Introduction
Ovarian reserve (OR) refers to the development and ability of ovarian follicles to produce fertilized oocytes in the cortex. OR is used to describe female reproductive potential and predict the response to controlled ovarian hyperstimulation (COH) in the context of assisted reproductive technology (ART) [1]. Diminished OR (DOR) is generally defined by a reduced reproductive ability in older and younger women of reproductive age with regular menses compared to women of comparable age [2] due to age and to metabolic, genetic, autoimmune, enzymatic, iatrogenic, toxic and infectious causes [1]. DOR affects 6% to 64% of infertile women of different ages [3] and leads to a decreased pregnancy rate and increased miscarriage rate in women undergoing ART [4–6]. Patients with DOR have increased embryo aneuploidy [7–9]. Previous research has shown that first trimester pregnancy loss occurred in 15–20% of clinically recognized pregnancies, and 50% of pregnancy losses resulted from embryonic chromosomal aberrations [10]. This loss significantly increases the physical and psychological burden and economic losses of ART patients, especially young women with DOR, who expect a better outcome than their older peers.
The definition of DOR was not standardized according to the Practice Committee of the American Society for Reproductive Medicine (ASRM) [11]. Generally, OR is evaluated using basal follicle-stimulating hormone (FSH), anti-Mullerian hormone (AMH) and antral follicle count (AFC) in women with regular periods [12–14]. Studies related to the influence of the age of DOR patients on their offspring revealed conflicting results. Most analyses indicated that advanced age increased the risk of DOR [15] and chromosomal aneuploidy in embryos [16] simultaneously, and DOR was associated with low embryo morphology grades and increased aneuploid miscarriages and viable aneuploid pregnancies [15, 17, 18]. However, conflicting investigations have shown that female age was not a significant predictor of clinical miscarriage [19]; there was no relevance between DOR and oocyte quality [18, 20–23], and oocytes from women with DOR had similar potential for euploid blastocyst development. These studies focused on the effects of DOR and female age on chromosomal aberrations in oocytes or transferred embryos, respectively, while very few examined chromosomal abnormalities in products of conception (POCs). At the same time, most studies of this issue have failed to account for other risk factors for miscarriage that are influenced by age and DOR, such AMH, AFC and FSH. Therefore, little is known about the etiology of DOR in women experiencing first-trimester miscarriage.
To evaluate this question, we enrolled a retrospective database of patients with DOR who experienced first-trimester miscarriage and obtained POCs collected for single nucleotide polymorphism (SNP) microarray techniques to investigate the role of age and controversial markers relating to OR and ART characteristics in chromosomal aberration-related miscarriage in DOR patients. Notably, as the methods for evaluating the chromosomes of spontaneously aborted embryos develop, SNP microarray is a first-line method for the genetic detection of individual developmental disorders and congenital dysplasia by the International Standards for Cytogenomic Arrays Consortium [24]. It provides whole genome-wide screening with a higher resolution and detects unbalanced copy number variations (CNVs) with sizes greater than 100 kb.
Results
Demographics of study subjects
There were 63 women with DOR in this study: 18 young (aged 20–32 years old) women and 45 old (aged over 32 years old) women. Table 1 presents the comparisons of different clinical characteristics and the ART strategies of enrolled couples in our study between the young and old groups. The larger proportion of female partners was in the old group (71.4%). There were no significant differences between the two maternal age groups in maternal-related parameters, such as BMI; serum basal E2, LH and thyroid-stimulating hormone (TSH) levels; gestational age at miscarriage (irrespective of fertilization method); fertilization method; the proportion of blastocysts transferred; and the number of embryos transferred. The median male age increased with the advancing age of their female partners, which was statistically significant in the two age groups (P < 0.001). The rates of prior gravidity (young group: 44.4%, old group: 73.3%, P = 0.040) and prior miscarriage (young group: 11.1%, old group: 44.4%, P = 0.039) were significantly different between the groups. In terms of reproductive potential, the young patients with DOR showed higher AMH (0.7 ± 0.2 vs. 0.5 ± 0.4, P = 0.040), higher AFC (3.8 ± 1.4 vs. 2.8 ± 2.1, P = 0.004), lower medians of FSH (9.3 vs. 10.5, P = 0.015) and more oocytes retrieved (7.2 ± 4.4 vs. 45.5 ± 2.8, P = 0.046) than the old group.
Table 1. Demographics of study objects grouped by age.
| Characteristics | Maternal age (years) | P-value | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total | 20–32 | ≥33 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No. of the cases | 63 | 18 (28.6) | 45 (71.4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Paternal parameters | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male age (years) | 33.6 ± 4.9 | 29.8 ± 4.0 | 36.0 ± 3.8 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Maternal parameters | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI (kg/m2) | NS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| <25 | 49 (77.8) | 14 (77.8) | 35 (77.8) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≥25 | 14 (22.2) | 4 (22.2) | 10 (22.2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TSH (mIU/mL) | 2.1 ± 0.9 | 2.0 ± 0.7 | 2.1 ± 1.0 | NS | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AMH (ng/mL) | 0.6 ± 0.4 | 0.7 ± 0.2 | 0.5 ± 0.4 | 0.040 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AFC | 3.1 ± 1.9 | 3.8 ± 1.4 | 2.8 ± 2.1 | 0.004 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Basal FSH level (mIU/mL) | 10.5 (8.7,12.4) | 9.3 (8.3,11.5) | 10.5 (8.9,12.9) | 0.015 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Basal E2 level (mIU/mL) | 34.8 (25.4,41.9) | 34.8 (28.5,41.9) | 34.8 (24.4,52.4) | NS | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Basal LH level (mIU/mL) | 4.9 ± 2.2 | 4.6 ± 2.1 | 5.1 ± 2.3 | NS | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Previous reproductive history | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Prior gravidity | 0.040 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0 | 22 (34.9) | 10 (55.6) | 12 (26.7) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1 | 21 (33.4) | 6 (33.3) | 15 (33.3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≥2 | 20 (31.7) | 2 (11.1) | 18 (40.0) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Prior miscarriage | 0.039 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0 | 41 (65.1) | 16 (88.9) | 25 (55.6) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1 | 18 (28.6) | 2 (11.1) | 16 (35.5) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≥2 | 4 (6.3) | 0 | 4 (8.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ART characteristics | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fertility method (%) | NS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IVF | 47 (74.6) | 11 (61.1) | 36 (80%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ICSI | 16 (25.4) | 7 (38.9) | 9 (20%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No. of ovum pick-up | 6.1±3.5 | 7.2 ± 4.4 | 5.5 ± 2.8 | 0.046 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Day of embryos transferred (%) | NS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| D3 | 59 (93.7) | 18 (100) | 41 (91.1) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| D5 | 4 (6.3) | 0 | 4 (8.9) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gestational age at miscarriage (weeks) | 8.5 ± 0.6 | 8.4 ± 0.5 | 8.5 ± 0.7 | NS | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SNP results | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender of POC (%) | NS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| male | 35 (55.6) | 10 (55.6) | 25 (55.6) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| female | 28 (44.4) | 8 (44.4) | 20 (44.4) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chromosomal abnormality of POC (%) | 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| normal | 16 (25.4) | 10 (55.6) | 6 (13.3) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| abnormal | 47 (74.6) | 8 (44.4) | 39 (86.7) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: Data are presented as numbers (%), means (95% CI), or median (25th, 75th percentile). BMI = body mass index; TSH = thyroid stimulating hormone; AMH = anti-Mullerian hormone; AFC = antral follicle count; FSH = follicle-stimulating hormone; LH = luteinizing hormone; ART = assisted reproductive technology; IU = international unit; IVF = in vitro fertilization; ICSI = intracytoplasmic sperm; SNP = single nucleotide polymorphism; POC = products of conception; NS = not significant; CI = Confidence interval. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Results of the SNP array analysis of POCs
SNP microarray analyses of 63 chorionic villus samples enrolled in our study identified 47 cases (74.6%) with chromosomal aberrations, indicating that the abnormalities were predicted to be causative of the miscarriage. The following chromosomal abnormalities were detected: 83.0% trisomy (39/47); 8.5% triploid (4/47); 4.3% monosomy (2/47); 2.1% structural abnormalities (1/47); and 2.1% mosaicism (1/47) (Table 2). The rate of chromosomal abnormality of POCs was significantly different between the young and old women with DOR (44.4% vs. 86.7%, P = 0.013). The sex distribution of POCs in the young group was similar to that in the old group (male POCs: 55.6% vs. 44.4%, P = 1) (Table 1).
Table 2. Spectrum of abnormal chromosomal karyotype: type and variations between maternal age groups.
| Variables | Maternal age (years) | Total frequency (%) | |
| 20–32 (n = 18) | ≥33 (n = 45) | ||
| Trisomy | 83.0 (39/47) | ||
| single | 4 | 32 | |
| double | 0 | 3 | |
| Triploidy | 2 | 2 | 8.5 (4/4) |
| Monosomy | 1 | 1 | 4.3 (2/47) |
| Structural abnormality | 1 | 0 | 2.1 (1/4) |
| Mosaicism | 0 | 1 | 2.1 (1/47) |
| Total | 8 | 39 | |
| Total frequency (%) | 44.4 (8/18) | 86.7 (39/45) | |
The most prevalent abnormality was trisomy. Single-chromosome trisomy was found in 36 cases and accounted for 92.3% (36/39) of all trisomies. Multiple trisomies (two or more chromosomes involved in trisomy) were observed in 3 chorionic villus samples, which constituted 7.7% (3/39). The most common trisomies were related to chromosomes 16 and 22 (18.5% and 16.2%, respectively), followed by 15 (9.3%), 20 (7.0%) and 21 (7.0%). No sex chromosome trisomy was observed, perhaps because of the small number of samples (Figure 1). Triploidy was found in 8.5% of samples (4/47). Three samples had a 69,XXY karyotype (75.0%), and 1 sample had a 69,XXX karyotype (25.0%). Monosomy was observed in 2 of 47 samples (4.3%), and both cases were on chromosome 21. There was 1 case of structural abnormality, related to duplication of chromosome 17, and 1 case of mosaicism, showing trisomy and monosomy. Representative examples of the SNP results of the POCs are shown in Figure 2.
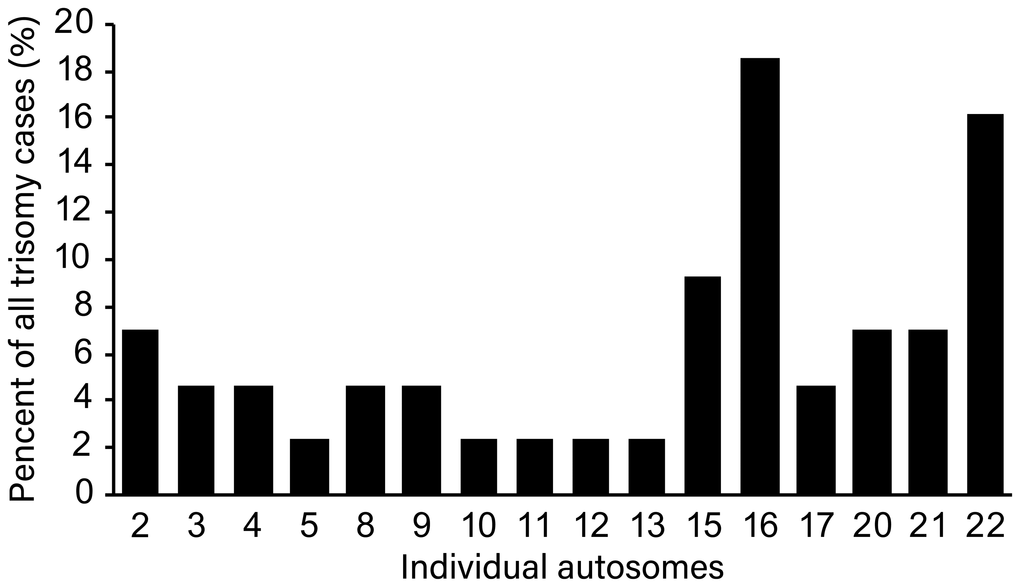
Figure 1. Distribution of trisomy of individual chromosome among all trisomy cases.
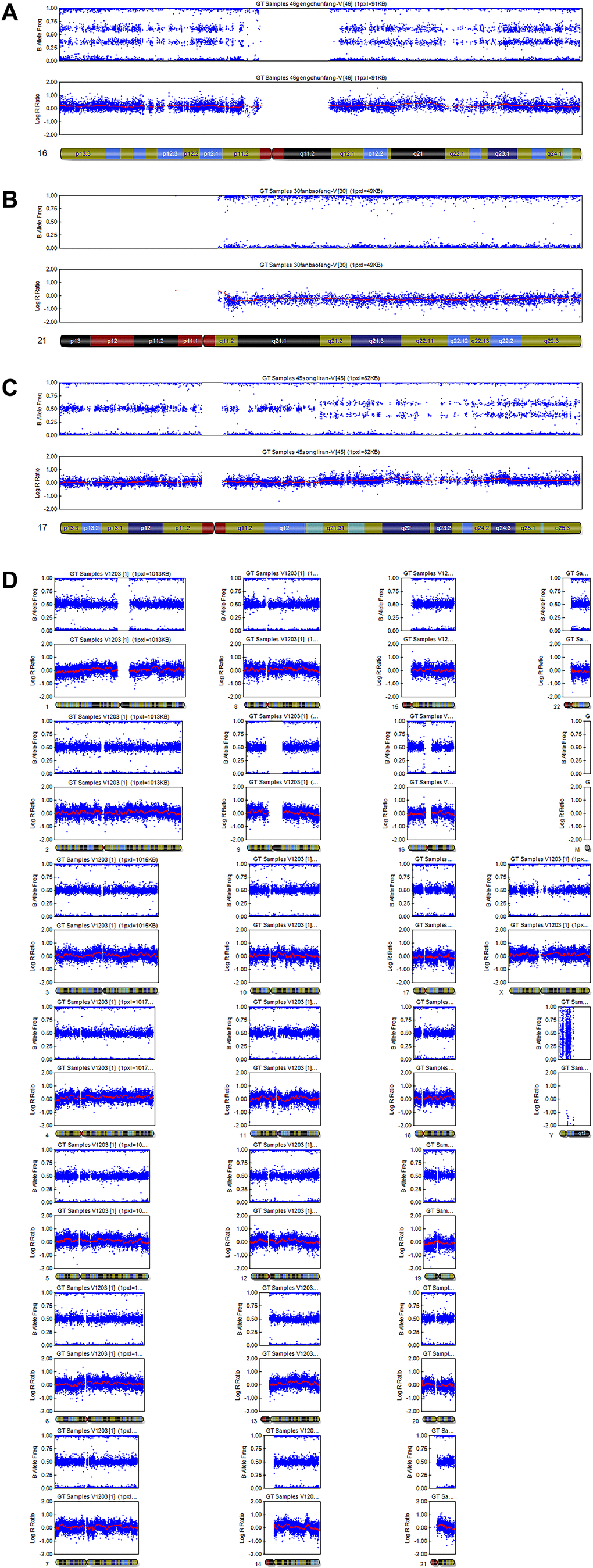
Figure 2. Representative examples of SNP results. (A) Single trisomy of chromosome 16. (B) Monosomy of chromosome 21. (C) Structural abnormality: duplication of chromosome 17. (D) Normal: arr (1–22) × 2, (X) × 2.
Discussion
Lower chromosomal abnormality frequencies in POCs of young patients (aged 20–32 years old) with DOR
Our study demonstrated that age is an independent risk factor for chromosomal abnormality after adjustment. The aberration-related miscarriages among women with DOR were more frequent in the old group (older than 32 years old) than in younger women (20–32 years old). The exact mechanism by which advanced age (older than 32 years old) relates to higher chromosomal abnormalities in POCs remains elusive. The decline in fertility among women with DOR is related to age and DOR simultaneously. Age-related decline is definitely characterized by a quantitative and qualitative decline in OR and adverse pregnancy outcomes due to mitochondrial dysfunction, telomere shortening, impaired DNA repair, epigenetic changes and metabolic/energetic disorders [25]. Previous researchers have focused on the effects of maternal age on chromosomal abnormality in eggs or transferred embryos, while we mainly examined the effect of female age on abnormal chromosomes in miscarried fetuses. We believe this current study provides a new view of the strategy of diagnosis and treatment regarding the link between age of women with DOR and fetal development. Furthermore, age could influence the pathogenesis and degree of DOR. Previous studies have demonstrated the association between age and DOR from clinical data and molecular mechanisms. Young patients with DOR have a significantly greater likelihood of acquiring transplantable embryos and high-quality embryos than old patients with DOR, and the clinical pregnancy outcome was good once eggs were acquired [26]. Woo et al. [27] identified different expression profiles of miRNAs that regulate genes related to reduced oocyte quality only in young women with DOR, including miRNAs involved in the WNT, TGF-b, P13K-Akt, and MAPK signaling pathways in granulosa cells (GCs). Similarly, Skiadas et al. [28] confirmed altered gene expression in GCs only among young women with DOR. We identified female age as an important factor leading to the chromosomal abnormality of POCs and speculate that the mechanisms governing quality parameters and follicular depletion might be divergent in young and old women with DOR.
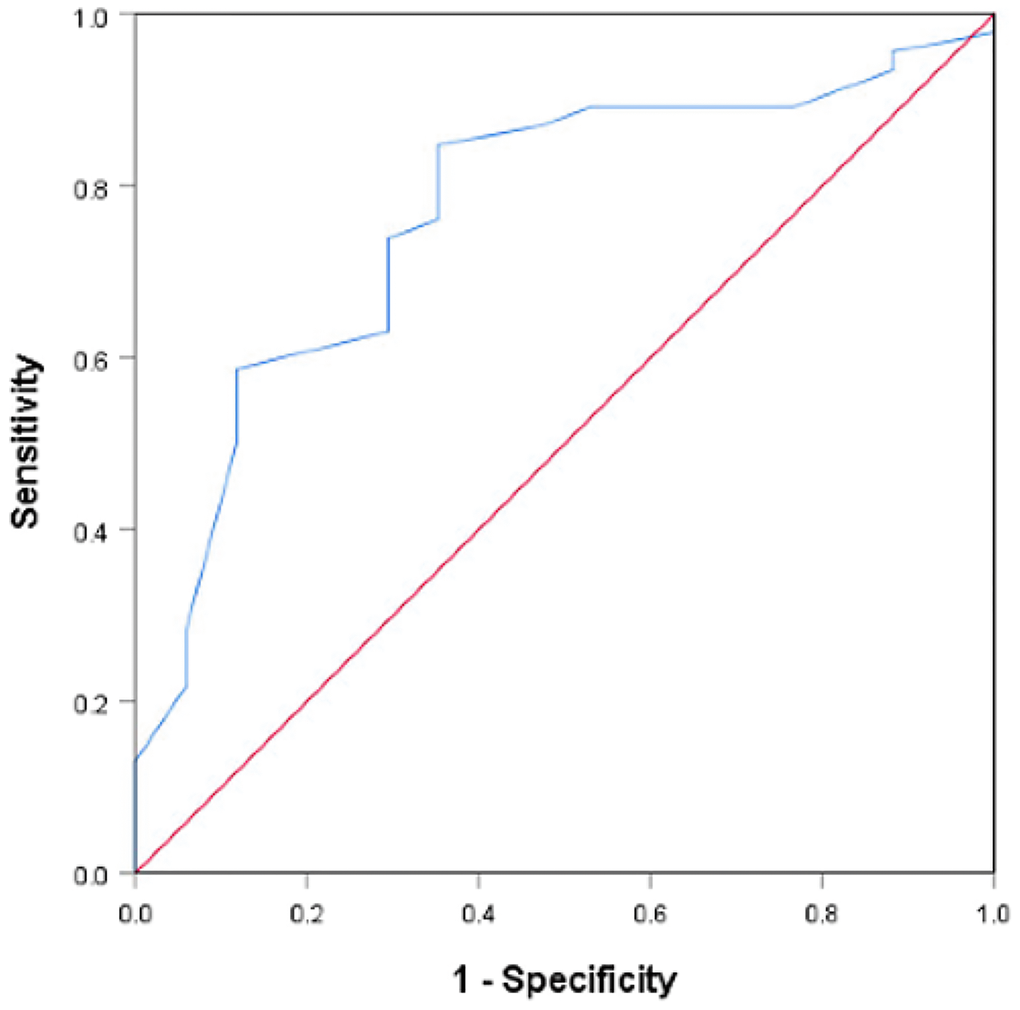
In addition, we identified 32 years old as a cut-off value for the prediction of aneuploid POCs for several reasons. (i) Receiver operator characteristic (ROC) curve. The ROC curves demonstrated the predictive utility of female age (AUC = 0.769). Female age exhibited a cut-off value of 32 years old and good sensitivity and specificity of 84.8% and 64.7%, respectively. Compared with 32 years old, 30 and 35 years old, which are important ages in fertility, presented poor sensitivity and specificity of 84.4% and 25.0% and 56.3% and 75.0%, respectively. (ii) Previous research. A recent study [29] reported that the chromosome errors (aneuploidy) in human eggs followed a U-shaped curve after chromosome segregation in human oocytes from females aged 9 to 43 years old, in which the group aged 20–32 years old had the smallest proportion of aneuploidy MII oocytes, and maternal age was the only significant factor that affected the aneuploidy of oocytes, consistent with our results. (iii) Other investigation of data from our reproductive center. There is no currently accepted medical definition of advanced age relevant to aneuploid miscarriages in women with DOR. Researchers used to define 30 and 35 years old as cut-off values of age. We investigated 63 POCs using a SNP microarray from women included in our study further and divided them into <30, 30–31, 32–34, ≥35–year-old group. The results showed that there was no significant difference between each pair of groups (<30 years old vs. ≥30 years old; <35 years old vs. ≥35 years old) (Figure 4). In addition, the data in our center investigated by Li et al. [30] showed that there was no significant difference in the rate of karyotypic abnormalities between the <30-year-old group and the 30- to 35-year-old group among women with normal OR. Our study indicated that chromosomal abnormalities of POCs might be primarily due to advanced age and relate to a new and younger cut-off value of age (32 years old). We speculate that women with DOR are more sensitive to advanced age compared with women with normal OR, and the impact of age on chromosomal aberrations might be greater than that of DOR.
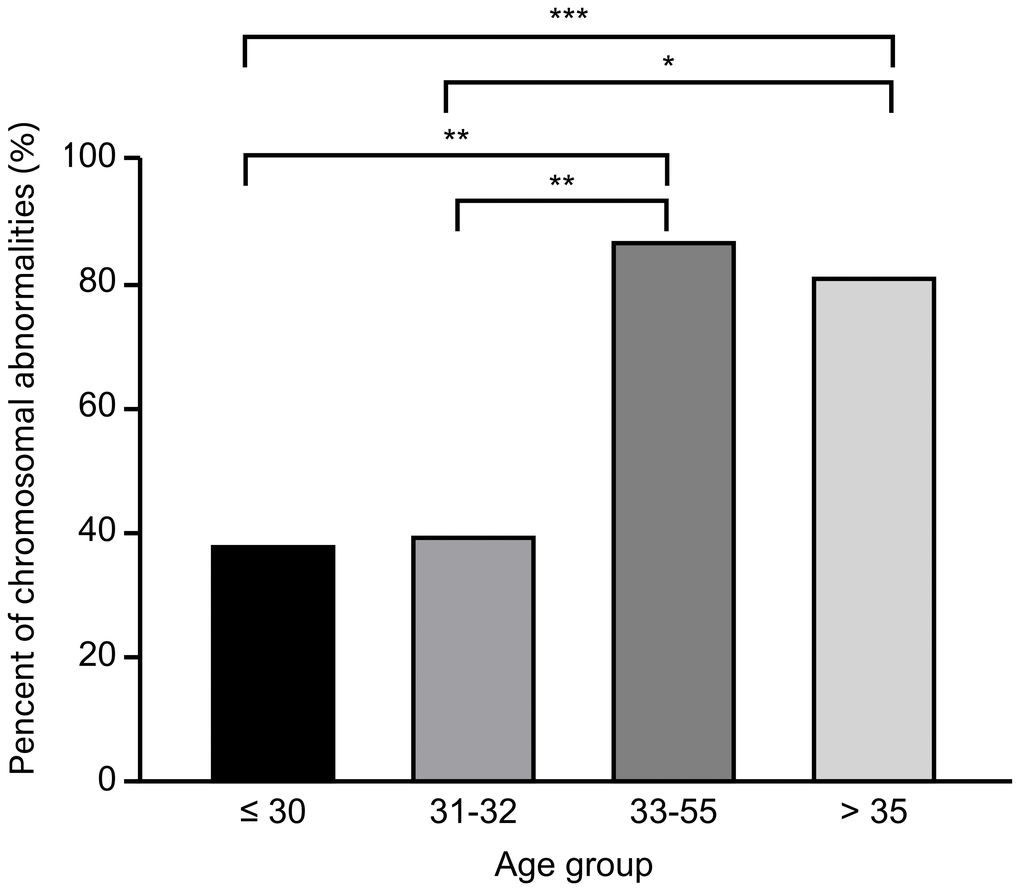
Figure 4. Distribution of chromosomal abnormalities in products of conception in different age groups.
Reproductive markers were not risk factors for a chromosomally aberrant fetus
In our study, several reproductive markers, such as AMH, AFC, basal FSH level, the number of oocytes retrieved and previous reproductive history, including prior gravidity and miscarriage, were significantly different between young and old patients with DOR in our study. The advanced age group showed lower AMH, AFC and number of oocytes retrieved and higher FSH, indicating poorer reproductive conditions. However, these factors were suggested not to be associated with abnormal chromosomes of chorionic villi after multivariate analysis. AMH, AFC, and basal FSH levels might reflect the quantity and quality of follicles and predict the number of transferred embryos through ovarian response to COS (controlled ovarian stimulation, COS). We included standardized and centralized measures of these reproductive markers, which were paired to a single IVF/ICSI-ET cycle leading to a clinical pregnancy to reduce the impact of subjectivity of the operator and within 1 year to weaken the influence of age-dependent variations. Furthermore, because the relationships of AMH, AFC and basal FSH levels with the number of retrieved oocytes have been proved, our study included the number of retrieved oocytes in multivariable analyses. Plante et al. [31] found that AMH levels did not differ between women with an aneuploid fetus and women with a euploid fetus. Notably, one study [17] confirmed a higher risk of abortion after IVF in women with low AMH (≤1.6 ng/ml) but only in older women (i.e., >34 years old). Another study found that AFC was not associated with any clinically significant increase in the risk of IVF pregnancy loss among women younger than 35 years of age [32]. Several studies were unable to confirm any independent association of AMH, AFC, basal FSH levels and the number of previous pregnancies and spontaneous miscarriages with aneuploidy in individuals who experienced pregnancy loss [20, 33–35], consistent with our study. Previous studies confirmed that DOR, as defined by baseline FSH, was not associated with any clinically significant increase in the risk of IVF pregnancy loss in women younger than 35 years old, suggesting that good quality oocytes could be obtained from young patients with DOR with high FSH levels. Our study showed no relationship of AMH, AFC, basal FSH level, the number of oocytes retrieved and previous reproductive history with chromosomal abnormalities in POCs, while the mechanism remains unclear, and prospective study of large samples is needed.
Male age was a suspected risk factor
Male age was significantly different between the two groups due to female age, and it was a suspected risk factor for chromosome aneuploidy of POCs. Studies have often found conflicting results. Antonarakis [36] showed that only 5% of fathers contributed to trisomy 21 in their offspring, and male factors did not influence aneuploidy rates once a blastocyst was obtained [37]. However, a recent study found that male partners aged 20–24 years old showed a higher risk and those aged 30 years old or older had negligible effects on chromosomal aberration-related miscarriages compared to the effects of being 25–29 years old. Notably, our study excluded men older than 40 years old and/or with an abnormal karyotype to reduce the effect of male age. As a result, male age likely did not affect our results.
Different age indicates different diagnoses and treatment focuses in women with DOR
Based on our study, we suggest diagnosing and treating first-trimester abortion differently in women with DOR at different ages. For young women (≤32 years old) with DOR, clinical doctors could primarily focus on maternal factors, such as immune, endocrine, infectious and prethrombotic states, and increase confidence once oocytes are obtained. However, for older women (older than 32 years old), there is a high risk of first-trimester abortion due to chromosomal aberrations of the embryo. Genetic consultation and preimplantation genetic testing of aneuploidies (PGT-A) might be advisable in future clinical practice.
Strengths and limitations
To our knowledge, this report is the first study to show the effects of female age on chromosomal aberrations of POCs in women with DOR as defined by serum AMH and basal FSH levels simultaneously. Our study divided patients innovatively according to the qualities of human eggs and found a novel cut-off value of age. Furthermore, we examined the POCs via SNP microarray, which is different from PGT-A, which is used generally in current studies. The former is applied on the POCs and aims to adjust diagnostic and treatment strategies for the next ART cycle among women experiencing miscarriage. The latter is a preimplantation genetic test for aneuploidy applied on blastocysts and to improve the success rate in current ART cycles. We excluded women with a history of recurrent pregnancy loss (RPL) (defined as ≥2), which correlates with embryo aneuploidy [38], to reduce confounding risk factors. However, there are several limitations of our study. First, our conclusions are limited due to the retrospective design involving a single medical center, and the sample size was suboptimal. Second, consensus on the diagnostic criteria for DOR/OR is lacking. A previous study showed that the predictive value of OR screening tests could be low in a younger population [39]. Third, the database did not include occupational characteristics, environmental information, psychological conditions or lifestyle data, which could contribute to miscarriage [40].
Materials and Methods
Study cohort and selection criteria
We retrospectively enrolled 869 patients who experienced involuntary first-trimester miscarriage after ART and were treated with dilation and curettage (D&C). The aborted villous and embryonic tissues were transferred to a preimplantation genetic diagnosis center for genetic analyses from September 2016 to September 2019. The data were based on the Clinical Reproductive Medicine Management System/Electronic Medical Record Cohort Database (CCRM/EMRCD) in the Reproductive Medicine Center of the First Affiliated Hospital of Zhengzhou University and Henan Key Laboratory of Reproduction and Genetics. The committee in our hospital approved this study, and all of the patients provided written informed consent at their first consultation. A total of 104 patients defined as having DOR by the Federal Register Notice according to FSH >10 mIU/ml and/or AMH <1.0 ng/ml [41] were included. As shown in Figure 5, we excluded couples with abnormal chromosome karyotypes: females with a history of RPL (defined as ≥2) [10]; females with immunological abnormalities; couples with endocrine disorders in either partner, such as polycystic ovary syndrome (PCOS), thyroid dysfunction, diabetes, congenital adrenal hyperplasia, 21-hydroxylase deficiency and hyperprolactinemia; patients with uterine abnormalities, such as endometriosis/adenomyosis, submucous myoma or genital malformation; multiple pregnancies; cycles involving donor oocytes or sperm; and males older than 40 years old. Parental congenital and/or chronic diseases were taken into consideration, but none were detected. According to a recently published study of aneuploid human eggs [29], the resulting 63 participants were divided into a young group (aged 20–32 years old) and an old group (older than 32 years old). The numbers of participants were 18 and 45, respectively. For all of the patients, clinical information was obtained, including paternal characteristics (i.e., age, body mass index (BMI), and basal sex hormone levels), ART process (i.e., fertilization method and categories of embryos transferred) and results of genetic analyses.
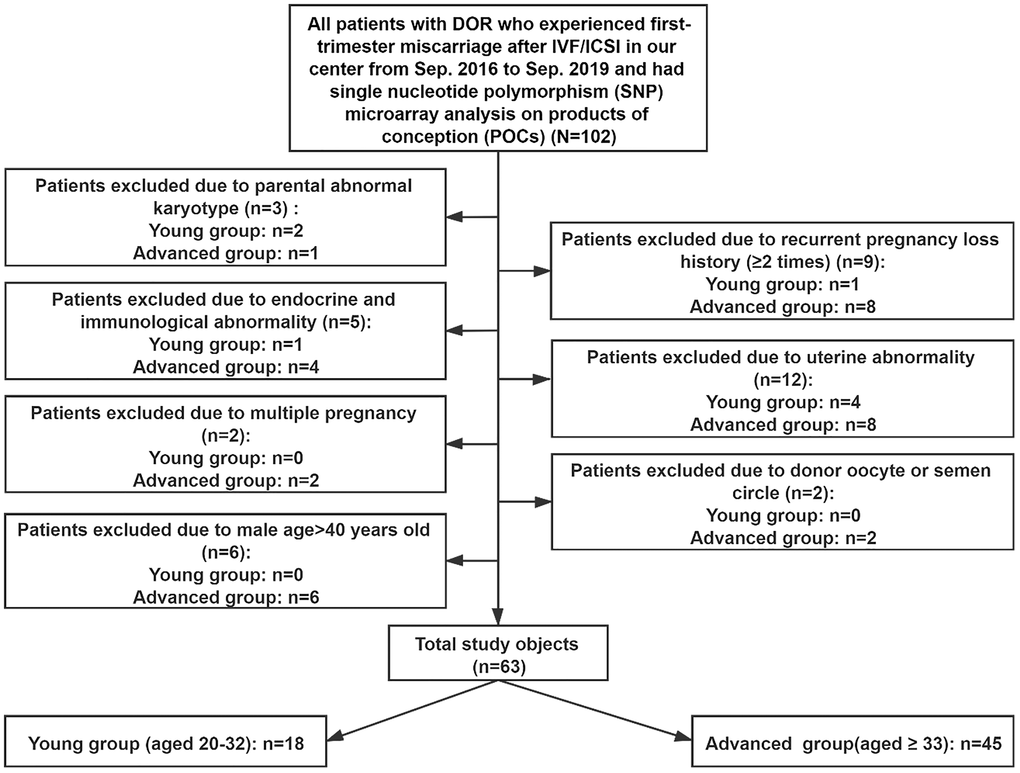
Figure 5. Study inclusion and exclusion.
ART protocol
Among the patients included in our study, 47 underwent in vitro fertilization (IVF), and 16 received intracytoplasmic sperm injection (ICSI). A previous study showed that different ART protocols produced no significant differences in the molecular karyotype of POC tissues [30]. For IVF/ICSI, ovarian stimulation was performed with a standard long protocol using gonadotropin releasing hormone (GnRH) agonists to prevent a premature luteinizing hormone (LH) surge and gonadotropins to stimulate follicle growth. When the diameter of the maximal follicle was greater than 20 mm, and more than 2/3 of the total follicles were >16 mm, human chorionic gonadotropin (hCG) was provided according to the serum FSH, LH, E2 and P levels during the cycles. Oocyte retrieval was performed under ultrasonic guidance 36–38 h later. Serum β-hCG levels were monitored on days 14 and 18. The outcome of pregnancy was defined by the detection of a gestational sac with a fetal heartbeat in the uterine cavity via ultrasound. Early missed abortion was defined by the absence of fetal cardiac pulsation in the uterine cavity after confirmation of clinical pregnancy.
DNA extraction and SNP microarray analysis
Chorionic villi from patients experiencing early missed abortion and undergoing D&C were sent to the preimplantation genetic diagnosis center for genetic analysis. Expert technicians cleaned the tissue with phosphate-buffered solution (PBS) to remove decidua and coagulated blood to avoid maternal genome contamination [42] and stored the samples at –80°C for DNA extraction.
Fresh DNA was extracted using a QIAamp DNA Mini Kit (Qiagen, Hilden, Germany), quantitated using Nanovue Plus (GE, Fairfield, CT, USA), and stored at -20° in preparation for subsequent SNP array analysis.
We used a Human CytoSNP-12v.21 Array (Illumina, San Diego, CA, USA) to detect molecular karyotypes and Genome-Studio (Illumina 2011) and Karyo-Studio software, version 1.4, to analyze the raw data. CNVs were mapped in the Database of Genomic Variants (DGV) (http://dgv.tcag.ca/dgv/app/faq) to identify candidate pathogenic CNVs. At least two independent technicians analyzed the data using strict criteria.
Statistical analysis
Statistical analyses were performed using SPSS software, version 22.0 (IBM Corp., Armonk, NY, USA). For demographics of the study subjects, normally distributed continuous variables are presented as the means ± SDs, and differences between groups were assessed using Student’s t-test. Continuous variables with skewed distributions are represented as medians (P25, P75) and were compared using the Kruskal-Wallis test. Categorical variables are expressed as frequencies (percentages) and were compared using the chi-square test. Logistic regression analysis was performed to identify the risk factors for chromosomal aberration of POCs in early pregnancy loss when adjusting for several confounding factors. Two-sided P values less than 0.05 were considered significant.
Author Contributions
WZ designed the study and performed data analyses and manuscript preparation. YG provided overall guidance. WN and JX performed the genetic analyses. JL, LZ, LY, XX, and BS contributed to the data collection. All of the authors reviewed and approved the final manuscript.
Acknowledgments
We thank all of the medical staff and patients for their work and participation in the Center for Reproductive Medicine at the First Affiliated Hospital of Zhengzhou University.
Conflicts of Interest
The authors declare that there are no conflicts of interest.
Funding
This study was supported by grant 81571409 from the National Natural Science Foundation of China.
References
- 1. Tal R, Seifer DB. Ovarian reserve testing: a user’s guide. Am J Obstet Gynecol. 2017; 217:129–40. https://doi.org/10.1016/j.ajog.2017.02.027 [PubMed]
- 2. Practice Committee of the American Society for Reproductive Medicine. Testing and interpreting measures of ovarian reserve: a committee opinion. Fertil Steril. 2015; 103:e9–17. https://doi.org/10.1016/j.fertnstert.2014.12.093 [PubMed]
- 3. Mutlu MF, Erdem A. Evaluation of ovarian reserve in infertile patients. J Turk Ger Gynecol Assoc. 2012; 13:196–203. https://doi.org/10.5152/jtgga.2012.28 [PubMed]
- 4. Busnelli A, Papaleo E, Del Prato D, La Vecchia I, Iachini E, Paffoni A, Candiani M, Somigliana E. A retrospective evaluation of prognosis and cost-effectiveness of IVF in poor responders according to the Bologna criteria. Hum Reprod. 2015; 30:315–22. https://doi.org/10.1093/humrep/deu319 [PubMed]
- 5. Oudendijk JF, Yarde F, Eijkemans MJ, Broekmans FJ, Broer SL. The poor responder in IVF: is the prognosis always poor?: a systematic review. Hum Reprod Update. 2012; 18:1–11. https://doi.org/10.1093/humupd/dmr037 [PubMed]
- 6. Lekamge DN, Barry M, Kolo M, Lane M, Gilchrist RB, Tremellen KP. Anti-Müllerian hormone as a predictor of IVF outcome. Reprod Biomed Online. 2007; 14:602–10. https://doi.org/10.1016/S1472-6483(10)61053-X [PubMed]
- 7. Harton GL, Munné S, Surrey M, Grifo J, Kaplan B, McCulloh DH, Griffin DK, Wells D, and PGD Practitioners Group. Diminished effect of maternal age on implantation after preimplantation genetic diagnosis with array comparative genomic hybridization. Fertil Steril. 2013; 100:1695–703. https://doi.org/10.1016/j.fertnstert.2013.07.2002 [PubMed]
- 8. Munné S, Chen S, Colls P, Garrisi J, Zheng X, Cekleniak N, Lenzi M, Hughes P, Fischer J, Garrisi M, Tomkin G, Cohen J. Maternal age, morphology, development and chromosome abnormalities in over 6000 cleavage-stage embryos. Reprod Biomed Online. 2007; 14:628–34. https://doi.org/10.1016/S1472-6483(10)61057-7 [PubMed]
- 9. Hardarson T, Hanson C, Lundin K, Hillensjö T, Nilsson L, Stevic J, Reismer E, Borg K, Wikland M, Bergh C. Preimplantation genetic screening in women of advanced maternal age caused a decrease in clinical pregnancy rate: a randomized controlled trial. Hum Reprod. 2008; 23:2806–12. https://doi.org/10.1093/humrep/den217 [PubMed]
- 10. Practice Committee of the American Society for Reproductive Medicine. Evaluation and treatment of recurrent pregnancy loss: a committee opinion. Fertil Steril. 2012; 98:1103–11. https://doi.org/10.1016/j.fertnstert.2012.06.048 [PubMed]
- 11. Practice Committee of the American Society for Reproductive Medicine. Testing and interpreting measures of ovarian reserve: a committee opinion. Fertil Steril. 2012; 98:1407–15. https://doi.org/10.1016/j.fertnstert.2012.09.036 [PubMed]
- 12. Ferraretti AP, Gianaroli L. The Bologna criteria for the definition of poor ovarian responders: is there a need for revision? Hum Reprod. 2014; 29:1842–45. https://doi.org/10.1093/humrep/deu139 [PubMed]
- 13. Ferraretti AP, La Marca A, Fauser BC, Tarlatzis B, Nargund G, Gianaroli L, and ESHRE working group on Poor Ovarian Response Definition. ESHRE consensus on the definition of ‘poor response’ to ovarian stimulation for in vitro fertilization: the Bologna criteria. Hum Reprod. 2011; 26:1616–24. https://doi.org/10.1093/humrep/der092 [PubMed]
- 14. Sills ES, Alper MM, Walsh AP. Ovarian reserve screening in infertility: practical applications and theoretical directions for research. Eur J Obstet Gynecol Reprod Biol. 2009; 146:30–36. https://doi.org/10.1016/j.ejogrb.2009.05.008 [PubMed]
- 15. van der Stroom EM, König TE, van Dulmen-den Broeder E, Elzinga WS, van Montfrans JM, Haadsma ML, Lambalk CB. Early menopause in mothers of children with Down syndrome? Fertil Steril. 2011; 96:985–90. https://doi.org/10.1016/j.fertnstert.2011.07.1149 [PubMed]
- 16. Klein J, Sauer MV. Assessing fertility in women of advanced reproductive age. Am J Obstet Gynecol. 2001; 185:758–70. https://doi.org/10.1067/mob.2001.114689 [PubMed]
- 17. Tarasconi B, Tadros T, Ayoubi JM, Belloc S, de Ziegler D, Fanchin R. Serum antimüllerian hormone levels are independently related to miscarriage rates after in vitro fertilization-embryo transfer. Fertil Steril. 2017; 108:518–24. https://doi.org/10.1016/j.fertnstert.2017.07.001 [PubMed]
- 18. Silberstein T, MacLaughlin DT, Shai I, Trimarchi JR, Lambert-Messerlian G, Seifer DB, Keefe DL, Blazar AS. Mullerian inhibiting substance levels at the time of HCG administration in IVF cycles predict both ovarian reserve and embryo morphology. Hum Reprod. 2006; 21:159–63. https://doi.org/10.1093/humrep/dei270 [PubMed]
- 19. Huang LN, Jun SH, Drubach N, Dahan MH. Predictors of in vitro fertilization outcomes in women with highest follicle-stimulating hormone levels ≥ 12 IU/L: a prospective cohort study. PLoS One. 2015; 10:e0124789. https://doi.org/10.1371/journal.pone.0124789 [PubMed]
- 20. Thum MY, Abdalla HI, Taylor D. Relationship between women’s age and basal follicle-stimulating hormone levels with aneuploidy risk in in vitro fertilization treatment. Fertil Steril. 2008; 90:315–21. https://doi.org/10.1016/j.fertnstert.2007.06.063 [PubMed]
- 21. Ebner T, Sommergruber M, Moser M, Shebl O, Schreier-Lechner E, Tews G. Basal level of anti-Müllerian hormone is associated with oocyte quality in stimulated cycles. Hum Reprod. 2006; 21:2022–26. https://doi.org/10.1093/humrep/del127 [PubMed]
- 22. Riggs R, Kimble T, Oehninger S, Bocca S, Zhao Y, Leader B, Stadtmauer L. Anti-Müllerian hormone serum levels predict response to controlled ovarian hyperstimulation but not embryo quality or pregnancy outcome in oocyte donation. Fertil Steril. 2011; 95:410–12. https://doi.org/10.1016/j.fertnstert.2010.07.1042 [PubMed]
- 23. Kushnir VA, Barad DH, Gleicher N. Association of abnormal ovarian reserve parameters with a higher incidence of aneuploid blastocysts. Obstet Gynecol. 2013; 121:1361. https://doi.org/10.1097/AOG.0b013e3182941ab0 [PubMed]
- 24. Miller DT, Adam MP, Aradhya S, Biesecker LG, Brothman AR, Carter NP, Church DM, Crolla JA, Eichler EE, Epstein CJ, Faucett WA, Feuk L, Friedman JM, et al. Consensus statement: chromosomal microarray is a first-tier clinical diagnostic test for individuals with developmental disabilities or congenital anomalies. Am J Hum Genet. 2010; 86:749–64. https://doi.org/10.1016/j.ajhg.2010.04.006 [PubMed]
- 25. Cimadomo D, Fabozzi G, Vaiarelli A, Ubaldi N, Ubaldi FM, Rienzi L. Impact of maternal age on oocyte and embryo competence. Front Endocrinol (Lausanne). 2018; 9:327. https://doi.org/10.3389/fendo.2018.00327 [PubMed]
- 26. Chang Y, Li J, Li X, Liu H, Liang X. Egg quality and pregnancy outcome in young infertile women with diminished ovarian reserve. Med Sci Monit. 2018; 24:7279–84. https://doi.org/10.12659/MSM.910410 [PubMed]
- 27. Woo I, Christenson LK, Gunewardena S, Ingles SA, Thomas S, Ahmady A, Chung K, Bendikson K, Paulson R, McGinnis LK. Micro-RNAs involved in cellular proliferation have altered expression profiles in granulosa of young women with diminished ovarian reserve. J Assist Reprod Genet. 2018; 35:1777–86. https://doi.org/10.1007/s10815-018-1239-9 [PubMed]
- 28. Skiadas CC, Duan S, Correll M, Rubio R, Karaca N, Ginsburg ES, Quackenbush J, Racowsky C. Ovarian reserve status in young women is associated with altered gene expression in membrana granulosa cells. Mol Hum Reprod. 2012; 18:362–71. https://doi.org/10.1093/molehr/gas008 [PubMed]
- 29. Gruhn JR, Zielinska AP, Shukla V, Blanshard R, Capalbo A, Cimadomo D, Nikiforov D, Chan AC, Newnham LJ, Vogel I, Scarica C, Krapchev M, Taylor D, et al. Chromosome errors in human eggs shape natural fertility over reproductive life span. Science. 2019; 365:1466–69. https://doi.org/10.1126/science.aav7321 [PubMed]
- 30. Li G, Jin H, Niu W, Xu J, Guo Y, Su Y, Sun Y. Effect of assisted reproductive technology on the molecular karyotype of missed abortion tissues. Biosci Rep. 2018; 38:BSR20180605. https://doi.org/10.1042/BSR20180605 [PubMed]
- 31. Plante BJ, Beamon C, Schmitt CL, Moldenhauer JS, Steiner AZ. Maternal antimullerian hormone levels do not predict fetal aneuploidy. J Assist Reprod Genet. 2010; 27:409–14. https://doi.org/10.1007/s10815-010-9433-4 [PubMed]
- 32. Bishop LA, Richter KS, Patounakis G, Andriani L, Moon K, Devine K. Diminished ovarian reserve as measured by means of baseline follicle-stimulating hormone and antral follicle count is not associated with pregnancy loss in younger in vitro fertilization patients. Fertil Steril. 2017; 108:980–87. https://doi.org/10.1016/j.fertnstert.2017.09.011 [PubMed]
- 33. Kline J, Kinney A, Reuss ML, Kelly A, Levin B, Ferin M, Warburton D. Trisomic pregnancy and the oocyte pool. Hum Reprod. 2004; 19:1633–43. https://doi.org/10.1093/humrep/deh310 [PubMed]
- 34. Weghofer A, Barad D, Li J, Gleicher N. Aneuploidy rates in embryos from women with prematurely declining ovarian function: a pilot study. Fertil Steril. 2007; 88:90–94. https://doi.org/10.1016/j.fertnstert.2006.11.081 [PubMed]
- 35. Havryliuk Y, Dragisic KG, Rosenwaks Z, Spandorfer S. O-49: baseline serum FSH levels in poor prognostic IVF patients: do abnormal levels predict fetal aneuploidy? Fertil Steril. 2006; 86:S21.
- 36. Antonarakis SE. Parental origin of the extra chromosome in trisomy 21 as indicated by analysis of DNA polymorphisms. Down Syndrome Collaborative Group. N Engl J Med. 1991; 324:872–76. https://doi.org/10.1056/NEJM199103283241302 [PubMed]
- 37. Mazzilli R, Cimadomo D, Vaiarelli A, Capalbo A, Dovere L, Alviggi E, Dusi L, Foresta C, Lombardo F, Lenzi A, Tournaye H, Alviggi C, Rienzi L, Ubaldi FM. Effect of the male factor on the clinical outcome of intracytoplasmic sperm injection combined with preimplantation aneuploidy testing: observational longitudinal cohort study of 1,219 consecutive cycles. Fertil Steril. 2017; 108:961–972.e3. https://doi.org/10.1016/j.fertnstert.2017.08.033 [PubMed]
- 38. Youssef A, Vermeulen N, Lashley EE, Goddijn M, van der Hoorn ML. Comparison and appraisal of (inter)national recurrent pregnancy loss guidelines. Reprod Biomed Online. 2019; 39:497–503. https://doi.org/10.1016/j.rbmo.2019.04.008 [PubMed]
- 39. Barnhart K, Osheroff J. Follicle stimulating hormone as a predictor of fertility. Curr Opin Obstet Gynecol. 1998; 10:227–32. https://doi.org/10.1097/00001703-199806000-00009 [PubMed]
- 40. Erlandsson L, Lindgren R, Nääv Å, Krais AM, Strandberg B, Lundh T, Boman C, Isaxon C, Hansson SR, Malmqvist E. Exposure to wood smoke particles leads to inflammation, disrupted proliferation and damage to cellular structures in a human first trimester trophoblast cell line. Environ Pollut. 2020; 264:114790. https://doi.org/10.1016/j.envpol.2020.114790 [PubMed]
- 41. Centers for Disease Control and Prevention. Reporting of Pregnancy Success Rates from Assisted Reproductive Technology (ART) Programs. Contract No. 2015–21108. Washington, DC: Department of Health and Human Services, 2015.
- 42. Lathi RB, Milki AA. Tissue sampling technique affects accuracy of karyotype from missed abortions. J Assist Reprod Genet. 2002; 19:536–38. https://doi.org/10.1023/A:1020916003666 [PubMed]