Introduction
Psoriasis is a chronic, systemic inflammatory skin condition that affects nearly 3% of the world’s population [1, 2]. The clinical manifestations of psoriasis are erythema and scaly skin, which can affect the entire body [3]. Psoriasis outbreaks are triggered by multiple factors, including hereditary factors, external environmental factors, upper respiratory tract infections, and external living conditions [4]. Psoriasis can seriously affect patient's quality of life; 20-30% of patients with psoriasis have joint pains [5]. In addition, patients with moderate or severe psoriasis are at an increased risk for metabolic syndrome and atherosclerotic cardiovascular disease [6]. Although current treatment measures for psoriasis are effective, a long-term remission cannot be achieved [7]. Therefore, it is necessary to develop novel therapeutic approaches for the treatment of psoriasis.
Curcumol is a polyphenol compound isolated from the rhizome of Rhizoma Curcumae; it is a pharmacological sesquiterpene with anti-tumor, anti-inflammatory, anti-viral, and anti-bacterial effects [8]. Curcumol inhibits proliferation of gastric cancer cells [9]. In addition, Li et al. indicated that curcumol reduces cigarette smoke extract-induced inflammatory injury in macrophages via reducing the release of intracellular reactive oxygen species (ROS) and pro-inflammatory factors [10]. Moreover, Chen et al. found that curcumol inhibits LPS-induced inflammatory response in RAW264.7 cells via suppressing the production of inflammatory cytokines, including TNF-α, IL-1β and IL-6 [11]. However, the biological role of curcumol in psoriasis remains unclear. Thus, it is urgent to investigate the role of curcumol in psoriasis.
The JAK/STAT signaling pathway is a cytokine stimulated signal transduction pathway that has been discovered in recent years. The transmission process of this signaling pathway is mainly composed of three components: tyrosine kinase associated receptor, Janus kinase and signal transducer and activator of transcription [12]. This pathway is involved in many important biological processes such as cell proliferation, differentiation, apoptosis, and immune regulation [13]. Activation of the JAK/STAT signal transduction pathway promotes cell growth and cell cycle transformation [14]. It's reported that matrine could inhibit the activation of JAK/STAT pathway to increase the inhibitory effect of drugs on lung cancer cells [15]. However, the mechanism by which curcumol regulates the occurrence and development of psoriasis remains unclear. This study aimed to investigate the role of curcumol during the progression of psoriasis.
M5 ((IL-1α, IL-17A, IL-22, oncostatin M, and TNF-α) can induce keratinocytes to exhibit some of the characteristics of psoriatic keratinocytes in vitro [16]. Meanwhile, it has been reported that IL-17 and IL-22 enhance cutaneous inflammation, leading to epidermal thickening of psoriatic lesions and adherence of psoriatic scales [17]. Therefore, in the present study, Normal human epidermal keratinocytes (NHEK) cells were treated with M5 to establish the model of psoriatic keratinocyte in vitro.
Results
Curcumol inhibits proliferation and cell cycle progression and promote apoptosis in stimulated human keratinocytes
First, we assessed the effect of curcumol on the viability of normal human epidermal keratinocyte (NHEK) cells using CCK-8 assay. As shown in Figure 1A, 24 h treatment with 5, 10, and 20 μM curcumol had a very limited effect on NHEK cell viability; however, 40 and 80 μM curcumol significantly reduced the cell viability. Thus, we used 10 and 20 μM curcumol in the following experiments. We then investigated the effect of curcumol on viability and proliferation of NHEK cells stimulated with a combination of proinflammatory cytokines characteristic for psoriasis (IL-1α, IL-17A, IL-22, oncostatin M, and TNF-α; mix M5). As shown in Figure 1B, 1C, M5 markedly increased the viability and proliferation of NHEK cells; however, these effects were reversed by curcumol. In addition, the result of apoptosis evaluation showed that M5 decreased the apoptosis of NHEK cells, while this phenomenon was notably reversed by curcumol (Figure 1D, 1E). Moreover, M5 notably decreased the percentage of NHEK cells in G0/G1 phases, but increased the percentage of cells in S-phase; however, these effects were reversed by curcumol treatment (Figure 1F). Furthermore, the expressions of cell cycle-associated proteins p27 Kip1 and CDK4 were determined using western blot assay. Consistently, M5 downregulated the level of p27 Kip1 and upregulated the level of CDK4; however, the effects of M5 on these proteins were all reversed by curcumol (Figure 1G–1I). All in all, these data suggested that curcumol could inhibit proliferation and cell cycle progression and promote apoptosis of M5-stimulated NHEK cells.
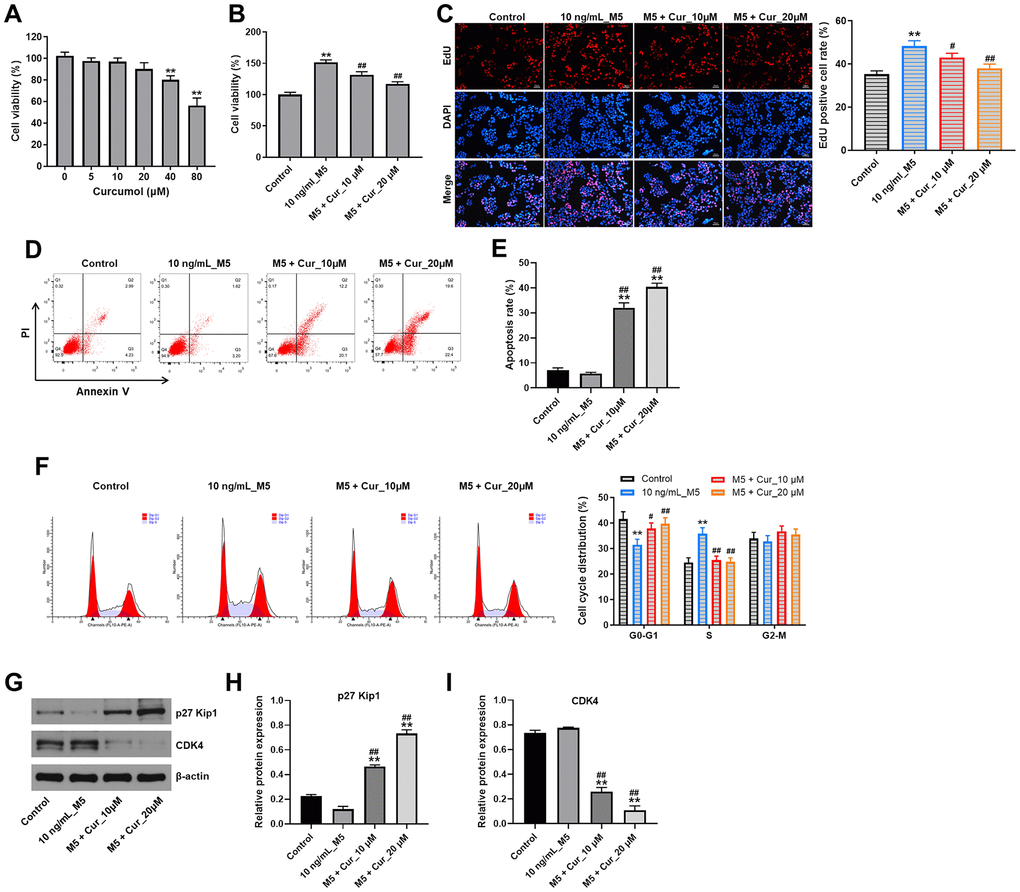
Figure 1. Curcumol inhibits proliferation and cell cycle progression and promote apoptosis in stimulated human keratinocytes. (A) CCK-8 cell viability assay in NHEK cells treated with curcumol (0, 5, 10, 20, 40 or 80 μM) for 24 h. (B) CCK-8 cell viability assay in NHEK cells pre-incubated with curcumol (10 or 20 μM) for 24 h, and stimulated with 10 ng/mL of M5 mix for 24 h. (C) Relative fluorescence levels were quantified by EdU and DAPI staining. (D, E) Flow cytometer assay was used to analyze cell apoptosis. (F) Cell cycle distribution was measured by flow cytometry. (G–I) Western blot was performed to measure the expressions of p27 Kip1 and CDK4 in NHEK cells. **P<0.01 compared with control group; #P<0.05, ##P<0.01 compared with 10 ng/mL_M5 group.
Curcumol suppresses inflammatory gene expression in M5-stimulated NHEK cells
Keratinocyte exposure to M5-proinflammatory cytokines induces expression of proinflammatory mediators, including cytokines, chemokines, and antimicrobial peptides [18]. Thus, we investigated whether curcumol could attenuate the inflammatory gene expression in M5-stimulated NHEK cells. As shown in Figure 2, M5 exposure significantly increased mRNA levels of cytokines (IL-6, IL-1β), chemokines (CXCL1, CXCL2), and antimicrobial peptides (LL37, β-defensin-2, S100A7 and S100A8) in NHEK cells. Importantly, this M5-induced inflammatory gene expression was notably suppressed by curcumol treatment, indicating that curcumol attenuates the inflammatory response in M5-stimulated NHEK cells.
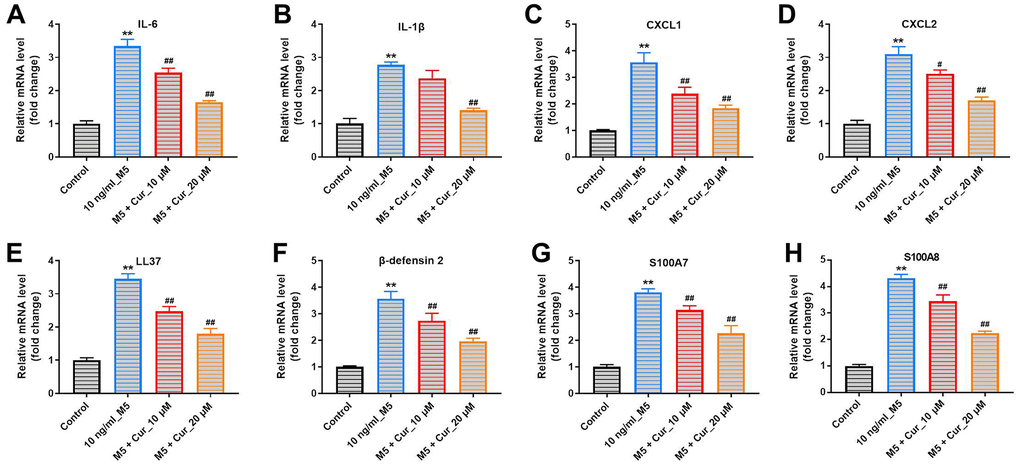
Figure 2. Curcumol suppresses inflammatory gene expression in M5-treated NHEK cells. NHEK cells were preincubated with curcumol (10 or 20 μM) for 24 h, stimulated with M5 (10 ng/mL) for 24 h, and gene expression of IL-6 (A), IL-1β (B), CXCL1 (C), CXCL2 (D), LL37 (E), β-defensin-2 (F), S100A7 (G), and S100A8 (H) was analyzed by RT-qPCR. **P<0.01 compared with control group; #P<0.05, ##P<0.01 compared with 10 ng/mL_M5 group.
Curcumol attenuates M5-induced oxidative stress damage in NHEK cells
We further investigated whether curcumol could protect NHEK cells against M5-induced oxidative stress damage. As illustrated in Figure 3A, curcumol dramatically inhibited M5-induced ROS production in NHEK cells. In addition, M5 significantly reduced the production of superoxide dismutase (SOD), glutathione (GSH), and catalase (CAT), and increased the production of malondialdehyde (MDA) in NHEK cells. However, these changes were markedly reversed by curcumol (Figure 3B–3E). These data indicated that curcumol could attenuate the M5-induced oxidative stress damage in NHEK cells.
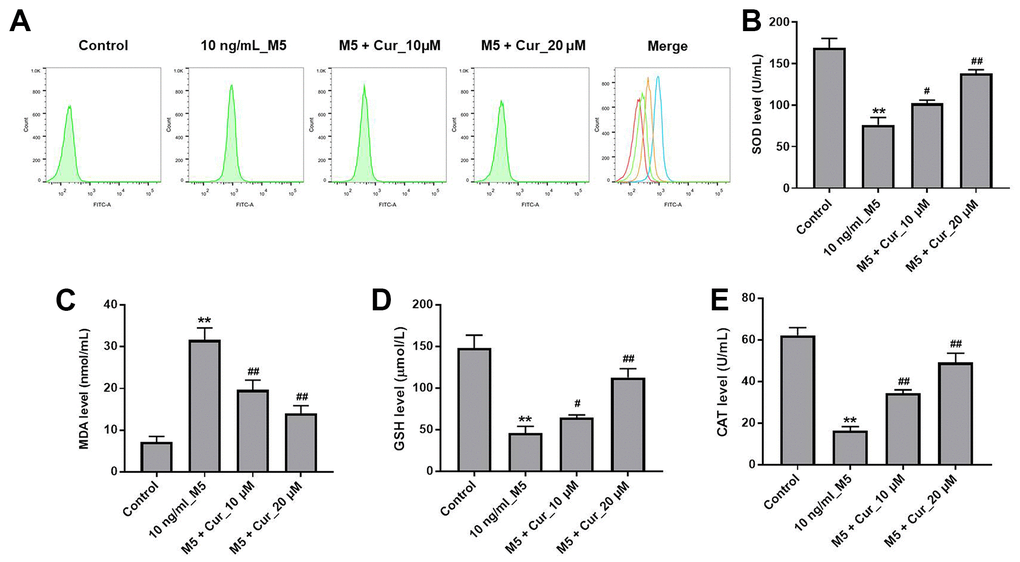
Figure 3. Curcumol attenuates M5-induced oxidative stress damage in NHEK cells. NHEK cells were treated with curcumol (10 or 20 μM) for 24 h, and then stimulated with M5 (10 ng/mL) for 24 h. (A) ROS production in NHEK cells was detected by flow cytometry. (B–E) ELISA was used to detect the levels of SOD (B), MDA (C), GSH (D), and CAT (E) in the supernatants of NHEK cells. **P<0.01 compared with control group; #P<0.05, ##P<0.01 compared with 10 ng/mL_M5 group.
Curcumol promotes differentiation of M5-stimulated NHEK cells
Psoriasis is characterized by the downregulation of keratinocyte differentiation markers [19]. Thus, we investigated whether curcumol could promote keratinocyte differentiation in M5-treated NHEK cells. As shown in Figure 4, curcumol reversed changes in protein levels of the differentiation markers keratin 1, keratin 5, keratin 10, filaggrin, and loricrin that were induced by M5 treatment in NHEK cells. These results indicate that curcumol promotes differentiation of M5-stimulated NHEK cells.
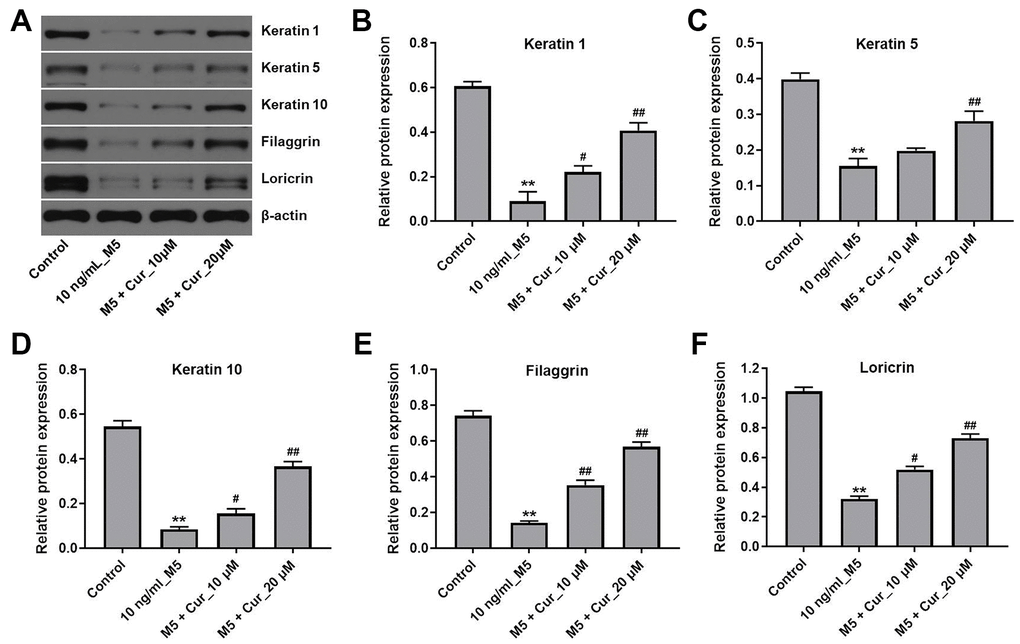
Figure 4. Curcumol promotes differentiation in M5-stimulated NHEK cells. NHEK cells were treated with curcumol (10 or 20 μM) for 24 h, and then stimulated with M5 (10 ng/mL) for 24 h. (A) Expression of keratin 1, keratin 5, keratin 10, filaggrin and loricrin in NHEK cells was analyzed by western blotting. (B–F) The relative expression of keratin 1 (B), keratin 5 (C), keratin 10 (D), filaggrin (E), and loricrin (F) quantified via normalization to β-actin. **P<0.01 compared with control group; #P<0.05, ##P<0.01 compared with 10 ng/mL_M5 group.
Curcumol inhibits proliferation of M5-stimulated NHEK cells via downregulation of p-STAT3/cyclin D2 axis
Since activation of STAT3 plays a critical role in psoriasis [20], we investigated the effect of curcumol on STAT3 signaling in M5-stimulated NHEK cells. As shown in Figure 5A, curcumol markedly decreased the level of p-STAT3 (phospho Y705) in M5-stimulated NHEK cells (Figure 5A). As the activated STAT3 translocates from cytoplasm to the nucleus, where it induces transcription of target genes, including cyclin D2 [21, 22], we also analyzed the levels of nuclear STAT3 after curcumol treatment. In addition, since Belsõ et al. found that the expression of cyclin D2 mRNA in psoriatic epidermis cells was significantly higher than that in normal epidermis cells [23]; thus, cyclin D2 was investigated as well. As shown in Figure 5B, curcumol reduced the nuclear STAT3 levels in M5-stimulated NHEK cells. Moreover, curcumol notably downregulated the mRNA and protein levels of cyclin D2 in M5-treated NHEK cells (Figure 5C, 5D), suggesting that curcumol inhibits proliferation of M5-treated NHEK cells via downregulation of the STAT3/cyclin D2 axis.
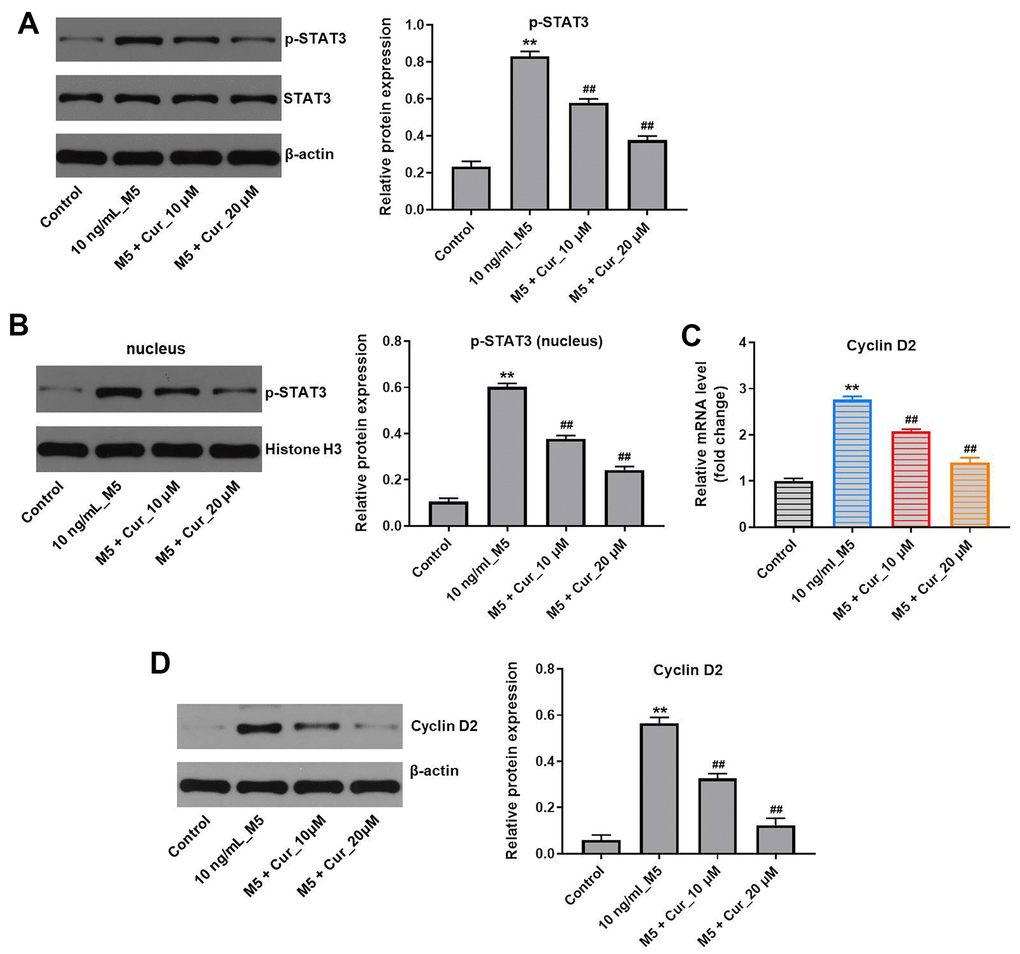
Figure 5. Curcumol inhibits proliferation of M5-treated NHEK cells via downregulation of p-STAT3/cyclin D2 axis. NHEK cells were treated with curcumol (10 or 20 μM) for 24 h, and then stimulated with M5 (10 ng/mL) for 24 h. (A) The expression level of p-STAT3 in NHEK cells was detected by western blotting, and normalized to STAT3. (B) The nuclear levels of p-STAT3 were analyzed by western blotting, and normalized to Histone H3. (C) Cyclin D2 mRNA levels were analyzed by RT-qPCR. (D) Cyclin D2 protein levels were analyzed by western blotting, and normalized to β-actin. **P<0.01 compared with control group; ##P<0.01 compared with 10 ng/mL_M5 group.
Curcumol inhibits proliferation of M5-stimulated NHEK cells via downregulation of JAK1/STAT3 signaling pathway
Since STAT3 activation is mediated by the Janus kinases (JAKs) [24], we investigated whether curcumol affects the JAK activity in M5-stimulated NHEK cells. As shown in Figure 6A, curcumol markedly decreased the phosphorylation level of JAK1 in M5-treated NHEK cells. In addition, a preincubation with the JAK1/2 inhibitor ruxolitinib reduced the phosphorylation levels of STAT3 in M5-stimulated NHEK cells, and this effect was further enhanced by curcumol (Figure 6B). Together, these data indicate that curcumol inhibits proliferation of stimulated human keratinocytes by suppressing the JAK1/STAT3 signaling pathway.
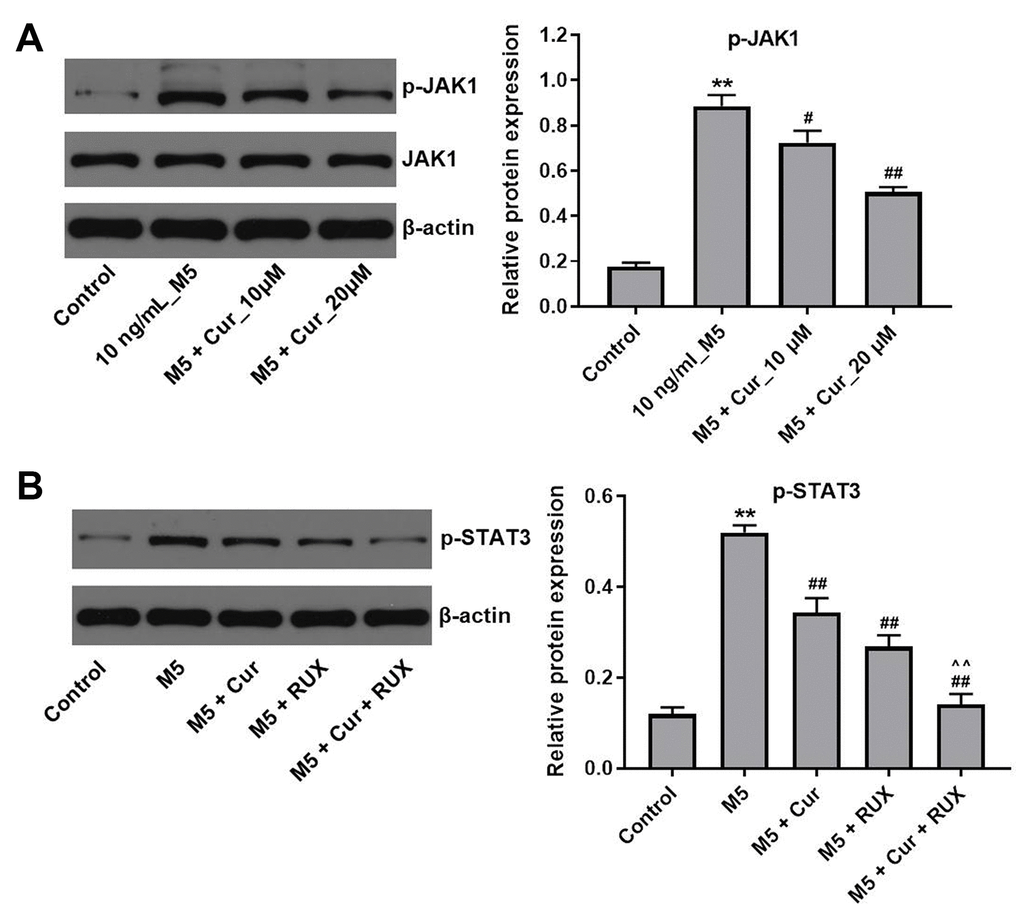
Figure 6. Curcumol inhibits proliferation of M5-treated NHEK cells via downregulation of JAK1/STAT3 signaling. (A) NHEK cells were treated with curcumol (10 or 20 μM; 24 h), and stimulated with M5 (10 ng/mL; 24 h). Levels of p-JAK1 were analyzed by western blotting, and normalized to JAK1. (B) NHEK cells were treated with 20 μM curcumol or/and 3μM ruxolitinib (RUX) for 24 h, and then stimulated with M5 (10 ng/mL) for 24 h. Levels of p-STAT3 were analyzed by western blotting, and normalized to STAT3. **P<0.01 compared with control group; #P<0.05, ##P<0.01 compared with M5 group; ^^P<0.01 compared with M5 + RUX group.
Discussion
The pathogenesis of psoriasis is extremely complex and involves numerous immune and inflammatory mediators, including IL-1α, TNF-α, IL-17A, and IL-22 [18, 25]. Thus, these cytokines are often used as in vitro inducers to stimulate keratinocyte hyperproliferation and inflammation [18]. In this study, a combination of IL-1α, IL-17A, IL-22, oncostatin M, and TNF-α (mix M5) was used to trigger psoriasis-like changes in NHEK cells in vitro. Our data demonstrated that M5 markedly promoted proliferation and cell cycle progression in NHEK cells. Importantly, curcumol significantly inhibited the M5-induced proliferation and cell cycle progression of NHEK cells. Sun et al. found that berberine inhibited proliferation of IL-22-stimulated keratinocytes [26]. This is consistent with our data that suggest that curcumol may alleviate psoriasis-like inflammation in keratinocytes by inhibiting their proliferation.
Previous studies have shown that M5 activates keratinocytes, leading to the release of cytokines, chemokines and antimicrobial peptides [27]. Our data demonstrated that M5 notably upregulated mRNA levels of cytokines (IL-6, IL-1β), chemokines (CXCL1, CXCL2), and antimicrobial peptides (LL37, β-defensin-2, S100A7 and S100A8) in NHEK cells. However, curcumol remarkably attenuated the expression of these inflammatory mediators in M5-treated NHEK cells. Liu et al. found that cimifugin inhibited TNFα-induced inflammatory response in NHEK cells [28]. In addition, betulinic acid attenuated psoriatic skin inflammation in a murine model of imiquimod-induced psoriasis via downregulation of pro-inflammatory mediators IL-6 and TNFα [29]. These findings indicate that curcumol might have a protective effect against M5-induced inflammatory injury in keratinocytes.
Previous studies have indicated an important role of STAT3 in the development and pathogenesis of psoriasis [30, 31]. IL-6 and IL-22 can induce STAT3 activation, leading to keratinocyte hyperproliferation and excessive inflammatory response [32, 33]. In addition, activation of STAT3 induces expression of cyclin D2, thereby promoting cell cycle progression [34]. Here, we found that curcumol reduced STAT3 phosphorylation and cyclin D2 expression in M5-stimulated NHEK cells, indicating that curcumol might block the STAT3-cyclin D2 signaling in keratinocytes. Curcumol was shown to suppress the activation of STAT3 in fibroblast-like synoviocytes in patients with rheumatoid arthritis [35]. However, the mechanisms remain largely unknown. Since JAKs are the upstream activators of STATs [36], inhibition of JAKs may block the activation of STAT3 in M5-stimulated NHEK cells. In this study, we found that curcumol decreased the phosphorylation level of JAK1 in M5-treated NHEK cells. In addition, curcumol enhanced the inhibitory effect of JAK1/2 inhibitor ruxolitinib on STAT3 activity, indicating that curcumol inhibits the STAT3 activation by inhibiting JAK1. Zuo et al. found that curcumol reduced the levels of phosphorylated STAT3 in hepatic cancer cells via JAK1 and JAK2 pathways [37]; this is in line with our results. Taken together, our results suggest that curcumol inhibits proliferation and inflammatory response in M5-stimulated NHEK cells via inhibition of the JAK1/STAT3/cyclin D2 pathway.
Obliviously, the present study contains certain limitations. For example, the detailed mechanism by which Curcumol regulates JAK1/STAT3 signaling pathway remains unclear; other signaling pathways associated with JAK1/STAT3 signaling pathway not been further explored. Therefore, additional studies are required in the future.
In summary, our results show that curcumol reduces proliferation and inflammatory response in keratinocytes stimulated with proinflammatory cytokines by inhibiting the JAK1/STAT3 pathway, suggesting that curcumol might provide a potential therapeutic option for the treatment of psoriasis.
Materials and Methods
Cell culture
Normal human epidermal keratinocytes (NHEK) cell line was purchased from ScienCell Research Laboratories (Carlsbad, CA, USA). Cells were cultured in Dulbecco's Modified Eagle Medium (DMEM, Thermo Fisher Scientific, Waltham, MA, USA) containing 10% fetal bovine serum (FBS, Thermo Fisher Scientific) and 100 U/ml penicillin and streptomycin (Thermo Fisher Scientific) at 37° C in an incubator with 5% CO2.
Cell counting kit-8 (CCK-8) assay
CCK-8 assay was performed to measure viability of NHEK cells. NHEK cells (5 x 103 cells/well) were seeded into a 96-well plate, and incubated overnight at 37° C. NHEK cells were pre-treated with 10 or 20 μM curcumol (MedChemExpress, Monmouth Junction, NJ, USA) for 24 h, and then exposed to 10 ng/mL mixture of five proinflammatory cytokines (M5; Sigma-Aldrich, St. Louis, MO, USA) for another 24 h. After that, 10 μL CCK-8 (Dojindo, Kumamoto, Japan) reagent was added into each well, and cells were incubated for another 2 h at 37° C. The absorbance of each well at 450 nm was measured using a microplate reader (Bio-Rad, Hercules, CA, USA).
5-Ethynyl-20-deoxyuridine (EdU) assay
EdU assay was performed using the EdU DNA Proliferation in vitro Detection kit (Ribobio Biology, Guangzhou, China). NHEK cells (4×105 cells/well) were placed into 24-well plate and incubated at 37° C overnight. Cells were then incubated with 50 μM EdU for 2 h at 37° C, stained with Apollo staining solution for 30 min at 37° C in dark, and observed under a fluorescence microscope.
Flow cytometry assay
Cells were collected, resuspended in PBS, and fixed overnight in 70% pre-cooled ethanol at 4° C. After that, fixed cells (1×106 cells/mL) were washed twice with PBS and stained with PI/RNase Staining Buffer (BD Biosciences, Franklin Lakes, NJ, USA) in the dark for 30 min at 37° C. The cell cycle distribution was analyzed using the FACScan™ flow cytometer (BD Biosciences).
Evaluation of cell apoptosis
NHEK cells were placed into 6-well plates as density of 3 × 105 cell/well. Then, Annexin V-FITC Apoptosis Detection Kit (Sungene Biotech, Tianjin, China) was used to detect the apoptosis of NHEK cells. NHEK cells were incubated with Annexin V-FITC (5 μl) and PI (5 μl) at 4° C for 15 min. Next, a BD AriaIII flow cytometry system (BD Biosciences, Franklin Lake, NJ, USA) was used to evaluate cell apoptosis.
Western blot assay
The BCA protein assay kit (Aspen Biotechnology, Wuhan, China) was used to determine protein concentration. Equal protein amounts (30 μg/lane) were separated by 10% SDS-PAGE, and transferred onto PVDF membranes (Millipore, Billerica, MA, USA). The membranes were blocked in 5% skimmed milk for 1 h at room temperature, and incubated at 4° C overnight with the following primary antibodies diluted 1:1000: anti-p27 (ab32034), anti-CDK4 (ab108357), anti-keratin 1 (ab185628), anti-keratin 5 (ab64081), anti-keratin 10 (ab237775), anti-filaggrin (sc-66192), anti-loricrin (ab137533), anti-p-STAT3 (phospho Y705) (ab76315), anti-cyclin D2 (ab207604), and anti-p-JAK1 (ab125051). Subsequently, the membranes were incubated with the corresponded secondary antibodies (1:5000) at room temperature for 1 h. Finally, the ECL kit (Thermo Fisher Scientific) was used to visualize the protein bands. Anti-filaggrin was provided by Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA). Other antibodies were purchased from Abcam (Cambridge, MA, USA).
Real-time quantitative PCR (RT-qPCR)
Total RNA was isolated from NHEK cells using Trizol reagent (ELK Biotechnology, Wuhan, China). EntiLink™ 1st Strand cDNA Synthesis Kit (ELK Biotechnology) was used for the synthesis of the first strand of cDNA. qPCR was performed on StepOne™ real-time PCR instrument (Life technologies) by using EnTurbo™ SYBR Green PCR SuperMix Kit (ELK Biotechnology). The following qPCR cycling conditions were used: 3 min 95° C, followed by 40 cycles of 10 s 95° C, 30 s 58° C, and 30 s 72° C. The primers are listed in Table 1. The 2–ΔΔCT method was used for data analysis.
Table 1. Primer sequences.
| Name | Primer sequences (5’-3’) | |
| IL-6 | Forward | ACTGGCAGAAAACAACCTGAAC |
| Reverse | TTGTACTCATCTGCACAGCTCTG | |
| IL-1β | Forward | ACGATGCACCTGTACGATCACT |
| Reverse | GAGAACACCACTTGTTGCTCCA | |
| CXCL1 | Forward | AACCGAAGTCATAGCCACACTC |
| Reverse | CTTCTCCTAAGCGATGCTCAAA | |
| CXCL2 | Forward | AAGGGGTTCGCCGTTCTC |
| Reverse | TGGCAGCGCAGTTCAGTG | |
| LL37 | Forward | CTCAGCCTCTGCGGAGAAAG |
| Reverse | CCACCTGAGCCCTATAAAAGATG | |
| β- defensin2 | Forward | ATGTCATCCAGTCTTTTGCCC |
| Reverse | TGCGTATCTTTGGACACCATAG | |
| S100A7 | Forward | GCACAAATTACCTCGCCGAT |
| Reverse | GACATTTTATTGTTCCTGGGGTC | |
| S100A8 | Forward | AAGTCCGTGGGCATCATGTT |
| Reverse | TCAGGTCATCCCTGTAGACGG | |
| CyclinD2 | Forward | GGATGAGGAAGTGAGCTCGC |
| Reverse | CTATTGAGGAGCACCGCCTC | |
| β-actin | Forward | GTCCACCGCAAATGCTTCTA |
| Reverse | TGCTGTCACCTTCACCGTTC |
ROS analysis
The Reactive Oxygen Species Assay Kit (Beyotime, Beijing, China) was used to detect the accumulation of intracellular ROS in NHEK cells. NHEK cells were stained with 2, 7-dichlorofluorescein diacetate staining solution at 37° C for 20 min. After that, the fluorescence intensity was analyzed by flow cytometry (BD Biosciences).
ELISA assay
Concentrations of SOD, MDA, GSH and CAT in NHEK cell supernatants were measured using ELISA kits (Nanjing Jiancheng Bioengineering Institute (Nanjing, China)) according to manufacturer's protocols. Superoxide Dismutase (SOD) assay kit (A001-3), Malondialdehyde (MDA) assay kit (A003-1), Reduced glutathione (GSH) assay kit (A006-2) and Catalase (CAT) assay kit (A007-1) were obtained from Nanjing Jiancheng Bioengineering Institute (Nanjing, China).
Statistical analysis
All statistical analyses were performed using the GraphPad Prism software (version 7.0, La Jolla, CA, USA). The comparison among multiple groups was analyzed with one-way analysis of variance (ANOVA) followed by Tukey’s test. All experiments were repeated three times. All data are shown as the mean ± SD; P<0.05 was considered as statistically significant.
Availability of data and materials
The datasets used and/or analyzed in this study are available from the corresponding author upon a reasonable request.
Author Contributions
ML made major contributions to the conception, design and manuscript drafting of this study. JS and FJ were responsible for data acquisition, data analysis, data interpretation and manuscript revision. JL made substantial contributions to conception and design of the study and revised the manuscript. All authors agreed to be accountable for all aspects of the work. All authors read and approved the final manuscript.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- 1. Schleicher SM. Psoriasis: Pathogenesis, Assessment, and Therapeutic Update. Clin Podiatr Med Surg. 2016; 33:355–66. https://doi.org/10.1016/j.cpm.2016.02.004 [PubMed]
- 2. Wang C, Zong J, Li Y, Wang X, Du W, Li L. MiR-744-3p regulates keratinocyte proliferation and differentiation via targeting KLLN in psoriasis. Exp Dermatol. 2019; 28:283–91. https://doi.org/10.1111/exd.13888 [PubMed]
- 3. Hoegler KM, John AM, Handler MZ, Schwartz RA. Generalized pustular psoriasis: a review and update on treatment. J Eur Acad Dermatol Venereol. 2018; 32:1645–51. https://doi.org/10.1111/jdv.14949 [PubMed]
- 4. Alwan W, Nestle FO. Pathogenesis and treatment of psoriasis: exploiting pathophysiological pathways for precision medicine. Clin Exp Rheumatol. 2015 (Suppl 93); 33:S2–6. [PubMed]
- 5. Boutet MA, Nerviani A, Gallo Afflitto G, Pitzalis C. Role of the IL-23/IL-17 Axis in Psoriasis and Psoriatic Arthritis: The Clinical Importance of Its Divergence in Skin and Joints. Int J Mol Sci. 2018; 19:530. https://doi.org/10.3390/ijms19020530 [PubMed]
- 6. Jung KJ, Kim TG, Lee JW, Lee M, Oh J, Lee SE, Chang HJ, Jee SH, Lee MG. Increased risk of atherosclerotic cardiovascular disease among patients with psoriasis in Korea: A 15-year nationwide population-based cohort study. J Dermatol. 2019; 46:859–66. https://doi.org/10.1111/1346-8138.15052 [PubMed]
- 7. Osmancevic A, Ståhle M. [Treatment of psoriasis: before and now]. Lakartidningen. 2017; 114:EU6Y. [PubMed]
- 8. Wei W, Rasul A, Sadiqa A, Sarfraz I, Hussain G, Nageen B, Liu X, Watanabe N, Selamoglu Z, Ali M, Li X, Li J. Curcumol: From Plant Roots to Cancer Roots. Int J Biol Sci. 2019; 15:1600–09. https://doi.org/10.7150/ijbs.34716 [PubMed]
- 9. Zang S, Tang Q, Dong F, Liu H, Li L, Guo F, Pan X, Lin H, Zeng W, Cai Z, Zhong Q, Zang N, Zang L. Curcumol inhibits the proliferation of gastric adenocarcinoma MGC-803 cells via downregulation of IDH1. Oncol Rep. 2017; 38:3583–91. https://doi.org/10.3892/or.2017.6028 [PubMed]
- 10. Li N, Liu TH, Yu JZ, Li CX, Liu Y, Wu YY, Yang ZS, Yuan JL. Curcumin and Curcumol Inhibit NF-κB and TGF- β1/Smads Signaling Pathways in CSE-Treated RAW246.7 Cells. Evid Based Complement Alternat Med. 2019; 2019:3035125. https://doi.org/10.1155/2019/3035125 [PubMed]
- 11. Chen X, Zong C, Gao Y, Cai R, Fang L, Lu J, Liu F, Qi Y. Curcumol exhibits anti-inflammatory properties by interfering with the JNK-mediated AP-1 pathway in lipopolysaccharide-activated RAW264.7 cells. Eur J Pharmacol. 2014; 723:339–45. https://doi.org/10.1016/j.ejphar.2013.11.007 [PubMed]
- 12. Morris R, Kershaw NJ, Babon JJ. The molecular details of cytokine signaling via the JAK/STAT pathway. Protein Sci. 2018; 27:1984–2009. https://doi.org/10.1002/pro.3519 [PubMed]
- 13. Zhu XY, Guo QY, Zhu M, Chen BG, Wang LY, Zhang DQ, Zhang L, Shao YP, Luo WD. HLX affects cell cycle and proliferation in AML cells via the JAK/STAT signaling pathway. Oncol Lett. 2020; 20:1888–96. https://doi.org/10.3892/ol.2020.11718 [PubMed]
- 14. Xiao C, Hong H, Yu H, Yuan J, Guo C, Cao H, Li W. MiR-340 affects gastric cancer cell proliferation, cycle, and apoptosis through regulating SOCS3/JAK-STAT signaling pathway. Immunopharmacol Immunotoxicol. 2018; 40:278–83. https://doi.org/10.1080/08923973.2018.1455208 [PubMed]
- 15. Chen SF, Zhang ZY, Zhang JL. Matrine increases the inhibitory effects of afatinib on H1975 cells via the IL-6/JAK1/STAT3 signaling pathway. Mol Med Rep. 2017; 16:2733–39. https://doi.org/10.3892/mmr.2017.6865 [PubMed]
- 16. Gao J, Chen F, Fang H, Mi J, Qi Q, Yang M. Daphnetin inhibits proliferation and inflammatory response in human HaCaT keratinocytes and ameliorates imiquimod-induced psoriasis-like skin lesion in mice. Biol Res. 2020; 53:48. https://doi.org/10.1186/s40659-020-00316-0 [PubMed]
- 17. Nguyen LT, Ahn SH, Nguyen UT, Yang IJ. Dang-Gui-Liu-Huang Tang a traditional herbal formula, ameliorates imiquimod-induced psoriasis-like skin inflammation in mice by inhibiting IL-22 production. Phytomedicine. 2018; 47:48–57. https://doi.org/10.1016/j.phymed.2018.04.051 [PubMed]
- 18. Rabeony H, Petit-Paris I, Garnier J, Barrault C, Pedretti N, Guilloteau K, Jegou JF, Guillet G, Huguier V, Lecron JC, Bernard FX, Morel F. Inhibition of keratinocyte differentiation by the synergistic effect of IL-17A, IL-22, IL-1α, TNFα and oncostatin M. PLoS One. 2014; 9:e101937. https://doi.org/10.1371/journal.pone.0101937 [PubMed]
- 19. Wang W, Yu X, Wu C, Jin H. IL-36γ inhibits differentiation and induces inflammation of keratinocyte via Wnt signaling pathway in psoriasis. Int J Med Sci. 2017; 14:1002–07. https://doi.org/10.7150/ijms.20809 [PubMed]
- 20. Nakajima K, Kataoka S, Sato K, Takaishi M, Yamamoto M, Nakajima H, Sano S. Stat3 activation in epidermal keratinocytes induces Langerhans cell activation to form an essential circuit for psoriasis via IL-23 production. J Dermatol Sci. 2019; 93:82–91. https://doi.org/10.1016/j.jdermsci.2018.11.007 [PubMed]
- 21. Xiong A, Yang Z, Shen Y, Zhou J, Shen Q. Transcription Factor STAT3 as a Novel Molecular Target for Cancer Prevention. Cancers (Basel). 2014; 6:926–57. https://doi.org/10.3390/cancers6020926 [PubMed]
- 22. Park SY, Lee CJ, Choi JH, Kim JH, Kim JW, Kim JY, Nam JS. The JAK2/STAT3/CCND2 Axis promotes colorectal Cancer stem cell persistence and radioresistance. J Exp Clin Cancer Res. 2019; 38:399. https://doi.org/10.1186/s13046-019-1405-7 [PubMed]
- 23. Belsõ N, Széll M, Pivarcsi A, Kis K, Kormos B, Kenderessy AS, Dobozy A, Kemény L, Bata-Csörgõ Z. Differential expression of D-type cyclins in HaCaT keratinocytes and in psoriasis. J Invest Dermatol. 2008; 128:634–42. https://doi.org/10.1038/sj.jid.5701059 [PubMed]
- 24. Parks OB, Pociask DA, Hodzic Z, Kolls JK, Good M. Interleukin-22 Signaling in the Regulation of Intestinal Health and Disease. Front Cell Dev Biol. 2016; 3:85. https://doi.org/10.3389/fcell.2015.00085 [PubMed]
- 25. Cheuk S, Wikén M, Blomqvist L, Nylén S, Talme T, Ståhle M, Eidsmo L. Epidermal Th22 and Tc17 cells form a localized disease memory in clinically healed psoriasis. J Immunol. 2014; 192:3111–20. https://doi.org/10.4049/jimmunol.1302313 [PubMed]
- 26. Sun S, Zhang X, Xu M, Zhang F, Tian F, Cui J, Xia Y, Liang C, Zhou S, Wei H, Zhao H, Wu G, Xu B, et al. Berberine downregulates CDC6 and inhibits proliferation via targeting JAK-STAT3 signaling in keratinocytes. Cell Death Dis. 2019; 10:274. https://doi.org/10.1038/s41419-019-1510-8 [PubMed]
- 27. Guilloteau K, Paris I, Pedretti N, Boniface K, Juchaux F, Huguier V, Guillet G, Bernard FX, Lecron JC, Morel F. Skin Inflammation Induced by the Synergistic Action of IL-17A, IL-22, Oncostatin M, IL-1{alpha}, and TNF-{alpha} Recapitulates Some Features of Psoriasis. J Immunol. 2010; 184:5263–70. https://doi.org/10.4049/jimmunol.0902464 [PubMed]
- 28. Liu A, Zhao W, Zhang B, Tu Y, Wang Q, Li J. Cimifugin ameliorates imiquimod-induced psoriasis by inhibiting oxidative stress and inflammation via NF-κB/MAPK pathway. Biosci Rep. 2020; 40:BSR20200471. https://doi.org/10.1042/BSR20200471 [PubMed]
- 29. Liu C, Chen Y, Lu C, Chen H, Deng J, Yan Y, Xu YY, Liu H, Huang H, Wei J, Han L, Dai Z. Betulinic acid suppresses Th17 response and ameliorates psoriasis-like murine skin inflammation. Int Immunopharmacol. 2019; 73:343–52. https://doi.org/10.1016/j.intimp.2019.05.030 [PubMed]
- 30. Calautti E, Avalle L, Poli V. Psoriasis: A STAT3-Centric View. Int J Mol Sci. 2018; 19:171. https://doi.org/10.3390/ijms19010171 [PubMed]
- 31. Wang A, Wei J, Lu C, Chen H, Zhong X, Lu Y, Li L, Huang H, Dai Z, Han L. Genistein suppresses psoriasis-related inflammation through a STAT3-NF-κB-dependent mechanism in keratinocytes. Int Immunopharmacol. 2019; 69:270–78. https://doi.org/10.1016/j.intimp.2019.01.054 [PubMed]
- 32. Miao X, Xiang Y, Mao W, Chen Y, Li Q, Fan B. TRIM27 promotes IL-6-induced proliferation and inflammation factor production by activating STAT3 signaling in HaCaT cells. Am J Physiol Cell Physiol. 2020; 318:C272–81. https://doi.org/10.1152/ajpcell.00314.2019 [PubMed]
- 33. Kwon TR, Lee SE, Kim JH, Na Jang Y, Kim SY, Mun SK, Kim CW, Na J, Kim BJ. 310 nm UV-LEDs attenuate imiquimod-induced psoriasis-like skin lesions in C57BL/6 mice and inhibit IL-22-induced STAT3 expression in HaCaT cells. Photochem Photobiol Sci. 2020; 19:1009–21. https://doi.org/10.1039/c9pp00444k [PubMed]
- 34. Sun J, Du Y, Song Q, Nan J, Guan P, Guo J, Wang X, Yang J, Zhao C. E2F is required for STAT3-mediated upregulation of cyclin B1 and Cdc2 expressions and contributes to G2-M phase transition. Acta Biochim Biophys Sin (Shanghai). 2019; 51:313–22. https://doi.org/10.1093/abbs/gmy174 [PubMed]
- 35. Wang H, Fang Y, Wang Y, Wang Z, Zou Q, Shi Y, Chen J, Peng D. Inhibitory Effect of Curcumol on Jak2-STAT Signal Pathway Molecules of Fibroblast-Like Synoviocytes in Patients with Rheumatoid Arthritis. Evid Based Complement Alternat Med. 2012; 2012:746426. https://doi.org/10.1155/2012/746426 [PubMed]
- 36. Nabavi SM, Ahmed T, Nawaz M, Devi KP, Balan DJ, Pittalà V, Argüelles-Castilla S, Testai L, Khan H, Sureda A, de Oliveira MR, Vacca RA, Xu S, et al. Targeting STATs in neuroinflammation: The road less traveled!. Pharmacol Res. 2019; 141:73–84. https://doi.org/10.1016/j.phrs.2018.12.004 [PubMed]
- 37. Zuo HX, Jin Y, Wang Z, Li MY, Zhang ZH, Wang JY, Xing Y, Ri MH, Jin CH, Xu GH, Piao LX, Ma J, Jin X. Curcumol inhibits the expression of programmed cell death-ligand 1 through crosstalk between hypoxia-inducible factor-1α and STAT3 (T705) signaling pathways in hepatic cancer. J Ethnopharmacol. 2020; 257:112835. https://doi.org/10.1016/j.jep.2020.112835 [PubMed]



