Introduction
Lipids account for nearly 60% of human brain tissue and the composition of fatty acids (FAs) seems to impact on brain integrity and function [1]. FAs are the elementary building blocks of triglycerides, phospholipids and cholesteryl esters. They function as energy sources and are important structural components of all cells. Polyunsaturated FAs (PUFAs) appear to play a key role in age-related cognitive decline and brain atrophy [2–4]. ω-3 and ω-6 PUFA include eicosapentaenoic acid (EPA, 20:5 ω-3), docosahexaenoic acid (DHA, 22:6 ω-3), linoleic acid (LA, 18:2 ω-6), γ-linolenic acid (GLA, 18:3 ω-6) and arachidonic acid (AA, 20:4 ω-6), respectively. While humans can synthesize saturated and monounsaturated FAs (MUFAs) they are not able to synthesize PUFA, such as GLA and LA or DHA and EPA, due to the absence of the conversion enzyme ω-3-desaturase [5].
In the brain, DHA is the most abundant PUFA species [6] accounting for more than 90% of all ω-3 PUFAs and for approximately 10–20% of all lipid species [7]. It is mainly incorporated in phosphatidylethanolamine, phosphatidylserine and in smaller amounts in phosphatidylcholine [8] at synaptic terminals, mitochondria and endoplasmic reticula. DHA is able to modulate cellular features and physiological processes, such as membrane fluidity, release of neurotransmitters, gene expression, myelination, neuroinflammation and neuronal growth [9]. The brain´s supply with PUFAs relies on their transfer across the blood brain barrier (BBB) by passive diffusion and specific transport mechanisms [10]. In blood, PUFAs circulate in their unbound form, bound to albumin [10] or esterified in triacylglycerols, phospholipids, cholesteryl esters or phosphatidylcholine [11]. Unbound free FAs (u-FFAs) are well correlated with total free FAs (t-FFAs), which include all non-esterified FAs. t-FFAs represent the FA fraction that can cross the BBB by passive diffusion and is readily available for metabolism [12]. This non-saturable process is regulated by plasma albumin so that plasma t-FFAs equilibrate to about 90% of maximal brain uptake within 15 min [13], and the higher the degree of unsaturation, the more rapid is the uptake [11].
The cerebral FA composition has been reported to influence cognitive function and structural damage of the aging brain [14]. In particular, FA saturation appears to be a critical determinant of AD risk [15, 16]. Cerebral FFAs have been related to an accelerated cognitive decline of dementia patients [2–4]. In-vitro experiments have demonstrated that FAs stimulate the assembly of amyloid β (Aβ) and tau filaments [17]. The proportion between ω-3 and ω-6 FA is thought to play a crucial pathogenic role in this context [18]. In line with this assumption, a diet rich in ω-3 FA, and/or low in ω-6 FA was found to lower the incidence of AD and other degenerative brain disorders [3, 19]. Other studies have measured plasma FAs in an attempt to estimate the risk of cognitive impairment and dementia that is attributable to imbalances in ω-3 and ω-6 FA composition [20–24]. The concept that ω-3 FAs protect against cognitive dysfunction and neurodegeneration, whereas ω-6 FA have detrimental effects, is supported by some investigations [2, 22–29] while others opposed this view [30–33]. Yagi et al. showed a positive correlation between EPA and the EPA/AA-ratio with performance on the Minimal Mental State Examination (MMSE) in Japanese coronary artery disease patients [24]. High plasma EPA concentrations have also been associated with better performance on specific cognitive functions, such as visual working memory [28], and were found to decrease the risk of dementia in general [25, 27]. The opposite was described for high plasma ω-6 PUFA concentrations [26] and unfavorable ratios between AA/DHA as well as ω-6/ω-3 PUFAs, which related to increased risk of dementia [25, 27, 29] and the progression of white matter lesions [23, 29]. These reports are contrasted by studies that failed to link ω-3 PUFA concentrations with cognitive decline, dementia or AD [30–32]. Moreover, a Cochrane Collaboration meta-analysis showed no benefit of ω-3 PUFA supplementation on cognitive function in cognitively healthy older people [33].
Existing studies are mainly limited by the measurement of total plasma FAs rather than the metabolically active t-FFAs. However, plasma t-FFA profiles may provide additional information about lipid metabolism and disease risk [34]. We here extend previous work by studying the association between plasma t-FFAs, cognitive function and brain structure in a well-characterized community-dwelling cohort of elderly individuals without stroke and dementia. The cognitive test battery was extensive, demanding and assessing different cognitive domains, and brain MRI was evaluated quantitatively with measurements of structural and microstructural brain alterations.
Materials and Methods
Study design
Stored EDTA plasma samples from 391 participants of the Austrian Stroke Prevention Study (ASPS) were used for t-FFA measurement as described below. Cognitive function was assessed with a comprehensive battery of neuropsychiatric tests capturing memory, executive function and visuopractical skills. Furthermore, structural neurodegenerative and vascular changes were analyzed in 167 individuals who underwent brain MRI on the day of cognitive testing.
Results were used to explore associations between FFAs, cognitive function and structural markers of neurodegeneration. Statistical analyses included only results from participants with a complete set of biochemical and cognitive test results who did not supplement fatty acids. Participants with a family history of dementia were excluded from the statistical analyses (n = 23). Written informed consent was obtained from all participants and the study was approved by the ethics committee of the Medical University of Graz, Austria.
Participants
The study population consisted of community-dwelling participants from the Austrian Stroke Prevention Study (ASPS) and the Austrian Stroke Prevention Family Study (ASPS-Fam). All participants had an unremarkable neurological status and no history of stroke and dementia. ASPS is a prospective single-center study examining the effects of vascular risk factors on brain structure and function [35, 36]. Randomly selected individuals from the community register of the city of Graz, Austria, were enrolled. ASPS-Fam is an extension of ASPS with a similar study protocol consisting of participants of ASPS and their first-grade relatives [37, 38].
Common risk factors assessed in all participants included arterial hypertension, diabetes mellitus, atrial fibrillation, hypercholesterolemia and hypertriglyceridemia. Hypertension was defined as history of hypertension or systolic blood pressure over 140 mmHg or a diastolic blood pressure over 90 mmHg [39] and current use of antihypertensive agents. Subjects were classified as diabetic on the basis of a documented history of diabetes, use of anti-diabetics or a fasting blood glucose level above 126 mg/dl (7.0 mmol/L) at the time of examination [40]. The presence of atrial fibrillation was confirmed by an electrocardiogram obtained during the study visit. Presence of hypercholesterolemia was confirmed if a participant had a history of hypercholesterolemia, was treated for hypercholesterolemia at the time of examination or if the total or LDL cholesterol was higher than 200 mg/dl or 130 mg/dl respectively. Hypertriglyceridemia was defined as history of hypertriglyceridemia or treatment for hypertriglyceridemia or triglyceride levels of at least 200 mg/dl.
Measurement of free fatty acids
Ten different FFAs were analyzed by a modified gas chromatography mass spectrometry (GC-MS) method with liquid-liquid sample extraction [41, 42]. Briefly, 50 μL of plasma were mixed with 600 μL of deionized water, 500 μL of 0.5 N methanolic hydrochloric acid and 4 mL Methyl tert-butyl ether (MTBE) in a 10 mL silanized glass tube. Furthermore, internal standard solutions (IS) containing 1 nmol of C18d35 and C21:0 were added. This mixture was vigorously vortexed for 30 s and subsequently centrifuged for 2 min at 1500 g. The upper organic layer was carefully transferred in a 10 mL silanized glass tube. The remaining aqueous layer was extracted again with 4 mL MTBE. After centrifugation, the MTBE phase of the second extraction was added to the first one. The combined extracts in the glass tube were dried under a gentle stream of nitrogen. In the next step, samples were derivatized using a modified protocol from Quehenberger et al. (2011). The dried extracts were resuspended and derivatized in 25 μL of 1% diisopropylethylamine in acetonitrile (DIPEA) and 25 μL of 10% pentafluorobenzyl bromide in acetonitrile (PFBBr) followed by 20 min of incubation at room temperature. Then, solvents were removed by evaporation under a gentle stream of nitrogen. The residues were again resuspended in 100 μL of pure methanol, vigorously vortexed for 10 s and then dried under a gentle stream of nitrogen. In the last step, the dried samples were dissolved in 200 μL of hexane, vigorously vortexed for 10 s and carefully transferred to an autosampler glass vial and placed into the autosampler of the GC-MS instrument.
For analysis a Trace GC Ultra-DSQ II GC-MS (Thermo Scientific) equipped with a 30 m TR-FAME column (30 m, 0,25 mm, 0,25 μm, Thermo Scientific) was used. From the purified samples, 1 μl was injected into the instrument in split/splitless mode and immediately evaporated in the injector, which was set at a temperature of 250°C. Methane was used as carrier gas at a flow rate of 2 ml/min. Subsequently, the evaporated samples were transferred from the injector onto the analytical column, which was kept at 150°C in the oven. After 1 min of equilibration, temperature was raised at a rate of 20°C/min to 200°C, which was kept for 2 min. Subsequently, the temperature was further increased with a rate of 30°C/min to a final temperature of 300°C and kept for 5 min for column bake out. The eluate from the column was sent through the transfer line, set at 310°C, into the source, where it was ionized by negative chemical ionization (NCI) with methane at a source temperature of 250°C. Lastly, ions were introduced into a single quadrupole mass spectrometer where FFAs were detected in single ion monitoring mode (SIM). The SIM masses and the retention times for all analytes including the IS are listed in Table 1.
Table 1. SIM masses and retention time of the measured fatty acids.
| Fatty acid | Mass (m/z) | SIM mass (m-181) (m/z) | Internal Standard | Retention time (min) |
| Palmitoleic acid (C16:0) | 256.2 | 255.2 | C18d35 | 6.16 |
| Palmitolenic acid (C16:1) | 254.2 | 253.2 | C18d35 | 6.36 |
| Stearic acid (C18:0) | 284.3 | 283.3 | C18d35 | 6.93 |
| Oleic acid (C18:1) | 282.3 | 281.3 | C18d35 | 7.05 |
| Linoleic acid (C18:2) | 280.2 | 279.2 | C18d35 | 7.26 |
| Gamma linoleic acid (C18:3) | 278.2 | 277.2 | C18d35 | 7.37 |
| Arachinoic acid (C20:0) | 312.3 | 311.3 | C21:0 | 7.51 |
| Arachidonic acid (C20:4) | 304.2 | 303.2 | C21:0 | 7.92 |
| Eicosapentaenoic acid (C20:5) | 302.2 | 301.2 | C21:0 | 8.11 |
| Docosahexaenoic acid (C22:6) | 328.2 | 327.2 | C21:0 | 8.56 |
| IS deuterated Stearic acid (C18d35) | 319.7 | 318.7 | 6.78 | |
| IS Heneicosylic acid (C21:0) | 326.3 | 325.3 | 7.76 |
Our method has been validated in accordance with FDA guidelines [43]. The measured concentrations of all FAs in our samples fell inside the validated analytical ranges for these compounds. Intra- and interassay imprecision were <15% for all compounds and across the entire analytical range.
Neuropsychological testing
Cognitive function was assessed by a comprehensive battery of neuropsychological tests capturing memory, executive function and visuopractical skills. A detailed description of these tests has been published previously [35, 44–49]. Individual tests were summarized in composite measures of the cognitive domains memory, executive function and visuopractical skills. These summary measures were calculated by converting test results to z-scores based on the mean and standard deviation of the combined ASPS and ASPS-Fam sample, and by computing the average z-scores within each cognitive domain.
Magnetic resonance imaging (MRI)
Study participants underwent MRI on a 3T whole-body MR system (TimTrio; Siemens Healthcare, Erlangen, Germany). The MRI subgroup included only those individuals in whom 3D T1 and FLAIR sequences were available.
Total, cortical and subcortical gray matter volume, hippocampal volume and lobar cortical volume, were computed from the T1 weighted MPRAGE images using FreeSurfer 5.3 [50, 51]. Based on the intensity of the voxels in the MRI image, the software automatically segments the brain into subcortical gray volumetric structures and cortical gray matter. Freesurfer divides the cerebral cortex into gyral based regions of interest and provides the cortical volume for each of these regions. Values of these regions were added up or averaged for volume, to obtain lobar measures. To correct for variations in individual head size, all measures were normalized for total intracranial volume.
Vascular lesions including white matter hyperintensities (WMH), silent non-lacunar and lacunar infarcts were assessed on FLAIR images by a blinded expert. WMH were outlined using a custom written IDL program (Exelis Visual Information Solutions, USA). Lesion areas were segmented by combined region growing and local thresholding following manual selection, as described earlier [52]. The total lesion volume (cubic millimeter) was calculated using the program FSLMATHS by multiplying the lesion area with the slice thickness and was normalized for head size. Due to a skewed distribution, the lesion load was logarithmically transformed. Lacunes were defined as focal lesions involving the basal ganglia, internal capsule, thalamus, brainstem, or the white matter, not exceeding a maximum diameter of 20 mm. We considered lesions with typical signal characteristics of infarcts following a typical vascular territory or located in a border zone between two vascular territories as non-lacunar infarcts.
To assess microstructural changes, we used the peak width of the skeletonized mean diffusivity (PSMD). This represents a new, robust, fully automated and easy-to-implement marker for cerebral small vessel disease based on diffusion tensor imaging, white matter tract skeletonization (as implemented in FSL-TBSS) and histogram analysis. This software package allows calculating PSMD from diffusion tensor imaging data
Statistical analysis
Statistical analysis was performed with the R statistical software package version 3.6.1 [53]. We assessed normality of continuous variables by visual inspection and Shapiro-Wilk’s test. Normally distributed variables are reported as mean ± standard deviation (std) and non-normally distributed variables as median and interquartile range (IQR). We used linear mixed models to determine the association between FFAs and cognition as well as MRI. All analyses were adjusted for age, sex, hypertension, diabetes, atrial fibrillation, hypercholesterolemia and hypertriglyceridemia. Cognition analyses were additionally adjusted for years of education, and, as we pooled ASPS and ASPS-Fam data, for cohort to adjust for any undetected differences between the two studies. ASPS-Fam is a family study and therefore the family structure was included in our models as a random effect using the lmekin function of the R package coxme [54]. The degree of relationship between any two individuals in the study is represented by a kinship matrix, which was generated using the R package kinship2 [55]. The results of linear mixed model analyses are presented as regression coefficient (β), standard error of the regression coefficient (SE) and p-values (p). For all p-values within Tables 2–4 we applied false discovery rate (FDR) correction [56] to compensate for the number of tests in the Table.
Table 2. Associations between free ω3 and ω6 polyunsaturated fatty acids (PUFA) and cognitive function.
| Area of cognitive function | ω3-PUFA | ω6-PUFA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N | β | se | p | pFDR | N | β | se | p | pFDR | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Executive function | 368 | –0.0094 | 0.0053 | 0.076 | 0.196 | 368 | –0.0016 | 0.0006 | 0.007 | 0.042 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Visuopractical skills | 368 | –0.0071 | 0.0083 | 0.395 | 0.593 | 368 | –0.0016 | 0.0009 | 0.098 | 0.196 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Memory | 368 | 0.0012 | 0.0086 | 0.891 | 0.891 | 368 | 0.0003 | 0.0010 | 0.753 | 0.891 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: N: sample size, beta: regression coefficient, se: standard error, p: p-value, pFDR: false discovery rate corrected p-value. Linear mixed model adjusted for age, sex, hypertension, diabetes, arterial fibrillation, hypercholesterolemia, hypertriglyceridemia and education. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 3. Significant associations between individual free plasma ω6 polyunsaturated fatty acids and executive function.
| Area of cognitive function | Executive function | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N | β | se | p | pFDR | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Linoleic Acid | 368 | –0.002 | 0.0007 | 0.01 | 0.02 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Arachidonic Acid | 368 | –0.008 | 0.0036 | 0.03 | 0.03 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: N: sample size, beta: regression coefficient, se: standard error, p: p-value, pFDR: false discovery rate corrected p-value. Linear mixed model adjusted for age, sex, hypertension, diabetes, arterial fibrillation, hypercholesterolemia and hypertriglyceridemia. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 4. Associations between ω3 and ω6 polyunsaturated fatty acids (PUFA) and MRI-derived morphologic features of cognitive impairment and dementia.
| Area of cognitive function | ω3-PUFA | ω6-PUFA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N | β | se | p | pFDR | N | β | se | p | pFDR | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total gray volume | 146 | 3.51E-04 | 3.71E-04 | 0.344 | 0.917 | 146 | –7.93E-05 | 5.58E-05 | 0.155 | 0.620 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hippocampus volume | 146 | 1.68E-06 | 4.96E-06 | 0.735 | 0.924 | 146 | 1.25E-07 | 7.21E-07 | 0.862 | 0.924 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Frontal lobe volume | 146 | 1.31E-05 | 1.23E-04 | 0.915 | 0.924 | 146 | –4.61E-05 | 1.81E-05 | 0.011 | 0.136 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Temporal lobe volume | 146 | 1.59E-04 | 6.65E-05 | 0.017 | 0.136 | 146 | –3.98E-06 | 1.02E-05 | 0.696 | 0.924 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Parietal lobe volume | 146 | 5.23E-05 | 8.49E-05 | 0.538 | 0.924 | 146 | –1.94E-05 | 1.27E-05 | 0.127 | 0.620 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Occipital lobe volume | 146 | –3.94E-06 | 4.11E-05 | 0.924 | 0.924 | 146 | –3.33E-06 | 6.15E-06 | 0.588 | 0.924 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WMH volume | 146 | –3.44E-05 | 1.16E-04 | 0.766 | 0.924 | 146 | –1.17E-05 | 1.72E-05 | 0.497 | 0.924 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PSMD | 68 | 2.91E-03 | 2.88E-03 | 0.313 | 0.917 | 68 | 6.27E-05 | 3.99E-04 | 0.875 | 0.924 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: N: sample size, beta: regression coefficient, se: standard error, p: p-value, pFDR: false discovery rate corrected p-value; WMH: white matter hyperintensities; PSMD: peak width of skeletonized mean diffusivity. Linear mixed model adjusted for age, sex, hypertension, diabetes, arterial fibrillation, hypercholesterolemia and hypertriglyceridemia. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Results
Descriptive statistics
Demographics, frequency of risk factors and laboratory findings are displayed in Table 5. The cohort included more females than males. Participants had a median age of 68 years. Hypercholesterolemia was the most prevalent vascular risk factor in the ASPS cohort affecting 321 (82.1%) individuals. Statins and other lipid lowering drugs were used by 15% of all subjects.
Table 5. Demographics and risk factors.
| Parameter | Result |
| N | 391 |
| Females, N (%) | 250 (63.9%) |
| Age (years), median (IQR) | 68 (62–74) |
| Education (years), median (IQR) | 10 (9–13) |
| BMI, median (IQR) | 26.5 (24.1–29.1) |
| Hypertension, N (%) | 276 (70.6%) |
| Diabetes, N (%) | 46 (11.8%) |
| Atrial fibrillation, N (%) | 24 (6.1%) |
| Statins, N (%) | 53 (13.6%) |
| Other Lipid lowering medication | 6 (1.5%) |
The median total FFA concentration was 307.5 μmol/L with a relative even distribution of saturated (31%), monounsaturated (39%) and polyunsaturated (30%) FFA. Table 6 shows the plasma concentrations of all measured FFA species. The plasma concentrations of ω-6 PUFA species were substantially higher than those of ω-3 PUFA species with a median ω-6/ω-3 ratio of 11.06 (IQR: 8.01–16.06).
Table 6. Median (IQR) concentrations of total and individual free fatty acids.
| Groups of FFA | ||
| Total FFA | (μmol/L) | 307.52 (243.65–415.86) |
| Saturated FFA | (μmol/L) | 94.91 (72.87–133.89) |
| Monounsaturated FFA | (μmol/L) | 118.37 (91.53–167.13) |
| Polyunsaturated FFA | (μmol/L) | 93.42 (76.39–119.34) |
| Individual species of saturated FFA | ||
| Palmitic Acid (C16:0) | (μmol/L) | 59.26 (43.03–84.39) |
| (% of total FFA) | 19.81 (16.19–22.51) | |
| Stearic Acid (C18:0) | median (IQR) | 36.99 (27.62–49.02) |
| (% of total FFA) | 11.43 (9.75–13.33) | |
| Arachinic Acid (C20:0) | median (IQR) | 0.15 (0.09–0.23) |
| (% of total FFA) | 0.05 (0.03–0.07) | |
| Individual species of monounsaturated FFA | ||
| Palmitoleic Acid (C16:1) | median (IQR) | 20.27 (13.24–30.54) |
| (% of total FFA) | 6.35 (4.35–8.18) | |
| Oleic Acid (C18:1) | median (IQR) | 94.25 (74.87–142.26) |
| (% of total FFA) | 31.52 (28.93–34.53) | |
| Individual species of polyunsaturated FFA | ||
| Linoleic Acid (C18:2) | median (IQR) | 68.89 (55.23–93.11) |
| (% of total FFA) | 22.47 (20.88–24.45) | |
| γ-Linolenic Acid (C18:3) | median (IQR) | 0.84 (0.48–1.36) |
| (% of total FFA) | 0.28 (0.16–0.45) | |
| Arachidonic Acid (C20:4) | median (IQR) | 14.82 (11.38–19.42) |
| (% of total FFA) | 4.66 (3.1–6.14) | |
| Eicosapentaenoic Acid(C20:5) | median (IQR) | 1.66 (1.13–2.43) |
| (% of total FFA) | 0.52 (0.33–0.77) | |
| Docosahexaenoic Acid (C22:6) | median (IQR) | 5.98 (4.25–8.29) |
| (% of total FFA) | 1.93 (1.3–2.75) | |
| ω3 and ω6 polyunsaturated FFA species | ||
| ω3PUFA (C22:6 + C20:5) | median (IQR) | 7.82 (5.63–11.01) |
| ω6 PUFA (C18:2 + C20:4) | median (IQR) | 85.02 (70.14–109.53) |
| ω6/ω3 ratio | median (IQR) | 11.06 (8.01–16.06) |
Association between plasma free PUFAs and cognitive test results
The associations between plasma PUFA concentrations and cognitive function were explored by using a linear mixed model adjusted for possible confounders (Table 2, Figure 1). This model showed a significant inverse relationship between ω-6 PUFA and executive function, which remained significant after correction for the expected false discovery rate due to multiple testing.
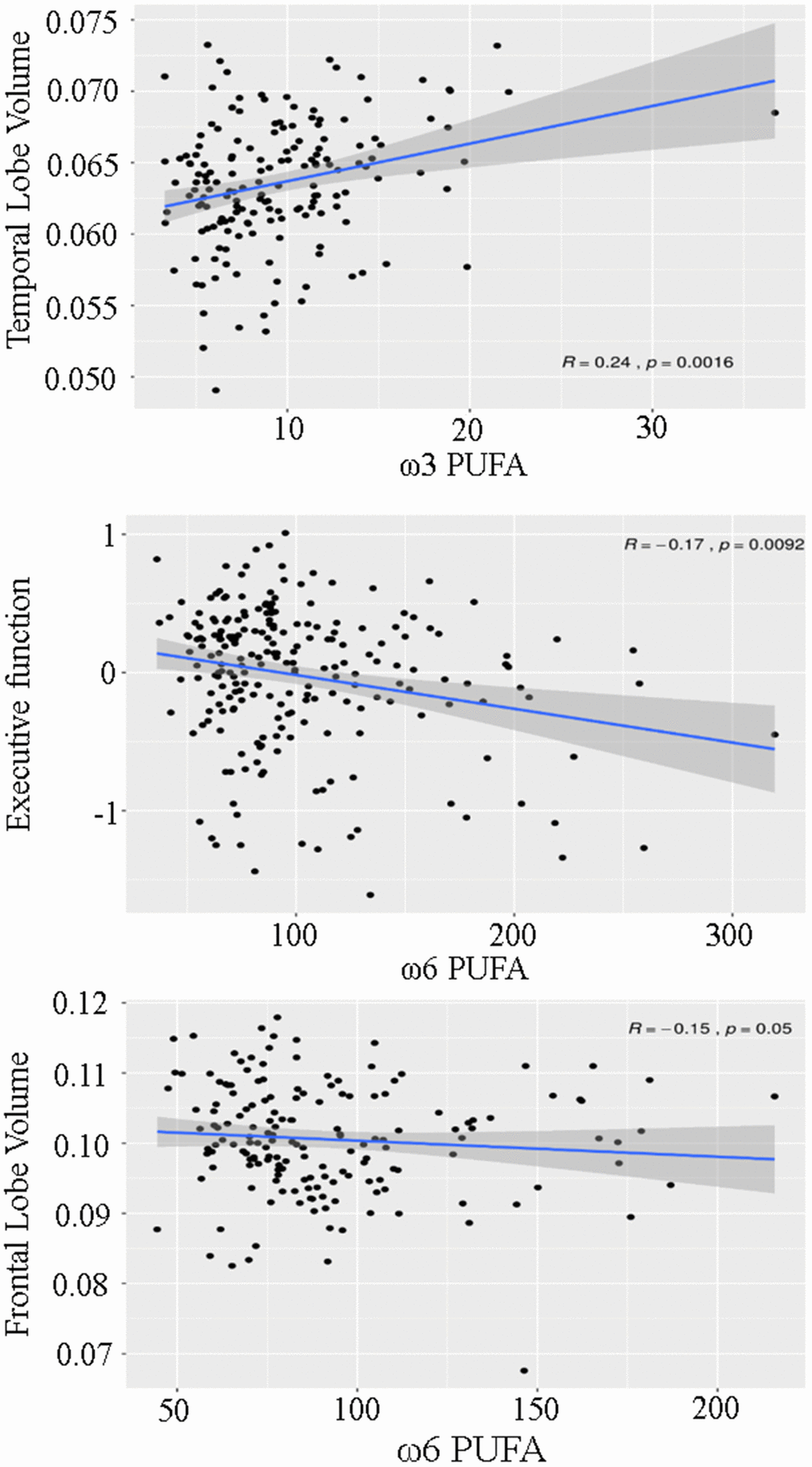
Figure 1. Scatterplot demonstrating the association between ω3 and ω6 polyunsaturated fatty acids (PUFA), temporal lobe volume, executive function and frontal lobe volume.
No significant association existed between free ω-6 PUFA concentrations, visuopractical skills and memory function. The link between ω-6 PUFA and executive function was primarily driven by the plasma concentration of linoleic acid (Table 3). Contrary to free ω-6 PUFA, the plasma concentrations of free ω-3 PUFA were not significantly related to any of the three cognitive domains.
Association between plasma free PUFAs and MRI findings
A higher plasma ω-6 PUFA concentration was significantly associated with a lower volume of the frontal lobe (Table 4, Figure 1) while lower ω-3 PUFA concentrations related positively to the volume of the temporal lobe. Both associations lost significance when p-values were corrected for the expected false discovery rate due to multiple testing. Furthermore, ω-3 and ω-6 PUFA were not associated with WMH and PSMD. However, only 17 individuals showed lacunes, 7 individuals had MRI signs of infarction, and PSMD results were available from 73 participants.
Discussion
Our results support the view that the composition of free plasma PUFAs is linked to cognitive function and the preservation of brain volume in non-demented elderly individuals. In ASPS, higher plasma concentrations of free ω-6 PUFAs, and in particular linoleic acid, were significantly associated with lower executive function. In line with this clinical finding, we also saw that the volume of the frontal lobes was smaller in the presence of higher ω-6 PUFA concentrations. In contrast, higher ω-3 PUFA concentrations were not significantly related to cognitive functioning and the association with larger temporal lobe volumes did not withstand correction for multiple comparisons. We did not find an association of PUFAs and WMH, lacunes, infarctions or PSMD. Yet the number of patients with vascular lesions was small as was the number of study participants who underwent diffusion tensor imaging.
Previous studies have demonstrated a predictive role of PUFAs for cognitive decline and the risk of dementia [18, 19, 24]. Most of these studies reported significant associations between ω-3 PUFA and cognitive function. For example, in the longitudinal Three City Study with over 1200 participants, Thomas et al. found a 13% decrease in the risk of dementia per one SD higher plasma ω-3 PUFA concentration [18]. Moreover, ω-3 PUFAs were also associated with global cognition and memory. Similar results were obtained in community-dwelling elderly Japanese [19] and in patients with coronary artery disease [24]. In a supplementation study by Soininen et al. the administration of Fortasyn Connect, a combination of ω-3 PUFAs and multiple vitamins, slowed down the cognitive decline in subjects with prodromal AD [57, 58]. In contrast, other studies failed to replicate these findings in both demented and non-demented individuals [59, 60]. Also, in our cohort, ω-3 PUFA plasma concentrations were not associated with cognitive function. Previous literature reported conflicting results for ω-6 PUFAs as well. Our results corroborate studies from Australia and Japan that found a higher concentration of total ω-6 PUFAs and AA in patients with cognitive impairment and dementia than in controls [61–63]. Others found no such differences [26, 64]. In a meta-analysis of 10 cross-sectional and case-control studies, Lin et al. reported higher EPA, DHA and total ω-3 PUFA concentrations in non-demented controls than in dementia patients but the ω-3/ω-6 PUFA ratio did not significantly differ between these groups [27]. However, most of the studies included in this meta-analysis were rather small case-control studies that compared mixed cohorts of demented patients and non-demented controls. Furthermore, dementia and cognitive function were differently assessed hampering a direct comparison between studies. The ASPS study extends previous work as it included a large cohort of 391 non-demented individuals that underwent in-depth cognitive testing which captured different domains of cognitive function. Moreover, an extensive panel of 10 FA species was studied with a fully validated and quality-controlled GC-MS method. This method detects t-FFAs including FAs that are loosely bound to albumin and FAs that circulate freely in blood. Another strength of ASPS is the inclusion of volumetric MRI measurements. The association between higher ω-6 PUFA concentrations with smaller frontal lobe volume and executive dysfunction is biologically plausible, because the frontal cortex is traditionally considered the major brain structure involved in executive functions [65–68]. It is of note, however that this association was not particularly strong and lost significance after correction for multiple testing. Another finding of interest was the positive relationship between ω-3 PUFAs and temporal lobe volume. Although in our analysis the association lost significance after correction for multiple testing, several previous reports made a similar observation [18, 28]. In 467 non-demented participants of the Three City Study with >1 MRI scan over a median follow-up of 4.0 years, higher EPA + DHA concentrations which are major constituents of the ω-3 PUFA fraction, were significantly associated with less atrophy of the temporal lobes and slower cognitive decline over the observational period [18]. The authors calculated that per 1 SD increase in ω-3 PUFA concentration, the loss of mean medial temporal lobe volume decreased by 0.02 cm3/year. Another investigation of the same group mainly confirmed their previous results [28]. An in vivo study on monkeys is also in line with these epidemiological data as it showed that the lipid composition of the diet has a direct influence on the PUFA pattern in the temporal lobe [69]. Importantly, the impact of dietary lipid intake on the PUFA composition at brain tissue level differs between specific brain regions [70, 71]. This might explain our observation that differences in free plasma PUFA concentrations affect some, but not all, brain regions. WMH and PSMD were both unrelated to free plasma PUFA concentrations. This observation supports the concept that PUFAs influence brain function and volume primarily through a modulatory effect on cerebral lipid composition. However, the low number of cases in whom these and other structural parameters were available limits statistical power.
PUFAs may contribute to brain aging and cognitive decline in several ways. The balance between ω-3 and ω-6 PUFAs is essential for neural development during childhood and adolescence, and remains important throughout life for membrane fluidity, the prevention of inflammatory states, and cardiovascular health [72]. For example, the brains from AD patients exhibit reductions in DHA and the DHA derived mediator neuroprotectin D1 (NPD1), which protects against cell injury-induced oxidative stress [73]. NPD1 is not only anti-inflammatory and neuroprotective, but also harbors anti-amyloidogenic activity [74]. In mice, administration of a DHA-rich diet increased the concentration of brain-derived neurotrophic factor (BDNF), which promotes synaptic plasticity and improves learning and memory function [75]. Furthermore, in mice DHA protects against β-amyloid (Aβ) production and deposition, and cerebral amyloid angiopathy [75–77].
Our results suggest that the frontal and temporal lobe are particularly sensitive to imbalances of ω-3 and ω-6 free PUFAs. It is important to note that the observed effects of ω-3 and ω-6 PUFAs on cognition and brain structure are subtle and might thus be of limited clinical relevance. Longitudinal studies should clarify the prospective risk of PUFA imbalances for accelerated cognitive decline and dementia. Moreover, the impact of the truly unbound PUFA fraction on cognitive function and the risk of dementia needs to be further explored.
Author Contributions
SS performed all measurements and quantifications. MH, WH and RS developed the study concept, were involved in the data analyses and manuscript writing. RS provided clinical and neuroimaging data of the study. EH performed all statistical analyses and was involved in the manuscript writing. SS and GF developed the method for the measurement of free fatty acids and were involved in the analysis of the results and the writing of the manuscript. GF supervised this study project. All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
References
- 1. Chang CY, Ke DS, Chen JY. Essential fatty acids and human brain. Acta Neurol Taiwan. 2009; 18:231–41. [PubMed]
- 2. Avallone R, Vitale G, Bertolotti M. Omega-3 Fatty Acids and Neurodegenerative Diseases: New Evidence in Clinical Trials. Int J Mol Sci. 2019; 20:4256. https://doi.org/10.3390/ijms20174256 [PubMed]
- 3. Oulhaj A, Jernerén F, Refsum H, Smith AD, de Jager CA. Omega-3 Fatty Acid Status Enhances the Prevention of Cognitive Decline by B Vitamins in Mild Cognitive Impairment. J Alzheimers Dis. 2016; 50:547–57. https://doi.org/10.3233/JAD-150777 [PubMed]
- 4. Weiser MJ, Butt CM, Mohajeri MH. Docosahexaenoic Acid and Cognition throughout the Lifespan. Nutrients. 2016; 8:99. https://doi.org/10.3390/nu8020099 [PubMed]
- 5. Simopoulos AP. The omega-6/omega-3 fatty acid ratio, genetic variation, and cardiovascular disease. Asia Pac J Clin Nutr. 2008 (Suppl 1); 17:131–34. [PubMed]
- 6. Chappus-McCendie H, Chevalier L, Roberge C, Plourde M. Omega-3 PUFA metabolism and brain modifications during aging. Prog Neuropsychopharmacol Biol Psychiatry. 2019; 94:109662. https://doi.org/10.1016/j.pnpbp.2019.109662 [PubMed]
- 7. Brenna JT, Diau GY. The influence of dietary docosahexaenoic acid and arachidonic acid on central nervous system polyunsaturated fatty acid composition. Prostaglandins Leukot Essent Fatty Acids. 2007; 77:247–50. https://doi.org/10.1016/j.plefa.2007.10.016 [PubMed]
- 8. Wassall SR, Leng X, Canner SW, Pennington ER, Kinnun JJ, Cavazos AT, Dadoo S, Johnson D, Heberle FA, Katsaras J, Shaikh SR. Docosahexaenoic acid regulates the formation of lipid rafts: A unified view from experiment and simulation. Biochim Biophys Acta Biomembr. 2018; 1860:1985–93. https://doi.org/10.1016/j.bbamem.2018.04.016 [PubMed]
- 9. Uauy R, Dangour AD. Nutrition in brain development and aging: role of essential fatty acids. Nutr Rev. 2006; 64:S24–33. https://doi.org/10.1301/nr.2006.may.s24-s33 [PubMed]
- 10. Liu JJ, Green P, John Mann J, Rapoport SI, Sublette ME. Pathways of polyunsaturated fatty acid utilization: implications for brain function in neuropsychiatric health and disease. Brain Res. 2015; 1597:220–46. https://doi.org/10.1016/j.brainres.2014.11.059 [PubMed]
- 11. Lagarde M, Bernoud N, Brossard N, Lemaitre-Delaunay D, Thiès F, Croset M, Lecerf J. Lysophosphatidylcholine as a preferred carrier form of docosahexaenoic acid to the brain. J Mol Neurosci. 2001; 16:201–04. https://doi.org/10.1385/JMN:16:2-3:201 [PubMed]
- 12. Wang SW, Wang M, Grossman BM, Martin RJ. Effects of dietary fat on food intake and brain uptake and oxidation of fatty acids. Physiol Behav. 1994; 56:517–22. https://doi.org/10.1016/0031-9384(94)90295-x [PubMed]
- 13. Rapoport SI. In vivo fatty acid incorporation into brain phosholipids in relation to plasma availability, signal transduction and membrane remodeling. J Mol Neurosci. 2001; 16:243–61. https://doi.org/10.1385/JMN:16:2-3:243 [PubMed]
- 14. Spencer SJ, Korosi A, Layé S, Shukitt-Hale B, Barrientos RM. Food for thought: how nutrition impacts cognition and emotion. NPJ Sci Food. 2017; 1:7. https://doi.org/10.1038/s41538-017-0008-y [PubMed]
- 15. Grant WB. Dietary links to Alzheimer's disease: 1999 update. J Alzheimers Dis. 1999; 1:197–201. https://doi.org/10.3233/jad-1999-14-501 [PubMed]
- 16. Patil S, Chan C. Palmitic and stearic fatty acids induce Alzheimer-like hyperphosphorylation of tau in primary rat cortical neurons. Neurosci Lett. 2005; 384:288–93. https://doi.org/10.1016/j.neulet.2005.05.003 [PubMed]
- 17. Wilson DM, Binder LI. Free fatty acids stimulate the polymerization of tau and amyloid beta peptides. In vitro evidence for a common effector of pathogenesis in Alzheimer's disease. Am J Pathol. 1997; 150:2181–95. [PubMed]
- 18. Thomas A, Baillet M, Proust-Lima C, Féart C, Foubert-Samier A, Helmer C, Catheline G, Samieri C. Blood polyunsaturated omega-3 fatty acids, brain atrophy, cognitive decline, and dementia risk. Alzheimers Dement. 2020. [Epub ahead of print]. https://doi.org/10.1002/alz.12195 [PubMed]
- 19. Nishihira J, Tokashiki T, Higashiuesato Y, Willcox DC, Mattek N, Shinto L, Ohya Y, Dodge HH. Associations between Serum Omega-3 Fatty Acid Levels and Cognitive Functions among Community-Dwelling Octogenarians in Okinawa, Japan: The KOCOA Study. J Alzheimers Dis. 2016; 51:857–66. https://doi.org/10.3233/JAD-150910 [PubMed]
- 20. Conklin SM, Manuck SB, Yao JK, Flory JD, Hibbeln JR, Muldoon MF. High omega-6 and low omega-3 fatty acids are associated with depressive symptoms and neuroticism. Psychosom Med. 2007; 69:932–34. https://doi.org/10.1097/PSY.0b013e31815aaa42 [PubMed]
- 21. Duan Q, Fan R, Lei R, Ma W, Ding B. Plasma fatty acid profile is related to cognitive function in obese Chinese populations (35-64 years): A cross-sectional study. Food Sci Nutr. 2020; 8:4773–81. https://doi.org/10.1002/fsn3.1738 [PubMed]
- 22. Phillips MA, Childs CE, Calder PC, Rogers PJ. Lower omega-3 fatty acid intake and status are associated with poorer cognitive function in older age: A comparison of individuals with and without cognitive impairment and Alzheimer's disease. Nutr Neurosci. 2012; 15:271–77. https://doi.org/10.1179/1476830512Y.0000000026 [PubMed]
- 23. Suwa M, Yamaguchi S, Komori T, Kajimoto S, Kino M. The Association between Cerebral White Matter Lesions and Plasma Omega-3 to Omega-6 Polyunsaturated Fatty Acids Ratio to Cognitive Impairment Development. Biomed Res Int. 2015; 2015:153437. https://doi.org/10.1155/2015/153437 [PubMed]
- 24. Yagi S, Hara T, Ueno R, Aihara K, Fukuda D, Takashima A, Hotchi J, Ise T, Yamaguchi K, Tobiume T, Iwase T, Yamada H, Soeki T, et al. Serum concentration of eicosapentaenoic acid is associated with cognitive function in patients with coronary artery disease. Nutr J. 2014; 13:112. https://doi.org/10.1186/1475-2891-13-112 [PubMed]
- 25. Chiu CC, Frangou S, Chang CJ, Chiu WC, Liu HC, Sun IW, Liu SI, Lu ML, Chen CH, Huang SY, Dewey ME, Stewart R. Associations between n-3 PUFA concentrations and cognitive function after recovery from late-life depression. Am J Clin Nutr. 2012; 95:420–27. https://doi.org/10.3945/ajcn.111.015784 [PubMed]
- 26. Conquer JA, Tierney MC, Zecevic J, Bettger WJ, Fisher RH. Fatty acid analysis of blood plasma of patients with Alzheimer's disease, other types of dementia, and cognitive impairment. Lipids. 2000; 35:1305–12. https://doi.org/10.1007/s11745-000-0646-3 [PubMed]
- 27. Lin PY, Chiu CC, Huang SY, Su KP. A meta-analytic review of polyunsaturated fatty acid compositions in dementia. J Clin Psychiatry. 2012; 73:1245–54. https://doi.org/10.4088/JCP.11r07546 [PubMed]
- 28. Samieri C, Maillard P, Crivello F, Proust-Lima C, Peuchant E, Helmer C, Amieva H, Allard M, Dartigues JF, Cunnane SC, Mazoyer BM, Barberger-Gateau P. Plasma long-chain omega-3 fatty acids and atrophy of the medial temporal lobe. Neurology. 2012; 79:642–50. https://doi.org/10.1212/WNL.0b013e318264e394 [PubMed]
- 29. Virtanen JK, Siscovick DS, Lemaitre RN, Longstreth WT, Spiegelman D, Rimm EB, King IB, Mozaffarian D. Circulating omega-3 polyunsaturated fatty acids and subclinical brain abnormalities on MRI in older adults: the Cardiovascular Health Study. J Am Heart Assoc. 2013; 2:e000305. https://doi.org/10.1161/JAHA.113.000305 [PubMed]
- 30. Araya-Quintanilla F, Gutiérrez-Espinoza H, Sánchez-Montoya U, Muñoz-Yañez MJ, Baeza-Vergara A, Petersen-Yanjarí M, Fernández-Lecaros L. Effectiveness of omega-3 fatty acid supplementation in patients with Alzheimer disease: A systematic review and meta-analysis. Neurologia (Engl Ed). 2020; 35:105–14. https://doi.org/10.1016/j.nrl.2017.07.009 [PubMed]
- 31. Devore EE, Grodstein F, van Rooij FJ, Hofman A, Rosner B, Stampfer MJ, Witteman JC, Breteler MM. Dietary intake of fish and omega-3 fatty acids in relation to long-term dementia risk. Am J Clin Nutr. 2009; 90:170–76. https://doi.org/10.3945/ajcn.2008.27037 [PubMed]
- 32. Kröger E, Verreault R, Carmichael PH, Lindsay J, Julien P, Dewailly E, Ayotte P, Laurin D. Omega-3 fatty acids and risk of dementia: the Canadian Study of Health and Aging. Am J Clin Nutr. 2009; 90:184–92. https://doi.org/10.3945/ajcn.2008.26987 [PubMed]
- 33. Sydenham E, Dangour AD, Lim WS. Omega 3 fatty acid for the prevention of cognitive decline and dementia. Cochrane Database Syst Rev. 2012; 6:CD005379. https://doi.org/10.1002/14651858.CD005379.pub3 [PubMed]
- 34. Huber AH, Kleinfeld AM. Unbound free fatty acid profiles in human plasma and the unexpected absence of unbound palmitoleate. J Lipid Res. 2017; 58:578–85. https://doi.org/10.1194/jlr.M074260 [PubMed]
- 35. Schmidt R, Fazekas F, Kapeller P, Schmidt H, Hartung HP. MRI white matter hyperintensities: three-year follow-up of the Austrian Stroke Prevention Study. Neurology. 1999; 53:132–39. https://doi.org/10.1212/wnl.53.1.132 [PubMed]
- 36. Schmidt R, Lechner H, Fazekas F, Niederkorn K, Reinhart B, Grieshofer P, Horner S, Offenbacher H, Koch M, Eber B, Schumacher M, Kapeller P, Freidl W, Dusek T. Assessment of cerebrovascular risk profiles in healthy persons: definition of research goals and the Austrian Stroke Prevention Study (ASPS). Neuroepidemiology. 1994; 13:308–13. https://doi.org/10.1159/000110396 [PubMed]
- 37. Ghadery C, Pirpamer L, Hofer E, Langkammer C, Petrovic K, Loitfelder M, Schwingenschuh P, Seiler S, Duering M, Jouvent E, Schmidt H, Fazekas F, Mangin JF, et al. R2* mapping for brain iron: associations with cognition in normal aging. Neurobiol Aging. 2015; 36:925–32. https://doi.org/10.1016/j.neurobiolaging.2014.09.013 [PubMed]
- 38. Seiler S, Schmidt H, Lechner A, Benke T, Sanin G, Ransmayr G, Lehner R, Dal-Bianco P, Santer P, Linortner P, Eggers C, Haider B, Uranues M, et al, and PRODEM Study Group. Driving cessation and dementia: results of the prospective registry on dementia in Austria (PRODEM). PLoS One. 2012; 7:e52710. https://doi.org/10.1371/journal.pone.0052710 [PubMed]
- 39. Redberg RF, Benjamin EJ, Bittner V, Braun LT, Goff DC
Jr , Havas S, Labarthe DR, Limacher MC, Lloyd-Jones DM, Mora S, Pearson TA, Radford MJ, Smetana GW, et al, and American Academy of Family Physicians, and American Association of Cardiovascular and Pulmonary Rehabilitation, and Preventive Cardiovascular Nurses Association. ACCF/AHA 2009 performance measures for primary prevention of cardiovascular disease in adults: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Performance Measures (Writing Committee to Develop Performance Measures for Primary Prevention of Cardiovascular Disease) developed in collaboration with the American Academy of Family Physicians; American Association of Cardiovascular and Pulmonary Rehabilitation; and Preventive Cardiovascular Nurses Association: endorsed by the American College of Preventive Medicine, American College of Sports Medicine, and Society for Women's Health Research. J Am Coll Cardiol. 2009; 54:1364–405. https://doi.org/10.1016/j.jacc.2009.08.005 [PubMed] - 40. American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2008 (Suppl 1); 31:S55–60. https://doi.org/10.2337/dc08-S055 [PubMed]
- 41. Matyash V, Liebisch G, Kurzchalia TV, Shevchenko A, Schwudke D. Lipid extraction by methyl-tert-butyl ether for high-throughput lipidomics. J Lipid Res. 2008; 49:1137–46. https://doi.org/10.1194/jlr.D700041-JLR200 [PubMed]
- 42. Quehenberger O, Armando AM, Dennis EA. High sensitivity quantitative lipidomics analysis of fatty acids in biological samples by gas chromatography-mass spectrometry. Biochim Biophys Acta. 2011; 1811:648–56. https://doi.org/10.1016/j.bbalip.2011.07.006 [PubMed]
- 43. Bioanalytical Method Validation. US Department of Health and Human Services. Food and Drug Administration. 2018. https://www.fda.gov/media/70858/download.
- 44. Army Individual Test Battery Manual of Directions and Scoring. Washington, DC: War Department, Adjutant General’s Office; 1944.
- 45. Wiener Reaktionsgerät. Mödling, Austria: Dr. Schuhfried Ges.mbH; 1991.
- 46. Bäumler G. Lern- und Gedächtnistest: LGT-3. Göttingen: Verlag für Psychologie Hogrefe; 1974.
- 47. Heaton RK. Wisconsin card sorting test manual. Odessa, Fl.: Psychological assessment resources; 1981.
- 48. Tewes U. Hamburg-Wechsler-Intelligenztest für Erwachsene: HAWIE-R. Switzerland, Bern: Huber; 1991.
- 49. Tiffin J, Asher EJ. The Purdue pegboard; norms and studies of reliability and validity. J Appl Psychol. 1948; 32:234–47. https://doi.org/10.1037/h0061266 [PubMed]
- 50. Dale AM, Fischl B, Sereno MI. Cortical surface-based analysis. I. Segmentation and surface reconstruction. Neuroimage. 1999; 9:179–94. https://doi.org/10.1006/nimg.1998.0395 [PubMed]
- 51. Fischl B, Salat DH, Busa E, Albert M, Dieterich M, Haselgrove C, van der Kouwe A, Killiany R, Kennedy D, Klaveness S, Montillo A, Makris N, Rosen B, Dale AM. Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron. 2002; 33:341–55. https://doi.org/10.1016/s0896-6273(02)00569-x [PubMed]
- 52. Plummer DL. DispImage: A Display and Analysis Tool for Medical Images. Rivista di Neuroradiologia. 1992; 5:489–95. https://doi.org/10.1177/197140099200500413
- 53. R Core Team. R: A Language and Environment for Statistical Computing (Version 3.6.1, R Foundation for Statistical Computing, Vienna, Austria). 2019. https://www.gbif.org/tool/81287/r-a-language-and-environment-for-statistical-computing.
- 54. Therneau TM. coxme: Mixed Effects Cox Models. 2018. https://cran.r-project.org/web/packages/coxme/index.html.
- 55. Sinwell J, Therneau TM. kinship2: Pedigree Functions. 2019. https://cran.r-project.org/web/packages/kinship2/index.html.
- 56. Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B (Methodological). 1995; 57:289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x
- 57. Soininen H, Solomon A, Visser PJ, Hendrix SB, Blennow K, Kivipelto M, Hartmann T, and LipiDiDiet clinical study group. 24-month intervention with a specific multinutrient in people with prodromal Alzheimer's disease (LipiDiDiet): a randomised, double-blind, controlled trial. Lancet Neurol. 2017; 16:965–75. https://doi.org/10.1016/S1474-4422(17)30332-0 [PubMed]
- 58. Soininen H, Solomon A, Visser PJ, Hendrix SB, Blennow K, Kivipelto M, Hartmann T, and LipiDiDiet clinical study group. 36-month LipiDiDiet multinutrient clinical trial in prodromal Alzheimer's disease. Alzheimers Dement. 2021; 17:29–40. https://doi.org/10.1002/alz.12172 [PubMed]
- 59. Arsenault LN, Matthan N, Scott TM, Dallal G, Lichtenstein AH, Folstein MF, Rosenberg I, Tucker KL. Validity of estimated dietary eicosapentaenoic acid and docosahexaenoic acid intakes determined by interviewer-administered food frequency questionnaire among older adults with mild-to-moderate cognitive impairment or dementia. Am J Epidemiol. 2009; 170:95–103. https://doi.org/10.1093/aje/kwp089 [PubMed]
- 60. Laurin D, Verreault R, Lindsay J, Dewailly E, Holub BJ. Omega-3 fatty acids and risk of cognitive impairment and dementia. J Alzheimers Dis. 2003; 5:315–22. https://doi.org/10.3233/jad-2003-5407 [PubMed]
- 61. Hirai K, Kozuki M, Miyanaga K, Miyagawa F, Takezoe R, Hasegawa M, Mori M. Lower Levels of Eicosapentaenoic Acid and the Ratio of Docosahexaenoic Acid to Arachidonic Acid in Sera of Patients with Multi-Infarct Dementia. Journal of Clinical Biochemistry and Nutrition. 2005; 36:83–89. https://doi.org/10.3164/jcbn.36.83
- 62. Milte CM, Sinn N, Street SJ, Buckley JD, Coates AM, Howe PR. Erythrocyte polyunsaturated fatty acid status, memory, cognition and mood in older adults with mild cognitive impairment and healthy controls. Prostaglandins Leukot Essent Fatty Acids. 2011; 84:153–61. https://doi.org/10.1016/j.plefa.2011.02.002 [PubMed]
- 63. Selley ML. A metabolic link between S-adenosylhomocysteine and polyunsaturated fatty acid metabolism in Alzheimer's disease. Neurobiol Aging. 2007; 28:1834–39. https://doi.org/10.1016/j.neurobiolaging.2006.08.003 [PubMed]
- 64. Boston PF, Bennett A, Horrobin DF, Bennett CN. Ethyl-EPA in Alzheimer's disease--a pilot study. Prostaglandins Leukot Essent Fatty Acids. 2004; 71:341–46. https://doi.org/10.1016/j.plefa.2004.07.001 [PubMed]
- 65. Personality changes with frontal and temporal lobe lesions. In: Psychiatric Aspects of Neurologic Disease. Benson D. Frank and Dietrich Blumer (editors).. New York: Grune & Stratton; 1975.
- 66. Leonard G, Jones L, Milner B. Residual impairment in handgrip strength after unilateral frontal-lobe lesions. Neuropsychologia. 1988; 26:555–64. https://doi.org/10.1016/0028-3932(88)90112-1 [PubMed]
- 67. Levin HS, Amparo E, Eisenberg HM, Williams DH, High WM
Jr , McArdle CB, Weiner RL. Magnetic resonance imaging and computerized tomography in relation to the neurobehavioral sequelae of mild and moderate head injuries. J Neurosurg. 1987; 66:706–13. https://doi.org/10.3171/jns.1987.66.5.0706 [PubMed] - 68. Luria AR. The human brain and psychological processes. New York: Harper & Row; 1966.
- 69. Miller LR, Jorgensen MJ, Kaplan JR, Seeds MC, Rahbar E, Morgan TM, Welborn A, Chilton SM, Gillis J, Hester A, Rukstalis M, Sergeant S, Chilton FH. Alterations in levels and ratios of n-3 and n-6 polyunsaturated fatty acids in the temporal cortex and liver of vervet monkeys from birth to early adulthood. Physiol Behav. 2016; 156:71–78. https://doi.org/10.1016/j.physbeh.2015.12.009 [PubMed]
- 70. Alashmali SM, Hopperton KE, Bazinet RP. Lowering dietary n-6 polyunsaturated fatty acids: interaction with brain arachidonic and docosahexaenoic acids. Curr Opin Lipidol. 2016; 27:54–66. https://doi.org/10.1097/MOL.0000000000000255 [PubMed]
- 71. Levant B, Ozias MK, Carlson SE. Diet (n-3) polyunsaturated fatty acid content and parity affect liver and erythrocyte phospholipid fatty acid composition in female rats. J Nutr. 2007; 137:2425–30. https://doi.org/10.1093/jn/137.11.2425 [PubMed]
- 72. Janssen CI, Kiliaan AJ. Long-chain polyunsaturated fatty acids (LCPUFA) from genesis to senescence: the influence of LCPUFA on neural development, aging, and neurodegeneration. Prog Lipid Res. 2014; 53:1–17. https://doi.org/10.1016/j.plipres.2013.10.002 [PubMed]
- 73. Zhao Y, Calon F, Julien C, Winkler JW, Petasis NA, Lukiw WJ, Bazan NG. Docosahexaenoic acid-derived neuroprotectin D1 induces neuronal survival via secretase- and PPARγ-mediated mechanisms in Alzheimer's disease models. PLoS One. 2011; 6:e15816. https://doi.org/10.1371/journal.pone.0015816 [PubMed]
- 74. Bazan NG, Molina MF, Gordon WC. Docosahexaenoic acid signalolipidomics in nutrition: significance in aging, neuroinflammation, macular degeneration, Alzheimer's, and other neurodegenerative diseases. Annu Rev Nutr. 2011; 31:321–51. https://doi.org/10.1146/annurev.nutr.012809.104635 [PubMed]
- 75. Wu A, Ying Z, Gomez-Pinilla F. Docosahexaenoic acid dietary supplementation enhances the effects of exercise on synaptic plasticity and cognition. Neuroscience. 2008; 155:751–59. https://doi.org/10.1016/j.neuroscience.2008.05.061 [PubMed]
- 76. Hooijmans CR, Rutters F, Dederen PJ, Gambarota G, Veltien A, van Groen T, Broersen LM, Lütjohann D, Heerschap A, Tanila H, Kiliaan AJ. Changes in cerebral blood volume and amyloid pathology in aged Alzheimer APP/PS1 mice on a docosahexaenoic acid (DHA) diet or cholesterol enriched Typical Western Diet (TWD). Neurobiol Dis. 2007; 28:16–29. https://doi.org/10.1016/j.nbd.2007.06.007 [PubMed]
- 77. Hooijmans CR, Van der Zee CE, Dederen PJ, Brouwer KM, Reijmer YD, van Groen T, Broersen LM, Lütjohann D, Heerschap A, Kiliaan AJ. DHA and cholesterol containing diets influence Alzheimer-like pathology, cognition and cerebral vasculature in APPswe/PS1dE9 mice. Neurobiol Dis. 2009; 33:482–98. https://doi.org/10.1016/j.nbd.2008.12.002 [PubMed]



