Introduction
The aging process is characterized by a dramatic decline in lean body or muscle mass over the decades of life and the accelerated loss of strength and function that are the hallmarks of sarcopenia [1]. The multiple adverse outcomes of sarcopenia include falls, multi-morbidity, impaired quality of life, disability and mortality [2]. As sarcopenia is potentially reversible, its effective treatment can have a dramatic impact on reducing the disease burden and increasing healthy lifespan of older people.
Age-related anabolic resistance -a blunted synthetic response to protein and exercise, are primary drivers of muscle mass loss in the aging process [3, 4]. Interventions targeting physical inactivity and malnutrition which are the primary causes of sarcopenia can thus potentially improve muscle quantity and quality and prevent or delay the progression of sarcopenia [5]. Clinical trial studies show that the effect of conventional nutritional interventions alone on muscle mass and strength in sarcopenic elderly subjects are limited [6, 7], but recent studies suggest that specific nutritional supplements such as leucine-enriched whey protein and vitamin D may increase muscle mass and muscle function in sarcopenic and malnourished older patients [8–11]. Physical exercise alone [12–14] or with nutritional intervention [15–25] are consistently shown to improve muscle mass, strength and gait speed in older adults with sarcopenia. Cognitive performance and decline are reportedly associated with handgrip strength and sarcopenia in older persons [26], and cognitive training has been shown to maintain and improve gait speed and balance [27, 28], but its effect on sarcopenia is largely unrecognized.
There is a paucity of studies that have explored changes in blood biomarkers of the physiological responses underlying sarcopenia reduction associated with lifestyle interventions. Chronic inflammation is regarded as a major pathophysiological mechanism underlying sarcopenia and has been investigated most commonly in lifestyle interventions for sarcopenia reduction [18, 29]. Exercise intervention reportedly reduce tumor necrosis factor alpha (TNF-α) mRNA and protein levels, while improving muscle strength in frail elderly [29]. Whey protein, amino acids, and vitamin D supplementation are reported to lower C-reactive protein (CRP) level in sarcopenic elderly [18]. Soy protein supplementation and exercise have been shown to result in a reduction in superoxide dismutase (SOD) levels, but did not alter either lipid or protein oxidation [25]. Increased cheese protein intake was shown to improve fasting insulin level in sarcopenic older persons [7]. The aetiology of sarcopenia is complex and multifaceted, involving homeostatic dysregulations in multiple physiological systems. They include imbalanced anabolic and catabolic metabolism that involves the actions of pancreatic, neuro-endocrine, muscle and adipocyte cytokine signaling, sex steroid anabolic homeostasis, hypothalamic–pituitary–adrenal (HPA) stress response, oxidative stress, and other mechanisms [30, 31]. The aim of this study was to explore the physiological responses associated with changes in muscle mass and function resulting from lifestyle intervention in frail older persons using a comprehensive range of blood biomarkers.
We previously reported a parallel group randomized controlled trial [32] multi-domain lifestyle interventions of six month duration (physical exercise, nutritional enrichment and cognitive training singly and in combination versus standard care control) among pre-frail and frail older persons living in the community. In that Frailty Intervention Trial (FIT) in Singapore, the effects of the different interventions on frailty outcomes (muscle strength, gait speed, body mass index, exhaustion and physical activity) at 3-month and 6-month has been reported [32]. Sarcopenia using DXA measure of appendicular lean muscle mass, muscle strength and functional performance was also assessed at baseline and follow up, and are the focus of examination of interventional outcome in this paper. We also collected archival baseline and follow-up blood specimens for analyses of known and potential blood biomarker indicators of molecular regulatory activity across multiple important physiological systems. In this study, we used these blood biomarkers to examine the physiological characteristics underlying the muscle mass and physical functional responses from lifestyle interventions in frail older adults.
Results
Baseline characterization of sarcopenia and functional status
Participants in this randomized controlled trial (n = 242) were pre-frail or frail by the Fried physical phenotype criteria, of Chinese ethnicity with an average age of 70.0 years (SD: 4.7 years). Sarcopenia and non-sarcopenia elderly were comparable in demographic variables including mean age, proportions of gender and formal education level. Lower BMI, ASMI, lower limb strength and higher frailty score were observed in participants with sarcopenia compared to those without sarcopenia (p < 0.001). The sarcopenic participants also tend to show lower gait speed and physical activity, but the differences did not reach statistical significance. No difference was observed in participants with and without sarcopenia with regard to cognitive function (MMSE), mental health (GDS), and lung function (FEV1/FVC% predicted). There were similar proportions of sarcopenia participants who received different interventions. Table 1.
Table 1. Baseline characteristics and interventions by sarcopenia groups.
| All subjects (n = 242) | Sarcopenic subgroups | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sarcopenia (n = 92) | Non-sarcopenia (n = 150) | t/χ2 | p | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years) | 69.97 ± 4.70 | 69.95 ± 4.72 | 69.98 ± 4.71 | –0.055 | 0.956 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender (female) | 150 (61.98) | 59 (64.13) | 91 (60.67) | 0.290 | 0.590 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Secondary and above education | 75 (30.99) | 33 (35.87) | 42 (28.00) | 1.651 | 0.199 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI (kg/m2) | 23.68 ± 3.47 | 21.31 ± 2.55 | 25.13 ± 3.15 | –9.820 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ASMI (kg/m2) | 6.11 ± 1.07 | 5.32 ± 0.82 | 6.60 ± 0.90 | –11.092 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lower limb strength (kg) | 14.17 ± 5.01 | 12.27 ± 3.11 | 15.33 ± 5.57 | –5.491 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gait speed (second) | 5.61 ± 1.65 | 5.76 ± 1.76 | 5.52 ± 1.57 | 1.074 | 0.284 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical activity (min/day) | 169.12 ± 12.63 | 158.20 ± 112.94 | 175.82 ± 112.29 | –1.182 | 0.238 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Frailty score | 2.05 ± 0.85 | 2.28 ± 0.87 | 1.90 ± 0.80 | 3.494 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Interventions (n (%)) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nutritional enrichment | 47 (19.42) | 16 (17.39) | 31 (20.67) | 6.187 | 0.186 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cognitive training | 49 (20.25) | 25 (27.17) | 24 (16.00) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical exercise | 48 (19.83) | 20 (21.74) | 28 (18.67) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Combined intervention | 49 (20.25) | 17 (18.48) | 32 (21.33) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard care | 49 (20.25) | 14 (15.22) | 35 (23.30) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Data are shown as mean ± SD or n (%). Abbreviations: BMI = body mass index; ASMI = Appendicular skeletal muscle index; MMSE = Mini-Mental State Examination; GDS = Geriatric Depression Scale. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Multi-domain lifestyle interventions reversed sarcopenia
As shown in Figure 1A, overall there were 92 participants who had sarcopenia at baseline, and this figure was reduced to 54 and 57 respectively after 3 months and 6 months of intervention; 34.3% (25) and 32.0% (24) of the participants showed reversal of sarcopenia at 3-month and 6-month respectively. The proportions of reversal at 3-month and 6-month were highest for low gait speed, with 85.9% (55) and 83.6% (51) of the participants respectively, followed by low lower limb strength (27.6% (51) and 30.4% (55)) and low ASMI (15.5% (13) and 14.3% (12)).
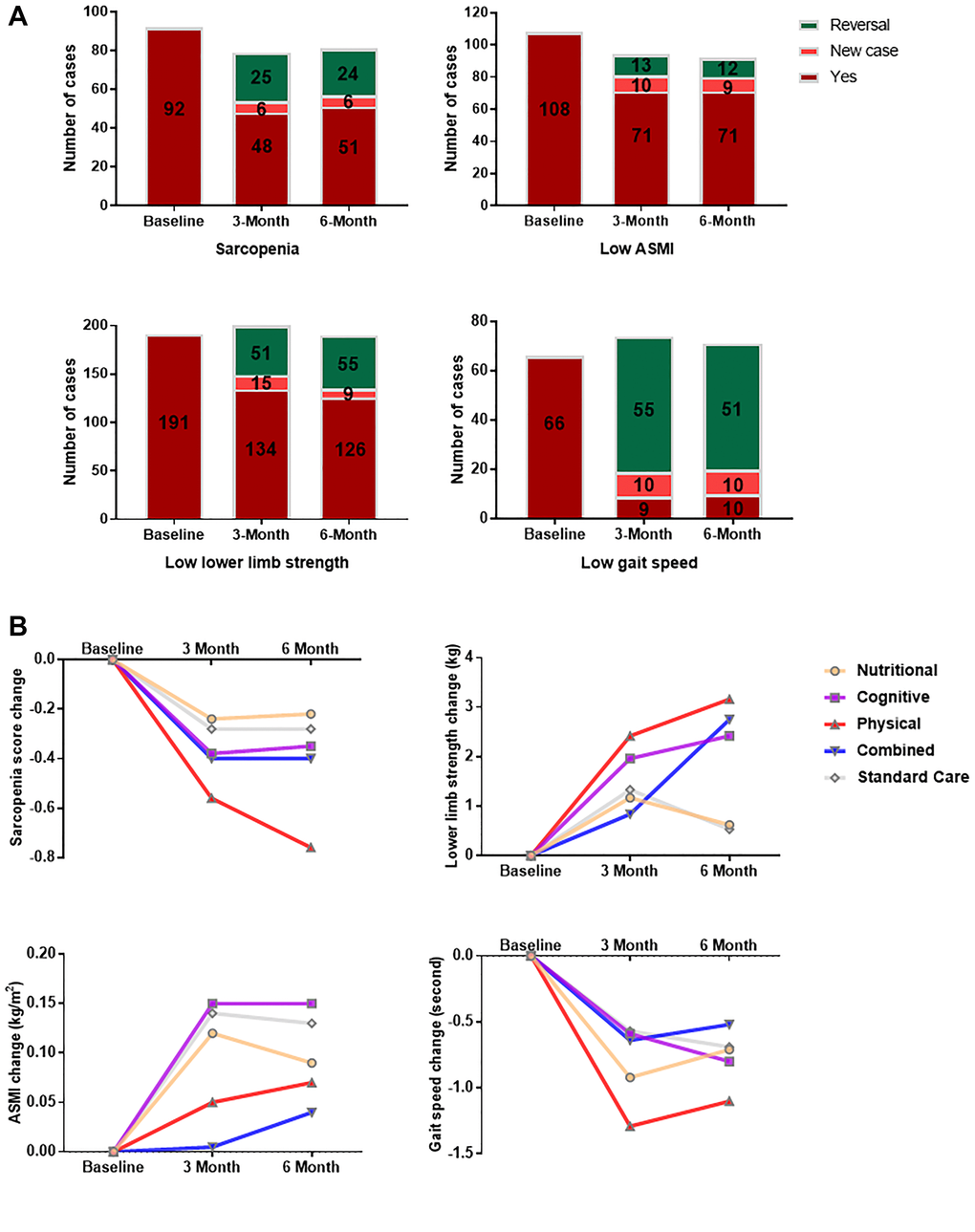
Figure 1. Changes in sarcopenia and its components after multi-domain lifestyle intervention. (A) Sarcopenia and component functional status among trial subjects (n = 242) at baseline, 3 month and 6 month. Reversal was defined as the presence at baseline and absence during follow-up. ASMI = Appendicular skeletal muscle index. (B) Changes in sarcopenia score and its components from baseline to 3-month and 6-month for each interventional arm. Sarcopenia score was calculated as the number of positive components for a participant. The components of sarcopenia included lower limb strength, ASMI, and gait speed. Participants were stratified by intervention groups consisting of nutritional enrichment (n = 47), cognitive training (n = 49), physical exercise (n = 48), combined intervention (n = 49), and standard care (n = 49) groups. ASMI = appendicular skeletal muscle index.
Mixed model analysis (Table 2) indicated a significant main effect of time (p < 0.001). There was a significant decrease of sarcopenia score (p < 0.001) and significant increases of lower limb strength (p < 0.001), ASMI (p < 0.01 at 3-month and p < 0.001 at 6-month), and gait speed (p < 0.001) at 3-month and 6-month of intervention. There was a significant time × group interaction for the improvement of lower limb strength (p < 0.05) and a borderline time × group interaction for the reduction of sarcopenia score (p = 0.059). Improvements in sarcopenia (mean change = –0.76, p < 0.01 at 6-month) and gait speed (mean change = –1.29 second, p < 0.05 at 3-month) were most evident in the physical exercise group in comparison with the standard care group. Combined intervention (mean change = 2.75 kg, p < 0.05), physical exercise (mean change = 3.16 kg, p < 0.01), and cognitive training (mean change = 2.42 kg, p < 0.05) significantly enhanced lower limb strength at 6-month of intervention versus the standard care group. Figure 1B summarizes the changes in sarcopenia score and its components from baseline to 3-month and 6-month for each interventional arm.
Table 2. Effects of multi-domain lifestyle interventions on sarcopenia and components at 3 month and 6 month.
| Lifestyle Interventions (mean ± SD) | Time | Group | Time*Group | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nutritional (n = 47) | Cognitive (n = 49) | Physical (n = 48) | Combined (n = 49) | Standard Care (n = 49) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sarcopenia score | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline | 1.47 ± 0.88 | 1.63 ± 0.86 | 1.75 ± 0.79 | 1.40 ± 0.79 | 1.33 ± 0.83 | <0.001 | 0.307 | 0.059 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 Month*** | 1.11 ± 0.94 | 1.25 ± 0.87 | 1.12 ± 0.75 | 1.00 ± 0.72 | 1.02 ± 0.73 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month*** | 1.14 ± 0.92 | 1.27 ± 0.75 | 0.93 ± 0.72 | 0.98 ± 0.88 | 1.02 ± 0.80 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change (mean (95% CI)) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 Month | –0.24 (–0.47, –0.02) | –0.38 (–0.65, –0.10) | –0.56 (–0.81, –0.32) | –0.40 (–0.62, –0.17) | –0.28 (–0.56, 0.01) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month | –0.22 (–0.45, 0.02) | –0.35 (–0.57, –0.13) | –0.76 (–0.99, –0.52)§§ | –0.40 (–0.60, –0.19) | –0.28 (–0.54, –0.01) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ASMI, kg/m2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline | 6.14 ± 1.25 | 5.70 ± 0.93 | 6.12 ± 0.98 | 6.41 ± 1.15 | 6.21 ± 0.92 | <0.001 | 0.066 | 0.635 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 Month** | 6.28 ± 1.26 | 5.92 ± 1.06 | 6.21 ± 1.01 | 6.40 ± 1.14 | 6.21 ± 0.85 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month*** | 6.26 ± 1.27 | 5.92 ± 1.09 | 6.24 ± 1.01 | 6.44 ± 1.14 | 6.19 ± 0.90 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change (mean (95% CI)) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 Month | 0.12 (0.03, 0.20) | 0.15 (–0.01, 0.30) | 0.05 (–0.03, 0.12) | –0.01(–0.15, 0.13) | 0.14 (0.03, 0.26) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month | 0.09 (0.01, 0.18) | 0.15 (–0.01, 0.30) | 0.07 (–0.00, 0.15) | 0.04 (–0.05, 0.12) | 0.13 (0.01, 0.25) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lower limb strength, kg | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline | 14.25 ± 5.86 | 12.71 ± 3.40 | 13.72 ± 4.17 | 15.10 ± 6.15 | 15.06 ± 4.74 | <0.001 | 0.255 | 0.012 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 Month*** | 15.43 ± 5.88 | 14.72 ± 5.77 | 16.13 ± 5.54 | 16.02 ± 6.17 | 16.22 ± 5.54 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month*** | 15.05 ± 4.79 | 15.16 ± 5.24 | 16.88 ± 5.47 | 17.77 ± 6.79 | 15.17 ± 4.47 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change (mean (95% CI)) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 Month | 1.17 (–0.21, 2.54) | 1.96 (0.37, 3.56) | 2.42 (1.05, 3.78) | 0.84 (–0.47, 2.15) | 1.33 (–0.38, 3.04) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month | 0.62 (–0.59, 1.83) | 2.42 (1.04, 3.80)§ | 3.16 (1.71, 4.61)§§ | 2.75 (1.46, 4.05)§ | 0.53 (–0.88, 1.95) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gait speed, second | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline | 5.75 ± 1.75 | 5.39 ± 1.17 | 6.08 ± 2.08 | 5.39 ± 1.25 | 5.47 ± 1.77 | <0.001 | 0.382 | 0.355 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 Month*** | 4.83 ± 1.20 | 4.76 ± 0.98 | 4.79 ± 0.89 | 4.74 ± 1.20 | 4.90 ± 1.73 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month*** | 4.99 ± 1.03 | 4.62 ± 0.81 | 4.97 ± 1.04 | 4.83 ± 1.13 | 4.79 ± 0.99 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change (mean (95% CI)) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 Month | –0.92 (–1.43, –0.41) | –0.59 (–0.98, –0.20) | –1.29 (–1.87, –0.71)§ | –0.64 (–1.06, –0.23) | –0.57 (–1.09, –0.04) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month | –0.71 (–1.22, –0.21) | –0.80 (–1.16, –0.43) | –1.10 (–1.73, –0.48) | –0.52 (–0.92, –0.11) | –0.69 (–1.19, –0.18) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: ASMI = Appendicular skeletal muscle index. **p < 0.01, ***p < 0.001 vs. baseline level; §p < 0.05, §§p < 0.01 vs. standard care group. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Responsiveness to interventions of sarcopenia versus non-sarcopenia participants
Although sarcopenic elderly were slower and more inactive at baseline, results (Table 3) showed that they were as responsive as their non-sarcopenia counterparts to the interventions. Both groups had overall significant improvements in lower limb strength (p < 0.001), ASMI (p < 0.01 for sarcopenia and p < 0.05 for non-sarcopenia), and gait speed (p < 0.001) at 3-month and 6-month of intervention. No difference between sarcopenia and non-sarcopenia groups was observed in the change of sarcopenia components at 3-month and 6-month, except that sarcopenia elderly had marginally more increase in 6-month ASMI than those without sarcopenia (p = 0.051). The comparable responsiveness of sarcopenia versus non-sarcopenia participants overall was replicated in the detailed data in each interventional arm shown in Table 4.
Table 3. Physical function improvements at 3-month and 6-month interventions in sarcopenia versus non-sarcopenia elderly.
| All subjects (n = 242) | Sarcopenic subgroups | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sarcopenia (n = 92) | Non-Sarcopenia (n = 150) | t | p | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lower limb strength, kg | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline (0M) | 14.17 ± 5.01 | 12.27 ± 3.11 | 15.33 ± 5.57 | –5.491 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 Month (3M) | 15.71 ± 5.76*** | 13.93 ± 4.13*** | 16.75 ± 6.31** | –4.132 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month (6M) | 16.04 ± 5.50*** | 14.08 ± 4.71*** | 17.17 ± 5.61*** | –4.256 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change (3M-0M)% | 15.57 ± 38.15 | 17.47 ± 36.55 | 14.47 ± 39.12 | 0.580 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change (6M-0M)% | 18.28 ± 36.22 | 19.05 ± 38.46 | 17.84 ± 34.99 | 0.243 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| F, p | 19.612, <0.001 | 10.160, <0.001 | 11.343, <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ASMI, kg/m2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline (0M) | 6.11 ± 1.07 | 5.32 ± 0.82 | 6.60 ± 0.90 | –11.092 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 Month (3M) | 6.21 ± 1.07** | 5.37 ± 0.74 | 6.66 ± 0.95* | –10.603 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month (6M) | 6.21 ± 1.09*** | 5.35 ± 0.76** | 6.68 ± 0.95 | –10.802 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change (3M-0M)% | 1.65 ± 6.18 | 2.20 ± 6.84 | 1.35 ± 5.80 | 0.928 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change (6M-0M)% | 1.69 ± 5.82 | 1.73 ± 7.04 | 1.68 ± 5.06 | 0.051 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| F, p | 8.104, <0.001 | 6.486, 0.002 | 3.513, 0.035 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gait speed, second | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline (0M) | 5.61 ± 1.65 | 5.76 ± 1.76 | 5.52 ± 1.57 | 1.074 | 0.284 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 Month (3M) | 4.84 ± 1.23*** | 4.72 ± 1.01*** | 4.85 ± 1.33*** | –0.834 | 0.405 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month (6M) | 4.84 ± 1.01*** | 4.92 ± 1.02*** | 4.79 ± 1.00*** | 0.942 | 0.347 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change (3M-0M)% | –10.43 ± 24.17 | –13.29 ± 23.56 | –8.79 ± 24.44 | –1.378 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change (6M-0M)% | –9.04 ± 24.45 | –9.37 ± 26.11 | –8.85 ± 23.53 | –0.155 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| F, p | 28.155, <0.001 | 13.688, <0.001 | 16.408, <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: ASMI = Appendicular skeletal muscle index. *p < 0.05, **p < 0.01, ***p < 0.001 vs. baseline level. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 4. Physical function changes of sarcopenia and non-sarcopenia elderly for each intervention arm.
| Sarcopenia | Non-Sarcopenia | t | p | Sarcopenia | Non-Sarcopenia | t | p | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nutritional Enrichment | Physical Exercise | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| n = 16 | n = 31 | n = 25 | n = 24 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lower limb strength, kg | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline | 11.02 ± 2.37 | 15.91 ± 6.44 | –3.765 | <0.001 | 11.97 ± 3.00 | 13.47 ± 3.67 | –1.566 | 0.124 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month | 12.53 ± 3.58 | 16.40 ± 4.87 | –2.708 | 0.010 | 13.52 ± 4.05 | 16.60 ± 5.80 | –2.032 | 0.048 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| t, p | –2.154, 0.049 | –0.073, 0.942 | –1.828, 0.083 | –3.069, 0.005 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ASMI,kg/m2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline | 4.87 ± 0.66 | 6.79 ± 0.94 | –7.247 | <0.001 | 5.20 ± 0.79 | 6.23 ± 0.78 | –4.559 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month | 4.77 ± 0.53 | 6.89 ± 0.91 | –8.898 | <0.001 | 5.36 ± 0.74 | 6.43 ± 1.12 | –3.511 | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| t, p | –0.383, 0.710 | –2.404, 0.024 | –1.868, 0.078 | –1.401, 0.177 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gait speed, second | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline | 6.39 ± 2.58 | 5.41 ± 1.03 | 1.457 | 0.163 | 5.59 ± 1.00 | 5.18 ± 1.32 | 1.214 | 0.231 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month | 5.53 ± 1.17 | 4.70 ± 0.83 | 2.690 | 0.010 | 4.87 ± 0.90 | 4.40 ± 0.66 | 1.988 | 0.053 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| t, p | 1.482, 0.160 | 2.935, 0.007 | 3.642, 0.002 | 2.785, 0.011 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cognitive Training | Combined Intervention | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| n = 20 | n = 28 | n = 17 | n = 32 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lower limb strength, kg | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline | 13.07 ± 2.79 | 14.18 ± 4.92 | –0.995 | 0.325 | 12.53 ± 3.99 | 16.46 ± 6.69 | –2.214 | 0.032 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month | 16.58 ± 6.26 | 17.09 ± 4.94 | –0.315 | 0.754 | 13.80 ± 3.91 | 19.63 ± 7.10 | –2.961 | 0.005 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| t, p | –2.818, 0.011 | –3.339, 0.002 | –1.873, 0.082 | –3.865, <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ASMI, kg/m2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline | 5.53 ± 0.80 | 6.54 ± 0.88 | –4.059 | <0.001 | 5.36 ± 0.71 | 6.98 ± 0.93 | –6.266 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month | 5.43 ± 0.75 | 6.66 ± 0.87 | –4.514 | <0.001 | 5.44 ± 0.79 | 6.95 ± 0.93 | –5.346 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| t, p | –0.600, 0.559 | –2.239, 0.034 | –2.065, 0.058 | 0.341, 0.736 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gait speed, second | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline | 5.76 ± 2.07 | 6.30 ± 2.09 | –0.894 | 0.376 | 5.33 ± 1.31 | 5.42 ± 1.24 | –0.239 | 0.812 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month | 4.84 ± 0.99 | 5.07 ± 1.08 | –0.733 | 0.467 | 4.79 ± 0.99 | 4.85 ± 1.21 | –0.176 | 0.861 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| t, p | 1.786, 0.090 | 3.145, 0.004 | 1.265, 0.226 | 2.188, 0.036 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard Care | Active Intervention | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| n = 14 | n = 35 | n = 78 | n = 115 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lower limb strength, kg | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline | 12.74 ± 3.22 | 15.98 ± 4.96 | –2.698 | 0.010 | 12.18 ± 3.11 | 15.13 ± 5.74 | –4.609 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month | 13.23 ± 4.07 | 15.94 ± 4.45 | –1.900 | 0.064 | 14.24 ± 4.83 | 17.54 ± 5.88 | –3.960 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| t, p | –0.446, 0.664 | –0.610, 0.546 | –4.328, <0.001 | –5.166, <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ASMI, kg/m2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline | 5.69 ± 0.97 | 6.41 ± 0.83 | –2.602 | 0.012 | 5.25 ± 0.77 | 6.66 ± 0.92 | –11.452 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month | 5.68 ± 0.79 | 6.41 ± 0.87 | –2.493 | 0.017 | 5.29 ± 0.75 | 6.75 ± 0.96 | –10.806 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| t, p | –0.151, 0.882 | –3.877, <0.001 | –2.513, 0.015 | –2.465, 0.015 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gait speed, second | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline | 5.85 ± 1.74 | 5.32 ± 1.78 | 0.945 | 0.349 | 5.74 ± 1.78 | 5.58 ± 1.50 | 0.656 | 0.513 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month | 4.59 ± 0.99 | 4.86 ± 1.00 | –0.836 | 0.407 | 4.98 ± 1.02 | 4.77 ± 1.00 | 1.382 | 0.169 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| t, p | 2.214, 0.047 | 1.770, 0.086 | 3.657, <0.001 | 5.385, <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: ASMI = Appendicular skeletal muscle index. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Baseline characteristics of subjects with post-interventional sarcopenia reversal
The features of sarcopenia reversal were explored by comparing the demographics, physical and related blood biomarkers in the groups with sarcopenia reversal (presence of sarcopenia at baseline and absence of sarcopenia at 6-month, n = 24) versus the sarcopenia non-reversal group (presence of sarcopenia at both baseline and 6-month, n = 51), and the non-sarcopenia group (absence of sarcopenia at both baseline and 6-month, n = 131) groups. There was a much higher proportion of male participants in the reversal group (54.2%) than in the sarcopenia non-reversal (25.5%) and the non-sarcopenia (38.2%) groups (p < 0.05). Figure 2A. Participants whose sarcopenia were reversed at 6-month had significantly higher baseline ASMI levels than those who remained sarcopenic at 6-month (p < 0.05). There was no significant difference in baseline lower limb strength between the sarcopenia reversal and non-sarcopenia groups (p > 0.05), but the sarcopenia non-reversal group had significantly lower baseline lower limb strength than those in the non-sarcopenia group (p < 0.001). No difference was observed for gait speed among the three groups (p > 0.05). Figure 2B.
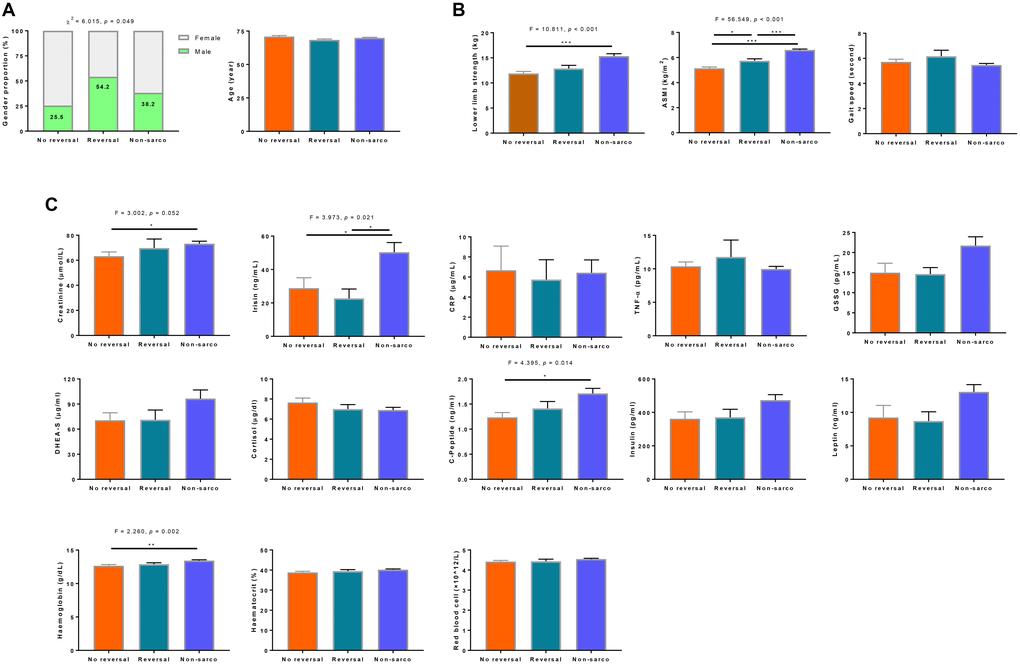
Figure 2. Characterization of demographics. (A) sarcopenia components, (B) and related biomarker, (C) features of elderly with sarcopenia reversal at the end of the intervention. No reversal was defined as the presence of sarcopenia at both baseline and 6-month (n = 51). Reversal group included elderly who had the presence of sarcopenia at baseline and absence of sarcopenia at 6-month (n = 24). Non-sarcopenia was classified as the absence of sarcopenia at both baseline and 6-month (n = 131). CRP = C-reactive protein, TNF-α = tumor necrosis factor alpha, DHEA-S = dehydroepiandrosterone sulfate.
Blood biomarkers of physiological functions
Cross-sectional differences by sarcopenia groups
At baseline, sarcopenic participants showed lower levels of circulating biomarker indicators of multiple physiological functions compared to non-sarcopenic participants, for those that were particularly related to muscle metabolism (creatinine: p < 0.05; irisin: p < 0.01), oxidative stress regulation (GSSG: p < 0.01), sex hormone and HPA stress (DHEA-S: p < 0.05), insulin signaling (c-peptide: p < 0.01; insulin: p < 0.05; leptin: p < 0.05), and tissue oxygenation (haemoglobin: p < 0.001; haematocrit: p < 0.05; red blood cell: p < 0.05). Table 5. The linear regression models also revealed the associations of these biomarkers with component measures of lower limb strength, ASMI and gait speed. Supplementary Table 1.
Table 5. Circulating biomarker levels in sarcopenia and non-sarcopenia subgroups at pre and post interventions.
| All subjects | Sarcopenia Subgroups | Pairwise comparison (6 month vs baseline) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sarcopenia | Non-Sarcopenia | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N = 242 | N = 92 | N = 150 | t | p | Mean | t | p | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Baseline | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Muscle metabolism | Creatinine (μmol/L) | 70.11 ± 22.72 | 65.46 ± 24.39 | 72.85 ± 21.29 | –2.347 | 0.020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Irisin (ng/mL) | 38.04 ± 47.08 | 24.99 ± 31.40 | 45.94 ± 52.84 | –3.130 | 0.002 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Inflammation and | CRP (μg/mL) | 6.17 ± 13.47 | 6.32 ± 13.78 | 6.08 ± 13.34 | 0.128 | 0.898 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Anti-oxidation | TNF-α (pg/mL) | 10.24 ± 5.05 | 10.69 ± 6.31 | 9.99 ± 4.17 | 0.977 | 0.329 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GSSG (pg/mL) | 18.55 ± 17.39 | 14.47 ± 11.36 | 21.02 ± 19.84 | –2.647 | 0.009 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Steroid hormone | DHEA-S (μg/ml) | 86.93 ± 86.32 | 69.62 ± 63.42 | 97.22 ± 99.75 | –2.339 | 0.021 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| regulation | Cortisol (μg/dl) | 6.99 ± 2.59 | 7.29 ± 2.60 | 6.81 ± 2.57 | 1.180 | 0.240 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Insulin signaling | C-peptide (ng/ml) | 1.54 ± 0.85 | 1.32 ± 0.59 | 1.67 ± 0.95 | –2.981 | 0.003 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Insulin (pg/ml) | 435.44 ± 282.99 | 375.15 ± 232.77 | 472.32 ± 304.05 | –2.171 | 0.031 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Leptin (ng/ml) | 11.13 ± 10.09 | 8.82 ± 9.30 | 12.54 ± 10.33 | –2.339 | 0.021 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue oxygenation | Haemoglobin (g/dL) | 13.26 ± 1.35 | 12.84 ± 1.18 | 13.51 ± 1.38 | –3.651 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Haematocrit (%) | 40.03 ± 3.61 | 39.30 ± 3.29 | 40.47 ± 3.73 | –2.116 | 0.036 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Redbloodcell (×1012/L) | 4.52 ± 0.38 | 4.44 ± 0.37 | 4.57 ± 0.39 | –2.115 | 0.036 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 Month | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Muscle metabolism | Creatinine (μmol/L) | 69.91 ± 23.34 | 67.94 ± 28.73 | 71.03 ± 19.73 | –0.759 | 0.450 | –0.327 | –0.560 | 0.576 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Irisin (ng/mL) | 36.08 ± 42.16 | 30.17 ± 42.98 | 39.38 ± 41.57 | –1.264 | 0.208 | –2.361 | –0.795 | 0.428 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Inflammation and | CRP (μg/mL) | 2.97 ± 3.81 | 2.69 ± 2.60 | 3.13 ± 4.36 | –0.707 | 0.480 | –3.259 | –3.365 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Anti-oxidation | TNF-α (pg/mL) | 9.52 ± 3.11 | 9.35 ± 3.42 | 9.62 ± 2.93 | 0.359 | 0.589 | –0.915 | –3.031 | 0.003 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GSSG (pg/mL) | 18.32 ± 18.29 | 13.33 ± 7.11 | 21.01 ± 21.65 | –3.054 | 0.003 | –0.807 | –0.626 | 0.532 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Steroid hormone | DHEA-S (μg/ml) | 84.51 ± 76.79 | 74.02 ± 66.55 | 90.49 ± 81.80 | –1.248 | 0.214 | –0.528 | –0.124 | 0.902 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Regulation | Cortisol (μg/dl) | 7.28 ± 2.51 | 7.84 ± 2.57 | 6.96 ± 2.43 | 2.047 | 0.043 | 0.143 | 0.555 | 0.580 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Insulin signaling | C-peptide (ng/ml) | 1.49 ± 0.72 | 1.34 ± 0.61 | 1.57 ± 0.77 | –1.879 | 0.062 | –0.090 | –1.377 | 0.171 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Insulin (pg/ml) | 414.79 ± 269.35 | 375.10 ± 226.08 | 437.91 ± 290.31 | –1.353 | 0.178 | –37.912 | –1.769 | 0.079 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Leptin (ng/ml) | 11.70 ± 12.71 | 9.97 ± 12.44 | 12.68 ± 12.82 | –1.247 | 0.214 | –0.180 | –0.252 | 0.802 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue oxygenation | Haemoglobin (g/dL) | 13.31 ± 1.36 | 12.92 ± 1.23 | 13.53 ± 1.39 | –2.907 | 0.004 | 0.075 | 1.321 | 0.188 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Haematocrit (%) | 40.52 ± 3.77 | 39.40 ± 3.53 | 41.21 ± 3.78 | –2.666 | 0.009 | 0.123 | 0.710 | 0.480 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Redbloodcell (×1012/L) | 4.55 ± 0.35 | 4.44 ± 0.33 | 4.60 ± 0.35 | –2.323 | 0.022 | –0.012 | –0.698 | 0.487 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: CRP = C - reactive protein, TNF-α = tumor necrosis factor alpha, DHEA-S = dehydroepiandrosterone sulfate. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Differences in pre- and post-interventional changes by sarcopenia groups
The differences in the levels of these biomarkers between the sarcopenia and non-sarcopenia groups that were observed at baseline pre-intervention were mostly reduced after 6 months of intervention, resulting in comparable levels of biomarkers of muscle metabolism (creatinine; irisin) and insulin signaling (c-peptide; insulin; leptin). There remained significant differences in the biomarker levels of anti-oxidation (GSSG) and tissue oxygenation (haemoglobin; haematocrit; red blood cell) between the two groups. With regard to sex hormone and HPA stress, the difference in DHEA-S levels disappeared while the difference in cortisol levels emerged at 6 months between sarcopenia and non-sarcopenia elderly. Table 5.
With regard to inflammatory biomarkers, no difference between sarcopenia and non-sarcopenia groups were observed at baseline or 6 months of intervention. There were substantial reductions in the levels of inflammation indicators such as CRP (from 6.17 μg/mL to 2.97 μg/mL) and TNF-α (from 10.24 pg/mL to 9.52 pg/mL), and this was especially pronounced in the sarcopenia group (CRP: from 6.32 μg/mL to 2.69 μg/mL; TNF-α: from 10.69 pg/mL to 9.35 pg/mL).
Differences by specific intervention domains
The level of TNF-α was most significantly reduced by combined intervention (p < 0.05) and the level of CRP was most significantly reduced by cognitive training (p < 0.05). Creatinine level decreased significantly in the standard care group (p <0.001), but was preserved in the physical exercise, combined intervention and nutritional enrichment groups (p > 0.05), or increased in the cognitive training group (p < 0.05). All active interventions also preserved c-peptide and insulin levels whereas they were significantly decreased in the standard care group (p < 0.05 for c-peptide and p < 0.01 for insulin). Supplementary Table 2.
Differences by sarcopenia reversal groups
We compared the baseline levels of blood biomarkers among non-sarcopenia, sarcopenia non-reversal and sarcopenia reversal groups. As shown in Figure 2C, sarcopenia reversal tended to be predicted by higher baseline creatinine, c-peptide, and haemoglobin levels.
Discussion
In this secondary analysis of data from our previous studies, we observed that multi-domain physical, nutritional and cognitive interventions among pre-frail and frail older adults were associated with favorable changes in sarcopenia and blood biomarkers underlying the muscle mass and physical functional response to intervention. As previously reported, the data are highly consistent with previous studies in showing that physical exercise alone or in combination with cognitive and nutritional intervention was most efficacious in improving muscle mass, lower limb strength and gait speed. The physical exercise in this study was of moderate and gradually increasing intensity and well tolerated with high adherence rate (85%). Perhaps unsurprisingly, there was limited effect observed with nutritional intervention delivered with a traditional oral nutrition supplement and not with a formulation with high content of leucine or whey protein or vitamin D, which have been shown in more recent studies to increase muscle mass and muscle function in sarcopenic and malnourished older patients [8–11]. The observed effects of cognitive training on enhancing gait speed is expected given the important role of cognitive processing especially executive functioning in balance and gait and functional mobility. The concurrent improvement in lower limb strength was somewhat surprising, and has not been reported by other interventional studies that only assessed gait and balance but not muscle strength. The mechanistic relationships are still not fully understood, although age-related loss of motor neuron can reduce muscle mass and strength, and sarcopenia is believed to be an important link between the comorbidity of cognitive impairment and physical functional impairment [28, 33]. We also speculate that the observed effect may be attributed to the extraordinary physical activity associated with participation in cognitive training classes in habitually sedentary frail individuals.
Chronic low-grade inflammation associated with oxidative stress is believed to be a major underlying mechanism of aging and aging-related diseases including sarcopenia [34]. Inflammatory markers such as CRP and IL-6 are reported to be associated with decreased muscle mass and strength [35, 36], and the reduction of inflammation is believed to directly or indirectly ameliorate age-related muscle loss [37]. In the present study, inflammatory levels are observed to be reduced especially by combined intervention, as evidenced by the significant drops in CRP and TNF-α levels. However, the levels of these inflammatory markers were not associated with sarcopenia status or reduction. Thus, the reduction of inflammation may not be the primary underlying mechanism of the response of sarcopenic elderly to lifestyle interventions. On the other hand, sarcopenic elderly showed lower levels of the powerful antioxidant, GSSG. Aging may predispose skeletal muscles to increased oxidative stress at rest and during disuse atrophy, leading to disuse- and sarcopenia-associated muscle loss. Previous studies have also reported the independent association of low circulating level of another antioxidant, carotenoids, with the decrease of skeletal muscle strength [38] and development of walking disability [39]. A randomized controlled trial has also shown that soy protein supplementation and exercise result in a reduction in superoxide dismutase (SOD) levels, but did not alter either lipid or protein oxidation [25].
Beyond inflammation and oxidative stress, we found that circulating biomarkers related to other physiological functions involved in the causative cascade of sarcopenia including muscle metabolism (creatinine, irisin), steroid hormone regulation (DHEA-S, cortisol), insulin signaling (c-peptide, insulin, leptin), and tissue oxygenation (haemoglobin, haematocrit, red blood cell) are linked to sarcopenia baseline status and sarcopenia reduction post-intervention. The association appears to be prominent for creatinine, c-peptide, and haemoglobin levels. Serum creatinine correlates well with dual-energy X-ray absorptiometry (DXA)-measured lean body mass and is regarded as a surrogate of muscle mass and a biomarker for sarcopenia [40, 41]. The observation that serum creatinine predicts sarcopenia reduction in this study suggests its possible prognostic value in interventional studies of sarcopenia.
Sufficient insulin secretion and efficient insulin signaling play a critical role in promoting protein synthesis, maintaining muscle function, and preventing muscle mass loss and sarcopenia [42, 43]. Consonantly in this study, both insulin and c-peptide levels were associated with sarcopenia status, ASMI, and lower limb strength, and were preserved by any of the active interventions but decreased in the standard care group. Furthermore, c-peptide was found to predict sarcopenia reduction. The levels of c-peptide are not affected by insulin injections or liver metabolization and are thus considered a better measure of portal insulin secretion than insulin itself [44].
Our observation of the association between haemoglobin and sarcopenia status and reduction, ASMI, muscle strength, and gait speed is consistent with reports of the association of haemoglobin and anemia with sarcopenia [45, 46]. Low haemoglobin concentration may impair tissue delivery of oxygen, creating sub-clinical hypoxia in skeletal muscle that in the long run impairs muscle strength and performance. Taken together, these circulating biomarkers associated with sarcopenia status and reduction may serve as easily accessible and cost-effective means of measuring physiological responses to interventions aimed at reducing sarcopenia in clinical research and practice.
Our analyses suggest that the effects of lifestyle interventions in reversing sarcopenia in community-dwelling older persons are accompanied by multi-level targeted physiological changes that include age-related inflammation and the anabolic and catabolic mechanisms of regulating hormones and tissue oxygenation involved in the causation of sarcopenia. Nevertheless, as the study was designed for frailty as the primary outcome among participants with pre-frailty and frailty, sarcopenia was not the primary readout. The findings obtained in this secondary analysis of data should therefore be validated in an independent dataset with sarcopenia as the primary outcome. The suggestive results of sarcopenia reduction based on multiple subgroup analyses are indirect observations of the effects of interventions, and should therefore be viewed with some circumspection. The exploratory nature of the study is emphasized, as we measured a wide array of biomarkers and evaluated all observable changes in physiological biomarkers associated with the interventions. Spurious significant results are possible, but some meaningful changes that stood out were highly consistent with previous reports in the literature.
Materials and Methods
Study design and participants
The parallel group randomized controlled trial of the Singapore Frailty Intervention Trial (FIT, http://clinicaltrials.gov/ identifier NCT00973258) has been described in a previous publication [32], and is briefly summarized here. The eligible study subjects were 246 community-dwelling older persons with the physical frailty phenotype of pre-frailty or frailty from Fried at al. [47]. These older persons were randomized to receive one of 5 interventions of 24-week duration each: physical exercise, nutritional enrichment, cognitive training, combined intervention, or standard care. The study was approved by National Health Group Domain Specific Review Board (DSRB) of Singapore, and all participants provided written informed consent.
Sarcopenia measurement
Sarcopenia, which is the primary outcome of this study, was determined based on appendicular lean mass, lower limb strength and gait speed, according to the Asian Working Group for Sarcopenia (AWGS) criteria [48] released in 2014. Appendicular lean body mass was measured by DXA scan with the Hologic® densitometer. Four participants refused to undergo the DXA scan and were removed from the analysis. A participant was categorized as having sarcopenia if he/she had both low appendicular skeletal muscle index (ASMI) and low knee extension strength (KES, less than or equal to 18 kg for men and less than or equal to 16 kg for women) and/or gait speed (GS, less than or equal to 0.8 m/s). Sarcopenia score was calculated as the number of positive components. KES was measured using the strap and strain gauge assembly component of the Physiological Profile Assessment and GS was assessed by the 6-meter fast gait speed test.
Multi-domain lifestyle interventions
Details of the interventions have been described in a previous publication [32] and are briefly summarized below:
Physical exercise
Participants performed resistance exercise integrated with functional tasks and balance training exercise involving functional strength, sensory input, and added attentional demands. Each program started with two sessions per week (90-minute each session) for 12 weeks in classes supervised by a qualified trainer, followed by home-based exercise for another 12 weeks. The exercise was tailored to the functional needs of the participants, maintaining a moderate intensity that focuses on long-term sustainability and enjoyment, with balance and strength as the key components.
Nutritional enrichment
Participants were provided nutritional advice and supervision with additional daily supplements of a commercial formula (Fortisip Multi Fibre, Nutricia: 200 mL liquid formula with 300 kcal carbohydrate (49%), fat (35%), protein (35%), and dietary fiber (4.6 g)), a calcium and vitamin D supplement tablet (Caltrate; 600 mg calcium and 200 IU vitamin D), an iron and folate capsule (Sangobion, Merck; 1 mg folate and 29 mg iron), and a vitamin B6 and B12 tablet (Neuroforte; 200 mg B6 and 200 μg B12) for 24 weeks. This regime was designed to increase caloric intake by 20% and provide additional 1/3 of the RADs of vitamins and minerals.
Cognitive training
Cognitive enhancing activities included learning strategies for recalling verbal and visual information, tasks such as “spot the differences”, categorical naming, coding to enhance attention and processing speed, and matrix reasoning exercises, mazes and tangram-like games aimed at enhancing reasoning and problem-solving abilities. Participants attended intensive cognitive training classes for the first 12 weeks followed by 12-week fortnightly 2-hour “booster” sessions where they reviewed the cognitive skills learned in the intensive classes.
Combined intervention
Elderly in this group underwent all three aforementioned interventions.
Standard care
Participants had access to standard community-based social, recreational and day care rehabilitation services and were given an equal volume of artificially sweetened, vanilla-flavored liquid (ingredients: non-dairy creamer, liquid caramel, sugar and water), two capsules, and one tablet (ingredients: corn-starch, lactose and magnesium stearate) that were identical in appearance to the active nutritional supplements. Both the active supplement and standard care placebo were administered by interventional nurses who had no knowledge of the participants’ group allocation.
Pre- and post-intervention assessments
At baseline pre-intervention and 3 months and 6 months post-intervention, participants underwent interviews and testing that included DXA scan, lower limb strength measurement, and 6-meter fast gait speed test for the assessment and diagnosis of sarcopenia. Blood was drawn after over-night fasting, and serum was isolated and stored in –80°C freezer until measurements.
Blood biomarkers
Archival serum specimens were thawed, and quantitative assays were performed for analytes of hormones, cytokines, metabolites, and other biomarkers that were identified from literature search to be involved in homeostatic processes associated with sarcopenia. Levels of CRP, cortisol, dehydroepiandrosterone sulfate (DHEA-S) and irisin were measured via the enzyme-linked immunosorbent assay (ELISA). Insulin, leptin and C-peptide levels were quantified using the Luminex assay (Merck Millipore). Measurements of creatinine, TNF-α, haemoglobin, haematocrit and red blood cell count were performed using standard clinical chemistry methods at NUH Referral Laboratories.
Statistical analysis
The efficacy of interventions was examined using intention-to-treat (ITT) analysis. Differences in sarcopenia versus non-sarcopenia groups were compared by independent t-test for continuous variables and chi-square test for categorical variables. The linear mixed model was applied to investigate the effects of treatment group, time and group × time interaction as fixed factors. For variables with significant group × time interaction indicating changing group effect over time, the simple main effect of treatment group was further evaluated at each time point using one-way analysis of variance (ANOVA) with Bonferroni post-hoc adjustments. All data analyses were performed using IBM SPSS 21 software (IBM, USA). Figures were generated using GraphPad Prism 7.
Supplementary Materials
Author Contributions
YL and TPN reviewed the literature, designed the study, analysed the data, drafted and revised the manuscript. MN, KBY, CTYT, MSN, LF, BYT, GC, SAK, SMC, PY, and AL contributed to the study design and data collection. All authors reviewed the results and drafts, and approved the final manuscript.
Acknowledgments
We thank the following voluntary welfare organizations for their support in kind: Geylang East Home for the Aged, Presbyterian Community Services, St Luke’s Eldercare Services, Thye Hua Kwan Moral Society (Moral Neighbourhood Links), Yuhua Neighbourhood Link, Henderson Senior Citizens’ Home, NTUC Eldercare Co-op Ltd, Thong Kheng Seniors Activity Centre (Queenstown Centre) and Redhill Moral Seniors Activity Centre.
Conflicts of Interest
All authors declare no real or potential conflicts of interest.
Funding
The study was supported by the National Medical Research Council [Grant: NMRC/1108/2007] and the Agency for Science Technology and Research [JCO Grant: 1434m00115].
References
- 1. Rosenberg IH. Sarcopenia: origins and clinical relevance. Clin Geriatr Med. 2011; 27:337–9. https://doi.org/10.1016/j.cger.2011.03.003 [PubMed]
- 2. Martone AM, Bianchi L, Abete P, Bellelli G, Bo M, Cherubini A, Corica F, Di Bari M, Maggio M, Manca GM, Marzetti E, Rizzo MR, Rossi A, et al. The incidence of sarcopenia among hospitalized older patients:results from the Glisten study. J Cachexia Sarcopenia Muscle. 2017; 8:907–14. https://doi.org/10.1002/jcsm.12224 [PubMed]
- 3. Markofski MM, Dickinson JM, Drummond MJ, Fry CS, Fujita S, Gundermann DM, Glynn EL, Jennings K, Paddon-Jones D, Reidy PT, Sheffield-Moore M, Timmerman KL, Rasmussen BB, et al. Effect of age on basal muscle protein synthesis and mTORC1 signaling in a large cohort of young and older men and women. Exp Gerontol. 2015; 65:1–7. https://doi.org/10.1016/j.exger.2015.02.015 [PubMed]
- 4. Bowen TS, Schuler G, Adams V. Skeletal muscle wasting in cachexia and sarcopenia: molecular pathophysiology and impact of exercise training. J Cachexia Sarcopenia Muscle. 2015; 6:197–207. https://doi.org/10.1002/jcsm.12043 [PubMed]
- 5. Cruz-Jentoft AJ, Landi F, Schneider SM, Zuniga C, Arai H, Boirie Y, Chen LK, Fielding RA, Martin FC, Michel JP, Sieber C, Stout JR, Studenski SA, et al. Prevalence of and interventions for sarcopenia in ageing adults:a systematic review. Report of the International Sarcopenia Initiative (EWGSOP and IWGS). Age Ageing. 2014; 43:748–59. https://doi.org/10.1093/ageing/afu115 [PubMed]
- 6. Milne AC, Avenell A, Potter J. Meta-analysis:protein and energy supplementation in older people. Ann Intern Med. 2006; 144:37–48. https://doi.org/10.7326/0003-4819-144-1-200601030-00008 [PubMed]
- 7. Alemán-Mateo H, Macías L, Esparza-Romero J, Astiazaran-García H, Blancas AL. Physiological effects beyond the significant gain in muscle mass in sarcopenic elderly men:evidence from a randomized clinical trial using a protein-rich food. Clin Interv Aging. 2012; 7:225–34. https://doi.org/10.2147/cia.s32356 [PubMed]
- 8. Cramer JT, Cruz-Jentoft AJ, Landi F, Hickson M, Zamboni M, Pereira SL, Hustead DS, Mustad VA. Impacts of High-Protein Oral Nutritional Supplements Among Malnourished Men and Women with Sarcopenia: A Multicenter, Randomized, Double-Blinded, Controlled Trial. J Am Med Dir Assoc. 2016; 17:1044–55. https://doi.org/10.1016/j.jamda.2016.08.009 [PubMed]
- 9. Takeuchi I, Yoshimura Y, Shimazu S, Jeong S, Yamaga M, Koga H. Effects of branched-chain amino acids and vitamin D supplementation on physical function, muscle mass and strength, and nutritional status in sarcopenic older adults undergoing hospital-based rehabilitation: A multicenter randomized controlled trial. Geriatr Gerontol Int. 2019; 19:12–7. https://doi.org/10.1111/ggi.13547 [PubMed]
- 10. Bo Y, Liu C, Ji Z, Yang R, An Q, Zhang X, You J, Duan D, Sun Y, Zhu Y, Cui H, Lu Q. A high whey protein, vitamin D and E supplement preserves muscle mass, strength, and quality of life in sarcopenic older adults: A double-blind randomized controlled trial. Clin Nutr. 2019; 38:159–64. https://doi.org/10.1016/j.clnu.2017.12.020 [PubMed]
- 11. Liberman K, Njemini R, Luiking Y, Forti LN, Verlaan S, Bauer JM, Memelink R, Brandt K, Donini LM, Maggio M, Mets T, Wijers SLJ, Sieber C, et al. Thirteen weeks of supplementation of vitamin D and leucine-enriched whey protein nutritional supplement attenuates chronic low-grade inflammation in sarcopenic older adults: the PROVIDE study. Aging Clin Exp Res. 2019; 31:845–54. https://doi.org/10.1007/s40520-019-01208-4 [PubMed]
- 12. Makizako H, Nakai Y, Tomioka K, Taniguchi Y, Sato N, Wada A, Kiyama R, Tsutsumimoto K, Ohishi M, Kiuchi Y, Kubozono T, Takenaka T. Effects of a Multicomponent Exercise Program in Physical Function and Muscle Mass in Sarcopenic/Pre-Sarcopenic Adults. J Clin Med. 2020; 9:1386. https://doi.org/10.3390/jcm9051386 [PubMed]
- 13. Tsekoura M, Billis E, Tsepis E, Dimitriadis Z, Matzaroglou C, Tyllianakis M, Panagiotopoulos E, Gliatis J. The Effects of Group and Home-Based Exercise Programs in Elderly with Sarcopenia: A Randomized Controlled Trial. J Clin Med. 2018; 7:480. https://doi.org/10.3390/jcm7120480 [PubMed]
- 14. Maruya K, Asakawa Y, Ishibashi H, Fujita H, Arai T, Yamaguchi H. Effect of a simple and adherent home exercise program on the physical function of community dwelling adults sixty years of age and older with pre-sarcopenia or sarcopenia. J Phys Ther Sci. 2016; 28:3183–8. https://doi.org/10.1589/jpts.28.3183 [PubMed]
- 15. Chang KV, Wu WT, Huang KC, Han DS. Effectiveness of early versus delayed exercise and nutritional intervention on segmental body composition of sarcopenic elders - A randomized controlled trial. Clin Nutr. 2020. [Epub ahead of print]. https://doi.org/10.1016/j.clnu.2020.06.037 [PubMed]
- 16. Liao CD, Chen HC, Huang SW, Liou TH. The Role of Muscle Mass Gain Following Protein Supplementation Plus Exercise Therapy in Older Adults with Sarcopenia and Frailty Risks: A Systematic Review and Meta-Regression Analysis of Randomized Trials. Nutrients. 2019; 11:1713. https://doi.org/10.3390/nu11081713 [PubMed]
- 17. Zhu LY, Chan R, Kwok T, Cheng KC, Ha A, Woo J. Effects of exercise and nutrition supplementation in community-dwelling older Chinese people with sarcopenia: a randomized controlled trial. Age Ageing. 2019; 48:220–8. https://doi.org/10.1093/ageing/afy179 [PubMed]
- 18. Rondanelli M, Klersy C, Terracol G, Talluri J, Maugeri R, Guido D, Faliva MA, Solerte BS, Fioravanti M, Lukaski H, Perna S. Whey protein, amino acids, and vitamin D supplementation with physical activity increases fat-free mass and strength, functionality, and quality of life and decreases inflammation in sarcopenic elderly. Am J Clin Nutr. 2016; 103:830–40. https://doi.org/10.3945/ajcn.115.113357 [PubMed]
- 19. Yoshimura Y, Wakabayashi H, Yamada M, Kim H, Harada A, Arai H. Interventions for Treating Sarcopenia: A Systematic Review and Meta-Analysis of Randomized Controlled Studies. J Am Med Dir Assoc. 2017; 18:553.e1–e16. https://doi.org/10.1016/j.jamda.2017.03.019 [PubMed]
- 20. Kim H, Kim M, Kojima N, Fujino K, Hosoi E, Kobayashi H, Somekawa S, Niki Y, Yamashiro Y, Yoshida H. Exercise and Nutritional Supplementation on Community-Dwelling Elderly Japanese Women With Sarcopenic Obesity: A Randomized Controlled Trial. J Am Med Dir Assoc. 2016; 17:1011–9. https://doi.org/10.1016/j.jamda.2016.06.016 [PubMed]
- 21. Maltais ML, Ladouceur JP, Dionne IJ. The Effect of Resistance Training and Different Sources of Postexercise Protein Supplementation on Muscle Mass and Physical Capacity in Sarcopenic Elderly Men. J Strength Cond Res. 2016; 30:1680–7. https://doi.org/10.1519/jsc.0000000000001255 [PubMed]
- 22. Zdzieblik D, Oesser S, Baumstark MW, Gollhofer A, Konig D. Collagen peptide supplementation in combination with resistance training improves body composition and increases muscle strength in elderly sarcopenic men: a randomised controlled trial. Br J Nutr. 2015; 114:1237–45. https://doi.org/10.1017/s0007114515002810 [PubMed]
- 23. Kim HK, Suzuki T, Saito K, Yoshida H, Kobayashi H, Kato H, Katayama M. Effects of exercise and amino acid supplementation on body composition and physical function in community-dwelling elderly Japanese sarcopenic women: a randomized controlled trial. J Am Geriatr Soc. 2012; 60:16–23. https://doi.org/10.1111/j.1532-5415.2011.03776.x [PubMed]
- 24. Kim H, Suzuki T, Saito K, Yoshida H, Kojima N, Kim M, Sudo M, Yamashiro Y, Tokimitsu I. Effects of exercise and tea catechins on muscle mass, strength and walking ability in community-dwelling elderly Japanese sarcopenic women: a randomized controlled trial. Geriatr Gerontol Int. 2013; 13:458–65. https://doi.org/10.1111/j.1447-0594.2012.00923.x [PubMed]
- 25. Shahar S, Kamaruddin NS, Badrasawi M, Sakian NI, Abd Manaf Z, Yassin Z, Joseph L. Effectiveness of exercise and protein supplementation intervention on body composition, functional fitness, and oxidative stress among elderly Malays with sarcopenia. Clin Interv Aging. 2013; 8:1365–75. https://doi.org/10.2147/cia.s46826 [PubMed]
- 26. Chang KV, Hsu TH, Wu WT, Huang KC, Han DS. Association Between Sarcopenia and Cognitive Impairment: A Systematic Review and Meta-Analysis. J Am Med Dir Assoc. 2016; 17:1164.e7–e15. https://doi.org/10.1016/j.jamda.2016.09.013 [PubMed]
- 27. Lipardo DS, Aseron AMC, Kwan MM, Tsang WW. Effect of Exercise and Cognitive Training on Falls and Fall-Related Factors in Older Adults With Mild Cognitive Impairment: A Systematic Review. Arch Phys Med Rehabil. 2017; 98:2079–96. https://doi.org/10.1016/j.apmr.2017.04.021 [PubMed]
- 28. Smith-Ray RL, Hughes SL, Prohaska TR, Little DM, Jurivich DA, Hedeker D. Impact of Cognitive Training on Balance and Gait in Older Adults. J Gerontol B Psychol Sci Soc Sci. 2015; 70:357–66. https://doi.org/10.1093/geronb/gbt097 [PubMed]
- 29. Greiwe JS, Cheng B, Rubin DC, Yarasheski KE, Semenkovich CF. Resistance exercise decreases skeletal muscle tumor necrosis factor alpha in frail elderly humans. FASEB J. 2001; 15:475–82. https://doi.org/10.1096/fj.00-0274com [PubMed]
- 30. Marty E, Liu Y, Samuel A, Or O, Lane J. A review of sarcopenia:Enhancing awareness of an increasingly prevalent disease. Bone. 2017; 105:276–86. https://doi.org/10.1016/j.bone.2017.09.008 [PubMed]
- 31. Walston J, Hadley EC, Ferrucci L, Guralnik JM, Newman AB, Studenski SA, Ershler WB, Harris T, Fried LP. Research agenda for frailty in older adults:toward a better understanding of physiology and etiology:summary from the American Geriatrics Society/National Institute on Aging Research Conference on Frailty in Older Adults. J Am Geriatr Soc. 2006; 54:991–1001. https://doi.org/10.1111/j.1532-5415.2006.00745.x [PubMed]
- 32. Ng TP, Feng L, Nyunt MS, Feng L, Niti M, Tan BY, Chan G, Khoo SA, Chan SM, Yap P, Yap KB. Nutritional, Physical, Cognitive, and Combination Interventions and Frailty Reversal Among Older Adults: A Randomized Controlled Trial. Am J Med. 2015; 128:1225–36.e1. https://doi.org/10.1016/j.amjmed.2015.06.017 [PubMed]
- 33. Tolea MI, Galvin JE. Sarcopenia and impairment in cognitive and physical performance. Clin Interv Aging. 2015; 10:663–71. https://doi.org/10.2147/cia.s76275 [PubMed]
- 34. Bottazzi B, Riboli E, Mantovani A. Aging, inflammation and cancer. Semin Immunol. 2018; 40:74–82. https://doi.org/10.1016/j.smim.2018.10.011 [PubMed]
- 35. Howard C, Ferrucci L, Sun K, Fried LP, Walston J, Varadhan R, Guralnik JM, Semba RD. Oxidative protein damage is associated with poor grip strength among older women living in the community. J Appl Physiol (1985). 2007; 103:17–20. https://doi.org/10.1152/japplphysiol.00133.2007 [PubMed]
- 36. Schaap LA, Pluijm SM, Deeg DJ, Visser M. Inflammatory markers and loss of muscle mass (sarcopenia) and strength. Am J Med. 2006; 119:526.e9–17. https://doi.org/10.1016/j.amjmed.2005.10.049 [PubMed]
- 37. Raimondo TM, Mooney DJ. Functional muscle recovery with nanoparticle-directed M2 macrophage polarization in mice. Proc Natl Acad Sci U S A. 2018; 115:10648–53. https://doi.org/10.1073/pnas.1806908115 [PubMed]
- 38. Lauretani F, Semba RD, Bandinelli S, Dayhoff-Brannigan M, Giacomini V, Corsi AM, Guralnik JM, Ferrucci L. Low plasma carotenoids and skeletal muscle strength decline over 6 years. J Gerontol A Biol Sci Med Sci. 2008; 63:376–83. https://doi.org/10.1093/gerona/63.4.376 [PubMed]
- 39. Semba RD, Varadhan R, Bartali B, Ferrucci L, Ricks MO, Blaum C, Fried LP. Low serum carotenoids and development of severe walking disability among older women living in the community: the women's health and aging study I. Age Ageing. 2007; 36:62–7. https://doi.org/10.1093/ageing/afl122 [PubMed]
- 40. Lien YH. Looking for Sarcopenia Biomarkers. Am J Med. 2017; 130:502–3. https://doi.org/10.1016/j.amjmed.2017.01.018 [PubMed]
- 41. Patel SS, Molnar MZ, Tayek JA, Ix JH, Noori N, Benner D, Heymsfield S, Kopple JD, Kovesdy CP, Kalantar-Zadeh K. Serum creatinine as a marker of muscle mass in chronic kidney disease: results of a cross-sectional study and review of literature. J Cachexia Sarcopenia Muscle. 2013; 4:19–29. https://doi.org/10.1007/s13539-012-0079-1 [PubMed]
- 42. Prod'homme M, Rieu I, Balage M, Dardevet D, Grizard J. Insulin and amino acids both strongly participate to the regulation of protein metabolism. Curr Opin Clin Nutr Metab Care. 2004; 7:71–7. https://doi.org/10.1097/00075197-200401000-00012 [PubMed]
- 43. Cleasby ME, Jamieson PM, Atherton PJ. Insulin resistance and sarcopenia: mechanistic links between common co-morbidities. J Endocrinol. 2016; 229:R67–81. https://doi.org/10.1530/joe-15-0533 [PubMed]
- 44. Shapiro ET, Tillil H, Rubenstein AH, Polonsky KS. Peripheral insulin parallels changes in insulin secretion more closely than C-peptide after bolus intravenous glucose administration. J Clin Endocrinol Metab. 1988; 67:1094–9. https://doi.org/10.1210/jcem-67-5-1094 [PubMed]
- 45. Hirani V, Naganathan V, Blyth F, Le Couteur DG, Seibel MJ, Waite LM, Handelsman DJ, Hsu B, Cumming RG. Low Hemoglobin Concentrations Are Associated With Sarcopenia, Physical Performance, and Disability in Older Australian Men in Cross-sectional and Longitudinal Analysis: The Concord Health and Ageing in Men Project. J Gerontol A Biol Sci Med Sci. 2016; 71:1667–75. https://doi.org/10.1093/gerona/glw055 [PubMed]
- 46. Hai S, Cao L, Wang H, Zhou J, Liu P, Yang Y, Hao Q, Dong B. Association between sarcopenia and nutritional status and physical activity among community-dwelling Chinese adults aged 60 years and older. Geriatr Gerontol Int. 2017; 17:1959–66. https://doi.org/10.1111/ggi.13001 [PubMed]
- 47. Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, Seeman T, Tracy R, Kop WJ, Burke G, McBurnie MA. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001; 56:M146–56. https://doi.org/10.1093/gerona/56.3.m146 [PubMed]
- 48. Chen LK, Liu LK, Woo J, Assantachai P, Auyeung TW, Bahyah KS, Chou MY, Chen LY, Hsu PS, Krairit O, Lee JS, Lee WJ, Lee Y, et al. Sarcopenia in Asia: consensus report of the Asian Working Group for Sarcopenia. J Am Med Dir Assoc. 2014; 15:95–101. https://doi.org/10.1016/j.jamda.2013.11.025 [PubMed]



