Introduction
Immune evasion assumes an important role in the genesis and progression of cancer, constituting one of the foremost hallmarks of cancer [1, 2]. In addition to modulating the immune system, immune checkpoints, which are made up of co-inhibitory and stimulatory signals, can shield cancer cells from immune destruction [1, 3]. Immune checkpoint inhibitors (ICIs), whether administered in isolation or in combination, can yield enduring antitumor effects by strategically reducing the production of negative immunomodulatory factors [4]. The Food and Drug Administration (FDA) has currently approved several ICIs (anti-PD-(L)1 and anti-CTLA-4 antibodies) for use in some cancer indications.
Nonetheless, the efficacy of ICI therapy varies contingent on the specific cancer type, typically spanning from a modest 10% to 40%. Notably, a substantial proportion of patients experience disease progression despite an initial positive response [4, 5]. Conversely, the administration of ICIs carries the risk of potential immune-related adverse events (irAEs), some of which can manifest as severe or even life-threatening [6]. The imperative of identifying, at an early stage, those individuals unlikely to benefit from ICI therapy, thereby averting ineffective interventions and mitigating the risk of severe irAEs, has emerged as a prominent concern in the field of cancer therapy. A diverse array of predictive markers related to the ICI response has been explored, encompassing parameters such as intratumoral PD-L1 expression, tumor mutational burden (TMB), and T-cell infiltration metrics [7]. Nevertheless, devising standardized criteria for the quantification of these markers remains a formidable challenge. Moreover, the complexity of procuring tumor samples before the initiation of therapy poses an additional hurdle. To date, regulatory companion diagnostic approval for ICI treatment has been granted solely for intratumoral PD-L1 detection [8]. Consequently, the quest for novel prognostic markers assumes paramount significance as a means to enhance the clinical outcomes of cancer patients undergoing ICI-based treatment. Cachexia, a condition characterised by progressive functional decline [9], manifests in nearly half of cancer patients [10] and contributes to 20% of cancer-related mortalities [11]. Cachexia is defined as a weight loss of > 5% over 6 months or a weight loss of >2% if the body mass index (BMI) is <20 kg/m2 in the absence of simple starvation [12]. Recent studies demonstrated that cachexia is related to reduced effectiveness of chemotherapy, surgery, and targeted treatment in tumor patients [13–15]. It is still unknown how cachexia affects the effectiveness of ICIs in tumor patients. Thus, the objective of our study was to comprehensively estimate the impact of cachexia on ICI-treated cancer patients. The outcomes of this research will contribute to the development of effective treatment strategies that enable precise and cost-effective therapies with minimal adverse effects.
Methods
Search strategy
The preferred reporting items for systematic reviews and meta-analyses criteria were used in this work [16]. From the beginning of each database through July 2023, we searched PubMed, EMBASE, and the Cochrane Library for published papers on the connection between cachexia and ICI efficacy. MeSH terms and keywords such as “Immune Checkpoint Inhibitors (MeSH)”, “PD-1 Inhibitors”, “PD-L1 Inhibitors”, “CTLA-4 Inhibitors”, “Cachexia” (MeSH), and “Weight Loss” (MeSH) were utilised, among others. The detailed search strategy is outlined in Supplementary File 1. Additionally, the grey literature was looked into using Google Scholar, as well as the reference lists of eligible research were manually evaluated. The search results were uploaded to Endnote 20, which allows for the automated removal of duplicates as well as the manual screening of abstracts and full-text publications.
Study selection
The following criteria were met by articles in English to be included in this study: (i) cancer patients who received ICIs; (ii) research evaluating the influence of cachexia before ICI therapy; (iii) outcomes such as overall survival (OS), progression-free survival (PFS), time to treatment failure (TTF), or objective response rate (ORR) were reported. ORR was defined as the percentage of individuals who achieved complete response or partial response as best response to treatment. Besides, the diagnosis of cachexia was based on the criteria established by Fearon et al. [12]. The following were the exclusion requirements: (i) study designs such as animal studies, case reports, and conference abstracts.
Data extraction
The data extraction mainly focused on the author, year, study design, study period, study region, treatment, type of cancer, sample size, age, male and female patients, definition of cachexia, and outcomes. HRs, ORs, and 95% CIs were primarily extracted from multivariate analyses, otherwise from univariate analyses or Engauge Digitizer to extract from the Kaplan-Meier survival curve. The Newcastle-Ottawa Scale was utilized to evaluate the standard of the included research. We assigned nine points worth of quality-related criteria to the domains of patient selection, study comparability, and study endpoints. Studies with a score ≥6 were deemed to be of high quality. The above process has been independently completed and cross-checked by two authors, with senior authors consulted on any disputes.
Statistical methods
Pooled analyses were performed using Stata 15.0. Heterogeneity was estimated using Cochran’s Q test and I2 statistics. A fixed-effect model with the Inverse Variance method was utilized when p > 0.1 and I2 <50% indicated non-significant heterogeneity; otherwise, the random-effect model with the DerSimonian-Laird method was applied [17, 18]. To investigate publication bias, the funnel plot, Egger tests, and Begg tests were utilized [19, 20]. By separately removing each study, a sensitivity analysis was done to gauge the stability of the conclusions. Subgroup analyses were performed for the Cox model and the kind of cancer.
Results
Literature search results
827 records were retrieved through the electronic database search for articles (Figure 1). Before screening for titles and abstracts, we eliminated duplicate entries and articles written in languages other than English. After carefully reviewing the full texts of the 27 studies that had been chosen, 12 studies totaling 1407 patients were eventually included in our analysis (Figure 1) [21–32].
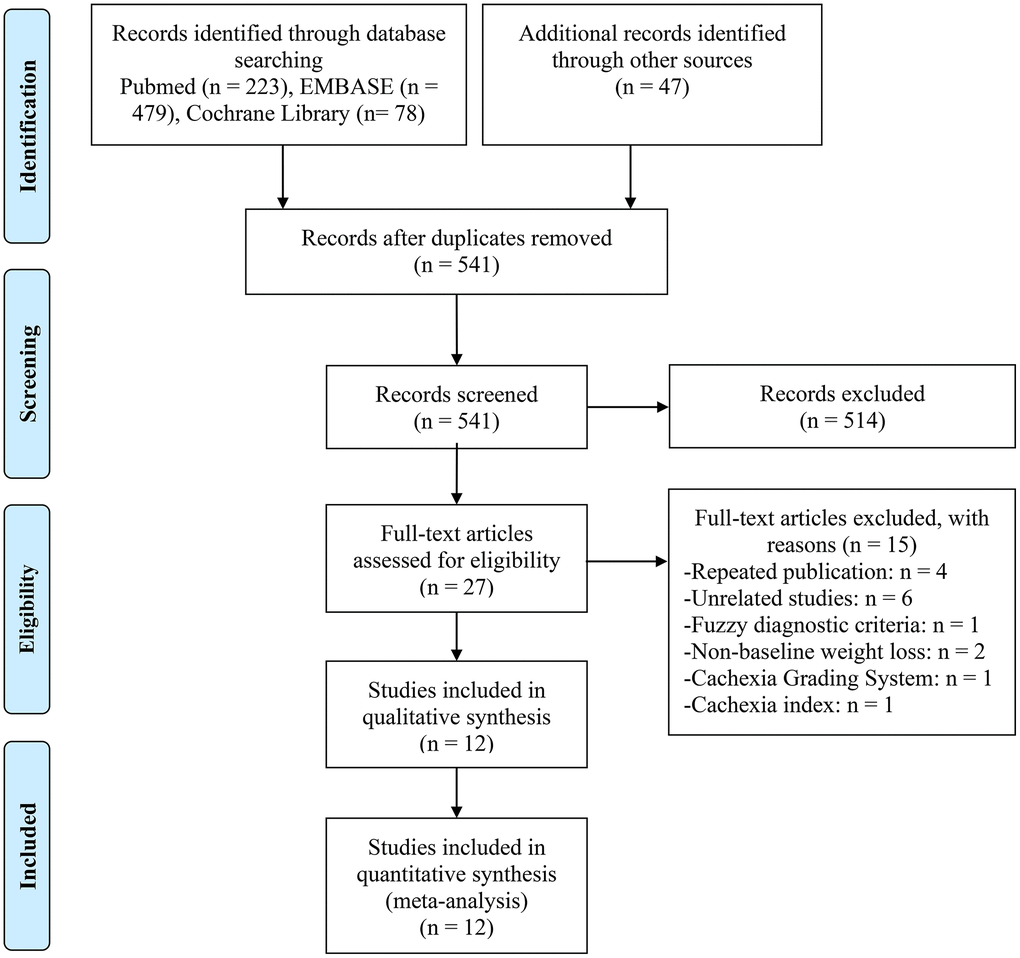
Figure 1. The flow diagram of identifying eligible studies.
Table 1 shows the characteristics of the included studies. Seven studies were performed in Japan, and one study each was conducted in the Netherlands, Italy, Greece, the USA, and France. Ten studies enrolled patients with NSCLC, and one study each enrolled patients with HNC and GC. Ten studies were retrospective, while two studies were prospective. Furthermore, the 12 studies received NOS scores ranging from 6 to 8, underscoring a minimal likelihood of bias (Table 1).
Table 1. Main characteristics of the studies included.
| Study | Study design | Study period | Study region | ICI treatment | Cancer type | Sample size | Age (years) | Sex (male/female) | Outcomes | NOS | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Willemsen et al. 2023 | R | 01/2014-03/2020 | Netherlands | Anti-CTLA-4 antibodies | HNC | 98 | 63 ± 8.0a | 83/15 | OS | 7 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Madeddu et al. 2023 | P | 04/2017-08/2021 | Italy | Pembrolizumab, Nivolumab | NSCLC | 74 | 69 ± 11.3a | 54/20 | OS, PFS | 7 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Matsuo et al. 2023 | R | 02/2016-10/2020 | Japan | Pembrolizumab, Nivolumab, Atezolizumab | NSCLC | 183 | - | 135/48 | OS, PFS, ORR | 8 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nishioka et al. 2022 | R | 05/2016-12/2018 | Japan | Pembrolizumab, Nivolumab, Atezolizumab | NSCLC | 74 | 68 (33–84)b | 58/16 | ORR | 7 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fujii et al. 2022 | R | 04/2014-06/2020 | Japan | Pembrolizumab | NSCLC | 53 | - | 42/11 | OS, ORR | 6 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Jo et al. 2022 | R | 03/2017-12/2018 | Japan | Pembrolizumab | NSCLC | 133 | - | 88/45 | OS, PFS, ORR | 7 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Miyawaki et al. 2022 | R | 12/2018-12/2020 | Japan | Anti-PD-(L)1 antibodies | NSCLC | 152 | 71 (35–88)b | 113/39 | OS, PFS | 7 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Morimoto et al. 2021 | R | 01/2019-12/2019 | Japan | Pembrolizumab, Bevacizumab, Atezolizumab | NSCLC | 196 | 69 (37–85)b | 142/54 | OS, PFS, ORR | 7 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mu et al. 2021 | R | 06/2011-08/2019 | USA | Anti-PD-(L)1 antibodies | NSCLC | 175 | 66 ± 12a | 96/79 | OS, PFS | 7 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rounis et al. 2021 | P | 2017–2020 | Greece | Pembrolizumab, Nivolumab, Atezolizumab | NSCLC | 83 | 66 (39–81)b | 70/13 | OS, PFS, ORR | 7 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Roch et al. 2020 | R | 07/2015-02/2017 | France | Pembrolizumab, Nivolumab | NSCLC | 142 | 64 ± 10.6a | 93/49 | OS, PFS | 8 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fujii et al. 2020 | R | 10/2017-12/2019 | Japan | Nivolumab | GC | 44 | - | 23/21 | OS, ORR | 6 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| amean ± standard deviation; bmedians (ranges). Abbreviations: R: retrospective study; P: prospective study; PD-(L)1: programmed cell death (ligand)-1; CTLA-4: cytotoxic T-lymphocyte-associated protein 4; HNC: head and neck cancer; NSCLC: non-small cell lung cancer; GC: gastric cancer; OS: overall survival; PFS: progression-free survival; ORR: overall response rate. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Pre-immunotherapy cachexia and OS
The effect of pre-treatment cachexia on OS in cancer patients treated with ICIs was examined in 12 cohorts, including 1333 participants. A fixed-effect model was used since the Cochran Q test and I2 statistics showed no significant heterogeneity (p = 0.214, I2 = 23.3%). The pooled results revealed that cancer patients with cachexia had significantly poorer OS than those without cachexia (HR = 1.88, 95% CI: 1.59–2.22, p < 0.001, Figure 2). NSCLC patients were recruited in 10 cohorts, and NSCLC patients with cachexia have shorter OS (I2 = 15.4%, p = 0.301; HR = 1.97, 95% CI: 1.64–2.36, p < 0.001, Supplementary Figure 1).
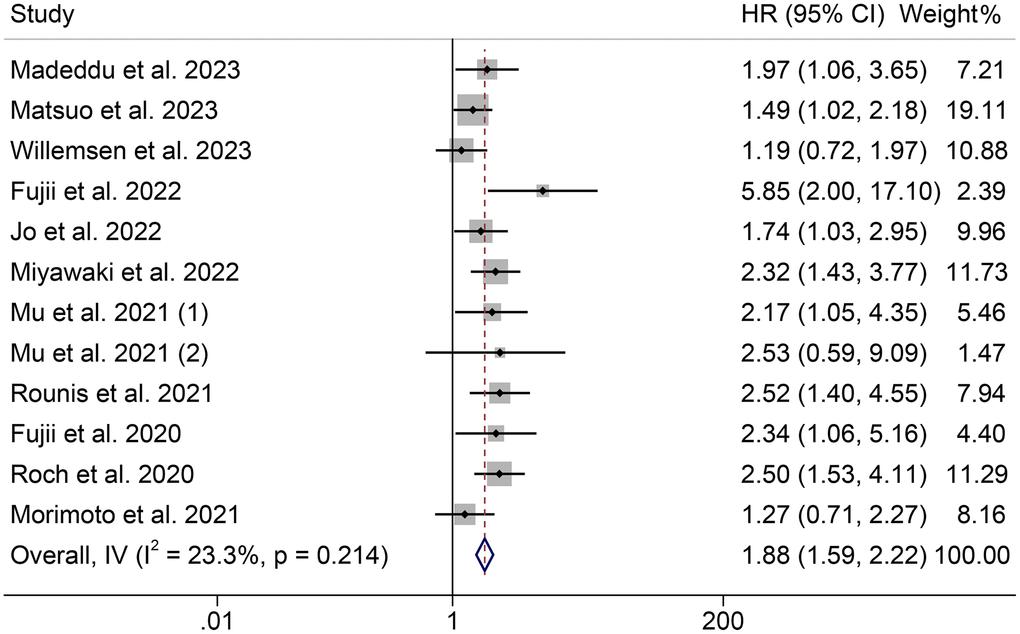
Figure 2. Forest plots of the relationship between cachexia and overall survival. Abbreviations: HR: hazard ratio; CI: confidence interval; IV: Inverse Variance method.
The HRs for OS according to the Cox proportional hazards model used are shown in Figure 3. Univariate and multivariate analyses were conducted in six cohorts, respectively. The HRs (95% CIs) were 2.01 (1.54–2.62) for univariate analyses and 1.80 (1.46–2.23) for multivariate analyses. There were no significant differences among the different models (P = 0.533).
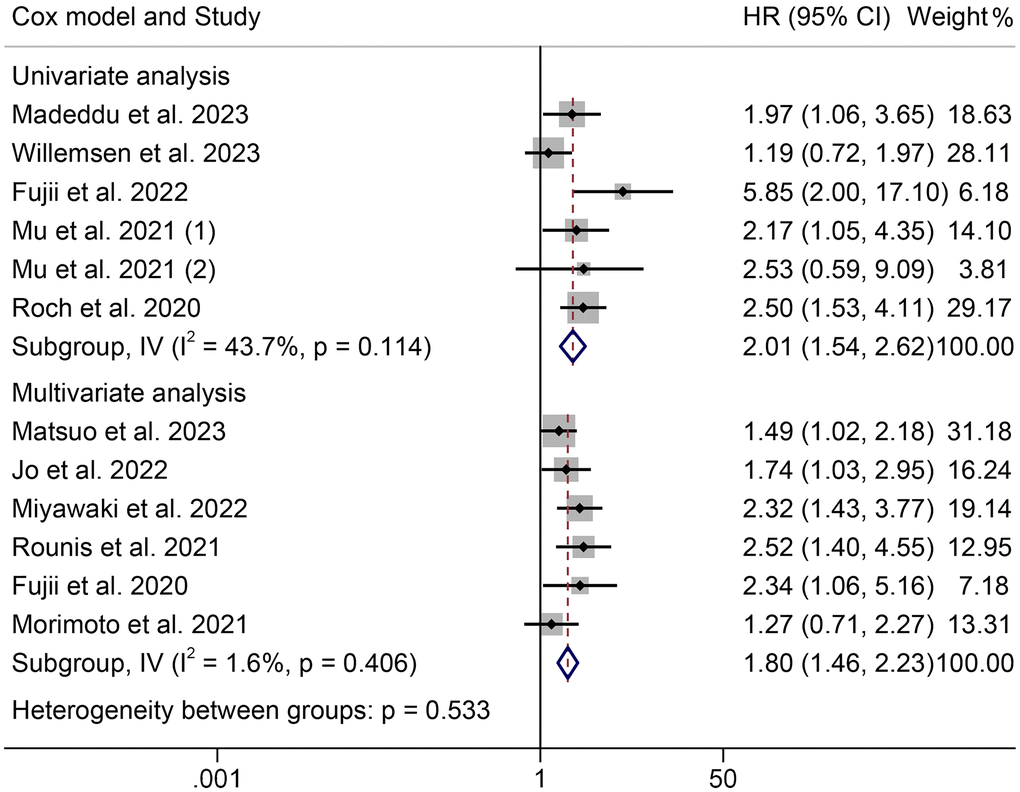
Figure 3. Forest plots of the association between cachexia and overall survival in the multivariate and univariate analysis. Abbreviations: HR: hazard ratio; CI: confidence interval; IV: Inverse Variance method.
Pre-treatment cachexia and PFS/TTF
The association between cachexia and PFS was investigated using survival data from nine studies with 1138 participants. Notably, those patients were all diagnosed with NSCLC. As shown in Figure 4, there was no significant heterogeneity among studies (I2 = 0.0%, p = 0.585), so a fixed-effect model was used. The results revealed that cachexia was significantly associated with worse PFS (HR = 1.84, 95% CI: 1.59–2.12, p < 0.001). The results were consistent with the above finding in univariate (HR = 1.90, 95% CI: 1.56–2.30, p < 0.001) and multivariate (HR = 1.76, 95% CI: 1.41–2.19, p < 0.001) analyses (Figure 5). Besides, we also found that patients with cachexia had considerably shorter TTF than those without cachexia (HR = 2.15, 95% CI: 1.32–3.50, p = 0.002, Supplementary Figure 2).
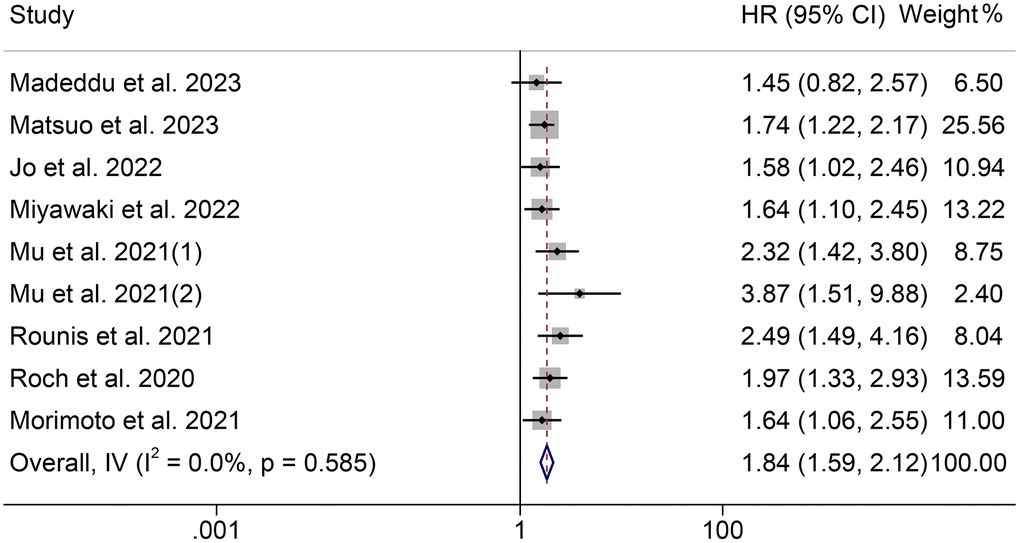
Figure 4. Forest plots of the relationship between cachexia and progression-free survival. Abbreviations: HR: hazard ratio; CI: confidence interval; IV: Inverse Variance method.
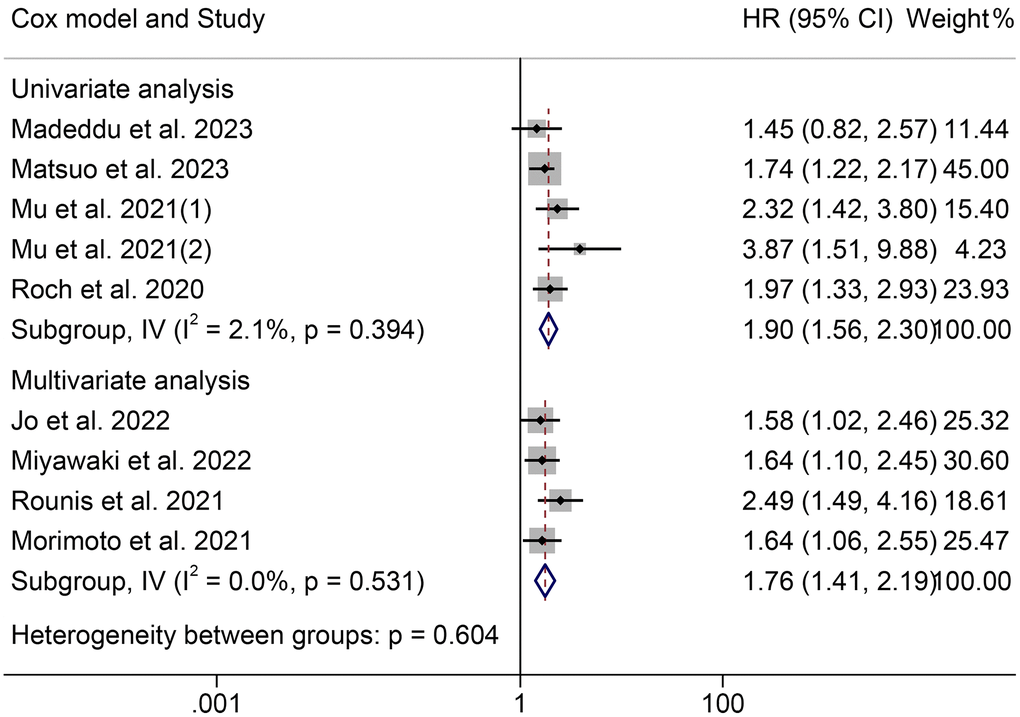
Figure 5. Forest plots of the association between cachexia and progression-free survival in the multivariate and univariate analysis. Abbreviations: HR: hazard ratio; CI: confidence interval; IV: Inverse Variance method.
Pre-immunotherapy cachexia and ORR
We also assessed the relationship between cachexia and the overall response rate. Due to significant heterogeneity, we used a random effects model (I2 = 61.6%, p = 0.016). The pooled analysis indicated that there was a trend towards a lower ORR in cancer patients with cachexia compared to those without cachexia, although not statistically significant (OR = 0.59, 95% CI: 0.32–1.09, p = 0.093, Figure 6). Besides, pooled analysis that included only NSCLC patients was consistent with the above findings (OR = 0.55, 95% CI: 0.29–1.06, p = 0.076, Supplementary Figure 3).
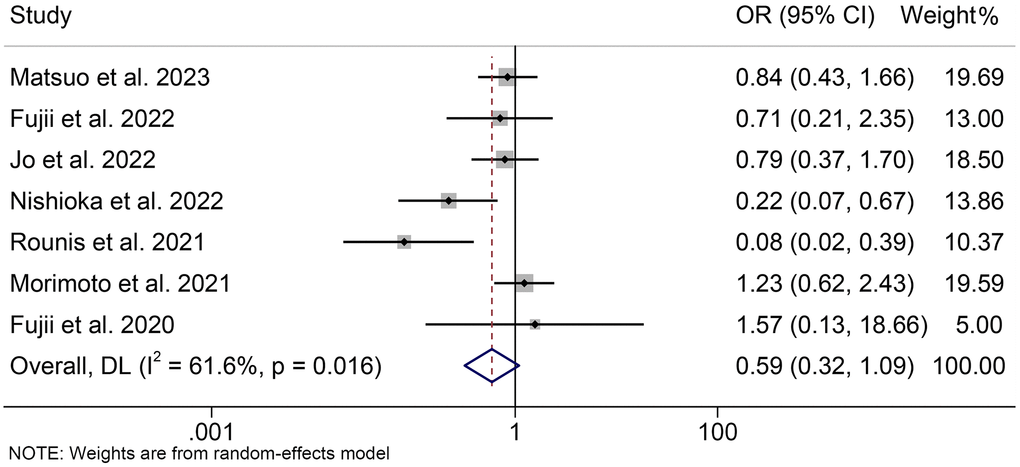
Figure 6. Forest plots of the relationship between cachexia and objective response rate. Abbreviations: OR: odds ratio; CI: confidence interval; DL: DerSimonian-Laird method.
Sensitivity analysis and publication bias
OS and PFS, the primary outcome indicators in this study, were subjected to sensitivity analyses and publication bias tests. The sensitivity analysis results are provided in Figure 7A, 7B. When any individual study was eliminated from the analysis, the pooled HRs for OS and PFS were similar. In addition, sensitivity analysis of ORR also confirmed that the above results are stable (Supplementary Figure 4). The funnel plot, Begg’s test (OS, p = 0.224; PFS, p = 0.602) and Egger’s test (OS, p = 0.077; PFS, p = 0.134) did not reveal any publication bias in OS (Supplementary Figure 5A) and PFS (Supplementary Figure 5B).
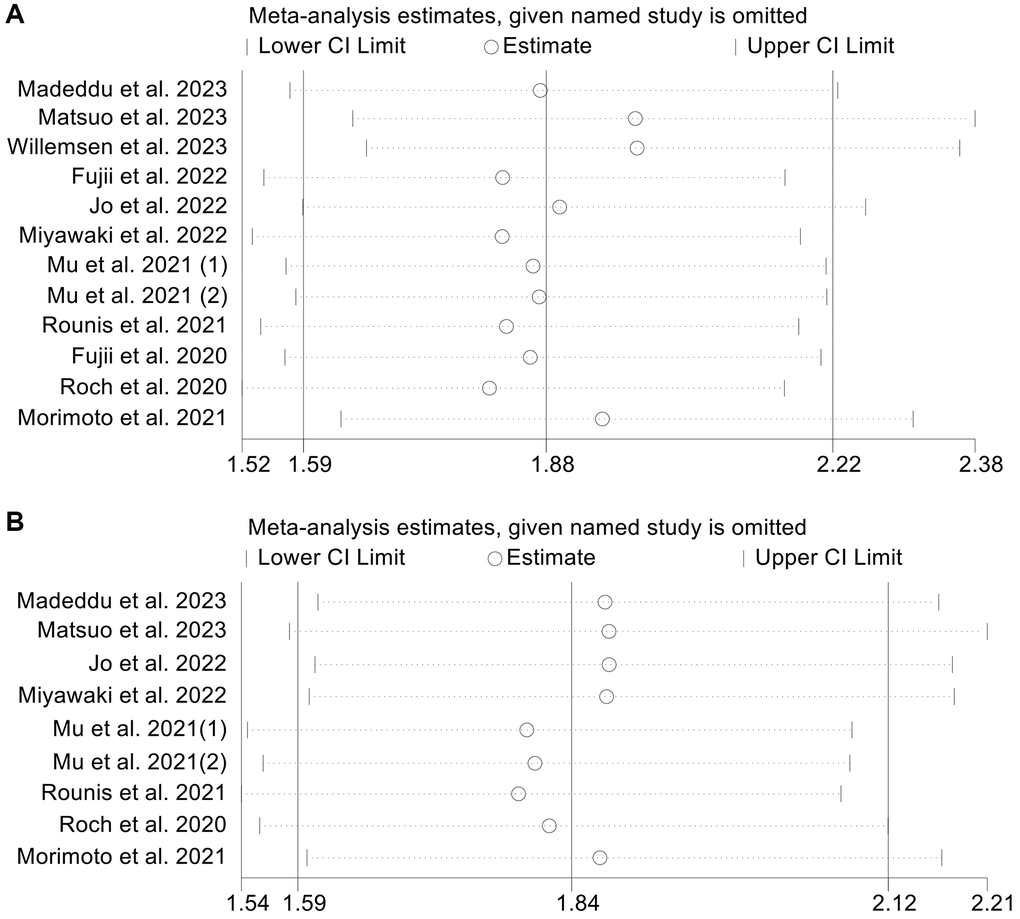
Figure 7. Sensitivity analysis of the association between cachexia and overall survival (A) and progression-free survival (B). Abbreviations: HR: hazard ratio; CI: confidence interval.
Discussion
With its outstanding efficacy and safety, immunotherapy using PD-(L)1 and CTLA-4 inhibitors has completely changed the way cancer patients are treated [33, 34]. However, it has been discovered that the therapeutic success of ICIs varies significantly among cancer patients, and there is still a lack of specific and reliable predictors of ICI efficacy. Cachexia, in particular, is frequent in cancer patients. This meta-analysis represents the first attempt, to our knowledge, to systematically assess the correlations between cachexia and the clinical outcomes of ICI-treated patients. The pooled data demonstrated that cachexia was significantly associated with a poorer OS and PFS.
The metabolic alterations linked to cachexia can reduce anti-tumor immunity. The release of pro-inflammatory cytokines, including TNF-α, IL-6, and IL-1, is provoked by cancer cells, which sets off a chain reaction that results in weight loss by causing the breakdown of skeletal muscle and adipose tissue as well as anorexia [12]. These factors also upregulate the release of corticotropin-releasing hormone while concurrently suppressing ghrelin, intensifying the loss of appetite [35]. Flint and colleagues demonstrated that tumor-induced IL-6 suppresses the production of hepatic ketone bodies, resulting in the significant secretion of glucocorticoids during periods of caloric deficiency [36]. Moreover, their research unveiled that this stress-induced hormonal response stifled immune activity within tumors, ultimately culminating in the failure of anticancer immunotherapy [36].
Cachexia may reduce the efficacy of ICIs in patients with NSCLC and high PD-L1 expression, according to a previous single-center retrospective analysis [37, 38]. This may be due to the fact that in NSCLC patients with cachexia, IL-6, IL-1, and TNF-α reduce CD8+ tumor infiltrating lymphocytes and anti-tumor immunity [37]. Studies have also confirmed that inhibition of cytokine pathways associated with cachexia formation has been shown to enhance the anti-cancer immune response [39, 40], and the combined blocking of specific cachexia-promoting mediators and the PD-1/PD-L1 axis has been shown to have a synergistic effect [41, 42]. Our conclusions are consistent with the above findings that cachexia shortens OS and PFS after cancer patients receiving ICI therapy.
In the context of cachexia management, a spectrum of therapeutic options, encompassing both pharmaceutical and non-pharmaceutical approaches, is available. Within the realm of pharmaceutical treatments, corticosteroids, non-steroidal anti-inflammatory drugs, and progesterone have demonstrated efficacy [43, 44]. It’s worth noting, however, that these treatments are accompanied by the risk of adverse events [43, 44]. Anamorelin, an orally administered, high-affinity, selective ghrelin receptor agonist, has exhibited the ability to significantly increase lean body mass in patients with advanced NSCLC, although it did not yield a significant improvement in handgrip strength [45]. Non-pharmacological interventions encompass dietary management [46] and structured physical exercise regimens [47]. Yet, physical exercise poses a challenge, as many advanced cancer patients tend to discontinue participation [48]. Additionally, interventions solely relying on either pharmaceutical or dietary approaches are less than fully effective [49]. Therefore, combining medication with diet management and physical exercise is an urgently needed holistic approach to improve ICI treatment outcomes for cancer patients with cachexia.
Some limitations of the present meta-analysis are to be addressed. First, most of the included studies were retrospective. Most of the included studies collected patient data retrospectively. The diagnostic criteria for cachexia were not entirely consistent between studies, although each study stated that their diagnostic criteria for cachexia referenced Fearon et al. [12]. Finally, although we found that cachexia was associated with a lower ORR, it was not statistically significant and more studies need to be included to explore the relationship.
Conclusion
Cancer cachexia is related to poor clinical outcomes in ICI-treated cancer patients and may be beneficial in identifying therapy indications. Early intervention to improve cachexia is thought to be significant for ICI treatment success and should be considered in the future.
Author Contributions
YY, LY, HT, WZ, and LJ conceived and designed the study. YY, LY, and HT were responsible for the collection and assembly of data, data analysis, and interpretation. YY, LY, and HT were involved in writing the manuscript. YY, LY, HT, WZ, and LJ revised the manuscript.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Funding
No funding was used for this paper.
References
- 1. Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011; 144:646–74. https://doi.org/10.1016/j.cell.2011.02.013 [PubMed]
- 2. Zhang J, Xiao Y, Zhang J, Yang Y, Zhang L, Liang F. Recent advances of engineered oncolytic viruses-based combination therapy for liver cancer. J Transl Med. 2024; 22:3. https://doi.org/10.1186/s12967-023-04817-w [PubMed]
- 3. Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012; 12:252–64. https://doi.org/10.1038/nrc3239 [PubMed]
- 4. Ribas A, Wolchok JD. Cancer immunotherapy using checkpoint blockade. Science. 2018; 359:1350–5. https://doi.org/10.1126/science.aar4060 [PubMed]
- 5. O'Donnell JS, Teng MWL, Smyth MJ. Cancer immunoediting and resistance to T cell-based immunotherapy. Nat Rev Clin Oncol. 2019; 16:151–67. https://doi.org/10.1038/s41571-018-0142-8 [PubMed]
- 6. Dolladille C, Ederhy S, Sassier M, Cautela J, Thuny F, Cohen AA, Fedrizzi S, Chrétien B, Da-Silva A, Plane AF, Legallois D, Milliez PU, Lelong-Boulouard V, Alexandre J. Immune Checkpoint Inhibitor Rechallenge After Immune-Related Adverse Events in Patients With Cancer. JAMA Oncol. 2020; 6:865–71. https://doi.org/10.1001/jamaoncol.2020.0726 [PubMed]
- 7. Schalper KA, Carleton M, Zhou M, Chen T, Feng Y, Huang SP, Walsh AM, Baxi V, Pandya D, Baradet T, Locke D, Wu Q, Reilly TP, et al. Elevated serum interleukin-8 is associated with enhanced intratumor neutrophils and reduced clinical benefit of immune-checkpoint inhibitors. Nat Med. 2020; 26:688–92. https://doi.org/10.1038/s41591-020-0856-x [PubMed]
- 8. Havel JJ, Chowell D, Chan TA. The evolving landscape of biomarkers for checkpoint inhibitor immunotherapy. Nat Rev Cancer. 2019; 19:133–50. https://doi.org/10.1038/s41568-019-0116-x [PubMed]
- 9. Fearon K, Arends J, Baracos V. Understanding the mechanisms and treatment options in cancer cachexia. Nat Rev Clin Oncol. 2013; 10:90–9. https://doi.org/10.1038/nrclinonc.2012.209 [PubMed]
- 10. Vagnildhaug OM, Balstad TR, Almberg SS, Brunelli C, Knudsen AK, Kaasa S, Thronæs M, Laird B, Solheim TS. A cross-sectional study examining the prevalence of cachexia and areas of unmet need in patients with cancer. Support Care Cancer. 2018; 26:1871–80. https://doi.org/10.1007/s00520-017-4022-z [PubMed]
- 11. Tisdale MJ. Cachexia in cancer patients. Nat Rev Cancer. 2002; 2:862–71. https://doi.org/10.1038/nrc927 [PubMed]
- 12. Fearon K, Strasser F, Anker SD, Bosaeus I, Bruera E, Fainsinger RL, Jatoi A, Loprinzi C, MacDonald N, Mantovani G, Davis M, Muscaritoli M, Ottery F, et al. Definition and classification of cancer cachexia: an international consensus. Lancet Oncol. 2011; 12:489–95. https://doi.org/10.1016/S1470-2045(10)70218-7 [PubMed]
- 13. von Haehling S, Anker SD. Prevalence, incidence and clinical impact of cachexia: facts and numbers-update 2014. J Cachexia Sarcopenia Muscle. 2014; 5:261–3. https://doi.org/10.1007/s13539-014-0164-8 [PubMed]
- 14. Brown LR, Sayers J, Yule MS, Drake TM, Dolan RD, McMillan DC, Laird BJA, Wigmore SJ, Skipworth RJE. The prognostic impact of pre-treatment cachexia in resectional surgery for oesophagogastric cancer: a meta-analysis and meta-regression. Br J Surg. 2023; 110:1703–11. https://doi.org/10.1093/bjs/znad239 [PubMed]
- 15. Nishie K, Yamamoto S, Nagata C, Koizumi T, Hanaoka M. Anamorelin for advanced non-small-cell lung cancer with cachexia: Systematic review and meta-analysis. Lung Cancer. 2017; 112:25–34. https://doi.org/10.1016/j.lungcan.2017.07.023 [PubMed]
- 16. Moher D, Liberati A, Tetzlaff J, Altman DG, and PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009; 6:e1000097. https://doi.org/10.1371/journal.pmed.1000097 [PubMed]
- 17. Lilong Z, Kuang T, Li M, Li X, Hu P, Deng W, Wang W. Sarcopenia affects the clinical efficacy of immune checkpoint inhibitors in patients with gastrointestinal cancers. Clin Nutr. 2024; 43:31–41. https://doi.org/10.1016/j.clnu.2023.11.009 [PubMed]
- 18. Zhang L, Ma W, Qiu Z, Kuang T, Wang K, Hu B, Wang W. Prognostic nutritional index as a prognostic biomarker for gastrointestinal cancer patients treated with immune checkpoint inhibitors. Front Immunol. 2023; 14:1219929. https://doi.org/10.3389/fimmu.2023.1219929 [PubMed]
- 19. Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997; 315:629–34. https://doi.org/10.1136/bmj.315.7109.629 [PubMed]
- 20. Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994; 50:1088–101. [PubMed]
- 21. Fujii H, Makiyama A, Iihara H, Okumura N, Yamamoto S, Imai T, Arakawa S, Kobayashi R, Tanaka Y, Yoshida K, Suzuki A. Cancer Cachexia Reduces the Efficacy of Nivolumab Treatment in Patients With Advanced Gastric Cancer. Anticancer Res. 2020; 40:7067–75. https://doi.org/10.21873/anticanres.14734 [PubMed]
- 22. Roch B, Coffy A, Jean-Baptiste S, Palaysi E, Daures JP, Pujol JL, Bommart S. Cachexia - sarcopenia as a determinant of disease control rate and survival in non-small lung cancer patients receiving immune-checkpoint inhibitors. Lung Cancer. 2020; 143:19–26. https://doi.org/10.1016/j.lungcan.2020.03.003 [PubMed]
- 23. Morimoto K, Uchino J, Yokoi T, Kijima T, Goto Y, Nakao A, Hibino M, Takeda T, Yamaguchi H, Takumi C, Takeshita M, Chihara Y, Yamada T, et al. Impact of cancer cachexia on the therapeutic outcome of combined chemoimmunotherapy in patients with non-small cell lung cancer: a retrospective study. Oncoimmunology. 2021; 10:1950411. https://doi.org/10.1080/2162402X.2021.1950411 [PubMed]
- 24. Mu W, Katsoulakis E, Whelan CJ, Gage KL, Schabath MB, Gillies RJ. Radiomics predicts risk of cachexia in advanced NSCLC patients treated with immune checkpoint inhibitors. Br J Cancer. 2021; 125:229–39. https://doi.org/10.1038/s41416-021-01375-0 [PubMed]
- 25. Rounis K, Makrakis D, Tsigkas AP, Georgiou A, Galanakis N, Papadaki C, Monastirioti A, Vamvakas L, Kalbakis K, Vardakis N, Kontogianni M, Gioulbasanis I, Mavroudis D, Agelaki S. Cancer cachexia syndrome and clinical outcome in patients with metastatic non-small cell lung cancer treated with PD-1/PD-L1 inhibitors: results from a prospective, observational study. Transl Lung Cancer Res. 2021; 10:3538–49. https://doi.org/10.21037/tlcr-21-460 [PubMed]
- 26. Fujii H, Araki A, Iihara H, Kaito D, Hirose C, Kinomura M, Yamazaki M, Endo J, Inui T, Yanase K, Sasaki Y, Gomyo T, Sakai C, et al. Cancer cachexia as a determinant of efficacy of first-line pembrolizumab in patients with advanced non-small cell lung cancer. Mol Clin Oncol. 2022; 16:91. https://doi.org/10.3892/mco.2022.2524 [PubMed]
- 27. Jo H, Yoshida T, Horinouchi H, Yagishita S, Matsumoto Y, Shinno Y, Okuma Y, Goto Y, Yamamoto N, Takahashi K, Motoi N, Ohe Y. Prognostic significance of cachexia in advanced non-small cell lung cancer patients treated with pembrolizumab. Cancer Immunol Immunother. 2022; 71:387–98. https://doi.org/10.1007/s00262-021-02997-2 [PubMed]
- 28. Miyawaki T, Naito T, Doshita K, Kodama H, Mori M, Nishioka N, Iida Y, Miyawaki E, Mamesaya N, Kobayashi H, Omori S, Ko R, Wakuda K, et al. Predicting the efficacy of first-line immunotherapy by combining cancer cachexia and tumor burden in advanced non-small cell lung cancer. Thorac Cancer. 2022; 13:2064–74. https://doi.org/10.1111/1759-7714.14529 [PubMed]
- 29. Nishioka N, Naito T, Miyawaki T, Yabe M, Doshita K, Kodama H, Miyawaki E, Iida Y, Mamesaya N, Kobayashi H, Omori S, Ko R, Wakuda K, et al. Impact of losing adipose tissue on outcomes from PD-1/PD-L1 inhibitor monotherapy in non-small cell lung cancer. Thorac Cancer. 2022; 13:1496–504. https://doi.org/10.1111/1759-7714.14421 [PubMed]
- 30. Madeddu C, Busquets S, Donisi C, Lai E, Pretta A, López-Soriano FJ, Argilés JM, Scartozzi M, Macciò A. Effect of Cancer-Related Cachexia and Associated Changes in Nutritional Status, Inflammatory Status, and Muscle Mass on Immunotherapy Efficacy and Survival in Patients with Advanced Non-Small Cell Lung Cancer. Cancers (Basel). 2023; 15:1076. https://doi.org/10.3390/cancers15041076 [PubMed]
- 31. Matsuo N, Azuma K, Murotani K, Murata D, Matama G, Kawahara A, Kojima T, Tokito T, Hoshino T. Prognostic effect of cachexia in patients with non-small cell lung cancer receiving immune checkpoint inhibitors. Thorac Cancer. 2023; 14:1362–7. https://doi.org/10.1111/1759-7714.14881 [PubMed]
- 32. Willemsen ACH, De Moor N, Van Dessel J, Baijens LWJ, Bila M, Hauben E, van den Hout MFC, Vander Poorten V, Hoeben A, Clement PM, Schols AMW. The predictive and prognostic value of weight loss and body composition prior to and during immune checkpoint inhibition in recurrent or metastatic head and neck cancer patients. Cancer Med. 2023; 12:7699–712. https://doi.org/10.1002/cam4.5522 [PubMed]
- 33. Boku N, Ryu MH, Kato K, Chung HC, Minashi K, Lee KW, Cho H, Kang WK, Komatsu Y, Tsuda M, Yamaguchi K, Hara H, Fumita S, et al. Safety and efficacy of nivolumab in combination with S-1/capecitabine plus oxaliplatin in patients with previously untreated, unresectable, advanced, or recurrent gastric/gastroesophageal junction cancer: interim results of a randomized, phase II trial (ATTRACTION-4). Ann Oncol. 2019; 30:250–8. https://doi.org/10.1093/annonc/mdy540 [PubMed]
- 34. Santini D, Zeppola T, Russano M, Citarella F, Anesi C, Buti S, Tucci M, Russo A, Sergi MC, Adamo V, Stucci LS, Bersanelli M, Mazzaschi G, et al. PD-1/PD-L1 checkpoint inhibitors during late stages of life: an ad-hoc analysis from a large multicenter cohort. J Transl Med. 2021; 19:270. https://doi.org/10.1186/s12967-021-02937-9 [PubMed]
- 35. Fujitsuka N, Asakawa A, Uezono Y, Minami K, Yamaguchi T, Niijima A, Yada T, Maejima Y, Sedbazar U, Sakai T, Hattori T, Kase Y, Inui A. Potentiation of ghrelin signaling attenuates cancer anorexia-cachexia and prolongs survival. Transl Psychiatry. 2011; 1:e23. https://doi.org/10.1038/tp.2011.25 [PubMed]
- 36. Flint TR, Janowitz T, Connell CM, Roberts EW, Denton AE, Coll AP, Jodrell DI, Fearon DT. Tumor-Induced IL-6 Reprograms Host Metabolism to Suppress Anti-tumor Immunity. Cell Metab. 2016; 24:672–84. https://doi.org/10.1016/j.cmet.2016.10.010 [PubMed]
- 37. Miyawaki T, Naito T, Kodama A, Nishioka N, Miyawaki E, Mamesaya N, Kawamura T, Kobayashi H, Omori S, Wakuda K, Ono A, Kenmotsu H, Murakami H, et al. Desensitizing Effect of Cancer Cachexia on Immune Checkpoint Inhibitors in Patients With Advanced NSCLC. JTO Clin Res Rep. 2020; 1:100020. https://doi.org/10.1016/j.jtocrr.2020.100020 [PubMed]
- 38. Kovács SA, Győrffy B. Transcriptomic datasets of cancer patients treated with immune-checkpoint inhibitors: a systematic review. J Transl Med. 2022; 20:249. https://doi.org/10.1186/s12967-022-03409-4 [PubMed]
- 39. Rautela J, Dagley LF, de Oliveira CC, Schuster IS, Hediyeh-Zadeh S, Delconte RB, Cursons J, Hennessy R, Hutchinson DS, Harrison C, Kita B, Vivier E, Webb AI, et al. Therapeutic blockade of activin-A improves NK cell function and antitumor immunity. Sci Signal. 2019; 12:eaat7527. https://doi.org/10.1126/scisignal.aat7527 [PubMed]
- 40. Tinoco R, Carrette F, Barraza ML, Otero DC, Magaña J, Bosenberg MW, Swain SL, Bradley LM. PSGL-1 Is an Immune Checkpoint Regulator that Promotes T Cell Exhaustion. Immunity. 2016; 44:1190–203. https://doi.org/10.1016/j.immuni.2016.04.015 [PubMed]
- 41. Li J, Xu J, Yan X, Jin K, Li W, Zhang R. Targeting Interleukin-6 (IL-6) Sensitizes Anti-PD-L1 Treatment in a Colorectal Cancer Preclinical Model. Med Sci Monit. 2018; 24:5501–8. https://doi.org/10.12659/MSM.907439 [PubMed]
- 42. Kaplanov I, Carmi Y, Kornetsky R, Shemesh A, Shurin GV, Shurin MR, Dinarello CA, Voronov E, Apte RN. Blocking IL-1β reverses the immunosuppression in mouse breast cancer and synergizes with anti-PD-1 for tumor abrogation. Proc Natl Acad Sci U S A. 2019; 116:1361–9. https://doi.org/10.1073/pnas.1812266115 [PubMed]
- 43. Ruiz Garcia V, López-Briz E, Carbonell Sanchis R, Gonzalvez Perales JL, Bort-Marti S. Megestrol acetate for treatment of anorexia-cachexia syndrome. Cochrane Database Syst Rev. 2013; 2013:CD004310. https://doi.org/10.1002/14651858.CD004310.pub3 [PubMed]
- 44. Madeddu C, Mantovani G, Gramignano G, Macciò A. Advances in pharmacologic strategies for cancer cachexia. Expert Opin Pharmacother. 2015; 16:2163–77. https://doi.org/10.1517/14656566.2015.1079621 [PubMed]
- 45. Temel JS, Abernethy AP, Currow DC, Friend J, Duus EM, Yan Y, Fearon KC. Anamorelin in patients with non-small-cell lung cancer and cachexia (ROMANA 1 and ROMANA 2): results from two randomised, double-blind, phase 3 trials. Lancet Oncol. 2016; 17:519–31. https://doi.org/10.1016/S1470-2045(15)00558-6 [PubMed]
- 46. Balstad TR, Solheim TS, Strasser F, Kaasa S, Bye A. Dietary treatment of weight loss in patients with advanced cancer and cachexia: a systematic literature review. Crit Rev Oncol Hematol. 2014; 91:210–21. https://doi.org/10.1016/j.critrevonc.2014.02.005 [PubMed]
- 47. Grande AJ, Silva V, Maddocks M. Exercise for cancer cachexia in adults: Executive summary of a Cochrane Collaboration systematic review. J Cachexia Sarcopenia Muscle. 2015; 6:208–11. https://doi.org/10.1002/jcsm.12055 [PubMed]
- 48. Oldervoll LM, Loge JH, Lydersen S, Paltiel H, Asp MB, Nygaard UV, Oredalen E, Frantzen TL, Lesteberg I, Amundsen L, Hjermstad MJ, Haugen DF, Paulsen Ø, Kaasa S. Physical exercise for cancer patients with advanced disease: a randomized controlled trial. Oncologist. 2011; 16:1649–57. https://doi.org/10.1634/theoncologist.2011-0133 [PubMed]
- 49. Del Fabbro E. More is better: a multimodality approach to cancer cachexia. Oncologist. 2010; 15:119–21. https://doi.org/10.1634/theoncologist.2010-0019 [PubMed]


