Introduction
Diabetic nephropathy (DN) is a microvascular disease caused by diabetes that leads to glomerular sclerosis and is a serious progressive complication of diabetes [1, 2]. Hyperglycaemia, hypertension and genetic susceptibility are major risk factors for the development of diabetic nephropathy [3]. The prevention and treatment of DN is a long-term problem for diabetic patients [4].
Previous studies have shown that the main pathological features of early renal injury include glomerular and tubular hypertrophy, basement membrane thickening, and progressive extracellular matrix (ECM) accumulation in the mesangial area [5]. MMP-9 is the main physiological regulator of the ECM, and it can degrade various components of ECM and slow the progression of glomerular sclerosis [6]. PPAR-γ is a ligand-activated nuclear transcription factor that regulates many important physiological responses, including glucose metabolism, inflammation, renal interstitial fibrosis, and insulin resistance [7]. Activated and phosphorylated NF-κB subunits (P65 and P50) translocate to the nucleus and induce the expression of DN-related target genes [8]. It has been reported that regulating NF-κB activity can improve renal ECM accumulation and fibrosis [9]. The chemical name of DCI is 1,2,4-hexahydroxycyclohexane. In recent years, DCI has been widely used in clinical research. However, evidence of its physiological and therapeutic properties is steadily emerging. First, DCI was thought to be an insulin sensitizer and islet analogue that promoted insulin sensitivity and reduced blood glucose levels. We have found that DCI also plays a key role in regulation of steroid production and other important processes, such as intercellular adhesion, inflammation and oxidation. Therefore, DCI is used to treat various diseases in the clinic [10, 11]. However, the specific pathophysiological mechanism by which DCI improves diabetic kidney injury still needs to be further explored. In the present study, we aimed to investigate the effect of DCI on fibrosis in DN. Therefore, we selected the db/db mouse model to solve these problems. The regulatory effect of DCI on glucose metabolism was evaluated by examining blood glucose, blood lipids and renal function. Subsequently, morphological changes in the mouse kidney were observed under a microscope, and changes in the expression levels of fibrosis-related proteins were measured to preliminarily explore the renal protective mechanism of DCI.
Results
Effects of DCI on the level of RBG in db/db mice
DCI RBG levels in db/db mice with type 2 diabetes (Table 1). Before administration, the RBG levels in db/db mice were significantly higher than those in db/m mice (P < 0.01), and the three groups were basically the same. After 6 weeks of administration, the RBG of HDCIG and LDCIG were decreased to some extent compared with those in the MCG. There was no significant difference between the HDCI and LDCI groups. Our findings indicated that DCI could effectively improve RBG levels in db/db mice.
Table 1. Effects of D-chiro-inositol on the level of blood glucose in db/db mice (mmol/L).
| Group | 0 weeks | 6 week | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NCG | 7.22 ± 0.32 | 6.42 ± 0.47 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MCG | 20.56 ± 3.46** | 27.58 ± 4.04** | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HDCIG | 20.52 ± 3.12 | 18.07 ± 4.26## | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDCIG | 20.97 ± 3.31 | 19.12 ± 4.33## | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mean ± SD, n = 10; *P < 0.05, **p < 0.01 vs. NCG; #p < 0.05, ##p < 0.01 vs. MCG. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Effects of DCI on serum Cr, BUN and renal weight index in db/db mice
As shown in Table 2, serum Cr and BUN levels in the MCG were significantly higher than those in the NCG. Compared with those in the MCG, serum Cr and BUN levels results in the HDCIG and LDCIG were reduced, and the effect on the HDCIG was more significant than in the other groups. The kidney index of db/db mice was significantly lower than that of db/m mice (P < 0.01). The kidney index of each drug-administered group was lower than that of the MCG group, however there was no significant difference. Our experimental results showed that DCI effectively alleviated kidney damage caused by diabetes and significantly improved renal function.
Table 2. Effects of D-chiro-inositol on Cr, BUN and renal weight index in db/db mice.
| Group | Cr (μmol/L) | BUN (mmol/L) | Renal weight index (mg/g, %) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NCG | 37.93 ± 8.77 | 187.02 ± 22.40 | 1.24 ± 0.085 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MCG | 50.62 ± 6.74** | 262.29 ± 32.20** | 0.70 ± 0.084** | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HDCIG | 39.44 ± 8.04## | 205.54 ± 23.62## | 0.69 ± 0.099 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDCIG | 43.22 ± 7.34# | 223.24 ± 22.88## | 0.65 ± 0.060 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mean ± SD, n = 10; *p < 0.05, **p < 0.01 vs. NCG; #p < 0.05, ##p < 0.01 vs. MCG. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Pathological staining of kidney tissues in db/db mice
DCI significantly ameliorated histological changes in the kidney (Figure 1). HE staining showed that the nuclei in kidney tissue in the NCG were stained blue, the cytoplasm was stained pink, and the structure of the renal tubules and glomeruli was normal. The MCG had severe inflammatory cell infiltration and significant oedema of renal tubular epithelial cells. The HDCIG and LDCIG reduced renal tissue damage. Masson staining showed a significant increase in glomerular and renal interstitial fibrosis in the MCG, while that in the HDCIG and LDCIG was decreased to varying degrees.
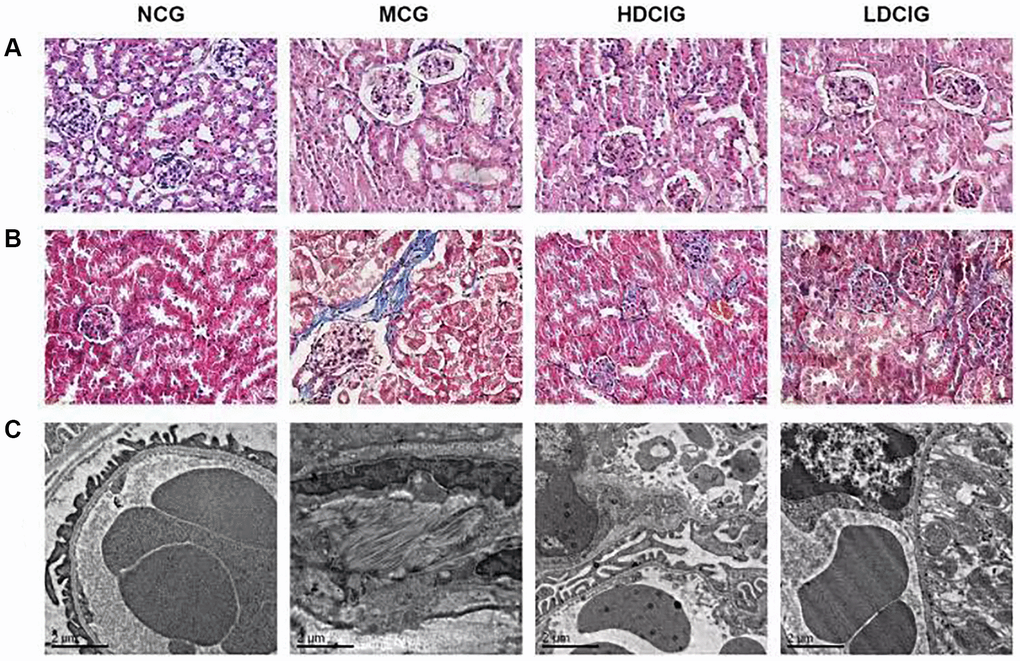
Figure 1. Morphological structure of kidney tissues in db/db mice. (A) HE staining; (B) Masson staining; (C) Electronic microscopy.
As observed by transmission electron microscopy, glomerular mesangial matrix proliferated in the MCG, which was accompanied by podocyte fusion or disappearance and basement membrane thickening. Renal mesangial matrix hyperplasia and basement membrane thickening were attenuated in the HDCIG and LDCIG, and the podocyte structure was improved compared with that in the MCG.
Immunohistochemical analysis of MMP-9, PPAR-γ and NF-κB protein expression
The immunohistochemical staining analysis are shown in Figure 2. MMP-9 and PPAR-γ proteins are expressed on mouse kidney glomerular cells. These proteins were highly expressed in the kidney tissue in the NCG, and the positive staining was brownish yellow and evenly distributed. The protein expression of MMP-9 and PPAR-γ in the MCG was lower than that in the NCG (P < 0.01). Moreover, the expression levels of MMP-9 and PPAR-γ in the HDCIG were significantly higher than those in the MCG. The NF-κB protein was expressed in the cytoplasm of tubular cells. The protein expression of NF-κB protein was lower in the NCG and higher in the MCG. The expression levels of NF-κB protein in the HDCIG and LDCIG were decreased, but there was no significant difference between the two groups.
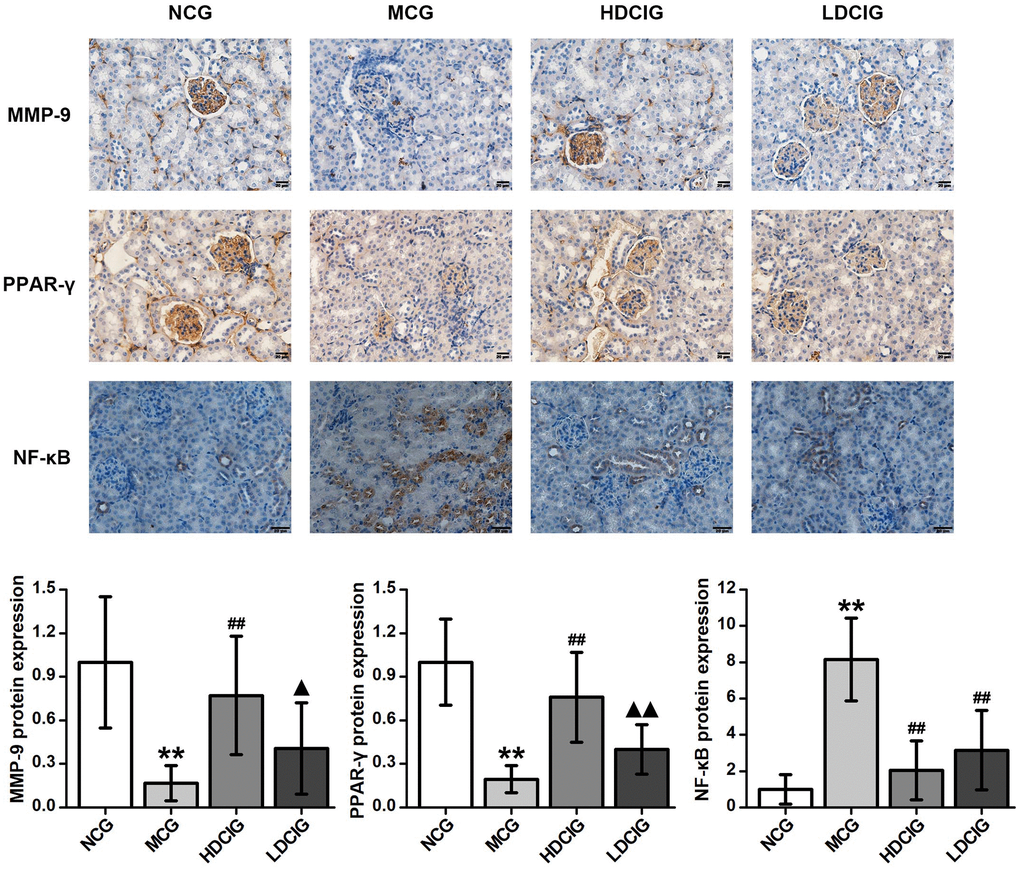
Figure 2. Immunohistochemistry determination of the relative protein expression in the kidney tissues of db/db mice (n = 4). The results obtained for the different groups were normalized to those reported in the normal control group (NCG). All values are expressed as the means ± SD. *p < 0.05, **p < 0.01 vs. NCG mice; #p < 0.05. ##p < 0.01 vs. MCG mice. ▲p < 0.05, ▲▲p < 0.01 vs. HDCIG mice.
Western-blot analysis of MMP-9, PPAR-γ and NF-κB protein expression
The results showed that the expression levels of MMP-9 and PPAR-γ protein in the MCG were lower than those in NCG (Figure 3). The protein expression of MMP-9 and PPAR-γ in the HDCIG was higher than that in the MCG. The expression of NF-κB protein in MCG was significantly higher than that in the NCG. Compared with those in the MCG, the protein expression levels of NF-κB in the HDCIG and LDCIG were decreased. These findings demonstrate the unique ability of DCI to mitigate renal fibrosis by acting on pathophysiological renal fibrosis events, as well as the proteins associated with renal fibrosis.
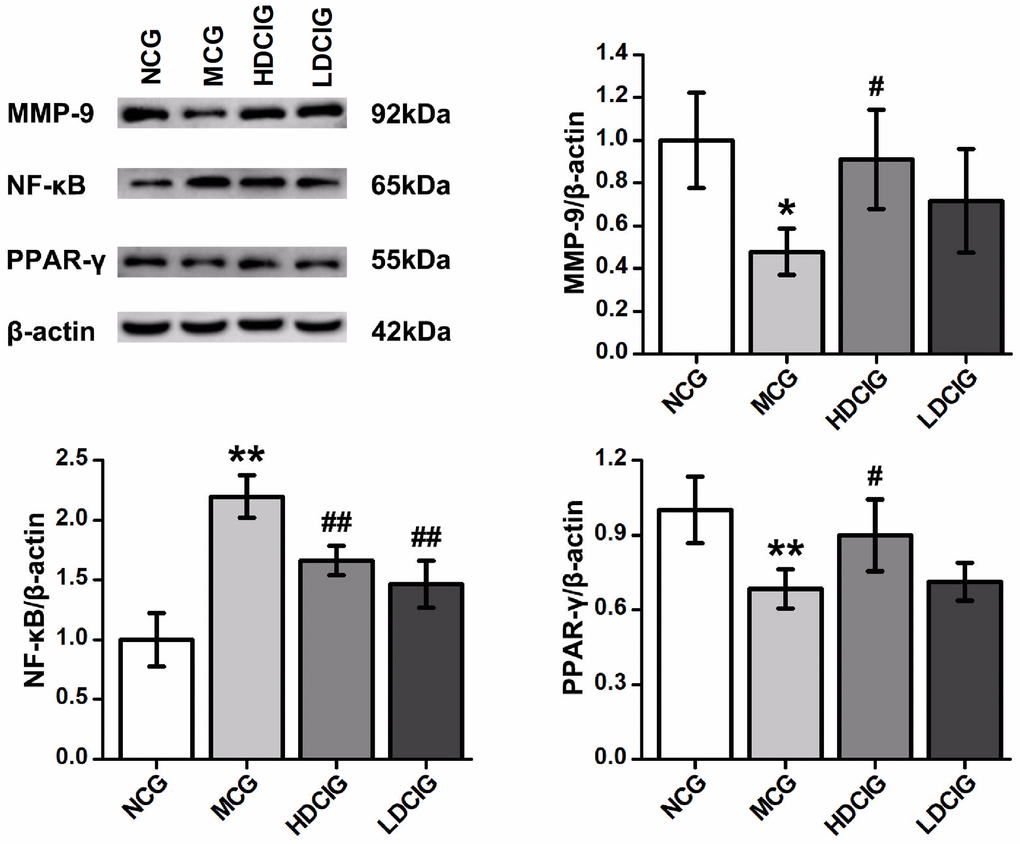
Figure 3. Effects of D-chiro-inositol on relative protein expression in the kidney tissues of db/db mice (n = 3). The results obtained for the different groups were normalized to those reported in the normal control group (NCG). All values are expressed as the means ± SD. *p < 0.05, **p < 0.01 vs. NCG mice; #p < 0.05. ##p < 0.01 vs. MCG mice. ▲p < 0.05, ▲▲p < 0.01 vs. HDCIG mice.
Discussion
DN is a serious complication associated with the development of diabetes and a major cause of end-stage renal disease [12]. The pathogenesis of DN is closely related to a variety of factors, including glucose and lipid metabolism disorders, hemodynamic abnormalities, oxidative stress, cytokines and inflammatory factors [13]. Many basic research studies have proven that renal fibrosis is the most important process in the progression of multiple chronic kidney diseases (including DN) to end-stage renal disease [14–16].
Renal fibrosis is dominated by tubulointerstitial fibrosis, which may manifest as glomerular sclerosis. Db/db mice are an ideal animal model that spontaneously develops type 2 diabetes [17]. This study investigated the effects of DCI on kidney tissue in db/db mice.
A previous study showed that DCI reduced blood glucose levels in db/db mice and protected the liver [18]. Combined with the blood glucose results of this experiment study, these results show that DCI has a good hypoglycaemic glycemic effect and improves the body’s glucose metabolism. Serum Cr and BUN levels are two important indicators for when clinically assessing kidney damage [19]. The serum results showed that DCI reduced the content levels of Cr and BUN in the serum of db/db mice and promoted glomerular filtration. Additionally, pathological staining showed that DCI reduced the proliferation of glomerular mesangial matrix in db/db mice and reduced the degree of renal fibrosis. This finding indicates that DCI alleviates kidney damage in db/db mice.
The MMP family is a major factor in the degradation of various ECM components [20], and of in this family, MMP-9 is a key enzyme that regulates ECM degradation [21], and is closely related to the occurrence and development of DN. Abnormal expression of MMP-9 is involved in proteinuria-related kidney diseases, including focal segmental glomerulosclerosis and anti-glomerular basement membrane inflammation [6]. Under high glucose stimulation, the gene expression of multiple mesangial matrix components is upregulated, and ECM synthesis is increased. In addition, MMP-9 expression is affected, and ECM degradation is reduced, promoting the process of renal fibrosis. Therefore, it is important to maintain the normal level of MMP-9. The transcription factor NF-κB is involved in the transcriptional regulation of many adhesion molecules and inflammatory cytokines, such as TNF-α, IL-10, and ICAM-1 [22, 23], so and increased levels of NF-κB leads to inflammatory reactions [24]. PPAR-γ plays an important role in improving insulin resistance, promoting glucose metabolism, lowering blood pressure, inhibiting fat accumulation in the kidney, increasing adiponectin expression, protecting podocytes, inhibiting renal interstitial fibrosis, and exerting anti-inflammatory and antioxidant effects [25–27]. Studies have shown that PPAR-γ inhibits NF-κB signalling by inhibiting the transcriptional activity of NF-κB, thereby inhibiting the expression of inflammatory factors associated with TNF-α [28]. In the current study, we found that DCI certainly enhanced the expression of PPAR-γ and MMP-9, decreased NF-κB activity, and reduced renal fibrosis in mice. These results suggested that enhancing PPAR-γ protein expression ameliorated fibrosis at least in part through by modulating the activity of the NF-κB and the expression of MMP-9. Therefore, based on our results, we speculate hypothesize that DCI protects the kidney by enhancing the expression of PPAR-γ and possibly alleviating renal fibrosis through the NF-κB/ MMP-9 signalling pathway.
Conclusions
In summary, DCI reduces blood glucose and kidney tissue damage in db/db mice. This outcome may be related to increases in the protein expression of MMP-9 and PPAR-γ and the inhibition of NF-κB protein expression in the kidney tissue of db/db mice. However, the mechanism of DCI may involve multiple targets and multiple pathways, and further research is needed.
Materials and Methods
Animal care
Male db/db mice (8 weeks, 29–35 g) and male db/m mice (8 weeks, 18–22 g) were purchased from Changzhou Cavens Laboratory Animal Co. Ltd. (Changzhou, China) (licence key: SCXK (Su) 2016-0010). Mouse granule feedstuff treated 60Co radiation was purchased from Nanjing Beisifu Feed Co. Ltd. (Nanjing, China). The animal experiments were performed in specific-pathogen-free barrier laboratory at the Experimental Animal Centre of North China University of Science and Technology (Tangshan, China). All procedures for animal experiments were approved by the Animal Ethics Committee of North China University of Science and Technology, according to the guidelines established by the European Union (Directive 2010/63/EU for animal experiments) and the National Institute of Health of the USA (NIH Publications No. 8023, revised 1978).
Grouping and administration
After adaptive feeding for 1 week, blood was drawn from the tails of the mice to determine the levels of blood glucose using a blood glucose metre (Roche, USA). Ten db/m mice were selected to establish the normal control group (NCG). Based on the RBG levels, the 30 db/db mice were randomly divided into three groups received different treatments: high-dose DCI group (HDCIG), low-dose DCI group (LDCIG) and model control group (MCG).
The mice in the HDCIG and LDCIG mice received DCI (purity: >98.0 %, I0632, TCI, Japan) at a dose of 70 mg/kg/d and 35 mg/kg/d, respectively. The NCG and MCG mice received an equal volume of pure water. All treatments were administered at 9 a.m. every day, and the duration of the entire experiment was 6 weeks.
Laboratory analyses
RBG measurements were conducted in each group at 0 and 6 weeks used tail vein blood after intragastric administration. Twenty-four hours after the last administration, blood samples were collected from the eye socket, and the serum was separated. Cr and BUN levels were measured using Assayan assay kit (Nanjing Jiancheng Bioengineering Institute, C011-2-1/1/C013-2-1, China) according to the Kit instructions.
Histopathological
After being weighed, 1 mm3 kidney tissue blocks were cut at the renal cortex, fixed with 0.025% glutaraldehyde solution for 12 h, incubated with phosphate buffer -buffered saline (PBS) for 8 h, and incubated with 0.01% osmic acid for 2 h. Then, the samples were rinsed and dehydrated, embedded with Epon 812, sliced (70 nm), and stained with lead citrate. Finally, the samples were observed by transmission electron microscopy (H-7650, Hitachi Limited, Japan).
The kidney tissue was fixed in 4% paraformaldehyde for 72 h, and subsequently subjected to conventional paraffin embedding. Then, the kidney tissue was cut into 3.5-μm-thick sections. After HE staining and Masson staining, the histological structure of the kidney tissue was observed under a BX50 microscope (Olympus, Japan).
Immunohistochemistry
Immunohistochemistry was performed using a universal two-step test kit (ZSGB-BIO, PV-9000, China). Paraffin-embedded sections of kidney tissues were dewaxed, hydrated, and boiled to repair antigens through ethylenediaminetetraacetic acid (EDTA) high-pressure heating. Following a wash with PBS, the sections were soaked with serum (ZSGB-BIO, ZLI-9022, China) and incubated with the following primary antibodies: anti-MMP-9 (E-11) (1:300, Santa Cruz, sc393859, USA), anti-PPAR-γ (1:200, Santa Cruz, sc7273, USA) and anti-NF-κB (1:200, Santa Cruz, sc8008, USA). After being incubated at 4°C overnight, the sections were incubated with the secondary antibody provided by the kit at 37°C. After being washed with PBS, the sections were dyed using 3,3′-diaminobenzidine (ZSGB-BIO, ZLI-9018, China) for 50 s. The nucleus was stained with hematoxylin for 1 min. In the negative control group, PBS was used instead of primary antibodies in the experiments. Finally, the positive proteins were colored brownish yellow, and the expression and distribution of indicated proteins were observed under a light microscope. The average integral absorbance was determined using Image-Pro Plus 6.0 software.
Western blot analysis
The kidney tissue was homogenized in protein lysate buffer (Leagene, PS0013, China). The supernatant was harvested after centrifugation at 4°C, and the protein was quantified using a bicinchoninic acid assay kit (Leagene, PT0006, China). The proteins were separated by gradient sodium dodecyl sulfate–polyacrylamide gel electrophoresis (Leagene, PE0017, China) and electrophoretically transferred to polyvinylidene difluoride blotting membranes (GE, 10600023, USA). The membranes were blocked with 5% non-fat dry milk for 2 h, and subsequently incubated with primary antibodies overnight at 4°C. The primary antibodies used were as follows: anti- MMP-9 antibody (1:500, Santa Cruz, sc393859), anti- PPAR-γ (1:500, Santa Cruz, sc7273), anti-NF-κB (1:500, SANTA, sc8008, USA) and anti-β-actin (1:5,000, Bioworld, BS6007M, USA). After being washed with TBST (Solarbio, T1081, China), the membranes were incubated with anti-mouse HRP labelled secondary antibodies (1:5,000, SeraCare, 5450-0011, USA). Signal quantification was performed using an Odyssey Infrared Imaging System (Li-COR, USA). The protein bands were normalized to the β-actin band in each sample.
Statistical analysis
Statistical analysis was performed using SPSS 19.0 software, and the experimental data are expressed as the means ± standard deviation (SD). Statistical significance was determined using one-way analysis of variance, followed by a least significant difference test for multiple comparisons. P < 0.05 denoted statistical significance.
Ethics statement
The experiments were approved by the Animal Ethics Welfare Committee of North China University of Science and Technology.
Author Contributions
Conceived and designed the study: GLH, HSY. Conducted the experiment: ZDD, ZJL. Drafted the manuscript: FCX. Analyzed the data: LJW, WY.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Funding
This research was supported by grants from the Natural Science Foundation of Hebei Province (Grant No.H2018209260). The authors would like to thank Hebei Key Laboratory for Chronic Diseases, Tangshan Key Laboratory for Preclinical and Basic Research on Chronic Diseases, PR China.
References
- 1. Bramham K. Diabetic Nephropathy and Pregnancy. Semin Nephrol. 2017; 37:362–9. https://doi.org/10.1016/j.semnephrol.2017.05.008 [PubMed]
- 2. Vervoort G. Treatment Goals in Diabetic Nephropathy. In: Roelofs J, Vogt L (eds). Diabetic Nephropathy. Springer, Cham. 2019; 435–50. https://doi.org/10.1007/978-3-319-93521-8_25
- 3. Gross JL, de Azevedo MJ, Silveiro SP, Canani LH, Caramori ML, Zelmanovitz T. Diabetic nephropathy: diagnosis, prevention, and treatment. Diabetes Care. 2005; 28:164–76. https://doi.org/10.2337/diacare.28.1.164 [PubMed]
- 4. Jakić M, Jakić M, Zibar L, Mihaljević D, Stipanić S, Teskera T. [Diabetic nephropathy and prevention of diabetic nephropathy caused chronic renal insufficiency]. Lijec Vjesn. 2009; 131:218–25. [PubMed]
- 5. Kolset SO, Reinholt FP, Jenssen T. Diabetic nephropathy and extracellular matrix. J Histochem Cytochem. 2012; 60:976–86. https://doi.org/10.1369/0022155412465073 [PubMed]
- 6. Huang HX, Jiang ZP, Shi YJ, Cheng XS, Chen XP, Han SY, Guo L. Effects of fermented buckwheat extract on vascular endothelial growth factor and matrix metalloproteinase-9 in renal tissues of db/db mice. Modern Preventive Medicine. 2017; 44:1102–6.
- 7. Odegaard JI, Ricardo-Gonzalez RR, Goforth MH, Morel CR, Subramanian V, Mukundan L, Red Eagle A, Vats D, Brombacher F, Ferrante AW, Chawla A. Macrophage-specific PPARgamma controls alternative activation and improves insulin resistance. Nature. 2007; 447:1116–20. https://doi.org/10.1038/nature05894 [PubMed]
- 8. Yi H, Peng R, Zhang LY, Sun Y, Peng HM, Liu HD, Yu LJ, Li AL, Zhang YJ, Jiang WH, Zhang Z. LincRNA-Gm4419 knockdown ameliorates NF-κB/NLRP3 inflammasome-mediated inflammation in diabetic nephropathy. Cell Death Dis. 2017; 8:e2583. https://doi.org/10.1038/cddis.2016.451 [PubMed]
- 9. Zhang L, Liu L, Bai M, Liu M, Wei L, Yang Z, Qian Q, Ning X, Sun S. Hypoxia-induced HE4 in tubular epithelial cells promotes extracellular matrix accumulation and renal fibrosis via NF-κB. FASEB J. 2020; 34:2554–67. https://doi.org/10.1096/fj.201901950R [PubMed]
- 10. Wang HY, Li Y. Research Status and Applied Prospect of Buckewheat. Food Science. 2004; 25:388–91.
- 11. Liu G, Hu TT, Zhang XY, Deng QJ, Liu XY, Zhou L, Wang SF, Wang ZG. Hypoglycemic Effect of Ethanol Extracts of Non-indigenous Black Buckwheat Stems and Leaves in Mice and Changes to Functional Components. Modern Food Science & Technology. 2017; 33:1–7. https://doi.org/10.13982/j.mfst.1673-9078.2017.2.001
- 12. Peters V, Yard B, Schmitt CP. Carnosine and Diabetic Nephropathy. Curr Med Chem. 2020; 27:1801–12. https://doi.org/10.2174/0929867326666190326111851 [PubMed]
- 13. Gaballa MR, Farag YMK. Predictors of diabetic nephropathy. Cent Eur J Med. 2013; 8:287–96. https://doi.org/10.2478/s11536-012-0055-3
- 14. Panizo S, Martínez-Arias L, Alonso-Montes C, Cannata P, Martín-Carro B, Fernández-Martín JL, Naves-Díaz M, Carrillo-López N, Cannata-Andía JB. Fibrosis in Chronic Kidney Disease: Pathogenesis and Consequences. Int J Mol Sci. 2021; 22:E408. https://doi.org/10.3390/ijms22010408 [PubMed]
- 15. Calle P, Hotter G. Macrophage Phenotype and Fibrosis in Diabetic Nephropathy. Int J Mol Sci. 2020; 21:E2806. https://doi.org/10.3390/ijms21082806 [PubMed]
- 16. Zeng LF, Xiao Y, Sun L. A Glimpse of the Mechanisms Related to Renal Fibrosis in Diabetic Nephropathy. Adv Exp Med Biol. 2019; 1165:49–79. https://doi.org/10.1007/978-981-13-8871-2_4 [PubMed]
- 17. Zhang H, Zhao T, Li Z, Yan M, Zhao H, Zhu B, Li P. Transcriptional Profile of Kidney from Type 2 Diabetic db/db Mice. J Diabetes Res. 2017; 2017:8391253. https://doi.org/10.1155/2017/8391253 [PubMed]
- 18. Fan CX, Wei M, Zhang DD, Gao QY, Huang HX, Wang JX, Han SY. Effect of D-chiro-inositol on hypoglycemic and liver protection in type 2 diabetic db/db mice and its mechanism. Chin Pharmacol Bull. 2018; 34:1713–18. https://doi.org/10.3969/j.issn.1001-1978.2018.12.018
- 19. Tahamtan M, Sheibani V, Shid Moosavi SM, Asadi-Shekaari M, Esmaeili-Mahani S, Aghaei I, Shabani M. Pre-Treatment with Erythropoietin Attenuates Bilateral Renal Ischemia-Induced Cognitive Impairments. Iran J Pharm Res. 2018; 17:601–12. [PubMed]
- 20. Castelló R, España F, Aznar J, Estellés A. Quantification of Several Components of Fibrinolytic and Matrix Metalloproteinase Systems in Primary. In: Wittwer C, Hahn M, Kaul K, (eds). Rapid Cycle Real-Time PCR — Methods and Applications. Springer, Berlin, Heidelberg. Breast Cancer. 2004; 117–24. https://doi.org/10.1007/978-3-642-18840-4_12
- 21. Monferran S, Paupert J, Dauvillier S, Salles B, Muller C. The membrane form of the DNA repair protein Ku interacts at the cell surface with metalloproteinase 9. EMBO J. 2004; 23:3758–68. https://doi.org/10.1038/sj.emboj.7600403 [PubMed]
- 22. Fakhrudin N, Waltenberger B, Cabaravdic M, Atanasov AG, Malainer C, Schachner D, Heiss EH, Liu R, Noha SM, Grzywacz AM, Mihaly-Bison J, Awad EM, Schuster D, et al. Identification of plumericin as a potent new inhibitor of the NF-κB pathway with anti-inflammatory activity in vitro and in vivo. Br J Pharmacol. 2014; 171:1676–86. https://doi.org/10.1111/bph.12558 [PubMed]
- 23. Renard P, Raes M. The proinflammatory transcription factor NFkappaB: a potential target for novel therapeutical strategies. Cell Biol Toxicol. 1999; 15:341–4. https://doi.org/10.1023/a:1007652414175 [PubMed]
- 24. Zheng L, Szabó C, Kern TS. Poly(ADP-ribose) polymerase is involved in the development of diabetic retinopathy via regulation of nuclear factor-kappaB. Diabetes. 2004; 53:2960–7. https://doi.org/10.2337/diabetes.53.11.2960 [PubMed]
- 25. Kanjanabuch T, Ma LJ, Chen J, Pozzi A, Guan Y, Mundel P, Fogo AB. PPAR-gamma agonist protects podocytes from injury. Kidney Int. 2007; 71:1232–9. https://doi.org/10.1038/sj.ki.5002248 [PubMed]
- 26. Tiwari S, Halagappa VK, Riazi S, Hu X, Ecelbarger CA. Reduced expression of insulin receptors in the kidneys of insulin-resistant rats. J Am Soc Nephrol. 2007; 18:2661–71. https://doi.org/10.1681/ASN.2006121410 [PubMed]
- 27. Yu Y, Du H, Wei S, Feng L, Li J, Yao F, Zhang M, Hatch GM, Chen L. Adipocyte-Derived Exosomal MiR-27a Induces Insulin Resistance in Skeletal Muscle Through Repression of PPARγ. Theranostics. 2018; 8:2171–88. https://doi.org/10.7150/thno.22565 [PubMed]
- 28. Mao JW, Tang HY, Wang YD. Influence of Rosiglitazone on the Expression of PPARγ, NF-κB, and TNF-α in Rat Model of Ulcerative Colitis. Gastroenterol Res Pract. 2012; 2012:845672. https://doi.org/10.1155/2012/845672 [PubMed]

