Introduction
End-stage renal disease (ESRD) is a dialysis-dependent status associated with a significant socioeconomic burden, and increasing age serves as a risk factor for mortality after dialysis initiation [1]. Every year, around 2.6 million people received renal replacement therapy due to ESRD worldwide [2], and Taiwan reportedly has a high prevalence of chronic kidney disease (15.46%) [3], along with the highest prevalence (0.33%) of treated ESRD in the world [4]. Among the etiologies of renal injury, drug-induced renal disorders play a major role, accounting for 18–27% of renal injuries [5].
Among the wide varieties of drugs that pose a threat to renal function, anti-vascular endothelial growth factor (VEGF) medications are often overlooked. Anti-VEGFs are widely used to prevent vascular proliferation in tumors and retinal diseases [6]. Anti-VEGF is extensively applied in the field of ophthalmology for indications that include exudative age-related macular degeneration (AMD), diabetic macular edema (DME), polypoidal choroidal vasculopathy (PCV), retinal vein occlusions (RVO), myopic choroid neovascularization (mCNV) and retinopathy of prematurity [7–9].
As the number of intravitreal anti-VEGF treatments has expanded in recent years [10–12], ophthalmologists are becoming aware of the associated adverse effects. Previous reports showed that hypertension is one of the adverse effects [13, 14], and risks of mortality and cerebrovascular accidents were found to be elevated in patients with poorer overall conditions [15, 16].
In vitro and animal studies have demonstrated that intravitreal anti-VEGF upregulates the inflammation in the kidney and retina [17], but current real-world evidence regarding renal complications after intravitreal anti-VEGF treatments remains scarce, and only case reports have demonstrated a possible correlation between intravitreal anti-VEGFs and kidney injuries [18–24]. Hence, by utilizing the National Health Insurance Research Database (NHIRD), a nationwide population-based dataset in Taiwan, a retrospective cohort study was conducted to investigate the risk of becoming dialysis-dependent after the administration of intravitreal anti-VEGFs. We hypothesized that the use of intravitreal anti-VEGFs was associated with increased risk of becoming dialysis dependent, compared with the control group.
Results
Baseline characteristics of the study cohort
A total of 2484 subjects who received intravitreal injections of anti-VEGF were included in the study group, and another 9936 subjects without anti-VEGF matched by age and sex served as the control group. In addition, 2447 patients in the anti-VEGF group were matched with 2447 patients that did not receive anti-VEGF therapy using propensity score matching (PSM) (Figure 1). The differences in the baseline characteristics between the anti-VEGF and control groups were summarized in Table 1. In the cohort matched by age and sex, the patients in the anti-VEGF group were significantly more likely to have diabetes mellitus. After PSM, indications for anti-VEGF injection and comorbidities were similarly distributed in the two groups. In both cohorts (age- and sex-matched and PSM), most indications for anti-VEGF were AMD/PCV (47%) and DME (42%), and approximately 75% of anti-VEGF-treated patients received ranibizumab treatment.
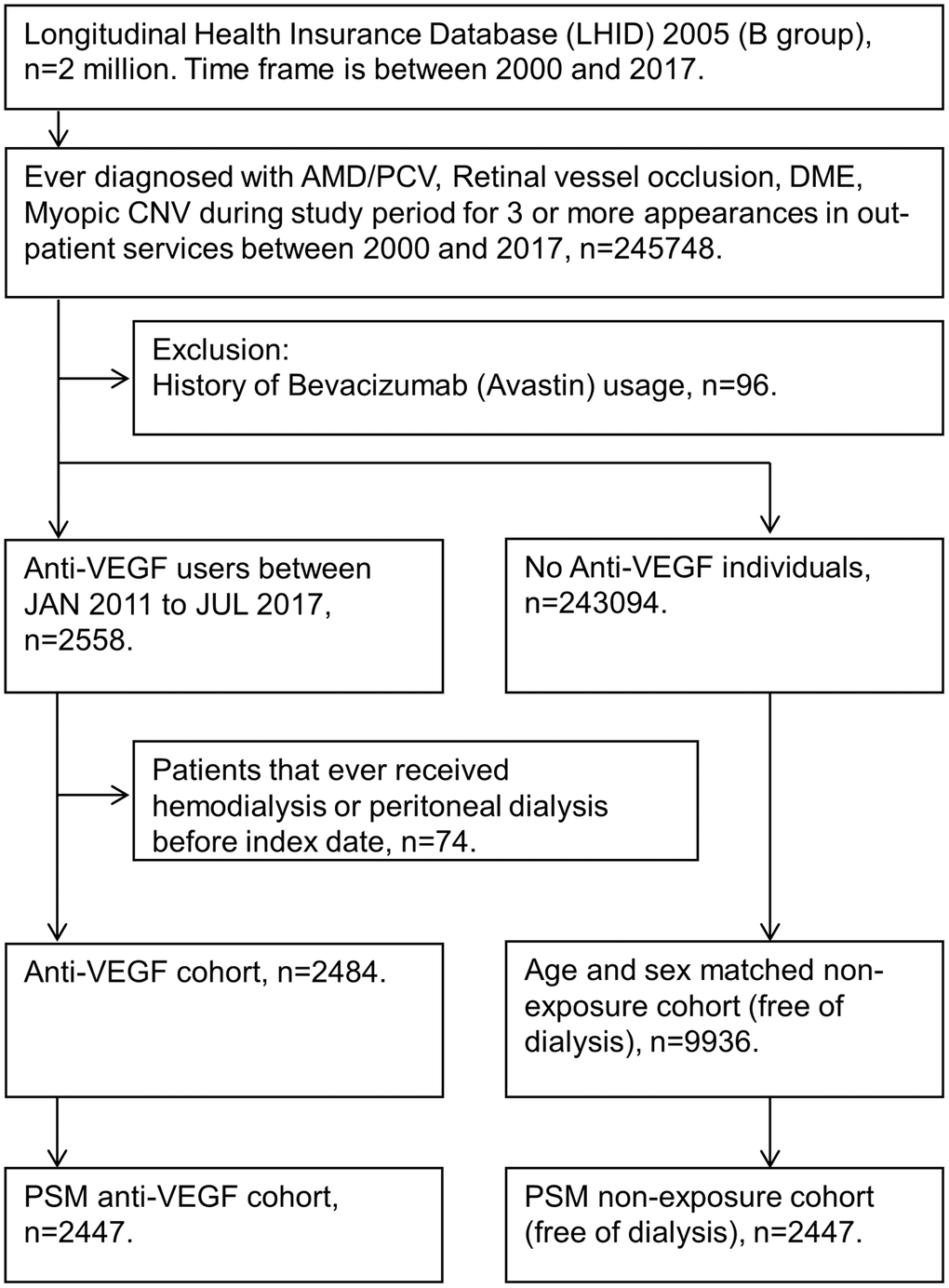
Figure 1. Flow diagram showing study participant selection. For patients who received intravitreal anti-vascular endothelial growth factor (VEGF) treatment, the index date was the day of the first intravitreal anti-vascular endothelial growth factor injection. For patients who did not receive intravitreal anti-vascular endothelial growth factor treatment, the index date was nested with the paired anti-VEGF patients. All study participants were at risk on the index date. Abbreviations: AMD: age-related macular degeneration; B group: 2005 Longitudinal Health Insurance Databases; CNV: choroidal neovascularization; DME: diabetic macular edema; PCV: polypoidal choroidal vasculopathy; PSM: propensity Score Matching; VEGF: vascular endothelial growth factor.
Table 1. Baseline characteristics.
| Variable | Age-and sex-matched | PSMa | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No anti-VEGF | Anti-VEGF | ASDb | No anti-VEGF | Anti-VEGF | ASD | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N = 9936 | N = 2484 | N = 2447 | N = 2447 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Year of index | 0.0000 | 0.0231 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2011–2013 | 2440 (24.56%) | 610 (24.56%) | 616 (25.17%) | 602 (24.60%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2014–2015 | 3728 (37.52%) | 932 (37.52%) | 907 (37.07%) | 918 (37.52%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2016–2017 | 3768 (37.92%) | 942 (37.92%) | 924 (37.76%) | 927 (37.88%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex | 0.0000 | 0.0117 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male | 5948 (59.86%) | 1487 (59.86%) | 1479 (60.44%) | 1465 (59.87%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female | 3988 (40.14%) | 997 (40.14%) | 968 (39.56%) | 982 (40.13%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age at index | 0.0000 | 0.0277 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 20–40 | 185 (1.86%) | 46 (1.85%) | 24 (0.98%) | 32 (1.31%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 40–60 | 2166 (21.80%) | 537 (21.62%) | 511 (20.88%) | 520 (21.25%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 60–80 | 5979 (60.18%) | 1501 (60.43%) | 1514 (61.87%) | 1495 (61.10%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 80–100 | 1606 (16.16%) | 400 (16.10%) | 398 (16.26%) | 400 (16.35%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Indication | 0.4971 | 0.0000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AMD/PCV | 3011 (30.30%) | 1176 (47.34%) | 1161 (47.45%) | 1151 (47.04%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| RVO | 687 (6.91%) | 129 (5.19%) | 120 (4.90%) | 129 (5.27%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DME | 4245 (42.72%) | 1052 (42.35%) | 1043 (42.62%) | 1040 (42.50%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Myopic CNV | 1993 (20.06%) | 127 (5.11%) | 123 (5.03%) | 127 (5.19%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Urbanization | 0.0226 | 0.0751 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Urban | 6211 (62.51%) | 1545 (62.20%) | 1555 (63.55%) | 1523 (62.24%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sub-urban | 2808 (28.26%) | 719 (28.95%) | 699 (28.57%) | 709 (28.97%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rural | 917 (9.23%) | 220 (8.86%) | 193 (7.89%) | 215 (8.79%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Insured unit type | 0.0937 | 0.0661 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Government | 819 (8.24%) | 177 (7.13%) | 166 (6.78%) | 176 (7.19%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Privately held company | 5105 (51.38%) | 1315 (52.94%) | 1323 (54.07%) | 1288 (52.64%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Agricultural organizations | 1873 (18.85%) | 470 (18.92%) | 450 (18.39%) | 467 (19.08%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Low-income | 59 (0.59%) | 15 (0.60%) | 11 (0.45%) | 14 (0.57%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-labor force | 1910 (19.22%) | 471 (18.96%) | 467 (19.08%) | 466 (19.04%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Others | 170 (1.71%) | 36 (1.45%) | 30 (1.23%) | 36 (1.47%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marital status | 0.0441 | 0.0643 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Single | 670 (6.74%) | 168 (6.76%) | 132 (5.39%) | 151 (6.17%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Married | 7957 (80.08%) | 1964 (79.07%) | 2013 (82.26%) | 1947 (79.57%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Divorced | 545 (5.49%) | 150 (6.04%) | 125 (5.11%) | 147 (6.01%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Spouse deceased | 764 (7.69%) | 202 (8.13%) | 177 (7.23%) | 202 (8.26%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Education | 0.1590 | 0.0519 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≤9 years | 4592 (46.22%) | 1122 (45.17%) | 1143 (46.71%) | 1121 (45.81%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 10–12 years | 1381 (13.90%) | 413 (16.63%) | 411 (16.80%) | 402 (16.43%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 13–15 years | 2890 (29.09%) | 777 (31.28%) | 737 (30.12%) | 754 (30.81%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≥15 years | 1073 (10.80%) | 172 (6.92%) | 156 (6.38%) | 170 (6.95%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Co-morbidities | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension | 5370 (54.05%) | 1430 (57.57%) | 0.0710 | 1398 (57.13%) | 1407 (57.50%) | 0.0074 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes mellitus | 5084 (51.17%) | 1397 (56.24%) | 0.1019 | 1371 (56.03%) | 1367 (55.86%) | 0.0033 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IHD | 744 (7.49%) | 173 (6.96%) | 0.0202 | 159 (6.50%) | 172 (7.03%) | 0.0212 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hyperlipidemia | 3516 (35.39%) | 953 (38.37%) | 0.0618 | 927 (37.88%) | 931 (38.05%) | 0.0034 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CHF | 462 (4.65%) | 108 (4.35%) | 0.0146 | 95 (3.88%) | 108 (4.41%) | 0.0266 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rheumatic disease | 103 (1.04%) | 21 (0.85%) | 0.0198 | 13 (0.53%) | 21 (0.86%) | 0.0394 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kidney disease | 1268 (12.76%) | 361 (14.53%) | 0.0516 | 323 (13.20%) | 348 (14.22%) | 0.0297 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CKD | 580 (5.84%) | 171 (6.88%) | 0.0429 | 169 (6.91%) | 165 (6.74%) | 0.0065 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Type of Anti-VEGF | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ranibizumab | 0 (0.00%) | 1874 (75.44%) | – | 0 (0.00%) | 1845 (75.40%) | – | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Aflibercept | 0 (0.00%) | 610 (24.56%) | – | 0 (0.00%) | 602 (24.60%) | – | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: AMD: Age-related macular degeneration; CHF: Congestive heart failure; CKD: Chronic kidney disease; CNV: Choroidal neovascularization; DME: Diabetic macular edema; IHD: Ischemic heart diseases; PCV: Polypoidal choroidal vasculopathy; RVO: Branch retinal vein occlusion or central retinal vein occlusion; VEGF: Vascular endothelial growth factor. aPropensity score matching (PSM) was done by matching: Year of index, Sex, Age at index, Indication, Urbanization, Insured unit type, Marital status, Education level, Co-morbidities (including Hypertension, Diabetes mellitus, Ischemic heart diseases, Hyperlipidemia, Congestive heart failure, and Rheumatic disease). bAbsolute standardized difference (ASD): >0.1 implies a meaningful imbalance between the groups. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Comparison of the incidence rates and cumulative risk of dialysis between anti-VEGF-administered patients and patients without anti-VEGF treatment
In the PSM cohort, the incidence rate of dialysis per 10,000 person-months was 17.58 (95% CI: 14.82–20.85) in the anti-VEGF group, which was significantly higher than that in the group without anti-VEGF treatment: 9.37 (95% CI: 7.41–11.84) (Table 2). There was a significant difference in the cumulative risk of dialysis between the cohorts over the entire Kaplan–Meier curve (p < 0.0001, log rank test; Figure 2), and the adjusted hazard ratio was 1.849 (95% CI: 1.378–2.482) in the PSM cohort (Table 2). In addition, hypertension, diabetes, and congestive heart failure were also associated with the increased risk of dialysis (Supplementary Table 1). Moreover, the results also revealed a higher risk of dialysis in the anti-VEGF group when the endpoints of hemodialysis (HD) and peritoneal dialysis (PD) were analyzed separately (Table 2).
Table 2. Incidence and risk of hemodialysis or peritoneal dialysis among study groups.
| Age-and sex-matched | PSMa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No anti-VEGF N = 9936 | Anti-VEGF N = 2484 | No anti-VEGF N = 2447 | Anti-VEGF N = 2447 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HD or PD | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Follow up person months | 302467 | 76142 | 74741 | 75080 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| New case | 281 | 137 | 70 | 132 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Incidence rateb (95% C.I.) | 9.29 (8.26–10.44) | 17.99 (15.22–21.27) | 9.37 (7.41–11.84) | 17.58 (14.82–20.85) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Adjusted Hazard ratio (95% C.I.) | Reference | 1.680 (1.358–2.078) | Reference | 1.849 (1.378–2.482) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hemodialysis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Follow up person months | 302506 | 76180 | 74752 | 75118 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| New case | 279 | 135 | 69 | 130 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Incidence rate (95% C.I.) | 9.22 (8.20–10.37) | 17.72 (14.97–20.98) | 9.23 (7.29–11.69) | 17.31 (14.57–20.55) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Adjusted Hazard ratio (95% C.I.) | Reference | 1.664 (1.343–2.061) | Reference | 1.839 (1.367–2.473) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Peritoneal dialysis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Follow up person months | 306222 | 78181 | 75680 | 77044 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| New case | 11 | 16 | 4 | 16 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Incidence rate (95% C.I.) | 0.36 (0.20–0.65) | 2.05 (1.25–3.34) | 0.53 (0.20–1.41) | 2.08 (1.27–3.39) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Adjusted Hazard ratio (95% C.I.) | Reference | 4.052 (1.754–9.358) | Reference | 3.426 (1.107–10.604) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: C.I.: confidence interval; HD: Hemodialysis; PD: Peritoneal dialysis; VEGF: Vascular endothelial growth factor. aPropensity score matching (PSM) was done by matching: Year of index, Sex, Age at index, Indication, Urbanization, Insured unit type, Marital status, Education level, Co-morbidities (including Hypertension, Diabetes mellitus, Ischemic heart diseases, Hyperlipidemia, Congestive heart failure, and Rheumatic disease). bIncidence rate, per 10000 person-month. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
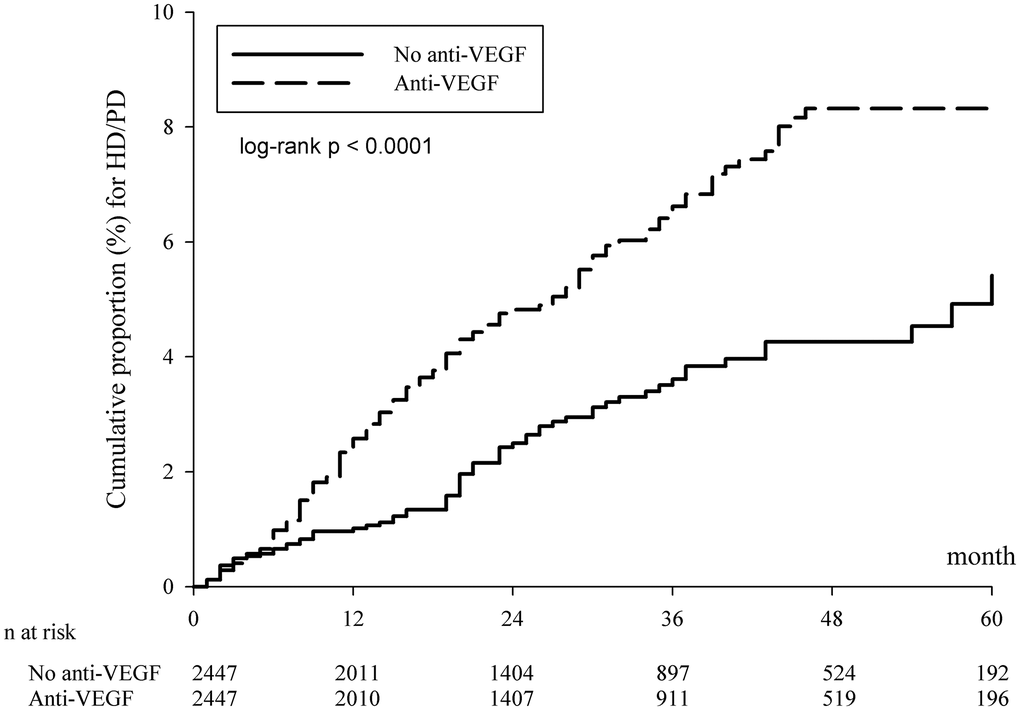
Figure 2. Kaplan-Meier curves of the cumulative proportion for hemodialysis/peritoneal dialysis in the propensity score matched cohort. Abbreviations: HD: hemodialysis; PD: peritoneal dialysis; VEGF: vascular endothelial growth factor.
Discussion
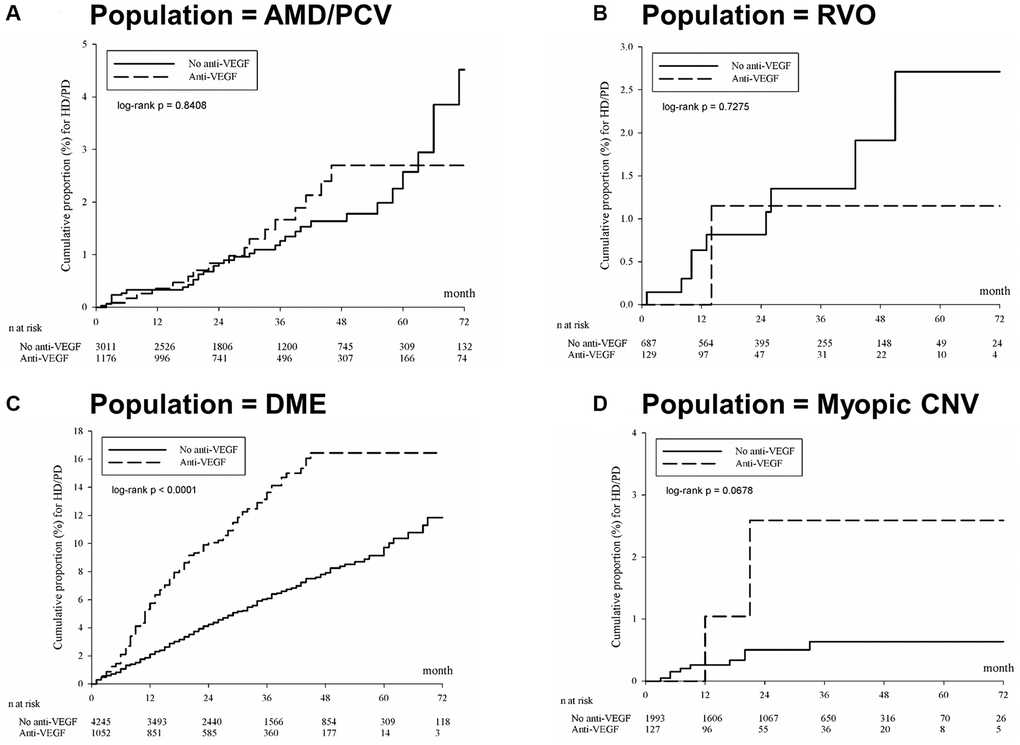
The present study demonstrated that compared to patients who never received intravitreal anti-VEGF, patients who ever received intravitreal anti-VEGF were associated with 85% higher risk of becoming dialysis-dependent according to the results of group comparison after PSM (Table 2). Given the increasing usage of intravitreal anti-VEGF and the large socioeconomic burden of being dialysis dependent, this minor difference may have a great impact on the current clinical practice. Subgroup analysis showed the risk of becoming dialysis dependent is 83% higher in the patients who received intravitreal anti-VEGFs for DME compared to the patients with DME but did not receive anti-VEGF treatment (Table 3).
Keir et al., [17] demonstrated that intravitreal injection of VEGF inhibits local complement factor H in the rodent kidney, thus inducing alternative complement pathway deposits in the kidney. In previous case reports, intravitreal injections of anti-VEGF agents were noted to impact renal function in patients with either healthy kidneys [20] or with already compromised kidneys [21, 23]. To our knowledge, this large-scale study is the first report to explore the role of intravitreally administered anti-VEGF in the cumulative risk of requiring maintenance dialysis by using a population-level database.
Our study showed that DME patients had a risk of dialysis dependence after the administration of intravitreal anti-VEGF agents (Table 3). Diabetes in the process of renal impairment seemed to be a confounder. The incidence of renal adverse events was reported to be 16–22% in diabetic patients of the Protocol T two-year database [25]. While some articles demonstrated a correlation between intravitreal injection of anti-VEGF agents and impairment of renal function and aggravation of proteinuria in diabetic kidneys [22–24], others showed no such correlation [26–28]. The influence of VEGF-A might be complex for the changes in retinal condition and renal function in diabetic patients, because whereas VEGF-A expression is often upregulated in the kidneys of diabetic rodents, the opposite pattern has been noted in humans [29].
The risk of major systemic adverse events was comparable among different anti-VEGF agents [30]. However, in our study, the patients receiving ranibizumab treatment in the anti-VEGF group had an elevated risk for dialysis-dependent status (adjusted hazard ratio: 1.896; 95% CI: 1.409–2.551), while no statistically significant risk was identified in those receiving aflibercept (adjusted hazard ratio: 1.166; 95% CI: 0.454–2.996) (Supplementary Table 2). Additionally, a slight dose-dependent trend was found in the ranibizumab-administered patients, which showed an elevated risk of being dialysis-dependent after administration of the second dose. Our finding contrasts with the hypothesis based solely on systemic VEGF-A inhibition. Different isoforms of VEGF-A may influence kidney disease development in diabetic patients [29]. Ranibizumab and aflibercept differ in their molecular structures, binding targets and binding affinities to their respective binding sites [31, 32], so theoretically, the risks of causing renal injury may differ from each drug. More clinical and laboratory investigations are required to further elucidate the mechanisms.
Our study results also demonstrated higher risk of PD after the uses of anti-VEGF drugs. The possible explanations of a higher risk of PD after the uses of anti-VEGF drugs when compared to HD might be due to the fact that the patients who were able to attend regular ophthalmologic exams were often with better overall performance and may prefer more autonomy when receiving renal replacement therapy. Hence, the risk of PD appeared higher after the uses of anti-VEGF drugs compared with HD.
For the Kaplan-Meier curves of the cumulative proportion for HD or PD in Figure 2, the survival line of anti-VEGF agents was unchanged after 48 months. The possible explanations might be as follows: The approved doses injections had to be administered within five years following the reimbursement approval under the policy of National Health Insurance reimbursement criteria for ranibizumab and aflibercept during the study period. Moreover, in recent real-world studies, [33, 34] investigators found that most injections were given in the first four years following the initialization of intravitreal anti-VEGF treatment. This may explain the reason why the survival line of anti-VEGF agents was unchanged after 48 months, since further renal adverse effects may not be present after the termination of regular intravitreal anti-VEGF treatment.
Strengths of this study include the large real-world cohort to evaluate possible side effects of anti-VEGF agents on risks of dialysis. Moreover, the similar findings in the age- and sex-matched cohort and PSM cohort re-ascertained our study results. Nonetheless, we do acknowledge some limitations to the presented study. First, the indications, demographic information and comorbidities of the included patients depended on the accuracy of International Classification of Diseases, 9th Revision, Clinical Modification (ICD-9-CM), and International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM) codes, so coding errors might exist and may lead to information bias. Second, due to the strict reimbursement criteria (Table 4) of the National Health Insurance, not all patients with indications of AMD/PCV, DME, RVO or myopic CNV were eligible to receive anti-VEGF treatment. Third, although ranibizumab and aflibercept are both covered by the National Health Insurance, some patients might receive self-paid anti-VEGF treatment, including off-label use of intravitreal bevacizumab. Fourth, DME accounted for over 40% of the indications, the current construction of the propensity score may not protect against potential residual confounding due to diabetes severity, since serial HbA1C is not available in the NHIRD. Fifth, as the primary outcome is receiving dialysis, the baseline renal function is a potential confounder. However, since the dataset of NHIRD did not contain the laboratory data of renal function, we could only adjust the chronic kidney disease in the analyses by relevant ICD codes. Even if our study results indicated a higher risk of developing dialysis in patients receiving intravitreal anti-VEGF agents, more studies, including randomized controlled trials, or new-user design with active comparator or systematic review and meta-analysis, are warranted to clarify the role of anti-VEGF agents in the pathophysiology of dialysis and to replicate our findings.
Table 4. The National Health Insurance reimbursement criteria for intravitreal ranibizumab and aflibercept in patients with different indications (released on March 1st, 2020).
| Initial date of reimbursement | Maximum doses/5 yrs | Age | CRT | HbA1c | BCVA 20/400-20/40 | FA | OCT | ICGA | High myopiaa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AMD/PCV | RBZ: Feb 2011/Dec 2017 AFL: Aug 2014/Nov 2016 | 7# | ≧50 yrs (nAMD) | V | V | V | V (PCV) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DME | RBZ: Feb 2013 AFL: Nov 2016 | 8# | ≧300 μm | <10% | V | V | V | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CRVO/BRVO | RBZ: Jul 2016/Dec 2017 AFL: Nov 2016/Dec 2017 | 7# | ≧18 yrs | ≧300 μm | V | V | V | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| mCNV | RBZ: Jul 2016 AFL: Dec 2017 | 3 | V | V | V | V | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: AFL: aflibercept; BCVA: best corrected visual acuity; BRVO: branch retinal vein occlusion; CRT: central retinal thickness; CRVO: central retinal vein occlusion; DME: diabetic macular edema; FA: fluorescein angiography; ICGA: indocyanine green angiography; mCNV: myopic choroidal neovascularization; AMD: age-related macular degeneration; OCT: optical coherence tomography; PCV: polypoidal choroidal vasculopathy; RBZ: ranibizumab. aHigh myopia: myopia over -6.00D and axial length >26 mm. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| The National Health Insurance reimbursement criteria of intravitreal ranibizumab and aflibercept in patients with different indications. In patients with a diagnosis of age-related macular degeneration (AMD)/polypoidal choroidal vasculopathy (PCV), the reimbursement criteria include: age 50 years or older; fluorescein angiography (FA), indocyanine green angiography (ICGA) (only for PCV) and optical coherence tomography (OCT) performed within the past month compatible with a diagnosis of neovascular AMD/PCV; and best-corrected visual acuity (BCVA) within 0.05–0.5 (20/400-20/40). In patients with a diagnosis of diabetic macular edema (DME), the reimbursement criteria include: central retinal thickness (CRT) above 300 μm; HbA1c level below 10%; FA, OCT performed within the past month compatible with a diagnosis of DME; and BCVA within 0.05–0.5 (20/400-20/40). In patients with a diagnosis of central retinal vein occlusion (CRVO) or branch retinal vein occlusion (BRVO), the reimbursement criteria include: age 18 years or older; central retinal thickness above 300 μm; FA, OCT performed within the past month compatible with a diagnosis of CRVO or BRVO; and BCVA within 0.05–0.5 (20/400-20/40). In patients with a diagnosis of myopic choroidal neovascularization (mCNV), the reimbursement criteria include: myopia over -6.00D and axial length >26 mm; FA, OCT performed within the past month compatible with a diagnosis of myopic CNV; and BCVA within 0.05–0.5 (20/400-20/40). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| #Patients with AMD/PCV, DME, CRVO/BRVO, and mCNV receive initial 3, 5, 3, and 3 injections respectively following the first reimbursement approval. Additional 4, 3, and 4 injections for AMD/PCV, DME, and CRVO/BRVO patients will be approved following the second reimbursement. All medication should be injected within five years following reimbursement approval. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
In summary, our study demonstrated an increased risk of becoming dialysis-dependent in patients receiving intravitreal anti-VEGF therapy. Furthermore, our data suggest that patients who receive anti-VEGF for DME treatment and those who receive repeated doses of ranibizumab are more susceptible to dialysis treatment. Based on these findings, a regular laboratory workup of the renal function in patients receiving multiple doses of anti-VEGF agents is imperative. Future work is warranted to develop a predictive model to determine the patients at risk of dialysis after anti-VEGF treatment, thereby helping clinicians choose appropriate treatment plans for each individual.
Methods
Data source
The retrospective population-based cohort study was approved by the Chung Shan Medical University Hospital Institutional Review Board (IRB No.: CS1-20108) and by the National Health Insurance Administration. The data extracted from the NHIRD, a database supported by the Taiwan National Health Research Institutes, contain all the medical information of insurance claims from the Taiwanese population. The information was anonymized before release, hence the Chung Shan Medical University Hospital Institutional Review Board obviated the necessity of obtaining participants’ informed consent for this study. We confirmed that all experiments were performed in accordance with relevant guidelines and regulations. The information includes date of birth, sex, place of residence, inpatient and outpatient services, details of medications, intervention procedures, date of admission and discharge, and diagnosis records (based on the ICD-9-CM and ICD-10-CM). In our present study, we used the data of 2 million individuals who were randomly sampled from the NHIRD, namely, the Longitudinal Health Insurance Database (LHID) (from 2000 to 2017). The accuracy of the NHIRD has already been demonstrated by several previous studies [35–37].
The national health insurance program implemented by the government in 1995, now covering 99% of the 23.5 million people residing in Taiwan, has approved the conditional reimbursement of intravitreal ranibizumab and aflibercept (Table 4) since 2011, provided that the application was approved by retinal experts in advance. Furthermore, the switch of anti-VEGF agents after approval was banned by the policy, that is, the patient received the same anti-VEGF throughout the treatment course. In view of the strict criteria of the reimbursement and the large sample size of Taiwan’s NHIRD, it is statistically feasible to assess the incidence of becoming dialysis-dependent status, which is a rare adverse event, among treated patients.
Study population
Individuals diagnosed with age-related macular degeneration or polypoidal choroidal vasculopathy (ICD-9-CM code 362.16, 362.50, 362.51, 362.52; ICD-10-CM code H35.05x, H35.30, H35.31, H35.32), retinal vessel occlusion (ICD-9-CM code 362.3x; ICD-10-CM code H34.x), diabetic macular edema (ICD-9-CM code 250.5x, 362.0x; ICD-10-CM code E08.3x, E09.3x, E10.3x, E11.3x, E13.3x), myopic choroidal neovascularization (ICD-9-CM code 360.21, 367.1; ICD-10-CM code H44.2x, H52.1x) with three more visits to outpatient services due to previously mentioned diseases during January 2000 to December 2017 were included. For patients who received intravitreal aflibercept (Anatomical Therapeutic Chemical (ATC) drug code: S01LA0) or ranibizumab (ATC drug code: S01LA04), the index date was the day of the first intravitreal anti-VEGF injection. For patients who did not receive intravitreal anti-VEGF treatment, the index date was nested with the paired anti-VEGF patients. All study participants were at risk on the index date. In the present study, we used the time distribution matching method to control immortal time bias [38]. To evaluate the association between intravitreal anti-VEGF and new-onset dialysis, the following exclusion criteria were defined: (1) use of bevacizumab and (2) HD or PD status before the index date. We excluded the patients using bevacizumab because bevacizumab was reimbursed for the indication of cancer treatment in Taiwan. Although systemic uses of aflibercept was approved for cancer treatment, it is not covered by Taiwan's National Health Insurance [39]. Hence, we did not exclude systemic uses of aflibercept in the present study. After the exclusions, 2484 patients who had undergone at least one intravitreal injection of anti-VEGF from January 2011 to July 2017 remained in the study cohort. The study design compared the difference in the risk of new-onset dialysis between anti-VEGF and without anti-VEGF cohorts. Each anti-VEGF patient was assigned to 4 controls matched by age and sex, and 9936 patients without anti-VEGF were included for the control cohort. Patients with and without anti-VEGF therapy were further matched with propensity scores at a 1:1 ratio to minimize selection bias. Propensity scores were calculated using logistic regression to estimate the probability of receiving intravitreal anti-VEGF based on the baseline variables of year of index, sex, age at index, indication, urbanization, insured unit type, marital status, education level, and comorbidities (hypertension, diabetes mellitus, ischemic heart diseases, hyperlipidemia, congestive heart failure, rheumatic disease, all kidney diseases, and specifically chronic kidney disease). After PSM, the groups with and without anti-VEGF therapy were comprised of 2447 patients (Figure 1).
Main outcome measurement
The primary outcome was new dialysis-dependent status following intravitreal anti-VEGF based on the charge master code for hemodialysis (58001C, 58002C, 58003C, 58007C, 58014C, 58018C, 58019C, 58020C, 58021C, 58022C, 58023C, 58024C, 58025C, 58027C, and 58029C) and peritoneal dialysis (58009B, 58010B, 58012B, 58011C, 58017C, and 58028C) after the index date. To avoid the emergent dialysis due to acute kidney injury, we defined the outcome of new dialysis as at least 2 times of records of dialysis. The study period was from January 1, 2000 to December 31, 2017. The varieties of injected agents and injection times were compared for analyzing their associations with newly performed dialysis. Subgroup analysis, including different indications of AMD/PCV, RVO, DME and myopic CNV, was also performed to identify relationships with newly performed dialysis.
Identification of comorbidities
Comorbidities were identified to evaluate the health status of each participant and to investigate the correlation between newly needed dialysis and comorbidities. Comorbidities included hypertension, diabetes mellitus, ischemic heart disease, hyperlipidemia, congestive heart failure, rheumatic disease, all kidney diseases, and specifically chronic kidney disease. The comorbidities were identified within 1 year before the index date by relevant ICD codes (Supplementary Table 3).
Statistical analysis
SAS version 9.4 (SAS Institute Inc., Cary, NC, USA) was used for all analyses. After 1:4 matching with age and sex and PSM, the absolute standardized difference (ASD) was used to evaluate the differences between the study and control groups. An ASD <0.1 indicates a negligible difference between two treatment groups. Chi-square tests or Fisher’s exact tests were used for categorical variables, and Student’s t-tests were used to compare continuous variables. Poisson assumptions were used to calculate the incidence rate. Cumulative incidence rates were calculated according to analysis of the cumulative incidence of outcome events. The adjusted HR integrates the patients’ demographic information and comorbidities and was analyzed with multiple Cox proportional hazards regressions. We also conducted subgroup analysis to assess the risk of dialysis-dependent status from intravitreal anti-VEGF treatment, grouped by indications (AMD/PCV, RVO, DME, and myopic CNV), anti-VEGF agents (ranibizumab and aflibercept) and number of injections. Dialysis was also divided into two subgroups: hemodialysis and peritoneal dialysis. The incidence rates of each subgroup were calculated. Statistical significance was defined as p < 0.05.
Ethics approval and consent to participate
The retrospective study was approved by the Chung Shan Medical University Hospital Institutional Review Board (IRB No.: CS1-20108) and by the National Health Insurance Administration. The information was anonymized before release, hence the Chung Shan Medical University Hospital Institutional Review Board obviated the necessity of obtaining participants’ informed consent for this study.
Consent for publication
The information was anonymized before release, hence the Chung Shan Medical University Hospital Institutional Review Board obviated the necessity of obtaining participants’ informed consent for this study.
Availability of data and materials
The datasets generated and/or analysed during the current study are available in the Taiwan National Health Insurance Research Database repository, (https://nhird.nhri.org.tw/en/How_to_cite_us.html).
Supplementary Materials
Author Contributions
Professor Shun-Fa Yang and Dr. Yu-Chen Su contributed equally as co-first authors. J.H.H. and S.F.Y. contributed to the study conception and design. All authors collected the clinical data. J.Y.H. carried out the statistical and data analyses. All authors participated in the interpretation of data. S.F.Y., S.M.H., L.W.W., J.H.H. supervised the research. The first draft of the manuscript was written by Y.C.S. and all authors commented on previous versions of the manuscript. All authors approved the submitted version, agreed to be accountable for their own contributions, and ensured that questions related to the accuracy or integrity of any part of the work, even ones in which the author was not personally involved, were appropriately investigated and resolved with the resolution documented in the literature.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Funding
This study was financially supported by funding from National Cheng Kung University Hospital, Taiwan [NCKUH-11102004], and the Ministry of Science and Technology [MOST 110-2314-B-006-086-MY3]; these grants were awarded to J.H. Hung.
References
- 1. Saeed F, Arrigain S, Schold JD, Nally JV
Jr , Navaneethan SD. What are the Risk Factors for One-Year Mortality in Older Patients with Chronic Kidney Disease? An Analysis of the Cleveland Clinic CKD Registry. Nephron. 2019; 141:98–104. https://doi.org/10.1159/000494298 [PubMed] - 2. Liyanage T, Ninomiya T, Jha V, Neal B, Patrice HM, Okpechi I, Zhao MH, Lv J, Garg AX, Knight J, Rodgers A, Gallagher M, Kotwal S, et al. Worldwide access to treatment for end-stage kidney disease: a systematic review. Lancet. 2015; 385:1975–82. https://doi.org/10.1016/S0140-6736(14)61601-9 [PubMed]
- 3. Tsai MH, Hsu CY, Lin MY, Yen MF, Chen HH, Chiu YH, Hwang SJ. Incidence, Prevalence, and Duration of Chronic Kidney Disease in Taiwan: Results from a Community-Based Screening Program of 106,094 Individuals. Nephron. 2018; 140:175–84. https://doi.org/10.1159/000491708 [PubMed]
- 4. Saran R, Robinson B, Abbott KC, Agodoa LYC, Bragg-Gresham J, Balkrishnan R, Bhave N, Dietrich X, Ding Z, Eggers PW, Gaipov A, Gillen D, Gipson D, et al. US Renal Data System 2018 Annual Data Report: Epidemiology of Kidney Disease in the United States. Am J Kidney Dis. 2019; 73:A7–8. https://doi.org/10.1053/j.ajkd.2019.01.001 [PubMed]
- 5. Taber SS, Pasko DA. The epidemiology of drug-induced disorders: the kidney. Expert Opin Drug Saf. 2008; 7:679–90. https://doi.org/10.1517/14740330802410462 [PubMed]
- 6. Hanna RM, Barsoum M, Arman F, Selamet U, Hasnain H, Kurtz I. Nephrotoxicity induced by intravitreal vascular endothelial growth factor inhibitors: emerging evidence. Kidney Int. 2019; 96:572–80. https://doi.org/10.1016/j.kint.2019.02.042 [PubMed]
- 7. Stewart MW. The expanding role of vascular endothelial growth factor inhibitors in ophthalmology. Mayo Clin Proc. 2012; 87:77–88. https://doi.org/10.1016/j.mayocp.2011.10.001 [PubMed]
- 8. Chen SN, Cheng CK, Yeung L, Chen JT, Chan WC, Liu JH, Sheu SJ, Wu WC, Lai CC. One-year real-world outcomes of ranibizumab 0.5 mg treatment in Taiwanese patients with polypoidal choroidal vasculopathy: a subgroup analysis of the REAL study. Int J Ophthalmol. 2018; 11:1802–8. https://doi.org/10.18240/ijo.2018.11.11 [PubMed]
- 9. Zhang Y, Han Q, Ru Y, Bo Q, Wei RH. Anti-VEGF treatment for myopic choroid neovascularization: from molecular characterization to update on clinical application. Drug Des Devel Ther. 2015; 9:3413–21. https://doi.org/10.2147/DDDT.S87920 [PubMed]
- 10. Sheu SJ, Cheng CK, Kuo HK, Tsai CY, Lin TC, Tan J, Chandwani H, Adena M, Chen SJ. Treatment patterns in diabetic macular edema in Taiwan: a retrospective chart review. Clin Ophthalmol. 2018; 12:2189–98. https://doi.org/10.2147/OPTH.S170089 [PubMed]
- 11. Owen CG, Jarrar Z, Wormald R, Cook DG, Fletcher AE, Rudnicka AR. The estimated prevalence and incidence of late stage age related macular degeneration in the UK. Br J Ophthalmol. 2012; 96:752–6. https://doi.org/10.1136/bjophthalmol-2011-301109 [PubMed]
- 12. Chang YS, Lee WJ, Lim CC, Wang SH, Hsu SM, Chen YC, Cheng CY, Teng YT, Huang YH, Lai CC, Tseng SH. Real-world use of ranibizumab for neovascular age-related macular degeneration in Taiwan. Sci Rep. 2018; 8:7486. https://doi.org/10.1038/s41598-018-25864-0 [PubMed]
- 13. Rasier R, Artunay O, Yuzbasioglu E, Sengul A, Bahcecioglu H. The effect of intravitreal bevacizumab (avastin) administration on systemic hypertension. Eye (Lond). 2009; 23:1714–8. https://doi.org/10.1038/eye.2008.360 [PubMed]
- 14. Risimic D, Milenkovic S, Nikolic D, Simeunovic D, Jaksic V, Stojkovic M, Stefanovic I, Jakovic N, Prostran M. Influence of intravitreal injection of bevacizumab on systemic blood pressure changes in patients with exudative form of age-related macular degeneration. Hellenic J Cardiol. 2013; 54:435–40. [PubMed]
- 15. Avery RL, Gordon GM. Systemic Safety of Prolonged Monthly Anti-Vascular Endothelial Growth Factor Therapy for Diabetic Macular Edema: A Systematic Review and Meta-analysis. JAMA Ophthalmol. 2016; 134:21–9. https://doi.org/10.1001/jamaophthalmol.2015.4070 [PubMed]
- 16. Hanhart J, Comaneshter DS, Freier-Dror Y, Vinker S. Mortality associated with bevacizumab intravitreal injections in age-related macular degeneration patients after acute myocardial infarct: a retrospective population-based survival analysis. Graefes Arch Clin Exp Ophthalmol. 2018; 256:651–63. https://doi.org/10.1007/s00417-018-3917-9 [PubMed]
- 17. Keir LS, Firth R, Aponik L, Feitelberg D, Sakimoto S, Aguilar E, Welsh GI, Richards A, Usui Y, Satchell SC, Kuzmuk V, Coward RJ, Goult J, et al. VEGF regulates local inhibitory complement proteins in the eye and kidney. J Clin Invest. 2017; 127:199–214. https://doi.org/10.1172/JCI86418 [PubMed]
- 18. Khneizer G, Al-Taee A, Bastani B. Self-limited membranous nephropathy after intravitreal bevacizumab therapy for age-related macular degeneration. J Nephropathol. 2017; 6:134–7. https://doi.org/10.15171/jnp.2017.23 [PubMed]
- 19. Touzani F, Geers C, Pozdzik A. Intravitreal Injection of Anti-VEGF Antibody Induces Glomerular Endothelial Cells Injury. Case Rep Nephrol. 2019; 2019:2919080. https://doi.org/10.1155/2019/2919080 [PubMed]
- 20. Pellé G, Shweke N, Duong Van Huyen JP, Tricot L, Hessaïne S, Frémeaux-Bacchi V, Hiesse C, Delahousse M. Systemic and kidney toxicity of intraocular administration of vascular endothelial growth factor inhibitors. Am J Kidney Dis. 2011; 57:756–9. https://doi.org/10.1053/j.ajkd.2010.11.030 [PubMed]
- 21. Cheungpasitporn W, Chebib FT, Cornell LD, Brodin ML, Nasr SH, Schinstock CA, Stegall MD, Amer H. Intravitreal Antivascular Endothelial Growth Factor Therapy May Induce Proteinuria and Antibody Mediated Injury in Renal Allografts. Transplantation. 2015; 99:2382–6. https://doi.org/10.1097/TP.0000000000000750 [PubMed]
- 22. Hanna RM, Lopez EA, Hasnain H, Selamet U, Wilson J, Youssef PN, Akladeous N, Bunnapradist S, Gorin MB. Three patients with injection of intravitreal vascular endothelial growth factor inhibitors and subsequent exacerbation of chronic proteinuria and hypertension. Clin Kidney J. 2019; 12:92–100. https://doi.org/10.1093/ckj/sfy060 [PubMed]
- 23. Huang YF, Chen SJ, Hsu MY, Hwang DK. Acute renal failure after intravitreal antivascular endothelial growth factor therapy. J Formos Med Assoc. 2017; 116:490–2. https://doi.org/10.1016/j.jfma.2016.09.010 [PubMed]
- 24. Georgalas I, Papaconstantinou D, Papadopoulos K, Pagoulatos D, Karagiannis D, Koutsandrea C. Renal injury following intravitreal anti-VEGF administration in diabetic patients with proliferative diabetic retinopathy and chronic kidney disease--a possible side effect? Curr Drug Saf. 2014; 9:156–8. https://doi.org/10.2174/1574886309666140211113635 [PubMed]
- 25. Wells JA, Glassman AR, Ayala AR, Jampol LM, Bressler NM, Bressler SB, Brucker AJ, Ferris FL, Hampton GR, Jhaveri C, Melia M, Beck RW, and Diabetic Retinopathy Clinical Research Network. Aflibercept, Bevacizumab, or Ranibizumab for Diabetic Macular Edema: Two-Year Results from a Comparative Effectiveness Randomized Clinical Trial. Ophthalmology. 2016; 123:1351–9. https://doi.org/10.1016/j.ophtha.2016.02.022 [PubMed]
- 26. Bagheri S, Dormanesh B, Afarid M, Sagheb MM. Proteinuria and Renal Dysfunction after Intravitreal Injection of Bevacizumab in Patients with Diabetic Nephropathy: A Prospective Observational Study. Galen Med J. 2018; 7:e1299. https://doi.org/10.22086/gmj.v0i0.1299 [PubMed]
- 27. O'Neill RA, Gallagher P, Douglas T, Little JA, Maxwell AP, Silvestri G, McKay G. Evaluation of long-term intravitreal anti-vascular endothelial growth factor injections on renal function in patients with and without diabetic kidney disease. BMC Nephrol. 2019; 20:478. https://doi.org/10.1186/s12882-019-1650-1 [PubMed]
- 28. Glassman AR, Liu D, Jampol LM, Sun JK, and Diabetic Retinopathy Clinical Research Network. Changes in Blood Pressure and Urine Albumin-Creatinine Ratio in a Randomized Clinical Trial Comparing Aflibercept, Bevacizumab, and Ranibizumab for Diabetic Macular Edema. Invest Ophthalmol Vis Sci. 2018; 59:1199–205. https://doi.org/10.1167/iovs.17-22853 [PubMed]
- 29. Majumder S, Advani A. VEGF and the diabetic kidney: More than too much of a good thing. J Diabetes Complications. 2017; 31:273–9. https://doi.org/10.1016/j.jdiacomp.2016.10.020 [PubMed]
- 30. Maloney MH, Payne SR, Herrin J, Sangaralingham LR, Shah ND, Barkmeier AJ. Risk of Systemic Adverse Events after Intravitreal Bevacizumab, Ranibizumab, and Aflibercept in Routine Clinical Practice. Ophthalmology. 2021; 128:417–24. https://doi.org/10.1016/j.ophtha.2020.07.062 [PubMed]
- 31. Avery RL, Castellarin AA, Steinle NC, Dhoot DS, Pieramici DJ, See R, Couvillion S, Nasir MA, Rabena MD, Maia M, Van Everen S, Le K, Hanley WD. Systemic Pharmacokinetics and Pharmacodynamics of Intravitreal Aflibercept, Bevacizumab, and Ranibizumab. Retina. 2017; 37:1847–58. https://doi.org/10.1097/IAE.0000000000001493 [PubMed]
- 32. Yang J, Wang X, Fuh G, Yu L, Wakshull E, Khosraviani M, Day ES, Demeule B, Liu J, Shire SJ, Ferrara N, Yadav S. Comparison of binding characteristics and in vitro activities of three inhibitors of vascular endothelial growth factor A. Mol Pharm. 2014; 11:3421–30. https://doi.org/10.1021/mp500160v [PubMed]
- 33. Chou YB, Chen MJ, Lin TC, Chen SJ, Hwang DK. Priority options of anti-vascular endothelial growth factor agents in wet age-related macular degeneration under the National Health Insurance Program. J Chin Med Assoc. 2019; 82:659–64. https://doi.org/10.1097/JCMA.0000000000000138 [PubMed]
- 34. Lo KJ, Chang JY, Chang HY, Chiou SH, Hwang DK, Chen SJ. Three-Year Outcomes of Patients with Neovascular Age-Related Macular Degeneration Treated with Aflibercept under the National Health Insurance Program in Taiwan. J Ophthalmol. 2020; 2020:4538135. https://doi.org/10.1155/2020/4538135 [PubMed]
- 35. Hsu CC, Lee CH, Wahlqvist ML, Huang HL, Chang HY, Chen L, Shih SF, Shin SJ, Tsai WC, Chen T, Huang CT, Cheng JS. Poverty increases type 2 diabetes incidence and inequality of care despite universal health coverage. Diabetes Care. 2012; 35:2286–92. https://doi.org/10.2337/dc11-2052 [PubMed]
- 36. Chen HL, Hsiao FY. Risk of hospitalization and healthcare cost associated with Diabetes Complication Severity Index in Taiwan's National Health Insurance Research Database. J Diabetes Complications. 2014; 28:612–6. https://doi.org/10.1016/j.jdiacomp.2014.05.011 [PubMed]
- 37. Keller JJ, Kang JH, Lin HC. Association between osteoporosis and psoriasis: results from the Longitudinal Health Insurance Database in Taiwan. Osteoporos Int. 2013; 24:1835–41. https://doi.org/10.1007/s00198-012-2185-5 [PubMed]
- 38. Karim ME, Gustafson P, Petkau J, Tremlett H, and Long-Term Benefits and Adverse Effects of Beta-Interferon for Multiple Sclerosis (BeAMS) Study Group. Comparison of Statistical Approaches for Dealing With Immortal Time Bias in Drug Effectiveness Studies. Am J Epidemiol. 2016; 184:325–35. https://doi.org/10.1093/aje/kwv445 [PubMed]
- 39. Kuo CN, Liao YM, Kuo LN, Tsai HJ, Chang WC, Yen Y. Cancers in Taiwan: Practical insight from epidemiology, treatments, biomarkers, and cost. J Formos Med Assoc. 2020; 119:1731–41. https://doi.org/10.1016/j.jfma.2019.08.023 [PubMed]