Introduction
Ischemic stroke is a devastating complication in patients with heart failure (HF). [1] HF is associated with over a 2-fold increased risk of ischemic stroke than those without. [2] Notably, risk of ischemic stroke could increase 17-fold within the first 30 days after HF diagnosis. [3, 4].
Stroke risk stratification using readily available clinical variables could help identify “low-risk” and “high-risk” subgroups in HF population for more regular anticoagulant strategies for stroke prevention. [5] CHADS2 and CHA2DS2-VASc scores was a widely useful scoring system for stratifying ischemic stroke risks in patients with atrial fibrillation (AF). [6, 7] CHADS2 and CHA2DS2-VASc, as a cluster of multiple stroke risk factors, was also extensively used to predict stroke in patients with heart failure with reduced ejection fraction (HFrEF) but without AF. [8–10] A potential limitation of these approaches is selection bias, because clinical features associated with non-use of anticoagulation are likely to influence stroke risk. Prior-stroke history has been shown as the most powerful independent risk factor for improving stroke prediction. However, prognostic ability of established risk stratification scores benefited from prior-stoke history may confer low-prognostic efficiency in patients without stroke history. Additionally, all patients, who have had an ischemic stroke history are at high risk of future stroke regardless of presence of AF, and for whom anticoagulation is strongly indicated. Arguably, clinicians are particularly interested in risk stratifying patients without stroke history for ischemic stroke and how much stroke risk can be reduced in those undergoing anticoagulant management.
Therefore, this study aims to develop a nomogram to predict the probability of first-ever ischemic stroke in HFrEF, to evaluate the effectiveness of anticoagulant use in patients with HFrEF for stroke prevention, and to compare the prognostic value between the resulting scoring model and CHADS2/CHA2DS2-VASc scores.
Results
Population characteristic
Clinical characteristics of the patients, stratified by the clinical outcome, are shown in Table 1. Totally, 6087 patients were enrolled in the present study. Among them, the mean age was 67.2±14.3 years, 71.0% were men, 48.7% had hypertension, 16.5% had diabetes, 21.2% had AF and 2.9% had deep vein thrombosis (DVT). LVEF at preoperative was 33.5±5.7%. Mean CHADS2 and CHA2DS2-VASc were 2.3±0.9 and 3.4±1.5 respectively. During the 8007.2 person-years follow up, 468 (5.8% events/pts-years) patients suffered from ischemic stroke.
Table 1. Characteristics of study population.
| Overall | Stroke (-) | Stroke (+) | P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6087 | 5619 | 468 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 67.2±14.3 | 67.0±14.4 | 69.2±12.0 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male | 4320 (71.0%) | 3979 (70.8%) | 341 (72.9%) | 0.368 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension | 2964 (48.7%) | 2711 (48.2%) | 253 (54.1%) | 0.016 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes | 1485 (24.4%) | 1351 (24.0%) | 134 (28.6%) | 0.029 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ICM | 2136 (35.1%) | 1985 (35.3%) | 151 (32.3%) | 0.190 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AF | 1291 (21.2%) | 1118 (19.9%) | 173 (37.0%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DVT | 174 (2.9%) | 137 (2.4%) | 137(7.9%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SEC/LVT | 327 (5.4%) | 288 (5.1%) | 39 (8.3%) | 0.005 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CKD | 1484 (24.4%) | 1387 (24.7%) | 97 (20.7%) | 0.057 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | 2083 (34.2%) | 1913 (34.0%) | 170 (36.3%) | 0.335 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Drinking | 1294 (21.3%) | 1168 (20.8%) | 126 (26.9%) | 0.003 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CHADS2 | 2.3±0.9 | 2.3±0.9 | 2.5±0.9 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CHA2DS2-VASc | 3.4±1.5 | 3.3±1.5 | 3.5±1.4 | 0.023 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hemoglobin, g/L | 120.5±29.5 | 120.2±29.8 | 124.5±25.5 | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Platelet, 109/L | 199.4±88.2 | 199.4±88.7 | 199.4±82.0 | 0.996 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BNP, ng/mL | 982.0 (390.0,2380.5) | 986.0 (388.0,2410.0) | 959.0 (390.8,1934.3) | 0.299 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TG, mmol/L | 1.4±0.9 | 1.4±0.9 | 1.4±1.0 | 0.891 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TC, mmol/L | 4.2±1.3 | 4.2±1.3 | 4.2±1.3 | 0.805 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDL-C, mmol/L | 2.5±1.0 | 2.5±1.0 | 2.5±0.9 | 0.339 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CRP, mg/L | 19.0 (6.6,51.4) | 18.9 (6.7,52.4) | 20.8 (5.7,46.7) | 0.925 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cr, μmol/L | 86.0 (69.0,118.0) | 86.0 (69.0,118.0) | 87.0 (68.0,114.0) | 0.343 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AST, U/L | 25.0 (15.0,46.0) | 25.0 (15.0,47.0) | 23.0 (14.0,44.0) | 0.124 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Glucose, mmol/L | 6.7±3.4 | 6.7±3.4 | 7.0±3.5 | 0.112 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| D-dimer | 1.3 (0.7, 2.5) | 1.2 (0.7, 2.5) | 1.4 (0.8, 3.3) | 0.003 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fibrinogen | 4.3±1.5 | 4.3±1.5 | 4.4±1.5 | 0272 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LVEF, % | 33.5±5.7 | 33.5±5.7 | 33.8±5.5 | 0.175 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Anticoagulant use | 750 (12.3%) | 729 (13.0%) | 21 (4.5%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antiplatelet use | 2811 (46.2%) | 2621 (46.6%) | 190(40.6%) | 0.012 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Statins use | 3150 (51.7%) | 2898 (51.6%) | 252 (53.8%) | 0.360 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Data are presented as mean±SD, median (interquartile range) or n (%). Abbreviations: ICM, ischemic cardiomyopathy; AF, atrial fibrillation; DVT, deep vein thrombosis; SEC, spontaneous echocardiographic contrast; LVT, left ventricular thrombus; CKD, chronic kidney disease; BNP, type B natriuretic peptide; TG, triglyceride; TC, total cholesterol, LDL-C, low-density lipoprotein cholesterol; CRP, C-reactive protein; Cr, creatinine; AST, aspartate aminotransferase; LVEF, left ventricular ejection fraction. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Compared with patients without ischemic stroke, those with ischemic stroke were older (69.2±12.0 vs 67.0±14.4 years, p <0.001), had a higher prevalence of hypertension (54.1% vs 48.2%; p=0.016), diabetes (28.6% vs 24.0%; p=0.029), AF (37.0% vs 19.9%; p<0.001), DVT (7.9% vs 2.4%; p<0.001) and SEC/LVT (8.3% vs 5.1%; p < 0.001), increased hemoglobin (124.5±25.5 vs 120.2±29.8g/L, p=0.001) and d-dimer levels [1.4 (0.8, 3.3) vs 1.2 (0.7, 2.5); p=0.003], but lower rate of anticoagulant use (4.5% vs 13.0%; p < 0.001) and antiplatelet use (40.6% vs 46.6%; p=0.012).
Independent predictors for first-ever ischemic stroke in HFrEF
As shown in Table 2, 9 variables (age, hypertension, diabetes, AF, DVT, SEC/LVT, d-dimer levels, anticoagulant use, antiplatelet use) entered the Cox regression model. In univariate Cox regression analysis, age per 1 year (HR=1.018, 95% CI 1.011–1.025, p<0.001), hypertension (HR=1.189, 95% CI 0.991-1.426, p=0.063), diabetes (HR=1.275, 95% CI 1.043–1.559, p<0.001), AF (HR=2.257, 95% CI 1.870–2.723, p<0.001), DVT (HR=4.131, 95% CI 2.945-5.794, p<0.001), SEC/LVT (HR=1.806, 95% CI 1.301-2.508, p<0.001), drinking (HR=1.246, 95% CI 1.016-1.529, p=0.035), hemoglobin per 1 unit (HR=1.003, 95% CI 1.000-1.007, p=0.047), d-dimer levels per 1 unit (HR=1.060, 95% CI 1.041–1.079, p<0.001), anticoagulant use (HR=0.294, 95% CI 0.190-0.455, p<0.001) were found to be potential risk factors for stroke. In multiple Cox regression analyses, 2 continuous variables (age and d-dimer) and 4 categorical variables (AF, DVT, SEC/LVT and anticoagulant use) remained independent predictors of ischemic stroke to construct the nomogram.
Table 2. Univariate cox analysis of potential clinical predictors of first-ever ischemic stroke in HFrEF.
| HR | 95%CI | P value | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.018 | 1.011-1.025 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension | 1.189 | 0.991-1.426 | 0.063 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes | 1.275 | 1.043-1.559 | 0.018 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AF | 2.257 | 1.870-2.723 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DVT | 4.131 | 2.945-5.794 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SEC/LVT | 1.806 | 1.301-2.508 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Drinking | 1.246 | 1.016-1.529 | 0.035 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hemoglobin | 1.003 | 1.000-1.007 | 0.047 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| D-dimer | 1.060 | 1.041-1.079 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Anticoagulant use | 0.294 | 0.190-0.455 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations as in Table 1. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Construction of the new scoring model
According to multivariate Cox regression (Table 3), the final model included age per 1 year (HR=1.029, 95% CI 1.011–1.049, p<0.001), AF (HR=2.874, 95% CI 2.284–3.616, p<0.001), DVT (HR=3.193, 95% CI 2.010-5.075, p<0.001), SEC/LVT (HR=1.722, 95% CI 1.113–2.665, p=0.015), and d-dimer levels per 1 unit (HR=1.050, 95% CI 1.025–1.075, p<0.001) and anticoagulant use (HR=0.206, 95% CI 0.130-0.326, p<0.001) through a prognostic nomogram (Figure 1). The nomogram was created by assigning a graphic preliminary score to each of the 6 predictors with a point range from 0 to 100.
Table 3. Multivariate analysis for the construction of the novel stroke risk score.
| HR | 95%CI | P value | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age per 1 year | 1.029 | 1.011-1.049 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AF | 2.874 | 2.284-3.616 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Anticoagulant use | 0.206 | 0.130-0.326 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DVT | 3.193 | 2.010-5.075 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| D-diner per 1 unit | 1.050 | 1.025-1.075 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SEC/LVT | 1.722 | 1.113-2.665 | 0.015 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations as in Table 1. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
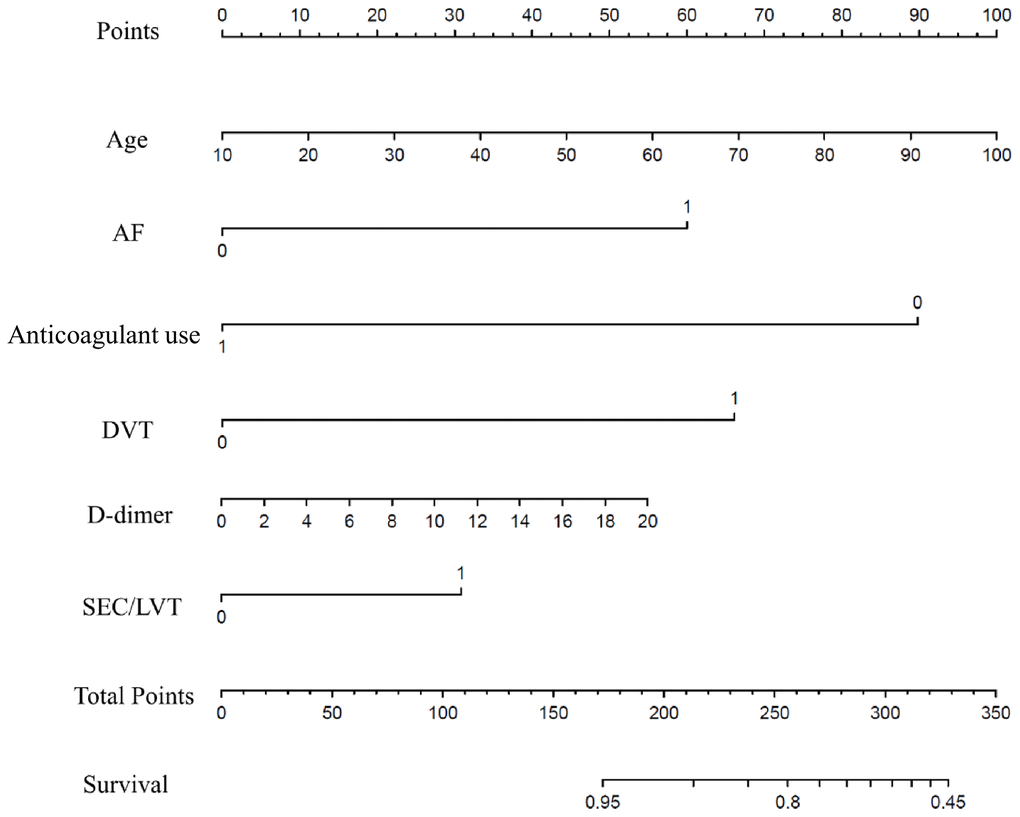
Figure 1. Nomogram model for first-ever ischemic stroke prediction in HFrEF. Abbreviations: HFrEF: heart failure with reduced ejection fraction.
Prognosis performance of this new scoring model
To use the nomogram, the first variable was located. A straight-line was then drawn upwards to the point’s axis to determine the points received for the variable. The process was repeated for the other variables and all points were then tallied to generate the total score. The sum numbers were located on the total points axis and a line was drawn downward to the survival axes and converted into an individual probability of stroke. According to the scores, subjects were divided into high, moderate and low-risk groups (0-150,150-200 and 200-350), with first-ever stroke incidence of 4.9%, 8.2% and 18.9%. Cumulative incidence curves for the entire population cohorts stratified by highest, moderate and low-risk groups are shown in Figure 2. During the first 6 months, patients with 200-350 scores had a significantly higher risk of ischemic stroke than individuals with 150-200 and 0-150 scores (P<0.001). As part of the assessment of its discriminator power, the new scoring system was compared to the CHADS2 and CHA2DS2-VASc score though area under the receiver operating curve (AUROC) analysis (Figure 3). AUROC of CHADS2 score and CHA2DS2-VASc score was 0.515 (95% CI 0.502-0.527) and 0.547 (95% CI 0.534-0.559) at hospitalization, 0.522 (95% CI 0.510-0.535) and 0.553 (95% CI 0.541-0.566) at 30-day follow-up, and 0.540 (95% CI 0.528-0.533) and 0.562 (95% CI 0.550-0.575) at 6-month follow-up. Compared with the two aforementioned scores, the new score exhibited a greater AUROC of 0.727 (95% CI 0.716-0.738), 0.728 (95% CI 0.717-0.739) and 0.714 (95% CI 0.703-0.725) for predicting first-ever ischemic stroke at hospitalization, during the 30-day and 6-month follow-up, respectively (all p<0.001).
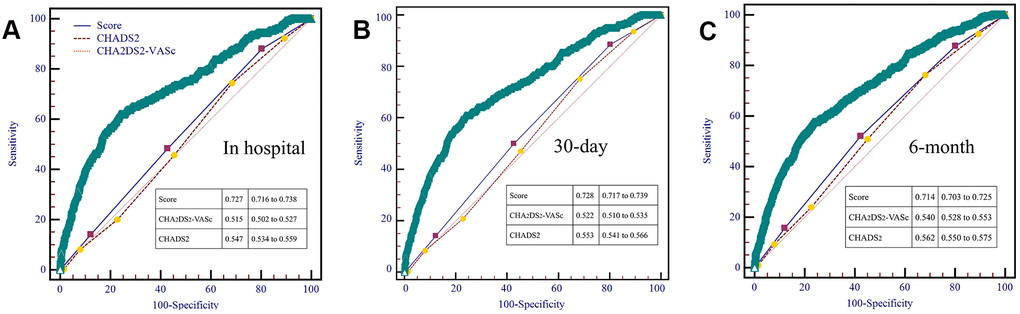
Figure 2. AUROC curves comparing prediction efficacy between our risk model and existing CHADS2 and CHA2DS2-VASc scores in hospital (A), 30-day (B) and 6-month (C) follow-up.
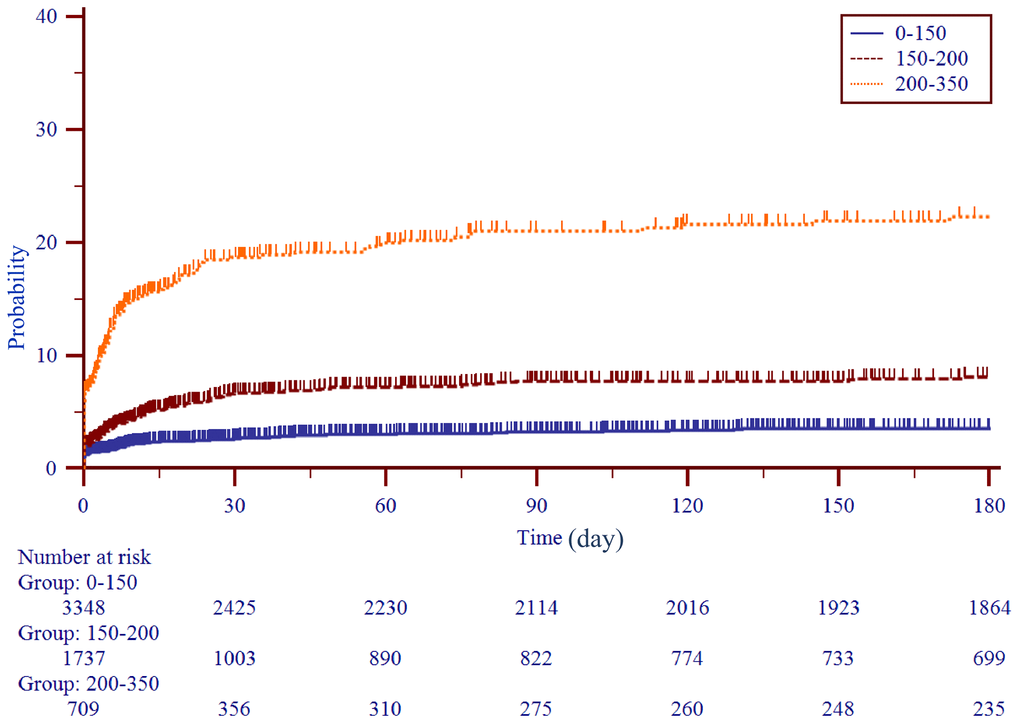
Figure 3. Kaplan-Meier curves stratified by three subgroups by score levels (0-150, 150-200, 200-350).
Discussion
Main finding
We developed a novel risk score for predicting first-ever stroke in a large cohort of HFrEF patients with or without anticoagulant use. The score included 2 continuous variables (age and d-dimer) and 4 categorical variables (AF, DVT, SEC/LVT and anticoagulant use). This scoring model predicted first-ever ischemic stroke with a significantly higher accuracy than the guideline-recommended CHADS2 and CHA2DS2-VASc risk model.
Risk factors from CHADS2 and CHA2DS2-VASc scores
As is well-known, age independently influences stroke outcome and was presented in CHA2DS2-VASc scores as 1 score for age 65–74 and 2 scores for age ≥75 years. In contrast to CHADS2 and CHA2DS2-VASc scores, using continuous variable, the score of age in this novel score is dynamic with the opportunity to allow monitoring of the patient's annual changes in risk of future events. The correlation between DVT and ischemic stroke was also addressed by recent literatures. [11–13] Potential underlying mechanisms include prothrombotic tendency, inflammation activity or endothelial damage [11–14]. In this study, DVT was associated with over 3-fold increased risk and was added into the construction of our resulting scoring model. Three previously identified clinical risk factors (gender, hypertension and diabetes) from CHA2DS2-VASc risk were not included in the present evaluation, as they have not displayed independent associations with stroke in presence of other risk factors. Thus, these factors were excluded from the construction of the scoring model since they do not provide additional prognostic value in risk prediction for stroke and.
SEC/LVT and d-dimer are two novel risk factors for stroke risk stratification in HFrEF
Patients with severe left ventricular dysfunction tend to have higher rates of SEC/LVT, which frequently was associated with higher ischemic stroke risk in patients with HFrEF. [15–18] Prognostic utility of LVT for stroke prediction also had been investigated among patients with dilated cardiomyopathy. [15] SEC is not infrequently observed with echocardiography in patients with HFrEF. Similar to LVT, SEC has also been recognized to exist in left ventricle with high incidence of thromboembolic events. [19–21] The presence of SEC/LVT may indicate fibrinogen concentration and a trigger mechanism for hypercoagulability [7, 15]. Moreover, d-dimer level also reflects increases in blood coagulation and degradation of fibrin, and thus, could be used as marker of thrombosis. [22, 23, 26] Additional studies have consistently shown that elevated d-dimer level is a determinant of the incidence of ischemic stroke not only in the general population but also in patients with HF. [23, 24].
Quantitative evaluation of antithrombotic agents for stroke risk in HFrEF
Due to lack of findings in randomized trials, anticoagulants have not been included in international treatment recommendations for HF patients without AF. [25–28] However, the WARCEF sub-study of HF patients in sinus rhythm reported that longer time in the therapeutic range among patients allocated to warfarin reduced the risk of the ischemic stroke and also improved net clinical benefit. [29] In our scoring model, quantitative evaluation can be conducted in the context of the potential benefit of anticoagulant use to determine optimal anticoagulant therapy. This can be mirrored by one paradigmatic example. A 70-year old (68 points) patient without stroke history, diagnosed as AF (60 points), DVT (70 points), SEC/LVT (30 points), with d-dimer>20 (58 points) and without anticoagulant use (0 points) would have a total nomogram score of 218 and a following probability of stroke >10%. Conversely, a patient underwent anticoagulant would arrive at a total nomogram score of 121 and a following probability of adverse outcome approximating <5%.
Limitation
This study has several limitations. Firstly, this study was retrospective and observational contributing to potential withdraw bias due to lost to follow-up. Secondly, this scoring model was developed from a single-center data without external validation, thereby limiting generalizability our findings. Future external validation studies should be performed in other cohorts and in patients of other ethnicities. Finally, it also remains uncertain about the influence of different dosage and duration of anticoagulant therapy for stroke prediction. High quality prospective research is needed to address unanswered questions to optimize anticoagulant therapy.
Conclusions
In summary, we derived the scoring model for stratifying the risk of first-ever ischemic stroke in patients with HFrEF. This novel score showed an improvement of discriminative accuracy when compared to existing scores.
Materials and Methods
Study design
Data from the electronic medical records database at the First Affiliated Hospital of Wenzhou Medical University, obtained between January 2009 and February 2019, were retrospectively analyzed, which contains age, gender, previous medical history, laboratory markers, diagnosis, treatment, and follow-up data. We included all patients with a baseline mean left ventricle ejection fraction (LVEF) of <40% by quantitative echocardiography assessment. We excluded patients with age of <18 years, history of stroke prior to the HF diagnosis, presence of thrombus in left atrium or right cardiac cavity, presence of prosthetic valves, infective endocarditis or cardiac tumors, incomplete echocardiography examination and laboratory parameters, or missing relevant clinical data including follow-up data. Overall, 18097 echocardiographic records with ejection fraction (EF) <40% were extracted. Of which, 8319 reports were excluded due to repetition from the same patients. As shown in Figure 4, 9778 patients were diagnosis as EF<40% at baseline was prescreened and then were subject to exclusion criteria above, 193 patients were excluded due to data error, age < 18 years or missing follow-up data. In addition, 2232 patients were excluded history of stroke prior to the HF diagnosis. Finally, 6087 heart failure patients with EF<40% were included for analysis. For each patient, the CHADS2 and CHA2DS2-VASc scores were calculated. [6, 7] Based on the CHADS2 score, patients were given one point for congestive heart failure (CHF), hypertension, age ≥75 years, and diabetes, and two points for previous stroke or thromboembolic events (TE). Based on the CHA2DS2-VASc score, patients were given one point for CHF, hypertension, diabetes, vascular disease, age 65–74, and female gender, and two points for previous stroke or TE and age ≥75 years. This investigation conforms to the principles outlined in the Declaration of Helsinki, and was approved by the ethical committee of the First Affiliated Hospital of Wenzhou Medical University Ethical Committee.
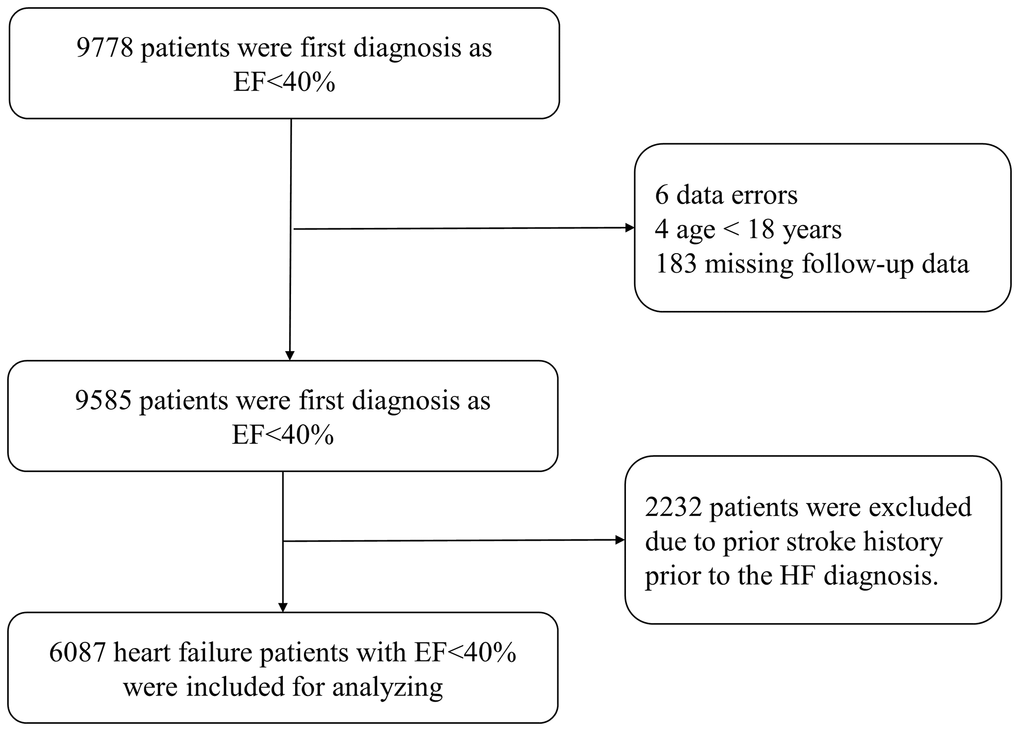
Figure 4. Study flow chart.
Definitions
The primary end point was defined as ischemic stroke at hospitalization, 30-day, 6-month and subsequently the remain period of follow-up. The nomogram was established based on outcomes at 30-day. Ischemic stroke diagnosed as clinically relevant focal neurological symptoms detected by computed tomography or magnetic resonance imaging and confirmed by a neurologist. Ischemic cardiomyopathy (ICM) was defined as CHF in the presence of cardiomyopathy associated with a documented history of myocardial infarction, coronary revascularization, or obstructive coronary artery disease (>50% stenosis) [30]. Chronic kidney disease (CKD) was defined as estimated glomerular filtration rate (eGFR) less than 60 mL/min/1.73m2. [31] LVEF was determined using the biplane Simpson’s method in the apical 2-chamber view. HFrEF was defined as EF with LVEF<40%. [32] Spontaneous echocardiographic contrast (SEC) was defined by dynamic smoke-like echoes with characteristic swirling motion distinct from white noise artifact. [33] LVT was diagnosed as an echogenic mass adjacent to but distinguishable from left ventricular endocardium in an area of wall-motion abnormality. Anticoagulants were used according to patients’ indications and contraindications from the current guidelines and in combination with the patients’ intentions. For high-risk patients, the use of anticoagulants would be taken under guidance of specialist physician. Some patients with atrial fibrillation (AF) or LVT were not on use of regular anticoagulant, the reasons were determined as high risk of bleeding, irregular medication, and refuse anticoagulation. Anticoagulants included warfarin and the novel oral anticoagulants (NOAC) such as dabigatran and rivaroxaban, which was available in Chinese markets.
Statistical analysis
Baseline characteristics were described using mean±SD for continuous variables and frequency (%) for categorical variables. Student’s t and chi-square tests were performed to determine significant differences between groups accordingly. Non-normally distributed variables were presented as median (quartile range) and were compared using the Mann Whitney U test. The associations of clinical meaningful variables were enrolled into the univariate Cox regression models. Then the variables, which were found to be significantly associated with stroke, were entered as potential independent variables in multivariate Cox regression models. Based on the regression coefficients obtained in the multivariable regression model, the survival nomogram was generated to obtain survival probability estimations. The predictive performance of the nomogram was assessed by the area under the receiver operating curve (AUROC) and Kaplan-Meier survival analyses. A two-side p<0.05 was considered as statistically significant. All analyses were performed with SPSS software (SPSS version 23.0 for Windows), MedCale software (MedCale version 11.4 for Windows) and R software (R 3.3.1 version Development Core Team).
Author Contributions
Study design: XD Zhou, WJ Huang, PR Shan. Data collection and processing: LF Yu, WZ Hu, YN Ji, CZ Zhou, CL Xue, GJ Yu. Data analysis: XD Zhou, LF Yu, RY Shi. Manuscript drafting: XD Zhou, LF Yu. Revision: PR Shan. Funding: WJ Huang, PR Shan.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This study was supported by the National Natural Science Foundation of China (81600341), the Natural Science Foundation of Zhejiang Province (LQ15H020005), Wenzhou Science Technology Bureau Foundation (Y20190616), and Medical Health Science and Technology Projects of Zhejiang Province (2021RC091).
References
- 1. Alberts VP, Bos MJ, Koudstaal P, Hofman A, Witteman JC, Stricker B, Breteler M. Heart failure and the risk of stroke: the Rotterdam Study. Eur J Epidemiol. 2010; 25:807–12. https://doi.org/10.1007/s10654-010-9520-y [PubMed]
- 2. Witt BJ, Gami AS, Ballman KV, Brown RD
Jr , Meverden RA, Jacobsen SJ, Roger VL. The incidence of ischemic stroke in chronic heart failure: a meta-analysis. J Card Fail. 2007; 13:489–96. https://doi.org/10.1016/j.cardfail.2007.01.009 [PubMed] - 3. Witt BJ, Brown RD
Jr , Jacobsen SJ, Weston SA, Ballman KV, Meverden RA, Roger VL. Ischemic stroke after heart failure: a community-based study. Am Heart J. 2006; 152:102–09. https://doi.org/10.1016/j.ahj.2005.10.018 [PubMed] - 4. Pullicino PM, McClure LA, Wadley VG, Ahmed A, Howard VJ, Howard G, Safford MM. Blood pressure and stroke in heart failure in the REasons for Geographic And Racial Differences in Stroke (REGARDS) study. Stroke. 2009; 40:3706–10. https://doi.org/10.1161/STROKEAHA.109.561670 [PubMed]
- 5. Holmes DR
Jr , Doshi SK, Kar S, Price MJ, Sanchez JM, Sievert H, Valderrabano M, Reddy VY. Left atrial appendage closure as an alternative to warfarin for stroke prevention in atrial fibrillation: a patient-level meta-analysis. J Am Coll Cardiol. 2015; 65:2614–23. https://doi.org/10.1016/j.jacc.2015.04.025 [PubMed] - 6. Gage BF, Waterman AD, Shannon W, Boechler M, Rich MW, Radford MJ. Validation of clinical classification schemes for predicting stroke: results from the National Registry of Atrial Fibrillation. JAMA. 2001; 285:2864–70. https://doi.org/10.1001/jama.285.22.2864 [PubMed]
- 7. Lip GY, Nieuwlaat R, Pisters R, Lane DA, Crijns HJ. Refining clinical risk stratification for predicting stroke and thromboembolism in atrial fibrillation using a novel risk factor-based approach: the euro heart survey on atrial fibrillation. Chest. 2010; 137:263–72. https://doi.org/10.1378/chest.09-1584 [PubMed]
- 8. Son MK, Lim NK, Park HY. Predicting stroke and death in patients with heart failure using CHA2 DS2-VASc score in Asia. BMC Cardiovasc Disord. 2019; 19:193. https://doi.org/10.1186/s12872-019-1178-0 [PubMed]
- 9. Melgaard L, Gorst-Rasmussen A, Lane DA, Rasmussen LH, Larsen TB, Lip GY. Assessment of the CHA2DS2-VASc score in predicting ischemic stroke, thromboembolism, and death in patients with heart failure with and without atrial fibrillation. JAMA. 2015; 314:1030–38. https://doi.org/10.1001/jama.2015.10725 [PubMed]
- 10. Kondo T, Yamada T, Morita T, Furukawa Y, Tamaki S, Iwasaki Y, Kawasaki M, Kikuchi A, Kawai T, Takahashi S, Ishimi M, Hakui H, Ozaki T, et al. The CHADS2 score predicts ischemic stroke in chronic heart failure patients without atrial fibrillation: comparison to other stroke risk scores. Heart Vessels. 2017; 32:193–200. https://doi.org/10.1007/s00380-016-0861-7 [PubMed]
- 11. Hu WS, Lin CL. The predictive role of CHA2DS2-VASc score between venous thromboembolism and ischemic stroke: a large-scale cohort study. J Hypertens. 2018; 36:628–33. https://doi.org/10.1097/HJH.0000000000001539 [PubMed]
- 12. Rinde LB, Småbrekke B, Mathiesen EB, Løchen ML, Njølstad I, Hald EM, Wilsgaard T, Brækkan SK, Hansen JB. Ischemic stroke and risk of venous thromboembolism in the general population: the tromsø study. J Am Heart Assoc. 2016; 5:e004311. https://doi.org/10.1161/JAHA.116.004311 [PubMed]
- 13. Balogun IO, Roberts LN, Patel R, Pathansali R, Kalra L, Arya R. Clinical and laboratory predictors of deep vein thrombosis after acute stroke. Thromb Res. 2016; 142:33–39. https://doi.org/10.1016/j.thromres.2016.04.002 [PubMed]
- 14. Goshgarian C, Gorelick PB. DVT prevention in stroke. Curr Neurol Neurosci Rep. 2017; 17:81. https://doi.org/10.1007/s11910-017-0782-6 [PubMed]
- 15. Crawford TC, Smith WT
4th , Velazquez EJ, Taylor SM, Jollis JG, Kisslo J. Prognostic usefulness of left ventricular thrombus by echocardiography in dilated cardiomyopathy in predicting stroke, transient ischemic attack, and death. Am J Cardiol. 2004; 93:500–03. https://doi.org/10.1016/j.amjcard.2003.10.056 [PubMed] - 16. Sharma ND, McCullough PA, Philbin EF, Weaver WD. Left ventricular thrombus and subsequent thromboembolism in patients with severe systolic dysfunction. Chest. 2000; 117:314–20. https://doi.org/10.1378/chest.117.2.314 [PubMed]
- 17. Srichai MB, Junor C, Rodriguez LL, Stillman AE, Grimm RA, Lieber ML, Weaver JA, Smedira NG, White RD. Clinical, imaging, and pathological characteristics of left ventricular thrombus: a comparison of contrast-enhanced magnetic resonance imaging, transthoracic echocardiography, and transesophageal echocardiography with surgical or pathological validation. Am Heart J. 2006; 152:75–84. https://doi.org/10.1016/j.ahj.2005.08.021 [PubMed]
- 18. Weinsaft JW, Kim HW, Shah DJ, Klem I, Crowley AL, Brosnan R, James OG, Patel MR, Heitner J, Parker M, Velazquez EJ, Steenbergen C, Judd RM, Kim RJ. Detection of left ventricular thrombus by delayed-enhancement cardiovascular magnetic resonance prevalence and markers in patients with systolic dysfunction. J Am Coll Cardiol. 2008; 52:148–57. https://doi.org/10.1016/j.jacc.2008.03.041 [PubMed]
- 19. Hijazi Z, Oldgren J, Siegbahn A, Granger CB, Wallentin L. Biomarkers in atrial fibrillation: a clinical review. Eur Heart J. 2013; 34:1475–80. https://doi.org/10.1093/eurheartj/eht024 [PubMed]
- 20. Chang KW, Hsu JC, Toomu A, Fox S, Maisel AS. Clinical Applications of Biomarkers in Atrial Fibrillation. Am J Med. 2017; 130:1351–57. https://doi.org/10.1016/j.amjmed.2017.08.003 [PubMed]
- 21. Heppell RM, Berkin KE, McLenachan JM, Davies JA. Haemostatic and haemodynamic abnormalities associated with left atrial thrombosis in non-rheumatic atrial fibrillation. Heart. 1997; 77:407–11. https://doi.org/10.1136/hrt.77.5.407 [PubMed]
- 22. Weitz JI, Fredenburgh JC, Eikelboom JW. A test in context: D-dimer. J Am Coll Cardiol. 2017; 70:2411–20. https://doi.org/10.1016/j.jacc.2017.09.024 [PubMed]
- 23. Hamatani Y, Nagai T, Nakai M, Nishimura K, Honda Y, Nakano H, Honda S, Iwakami N, Sugano Y, Asaumi Y, Aiba T, Noguchi T, Kusano K, et al, and NaDEF Investigators. Elevated Plasma D-Dimer Level Is Associated With Short-Term Risk of Ischemic Stroke in Patients With Acute Heart Failure. Stroke. 2018; 49:1737–40. https://doi.org/10.1161/STROKEAHA.118.021899 [PubMed]
- 24. Sadanaga T, Sadanaga M, Ogawa S. Evidence that D-dimer levels predict subsequent thromboembolic and cardiovascular events in patients with atrial fibrillation during oral anticoagulant therapy. J Am Coll Cardiol. 2010; 55:2225–31. https://doi.org/10.1016/j.jacc.2009.12.049 [PubMed]
- 25. Cokkinos DV, Haralabopoulos GC, Kostis JB, Toutouzas PK, and HELAS Investigators. Efficacy of antithrombotic therapy in chronic heart failure: the HELAS study. Eur J Heart Fail. 2006; 8:428–32. https://doi.org/10.1016/j.ejheart.2006.02.012 [PubMed]
- 26. Cleland JG, Findlay I, Jafri S, Sutton G, Falk R, Bulpitt C, Prentice C, Ford I, Trainer A, Poole-Wilson PA. The Warfarin/Aspirin Study in Heart failure (WASH): a randomized trial comparing antithrombotic strategies for patients with heart failure. Am Heart J. 2004; 148:157–64. https://doi.org/10.1016/j.ahj.2004.03.010 [PubMed]
- 27. Meschia JF, Bushnell C, Boden-Albala B, Braun LT, Bravata DM, Chaturvedi S, Creager MA, Eckel RH, Elkind MS, Fornage M, Goldstein LB, Greenberg SM, Horvath SE, et al, and American Heart Association Stroke Council, and Council on Cardiovascular and Stroke Nursing, and Council on Clinical Cardiology, and Council on Functional Genomics and Translational Biology, and Council on Hypertension. Guidelines for the primary prevention of stroke: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2014; 45:3754–832. https://doi.org/10.1161/STR.0000000000000046 [PubMed]
- 28. Massie BM, Collins JF, Ammon SE, Armstrong PW, Cleland JG, Ezekowitz M, Jafri SM, Krol WF, O’Connor CM, Schulman KA, Teo K, Warren SR, and WATCH Trial Investigators. Randomized trial of warfarin, aspirin, and clopidogrel in patients with chronic heart failure: the Warfarin and Antiplatelet Therapy in Chronic Heart Failure (WATCH) trial. Circulation. 2009; 119:1616–24. https://doi.org/10.1161/CIRCULATIONAHA.108.801753 [PubMed]
- 29. Homma S, Thompson JL, Pullicino PM, Levin B, Freudenberger RS, Teerlink JR, Ammon SE, Graham S, Sacco RL, Mann DL, Mohr JP, Massie BM, Labovitz AJ, et al, and WARCEF Investigators. Warfarin and aspirin in patients with heart failure and sinus rhythm. N Engl J Med. 2012; 366:1859–69. https://doi.org/10.1056/NEJMoa1202299 [PubMed]
- 30. Samman Tahhan A, Hammadah M, Sandesara PB, Hayek SS, Kalogeropoulos AP, Alkhoder A, Mohamed Kelli H, Topel M, Ghasemzadeh N, Chivukula K, Ko YA, Aida H, Hesaroieh I, et al. Progenitor cells and clinical outcomes in patients with heart failure. Circ Heart Fail. 2017; 10:e004106. https://doi.org/10.1161/CIRCHEARTFAILURE.117.004106 [PubMed]
- 31. Levey AS, Stevens LA, Coresh J. Conceptual model of CKD: applications and implications. Am J Kidney Dis. 2009; 53:S4–16. https://doi.org/10.1053/j.ajkd.2008.07.048 [PubMed]
- 32. Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JG, Coats AJ, Falk V, González-Juanatey JR, Harjola VP, Jankowska EA, Jessup M, Linde C, Nihoyannopoulos P, et al, and Authors/Task Force Members, and Document Reviewers. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2016; 18:891–975. https://doi.org/10.1002/ejhf.592 [PubMed]
- 33. Black IW. Spontaneous echo contrast: where there’s smoke there’s fire. Echocardiography. 2000; 17:373–82. https://doi.org/10.1111/j.1540-8175.2000.tb01153.x [PubMed]


