementary Figures 5, 6), but were not associated with the risk of cardiovascular mortality (both P values >0.05; Supplementary Figures 7, 8) after adjusting for covariables. Although patients with ERP-inf +/lat + were associated with increased risk of all-cause mortality, the data was not sufficient to make a strong conclusion because only 5 deaths were reported in this group during the 10-year follow-up.
Table 2. Association between ERP and all-cause mortality*.
| Hazard ratio (95% CI) | P-value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ERP (reference: negative) | 0.98 (0.81, 1.20) | 0.877 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender (reference: male) | 0.71 (0.57, 0.87) | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.11 (1.10, 1.12) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI (kg/m2) | 0.96 (0.94, 0.98) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Systolic BP (mmHg) | 1.01 (1.01, 1.02) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diastolic BP (mmHg) | 0.99 (0.98, 0.99) | 0.003 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking (reference: current) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Quit | 0.63 (0.50, 0.79) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Never | 0.50 (0.40, 0.63) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes mellitus | 1.51 (1.26, 1.81) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stroke | 1.94 (1.52, 2.48) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chronic kidney disease | 1.35 (1.10, 1.66) | 0.005 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| QTc | 1.01 (1.00, 1.01) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age-stratified | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 55-64 y | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ERP (reference: negative) | 1.04 (0.58, 1.86) | 0.886 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender (reference: male) | 0.34 (0.16, 0.73) | 0.006 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking (reference: current) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Quit | 0.63 (0.31, 1.27) | 0.198 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Never | 0.40 (0.21, 0.79) | 0.008 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| QTc | 1.01 (1.00, 1.03) | 0.012 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 65-74 y | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ERP (reference: negative) | 1.01 (0.73, 1.39) | 0.953 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender (reference: Male) | 0.56 (0.39, 0.80) | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Systolic BP (mmHg) | 1.02 (1.00, 1.03) | 0.008 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diastolic BP (mmHg) | 0.97 (0.96, 0.99) | 0.004 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking (reference: current) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Quit | 0.66 (0.45, 0.97) | 0.032 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Never | 0.44 (0.31, 0.64) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stroke | 1.98 (1.33, 2.95) | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| QTc | 1.01 (1.00, 1.01) | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≥75 y | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ERP (reference: negative) | 0.87 (0.65, 1.17) | 0.357 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI (kg/m2) | 0.92 (0.89, 0.95) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Systolic BP (mmHg) | 1.02 (1.01, 1.03) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diastolic BP (mmHg) | 0.98 (0.97, 0.99) | 0.002 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking (reference: current) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Quit | 0.78 (0.55, 1.09) | 0.150 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Never | 0.68 (0.48, 0.96) | 0.030 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes mellitus | 1.42 (1.11, 1.81) | 0.005 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stroke | 1.89 (1.35, 2.64) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| QTc | 1.01 (1.00, 1.01) | 0.008 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ERP: early repolarization pattern; BMI: body mass index; BP: blood pressure; CI, confidence interval; *Except for ERP, only significant variables (P<0.05) are shown here; QTc was calculated by Bazett’s equations. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 3. Association between ERP and cardiovascular mortality*.
| Hazard ratio (95% CI) | P-value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ERP (reference: negative) | 1.27 (0.74, 2.20) | 0.386 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.13 (1.10, 1.17) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stroke | 2.11 (1.01, 4.42) | 0.048 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age-stratified | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 55-64 y | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ERP (reference: negative) | 1.08 (0.21, 5.68) | 0.928 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI | 1.23 (1.02, 1.47) | 0.027 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CKD | 4.83 (1.03, 22.61) | 0.046 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 65-74 y | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ERP (reference: negative) | 0.83 (0.28, 2.47) | 0.744 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stroke | 3.59 (1.30, 9.92) | 0.014 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| QTc | 1.02 (1.00, 1.04) | 0.017 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≥75 y | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ERP (reference: negative) | 1.54 (0.77, 3.07) | 0.219 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Systolic BP (mmHg) | 1.03 (1.01, 1.05) | 0.014 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ERP: early repolarization pattern; BMI: body mass index; BP: blood pressure; CKD: chronic kidney disease; *Except for ERP, only significant variables (P<0.05) are shown here. QTc was calculated by Bazett’s equations. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Since age is a strong predictor of both all-cause and cardiovascular mortality [18], we further compared mortality rates by stratifying ERP (+) and ERP (-) individuals into 3 age subgroups (55-64, 65-74, and ≥75 years). Multivariate Cox proportional hazard analyses after adjusting for covariables showed that ERP was not a risk factor for both all-cause and cardiovascular mortality in all the age subgroups (all P values >0.05; Tables 2, 3, Supplementary Figures 9–14). Moreover, study subjects with ERP in inferior leads alone (ERP-inf +), lateral leads alone (ERP-lat +), or both inferior and lateral leads (ERP-inf +/lat +) were not associated with increased risk of both all-cause and cardiovascular mortality in all age subgroups (all P values >0.05; Supplementary Figure 15–32).
Comparisons of the prevalence and clinical outcomes of ERP in community-based studies worldwide
As shown in Table 4, the prevalence of ERP ranged from ~1% to 25% in Caucasians and 3.5%-24% in Japanese according to the community based population studies worldwide [10, 13, 14, 16]. However, the association between all-cause and cardiovascular mortality and EPR was not consistent among the published studies. Most studies showed that ERP was not associated with all-cause mortality but was associated with cardiovascular mortality. Our study cohort included older middle-aged and geriatric individuals ≥55 years with an average age of 69.1±8.2 years (range: 55-103 years), which was the highest age average of a study cohort when compared with previous reports. Moreover, the prevalence of ERP was 19.26% in our study cohort. This was within the prevalence range reported by other studies (0.99-24.79%), but was higher than the mean worldwide prevalence of 6.42%. Overall, our study shows that ERP is not associated with increased risk of all-cause and cardiovascular mortality in older middle aged and geriatric Han-Chinese population in Taiwan.
Table 4. Summary of results from community-based studies worldwide regarding the prevalence of ERP, age distribution, and relationship with all-cause and cardiovascular mortality.
| Study | Year | Country | Screened population N | Male (%) | Age (years) | ERP (+) | All-cause mortality | Cardiovascular mortality | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N (%) | M (%) | F (%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| North America | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Klatsky et al.[10] | 2003 | USA | 73,088 | 43.88 | NA | 670 (0.92) | 583 (1.82) | 87 (0.21) | Not increased | Increased | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Olson et al.[19] | 2011 | USA | 15,141 | 44.30 | 54.1 | 1866 (12.3) | 1420 (21.17) | 446 (5.29) | Not increased | SCD increased in whites and females | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Uberoi et al.[40] | 2011 | USA | 29,281 | 87.24 | 55±15 | 714 (2.44) | 661 (2.59) | 53 (1.42) | NA | Not increased | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Perez et al.[41] | 2012 | USA | 29,281 | 87 | 55 | 664 (2.27) | NA | NA | NA | Increased in non-African Americans but not in African Americans | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ilkhanoff et al.[13] | 2014 | USA | 5,039 | 45.54 | 25 (18-30) | 1249 (24.79) | 1139 (49.63) | 110 (4.01) | Not increased | Not increased | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pargaonkar et al.[42] | 2015 | USA | 20,661 | 90.53 | 20-55 | 4219 (20) | 3840 (20.53) | 379 (19.38) | NA | Not increased | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Leiderman et al.[43] | 2019 | USA | 17,901 | 38.57 | 53±13 | 995 (5.6) | 489 (7.08) | 506 (4.60) | Not increased | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Europe | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tikkanen et al.[25] | 2009 | Finland | 10,864 | 52.40 | 44±8 | 630 (5.8) | 407 (7.15) | 223 (4.31) | NA | Increased | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sinner et al.[15] | 2010 | Germany | 6,213 | 48.85 | 35-74 | 812 (13.1) | 439 (14.46) | 373 (11.74) | Increased | Increased | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rollin et al.[20] | 2012 | France | 1,161 | 51.59 | 35-64 | 159 (13.7) | 126 (21.04) | 33 (5.87) | Increased | Increased | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Asia | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Haruta et al.[16] | 2011 | Japan | 5,676 | 43.71 | 47.2±15.4 | 779 (23.9) | 815 (31.20) | 614 (18.25) | Decreased | Decreased | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hisamatsu et al.[14] | 2013 | Japan | 7,630 | 40.73 | 52.4 (30-95) | 264 (3.5) | 253 (8.14) | 11 (0.24) | NA | Cardiac death and death from CAD increased | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Juang et al.present study | 2020 | Taiwan | 4,615 | 47.78 | 69.1±8.2 (55-103) | 889 (19.26) | 489 (7.08) | 506 (4.60) | Not increased | Not increased | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total | - | - | 226,851 | - | 14,560 (6.42) | 10,642 (9.72) | 3,254 (3.69) | - | - | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ERP: early repolarization pattern; NA: not available, means no number was provided in the papers; USA: United States of America; M: male; F: female; SCD: sudden cardiac death; CAD: coronary artery disease | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Discussion
The prevalence of cardiovascular disease is expected to increase because of a constant rise in the proportion of older individuals worldwide. ERP is not a rare event and has been reported in studies related to several ethnicities [19, 20], and has been associated with sudden cardiac arrest [15, 20]. To the best of our knowledge, this is the first study to examine the prevalence and prognostic value of ERP in a large cohort of older middle-aged and elderly individuals with a mean age ≥65 years.
Previous studies show that prevalence of ERP varies from ~1% to 25% in a general population [10, 13, 14, 16]. This wide range of ERP prevalence reflects differences in age range, proportions of male subjects, and inconsistent definition of ERP between studies. In the MONICA/KORA study that included 6,213 participants between 35-74 years, ERP prevalence of individuals above 55 years was 6.29% [15]. In the Japanese NIPPON DATA90 study with 7,630 participants aged 30-95 years, ERP prevalence of individuals above 60 years was 2.5% [14]. The number of participants in the older middle-aged and elderly population (aged 55-103 years) was highest in our study compared to previous studies. The prevalence of ERP in this age group was 19.2%— the highest overall ERP prevalence among all community-based studies that included study subjects with a mean age > 55 years. This may suggest that aging causes degeneration of the cardiac conduction pathways [21] or increased fibrosis and fat deposition within the heart of elderly patients [22].
In the past years, the association between ERP and mortality has been tested in general populations by several epidemiologic studies [13, 15, 23–25]. The results are inconsistent and conflicting. The MONICA/KORA study screened 6,213 individuals aged 35-74 years and showed increased all-cause and cardiovascular mortality in individuals with ERP [15]. The CARDIA study enrolled 5,039 biracial young adults aged 18-30 years with a follow-up time >20 years and demonstrated that ERP was not associated with all-cause and cardiovascular mortality [13]. The CHD study consisting of 10,864 individuals aged 30-59 years concluded that ERP-positive in the inferior leads was associated with increased risk of cardiac mortality, but was not related to all-cause mortality [25]. A meta-analysis of 16 studies including 4 case-control and 12 prospective or retrospective studies (334,524 individuals from multiple ethnicities) showed that ERP was an electrocardiographic risk marker for deaths related to arrhythmia, cardiac diseases, and all-causes [23, 24]. However, there was considerable heterogeneity because of differences in study designs, ethnicity and potential misdiagnosis of ERP. Moreover, majority of these duties did not analyze older middle-aged and geriatric individuals. In the Japanese NIPPON DATA90 study of 2,433 individuals older than 60 years, subgroup analysis showed that J-point elevation was not associated with cardiovascular death or death from coronary artery disease [14]. Our random-sampling community-based cohort study specifically enrolled older middle-aged and elderly individuals from the Han Chinese population, and prospectively followed up these individuals for 11years with less than 1% dropouts. Our analysis showed that ERP was not associated with both all-cause and cardiovascular mortality.
ECG is globally used as an inexpensive and noninvasive technique to detect electric abnormalities of the heart. Several individuals receive annual health examinations including ECG. ERP is an incidental and common finding on an ECG during a routine health examination. Our study provides an important reference for clinicians or health care providers when they encounter asymptomatic elderly individuals with an incidental ERP and without a family history of SCD in members younger than 40 years.
The results of the association between ERP and mortality in several epidemiologic studies are inconsistent and conflicting [13, 15, 23–25]. The possible reasons may be due to heterogeneity in observational studies, differences in study designs, as well as differences in age and ethnicity of the study subjects. Therefore, prospective long-term randomized clinical trials (RCTs) in different age groups (30-40 year or 60-70 year-olds) in specific ethnic populations are required in the future to minimize all possible confounding factors. Moreover, combining ERP with other ECG risk parameters (such as QTc interval) may provide better risk stratification of large community-based cohorts compared to ERP as a single ECG risk factor.
There are several limitations in our study. Firstly, our findings may not apply to younger individuals because our study consisted of individuals above the age of 55 years. Secondly, the HALST cohort was restricted to individuals of Han Chinese ethnicity, and may not be applicable to other ethnicities. Thirdly, detailed clinical information including echocardiographic assessments, coronary angiography, and medications were not available in the HALST database because this data was obtained during screening.
In summary, our study shows that prevalence of ERP in a standard 12-lead ECG is common in relatively healthy, community-dwelling, ambulatory individuals above 55 years. Moreover, ERP alone is not associated with all-cause and cardiovascular mortality.
Materials and Methods
Study subjects and inclusion criteria
Majority (>95%) of Taiwanese are of Han Chinese ancestry and nearly 2% are of aboriginal Austronesian ancestry [26]. In this study, we initially included 5,380 Han Chinese individuals of the HALST study cohort and excluded all aboriginal Taiwanese subjects [27–29]. This study was approved by the Ethics Committee of the National Health Research Institutes and conducted according to the principles of the Declaration of Helsinki. We obtained signed informed consent from the study subjects. The study cohort represented a random sample of the entire national population and was selected based on citizen IDs from 7 communities in the Northern, Central, Southern, and Eastern regions of Taiwan. The eligible and willing participants were enrolled from December 2008 to March 2013. At the time of enrollment, we prospectively collected three serial 12-lead ECGs and other relevant clinical and demographic information from the study subjects.
We then excluded individuals with cancer, significant heart diseases such as myocardial infarction and severe valvular diseases, ventricular conduction delay (left or right bundle branch block or QRS>120 ms), atrial fibrillation or flutter, QTc interval corrected for heart rate with Bazett's formula (QTc) of less than 340 msec (short QT interval) or more than 440 msec (long QT interval) at baseline and before arrhythmia [30, 31], Brugada type 1 ECG [32], catecholaminergic arrhythmias [33], ventricular pre-excitation, and implanted pacemakers. The final study cohort consisted of 4,615 individuals, which was followed up for 95.1±21.9 months. During follow-up, 770 study subjects died of cardiac (n=87) or other causes (n=683).
All participants were prospectively followed on a regular basis according to the HALST study framework guidelines and the follow-up information was available until April 2019. We obtained death certificates from the National Taiwan Ministry of Health and Welfare and evaluated the cause using the 10th revision of the International Classification of Diseases (ICD-10). The death from cardiac causes was defined by ICD codes, I01-I02.0, I05-I09, I20-I25, I27, and I30-I52.
ECG recording and definition of ERP
The 12-lead ECGs (Hewlett Packard, USA) were recorded using standard settings of 10 mm/mV and 25 mm/s. PR and QRS were computed automatically, whereas, QTc was computed using the Bazett’s formula [34]. ERP was defined as a J-point (QRS-ST junction) elevation of at least ≥0.1 mV from baseline in ≥2 adjacent leads with either QRS slurring or notching morphology, as described by Haissaguerre et al. and the 2015 consensus criteria [7, 35]. The anterior precordial leads (V1 to V3) were excluded from the analysis to avoid inclusion of patients with right ventricular arrhythmogenic dysplasia or the Brugada syndrome [36, 37].
ECGs were displayed on a 24-inch computer screen in multiple formats to enable careful classification of slurring on the downslope of the R and J waves. All ECGs were independently analyzed and interpreted in a random order by two trained cardiologists who were blinded to clinical data and the follow-up status. In case of divergent results, a third blinded cardiologist re-interpreted the ECG and a preliminary ERP status was achieved by a majority vote. After the preliminary decision on ERP status, two trained cardiologists jointly reassessed all the ECGs that were considered ERP-positive and reached a final consensus decision on the ERP status.
Long-term prognosis and follow-up
We collected clinical data regarding history of unexplained syncope, circumstances of sudden cardiac arrest, and a family history of unexplained sudden death (at <40 years of age).
The primary end point was death from cardiac causes, and the secondary end point was death from any cause before April 2019. An annual follow-up telephone interview of all the study participants was carried out by the study nurses to determine any new cardiovascular or frailty-related events after the initial enrollment. The cause of deaths was determined by examining the death certificates from the National Taiwan Institute of Health and Welfare and reviewed by 2 clinicians blinded to the ECG results. The cause of sudden death from arrhythmia was adjudicated by a committee of experienced cardiologists who were blinded to the data from the ECG analyses.
Statistical analysis
We used the Fisher exact test to compare categorical variables and Student’s t test to compare continuous data between the groups. Linear regression was used for continuous variables and logistic regression was used for dichotomous variables to determine the relationship between ERP and mortality. Multivariate analysis was performed to determine hazard ratios (HRs) and confidence intervals (CIs). The models were primarily adjusted for age and sex, and further adjusted for covariates that were selected on the basis of previous evidence of an association with death from cardiovascular causes or other causes. This included continuous variables such as BMI, SBP, DBP, and QTc, and categorical variables such as smoking status (current, quit, never), hypertension, diabetes mellitus, hyperlipidemia, stroke, and chronic kidney disease. The relationship between ERP and mortality was calculated using a weighted Cox proportional hazards model [38]. Kaplan-Meier survival curves and log rank tests were used to determine survival times of different groups of individuals. Clinical factors such as gender, age, smoking, SBP, DBP, BMI, stroke, diabetes mellitus, hypertension, hyperlipidemia, and chronic kidney disease were used for multivariable analysis. P < 0.05 was considered statistically significant. Inter-observer agreement was based on the overall proportion of agreements and the Kappa statistic across all ECGs. Statistical analyses were performed using the R software, version 4.0.1 (R Foundation for Statistical Computing) [39].
Author Contributions
Conceptualization and study design: SFSY, JJMJ, and CYJC; Enrollment of study subjects, data collection, and laboratory work: CAH, ISC, ICW, CCH, TYC, and WTT; Data analysis and interpretation: SFSY, JJMJ, CYJC; Resources and supervision: CAH and JJMJ; Manuscript writing, review, and editing: SFSY and JJMJ.
Acknowledgments
We are sincerely grateful to the advisory committee of the HALST study (Drs. Luigi Ferrucci, Jack M. Guralnik, Dilip V. Jeste, and Kung-Yee Liang) for their valuable suggestions. We thank all study participants. We also thank members of the HALST study group, including Drs. Chao-Agnes Hsiung, Chih-Cheng Hsu, I-Chien Wu, Hsing-Yi Chang, Chu-Chih Chen, Yen-Feng Chiu, Hui-Ju Tsai, and Ken N. Kuo, of National Heath Research Institutes; Dr. Ching-Yu Chen of National Taiwan University; Dr. Kiang Liu of Northwestern University Medical School; Dr. Marion Lee of University of California at San Francisco; Dr. Ida Chen of Cedars-Sinai Medical Center; Dr. Shu-Han Yu of Chung Shan Medical University; Dr. Kai-Ting Ko of Mackay Memorial Hospital; Dr. Tzuo-Yun Lan of National Yang-Ming University; Dr. Hou-Chang Chiu of Shin Kong Wu Ho-Su Memorial Hospital; Dr. Wen-Jin Liaw of Yee Zen General Hospital; Dr. Yo-Hann Liu of Hope Doctors Hospital; Dr. I-Ching Lin of Changhua Christian Hospital; Dr. Ping-An Wu of Potz General Hospital; Dr. Chon-Chou Juan of Yuan's General Hospital; Drs. Chi-Chung Wang and Shi-Chen Shen of Mennonite Christian Hospital; and Dr. Huey-Ming Lo of Fu-Jen Catholic University Hospital. We thank the staff of the Sixth Core Lab, Department of Medical Research, National Taiwan University Hospital, and Taiwan Health foundation for technical support. We also thank Melissa Stauffer of the Scientific Editing Solutions for editing the manuscript.
Conflicts of Interest
The authors declare that there are no conflicts of interest.
Funding
This study was supported financially by the grants from the National Health Research Institutes of Taiwan (Grant numbers: PH-101-SP-01, PH-102-SP-01, and PH-103-SP-01), Ministry of Science and Technology of Taiwan (Grant numbers: MOST 106-2314-B-002-134-MY2, MOST 106-2314-B-002-206, MOST 107-2314-B-002-009, and MOST 107-2314-B-002-261-MY3), the Taiwan Health Foundation, and National Taiwan University Hospital (Grant numbers: NTUH-UN105-012 and NTUH 106-018).
References
- 1. Chugh SS, Reinier K, Teodorescu C, Evanado A, Kehr E, Al Samara M, Mariani R, Gunson K, Jui J. Epidemiology of sudden cardiac death: clinical and research implications. Prog Cardiovasc Dis. 2008; 51:213–28. https://doi.org/10.1016/j.pcad.2008.06.003 [PubMed]
- 2. Stecker EC, Reinier K, Marijon E, Narayanan K, Teodorescu C, Uy-Evanado A, Gunson K, Jui J, Chugh SS. Public health burden of sudden cardiac death in the United States. Circ Arrhythm Electrophysiol. 2014; 7:212–7. https://doi.org/10.1161/CIRCEP.113.001034 [PubMed]
- 3. Zipes DP, Wellens HJ. Sudden cardiac death. Circulation. 1998; 98:2334–51. https://doi.org/10.1161/01.cir.98.21.2334 [PubMed]
- 4. Survivors of out-of-hospital cardiac arrest with apparently normal heart. Need for definition and standardized clinical evaluation. Consensus statement of the joint steering committees of the unexplained cardiac arrest registry of Europe and of the idiopathic ventricular fibrillation registry of the United States. Circulation. 1997; 95:265–72. https://doi.org/10.1161/01.cir.95.1.265 [PubMed]
- 5. Jimmy JJ, Chen CY, Yeh HM, Chiu WY, Yu CC, Liu YB, Tsai CT, Lo LW, Yeh SF, Lai LP. Clinical characteristics of patients with congenital long QT syndrome and bigenic mutations. Chin Med J (Engl). 2014; 127:1482–86. [PubMed]
- 6. Gussak I, Antzelevitch C. Early repolarization syndrome: clinical characteristics and possible cellular and ionic mechanisms. J Electrocardiol. 2000; 33:299–309. https://doi.org/10.1054/jelc.2000.18106 [PubMed]
- 7. Haïssaguerre M, Derval N, Sacher F, Jesel L, Deisenhofer I, de Roy L, Pasquié JL, Nogami A, Babuty D, Yli-Mayry S, De Chillou C, Scanu P, Mabo P, et al. Sudden cardiac arrest associated with early repolarization. N Engl J Med. 2008; 358:2016–23. https://doi.org/10.1056/NEJMoa071968 [PubMed]
- 8. Nam GB, Kim YH, Antzelevitch C. Augmentation of J waves and electrical storms in patients with early repolarization. N Engl J Med. 2008; 358:2078–79. https://doi.org/10.1056/NEJMc0708182 [PubMed]
- 9. Rosso R, Kogan E, Belhassen B, Rozovski U, Scheinman MM, Zeltser D, Halkin A, Steinvil A, Heller K, Glikson M, Katz A, Viskin S. J-point elevation in survivors of primary ventricular fibrillation and matched control subjects: incidence and clinical significance. J Am Coll Cardiol. 2008; 52:1231–38. https://doi.org/10.1016/j.jacc.2008.07.010 [PubMed]
- 10. Klatsky AL, Oehm R, Cooper RA, Udaltsova N, Armstrong MA. The early repolarization normal variant electrocardiogram: correlates and consequences. Am J Med. 2003; 115:171–77. https://doi.org/10.1016/s0002-9343(03)00355-3 [PubMed]
- 11. Mehta M, Jain AC, Mehta A. Early repolarization. Clin Cardiol. 1999; 22:59–65. https://doi.org/10.1002/clc.4960220203 [PubMed]
- 12. Mehta MC, Jain AC. Early repolarization on scalar electrocardiogram. Am J Med Sci. 1995; 309:305–11. https://doi.org/10.1097/00000441-199506000-00001 [PubMed]
- 13. Ilkhanoff L, Soliman EZ, Prineas RJ, Walsh JA
3rd , Ning H, Liu K, Carr JJ, Jacobs DRJr , Lloyd-Jones DM. Clinical characteristics and outcomes associated with the natural history of early repolarization in a young, biracial cohort followed to middle age: the Coronary Artery Risk Development in Young Adults (CARDIA) study. Circ Arrhythm Electrophysiol. 2014; 7:392–9. https://doi.org/10.1161/CIRCEP.113.000874 [PubMed] - 14. Hisamatsu T, Ohkubo T, Miura K, Yamamoto T, Fujiyoshi A, Miyagawa N, Kadota A, Takashima N, Nagasawa SY, Kita Y, Murakami Y, Okayama A, Horie M, et al, and NIPPON DATA90 Research Group. Association between J-point elevation and death from coronary artery disease—15-year follow up of the NIPPON DATA90. Circ J. 2013; 77:1260–66. https://doi.org/10.1253/circj.cj-12-1273 [PubMed]
- 15. Sinner MF, Reinhard W, Müller M, Beckmann BM, Martens E, Perz S, Pfeufer A, Winogradow J, Stark K, Meisinger C, Wichmann HE, Peters A, Riegger GA, et al. Association of early repolarization pattern on ECG with risk of cardiac and all-cause mortality: a population-based prospective cohort study (MONICA/KORA). PLoS Med. 2010; 7:e1000314. https://doi.org/10.1371/journal.pmed.1000314 [PubMed]
- 16. Haruta D, Matsuo K, Tsuneto A, Ichimaru S, Hida A, Sera N, Imaizumi M, Nakashima E, Maemura K, Akahoshi M. Incidence and prognostic value of early repolarization pattern in the 12-lead electrocardiogram. Circulation. 2011; 123:2931–37. https://doi.org/10.1161/CIRCULATIONAHA.110.006460 [PubMed]
- 17. Haïssaguerre M, Sacher F, Nogami A, Komiya N, Bernard A, Probst V, Yli-Mayry S, Defaye P, Aizawa Y, Frank R, Mantovan R, Cappato R, Wolpert C, et al. Characteristics of recurrent ventricular fibrillation associated with inferolateral early repolarization role of drug therapy. J Am Coll Cardiol. 2009; 53:612–19. https://doi.org/10.1016/j.jacc.2008.10.044 [PubMed]
- 18. Menotti A, Mulder I, Nissinen A, Feskens E, Giampaoli S, Tervahauta M, Kromhout D. Cardiovascular risk factors and 10-year all-cause mortality in elderly European male populations; the FINE study. Finland, Italy, Netherlands, Elderly. Eur Heart J. 2001; 22:573–9. https://doi.org/10.1053/euhj.2000.2402 [PubMed]
- 19. Olson KA, Viera AJ, Soliman EZ, Crow RS, Rosamond WD. Long-term prognosis associated with J-point elevation in a large middle-aged biracial cohort: the ARIC study. Eur Heart J. 2011; 32:3098–106. https://doi.org/10.1093/eurheartj/ehr264 [PubMed]
- 20. Rollin A, Maury P, Bongard V, Sacher F, Delay M, Duparc A, Mondoly P, Carrié D, Ferrières J, Ruidavets JB. Prevalence, prognosis, and identification of the Malignant form of early repolarization pattern in a population-based study. Am J Cardiol. 2012; 110:1302–08. https://doi.org/10.1016/j.amjcard.2012.06.033 [PubMed]
- 21. Thrainsdottir IS, Hardarson T, Thorgeirsson G, Sigvaldason H, Sigfusson N. The epidemiology of right bundle branch block and its association with cardiovascular morbidity—the reykjavik study. Eur Heart J. 1993; 14:1590–96. https://doi.org/10.1093/eurheartj/14.12.1590 [PubMed]
- 22. Biernacka A, Frangogiannis NG. Aging and Cardiac Fibrosis. Aging Dis. 2011; 2:158–173. [PubMed]
- 23. Cheng YJ, Lin XX, Ji CC, Chen XM, Liu LJ, Tang K, Wu SH. Role of early repolarization pattern in increasing risk of death. J Am Heart Assoc. 2016; 5:e003375. https://doi.org/10.1161/JAHA.116.003375 [PubMed]
- 24. Wu SH, Lin XX, Cheng YJ, Qiang CC, Zhang J. Early repolarization pattern and risk for arrhythmia death: a meta-analysis. J Am Coll Cardiol. 2013; 61:645–50. https://doi.org/10.1016/j.jacc.2012.11.023 [PubMed]
- 25. Tikkanen JT, Anttonen O, Junttila MJ, Aro AL, Kerola T, Rissanen HA, Reunanen A, Huikuri HV. Long-term outcome associated with early repolarization on electrocardiography. N Engl J Med. 2009; 361:2529–37. https://doi.org/10.1056/NEJMoa0907589 [PubMed]
- 26. Executive Yuan tRoCT. The Republic of China Yearbook., 2013.
- 27. Wu IC, Chang HY, Hsu CC, Chiu YF, Yu SH, Tsai YF, Shen SC, Kuo KN, Chen CY, Liu K, Lee MM, Hsiung CA. Association between dietary fiber intake and physical performance in older adults: a nationwide study in Taiwan. PLoS One. 2013; 8:e80209. https://doi.org/10.1371/journal.pone.0080209 [PubMed]
- 28. Juang JM, Chen CY, Chen YH, Wu IC, Hsu CC, Chen LN, Tang FC, Wang CC, Juan CC, Chiu HC, Lo HM, Chang IS, Hwang JJ, et al. Prevalence and prognosis of brugada electrocardiogram patterns in an elderly Han Chinese population: a nation-wide community-based study (HALST cohort). Europace. 2015 (Suppl 2); 17:ii54–62. https://doi.org/10.1093/europace/euv141 [PubMed]
- 29. Chen CJ, Juang JJ, Chen YH, Wu IC, Hsu CC, Wu RC, Chen KC, Liaw WJ, Tsai TL, Lin LY, Hwang JJ, Ho LT, Yu CC, et al. Comparisons of clinical impacts on individuals with brugada electrocardiographic patterns defined by ISHNE criteria or EHRA/HRS/APHRS criteria: a nationwide community-based study. Ann Med. 2018; 50:7–15. https://doi.org/10.1080/07853890.2017.1353222 [PubMed]
- 30. Moss AJ, Schwartz PJ, Crampton RS, Locati E, Carleen E. The long QT syndrome: a prospective international study. Circulation. 1985; 71:17–21. https://doi.org/10.1161/01.cir.71.1.17 [PubMed]
- 31. Gaita F, Giustetto C, Bianchi F, Wolpert C, Schimpf R, Riccardi R, Grossi S, Richiardi E, Borggrefe M. Short QT Syndrome: a familial cause of sudden death. Circulation. 2003; 108:965–70. https://doi.org/10.1161/01.CIR.0000085071.28695.C4 [PubMed]
- 32. Berne P, Brugada J. Brugada syndrome 2012. Circ J. 2012; 76:1563–71. https://doi.org/10.1253/circj.cj-12-0717 [PubMed]
- 33. Cerrone M, Napolitano C, Priori SG. Catecholaminergic polymorphic ventricular tachycardia: a paradigm to understand mechanisms of arrhythmias associated to impaired Ca(2+) regulation. Heart Rhythm. 2009; 6:1652–59. https://doi.org/10.1016/j.hrthm.2009.06.033 [PubMed]
- 34. Bazett HC. An analysis of the time-relations of electrocardiograms. Annals of Noninvasive Electrocardiology. 1997; 2:177–194. https://doi.org/10.1111/j.1542-474X.1997.tb00325.x
- 35. Macfarlane PW, Antzelevitch C, Haissaguerre M, Huikuri HV, Potse M, Rosso R, Sacher F, Tikkanen JT, Wellens H, Yan GX. The Early Repolarization Pattern: A Consensus Paper. J Am Coll Cardiol. 2015; 66:470–7. https://doi.org/10.1016/j.jacc.2015.05.033 [PubMed]
- 36. Corrado D, Basso C, Thiene G. Sudden cardiac death in young people with apparently normal heart. Cardiovasc Res. 2001; 50:399–408. https://doi.org/10.1016/s0008-6363(01)00254-1 [PubMed]
- 37. Brugada J, Brugada R, Brugada P. Right bundle-branch block and ST-segment elevation in leads V1 through V3: a marker for sudden death in patients without demonstrable structural heart disease. Circulation. 1998; 97:457–60. https://doi.org/10.1161/01.cir.97.5.457 [PubMed]
- 38. Cox DR. Regression Models and Life-Tables. Journal of the Royal Statistical Society Series B (Methodological). 1972; 34:187–220. https://doi.org/10.1111/j.2517-6161.1972.tb00899.x
- 39. (2020) RCT. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.r-project.org/.
- 40. Uberoi A, Jain NA, Perez M, Weinkopff A, Ashley E, Hadley D, Turakhia MP, Froelicher V. Early repolarization in an ambulatory clinical population. Circulation. 2011; 124:2208–14. https://doi.org/10.1161/CIRCULATIONAHA.111.047191 [PubMed]
- 41. Perez MV, Uberoi A, Jain NA, Ashley E, Turakhia MP, Froelicher V. The prognostic value of early repolarization with ST-segment elevation in African Americans. Heart Rhythm. 2012; 9:558–65. https://doi.org/10.1016/j.hrthm.2011.11.020 [PubMed]
- 42. Pargaonkar VS, Perez MV, Jindal A, Mathur MB, Myers J, Froelicher VF. Long-term prognosis of early repolarization with j-wave and QRS slur patterns on the resting electrocardiogram: a cohort study. Ann Intern Med. 2015; 163:747–55. https://doi.org/10.7326/M15-0598 [PubMed]
- 43. Leiderman E, Kargoli F, Shulman E, Aagaard P, Hoch E, Zaremski L, Di Biase L, Kim SG, Gross JN, Ferrick KJ, Fisher J, Krumerman A. Early repolarization pattern in an ethnically diverse population: increased risk in hispanics. Pacing Clin Electrophysiol. 2020; 43:30–36. https://doi.org/10.1111/pace.13827 [PubMed]


