Introduction
Increasing lifespan and promoting healthy living into old age are among top priorities of health care systems all over the world. Although lifespan is steadily improving, an increasing proportion of the population is affected by multiple chronic diseases. Moreover, in recent years, the global COVID-19 pandemic poses a heavy burden on public health and accounts for substantial mortality and morbidity [1, 2]. According to the estimation of World Health Organization, around 14.83 million excess deaths globally were due to COVID-19 [1].
Proteins are the building blocks of life, important for etiology of disease and often used as drug targets for treatment. However, the specific proteins involved in the complex ageing process and COVID-19 risk are largely unclear. The dysregulation in inflammatory and immunomodulatory response has been thought to play a key role in multiple diseases and disorders [3], as well as in COVID-19 [4]. Several proteins related to inflammation and immune function, such as CXCL14 [5] and soluble HLA-G (sHLA-G) [6], have been identified by comparing patients with and without COVID-19. However, these associations may be confounded by some other characteristics, such as socioeconomic position. Moreover, proteins in other pathways, such as cardiometabolism, may also affect COVID-19 and longevity. Therefore, a comprehensive examination of proteins using advanced causal inference methods robust to confounding is needed.
Mendelian randomization (MR) uses genetic variants as instrument variables (IVs) to assess the causal role of proteins in COVID-19 and lifespan. As the genotypes are determined at conception and randomly allocated, MR estimates are thus not subject to residual confounding [7]. Using MR, some studies explored the associations of proteins with COVID-19 [8, 9] or with healthspan [10], but not both. In addition, the genetic instruments of these proteins are generally based on relatively smaller genome-wide association study (GWAS) (~3,301 people) [8–10]. Recently, the UK Biobank Pharma Proteomics Project (UKB-PPP), a collaboration between the UK Biobank (UKB) and thirteen biopharmaceutical companies has measured the plasma proteomic profiles in 54,306 UKB participants. In this study, we conducted MR analysis using genetic instruments from much larger GWAS of proteins and applied to large GWAS of COVID-19 (severe COVID-19, COVID-19 hospitalization and SARS-COV2 infection), healthspan and lifespan (mother’s and father’s attained age), to identify proteins causally related to COVID-19, healthspan and lifespan. We included both COVID-19 and healthspan and lifespan in the outcome, because COVID-19 which occurred in recent years reflects a new threat to longevity, whilst healthspan and lifespan reflect overall morbidity and mortality.
Methods
Study design
We used proteome-wide MR study to identify proteins causally related to COVID-19, healthspan and lifespan. To better understand the function of these proteins, we checked the function of each selected protein. To assess the drug repurposing opportunity, we also checked whether these proteins are targeted by existing drugs. The flow chart of the study design was shown in Figure 1.
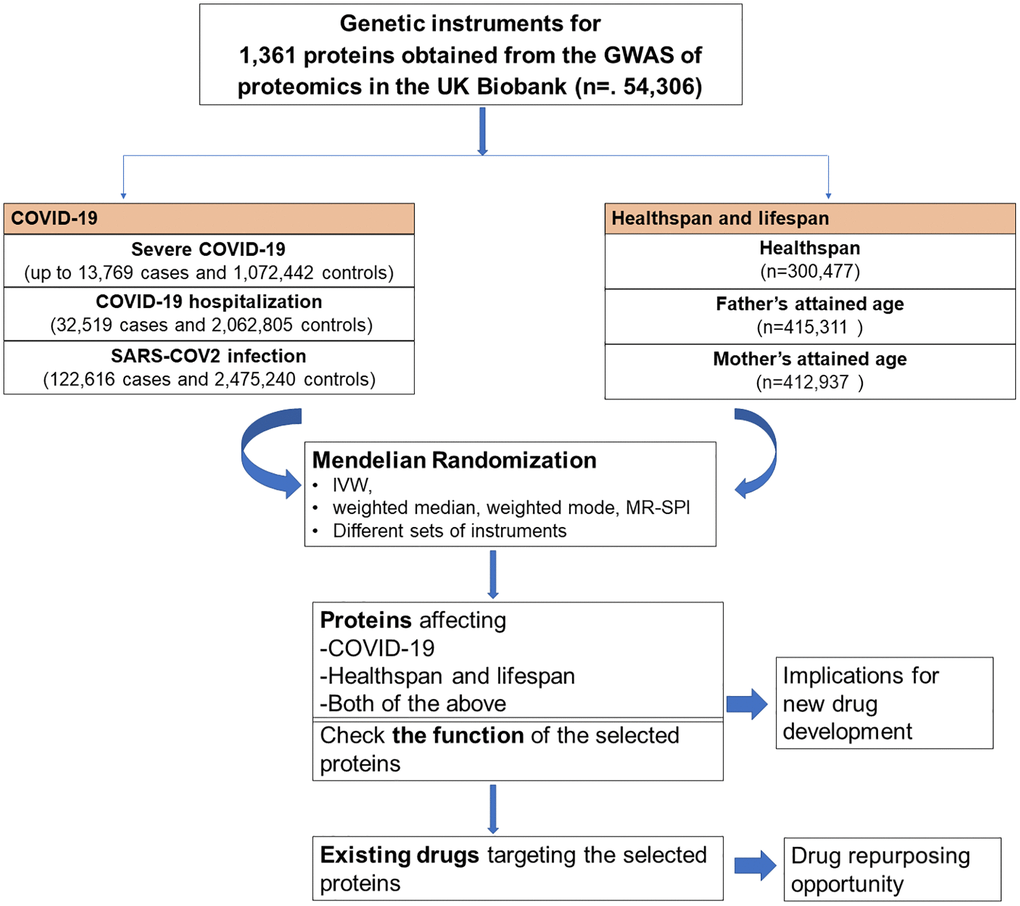
Figure 1. Flow chart of the study design.
Proteomics data from UK Biobank
The proteomic profiling measured 1,472 protein analytes and captured 1,463 unique proteins using the Olink Explore 1536 platform in 54,306 plasma samples in the UK Biobank. The blood samples were from a randomised subset of 46,673 UKB participants at baseline visit, 6,385 individuals at baseline selected by the UKB-PPP consortium and 1,268 individuals who participated in the COVID-19 repeat imaging study at multiple visits. No batch effects, plate effects or abnormalities in protein coefficients of variation were observed [11]. We used significant (p < 3.4 × 10−11) and independent (r2 < 0.01) single nucleotide polymorphisms (SNPs) as instruments provided by the study of Sun et al. [1]. The details of the samples and sample selection in UKB-PPP were shown in Supplementary Methods. Up to 1,361 proteins with genetic instruments available in at least one of the outcome datasets were included in the analysis. The genetic instruments for proteins used in the MR analysis are provided in Supplementary Table 1.
Genetic associations with COVID-19
We obtained genetic associations of these genetic instruments with severe COVID-19 (up to 13,769 cases and 1,072,442 controls), hospitalization (up to 32,519 cases and 2,062,805 controls) and SARS-COV2 infection (up to 122,616 cases and 2,475,240 controls), from the latest GWAS results provided by the COVID-19 host genetics initiative [12]. Severe COVID-19 was defined as (1) hospitalization with laboratory confirmed SARS-CoV-2 infection based on RNA and/or serology and (2) hospitalization with COVID19 as the primary reason for admission, and (3) followed by death or respiratory support. COVID-19 hospitalization was defined as hospitalization with laboratory confirmed SARS-CoV-2 infection due to corona-related symptoms. COVID-19 infection was defined as (1) laboratory confirmed SARS-CoV-2 infection (RNA and/or serology based), or (2) physician diagnosis of COVID-19, or (3) self-report as COVID-19 positive. The GWAS was adjusted for age, age square, sex and the interaction of age and sex [12].
Genetic associations with healthspan and lifespan (mother’s and father’s attained age)
We obtained summary statistics for healthspan from a large GWAS conducted in 300,477 people of British ancestry in the UK Biobank. Healthspan was defined as the age of the first incidence of congestive Heart Failure (CHF), Myocardial Infarction (MI), Chronic Obstructive Pulmonary Disease (COPD), stroke, dementia, diabetes, cancer, and death [13]. The GWAS used Cox-Gompertz survival models, with adjustment for sex, the first genetic principal components variables, assessment centre and genotyping batch [13].
We obtained genetic associations with parental survival (attained age) from a GWAS of parental longevity in European descent UK Biobank participants (n = 415,311 for father’s attained age, n = 412,937 for mother’s attained age) [14]. The GWAS used a Cox proportional hazards model to estimate offspring genetic variant effects on parental survival, stratified by sex, and adjusted for age and 10 principal components of ancestry. As the effect sizes obtained using genetic data from offspring are half of the actual variant effect size in the parent, they were doubled to reflect the expected genetic effects in parents [14].
In both GWAS, the summary statistics from the GWAS were reported as log hazard ratios, for ease of understanding, these were converted to years of life by inverting the sign and multiplied by 10 [13, 14].
Statistical analysis
MR estimates were based on the genetic association with each type of COVID-19 (severe COVID-19, COVID-19 hospitalization or SARS-COV2 infection) divided by the genetic association with each protein), i.e., the Wald ratio estimates. Similarly, we obtained the MR estimates for the association of each protein with healthspan and lifespan. The genetic variant specific estimates were meta-analysed using inverse variance weighting (IVW). Multiplicative random effects model was used when three or more genetic variants were used as instruments, and fixed effects model was used when less than three genetic variants were used as instruments. To account for multiple testing, we used Bonferroni corrected significance (p-value < 0.05/1,361 = 3.7 × 10−5) as the cut-off. Heterogeneity test was also conducted for the identified protein-outcome associations. In sensitivity analysis, for proteins with three or more genetic variants as instruments, we used different MR methods under different assumptions from IVW, including weighted median, weighted mode and MR-SPI. The weighted median method can provide consistent estimates even when up to 50% of the information comes from invalid genetic variants [15]. The weighted mode is based on the assumption that a plurality of genetic variants are valid instruments, i.e., no larger subset of invalid instruments estimating the same causal parameter than the subset of valid instruments exists [16]. Also based on the plurality assumption, MR-SPI can automatically select genetic variants as valid instruments and provide robust inference in finite samples [17]. Given that some proteins lack cis-SNPs as instruments, we used cis- and trans-SNPs as instruments in the main analysis and cis-SNPs in the sensitivity analysis.
Identification of drug repurposing opportunities
To have a better understanding of the biological function of each identified protein, we also looked into their functions in STRING [18], a database with comprehensive information on protein network and function, and UNIPROT (https://www.uniprot.org/). To identify potential drug repurposing opportunity, we checked whether the proteins are targeted by currently available drugs, by searching in DrugBank, a publicly available resource with drug and drug target information on over 13,000 drugs (https://www.drugbank.ca/).
All statistical analysis were performed using R (Foundation for Statistical Computing, Vienna, Austria; Version 4.1.1) and “TwoSampleMR”, “MendelianRandomization”, “ggplot2”, “MR.SPI” R packages.
Availability of data and materials
All the data used in the study are publicly available. The data sources have been specified in the methods.
Results
Drug repurposing opportunity
Table 1 shows the proteins targeted by existing drugs. In this study, we found three proteins that affect the risk of COVID-19 in MR analysis have already been targeted by drugs currently used for the treatment of epilepsy, glaucoma, rheumatoid arthritis and neoplasm. Seven proteins that affect healthspan or lifespan have been targeted by drugs currently used for the treatment of cardiovascular disease, atherosclerosis, and neoplasm. This suggests that these existing drugs have the repurposing potential for lowering the risk of COVID-19 or improving healthspan/lifespan.
Table 1. Proteins targeted by existing drugs with potential for drug repurposing.
| Protein | Findings in this MR | Drug(s) targeting this protein | Disease of treatment | Action type |
| CA4 | Increases the risk of COVID-19 hospitalization and SARS-COV2 infection | METHAZOLAMIDE | Open-angle glaucoma | Inhibitor |
| ACETAZOLAMIDE | Epilepsy | Inhibitor | ||
| TOPIRAMATE | Migraine disorder | Inhibitor | ||
| SULTHIAME | Epilepsy | Inhibitor | ||
| DICHLORPHENAMIDE | Glaucoma | Inhibitor | ||
| FGFR2 | Increases the risk of SARS-COV2 infection | NINTEDANIB | Pulmonary fibrosis; neoplasm | Inhibitor |
| REGORAFENIB | Metastatic colorectal cancer | Inhibitor | ||
| ERDAFITINIB | Neoplasm | Inhibitor | ||
| TDGF1 | Lowers the risk of three COVID-19 related traits | BIIB-015 | Neoplasm | Binding agent |
| CD74 | Lowers father’s lifespan | MILATUZUMAB | Multiple myeloma; chronic lymphocytic leukemia | Antagonist |
| GP1BA | Lowers father’s lifespan | ANFIBATIDE | Myocardial infarction | Antagonist |
| GPNMB | Increases healthspan | GLEMBATUMUMAB VEDOTIN; GLEMBATUMUMAB | Breast cancer; melanoma | Binding agent |
| KIR2DL3 | Lowers father’s lifespan | IPH-2101; LIRILUMAB | Multiple myeloma | Inhibitor |
| PCSK9 | Lowers father’s lifespan | PCSK9 inhibitors, such as EVOLOCUMAB and INCLISIRAN | Cardiovascular disease | Inhibitor |
| PLA2G7 | Lowers healthspan | DARAPLADIB | Atherosclerosis | Inhibitor |
| RILAPLADIB | Atherosclerosis; Alzheimer’s disease | Inhibitor | ||
| TNFRSF8 | Lowers mother’s lifespan | BRENTUXIMAB VEDOTIN | Lymphoma | Inhibitor |
Discussion
Using MR to minimize confounding bias, we not only confirmed some proteins reported in previous studies, such as SFTPD lowering the risk of severe COVID-19 [8], but also identified several novel proteins which are associated with the risk of COVID-19, with functions involved in inflammation and immunity, apoptosis and metabolism. We also identified novel proteins related to healthspan and lifespan, and some of them are also involved in inflammation and immunity, such as CXCL9, HLA-DRA, LILRB4, IL19 and TNFRSF8, in apoptosis, such as FOXO3, and involved in metabolism, such as PCSK9 and LDLR. This finding implies that drugs targeting on these proteins may be used for disease prevention and treatment. For example, PCSK9 inhibitors, which have been used to treat hyperlipidemia, increase years of life. The identification of these proteins deepened the understanding of molecular mechanisms and provided new targets, with relevance to new drug development. Notably, we found 3 proteins affecting COVID-19 and 7 proteins affecting healthspan or lifespan are targeted by existing drugs, suggesting a great potential of drug repurposing. In the following discussion, we discussed in detail these proteins involved in these functions, as well as proteins targeted by existing drugs.
Proteins involved in inflammation and immune function
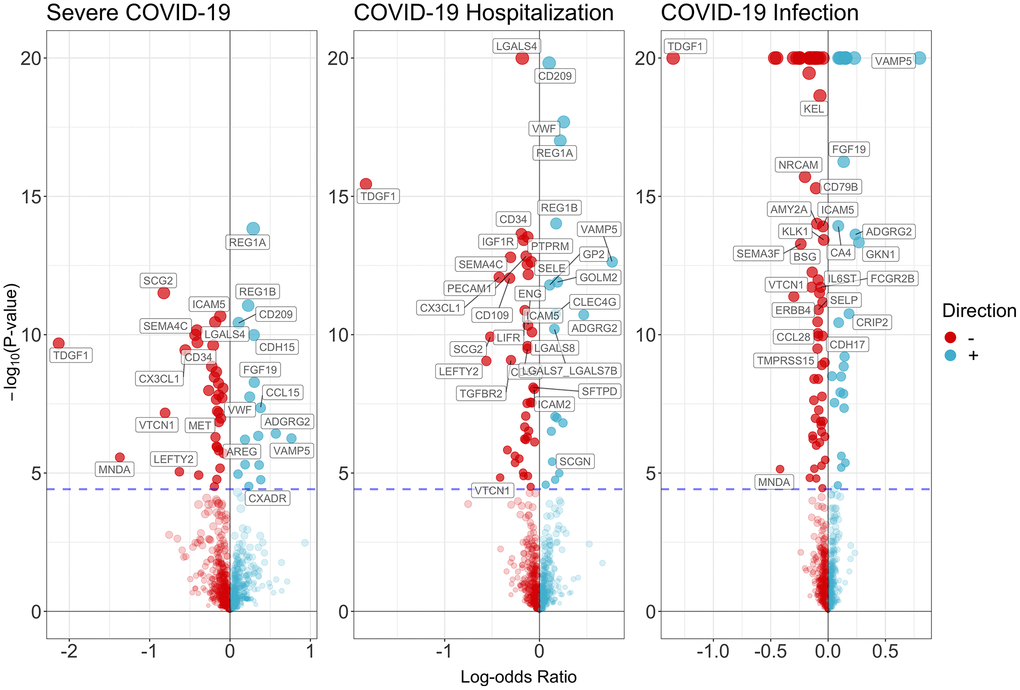
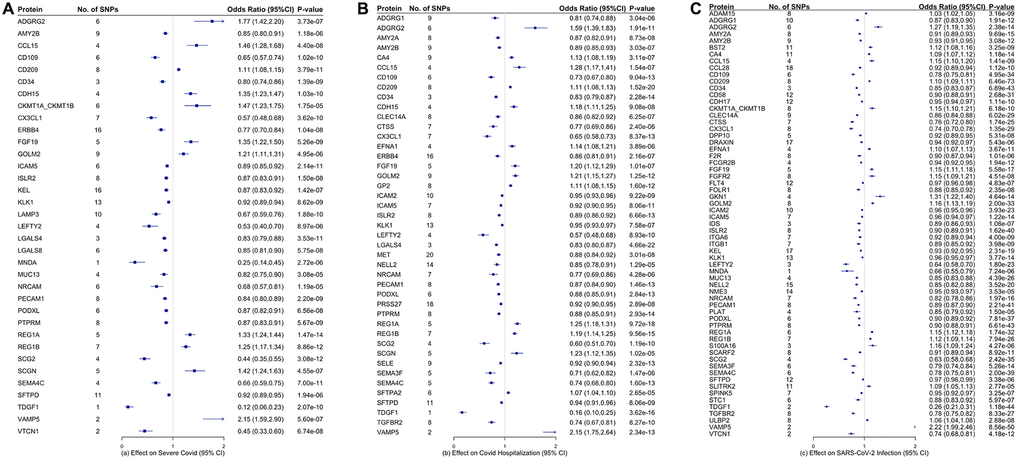
In the analysis of COVID-19, our findings are consistent with a previous MR study showing SFTPD is related to lower severe COVID-19 [8]. The protein is part of the innate immune response, the protein and the gene encoding this protein protects the lung against inhaled microorganisms and chemicals [19]. SFTPD also interacts with COVID-19 spike proteins [20]. Partly consistent with a previous MR study showing sICAM-2 lowers the risk of severe COVID-19 [21] and in line with another MR study showing ICAM5 lowers the risk of severe COVID-19 [22], we found ICAM-2 and ICAM-5 lower the risk of COVID-19 hospitalization. ICAM-2 and ICAM-5 both belong to the Ig-like cell adhesion molecule family which may play a role in lymphocyte recirculation by blocking LFA-1-dependent cell adhesion. They mediate adhesive interactions important for antigen-specific immune response, NK-cell mediated clearance, lymphocyte recirculation, and other cellular interactions important for immune response and surveillance. The gene, ICAM5, is also related to severe COVID-19 [23].
Moreover, we identified several other inflammation-related proteins that are linked to COVID-19, such as CD209, CD58, CCL15, CCL28, and MNDA. CD209 is a pathogen-recognition receptor expressed on the surface of immature dendritic cells (DCs) and involved in initiation of primary immune response. In vitro experiment shows that CD209 also serves as alternative receptors for SARS-CoV-2 in disease-relevant cell types, including the vascular system [24]. This is in line with our findings that CD209 increased the risk of severe COVID-19, COVID-19 hospitalization and SARS-COV2 infection. CD58 is involved in activation of NK and T cells, a reduction in the expression of CD58 results in reduced activation of NK and cytotoxic T cells and may play a role in COVID-19 [25]. This may explain why we found CD58 lowers the risk of severe COVID-19 in our MR study. CCL15, a chemokine involved in leukocyte trafficking, was identified as predictor for severe COVID-19 [26]. CCL28 displays strong homing capabilities for B and T cells and orchestrates the trafficking and functioning of lymphocytes [27]. CCL28 may be used as an indicator for mucosal immune responses in people with SARS-COV2 infection [28]. In our study, we found CCL28 lowers the risk of SARS-COV2 infection. MNDA plays a role in the granulocyte/monocyte cell-specific response to interferon. It is required for INFα production from human blood cells in response to viruses [29], and may contribute to the immune response to SARS-CoV-2 [30].
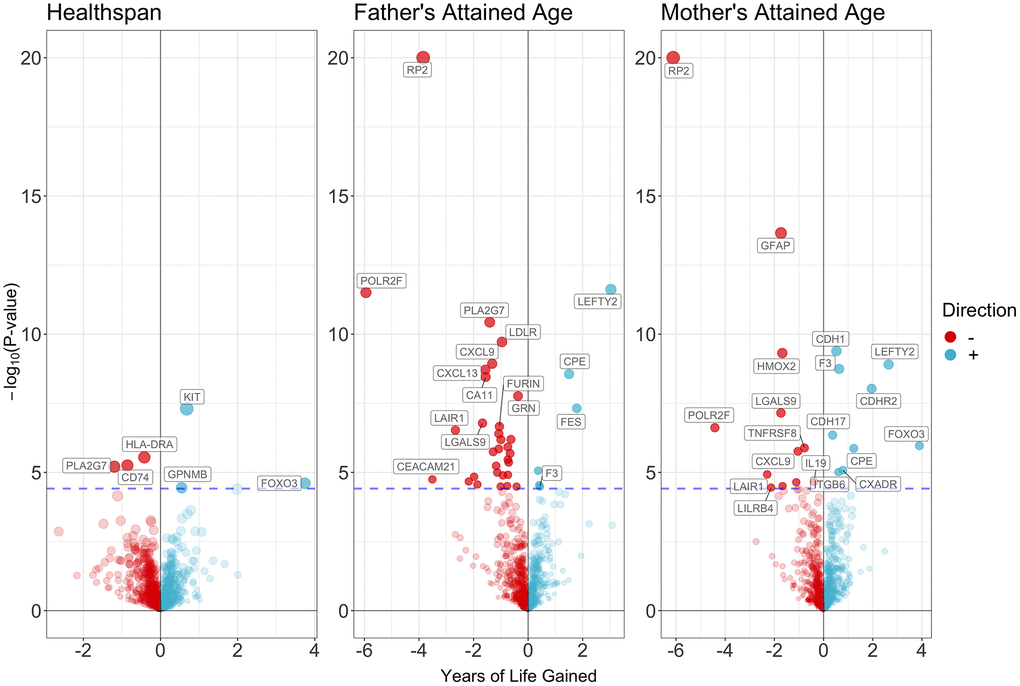
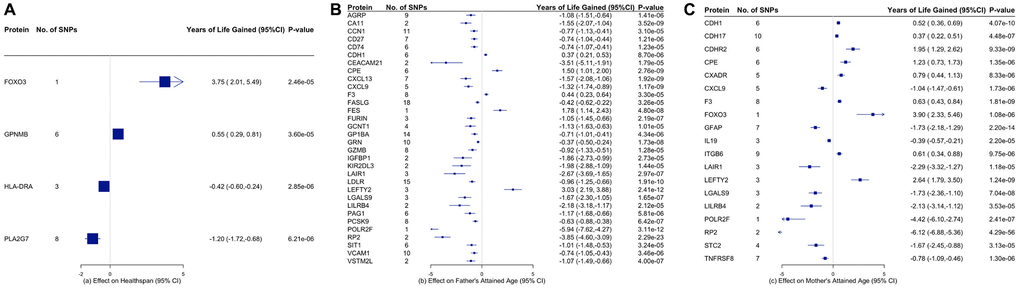
Meanwhile, in the analysis for healthspan and lifespan, we also found several proteins which play a role in inflammation and immune function and have not been reported in previous MR studies, including CXCL9, HLA-DRA, LILRB4, IL19 and TNFRSF8. CXCL9 was involved in T cell trafficking. In animal experiments, it increased with aging, which can be prevented by calorie restriction, an established approach of increasing longevity [31]. Consistently, we found CXCL9 was related to shorter years of life. HLA-DRA was expressed on the surface of various antigen presenting cells such as B lymphocytes, dendritic cells, and monocytes/macrophages, and plays a central role in the immune system. The expression of HLA-DRA was higher in older people [32]. LILRB4 plays an important role in adaptive immunity, and increases with age in animal experiments [33]. In our study, we further suggested that increased HLA-DRA and LILRB4 lower healthspan and lifespan, respectively. TNFRSF8 may play a role in the regulation of cellular growth, transformation of activated lymphoblasts. Its regulating gene, TNFRSF8, has also been shown to relate to ageing [34]. IL19 promotes the production of IL6, which increased with age and its in vitro synthesis is prevented by diet restriction [35]. In our study, for the first time, we clearly showed that TNFRSF8 and IL19 possibly lowered lifespan.
Proteins involved in apoptosis
Apoptosis is an important process in aging. Consistent with previous studies [36], we found FOXO3 affects healthspan and maternal lifespan. The FOXO3 gene functions as a trigger for apoptosis through expression of genes necessary for cell death. Notably, in the proteins related to COVID-19, we also found several proteins involved in apoptosis, such as FGFR2 and ERBB4. FGFR2 promotes gastric cancer progression by inhibiting the expression of Thrombospondin4 via PI3K-Akt-Mtor pathway [37]. Drug targeting FGFR2 has been hypothesized to be used as treatments for COVID-19 [38] but this has not been tested in trials. ERBB4 is a tyrosine-protein kinase that plays an essential role as cell surface receptor for neuregulins and EGF family members and regulates cell proliferation, differentiation, migration and apoptosis. ERBB4 was downregulated after the coronavirus infection, and was upregulated by using the Wortmannin, which may inhibit the pathological cycle and development of SARS-CoV-2 in the human hosts [39].
Notably, we also found LEFTY2 contributes to both COVID-19 and lifespan. LEFTY2 lowers the risk of all COVID-19 phenotypes and increases paternal and maternal lifespan. LEFTY2 encodes a secreted ligand of the transforming growth factor-beta (TGF-beta) superfamily of proteins, which acts as a crucial regulator of cell growth, proliferation, differentiation and apoptosis [40]. More studies are needed to clarify the pathways underlying the effects of LEFTY2, to provide insights for clinical practice and healthcare.
Proteins involved in metabolism
Metabolic syndrome is a known risk factor for COVID-19 and leads to multiple chronic diseases and mortality. Interestingly, we identified several proteins regulating metabolism. For example, we found AMY2B lowers the risk of severe COVID-19 and AMY2A lowers the risk of hospitalization. AMY2A and AMY2B both play a role in carbohydrate metabolism; the former (AMY2A) was FDA approved drug target. Evidently, the levels of AMY2A and AMY2B were relatively higher in survivors of COVID-19 compared to those who died of COVID-19 [41].
Among the proteins for aging, we found PLA2G7, PCSK9 and LDLR, which are all related to lipid metabolism, lower healthspan and/or lifespan. PLA2G7 has been associated with atherosclerosis, diabetes, and cardiovascular disease, and considered as a potential target that is a nexus between the immune, metabolic, and cardiovascular pathways of aging [42, 43]. PCSK9 and LDLR play an important role in lipid metabolism, and lead to higher risk of IHD, the leading cause of mortality. PCSK9 inhibitors, the drugs targeting PCSK9, are getting arising attention. In this novel study, we showed that PCSK9 inhibitors not only lower the cardiovascular risk, but also should be considered as a treatment for aging.
Drug repurposing opportunity
Importantly, we found that several proteins are targeted by existing drugs, which provided novel insights into drug repurposing. For example, we found that PLA2G7 which lowers healthspan is targeted by PLA2G7 inhibitors which are currently used in the treatment for atherosclerosis and Alzheimer disease. This suggests that PLA2G7 inhibitors may be repurposed to increase healthspan. Similarly, we suggested several drugs, such as FGFR2 inhibitors which have been used for the treatment of neoplasm can be considered to lower the risk of COVID-19. These novel findings are worthwhile to be tested in future randomized controlled trials.
Strengths and limitations
Using MR enables us to minimize unmeasured confounding bias in cohort studies and case-control studies. In this study, we used by far the largest available GWAS of proteomics, COVID-19, lifespan and healthspan, which improves the power to identify proteins causally related to these outcomes. Despite, we also acknowledge a few limitations. First, MR is based on three core assumptions, i.e., the relevance, independence, and exclusion-restriction assumption [49]. To satisfy these assumptions, we used genetic variants strongly associated with these proteins, with Bonferroni-corrected significance. We also used multiple sensitivity analysis methods robust to pleiotropy and checked the directions of associations from different genetic instruments (trans plus cis SNPs versus cis SNPs). In the situation where heterogeneity test suggested large heterogeneity, estimates from methods robust to pleiotropy are more valid than IVW. Considering that the measurements of proteins at baseline were conducted before the occurrence of the outcomes, reverse causality is not a main concern, so we did not perform bi-directional MR analysis. Second, MR studies of COVID-19 might be subject to survivor bias (selection bias), i.e., people might have died of COVID-19 or other diseases before recruitment. So, the causal effects might be underestimated. Third, population stratification might affect MR estimates. However, the GWAS data used in this study were derived from people largely of European ancestry. Meanwhile, as the study was based on people of European ancestry, the findings may not be generalizable to other ancestries. Fourth, the samples for proteins and outcomes both included UK Biobank. The partly overlapping in samples may bias two-sample MR estimates [2], but a simulation study shows two-sample MR can be safely conducted in a single large dataset [3], such as UK Biobank. So, the sample overlapping may not be a main concern, but the estimates need to be interpreted more cautiously. Finally, as we used summary statistics, we cannot assess the potential nonlinear association of these proteins with COVID-19, healthspan and lifespan.
Conclusions
In this study, using genetic instruments from by far the largest GWAS of proteomics, as well as by far the largest GWAS of COVID-19, healthspan and lifespan, we identified multiple proteins affecting COVID-19 and aging. We not only confirmed proteins identified in previous studies, such as SFTPD which affects COVID-19 and FOXO3 which affects healthspan, notably we identified several proteins with functions involved in inflammation and immune function, apoptosis, metabolism and hormone regulation. These findings provide novel insights into disease pathogenesis and pinpoint targets for therapeutic development or drug repurposing. For example, our findings of PCSK9 shortening lifespan and PLA2G7 shortening healthspan suggested that PCSK9 inhibitors, such as Evolocumab, and drugs inhibiting PLA2G7, such as Darapladib, may be beneficial for increasing lifespan and healthspan.
Supplementary Materials
Author Contributions
The manuscript was drafted by JVZ. Data analysis was conducted by MY, with the help of ZL and JVZ. JVZ developed the study conception, JVZ and ZL designed the analysis, and interpreted the results. All authors read and approved the final manuscript.
Acknowledgments
This research was conducted using the summary statistics from COVID-19 host genetics consortium and large genome-wide association studies of healthspan and lifespan. The authors would like to thank all participants in the study and investigators for sharing the valuable data.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Ethical Statement
The study is an analysis using publicly available summary data that does not require ethical approval.
Funding
No funding was used for this paper.
References
- 1. Msemburi W, Karlinsky A, Knutson V, Aleshin-Guendel S, Chatterji S, Wakefield J. The WHO estimates of excess mortality associated with the COVID-19 pandemic. Nature. 2023; 613:130–7. https://doi.org/10.1038/s41586-022-05522-2 [PubMed]
- 2. Aburto JM, Kashyap R, Schöley J, Angus C, Ermisch J, Mills MC, Dowd JB. Estimating the burden of the COVID-19 pandemic on mortality, life expectancy and lifespan inequality in England and Wales: a population-level analysis. J Epidemiol Community Health. 2021; 75:735–40. https://doi.org/10.1136/jech-2020-215505 [PubMed]
- 3. Furman D, Campisi J, Verdin E, Carrera-Bastos P, Targ S, Franceschi C, Ferrucci L, Gilroy DW, Fasano A, Miller GW, Miller AH, Mantovani A, Weyand CM, et al. Chronic inflammation in the etiology of disease across the life span. Nat Med. 2019; 25:1822–32. https://doi.org/10.1038/s41591-019-0675-0 [PubMed]
- 4. Blanco-Melo D, Nilsson-Payant BE, Liu WC, Uhl S, Hoagland D, Møller R, Jordan TX, Oishi K, Panis M, Sachs D, Wang TT, Schwartz RE, Lim JK, et al. Imbalanced Host Response to SARS-CoV-2 Drives Development of COVID-19. Cell. 2020; 181:1036–45.e9. https://doi.org/10.1016/j.cell.2020.04.026 [PubMed]
- 5. Bi X, Liu W, Ding X, Liang S, Zheng Y, Zhu X, Quan S, Yi X, Xiang N, Du J, Lyu H, Yu D, Zhang C, et al. Proteomic and metabolomic profiling of urine uncovers immune responses in patients with COVID-19. Cell Rep. 2022; 38:110271. https://doi.org/10.1016/j.celrep.2021.110271 [PubMed]
- 6. Al-Bayatee NT, Ad'hiah AH. Soluble HLA-G is upregulated in serum of patients with severe COVID-19. Hum Immunol. 2021; 82:726–32. https://doi.org/10.1016/j.humimm.2021.07.007 [PubMed]
- 7. Lawlor DA, Harbord RM, Sterne JA, Timpson N, Davey Smith G. Mendelian randomization: using genes as instruments for making causal inferences in epidemiology. Stat Med. 2008; 27:1133–63. https://doi.org/10.1002/sim.3034 [PubMed]
- 8. Palmos AB, Millischer V, Menon DK, Nicholson TR, Taams LS, Michael B, Sunderland G, Griffiths MJ, Hübel C, Breen G, and COVID Clinical Neuroscience Study Consortium. Proteome-wide Mendelian randomization identifies causal links between blood proteins and severe COVID-19. PLoS Genet. 2022; 18:e1010042. https://doi.org/10.1371/journal.pgen.1010042 [PubMed]
- 9. Gaziano L, Giambartolomei C, Pereira AC, Gaulton A, Posner DC, Swanson SA, Ho YL, Iyengar SK, Kosik NM, Vujkovic M, Gagnon DR, Bento AP, Barrio-Hernandez I, et al, and VA Million Veteran Program COVID-19 Science Initiative. Actionable druggable genome-wide Mendelian randomization identifies repurposing opportunities for COVID-19. Nat Med. 2021; 27:668–76. https://doi.org/10.1038/s41591-021-01310-z [PubMed]
- 10. Perrot N, Pelletier W, Bourgault J, Couture C, Li Z, Mitchell PL, Ghodsian N, Bossé Y, Thériault S, Mathieu P, Arsenault BJ. A trans-omic Mendelian randomization study of parental lifespan uncovers novel aging biology and therapeutic candidates for chronic diseases. Aging Cell. 2021; 20:e13497. https://doi.org/10.1111/acel.13497 [PubMed]
- 11. Sun BB, Chiou J, Traylor M, Benner C, Hsu YH, Richardson TG, Surendran P, Mahajan A, Robins C, Vasquez-Grinnell SG, Hou L, Kvikstad EM, Burren OS, et al. Genetic regulation of the human plasma proteome in 54,306 UK Biobank participants. bioRxiv. 2022. https://doi.org/10.1101/2022.06.17.496443
- 12. The COVID-19 Host Genetics Initiative. https://www.covid19hg.org/.
- 13. Zenin A, Tsepilov Y, Sharapov S, Getmantsev E, Menshikov LI, Fedichev PO, Aulchenko Y. Identification of 12 genetic loci associated with human healthspan. Commun Biol. 2019; 2:41. https://doi.org/10.1038/s42003-019-0290-0 [PubMed]
- 14. Pilling LC, Kuo CL, Sicinski K, Tamosauskaite J, Kuchel GA, Harries LW, Herd P, Wallace R, Ferrucci L, Melzer D. Human longevity: 25 genetic loci associated in 389,166 UK biobank participants. Aging (Albany NY). 2017; 9:2504–20. https://doi.org/10.18632/aging.101334 [PubMed]
- 15. Bowden J, Davey Smith G, Haycock PC, Burgess S. Consistent Estimation in Mendelian Randomization with Some Invalid Instruments Using a Weighted Median Estimator. Genet Epidemiol. 2016; 40:304–14. https://doi.org/10.1002/gepi.21965 [PubMed]
- 16. Hartwig FP, Davey Smith G, Bowden J. Robust inference in summary data Mendelian randomization via the zero modal pleiotropy assumption. Int J Epidemiol. 2017; 46:1985–98. https://doi.org/10.1093/ije/dyx102 [PubMed]
- 17. Yao M, Guo Z, Liu Z. Robust Mendelian Randomization Analysis by Automatically Selecting Valid Genetic Instruments for Inferring Causal Relationships between Complex Traits and Diseases. medRxiv. 2023. https://doi.org/10.1101/2023.02.20.23286200
- 18. Szklarczyk D, Gable AL, Lyon D, Junge A, Wyder S, Huerta-Cepas J, Simonovic M, Doncheva NT, Morris JH, Bork P, Jensen LJ, Mering CV. STRING v11: protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2019; 47:D607–13. https://doi.org/10.1093/nar/gky1131 [PubMed]
- 19. Ortega FJ, Pueyo N, Moreno-Navarrete JM, Sabater M, Rodriguez-Hermosa JI, Ricart W, Tinahones FJ, Fernández-Real JM. The lung innate immune gene surfactant protein-D is expressed in adipose tissue and linked to obesity status. Int J Obes (Lond). 2013; 37:1532–8. https://doi.org/10.1038/ijo.2013.23 [PubMed]
- 20. Chen L, Zheng S. Understand variability of COVID-19 through population and tissue variations in expression of SARS-CoV-2 host genes. Inform Med Unlocked. 2020; 21:100443. https://doi.org/10.1016/j.imu.2020.100443 [PubMed]
- 21. Zhu J, Wu C, Wu L. Associations Between Genetically Predicted Protein Levels and COVID-19 Severity. J Infect Dis. 2021; 223:19–22. https://doi.org/10.1093/infdis/jiaa660 [PubMed]
- 22. Zheng J, Zhang Y, Zhao H, Liu Y, Baird D, Karim MA, Ghoussaini M, Schwartzentruber J, Dunham I, Elsworth B, Roberts K, Compton H, Miller-Molloy F, et al. Multi-ancestry Mendelian randomization of omics traits revealing drug targets of COVID-19 severity. EBioMedicine. 2022; 81:104112. https://doi.org/10.1016/j.ebiom.2022.104112 [PubMed]
- 23. Velavan TP, Pallerla SR, Rüter J, Augustin Y, Kremsner PG, Krishna S, Meyer CG. Host genetic factors determining COVID-19 susceptibility and severity. EBioMedicine. 2021; 72:103629. https://doi.org/10.1016/j.ebiom.2021.103629 [PubMed]
- 24. Amraei R, Yin W, Napoleon MA, Suder EL, Berrigan J, Zhao Q, Olejnik J, Chandler KB, Xia C, Feldman J, Hauser BM, Caradonna TM, Schmidt AG, et al. CD209L/L-SIGN and CD209/DC-SIGN Act as Receptors for SARS-CoV-2. ACS Cent Sci. 2021; 7:1156–65. https://doi.org/10.1021/acscentsci.0c01537 [PubMed]
- 25. van Eeden C, Khan L, Osman MS, Cohen Tervaert JW. Natural Killer Cell Dysfunction and Its Role in COVID-19. Int J Mol Sci. 2020; 21:6351. https://doi.org/10.3390/ijms21176351 [PubMed]
- 26. Lucas C, Wong P, Klein J, Castro TBR, Silva J, Sundaram M, Ellingson MK, Mao T, Oh JE, Israelow B, Takahashi T, Tokuyama M, Lu P, et al, and Yale IMPACT Team. Longitudinal analyses reveal immunological misfiring in severe COVID-19. Nature. 2020; 584:463–9. https://doi.org/10.1038/s41586-020-2588-y [PubMed]
- 27. Mohan T, Deng L, Wang BZ. CCL28 chemokine: An anchoring point bridging innate and adaptive immunity. Int Immunopharmacol. 2017; 51:165–70. https://doi.org/10.1016/j.intimp.2017.08.012 [PubMed]
- 28. Yan Y, Jiang X, Wang X, Liu B, Ding H, Jiang M, Yang Z, Dai Y, Ding D, Yu H, Zhang S, Liu J, Sha M, et al. CCL28 mucosal expression in SARS-CoV-2-infected patients with diarrhea in relation to disease severity. J Infect. 2021; 82:e19–21. https://doi.org/10.1016/j.jinf.2020.08.042 [PubMed]
- 29. Gu L, Casserly D, Brady G, Carpenter S, Bracken AP, Fitzgerald KA, Unterholzner L, Bowie AG. Myeloid cell nuclear differentiation antigen controls the pathogen-stimulated type I interferon cascade in human monocytes by transcriptional regulation of IRF7. Nat Commun. 2022; 13:14. https://doi.org/10.1038/s41467-021-27701-x [PubMed]
- 30. Beltrán-Camacho L, Eslava-Alcón S, Rojas-Torres M, Sánchez-Morillo D, Martinez-Nicolás MP, Martín-Bermejo V, de la Torre IG, Berrocoso E, Moreno JA, Moreno-Luna R, Durán-Ruiz MC. The serum of COVID-19 asymptomatic patients up-regulates proteins related to endothelial dysfunction and viral response in circulating angiogenic cells ex-vivo. Mol Med. 2022; 28:40. https://doi.org/10.1186/s10020-022-00465-w [PubMed]
- 31. Trott DW, Lesniewski LA, Donato AJ. Selected life-extending interventions reduce arterial CXCL10 and macrophage colony-stimulating factor in aged mouse arteries. Cytokine. 2017; 96:102–6. https://doi.org/10.1016/j.cyto.2017.03.008 [PubMed]
- 32. de Almeida Chuffa LG, Freire PP, Dos Santos Souza J, de Mello MC, de Oliveira Neto M, Carvalho RF. Aging whole blood transcriptome reveals candidate genes for SARS-CoV-2-related vascular and immune alterations. J Mol Med (Berl). 2022; 100:285–301. https://doi.org/10.1007/s00109-021-02161-4 [PubMed]
- 33. Jonker MJ, Melis JP, Kuiper RV, van der Hoeven TV, Wackers PFK, Robinson J, van der Horst GT, Dollé ME, Vijg J, Breit TM, Hoeijmakers JH, van Steeg H. Life spanning murine gene expression profiles in relation to chronological and pathological aging in multiple organs. Aging Cell. 2013; 12:901–9. https://doi.org/10.1111/acel.12118 [PubMed]
- 34. Tanaka T, Biancotto A, Moaddel R, Moore AZ, Gonzalez-Freire M, Aon MA, Candia J, Zhang P, Cheung F, Fantoni G, Semba RD, Ferrucci L, and CHI consortium. Plasma proteomic signature of age in healthy humans. Aging Cell. 2018; 17:e12799. https://doi.org/10.1111/acel.12799 [PubMed]
- 35. Ershler WB, Sun WH, Binkley N, Gravenstein S, Volk MJ, Kamoske G, Klopp RG, Roecker EB, Daynes RA, Weindruch R. Interleukin-6 and aging: blood levels and mononuclear cell production increase with advancing age and in vitro production is modifiable by dietary restriction. Lymphokine Cytokine Res. 1993; 12:225–30. [PubMed]
- 36. Willcox BJ, Tranah GJ, Chen R, Morris BJ, Masaki KH, He Q, Willcox DC, Allsopp RC, Moisyadi S, Poon LW, Rodriguez B, Newman AB, Harris TB, et al. The FoxO3 gene and cause-specific mortality. Aging Cell. 2016; 15:617–24. https://doi.org/10.1111/acel.12452 [PubMed]
- 37. Huang T, Liu D, Wang Y, Li P, Sun L, Xiong H, Dai Y, Zou M, Yuan X, Qiu H. FGFR2 Promotes Gastric Cancer Progression by Inhibiting the Expression of Thrombospondin4 via PI3K-Akt-Mtor Pathway. Cell Physiol Biochem. 2018; 50:1332–45. https://doi.org/10.1159/000494590 [PubMed]
- 38. Sonkar C, Doharey PK, Rathore AS, Singh V, Kashyap D, Sahoo AK, Mittal N, Sharma B, Jha HC. Repurposing of gastric cancer drugs against COVID-19. Comput Biol Med. 2021; 137:104826. https://doi.org/10.1016/j.compbiomed.2021.104826 [PubMed]
- 39. Selvaraj G, Kaliamurthi S, Peslherbe GH, Wei DQ. Identifying potential drug targets and candidate drugs for COVID-19: biological networks and structural modeling approaches. F1000Res. 2021; 10:127. https://doi.org/10.12688/f1000research.50850.3 [PubMed]
- 40. Lu WQ, Qiu JL, Huang ZL, Liu HY. Enhanced circulating transforming growth factor beta 1 is causally associated with an increased risk of hepatocellular carcinoma: a mendelian randomization meta-analysis. Oncotarget. 2016; 7:84695–704. https://doi.org/10.18632/oncotarget.13218 [PubMed]
- 41. Filbin MR, Mehta A, Schneider AM, Kays KR, Guess JR, Gentili M, Fenyves BG, Charland NC, Gonye ALK, Gushterova I, Khanna HK, LaSalle TJ, Lavin-Parsons KM, et al. Longitudinal proteomic analysis of severe COVID-19 reveals survival-associated signatures, tissue-specific cell death, and cell-cell interactions. Cell Rep Med. 2021; 2:100287. https://doi.org/10.1016/j.xcrm.2021.100287 [PubMed]
- 42. Cao F, Lee RT. PLA2G7, caloric restriction and cardiovascular aging. J Cardiovasc Aging. 2022; 2:19. https://doi.org/10.20517/jca.2022.08 [PubMed]
- 43. Spadaro O, Youm Y, Shchukina I, Ryu S, Sidorov S, Ravussin A, Nguyen K, Aladyeva E, Predeus AN, Smith SR, Ravussin E, Galban C, Artyomov MN, Dixit VD. Caloric restriction in humans reveals immunometabolic regulators of health span. Science. 2022; 375:671–7. https://doi.org/10.1126/science.abg7292 [PubMed]
- 44. Aschenbrenner AC, Mouktaroudi M, Krämer B, Oestreich M, Antonakos N, Nuesch-Germano M, Gkizeli K, Bonaguro L, Reusch N, Baßler K, Saridaki M, Knoll R, Pecht T, et al, and German COVID-19 Omics Initiative (DeCOI). Disease severity-specific neutrophil signatures in blood transcriptomes stratify COVID-19 patients. Genome Med. 2021; 13:7. https://doi.org/10.1186/s13073-020-00823-5 [PubMed]
- 45. Bonilla C, Lewis SJ, Rowlands MA, Gaunt TR, Davey Smith G, Gunnell D, Palmer T, Donovan JL, Hamdy FC, Neal DE, Eeles R, Easton D, Kote-Jarai Z, et al, and PRACTICAL consortium. Assessing the role of insulin-like growth factors and binding proteins in prostate cancer using Mendelian randomization: Genetic variants as instruments for circulating levels. Int J Cancer. 2016; 139:1520–33. https://doi.org/10.1002/ijc.30206 [PubMed]
- 46. Larsson SC, Michaëlsson K, Burgess S. IGF-1 and cardiometabolic diseases: a Mendelian randomisation study. Diabetologia. 2020; 63:1775–82. https://doi.org/10.1007/s00125-020-05190-9 [PubMed]
- 47. Ilnytska O, Argyropoulos G. The role of the Agouti-Related Protein in energy balance regulation. Cell Mol Life Sci. 2008; 65:2721–31. https://doi.org/10.1007/s00018-008-8104-4 [PubMed]
- 48. Redmann SM
Jr , Argyropoulos G. AgRP-deficiency could lead to increased lifespan. Biochem Biophys Res Commun. 2006; 351:860–4. https://doi.org/10.1016/j.bbrc.2006.10.129 [PubMed] - 49. Davies NM, Holmes MV, Davey Smith G. Reading Mendelian randomisation studies: a guide, glossary, and checklist for clinicians. BMJ. 2018; 362:k601. https://doi.org/10.1136/bmj.k601 [PubMed]