Introduction
Atrial fibrillation (AF) is the most common arrhythmia in clinical practice. Its incidence increases dramatically with increasing age, also due to the aging-related cardiac remodeling [1]. Prevalence of AF in older adults is approximately 7.3% and subjects aged 80 years or older represent more than half of all cases of AF, with increasing estimates in coming decades [2]. However, the real prevalence of AF in the population, especially in older adults, is likely to be underestimated, as AF is often asymptomatic. Opportunistic pulse palpation or electrocardiogram (ECG) strips are most often unable to detect short AF episodes [3–5]. This is particularly true in high risk populations, such as patients with cryptogenic stroke, in which longer monitoring times showed to improve the detection rate of intermittent AF [4, 6]. Up to now, the latest European Society of Cardiology (ESC) guidelines on AF recommend opportunistic screening for AF by pulse palpation or ECG rhythm strip in patients ≥ 65 years and suggest more intensive ECG screening in individuals aged ≥ 75 years or those at high risk of stroke [7]. In hospital settings outside the intensive care units, continuous ECG monitoring and cardiac telemetry are not routinely used. However, several acute conditions could trigger supraventricular arrhythmias during hospitalization, even in patients with no history of AF. Furthermore, these events may be asymptomatic and therefore unrecognized. Older patients are often hospitalized for electrolyte disturbances, hypoxemia, acute organ failure and infections, and these abnormalities are associated to a greater risk of new-onset AF [8–10].
Subclinical AF (SCAF) is defined as AF episodes detected by implantable or wearable cardiac monitors and confirmed by visually reviewed intracardiac electrograms or ECG-recorded rhythm in subjects without symptoms attributable to AF, in whom clinical AF has not been previously detected [7]. Most of the data regarding SCAF derive from long-term studies on adult outpatients with known heart disease and implanted devices [11, 12] or continuous ECG monitoring in community-dwelling or selected populations (i.e. cryptogenic stroke) [4, 13, 14]. Conversely, short-term evidence on SCAF in older populations is scarce, especially in the hospital setting.
We performed an exploratory study to investigate both the prevalence and predictors of SCAF in multimorbid older adults hospitalized for acute medical diseases.
Results
General characteristics
Sixty-five patients have been enrolled. However, five patients were excluded because they did not meet the minimum quality criteria for a satisfactory ECG recording (legible and artifact-free recording for at least 48 hours). Therefore, all the analyses were conducted on 60 patients. None of the enrolled patients had any symptoms attributable to AF episodes during hospitalization [15]. None of the enrolled patients died during ECG recording. Mean age was 85.7±4.9 years, with female prevalence (58.3%). General characteristics of the study population are summarized in Table 1. Anemia (Hbg < 12 g/dl) was found in 43 patients (71.7%) and eGFR < 60 ml/min/1.73mq was found in 27 patients (45.0%). No patients took oral anticoagulants. All enrolled patients had a CHA2DS2-VASc score ≥3. Echocardiographic characteristics of the study population are described in Table 2. More than half of the study population (57.1%) had left ventricular hypertrophy (LVH) and 56.8% had LV diastolic dysfunction. Most patients (82.5%) had a preserved left ventricular ejection fraction (LVEF ≥ 50%). The burden of co-morbidities was high in almost all of the subjects. The more prevalent co-morbidities were hypertension, heart failure (HF), diabetes mellitus, chronic obstructive pulmonary disease (COPD) and cognitive impairment.
Table 1. General characteristics of the entire study population and according to SCAF status.
| Clinical characteristics | Study population (n° 60) | Absence of SCAF (n° 44) | Presence of SCAF (n° 16) | p* | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years) | 85.7±4.9 | 85.5±4.2 | 86.3±6.5 | 0.671 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex (females) | 58.3% | 61.3% | 50.0% | 0.430 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI (kg/mq) | 26.9±4.3 | 26.7±4.1 | 26.9±5.0 | 0.969 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Arterial hypertension | 83.1% | 84.1% | 80.0% | 0.715 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Type 2 Diabetes Mellitus | 33.9% | 34.1% | 33.3% | 0.957 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History of TIA/Stroke | 10.2% | 11.4% | 6.7% | 0.603 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History of CAD | 11.9% | 13.6% | 6.7% | 0.471 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History of chronic HF | 42.9% | 52.3% | 40.0% | 0.412 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History of COPD | 33.9% | 36.4% | 26.7% | 0.493 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cognitive impairment | 34.5% | 34.9% | 33.3% | 0.913 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GIC (High comorbidity) | 87.9% | 90.7% | 80.0% | 0.273 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ADL Hierarchy Scale: Assistance required | 43.1% | 44.2% | 40.0% | 0.854 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ADL Hierarchy Scale: Dependence | 41.4% | 41.9% | 40.0% | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Polypharmacy | 77.6% | 81.6% | 63.6% | 0.209 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CHA2DS2-VASc score | 4.6±1.2 | 4.8±1.2 | 4.1±1.2 | 0.060 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Systolic BP (mmHg) | 128.5±25.0 | 130.7±25.3 | 121.9±23.7 | 0.278 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diastolic BP (mmHg) | 68.1±11.5 | 68.3±12.7 | 67.5±7.1 | 0.817 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heart rate (bpm) | 80.0±14.5 | 78.5±13.1 | 83.8±17.7 | 0.232 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Main admission diagnoses** | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Acute Decompensated HF | 54.5% | 61.0% | 35.7% | 0.101 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| COPD exacerbation | 22.0% | 22.7% | 20.0% | 0.826 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pneumonia | 33.9% | 31.8% | 40.0% | 0.563 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Acute kidney injury | 10.2% | 13.6% | 0.0% | 0.131 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Acute respiratory failure | 55.9% | 59.1% | 46.7% | 0.403 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| UTI or other infections | 35.6% | 40.9% | 20.0% | 0.144 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Admission lab parameters | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NT-proBNP (pg/ml) | 2082 (426-5160) | 2366 (759-5136) | 1075 (354-6904) | 0.297 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hgb (g/dl) | 11.0±1.6 | 10.9±1.6 | 11.3±1.7 | 0.425 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WBC (n/mm3) | 8595 (6235-11413) | 8690 (6323-12682) | 8290 (3343-10080) | 0.292 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| eGFR (ml/min/1.73m2) | 66.3±31.3 | 63.5±30.4 | 73.8±33.5 | 0.263 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Serum sodium (mEq/l) | 140.0±4.1 | 139.8±4.4 | 140.3±3.1 | 0.722 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Serum potassium (mEq/l) | 4.3±0.5 | 4.2±0.5 | 4.3±0.4 | 0.629 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Albumin (g/dl) | 3.3±0.5 | 3.2±0.5 | 3.6±0.5 | 0.387 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total cholesterol (mg/dl) | 142.0±41.4 | 139.1±40.6 | 150.5±44.1 | 0.362 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CRP (mg/dl) | 8.0 (2.5-16.3) | 8.0 (2.7-16.8) | 6.7 (2.2-16.2) | 0.525 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Admission ABG parameters*** | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| pH | 7.46±0.06 | 7.46±0.05 | 7.46±0.09 | 0.890 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| pO2 (mmHg) | 58.5±15.1 | 56.6±14.4 | 64.3±16.2 | 0.128 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| pCO2 (mmHg) | 42.0±13.0 | 42.2±14.2 | 41.3±8.6 | 0.809 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| P/F | 253.9±63.7 | 247.3±59.0 | 269.5±76.7 | 0.300 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HCO3- (mmol/l) | 28.5±6.6 | 28.4±7.2 | 28.9±5.0 | 0.813 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lactates (mmol/l) | 1.4±0.7 | 1.5±0.8 | 1,3±0.4 | 0.391 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cardiovascular therapy on admission | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ACE-I/ARBs | 51.0% | 50.0% | 54.5% | 0.791 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diuretics | 63.3% | 63.2% | 63.6% | 0.977 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Beta blockers | 40.8% | 44.7% | 27.3% | 0.299 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dihydropyridine calcium channel blockers | 16.3% | 18.4% | 9.1% | 0.461 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mineralocorticoid receptor antagonists | 12.2% | 13.2% | 9.1% | 0.717 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antiplatelet drugs | 40.0% | 43.2% | 31.3% | 0.404 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Statins | 20.4% | 18.4% | 27.3% | 0.521 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| *Comparison between patients with SCAF and without SCAF. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| **Each patient could have more than one admission diagnosis. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ***Performed in 48 patients. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Continuous variables were expressed as mean ± standard deviation, or as median and interquartile range if markedly skewed. Categorical variables were expressed as percentage. Polypharmacy was defined as the use of 5 or more drugs. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SCAF: subclinical atrial fibrillation; BMI: body mass index; TIA: transient ischemic attack; CAD: coronary artery disease; HF: heart failure; COPD: chronic obstructive pulmonary disease; GIC: geriatric index of comorbidity; ADL: activities of daily living; BP: blood pressure; UTI: urinary tract infection; NT-proBNP: N-terminal pro–B-type natriuretic peptide; Hgb: haemoglobin; WBC: white blood cells; eGFR: estimated glomerular filtration rate; CRP: C reactive protein; ABG: arterial blood gas; ACE-I: angiotensin-converting enzyme inhibitors; ARBs: angiotensin II receptor blockers. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 2. Echocardiographic and continuous ECG monitoring parameters of the entire study population and according to SCAF status.
| Study population (n° 60) | Absence of SCAF (n° 44) | Presence of SCAF (n° 16) | p* | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Echocardiographic parameters | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LVMI (g/mq) | 99.4±32.2 | 96.0±30.5 | 115.2±39.0 | 0.233 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LVEF (%) | 55.8±9.5 | 54.9±9.3 | 58.4±10.1 | 0.323 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LAVI (ml/mq) | 36.4±11.2 | 36.4±11.3 | 36.6±11.4 | 0.977 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| E/E’ | 12.5±4.4 | 13.0±4.5 | 9.6±2.6 | 0.118 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TAPSE (mm) | 19.7±4.4 | 20.2±3.9 | 17.5±5.6 | 0.122 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PAPs (mmHg) | 32.8±7.9 | 33.0±8.0 | 32.0±8.4 | 0.799 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Continuous ECG monitoring parameters | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Interatrial block (yes)** | 36.7% | 29.5% | 56.3% | 0.058 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mean HR (bpm) | 72.6±12.2 | 71.0±11.7 | 77.1±12.9 | 0.087 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N° VEBs/24h | 1225 (278-2766) | 764 (242-2355) | 1996 (401-6002) | 0.176 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| VEBs burden (%) | 1.0 (0.0-3.0) | 1.0 (0.0-2.0) | 2.0 (1.0-5.5) | 0.033 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N° SVEBs/24h | 724 (284-3941) | 627 (249-2347) | 2726 (443-6591) | 0.032 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SVEBs burden (%) | 1.0 (0.0-3.8) | 1.0 (0.0-2.8) | 2.5 (1.0-5.8) | 0.020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Presence of PSVT or VT | 38.3% | 18.8% | 45.5% | 0.060 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| *Comparison between patients with SCAF and without SCAF. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| **Referred to ECG at admission. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Continuous variables were expressed as mean ± standard deviation, or as median and interquartile range if markedly skewed. Categorical variables were expressed as percentage. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SCAF: subclinical atrial fibrillation; ECG: electrocardiogram; LVMI: left ventricular mass index; LVEF: left ventricular ejection fraction; LAVI: left atrial volume index; TAPSE: tricuspid annular plane excursion; PAPs: systolic pulmonary artery pressure; HR: heart rate; VEBs: ventricular ectopic beats; SVEBs: supraventricular ectopic beats; PSVT: paroxysmal supraventricular tachycardia; VT: ventricular tachycardia. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Prevalence and predictors of subclinical atrial fibrillation
The mean duration of the valid ECG recording was 4.2±1.4 days. A SCAF was detected in 16 patients (26.7%). The majority of these patients had at least a SCAF episode lasting 6 minutes or longer (Figure 1). Maximum heart rate during SCAF episodes was 183.3±45.4 bpm and the median duration of the episodes was 4.7 (1.7-10.3) minutes. The median of SCAF burden, defined by the percentage of analysable time spent in AF, was 3.7% (0.4-6.0%). No significant difference emerged between patients with or without SCAF regarding baseline clinical characteristics, admission diagnosis, laboratory and arterial blood gas (ABG) parameters or cardiovascular (CV) therapy on admission (Table 1). Moreover, both prevalence of anemia (72.7% vs 68.8%, p=0.762) and estimated glomerular filtration rate (eGFR) < 60 ml/min/1.73mq (50.0% vs 25.0%, p= 0.084) did not differ between patients with or without SCAF. The two groups showed similar echocardiographic parameters (Table 2), without differences regarding prevalence of LVH (57.7% vs 55.6%, p=0.911) and LV diastolic dysfunction (58.6% vs 50.0%, p=0.663). Patients with SCAF had almost double prevalence of interatrial block at baseline ECG compared to patients without SCAF, although without reaching statistical significance (Table 2). Patients with SCAF had both a higher daily number and burden of supraventricular ectopic beats (SVEBs) and a higher burden of ventricular ectopic beats (VEBs), as described in Table 2. After categorizing the number of SVEBs/24h into tertiles, patients with ≥ 2004 SVEBs/24h (3° tertile) had more than 6-fold higher prevalence of SCAF compared to patients with < 411 SVEBs/24h (1° tertile) [OR 6.2 (95% CI: 1.1-34.7), p=0.038]. Patients with SCAF had a higher mean duration of valid ECG recording, although not statistically significant (4.9±1.4 vs 4.1±1.4 days, p=0.067). Time to first SCAF episode was found within 3 days of ECG recording in all enrolled patients (Figure 2).
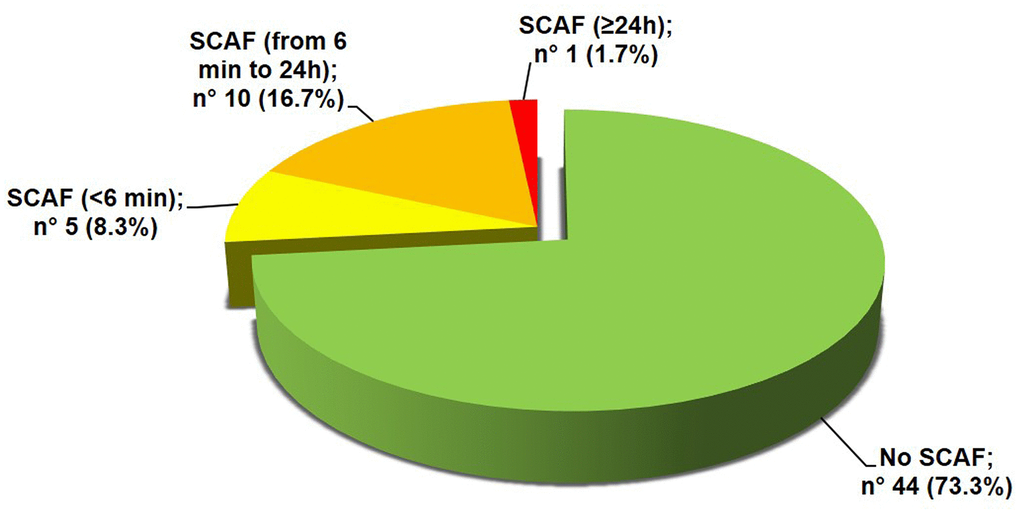
Figure 1. Prevalence of SCAF. SCAF: subclinical atrial fibrillation.
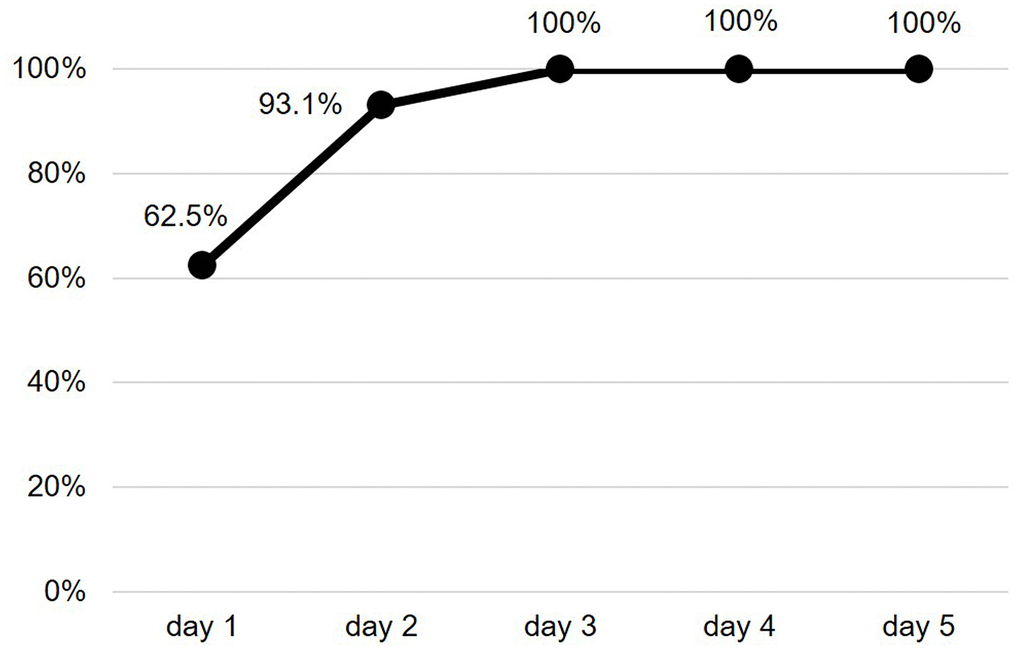
Figure 2. Cumulative frequency of SCAF detection. SCAF: subclinical atrial fibrillation.
Discussion
In our study, we investigated both the prevalence and predictors of SCAF in multimorbid older adults with no history of AF and hospitalized for acute medical diseases. More than a quarter of study population had a SCAF episode during hospitalization, often lasting more than 6 minutes, but less than 24 hours.
Prevalence of SCAF in clinical studies
Several previous studies investigated the prevalence of SCAF both in community-dwelling and selected high-risk populations, finding different results according to continuous or intermittent ECG recording, population screened, device used and duration of monitoring [3, 16]. A recent meta-analysis of studies, that performed a single time point screen for AF in the general population, found progressive increase in new AF detection rate with age, ranging from 0.34% (<60 years) to 2.73% (≥85 years) [17]. Studies on cardiac implanted electronic devices found high prevalence of SCAF, also because they often involved older subjects with significant structural heart disease, well-known risk factors for AF [18]. The ASSERT-II trial investigated the prevalence of SCAF ≥5 minutes, using subcutaneous electrocardiographic monitors, in older outpatients with risk factors, such as sleep apnea/obesity and left atrial enlargement, finding a detection rate of 34.4%/year [19]. In this trial, the median interval between the first episode of SCAF and clinical diagnosis of AF was only about 3 months [19], thus highlighting the propensity of these patients to later develop symptomatic disease. Lower rates were found using intermittent monitoring [20, 21]. A community-dwelling elderly population study (mean age: 79±5 years) found a prevalence of SCAF of 2.3% with 2 weeks of ambulatory ECG monitoring that increased to 4.1% with 4 weeks of monitoring [22]. Very recently, Gladstone DJ et al. detected a prevalence of 5.3% of asymptomatic AF lasting >5 minutes using a 2-week continuous wearable ECG patch monitor in individuals aged 75 years or older with hypertension and without known AF from primary care [23]. In the ASSERT-III, focused on older patients aged ≥80 years with hypertension and at least one additional AF risk factor who have been monitored continuously for 30–60 days, the authors found 15% of patients with at least one episode of SCAF ≥6 min [14]. The prevalence found in our study is likely the result of the peculiar setting in which our study took place. To the best of our knowledge, this is the first study that evaluated the prevalence of SCAF in such an older population (mean age: 85.7±4.9 years) in a hospital setting outside of intensive care units, using a continuous ECG recording.
New-onset atrial fibrillation in the hospital setting
New-onset AF is defined as a new onset or a first detectable episode of AF, whether symptomatic or not, and not necessarily detected through screening [7]. Several acute conditions may be associated with new-onset AF during hospitalization. This could explain at least in part the high prevalence of SCAF found in our study. Previous studies found that patients admitted for both pneumonia and other infections, such as urinary tract or intra-abdominal infections, had a greater risk of new-onset AF during hospitalization [8]. New-onset AF risk is directly correlated with the severity of the infection and linked to both prolonged hospitalization and increased mortality risk [24, 25]. Pneumonia is often followed by CV complications, included AF. A recent report found a new-onset AF in 10.1% of patients admitted for pneumonia occurring early during admission, particularly associated with CHA2DS2-VASc score >3 and high burden of co-morbidities [26]. Overall, new-onset AF is identified in about 2% of all hospitalizations and the main risk factors include older age, comorbidity index, male sex, history of hypertension, myocardial infarction, HF, cerebrovascular disease, chronic lung disease and laboratory abnormalities (increased creatinine and decreased serum albumin level) [27]. The AF linked to acute conditions shows a high risk of recurrence and a worse long-term outcome [8, 27]. In addition to infections and the high burden of cardiorespiratory comorbidities, more than half of our older patients had cardiac or respiratory failure on admission. Acute systemic inflammation, cardiac overload, hypoxemia, neuro-hormonal activation, autonomic dysfunctions, typical of all these acute conditions, predispose to cardiac complications and arrhythmias [28–30].
Possible clinical implications of SCAF
The clinical significance of SCAF is still uncertain. While SCAF episodes of shorter duration could be considered clinically irrelevant, previous studies found that SCAF episodes lasting more than 5-6 minutes are associated with an increased risk of clinical AF, ischemic stroke, CV events and mortality [11, 31–34]. Hence, the choice to identify and highlight those patients having a SCAF duration ≥ 6 minutes in our study, in agreement with previous studies [35], suggesting the possibility to follow these older patients more closely in order to prevent future adverse events. However, the risk of stroke in SCAF and the cost/benefit ratio of anticoagulation is still an open debate, given the lack of agreement in the published literature, especially in older people [36]. To date, there are no clear parameters of SCAF to initiate oral anticoagulation. In a post-hoc analysis of ASSERT study, only SCAF > 24h was associated with a significant increase in risk of stroke or thromboembolism [35]. However, a recent meta-analysis showed that it is not possible to define a SCAF duration threshold, because the relationship between SCAF duration and risk of stroke is likely to be continuous [37]. Overall, the risk of stroke would likely be lower than that of clinical AF [31]. It is important to note that shorter SCAF episodes are associated with higher probability of subsequent longer SCAF episodes and the progression to clinical AF over 2-year follow-up was found to be around 16% for detected SCAF lasting between 6 minutes and 24 hours [38, 39]. Moreover, patients with SCAF progression are also at greater risk for HF hospitalization [39]. Hence, a closer follow up of these older patients with short SCAF episodes could be advised. There is not always a temporal relationship between SCAF episodes and ischemic events, suggesting that SCAF, and in general AF, may be an epiphenomenon/marker of high risk patients, more than a direct risk factor [40]. At the same time, there are doubts regarding the real performance of CHA2DS2-VASc score in SCAF [18], although recent evidence showed a direct relationship between rate of stroke and CHA2DS2-VASc score also in SCAF [41]. Kaplan RM et al found that patients with a CHA2DS2-VASc score of 3 to 4 and an AF duration of >6 minutes had a risk of stroke >1%/year, an hypothetical threshold proposed for anticoagulant initiation in previous studies [41, 42]. It is important to note that all patients had a CHA2DS2-VASc score ≥3 in our study. Further studies may clarify the real benefits and indications of anticoagulation in older people with SCAF, especially regarding short SCAF episodes (≥ 6 minutes and <24 hours) [18, 43, 44]. The latest ESC Guidelines on AF suggest oral anticoagulation in selected patients at high risk of stroke (i.e. with previous stroke and/or age ≥75 years, or CHA2DS2-VASc ≥3) and a longer SCAF (≥5.5 hours), especially if high burden, mainly referring to SCAF detected by cardiac implantable electronic devices or cardiac monitors. However, a close follow-up for progression and risk factors control is also suggested for those with high risk of stroke and shorter SCAF episodes [7].
Besides a higher risk of stroke, HF hospitalization and mortality, older patients with AF have also a significantly lower cognitive performance than their counterparts in sinus rhythm, even after adjusting for age and the other CV risk factors [45, 46]. Both paroxysmal and persistent AF are associated with more than a 2-fold increased risk of silent cerebral vascular lesions [47] and there is an association between these findings in computed tomography or magnetic resonance in AF patients and risk of cognitive decline, even in the absence of manifest stroke [48]. Therefore, unrecognized AF, including SCAF, could contribute to accelerate cognitive decline in older subjects leading to disability and worse prognosis [49].
Predictors of SCAF and ECG monitoring techniques
Previous population-based studies on outpatients found that increased LA volume and left atrial enlargement were associated with increased rate of SCAF [22, 50]. Furthermore, older age, male sex, hypertension, history of HF and higher CHA2DS2-VASc score were found to be independent predictors of SCAF [19, 33, 51]. In our study, we found no significant clinical or echocardiographic predictors of SCAF, likely due to the small sample analyzed, the very old age and the high prevalence of both CV risks and CV disease in our population. Interatrial block (IAB) is a conduction delay between the atria, detectable on the ECG. Its prevalence increases with age and has been found to be associated with an increased risk of AF [52]. In agreement with previous studies, we found higher prevalence of SCAF in patients with IAB in our study, although without reaching the statistical significance, likely due to the small sample size [53].
Frequent atrial premature beats (APBs) were found to predict paroxysmal AF during follow-up in patients with acute ischemic stroke and without known AF [54]. In EMBRACE trial, atrial premature beat count on 24h Holter ECG (APBs/24h) was the only significant predictor of SCAF, with a probability of AF that increased from 7-9% in patients with <100 APBs/24h to 40% in patients with ≥1500 APBs/24h [55]. Not by chance, the only predictor of SCAF in our study was the number of SVEB/24h. The identification of patients at higher risk of developing AF during hospitalization could help improving cardiac monitoring strategies and management.
In the outpatient setting, the longer the ECG monitoring, the higher the probability of finding AF episodes, although the optimal duration of continuous ECG monitoring in high risk populations is still unclear [56]. In our study, we found that SCAF episodes during hospitalization often had both a low burden and a short duration (median duration of about 5 minutes), suggesting that routine clinical examination (pulse palpation, auscultation and blood pressure measurement) or short-duration, standard 12-lead ECG are likely unable to detect SCAF in this setting. Interestingly, the first SCAF episode was within the first 3 days of ECG monitoring in the entire sample of our study. This finding could affect the duration of continuous ECG recording in future studies on this topic.
Screening campaigns in high-risk populations, such as subjects with previous stroke or outpatients aged more than 75 years, have been found cost-effective [57], although duration of screening, screening device, population to screen, and features of AF associated with significant stroke risk are still debated topics [3, 58]. The clinical significance and the correct approach/management of SCAF in older adults is still unclear, although it may be an opportunity to reduce both CV morbidity and mortality, by preventing new onset of stroke and hospitalizations [18].
Study limits
The population taken into account (multimorbid hospitalized patients aged 80+ with no history of AF), almost never investigated in previous reports on this topic, represents the main novelty of our research. On the other hand, the small sample size due to this very particular population analyzed is the main limitation of our study. Future studies on larger samples could better clarify the prevalence and predictors of SCAF in older inpatients and, more importantly, could help understanding the implications of SCAF on clinical management and outcomes. Last, possible confounding risk factors occurred during hospitalization may not have been taken into account in the analyses.
Conclusions
We found a high prevalence of SCAF in older adults hospitalized for acute medical diseases with no history of AF. Detecting a SCAF could significantly affect the clinical management of these subjects, in terms of both risk factors control and prevention of AF progression and complications. The latest ESC Guidelines on AF emphasize the role of a multidisciplinary approach with an integrated AF management team. In this context, geriatricians play a key role in managing older patients with AF and multiple morbidities. Our findings could foster larger multicenter studies on hospitalized older populations, in order to better clarify the clinical meaning and implications of this unrecognized condition, including anticoagulant therapy indications.
Materials and Methods
Study design and population
We performed a cross-sectional study on older adults consecutively admitted for acute medical diseases, from January 2019 to July 2019, to the Internal Medicine and Geriatrics Unit of the Italian National Institute of Health and Science on Ageing (INRCA: Istituto Nazionale di Riposo e Cura per Anziani), which is the only organization specifically focused on geriatric care and gerontological research in Italy. Indeed, our hospital is dedicated to scientific research and care of older subjects (mostly aged 80 years or older), which are usually still excluded from clinical trials and in which scientific evidence is scarce. We took into account the following inclusion criteria: age ≥80 years, sinus rhythm on admission ECG, no history of AF reported by patient or by his medical records. We excluded patients having conditions with a life expectancy of less than 1 year (end-stage renal disease or dialysis, decompensated cirrhosis, advanced cancer, severe dementia or bed rest syndrome), decompensated hypo/hyperthyroidism, presence of implanted cardiac electronic devices (pacemaker or loop recorder), history of long QT syndrome or evidence of corrected QT interval (QTc) duration >500 milliseconds by the Fridericia formula. Patients admitted for acute diseases requiring continuous ECG monitoring (i.e. suspected cardiac syncope) were also excluded. All participants, or their legal representatives, gave their informed written consent and clinical investigations have been conducted according to the principles expressed in the Declaration of Helsinki. This study was approved by the local institutional ethics committee.
Proceedings
A 12-lead ECG was performed in all patients on admission to evaluate the presence of sinus rhythm, according to our routine clinical practice. After taking into account the inclusion and exclusion criteria, a continuous ECG monitoring (LIFECARD CF, Spacelabs Healthcare Limited, Hertford, United Kingdom) was performed in all enrolled patients, within the first 12 hours from admission, for 5 days. The choice of 5 days was based on the mean length of stay in our ward found in previous studies [59]. This device allows a continuous ECG recording up to a maximum of 7 days using 3 electrodes and 3 channels. It notifies the patient after the displacement of an electrode allowing the correct replacement. Moreover, the correct positioning of the electrodes has been monitored 3 times a day by both the medical and nursing staff. At the end of the recording, data have been elaborated using a dedicated software and interpreted by a single expert cardiologist (LP) of the Cardiology Unit, in order to identify SCAF episodes. A valid diagnostic ECG monitoring needed at least 48 hours of readable recording. Minimum quality criteria for a satisfactory ECG recording were based on the 2017 ISHNE-HRS expert consensus statement on ambulatory ECG and external cardiac monitoring/telemetry [60]. During hospitalization, all patients were managed according to the usual “good clinical practice”, regardless of whether they have participated to the study or not.
Clinical parameters
AF was defined by an irregular rhythm with absent P-waves lasting ≥ 30 seconds. Medical history and laboratory parameters were collected in each enrolled patient on admission. We took into account the following laboratory parameters: hemoglobin (Hgb), white blood cell count (WBC), creatinine, estimated glomerular filtration rate (eGFR), serum sodium and potassium, N-terminal pro-B-type natriuretic peptide (NT-proBNP), glycemia, C-reactive protein, albumin, total cholesterol. The eGFR was estimated using the CKD-EPI equation. Body mass index (BMI) was defined as the body mass divided by the square of the body height and was expressed in units of kg/m2. The CHA2DS2-VASc score was calculated according to the 2020 ESC Guidelines on AF [7]. The age-adjusted NT-proBNP cut-off of 1800 pg/mL, proposed by Januzzi et al, was used to diagnose acute decompensated heart failure (HF) [61]. An arterial blood gas (ABG) analysis was performed on admission as per clinical indication. During the hospitalization, a transthoracic echocardiographic evaluation was performed by the same physician, following a standardized protocol, to avoid inter-observer bias. The following main echocardiogram parameters were collected: left ventricular mass indexed to body surface area (g/m2) (LVMI), left ventricular ejection fraction (LVEF), left atrial volume index (LAVI), tricuspid annular plane excursion (TAPSE), systolic pulmonary artery pressure (PAPs), E/E’. Left ventricular hypertrophy (LVH) was defined as LV mass/body surface area (BSA) in mg/m2 > 115 (men) and > 95 (women) according to the 2018 ESC/ESH Guidelines [62, 63]. Left ventricular diastolic dysfunction was defined according to the 2016 ESC recommendations [64]. Regarding admission ECG, IAB was defined according to the following criteria: P-wave duration >120 msec without biphasic morphology in the inferior leads (II, III and aVF) for partial IAB, and P-wave duration >120 msec with biphasic morphology in the inferior leads for advanced IAB [53].
Geriatric comprehensive assessment
As previously reported [65], to evaluate patients’ functional status, the 7-point MDS Activities of Daily Living (ADL) Hierarchy scale was used. The ADL Hierarchy scale groups activities of daily living according to the stage of the disablement process in which they occur [66]. The ADL Hierarchy Scale ranges from 0 (no dependence) to 6 (total dependence). ADL disability was categorized as follows: no impairment (ADL Hierarchy Scale score <2), assistance required (ADL Hierarchy Scale score 2-4), and dependence (ADL Hierarchy Scale score ≥5). Cognitive impairment was based on a previous documented diagnosis, given that the result of any cognitive test could have been altered by the acute phase. The Geriatric Index of Comorbidity (GIC) was used to determine the burden of comorbidities and it was categorized as low comorbidity (GIC classes 1 or 2) and high comorbidity (GIC classes 3 or 4) [67]. Polypharmacy was defined as the use of 5 or more drugs.
Statistical analysis
Data were analyzed with the Statistical Package for Social Science version 13 (SPSS Inc. Chicago, Illinois, USA). A p-value less than 0.05 was defined as statistically significant. Continuous variables were checked for normality. Normal continuous variables were expressed as mean ± SD. Skewed variables were expressed as median and interquartile range. Categorical variables were expressed as percentages. The χ2 test was used to analyze the differences between categorical variables. The unpaired t test and Mann-Whitney test were used to compare quantitative variables.
Logistic regression analysis was used to evaluate the association between SCAF and number of SVEBs/24h categorized into tertiles.
Author Contributions
FS: conception and design, analysis and interpretation of data, drafting the article, agreement to be accountable for all aspects of the work and final approval of the version to be published; FG: analysis and interpretation of data, drafting the article, agreement to be accountable for all aspects of the work and final approval of the version to be published; LP: acquisition of data, analysis and interpretation of data, revising the article, agreement to be accountable for all aspects of the work and final approval of the version to be published; FEL: acquisition of data, agreement to be accountable for all aspects of the work and final approval of the version to be published; SR: acquisition of data, agreement to be accountable for all aspects of the work and final approval of the version to be published; PS: acquisition of data, agreement to be accountable for all aspects of the work and final approval of the version to be published; GD: acquisition of data, agreement to be accountable for all aspects of the work and final approval of the version to be published; RA: revising the article, agreement to be accountable for all aspects of the work and final approval of the version to be published; RS: conception and design, analysis and interpretation of data, revising the article, agreement to be accountable for all aspects of the work and final approval of the version to be published.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This research was funded by University “Politecnica delle Marche” (Ricerca di Ateneo to R. Sarzani) and a liberal unrestricted donation from Daiichi-Sankyo Italy.
References
- 1. Ravassa S, Ballesteros G, Díez J. Aging and atrial fibrillation: a matter of fibrosis. Aging (Albany NY). 2019; 11:9965–66. https://doi.org/10.18632/aging.102501 [PubMed]
- 2. Di Carlo A, Bellino L, Consoli D, Mori F, Zaninelli A, Baldereschi M, Cattarinussi A, D’Alfonso MG, Gradia C, Sgherzi B, Pracucci G, Piccardi B, Polizzi B, Inzitari D, and National Research Program: Progetto FAI. La Fibrillazione Atriale in Italia. Prevalence of atrial fibrillation in the Italian elderly population and projections from 2020 to 2060 for Italy and the European Union: the FAI Project. Europace. 2019; 21:1468–75. https://doi.org/10.1093/europace/euz141 [PubMed]
- 3. Jones NR, Taylor CJ, Hobbs FD, Bowman L, Casadei B. Screening for atrial fibrillation: a call for evidence. Eur Heart J. 2020; 41:1075–85. https://doi.org/10.1093/eurheartj/ehz834 [PubMed]
- 4. Gladstone DJ, Spring M, Dorian P, Panzov V, Thorpe KE, Hall J, Vaid H, O’Donnell M, Laupacis A, Côté R, Sharma M, Blakely JA, Shuaib A, et al, and EMBRACE Investigators and Coordinators. Atrial fibrillation in patients with cryptogenic stroke. N Engl J Med. 2014; 370:2467–77. https://doi.org/10.1056/NEJMoa1311376 [PubMed]
- 5. Turakhia MP, Shafrin J, Bognar K, Trocio J, Abdulsattar Y, Wiederkehr D, Goldman DP. Estimated prevalence of undiagnosed atrial fibrillation in the United States. PLoS One. 2018; 13:e0195088. https://doi.org/10.1371/journal.pone.0195088 [PubMed]
- 6. Ritter MA, Kochhäuser S, Duning T, Reinke F, Pott C, Dechering DG, Eckardt L, Ringelstein EB. Occult atrial fibrillation in cryptogenic stroke: detection by 7-day electrocardiogram versus implantable cardiac monitors. Stroke. 2013; 44:1449–52. https://doi.org/10.1161/STROKEAHA.111.676189 [PubMed]
- 7. Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, Blomström-Lundqvist C, Boriani G, Castella M, Dan GA, Dilaveris PE, Fauchier L, Filippatos G, Kalman JM, et al, and ESC Scientific Document Group. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur Heart J. 2021; 42:373–498. https://doi.org/10.1093/eurheartj/ehaa612 [PubMed]
- 8. Walkey AJ, Benjamin EJ, Lubitz SA. New-onset atrial fibrillation during hospitalization. J Am Coll Cardiol. 2014; 64:2432–33. https://doi.org/10.1016/j.jacc.2014.09.034 [PubMed]
- 9. Sibley S, Muscedere J. New-onset atrial fibrillation in critically ill patients. Can Respir J. 2015; 22:179–82. https://doi.org/10.1155/2015/394961 [PubMed]
- 10. Moss TJ, Calland JF, Enfield KB, Gomez-Manjarres DC, Ruminski C, DiMarco JP, Lake DE, Moorman JR. New-Onset Atrial Fibrillation in the Critically Ill. Crit Care Med. 2017; 45:790–97. https://doi.org/10.1097/CCM.0000000000002325 [PubMed]
- 11. Healey JS, Connolly SJ, Gold MR, Israel CW, Van Gelder IC, Capucci A, Lau CP, Fain E, Yang S, Bailleul C, Morillo CA, Carlson M, Themeles E, et al, and ASSERT Investigators. Subclinical atrial fibrillation and the risk of stroke. N Engl J Med. 2012; 366:120–29. https://doi.org/10.1056/NEJMoa1105575 [PubMed]
- 12. Martin DT, Bersohn MM, Waldo AL, Wathen MS, Choucair WK, Lip GY, Ip J, Holcomb R, Akar JG, Halperin JL, and IMPACT Investigators. Randomized trial of atrial arrhythmia monitoring to guide anticoagulation in patients with implanted defibrillator and cardiac resynchronization devices. Eur Heart J. 2015; 36:1660–68. https://doi.org/10.1093/eurheartj/ehv115 [PubMed]
- 13. Sanna T, Diener HC, Passman RS, Di Lazzaro V, Bernstein RA, Morillo CA, Rymer MM, Thijs V, Rogers T, Beckers F, Lindborg K, Brachmann J, and CRYSTAL AF Investigators. Cryptogenic stroke and underlying atrial fibrillation. N Engl J Med. 2014; 370:2478–86. https://doi.org/10.1056/NEJMoa1313600 [PubMed]
- 14. Gorenek B, Bax J, Boriani G, Chen SA, Dagres N, Glotzer TV, Healey JS, Israel CW, Kudaiberdieva G, Levin LÅ, Lip GY, Martin D, Okumura K, et al, and ESC Scientific Document Group. Device-detected subclinical atrial tachyarrhythmias: definition, implications and management-an European Heart Rhythm Association (EHRA) consensus document, endorsed by Heart Rhythm Society (HRS), Asia Pacific Heart Rhythm Society (APHRS) and Sociedad Latinoamericana de Estimulación Cardíaca y Electrofisiología (SOLEACE). Europace. 2017; 19:1556–78. https://doi.org/10.1093/europace/eux163 [PubMed]
- 15. Lévy S, Santini L, Cappato R, Steinbeck G, Capucci A, Saksena S. Clinical classification and the subclinical atrial fibrillation challenge: a position paper of the European Cardiac Arrhythmia Society. J Interv Card Electrophysiol. 2020; 59:495–507. https://doi.org/10.1007/s10840-020-00859-y [PubMed]
- 16. Khurshid S, Healey JS, McIntyre WF, Lubitz SA. Population-Based Screening for Atrial Fibrillation. Circ Res. 2020; 127:143–54. https://doi.org/10.1161/CIRCRESAHA.120.316341 [PubMed]
- 17. Lowres N, Olivier J, Chao TF, Chen SA, Chen Y, Diederichsen A, Fitzmaurice DA, Gomez-Doblas JJ, Harbison J, Healey JS, Hobbs FD, Kaasenbrood F, Keen W, et al. Estimated stroke risk, yield, and number needed to screen for atrial fibrillation detected through single time screening: a multicountry patient-level meta-analysis of 141,220 screened individuals. PLoS Med. 2019; 16:e1002903. https://doi.org/10.1371/journal.pmed.1002903 [PubMed]
- 18. Noseworthy PA, Kaufman ES, Chen LY, Chung MK, Elkind MS, Joglar JA, Leal MA, McCabe PJ, Pokorney SD, Yao X, and American Heart Association Council on Clinical Cardiology Electrocardiography and Arrhythmias Committee, and Council on Arteriosclerosis, Thrombosis and Vascular Biology, and Council on Cardiovascular and Stroke Nursing, and Stroke Council. Subclinical Device-Detected Atrial Fibrillation: Pondering the Knowledge Gap: A Scientific Statement From the American Heart Association. Circulation. 2019; 140:e944–63. https://doi.org/10.1161/CIR.0000000000000740 [PubMed]
- 19. Healey JS, Alings M, Ha A, Leong-Sit P, Birnie DH, de Graaf JJ, Freericks M, Verma A, Wang J, Leong D, Dokainish H, Philippon F, Barake W, et al, and ASSERT-II Investigators. Subclinical Atrial Fibrillation in Older Patients. Circulation. 2017; 136:1276–83. https://doi.org/10.1161/CIRCULATIONAHA.117.028845 [PubMed]
- 20. Halcox JP, Wareham K, Cardew A, Gilmore M, Barry JP, Phillips C, Gravenor MB. Assessment of Remote Heart Rhythm Sampling Using the AliveCor Heart Monitor to Screen for Atrial Fibrillation: The REHEARSE-AF Study. Circulation. 2017; 136:1784–94. https://doi.org/10.1161/CIRCULATIONAHA.117.030583 [PubMed]
- 21. Svennberg E, Engdahl J, Al-Khalili F, Friberg L, Frykman V, Rosenqvist M. Mass Screening for Untreated Atrial Fibrillation: The STROKESTOP Study. Circulation. 2015; 131:2176–84. https://doi.org/10.1161/CIRCULATIONAHA.114.014343 [PubMed]
- 22. Rooney MR, Soliman EZ, Lutsey PL, Norby FL, Loehr LR, Mosley TH, Zhang M, Gottesman RF, Coresh J, Folsom AR, Alonso A, Chen LY. Prevalence and Characteristics of Subclinical Atrial Fibrillation in a Community-Dwelling Elderly Population: The ARIC Study. Circ Arrhythm Electrophysiol. 2019; 12:e007390. https://doi.org/10.1161/CIRCEP.119.007390 [PubMed]
- 23. Gladstone DJ, Wachter R, Schmalstieg-Bahr K, Quinn FR, Hummers E, Ivers N, Marsden T, Thornton A, Djuric A, Suerbaum J, von Grünhagen D, McIntyre WF, Benz AP, et al, and SCREEN-AF Investigators and Coordinators. Screening for Atrial Fibrillation in the Older Population: A Randomized Clinical Trial. JAMA Cardiol. 2021; 6:558–67. https://doi.org/10.1001/jamacardio.2021.0038 [PubMed]
- 24. Walkey AJ, Wiener RS, Ghobrial JM, Curtis LH, Benjamin EJ. Incident stroke and mortality associated with new-onset atrial fibrillation in patients hospitalized with severe sepsis. JAMA. 2011; 306:2248–54. https://doi.org/10.1001/jama.2011.1615 [PubMed]
- 25. Klein Klouwenberg PM, Frencken JF, Kuipers S, Ong DS, Peelen LM, van Vught LA, Schultz MJ, van der Poll T, Bonten MJ, Cremer OL, and MARS Consortium *. Incidence, Predictors, and Outcomes of New-Onset Atrial Fibrillation in Critically Ill Patients with Sepsis. A Cohort Study. Am J Respir Crit Care Med. 2017; 195:205–11. https://doi.org/10.1164/rccm.201603-0618OC [PubMed]
- 26. Pieralli F, Biondo B, Vannucchi V, Falcone M, Antonielli E, De Marzi G, Casati C, Maddaluni L, Nozzoli C, Olivotto I. Performance of the CHA2DS2-VASc score in predicting new onset atrial fibrillation during hospitalization for community-acquired pneumonia. Eur J Intern Med. 2019; 62:24–28. https://doi.org/10.1016/j.ejim.2019.01.012 [PubMed]
- 27. Massera D, Wang D, Vorchheimer DA, Negassa A, Garcia MJ. Increased risk of stroke and mortality following new-onset atrial fibrillation during hospitalization. Europace. 2017; 19:929–36. https://doi.org/10.1093/europace/euw110 [PubMed]
- 28. Corrales-Medina VF, Musher DM, Wells GA, Chirinos JA, Chen L, Fine MJ. Cardiac complications in patients with community-acquired pneumonia: incidence, timing, risk factors, and association with short-term mortality. Circulation. 2012; 125:773–81. https://doi.org/10.1161/CIRCULATIONAHA.111.040766 [PubMed]
- 29. Gorenek B, Halvorsen S, Kudaiberdieva G, Bueno H, Van Gelder IC, Lettino M, Marin F, Masip J, Mueller C, Okutucu S, Poess J, Potpara TS, Price S, Lip GY. Atrial fibrillation in acute heart failure: A position statement from the Acute Cardiovascular Care Association and European Heart Rhythm Association of the European Society of Cardiology. Eur Heart J Acute Cardiovasc Care. 2020; 9:348–57. https://doi.org/10.1177/2048872619894255 [PubMed]
- 30. Maida CD, Vasto S, Di Raimondo D, Casuccio A, Vassallo V, Daidone M, Del Cuore A, Pacinella G, Cirrincione A, Simonetta I, Della Corte V, Rizzica S, Geraci G, et al. Inflammatory activation and endothelial dysfunction markers in patients with permanent atrial fibrillation: a cross-sectional study. Aging (Albany NY). 2020; 12:8423–33. https://doi.org/10.18632/aging.103149 [PubMed]
- 31. Mahajan R, Perera T, Elliott AD, Twomey DJ, Kumar S, Munwar DA, Khokhar KB, Thiyagarajah A, Middeldorp ME, Nalliah CJ, Hendriks JM, Kalman JM, Lau DH, Sanders P. Subclinical device-detected atrial fibrillation and stroke risk: a systematic review and meta-analysis. Eur Heart J. 2018; 39:1407–15. https://doi.org/10.1093/eurheartj/ehx731 [PubMed]
- 32. Pastori D, Miyazawa K, Li Y, Székely O, Shahid F, Farcomeni A, Lip GY. Atrial high-rate episodes and risk of major adverse cardiovascular events in patients with cardiac implantable electronic devices. Clin Res Cardiol. 2020; 109:96–102. https://doi.org/10.1007/s00392-019-01493-z [PubMed]
- 33. Gonzalez M, Keating RJ, Markowitz SM, Liu CF, Thomas G, Ip JE, Lerman BB, Cheung JW. Newly detected atrial high rate episodes predict long-term mortality outcomes in patients with permanent pacemakers. Heart Rhythm. 2014; 11:2214–21. https://doi.org/10.1016/j.hrthm.2014.08.019 [PubMed]
- 34. Glotzer TV, Hellkamp AS, Zimmerman J, Sweeney MO, Yee R, Marinchak R, Cook J, Paraschos A, Love J, Radoslovich G, Lee KL, Lamas GA, and MOST Investigators. Atrial high rate episodes detected by pacemaker diagnostics predict death and stroke: report of the Atrial Diagnostics Ancillary Study of the MOde Selection Trial (MOST). Circulation. 2003; 107:1614–19. https://doi.org/10.1161/01.CIR.0000057981.70380.45 [PubMed]
- 35. Van Gelder IC, Healey JS, Crijns HJ, Wang J, Hohnloser SH, Gold MR, Capucci A, Lau CP, Morillo CA, Hobbelt AH, Rienstra M, Connolly SJ. Duration of device-detected subclinical atrial fibrillation and occurrence of stroke in ASSERT. Eur Heart J. 2017; 38:1339–44. https://doi.org/10.1093/eurheartj/ehx042 [PubMed]
- 36. Wilkinson C, Clegg A, Todd O, Rockwood K, Yadegarfar ME, Gale CP, Hall M. Atrial fibrillation and oral anticoagulation in older people with frailty: a nationwide primary care electronic health records cohort study. Age Ageing. 2021; 50:772–79. https://doi.org/10.1093/ageing/afaa265 [PubMed]
- 37. Rahimi K. Subclinical atrial fibrillation in need of more assertive evidence. Eur Heart J. 2017; 38:1345–47. https://doi.org/10.1093/eurheartj/ehx122 [PubMed]
- 38. Swiryn S, Orlov MV, Benditt DG, DiMarco JP, Lloyd-Jones DM, Karst E, Qu F, Slawsky MT, Turkel M, Waldo AL, and RATE Registry Investigators. Clinical Implications of Brief Device-Detected Atrial Tachyarrhythmias in a Cardiac Rhythm Management Device Population: Results from the Registry of Atrial Tachycardia and Atrial Fibrillation Episodes. Circulation. 2016; 134:1130–40. https://doi.org/10.1161/CIRCULATIONAHA.115.020252 [PubMed]
- 39. Wong JA, Conen D, Van Gelder IC, McIntyre WF, Crijns HJ, Wang J, Gold MR, Hohnloser SH, Lau CP, Capucci A, Botto G, Grönefeld G, Israel CW, et al. Progression of Device-Detected Subclinical Atrial Fibrillation and the Risk of Heart Failure. J Am Coll Cardiol. 2018; 71:2603–11. https://doi.org/10.1016/j.jacc.2018.03.519 [PubMed]
- 40. Brambatti M, Connolly SJ, Gold MR, Morillo CA, Capucci A, Muto C, Lau CP, Van Gelder IC, Hohnloser SH, Carlson M, Fain E, Nakamya J, Mairesse GH, et al, and ASSERT Investigators. Temporal relationship between subclinical atrial fibrillation and embolic events. Circulation. 2014; 129:2094–99. https://doi.org/10.1161/CIRCULATIONAHA.113.007825 [PubMed]
- 41. Kaplan RM, Koehler J, Ziegler PD, Sarkar S, Zweibel S, Passman RS. Stroke Risk as a Function of Atrial Fibrillation Duration and CHA2DS2-VASc Score. Circulation. 2019; 140:1639–46. https://doi.org/10.1161/CIRCULATIONAHA.119.041303 [PubMed]
- 42. Eckman MH, Singer DE, Rosand J, Greenberg SM. Moving the tipping point: the decision to anticoagulate patients with atrial fibrillation. Circ Cardiovasc Qual Outcomes. 2011; 4:14–21. https://doi.org/10.1161/CIRCOUTCOMES.110.958108 [PubMed]
- 43. Lopes RD, Alings M, Connolly SJ, Beresh H, Granger CB, Mazuecos JB, Boriani G, Nielsen JC, Conen D, Hohnloser SH, Mairesse GH, Mabo P, Camm AJ, Healey JS. Rationale and design of the Apixaban for the Reduction of Thrombo-Embolism in Patients With Device-Detected Sub-Clinical Atrial Fibrillation (ARTESiA) trial. Am Heart J. 2017; 189:137–45. https://doi.org/10.1016/j.ahj.2017.04.008 [PubMed]
- 44. Kirchhof P, Blank BF, Calvert M, Camm AJ, Chlouverakis G, Diener HC, Goette A, Huening A, Lip GY, Simantirakis E, Vardas P. Probing oral anticoagulation in patients with atrial high rate episodes: Rationale and design of the Non-vitamin K antagonist Oral anticoagulants in patients with Atrial High rate episodes (NOAH-AFNET 6) trial. Am Heart J. 2017; 190:12–18. https://doi.org/10.1016/j.ahj.2017.04.015 [PubMed]
- 45. Diener HC, Hart RG, Koudstaal PJ, Lane DA, Lip GY. Atrial Fibrillation and Cognitive Function: JACC Review Topic of the Week. J Am Coll Cardiol. 2019; 73:612–19. https://doi.org/10.1016/j.jacc.2018.10.077 [PubMed]
- 46. Kim D, Yang PS, Yu HT, Kim TH, Jang E, Sung JH, Pak HN, Lee MY, Lee MH, Lip GY, Joung B. Risk of dementia in stroke-free patients diagnosed with atrial fibrillation: data from a population-based cohort. Eur Heart J. 2019; 40:2313–23. https://doi.org/10.1093/eurheartj/ehz386 [PubMed]
- 47. Kalantarian S, Ay H, Gollub RL, Lee H, Retzepi K, Mansour M, Ruskin JN. Association between atrial fibrillation and silent cerebral infarctions: a systematic review and meta-analysis. Ann Intern Med. 2014; 161:650–58. https://doi.org/10.7326/M14-0538 [PubMed]
- 48. Knecht S, Oelschläger C, Duning T, Lohmann H, Albers J, Stehling C, Heindel W, Breithardt G, Berger K, Ringelstein EB, Kirchhof P, Wersching H. Atrial fibrillation in stroke-free patients is associated with memory impairment and hippocampal atrophy. Eur Heart J. 2008; 29:2125–32. https://doi.org/10.1093/eurheartj/ehn341 [PubMed]
- 49. Spannella F, Giulietti F, Cocci G, Landi L, Lombardi FE, Borioni E, Cenci A, Giordano P, Sarzani R. Acute Exacerbation of Chronic Obstructive Pulmonary Disease in Oldest Adults: Predictors of In-Hospital Mortality and Need for Post-acute Care. J Am Med Dir Assoc. 2019; 20:893–98. https://doi.org/10.1016/j.jamda.2019.01.125 [PubMed]
- 50. Bertelsen L, Diederichsen SZ, Haugan KJ, Brandes A, Graff C, Krieger D, Kronborg C, Køber L, Højberg S, Vejlstrup N, Svendsen JH. Left atrial volume and function assessed by cardiac magnetic resonance imaging are markers of subclinical atrial fibrillation as detected by continuous monitoring. Europace. 2020; 22:724–31. https://doi.org/10.1093/europace/euaa035 [PubMed]
- 51. Israel C, Kitsiou A, Kalyani M, Deelawar S, Ejangue LE, Rogalewski A, Hagemeister C, Minnerup J, Schäbitz WR. Detection of atrial fibrillation in patients with embolic stroke of undetermined source by prolonged monitoring with implantable loop recorders. Thromb Haemost. 2017; 117:1962–69. https://doi.org/10.1160/TH17-02-0072 [PubMed]
- 52. Ariyarajah V, Puri P, Kranis M, Wilner DA, Spodick DH. Prevalence of interatrial block in the Program of All-Inclusive Care for the Elderly (PACE). Am J Geriatr Cardiol. 2006; 15:174–77. https://doi.org/10.1111/j.1076-7460.2006.04518.x [PubMed]
- 53. Boccanelli A, Mureddu GF, Cesaroni G, Prati F, Rangoni F, Agabiti N, Davoli M, Scardovi AB, Latini R. Predictive value of interatrial block for atrial fibrillation in elderly subjects enrolled in the PREDICTOR study. J Electrocardiol. 2019; 54:22–27. https://doi.org/10.1016/j.jelectrocard.2019.02.011 [PubMed]
- 54. Wallmann D, Tüller D, Wustmann K, Meier P, Isenegger J, Arnold M, Mattle HP, Delacrétaz E. Frequent atrial premature beats predict paroxysmal atrial fibrillation in stroke patients: an opportunity for a new diagnostic strategy. Stroke. 2007; 38:2292–94. https://doi.org/10.1161/STROKEAHA.107.485110 [PubMed]
- 55. Gladstone DJ, Dorian P, Spring M, Panzov V, Mamdani M, Healey JS, Thorpe KE, and EMBRACE Steering Committee and Investigators. Atrial premature beats predict atrial fibrillation in cryptogenic stroke: results from the EMBRACE trial. Stroke. 2015; 46:936–41. https://doi.org/10.1161/STROKEAHA.115.008714 [PubMed]
- 56. Dussault C, Toeg H, Nathan M, Wang ZJ, Roux JF, Secemsky E. Electrocardiographic monitoring for detecting atrial fibrillation after ischemic stroke or transient ischemic attack: systematic review and meta-analysis. Circ Arrhythm Electrophysiol. 2015; 8:263–69. https://doi.org/10.1161/CIRCEP.114.002521 [PubMed]
- 57. Aronsson M, Svennberg E, Rosenqvist M, Engdahl J, Al-Khalili F, Friberg L, Frykman-Kull V, Levin LÅ. Cost-effectiveness of mass screening for untreated atrial fibrillation using intermittent ECG recording. Europace. 2015; 17:1023–29. https://doi.org/10.1093/europace/euv083 [PubMed]
- 58. Guerra F, Stronati G. Risk prediction models in atrial fibrillation: from theory to practice. Eur J Prev Cardiol. 2021; 28:584–85. https://doi.org/10.1093/eurjpc/zwaa133 [PubMed]
- 59. Spannella F, Giulietti F, Cocci G, Landi L, Borioni E, Lombardi FE, Rosettani G, Bernardi B, Bordoni V, Giordano P, Bordicchia M, Sarzani R. N-terminal pro B-Type natriuretic peptide is inversely correlated with low density lipoprotein cholesterol in the very elderly. Nutr Metab Cardiovasc Dis. 2018; 28:629–35. https://doi.org/10.1016/j.numecd.2018.02.013 [PubMed]
- 60. Steinberg JS, Varma N, Cygankiewicz I, Aziz P, Balsam P, Baranchuk A, Cantillon DJ, Dilaveris P, Dubner SJ, El-Sherif N, Krol J, Kurpesa M, La Rovere MT, et al. 2017 ISHNE-HRS expert consensus statement on ambulatory ECG and external cardiac monitoring/telemetry. Heart Rhythm. 2017; 14:e55–96. https://doi.org/10.1016/j.hrthm.2017.03.038 [PubMed]
- 61. Sarzani R, Spannella F, Giulietti F, Fedecostante M, Giordano P, Gattafoni P, Espinosa E, Busco F, Piccinini G, Dessì-Fulgheri P. NT-proBNP and Its Correlation with In-Hospital Mortality in the Very Elderly without an Admission Diagnosis of Heart Failure. PLoS One. 2016; 11:e0153759. https://doi.org/10.1371/journal.pone.0153759 [PubMed]
- 62. Williams B, Mancia G, Spiering W, Agabiti Rosei E, Azizi M, Burnier M, Clement D, Coca A, De Simone G, Dominiczak A, Kahan T, Mahfoud F, Redon J, et al, and List of authors/Task Force members:. 2018 Practice Guidelines for the management of arterial hypertension of the European Society of Hypertension and the European Society of Cardiology: ESH/ESC Task Force for the Management of Arterial Hypertension. J Hypertens. 2018; 36:2284–309. https://doi.org/10.1097/HJH.0000000000001961 [PubMed]
- 63. Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JG, Coats AJ, Falk V, González-Juanatey JR, Harjola VP, Jankowska EA, Jessup M, Linde C, Nihoyannopoulos P, et al, and Authors/Task Force Members, and Document Reviewers. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2016; 18:891–975. https://doi.org/10.1002/ejhf.592 [PubMed]
- 64. Nagueh SF, Smiseth OA, Appleton CP, Byrd BF 3rd, Dokainish H, Edvardsen T, Flachskampf FA, Gillebert TC, Klein AL, Lancellotti P, Marino P, Oh JK, Alexandru Popescu B, Waggoner AD, and Houston, Texas; Oslo, Norway; Phoenix, Arizona; Nashville, Tennessee; Hamilton, Ontario, Canada; Uppsala, Sweden; Ghent and Liège, Belgium; Cleveland, Ohio; Novara, Italy; Rochester, Minnesota; Bucharest, Romania; and St. Louis, Missouri. Recommendations for the Evaluation of Left Ventricular Diastolic Function by Echocardiography: An Update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2016; 17:1321–60. https://doi.org/10.1093/ehjci/jew082 [PubMed]
- 65. Spannella F, Giulietti F, Balietti P, Cocci G, Landi L, Lombardi FE, Borioni E, Bernardi B, Rosettani G, Bordoni V, Sarzani R. Renin-Angiotensin System Blockers and Statins Are Associated With Lower In-Hospital Mortality in Very Elderly Hypertensives. J Am Med Dir Assoc. 2018; 19:342–47. https://doi.org/10.1016/j.jamda.2017.09.023 [PubMed]
- 66. Morris JN, Fries BE, Morris SA. Scaling ADLs within the MDS. J Gerontol A Biol Sci Med Sci. 1999; 54:M546–53. https://doi.org/10.1093/gerona/54.11.m546 [PubMed]
- 67. Rozzini R, Frisoni GB, Ferrucci L, Barbisoni P, Sabatini T, Ranieri P, Guralnik JM, Trabucchi M. Geriatric Index of Comorbidity: validation and comparison with other measures of comorbidity. Age Ageing. 2002; 31:277–85. https://doi.org/10.1093/ageing/31.4.277 [PubMed]


