Introduction
Obesity is a chronic metabolic disease caused by a variety of factors, characterized by disorders of body energy metabolism and excessive fat accumulation. It is closely related to the occurrence of chronic diseases such as type 2 diabetes, hypertension, cardiovascular disease and tumor, and has become a major risk factor threatening human health [1, 2]. The latest data from the World Health Organization (WHO) in 2016 show that more than 1.9 billion adults worldwide are overweight, of which more than 650 million are obese [3]. Obesity has become widespread worldwide and is currently one of the health crises in the world.
Obesity is the result of the interaction of genetic, environmental, and behavioral factors. Family and twin studies have shown that heritability estimates of 40-70% [4], which allows modern molecular genetics to deeply explore the mechanism of the occurrence and development of obesity. DNA methylation is one of the most deeply studied epigenetic modifications, which is highly related to adipocyte differentiation and the occurrence of obesity [5, 6]. It affects the growth and development of adipose tissue by regulating the expression of adipocyte differentiation transcription factors, transcription cofactors, and other adipocytes metabolism-related genes [7].
Cell death-including DFF45-like effector B (CIDEB), located on human chromosome 14q11, can affect gene expression in multiple metabolic pathways and signaling networks, such as lipid droplet formation, lipogenesis, glycolysis, and gluconeogenesis and so on [8–10]. As an important lipid droplet surface protein, CIDEB promotes the lipidation and maturation of very low density lipoprotein (VLDL) by binding to apolipoprotein B and then regulates fat metabolism [11]. The study found that CIDEB-null mice displayed significantly increased body metabolism, which could resist obesity and liver fat degeneration induced by high-fat diet [12]. Compared with normal wild-type mice, CIDEB-null mice have smaller lipid droplets, lower proportion of white adipose tissue (WAT), and significantly lower plasma triglyceride (TG) and fatty acid contents [12, 13]. Therefore, the CIDEB gene plays an important role in maintaining lipid homeostasis and energy metabolism of the whole organism [14].
At present, related studies on CIDEB and lipid metabolism are mostly focused on gene knockout, and most of them are animal experiments on mice. The study on the methylation level of CIDEB gene promoter in obesity has not been reported. The purpose of this study is to explore the relationship between CIDEB and overweight/obesity by comparing the CIDEB gene promoter methylation level in abdominal subcutaneous adipose tissue (SAT) and omental adipose tissue (OAT) between normal-weight and overweight/obesity groups, as to provide a theoretical basis for gene-targeted therapy of obesity.
Materials and Methods
Materials
Subjects
According to the inclusion and exclusion criteria, 61 patients with abdominal surgery from a municipal hospital of Henan province in China were selected, with an average age of 51.87±14 years, including 10 males and 51 females. The case data of the subjects were collected, including the patient's height, weight, waist circumference (WC), hip circumference, blood pressure, fasting plasma glucose (FPG), blood lipids, and other indicators. According to the Chinese adult obesity diagnostic criteria [15], the subjects were divided into two groups: 28 cases of normal-weight group (18.5kg/m2 ≤ BMI < 24kg/m2) and 33 cases of overweight/obesity group (BMI ≥ 24.0kg/m2). The study was approved by the ethics committee of Zhengzhou University.
Inclusion criteria
(1) Adults to undergo abdominal surgery, such as appendicitis surgery, abdominal external hernia surgery, gallstone surgery; (2) The patient is mentally normal, without consciousness disorder, and can communicate fluently; (3) The patient voluntarily participates and signs an informed consent.
Exclusion criteria
(1) Pregnant women, patients with disabilities or mental disorders, secondary obesity, malignant tumors; (2) Patients with infectious diseases such as hepatitis B, AIDS, and tuberculosis; (3) Patients taking adrenergic receptor blockers, antidepressants, or psychoactive drugs.
Methods
DNA extraction
During the operation, 1-2cm2 of abdominal SAT and OAT were taken out from the subjects, immediately washed with sterile saline and stored in liquid nitrogen.
Methylation detection of CIDEB gene promoter
DNA extraction kit (TIANGEN, Beijing, China) was used to extract DNA, and the CIDEB promoter methylation level was detected by the MethylTarget method. The methylation detection was assisted by Genesky Biotechnologies Inc., Shanghai. Specific steps are as follows: (1) Agarose gel electrophoresis was used to detect the integrity of genomic DNA, and then Nanodrop 2000 (NanoDrop Technologies, Wilmington, DE, USA) was used to detect the quality of genomic DNA. (2) Design and optimization of primers. (3) Panel optimization of multiplex PCR primers: The primers optimized by step (2) were mixed into multiple PCR primers panel, and then the composition and concentration of primers in multiplex PCR panel were optimized by capillary electrophoresis. (4) Bisulfite treatment: Sample processing was performed using EZ DNA Methylation-Gold Kit (Zymo, Irvine, CA, USA) and convert cytosine C, which has not been methylated by genomic DNA, into uracil U. (5) Multiple PCR response of sample target fragment: The optimized multiplex PCR primer panel was used for multiplex PCR amplification with the transformed sample genome as the template. After quality control, the amplified products of all multiplex PCR primer panels with the same sample genomic DNA as template were mixed, and the amount of amplified products of each primer site was ensured to be equivalent. (6) Adding specific tag sequence to the sample: Using the primers with Index sequence, the specific tag sequence compatible with the Illumina platform (Illumina, San Diego, CA, USA) was introduced to the end of the library by PCR amplification. (7) High throughput sequencing: The Index PCR products of all samples were mixed equally to obtain the final MethylTarget sequencing library, whose fragment length distribution was verified by Agilent 2100 bioAnalyzer (Agilent Technologies, Santa Clara, CA, USA). After accurate quantification of the library molar concentration, the high-throughput sequencing was carried out on the Illumina Hiseq platform (Illumina, San Diego, CA, USA) with a 2×150bp double-terminal sequencing mode.
Statistical analysis
The data were analyzed with IBM SPSS Statistics 23.0 (IBM Corp, Armonk, NY, USA). Basic characteristics of the subjects were described with mean and standard deviation
Availability of data and materials
The datasets generated and/or analyzed during the current study are available.
Ethics approval
The study was approved by the ethics committee of Zhengzhou University.
Results
The characteristics of subjects
According to the inclusion and exclusion criteria, 61 adult patients were included, of which both abdominal SAT and OAT were collected from 42 patients, and only one type of adipose tissue was collected from 19 patients. The average age of subjects was 51.87 years old. There were 28 cases in the normal-weight group, including 5 males and 23 females; 33 cases in the overweight/obesity group, including 5 males and 28 females. There was no statistical difference in gender distribution between two groups (F0632=0.081, P=0.776). The results of two independent sample t test showed that weight and BMI of overweight/obesity group were higher than normal-weight group (P<0.05), but there was no statistical difference in indicators such as age, height, waist-to-hip ratio, FPG, total cholesterol (TC), total triglyceride (TG), high density lipoprotein (HDL), low density lipoprotein (LDL) (P>0.05). Details were shown in Table 1.
Table 1. Comparison of general information between different BMI groups
| Index | Normal-weight group | Overweight/obesity group | t | P | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (n=28) | (n=33) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 54.33±16.54 | 49.85±11.38 | 1.241 | 0.220 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Height (cm) | 160.65±6.34 | 161.65±6.00 | -0.628 | 0.532 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Weight (kg) | 53.63±6.52 | 70.77±8.28 | -7.225 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI (kg/m2) | 21.91±1.42 | 27.05±2.50 | -9.638 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WHR | 0.90±0.05 | 0.92±0.07 | -0.849 | 0.399 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SBP (mmHg) | 123.56±15.99 | 124.39±12.84 | -0.225 | 0.823 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DBP (mmHg) | 74.22±9.56 | 74.27±7.60 | -0.023 | 0.982 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FPG (mmol/L) | 5.30±1.31 | 6.45±4.13 | -1.501 | 0.141 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TC (mmol/L) | 4.23±1.14 | 4.59±0.98 | -1.324 | 0.191 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TG (mmol/L) | 1.59±1.36 | 2.24±1.64 | -1.665 | 0.101 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HDL (mmol/L) | 1.06±0.31 | 1.10±0.26 | -0.543 | 0.589 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDL (mmol/L) | 2.55±1.25 | 2.27±0.94 | 1.004 | 0.320 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI, body mass index; WHR, waist-hip ratio; SBP, systolic blood pressure; DBP, diastolic blood pressure; FPG, fasting plasma glucose; TC, total cholesterol; TG, total triglyceride; HDL, high density lipoprotein; LDL, low density lipoprotein. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
CIDEB gene promoter CpG methylation sites and the methylation level heatmap
In this study, 16 CpG methylation sites in CIDEB gene (Chr14: 24780614 to Chr14: 24780744) were detected. The various CpG sites and their corresponding physical locations in the chromosome were shown in Table 2. The methylation heatmap of CpG sites was shown in Figure 1.
Table 2. CpG sites of CIDEB gene promoter and their corresponding physical locations in the chromosome.
| CpG sites | Locations in chromosome | CpG sites | Locations in chromosome |
| 30 | Chr14: 24780744 | 100 | Chr14: 24780674 |
| 39 | Chr14: 24780735 | 108 | Chr14: 24780666 |
| 49 | Chr14: 24780725 | 114 | Chr14: 24780660 |
| 57 | Chr14: 24780717 | 124 | Chr14: 24780650 |
| 82 | Chr14: 24780692 | 139 | Chr14: 24780635 |
| 91 | Chr14: 24780683 | 151 | Chr14: 24780623 |
| 95 | Chr14: 24780679 | 156 | Chr14: 24780618 |
| 97 | Chr14: 24780677 | 160 | Chr14: 24780614 |
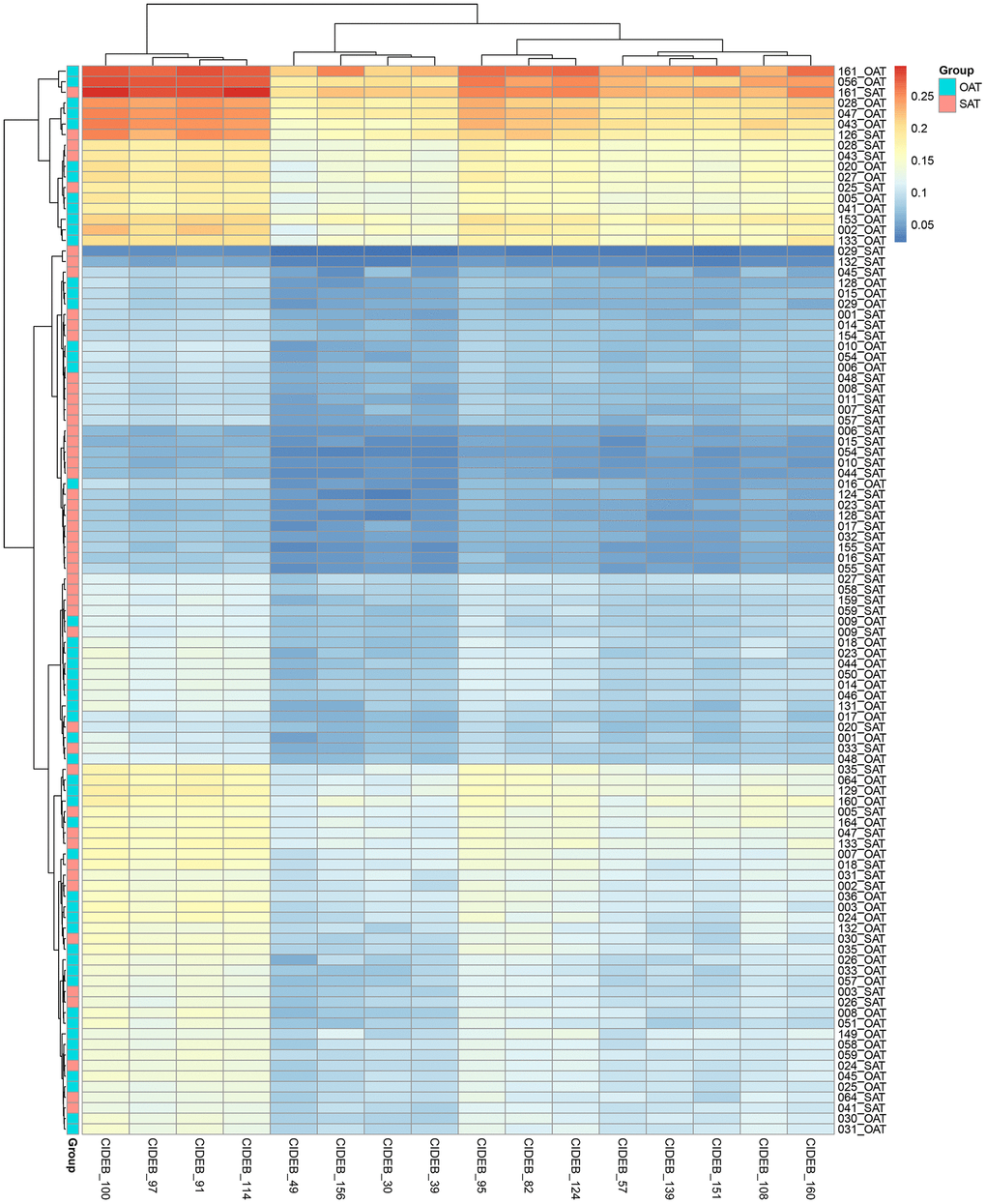
Figure 1. Methylation heatmap of CIDE-B gene promoter CpG sites.
Comparison of the methylation level of CIDEB gene promoter CpG sites between normal-weight and overweight/obesity groups
The results of two independent sample t test showed that there was no significant difference in CpG site methylation level between normal-weight and overweight/obesity groups (P>0.05). Details were shown in Table 3.
Table 3. Comparison of CIDEB gene promoter methylation level between different BMI groups
| Adipose tissue | Methylation sites | Normal-weight group | Overweight/obesity group | P | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| n | n | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SAT | Chr14: 24780744 | 23 | 0.075±0.044 | 26 | 0.088±0.033 | 0.240 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780735 | 23 | 0.071±0.043 | 26 | 0.083±0.035 | 0.314 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780725 | 23 | 0.069±0.039 | 26 | 0.078±0.031 | 0.325 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780717 | 23 | 0.082±0.045 | 26 | 0.093±0.035 | 0.332 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780692 | 23 | 0.091±0.049 | 26 | 0.105±0.039 | 0.280 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780683 | 23 | 0.110±0.053 | 26 | 0.124±0.044 | 0.337 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780679 | 23 | 0.098±0.049 | 26 | 0.111±0.040 | 0.308 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780677 | 23 | 0.106±0.052 | 26 | 0.117±0.042 | 0.405 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780674 | 23 | 0.114±0.055 | 26 | 0.127±0.045 | 0.393 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780666 | 23 | 0.087±0.042 | 26 | 0.099±0.036 | 0.314 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780660 | 23 | 0.110±0.055 | 26 | 0.120±0.044 | 0.482 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780650 | 23 | 0.091±0.049 | 26 | 0.104±0.039 | 0.310 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780635 | 23 | 0.080±0.043 | 26 | 0.092±0.034 | 0.268 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780623 | 23 | 0.077±0.045 | 26 | 0.093±0.034 | 0.155 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780618 | 23 | 0.071±0.045 | 26 | 0.084±0.032 | 0.243 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780614 | 23 | 0.085±0.049 | 26 | 0.098±0.035 | 0.305 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OAT | Chr14: 24780744 | 25 | 0.096±0.037 | 27 | 0.109±0.039 | 0.247 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780735 | 25 | 0.100±0.040 | 27 | 0.107±0.039 | 0.489 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780725 | 25 | 0.087±0.038 | 27 | 0.096±0.037 | 0.426 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780717 | 25 | 0.109±0.042 | 27 | 0.115±0.041 | 0.626 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780692 | 25 | 0.126±0.046 | 27 | 0.134±0.044 | 0.516 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780683 | 25 | 0.149±0.047 | 27 | 0.157±0.047 | 0.533 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780679 | 25 | 0.133±0.046 | 27 | 0.142±0.046 | 0.473 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780677 | 25 | 0.143±0.047 | 27 | 0.150±0.047 | 0.574 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780674 | 25 | 0.155±0.046 | 27 | 0.161±0.048 | 0.680 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780666 | 25 | 0.117±0.041 | 27 | 0.125±0.042 | 0.496 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780660 | 25 | 0.145±0.046 | 27 | 0.154±0.047 | 0.461 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780650 | 25 | 0.122±0.046 | 27 | 0.132±0.046 | 0.428 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780635 | 25 | 0.109±0.044 | 27 | 0.119±0.041 | 0.430 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780623 | 25 | 0.105±0.047 | 27 | 0.115±0.040 | 0.400 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780618 | 25 | 0.097±0.046 | 27 | 0.108±0.041 | 0.360 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780614 | 25 | 0.115±0.048 | 27 | 0.126±0.046 | 0.431 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SAT, subcutaneous adipose tissue; OAT, omental adipose tissue. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Comparison of the methylation level of CIDEB gene promoter between SAT and OAT
The results of paired t test showed that 16 CpG sites detected were statistically different between two groups, and the methylation level of these CpG sites in the OAT was higher than that in the SAT (Table 4).
Table 4. CIDEB gene methylation sites different statistically between SAT and OAT
| Methylation sites | SAT | OAT | t | P | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780744 | 0.083±0.039 | 0.100±0.038 | 4.385 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780735 | 0.079±0.037 | 0.102±0.041 | 5.564 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780725 | 0.076±0.035 | 0.089±0.038 | 3.878 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780717 | 0.089±0.040 | 0.110±0.041 | 4.836 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780692 | 0.099±0.043 | 0.127±0.046 | 5.994 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780683 | 0.119±0.047 | 0.151±0.047 | 6.423 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780679 | 0.107±0.044 | 0.135±0.046 | 6.484 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780677 | 0.114±0.046 | 0.144±0.046 | 6.299 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780674 | 0.122±0.049 | 0.155±0.047 | 6.398 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780666 | 0.095±0.038 | 0.118±0.040 | 5.794 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780660 | 0.117±0.048 | 0.147±0.047 | 5.928 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780650 | 0.099±0.044 | 0.124±0.046 | 5.882 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780635 | 0.088±0.038 | 0.112±0.042 | 6.149 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780623 | 0.087±0.040 | 0.108±0.044 | 5.207 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780618 | 0.080±0.039 | 0.100±0.044 | 5.147 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chr14: 24780614 | 0.094±0.042 | 0.118±0.047 | 5.772 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SAT, subcutaneous adipose tissue; OAT, omental adipose tissue. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Comparison of CIDEB gene methylation haplotypes between normal-weight group and overweight/obesity group
Table 5 showed that there were 3 haplotypes with statistical differences between normal-weight and overweight/obesity groups, including 2 in SAT and 1 in OAT.
Table 5. Haplotypes of CIDEB gene promoter methylation different statistically between different BMI groups.
| Adipose tissue | Haplotype | Normal-weight group (n = 23) | Overweight/obesity group (n = 26) | P | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SAT | ttttttttttcttttt | 0.010±0.003 | 0.008±0.002 | 0.011 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ttttttttttttttct | 0.006±0.002 | 0.005±0.001 | 0.016 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OATa | ttttttttcttttttt | 0.010±0.002 | 0.008±0.002 | 0.027 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SAT, subcutaneous adipose tissue; OAT, omental adipose tissue. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| aOAT samples in normal-weight group and overweight/obesity group are 25,27 respectively. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Discussion
In recent years, the role of epigenetics has attracted more and more attention in the process of studying the pathogenesis of diseases. DNA methylation is a well-studied epigenetic modification, which only affects the transcription activity of genes and does not change the DNA sequence. A genome-wide analysis found that methylation levels of some genes were associated with different somatotypes and body composition of preschool children [16]. The previous study of our group showed that methylation level of PRDM16 gene promoter was higher in overweight/obese people [17]. In a large-scale study investigating DNA methylation in CD4+ T-cells [18], 8 CpG sites were associated with BMI and five with WC. Therefore, DNA methylation can regulate the growth and development of adipose tissue, which plays an important role in the occurrence and development of metabolic diseases related to obesity [19, 20].
There are three members of the CIDE protein family, including CIDEA, CIDEB, and CIDEC/fat-specific protein 27 (FSP27, in rodent), which were originally thought to be involved in apoptosis in mammals [21]. Along with the deepening of the study, it has been discovered that CIDE proteins are key factors in controlling multiple lipid metabolism pathways and maintaining lipid homeostasis [22], and are closely related to the occurrence and development of obesity, diabetes, and fatty liver [9, 23, 24]. Whether in brown or white adipocytes, the lack of CIDE family proteins can cause the lipid droplets in these cells to decrease in size, increase in number, and accelerate fat degradation [22, 25].
Our study found that methylation value of each CpG site of CIDEB gene in overweight/obesity group was higher than that of normal-weight group, but there was no statistical difference. Possible reasons are as follows: (1) Due to human subjects, it is difficult to obtain adipose tissue, resulting in a small sample size; (2) Obesity is a complex disease, and its occurrence is the result of the interaction of multiple genes [26, 27], so methylation status of a single gene promoter sequence may not be significant. Therefore, the combined study of multiple gene promoter methylation may be more helpful to explain the molecular mechanism of the occurrence and development of obesity, and DNA methylation can be explored at the genome-wide level in future studies.
This is the first study of the CIDEB gene methylation level in adipose tissue. We found that methylation levels of the CIDEB promoter in OAT were higher than that in SAT, indicating that expression of CIDEB in SAT was more than that of OAT, which was consistent with the fact that subcutaneous fat storage is higher than that of OAT. According to functional and anatomical differences, WAT is divided into SAT and OAT [28]. SAT is the largest fat storage tissue. When the energy storage of SAT is saturated, fat is ectopically stored in viscera [29]. Therefore, higher level of CIDEB expression in the SAT made white fat accumulate preferentially in the subcutaneous layer.
In terms of genetics, chronic non-communicable diseases are caused by the interaction of multiple genes or mutations within genes, rather than the isolated effect of a single polymorphic site. Haplotype refers to the combination of alleles at multiple sites that are inherited together on the same chromosome. Haplotype analysis is an accurate and reliable method for discovering the association between genome structure and diseases [30]. We found that 3 haplotypes were related to overweight or obesity, which indicated that regulation of CIDEB expression is not a single-site mutation, but a combination of multiple-site mutations.
This study is the first to report the association of the CIDEB gene promoter methylation level with overweight or obesity in abdominal adipose tissue of adults, which is a significant exploration to elucidate the epigenetic mechanism of obesity development. Moreover, the MethylTarget technology we used can accurately calculate the methylation level of each CpG site, which has the advantages of high accuracy, strong flexibility, and high-cost performance.
The relatively small sample size was a limitation of this study. Following the principle of informed consent, human tissue sampling is inconvenient, and it is indeed difficult to obtain both SAT and OAT. Moreover, due to the lack of relevant studies on methylation of CIDEB gene at present, the sample content cannot be determined by literature method. Through literature review, we found that the sample size of DNA methylation studies involving human tissues was generally small, while the sample size of studies obtaining both types of adipose tissue was even smaller. For example, a Spanish study [31] collected visceral adipose tissues (VAT) from 57 patients with colorectal cancer and 50 healthy controls to explore vitamin D receptor (VDR) expression and methylation in colorectal cancer. Helene A. Fachim et al. [32] performed SAT biopsy in 20 patients with impaired glucose regulation to evaluate the effect of lifestyle intervention on Caveolin-1 gene methylation and provided insights for targeted treatment of diabetes. To investigate the relationship between obesity and insulin resistance, Aneta Cierzniak et al. [33] collected VAT and SAT from 45 patients undergoing abdominal surgery. Tarryn Willmer et al. [34] obtained abdominal SAT and gluteal SAT from 27 obese and 27 normal weight urban-dwelling South African women and found that the methylation level of FK506-binding protein 51 kDa (FKBP5) gene was associated with obesity and insulin resistance. In conclusion, the sample size of this study is moderate, and the results of this study have a certain reference value, which can provide a theoretical basis for finding targets for the treatment of obesity.
Conclusions
The methylation level of CIDEB gene promoter in abdominal SAT and OAT may be related to overweight or obesity in adults, and the specific regulatory mechanism needs to be further studied.
Author Contributions
Zhiguang Ping contributed to the study design. Yanzi Chen carried out experiments. Ming Lu collected clinical specimens. Zhaoyan Guo analyzed the data. Zhiguang Ping and Zhaoyan Guo drafted the manuscript. Li Liu contributed to the interpretation and discussion of the manuscript. Li Liu and Zhiguang Ping contributed to review/edit the manuscript. All authors read and approved the final manuscript.
Acknowledgments
We thank all the subjects for their important contribution in this study.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This work was supported by the Natural Science Foundation of Henan Province (Grant number 182300410303); National natural science foundations of China (Grant number 81001280); National natural science foundations of China (Grant number 81202277).
References
- 1. Iyengar NM, Gucalp A, Dannenberg AJ, Hudis CA. Obesity and Cancer Mechanisms: Tumor Microenvironment and Inflammation. J Clin Oncol. 2016; 34:4270–6. https://doi.org/10.1200/JCO.2016.67.4283 [PubMed]
- 2. Nimptsch K, Konigorski S, Pischon T. Diagnosis of obesity and use of obesity biomarkers in science and clinical medicine. Metabolism. 2019; 92:61–70. https://doi.org/10.1016/j.metabol.2018.12.006 [PubMed]
- 3. World Health Organization. https://www.who.int/en/news-room/fact-sheets/detail/obesity-and-overweight. 2021.
- 4. Stryjecki C, Alyass A, Meyre D. Ethnic and population differences in the genetic predisposition to human obesity. Obes Rev. 2018; 19:62–80. https://doi.org/10.1111/obr.12604 [PubMed]
- 5. Kamei Y, Suganami T, Ehara T, Kanai S, Hayashi K, Yamamoto Y, Miura S, Ezaki O, Okano M, Ogawa Y. Increased expression of DNA methyltransferase 3a in obese adipose tissue: studies with transgenic mice. Obesity (Silver Spring). 2010; 18:314–21. https://doi.org/10.1038/oby.2009.246 [PubMed]
- 6. Xu X, Su S, Barnes VA, De Miguel C, Pollock J, Ownby D, Shi H, Zhu H, Snieder H, Wang X. A genome-wide methylation study on obesity: differential variability and differential methylation. Epigenetics. 2013; 8:522–33. https://doi.org/10.4161/epi.24506 [PubMed]
- 7. D’Alessio AC, Weaver IC, Szyf M. Acetylation-induced transcription is required for active DNA demethylation in methylation-silenced genes. Mol Cell Biol. 2007; 27:7462–74. https://doi.org/10.1128/MCB.01120-07 [PubMed]
- 8. Gong J, Sun Z, Li P. CIDE proteins and metabolic disorders. Curr Opin Lipidol. 2009; 20:121–6. https://doi.org/10.1097/MOL.0b013e328328d0bb [PubMed]
- 9. Chen FJ, Yin Y, Chua BT, Li P. CIDE family proteins control lipid homeostasis and the development of metabolic diseases. Traffic. 2020; 21:94–105. https://doi.org/10.1111/tra.12717 [PubMed]
- 10. Xu W, Wu L, Yu M, Chen FJ, Arshad M, Xia X, Ren H, Yu J, Xu L, Xu D, Li JZ, Li P, Zhou L. Differential Roles of Cell Death-inducing DNA Fragmentation Factor-α-like Effector (CIDE) Proteins in Promoting Lipid Droplet Fusion and Growth in Subpopulations of Hepatocytes. J Biol Chem. 2016; 291:4282–93. https://doi.org/10.1074/jbc.M115.701094 [PubMed]
- 11. Ye J, Li JZ, Liu Y, Li X, Yang T, Ma X, Li Q, Yao Z, Li P. Cideb, an ER- and lipid droplet-associated protein, mediates VLDL lipidation and maturation by interacting with apolipoprotein B. Cell Metab. 2009; 9:177–90. https://doi.org/10.1016/j.cmet.2008.12.013 [PubMed]
- 12. Li JZ, Ye J, Xue B, Qi J, Zhang J, Zhou Z, Li Q, Wen Z, Li P. Cideb regulates diet-induced obesity, liver steatosis, and insulin sensitivity by controlling lipogenesis and fatty acid oxidation. Diabetes. 2007; 56:2523–32. https://doi.org/10.2337/db07-0040 [PubMed]
- 13. Li JZ, Lei Y, Wang Y, Zhang Y, Ye J, Xia X, Pan X, Li P. Control of cholesterol biosynthesis, uptake and storage in hepatocytes by Cideb. Biochim Biophys Acta. 2010; 1801:577–86. https://doi.org/10.1016/j.bbalip.2010.01.012 [PubMed]
- 14. Gao G, Chen FJ, Zhou L, Su L, Xu D, Xu L, Li P. Control of lipid droplet fusion and growth by CIDE family proteins. Biochim Biophys Acta Mol Cell Biol Lipids. 2017; 1862:1197–204. https://doi.org/10.1016/j.bbalip.2017.06.009 [PubMed]
- 15. China WGoOi. Guidelines for the prevention and control of overweight and obesity in China (excerpt). Acta Nutr Sin 2004; 26:1–4.
- 16. Rzehak P, Covic M, Saffery R, Reischl E, Wahl S, Grote V, Weber M, Xhonneux A, Langhendries JP, Ferre N, Closa-Monasterolo R, Escribano J, Verduci E, et al. DNA-Methylation and Body Composition in Preschool Children: Epigenome-Wide-Analysis in the European Childhood Obesity Project (CHOP)-Study. Sci Rep. 2017; 7:14349. https://doi.org/10.1038/s41598-017-13099-4 [PubMed]
- 17. Liu L, Chen Y, Chen J, Lu M, Guo R, Han J, Zhang Y, Pei X, Ping Z. The relationship between PRDM16 promoter methylation in abdominal subcutaneous and omental adipose tissue and obesity. Clin Nutr. 2021; 40:2278–84. https://doi.org/10.1016/j.clnu.2020.10.016 [PubMed]
- 18. Aslibekyan S, Demerath EW, Mendelson M, Zhi D, Guan W, Liang L, Sha J, Pankow JS, Liu C, Irvin MR, Fornage M, Hidalgo B, Lin LA, et al. Epigenome-wide study identifies novel methylation loci associated with body mass index and waist circumference. Obesity (Silver Spring). 2015; 23:1493–501. https://doi.org/10.1002/oby.21111 [PubMed]
- 19. Ling C, Rönn T. Epigenetics in Human Obesity and Type 2 Diabetes. Cell Metab. 2019; 29:1028–44. https://doi.org/10.1016/j.cmet.2019.03.009 [PubMed]
- 20. Samblas M, Milagro FI, Martínez A. DNA methylation markers in obesity, metabolic syndrome, and weight loss. Epigenetics. 2019; 14:421–44. https://doi.org/10.1080/15592294.2019.1595297 [PubMed]
- 21. Park HH. Structural insight into CIDE domains: the Janus face of CIDEs. Apoptosis. 2015; 20:240–9. https://doi.org/10.1007/s10495-014-1067-z [PubMed]
- 22. Xu L, Zhou L, Li P. CIDE proteins and lipid metabolism. Arterioscler Thromb Vasc Biol. 2012; 32:1094–8. https://doi.org/10.1161/ATVBAHA.111.241489 [PubMed]
- 23. Slayton M, Gupta A, Balakrishnan B, Puri V. CIDE Proteins in Human Health and Disease. Cells. 2019; 8:238. https://doi.org/10.3390/cells8030238 [PubMed]
- 24. Zhou L, Yu M, Arshad M, Wang W, Lu Y, Gong J, Gu Y, Li P, Xu L. Coordination Among Lipid Droplets, Peroxisomes, and Mitochondria Regulates Energy Expenditure Through the CIDE-ATGL-PPARα Pathway in Adipocytes. Diabetes. 2018; 67:1935–48. https://doi.org/10.2337/db17-1452 [PubMed]
- 25. Nishino N, Tamori Y, Tateya S, Kawaguchi T, Shibakusa T, Mizunoya W, Inoue K, Kitazawa R, Kitazawa S, Matsuki Y, Hiramatsu R, Masubuchi S, Omachi A, et al. FSP27 contributes to efficient energy storage in murine white adipocytes by promoting the formation of unilocular lipid droplets. J Clin Invest. 2008; 118:2808–21. https://doi.org/10.1172/JCI34090 [PubMed]
- 26. Rankinen T, Zuberi A, Chagnon YC, Weisnagel SJ, Argyropoulos G, Walts B, Pérusse L, Bouchard C. The human obesity gene map: the 2005 update. Obesity (Silver Spring). 2006; 14:529–644. https://doi.org/10.1038/oby.2006.71 [PubMed]
- 27. Sayols-Baixeras S, Subirana I, Fernández-Sanlés A, Sentí M, Lluís-Ganella C, Marrugat J, Elosua R. DNA methylation and obesity traits: An epigenome-wide association study. The REGICOR study. Epigenetics. 2017; 12:909–16. https://doi.org/10.1080/15592294.2017.1363951 [PubMed]
- 28. Jové M, Moreno-Navarrete JM, Pamplona R, Ricart W, Portero-Otín M, Fernández-Real JM. Human omental and subcutaneous adipose tissue exhibit specific lipidomic signatures. FASEB J. 2014; 28:1071–81. https://doi.org/10.1096/fj.13-234419 [PubMed]
- 29. Le Jemtel TH, Samson R, Milligan G, Jaiswal A, Oparil S. Visceral Adipose Tissue Accumulation and Residual Cardiovascular Risk. Curr Hypertens Rep. 2018; 20:77. https://doi.org/10.1007/s11906-018-0880-0 [PubMed]
- 30. Gabriel SB, Schaffner SF, Nguyen H, Moore JM, Roy J, Blumenstiel B, Higgins J, DeFelice M, Lochner A, Faggart M, Liu-Cordero SN, Rotimi C, Adeyemo A, et al. The structure of haplotype blocks in the human genome. Science. 2002; 296:2225–9. https://doi.org/10.1126/science.1069424 [PubMed]
- 31. Castellano-Castillo D, Morcillo S, Clemente-Postigo M, Crujeiras AB, Fernandez-García JC, Torres E, Tinahones FJ, Macias-Gonzalez M. Adipose tissue inflammation and VDR expression and methylation in colorectal cancer. Clin Epigenetics. 2018; 10:60. https://doi.org/10.1186/s13148-018-0493-0 [PubMed]
- 32. Fachim HA, Siddals K, Malipatil N, Donn RP, Moreno GY, Dalton CF, Adam S, Soran H, Gibson JM, Heald AH. Lifestyle intervention in individuals with impaired glucose regulation affects Caveolin-1 expression and DNA methylation. Adipocyte. 2020; 9:96–107. https://doi.org/10.1080/21623945.2020.1732513 [PubMed]
- 33. Cierzniak A, Pawelka D, Kaliszewski K, Rudnicki J, Dobosz T, Malodobra-Mazur M. DNA methylation in adipocytes from visceral and subcutaneous adipose tissue influences insulin-signaling gene expression in obese individuals. Int J Obes (Lond). 2021; 45:650–8. https://doi.org/10.1038/s41366-020-00729-7 [PubMed]
- 34. Willmer T, Goedecke JH, Dias S, Louw J, Pheiffer C. DNA methylation of FKBP5 in South African women: associations with obesity and insulin resistance. Clin Epigenetics. 2020; 12:141. https://doi.org/10.1186/s13148-020-00932-3 [PubMed]



