Introduction
The latest epidemiological data showed that stroke is the leading cause of death and disability globally among the 18 neurological disorders [1]. Of all stroke types, ischemic stroke (IS) accounts for almost 80% [2]. Despite intravascular thrombectomy (ET) has broad application prospects in the new era of intravascular therapy, intravenous thrombolysis (IVT) remains the preferred and fundamental treatment for acute ischemic stroke (AIS) within 4.5 h of symptom onset as recommended by the guidelines [3, 4]. Nevertheless, higher risks of hemorrhagic transformation (HT) and neuroinflammation are related to IVT treatment due to the ischemia-reperfusion injury, rt-PA toxicity, and comorbidities, which may lead to worse functional outcomes in AIS patients, thus rendering this life-saving therapy a double-edged sword [5, 6].
Left ventricular systolic dysfunction (LVSD) has been reported as an important clinical predictor of higher morbidity and mortality in AIS patients [7, 8]. Left ventricular ejection fraction (LVEF) is a common parameter for the evaluation of left ventricular systolic function, and is widely used in clinical diagnosis and experimental research [9–11]. Previous studies prior to IVT or ET have indicated that reduced LVEF was associated with higher NIHSS scores at admission, poorer functional outcomes and higher in-hospital mortality rates [8, 12]. LVSD may involve in the pathophysiology of AIS through different mechanisms. It has been suggested that LVSD may predispose to in situ thrombosis as well as proximal thrombosis formation, leading to intracranial vascular occlusion after detachment which decreases global cerebral blood flow. In addition, LVSD causes a state of global cerebral hypoperfusion with decreased brain blood flow and cerebrovascular reactivity [13, 14], which contributes to a higher risk of ischemic insult.
Although LVEF has been routinely investigated during hospitalization in AIS patients, its impact on clinical outcomes remains controversial due to the lack of consistent LVEF classification standards. Moreover, few studies focused on the association between LVSD and functional prognosis in AIS patients undergoing IVT. Therefore, in current prospective cohort registry for AIS undergoing IVT, we aimed to investigate the relationship between LVSD and clinical outcomes and to explore the optimal nodes for predicting poor prognosis with reduced LVEF.
Materials and Methods
Study population
From June 2021 to May 2023, we prospectively collected a total of 310 patients treated with intravenous rt-PA (dose of 0.9 mg/kg, maximum 90mg) within 4.5 h of AIS onset in the National Stroke Prevention and Treatment Engineering Management Special Database. Of these, twelve patients who underwent intravenous thrombolysis combined with ET, and twenty-five patients with lack of LVEF data were excluded. Ultimately, 273 patients were eligible for inclusion in the final analysis (Figure 1).
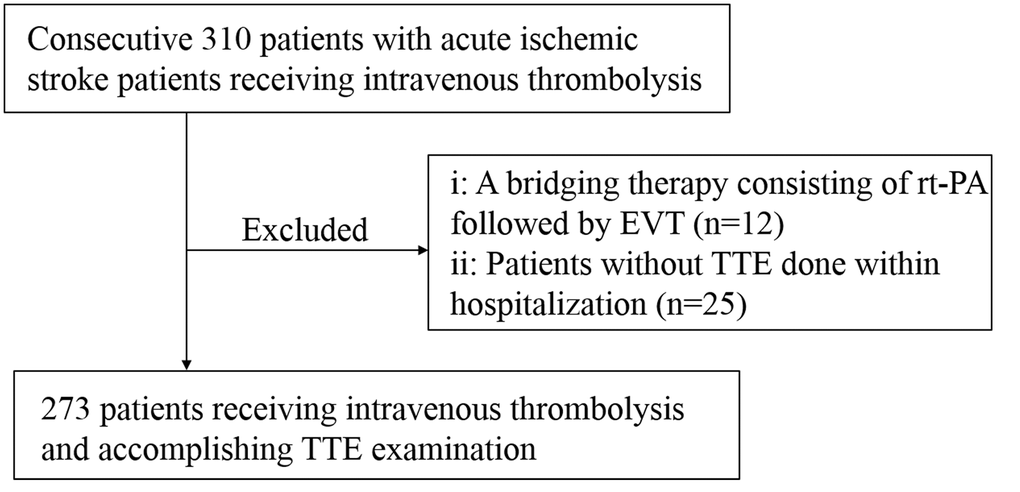
Figure 1. Flowchart of study population selection. rt-PA, recombinant tissue plasminogen activator; EVT, Endovascular therapy; TTE, Transthoracic echocardiography.
Data collection
Data regarding the demographic and clinical characteristics, age, sex, blood pressure, onset-to-needle time (ONT), stroke subtype, lifestyle risk factors (smoking and drinking alcohol), past medical history (hypertension, diabetes mellitus, hyperlipemia, previous stroke, coronary heart disease and atrial fibrillation) were collected from electronic medical records. ONT is further subdivided into less than 3 h and 3 to 4.5 h. Stroke subtype was determined based on the TOAST criteria [15]. The severity of AIS was assessed by professional clinicians via the National Institutes of Health Stroke Scale (NIHSS) score at admission and 24 h after IVT [16]. The modified Rankin Scale (mRS) score with seven levels was used to assess neurological functional outcomes at the 3-month follow-up [17].
Clinical measurement and definition of LVSD
LVSD was measured by the evaluation of the LVEF on transthoracic echocardiography (TTE) using Simpson’s biplane method of discs in line with international guidelines.
This procedure was based on the tracking of the central intimal boundary in both the apical four- and two-chamber views at the end of systole and diastole performed by a trained echocardiologist. Echocardiography and data extraction were completed within 24 h after admission. LVSD was defined as LVEF <50% according to previous descriptions.
Clinical outcome
The severity of AIS was assessed by well-trained neurologists at baseline and 24 h after rt-PA thrombolysis using the National Institutes of Health Stroke Scale (NIHSS) score [16]. Early neurological deterioration (END) was defined as an increment in the total NIHSS score of ≥ 4 points within 24 h after thrombolysis [18]. Complications of rt-PA intravenous thrombolysis, including symptomatic intracranial hemorrhage (sICH) and hemorrhagic transformation (HT) were evaluated in accordance with the European Cooperative Acute Stroke Study II (ECASS-II) criteria [19]. In brief, sICH was defined as a newly occurring hyper-density around the infarction on CT with neurological deterioration (NIHSS score ≥4) within 24 hours after intravenous thrombolysis. The Modified Rankin Scale (mRS) was utilized to assess functional outcomes after 90 days, with a score ≤ 2 representing a good outcome, whereas a score of 3~6 represents a poor outcome [17]. The primary outcome was deemed as a good functional outcome with a 90-day mRS score of 0~2, while the secondary outcomes evaluated were END, sICH, and HT.
Statistical analysis
Statistical analyses were conducted using SPSS version 17.0 software (SPSS Inc., Chicago, IL, USA) and R version 4.1.0 (R Foundation for Statistical Computing, Vienna, Austria). Continuous variables conforming to normal distribution were shown as mean ± standard deviation (SD) and compared using Student’s t-test or One-way ANOVA. Categorical variables were presented as percentages and analyzed by using Chi-square test. After adjusting for confounding factors, multivariable ordinal logistic regression model was used to analyze the association between LVEF and functional outcome at three months based on variables with a p-value < 0.05 in univariate analyses. A nonlinear logistic regression with restricted cubic spline (RCS) was used to examine the shape of the association between LVEF and primary outcome with four knots (defined at the 5th, 35th, 65th, and 95th percentiles). Subgroup analysis was also performed to further verify the reliability and practicability of the results and the multiplicative interaction terms were tested by the likelihood ratio test. A 2-tailed p-value < 0.05 was considered as the level for statistical significance.
Results
Baseline characteristics of study samples
As shown in Table 1, a total of 273 AIS patients (169 men and 104 women) were finally included after screening, with a mean age of 69.2 ±12.5 years, then divided into two groups (LVEF ≥50% group and LVEF<50% group). LVSD was observed in 41 (15.0%) patients. There were no significant differences in age, sex, stroke risk factors (CHD, smoking, and drinking), time from onset to intravenous thrombolysis (ONT), as well as sICH (p>0.05). However, hyperlipidemia, DM, coronary heart disease, and initial higher NIHSS scores were more commonly found in LVSD patients compared to non-LVSD patients (p<0.05).
Table 1. Baseline characteristics of patients stratified by the degree of LVEF.
| Variable | Total (n=273) | LVEF ≥ 50% (n=232) | LVEF < 50% (n=41) | P-value |
| Demographic data | ||||
| Age, years mean (SD) | 69.2±12.5 | 68.8±12.8 | 71.7±10.5 | 0.082 |
| Male [n(%)] | 169 (61.9) | 146 (63.0) | 23 (56.1) | 0.406 |
| Stroke risk factors [n(%)] | ||||
| Hypertension | 136 (58.6) | 108 (46.6) | 26 (63.4) | 0.034 |
| Diabetes | 66 (28.4) | 51 (22.0) | 15 (36.6) | 0.044 |
| Hyperlipidemia | 110 (40.3) | 97 (41.8) | 18 (43.9) | 0.803 |
| CHD | 100 (43.1) | 83 (35.8) | 19 (46.3) | 0.197 |
| Smoking | 90 (33.0) | 81 (35.0) | 9 (22.0) | 0.104 |
| Drinking | 84 (30.8) | 73 (31.5) | 11 (26.8) | 0.553 |
| History of stroke or TIA [n(%)] | 38 (13.9) | 32 (13.8) | 6 (14.6) | |
| Stroke etiology, TOAST [n(%)] | 0.502 | |||
| Large artery atherosclerosis | 167 (60.9) | 144 (61.8) | 23 (56.1) | |
| Cardioembolism | 13 (4.7) | 9 (3.9) | 4 (9.8) | |
| Small artery occlusion | 80 (29.2) | 69 (29.6) | 11 (26.8) | |
| Other determined | 10 (3.6) | 8 (3.4) | 2 (4.9) | |
| Undetermined | 4 (1.5) | 3 (1.3) | 1 (2.4) | |
| ONT [n(%)] | 0.698 | |||
| < 3 h | 186 (68.1) | 157 (67.7) | 29 (70.7) | |
| 3 to 4.5 h | 87 (31.9) | 75 (32.3) | 12 (29.3) | |
| Stroke evaluation | ||||
| NIHSS on arrival, median (IQR) | 6 (5, 8) | 6 (4, 8) | 8 (6, 10) | <0.001 |
| NIHSS at 24 h, median (IQR) | 4 (2, 7) | 4 (2, 6) | 8 (5, 10) | <0.001 |
| Outcomes [n(%)] | ||||
| mRS (0~2) at 90 days | 154 (56.4) | 143 (61.6) | 11 (26.8) | 0.003 |
| sICH or HT | 8 (2.9) | 5 (2.2) | 3 (7.3) | 0.071 |
| END | 11 (4.0) | 6 (2.6) | 5 (12.2) | 0.004 |
LVSD correlations with clinical outcome
Fewer LVSD patients achieved a good functional outcome at 90 days (mRS≤2), however, there was no significant difference in the proportion of patients with sICH or HT compared with the non-LVSD group. By analyzing the distribution of mRS between the two groups, the relationship between LVEF levels and prognosis emerged. LVSD patients had significantly worse mRS scores after IVT in comparison with LVEF ≥ 50% patients (Figure 2). After adjusting for age, gender, and other conventional confounders, multivariate logistic regression analysis showed that higher NIHSS score (OR 1.08, 95% CI 1.04 to 1.11; p<0.001), pre-existing diabetes (OR 2.08, 95% CI 1.11 to 3.90; p=0.023), and presence of LVSD (OR 2.78, 95% CI 1.23 to 6.26; p=0.014) were associated with the 90-day poor functional outcomes (Table 2).
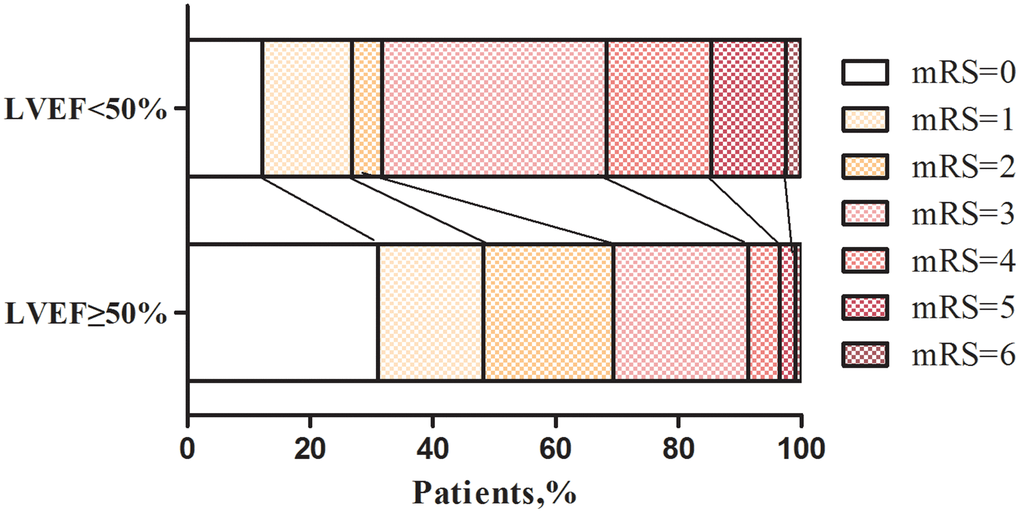
Figure 2. Distribution of mRS scores at three months according to LVEF levels after IVT in AIS patients. LVEF, left ventricular ejection function.
Table 2. Univariate and multivariate analyses of predictors of poor functional outcomes in AIS patients.
| Variable | Univariate analysis | Multivariate analysis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| mRS (0~2) | mRS (3~6) | OR (95%CI) | P-value | Adjusted OR (95%CI) | P-value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years), mean (SD) | 68.3±12.3 | 70.4±12.8 | 1.01 (0.99, 1.03) | 0.181 | 1.00 (0.98, 1.02) | 0.922 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male [n(%)] | 101 (59.8) | 68 (40.2) | 0.70 (0.43, 1.15) | 0.155 | 0.80 (0.45, 1.42) | 0.453 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension, [n(%)] | 66 (48.5) | 70 (51.5) | 1.40 (0.86, 2.26) | 0.173 | 1.07 (0.62, 1.84) | 0.814 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes, [n(%)] | 24 (36.4) | 42 (63.6) | 2.96 (1.66, 5.25) | <0.001 | 2.08 (1.11, 3.90) | 0.023 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hyperlipidemia, [n(%)] | 71 (64.5) | 39 (35.5) | 0.69 (0.42, 1.12) | 0.130 | 0.66 (0.38, 1.16) | 0.145 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CHD, [n(%)] | 44 (44.0) | 56 (56.0) | 1.25 (0.76, 2.05) | 0.372 | 1.07 (0.61, 1.87) | 0.822 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| History of stroke or TIA, [n(%)] | 18 (47.4) | 20 (52.6) | 1.44 (0.72, 2.88) | 0.308 | 1.15 (0.52, 2.54) | 0.732 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IVT 3h [n(%)] | 45 (51.7) | 42 (48.3) | 1.18 (0.70, 1.98) | 0.528 | 1.32 (0.74, 2.36) | 0.347 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NIHSS on arrival, median (IQR) | 6 (4, 7) | 7 (6, 9) | 1.40 (1.24, 1.58) | <0.001 | 1.31 (1.21, 1.49) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reduced LVEF, [n(%)] | 11 (26.8) | 30 (73.2) | 4.38 (2.09, 9.18) | <0.001 | 2.78 (1.23, 6.26) | 0.014 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LVEF by category, [n(%)] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LVEF (≥50%) | 143 (52.3) | 89 (32.6) | (Reference) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LVEF (41~49%) | 10 (3.7) | 19 (7.0) | 3.21 (1.44, 7.18) | 0.009 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LVEF (≤40%) | 1 (0.4) | 11 (4.0) | 16.07 (2.02, 127.66) | 0.004 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LVEF, Left ventricular ejection fraction; NIHSS, National Institutes of Health Stroke Scale; TIA, Transient ischemic attack; CHD, Coronary heart disease; IVT, Intravenous thrombolysis. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
RCS regression and subgroup analysis for the clinical outcomes
In the further multivariable logistic regression model with RCS analysis of AIS patients based on LVEF stratified, the proportion of patients achieving good functional outcomes at 90 days significantly decreased with greater degree of LVSD (P for nonlinearity = 0.414; Figure 3). The optimal cutoff values of the LVEF after rt-PA treatment for predicting 90-days poorer functional outcomes were 0.48 (Figure 3). The results of subgroup analyses classified by age (< 65 or ≥ 65 years), sex (female or male), NIHSS on arrival (< 8 or ≥ 8 scores), diabetes mellitus and coronary heart disease are depicted in Figure 4. Obviously, reduced LVEF levels after IVT were discovered to be related with poor outcomes, especially in elderly individuals and higher NIHSS scores (both p=0.023), male (p=0.029), and coronary heart disease (p=0.030).
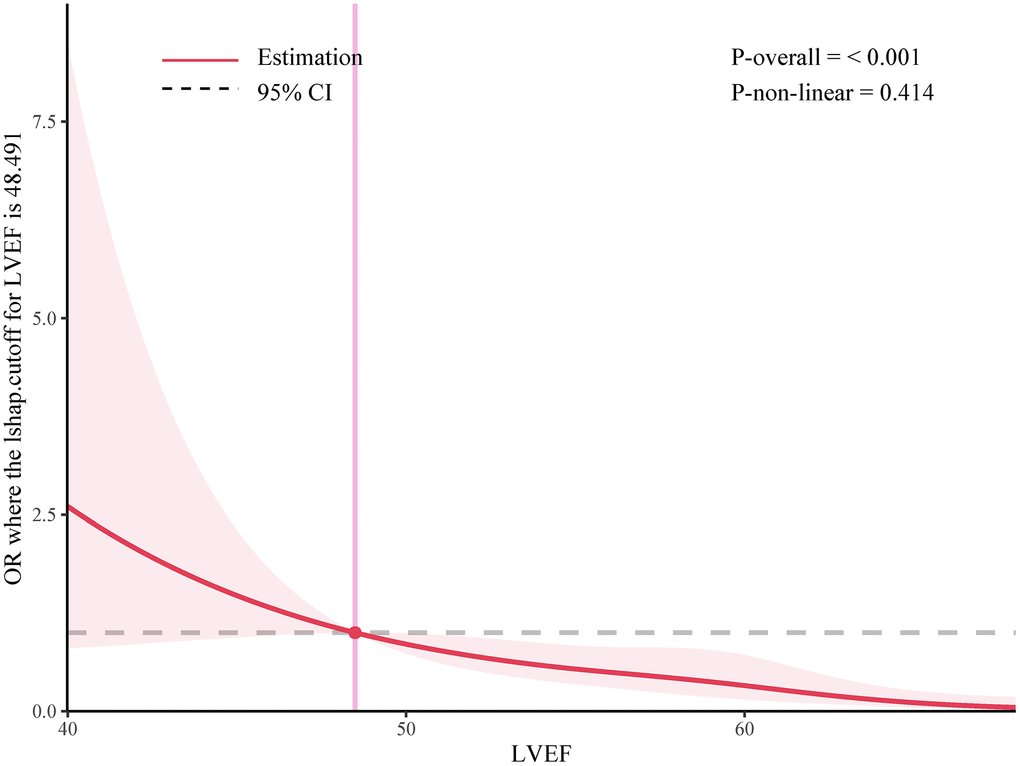
Figure 3. Adjusted ORs of the 3-month primary outcome according to LVEF levels after IVT. OR and 95% CI derived from restricted cubic spline regression, with knots placed at the 5th, 35th, 65th, and 95th percentiles of the levels of LVEF after IVT.
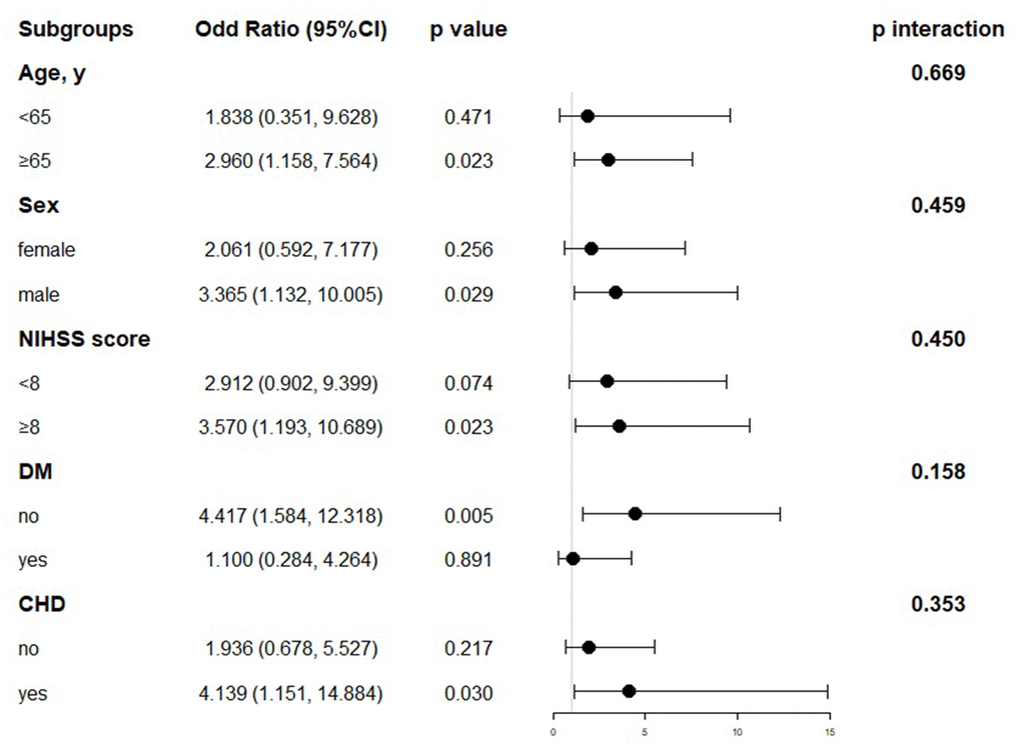
Figure 4. Subgroup analyses for poor outcomes by LVEF after IVT. NIHSS, National Institutes of Health Stroke Scale; DM, Diabetes mellitus; CHD, Coronary heart disease.
Discussion
In this prospective interventional cohort study, we explored the relationship between LVSD and prognosis of AIS patients with 273 cases from the Hefei Hospital Affiliated to Anhui Medical University. The main light of our findings was as follows: (1) The reduced LVEF was significantly associated with END, in-hospital mortality and poor outcomes at 90 days, even after adjusting for sex, age and other comorbidities. (2) RCS analysis showed a negative linear dose-response association between LVEF and functional outcomes at 90 days. (3) The optimal cutoff values of the LVEF after rt-PA treatment for predicting 90-days poorer functional outcomes were 0.48.
Despite the continuous updating of guidelines and widespread promotion of endovascular therapy, intravenous thrombolysis is considered as a preferred and fundamental care for AIS patients who meet certain time-window criteria. Previous studies have reported a correlation between heart failure and cerebral ischemic lesions [20, 21], which indicates a common pathophysiological mechanism underlying AIS and cardiac dysfunction. Data showed that the prevalence of LVSD in AIS patients is as high as 22% [22]. Surprisingly, the majority of contemporary trials and IVT registries did not report or focus on baseline data on cardiac diseases [23–25]. Therefore, few evidence are available about the neurofunctional predictive role of LVSD or heart failure in AIS patients; besides, most studies involved those patients who have not been treated with IVT [26–28].
Studies related to the effect of LVSD or heart failure on functional outcomes and mortality in AIS patients who underwent IVT showed conflicting results. Wahlgren et al. reported that a history of cardiac failure was associated with a 36% increased risk of 90-day mortality [29]. However, recall records of heart failure may underestimate the actual clinical presence of LVSD [30]. In our cohort, 23% of AIS patients had LVSD confirmed by echocardiography vs only 10% with clinical diagnosis of heart failure as determined from self-report of hospital records. While the study by Wei et al. suggested that LVEF<40% was only able to influence mortality, but not neurological outcomes (disability) in AIS patients undergoing IVT [7]. The negative result of the latter study may be attributed to the inclusion of relatively severe heart failure patients (2.4% patients with LVEF<50%), compared with our cohort of AIS patients with LVSD (15.0% patients with LVEF<50%). Therefore, in our view, these inconsistent results may be partially due to different definitions of heart failure and categories of LVEF levels. In the current study, we evaluated the association of LVSD with clinical outcomes in AIS patients against the latest international guidelines for heart failure [31]. Our findings were consistent with a recent study that had similar baseline LVEF characteristics and reported adverse functional outcomes at 90 days in AIS patients treated with IV thrombolysis [32]. In contrast, we added subgroup analysis to improve the stability of the results, and found the best LVEF predictors (48%) of adverse neurological outcomes through restricted cubic spline analysis.
Moreover, previous studies have shown that post-thrombolysis early neurological function changes, especially END, could lead to prolonged hospitalization and severe poor prognosis [33, 34]. In addition to previously identified risk factors, including diabetes [18], NIHSS on admission [35], and systolic blood pressure [36] associated with END after IVT, our study also found that LVSD patients were more prone to END and were closely associated with a poor 90 days functional prognosis.
The pathophysiological mechanism underlying the association between LVSD and poor outcomes in AIS patients was unclear and may be multifactorial. A reasonable hypothesis is that LVSD has a bidirectional effect on both the brain and heart [37, 38]. Although IVT helps to improve cerebral reperfusion, our findings instead support the phenomenon that reduced LVEF may still lead to poor cerebral perfusion, which possibly correlates with decreased brain autoregulation function and network structure after stroke onset [39]. Furthermore, other possible mechanisms for brain-heart interaction include sympathetic and parasympathetic regulation [40], neuroendocrine factors surge [41], and immune and inflammation response [42], negatively impacting the neurorehabilitation process and thus functional outcomes at 90 days. Furthermore, an increase in NIHSS values indicated the extent of cardiovascular autonomic control dysfunction [43]. In this way, either autonomic changes in the brain or heart may put AIS patients at an increased risk of cardiovascular complications and poor prognosis.
Interestingly, after adjusting for relevant confounding variables in the multivariate analysis, apart from LVSD, diabetes mellitus (DM) remained another significant risk factor related with poor outcomes in the current study. This finding was consistent with several prior studies that reported negative outcomes at 90-day in AIS patients treated with IVT or ET [44, 45]. Of note, DM or hyperglycemia was strongly associated with an increased risk of sICH after IVT [46]. A reasonable explanation was that vascular endothelial cell dysfunction and tissue acidosis as a result of central energy metabolism disruption lead to increased blood-brain barrier (BBB) permeability, resulting in cerebral edema, thereby increasing the risk of intracranial hemorrhage [47]. Of note, lower blood glucose levels could also induce sICH after IVT [48, 49]. While Zhang et al. reported that the effective rate after 24h IV was obviously when maintaining blood within the range of 7.0~9.0 mmol/L [48]. Moreover, American Heart Association (AHA) / American Stroke Association (ASA) guidelines suggested that stroke patients with co-existing DM may benefit most from IV time window controlling within 3 hours [50].
Identifying factors that evaluate the effectiveness of thrombolysis in AIS patients may help clinicians predict outcomes, guide early treatment decisions, and inform the development of new interventions. We provide greater insights to this field based on current prospective cohort studies that found a link between readily available clinical indicators of LVEF and the prognosis of AIS patients undergoing IVT. However, some limitations of this study still need to be addressed. Firstly, due to the limited time-window for IVT, LVEF measurement may be inconsistent if performed before as compared to after the stroke events. Secondly, cardiac assessment and echocardiogram at baseline were not always conducted by the same cardiologist, and the lack of validation of heart structure, vascular status, or serological markers (NT pro-BNP) may reduce statistical efficacy. Thirdly, this study did not collect detailed follow-up treatment information, such as prescribed doses of antiplatelet, anticoagulant, or antihypertensive agents, which confounding factors may affect clinical prognosis.
Conclusions
Taken together, our findings indicated that LVSD was a stronger predictor of 90-day poor functional outcomes in AIS patients after IVT, suggesting the need to monitor and optimize LVEF in stroke management.
Author Contributions
Yu Wang and Long Wang were involved in the study design. Chi Zhang contributed to collect the data, perform experiments and conduct statistical analyses. Jun-Cang Wu, Zheng Tan, and Xiao-Lu He helped with recruitment and data acquisition. Fei Li helped analyze and interpret the data. Chi Zhang was involved in the editing of the manuscript. All authors have read and agreed to the submission and publication of the manuscript.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Ethical Statement and Consent
The study procedures were conducted in accordance with the Declaration of Helsinki. Approval was obtained from the Ethical Committee of Second People’s Hospital of Hefei (No. KY2021-0112). All patients provided written consent.
Funding
This study was supported by Hefei Health and Wellness Applied Medicine Program (Hwk2023zd022) and Hefei Key Common Technology R&D Project (GJ2022SM07).
References
- 1. GBD 2019 Stroke Collaborators. Global, regional, and national burden of stroke and its risk factors, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 2021; 20:795–820. https://doi.org/10.1016/S1474-4422(21)00252-0 [PubMed]
- 2. Liu C, Han S, Zheng J, Wang H, Li S, Li J. EphA4 regulates white matter remyelination after ischemic stroke through Ephexin-1/RhoA/ROCK signaling pathway. Glia. 2022; 70:1971–91. https://doi.org/10.1002/glia.24232 [PubMed]
- 3. Liu Y, Gebrezgiabhier D, Reddy AS, Davis E, Zheng Y, Arturo Larco JL, Shih AJ, Pandey AS, Savastano LE. Failure modes and effects analysis of mechanical thrombectomy for stroke discovered in human brains. J Neurosurg. 2021; 136:197–204. https://doi.org/10.3171/2020.11.JNS203684 [PubMed]
- 4. Paul S, Candelario-Jalil E. Emerging neuroprotective strategies for the treatment of ischemic stroke: An overview of clinical and preclinical studies. Exp Neurol. 2021; 335:113518. https://doi.org/10.1016/j.expneurol.2020.113518 [PubMed]
- 5. Tsivgoulis G, Katsanos AH, Sandset EC, Turc G, Nguyen TN, Bivard A, Fischer U, Khatri P. Thrombolysis for acute ischaemic stroke: current status and future perspectives. Lancet Neurol. 2023; 22:418–29. https://doi.org/10.1016/S1474-4422(22)00519-1 [PubMed]
- 6. Liu Q, Shi K, Wang Y, Shi FD. Neurovascular Inflammation and Complications of Thrombolysis Therapy in Stroke. Stroke. 2023; 54:2688–97. https://doi.org/10.1161/STROKEAHA.123.044123 [PubMed]
- 7. Wei N, Wei Y, Nie X, Liu X, Xiang X, Pan Y, Meng X, Liu L, Wang Y. Effect of left ventricular ejection fraction Spectrum on 1-Year mortality in patients with acute ischemic stroke or transient ischemic attack. CNS Neurosci Ther. 2023; 29:3518–27. https://doi.org/10.1111/cns.14285 [PubMed]
- 8. Tan BYQ, Leow AS, Lee TH, Gontu VK, Andersson T, Holmin S, Wong HF, Lin CM, Cheng CK, Sia CH, Ngiam N, Ng ZX, Yeo J, et al. Left ventricular systolic dysfunction is associated with poor functional outcomes after endovascular thrombectomy. J Neurointerv Surg. 2021; 13:515–8. https://doi.org/10.1136/neurintsurg-2020-016216 [PubMed]
- 9. Li Y, Fitzgibbons TP, McManus DD, Goddeau RP
Jr , Silver B, Henninger N. Left Ventricular Ejection Fraction and Clinically Defined Heart Failure to Predict 90-Day Functional Outcome After Ischemic Stroke. J Stroke Cerebrovasc Dis. 2019; 28:371–80. https://doi.org/10.1016/j.jstrokecerebrovasdis.2018.10.002 [PubMed] - 10. Baker AD, Schwamm LH, Sanborn DY, Furie K, Stretz C, Mac Grory B, Yaghi S, Kleindorfer D, Sucharew H, Mackey J, Walsh K, Flaherty M, Kissela B, et al. Acute Ischemic Stroke, Depressed Left Ventricular Ejection Fraction, and Sinus Rhythm: Prevalence and Practice Patterns. Stroke. 2022; 53:1883–91. https://doi.org/10.1161/STROKEAHA.121.036706 [PubMed]
- 11. Rojek A, Gąsecki D, Fijałkowski M, Kowalczyk K, Kwarciany M, Wolf J, Nyka W, Boutouyrie P, Laurent S, Narkiewicz K. Left ventricular ejection fraction and aortic stiffness are independent predictors of neurological outcome in acute ischemic stroke. J Hypertens. 2016; 34:2441–8. https://doi.org/10.1097/HJH.0000000000001095 [PubMed]
- 12. Gentile L, Pracucci G, Saia V, Falcou A, Biraschi F, Zini A, Simonetti L, Riva L, Bigliardi G, Vallone S, Nencini P, Limbucci N, Diomedi M, et al, and Italian Registry of Endovascular Treatment in Acute Stroke (IRETAS) Collaborators. Mechanical thrombectomy in patients with heart failure: the Italian registry of Endovascular Treatment in Acute Stroke. Neurol Sci. 2023; 44:3577–85. https://doi.org/10.1007/s10072-023-06830-9 [PubMed]
- 13. Serber SL, Rinsky B, Kumar R, Macey PM, Fonarow GC, Harper RM. Cerebral blood flow velocity and vasomotor reactivity during autonomic challenges in heart failure. Nurs Res. 2014; 63:194–202. https://doi.org/10.1097/NNR.0000000000000027 [PubMed]
- 14. Chen X, Lu D, Guo N, Kang Z, Zhang K, Wang J, Men X, Lu Z, Qiu W. Left ventricular ejection fraction and right atrial diameter are associated with deep regional CBF in arteriosclerotic cerebral small vessel disease. BMC Neurol. 2021; 21:67. https://doi.org/10.1186/s12883-021-02096-w [PubMed]
- 15. Adams HP
Jr , Bendixen BH, Kappelle LJ, Biller J, Love BB, Gordon DL, Marsh EE 3rd. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke. 1993; 24:35–41. https://doi.org/10.1161/01.str.24.1.35 [PubMed] - 16. Lyden P, Raman R, Liu L, Emr M, Warren M, Marler J. National Institutes of Health Stroke Scale certification is reliable across multiple venues. Stroke. 2009; 40:2507–11. https://doi.org/10.1161/STROKEAHA.108.532069 [PubMed]
- 17. Banks JL, Marotta CA. Outcomes validity and reliability of the modified Rankin scale: implications for stroke clinical trials: a literature review and synthesis. Stroke. 2007; 38:1091–6. https://doi.org/10.1161/01.STR.0000258355.23810.c6 [PubMed]
- 18. Che F, Wang A, Ju Y, Ding Y, Duan H, Geng X, Zhao X, Wang Y. Early neurological deterioration in acute ischemic stroke patients after intravenous thrombolysis with alteplase predicts poor 3-month functional prognosis - data from the Thrombolysis Implementation and Monitor of Acute Ischemic Stroke in China (TIMS-China). BMC Neurol. 2022; 22:212. https://doi.org/10.1186/s12883-022-02737-8 [PubMed]
- 19. Hacke W, Kaste M, Fieschi C, von Kummer R, Davalos A, Meier D, Larrue V, Bluhmki E, Davis S, Donnan G, Schneider D, Diez-Tejedor E, Trouillas P. Randomised double-blind placebo-controlled trial of thrombolytic therapy with intravenous alteplase in acute ischaemic stroke (ECASS II). Second European-Australasian Acute Stroke Study Investigators. Lancet. 1998; 352:1245–51. https://doi.org/10.1016/s0140-6736(98)08020-9 [PubMed]
- 20. Barkhudaryan A, Doehner W, Scherbakov N. Ischemic Stroke and Heart Failure: Facts and Numbers. An Update. J Clin Med. 2021; 10:1146. https://doi.org/10.3390/jcm10051146 [PubMed]
- 21. Katsanos AH, Parissis J, Frogoudaki A, Vrettou AR, Ikonomidis I, Paraskevaidis I, Triantafyllou N, Kargiotis O, Voumvourakis K, Alexandrov AV, Tsivgoulis G. Heart failure and the risk of ischemic stroke recurrence: A systematic review and meta-analysis. J Neurol Sci. 2016; 362:182–7. https://doi.org/10.1016/j.jns.2016.01.053 [PubMed]
- 22. Devulapalli S, Dunlap S, Wilson N, Cockburn S, Kurukumbi M, Mehrotra P, Jayam-Trouth A. Prevalence and clinical correlation of left ventricular systolic dysfunction in african americans with ischemic stroke. J Stroke Cerebrovasc Dis. 2014; 23:1965–8. https://doi.org/10.1016/j.jstrokecerebrovasdis.2014.01.026 [PubMed]
- 23. Nagakane Y, Christensen S, Brekenfeld C, Ma H, Churilov L, Parsons MW, Levi CR, Butcher KS, Peeters A, Barber PA, Bladin CF, De Silva DA, Fink J, et al, and EPITHET Investigators. EPITHET: Positive Result After Reanalysis Using Baseline Diffusion-Weighted Imaging/Perfusion-Weighted Imaging Co-Registration. Stroke. 2011; 42:59–64. https://doi.org/10.1161/STROKEAHA.110.580464 [PubMed]
- 24. Schlemm L, Braemswig TB, Boutitie F, Vynckier J, Jensen M, Galinovic I, Simonsen CZ, Cheng B, Cho TH, Fiehler J, Puig J, Thijs V, Fiebach J, et al, and WAKE-UP Investigators. Cerebral Microbleeds and Treatment Effect of Intravenous Thrombolysis in Acute Stroke: An Analysis of the WAKE-UP Randomized Clinical Trial. Neurology. 2022; 98:e302–14. https://doi.org/10.1212/WNL.0000000000013055 [PubMed]
- 25. Menon BK, Buck BH, Singh N, Deschaintre Y, Almekhlafi MA, Coutts SB, Thirunavukkarasu S, Khosravani H, Appireddy R, Moreau F, Gubitz G, Tkach A, Catanese L, et al, and AcT Trial Investigators. Intravenous tenecteplase compared with alteplase for acute ischaemic stroke in Canada (AcT): a pragmatic, multicentre, open-label, registry-linked, randomised, controlled, non-inferiority trial. Lancet. 2022; 400:161–9. https://doi.org/10.1016/S0140-6736(22)01054-6 [PubMed]
- 26. Sung PH, Chen KH, Lin HS, Chu CH, Chiang JY, Yip HK. The Correlation between Severity of Neurological Impairment and Left Ventricular Function in Patients after Acute Ischemic Stroke. J Clin Med. 2019; 8:190. https://doi.org/10.3390/jcm8020190 [PubMed]
- 27. Kim WJ, Nah HW, Kim DH, Cha JK. Association between Left Ventricular Dysfunction and Functional Outcomes at Three Months in Acute Ischemic Stroke. J Stroke Cerebrovasc Dis. 2016; 25:2247–52. https://doi.org/10.1016/j.jstrokecerebrovasdis.2016.05.004 [PubMed]
- 28. Takahashi S, Ishizuka K, Hoshino T, Mizuno T, Nishimura A, Toi S, Kitagawa K. Long-Term Outcome in Patients With Acute Ischemic Stroke and Heart Failure. Circ J. 2023; 87:401–8. https://doi.org/10.1253/circj.CJ-21-0937 [PubMed]
- 29. Wahlgren N, Ahmed N, Eriksson N, Aichner F, Bluhmki E, Dávalos A, Erilä T, Ford GA, Grond M, Hacke W, Hennerici MG, Kaste M, Köhrmann M, et al, and Safe Implementation of Thrombolysis in Stroke-MOnitoring STudy Investigators. Multivariable analysis of outcome predictors and adjustment of main outcome results to baseline data profile in randomized controlled trials: Safe Implementation of Thrombolysis in Stroke-MOnitoring STudy (SITS-MOST). Stroke. 2008; 39:3316–22. https://doi.org/10.1161/STROKEAHA.107.510768 [PubMed]
- 30. Palumbo V, Baldasseroni S, Nencini P, Pracucci G, Arba F, Piccardi B, Marella GA, Di Bari M, Gensini GF, Marchionni N, Inzitari D. The coexistence of heart failure predicts short term mortality, but not disability, in patients with acute ischemic stroke treated with thrombolysis: the Florence area Registry. Eur J Intern Med. 2012; 23:552–7. https://doi.org/10.1016/j.ejim.2012.03.015 [PubMed]
- 31. Kosaraju A, Goyal A, Grigorova Y, Makaryus AN: Left Ventricular Ejection Fraction. In: StatPearls. edn. Treasure Island (FL) ineligible companies. Disclosure: Amandeep Goyal declares no relevant financial relationships with ineligible companies. Disclosure: Yulia Grigorova declares no relevant financial relationships with ineligible companies. Disclosure: Amgad Makaryus declares no relevant financial relationships with ineligible companies. 2023.
- 32. Chee RCH, Lin NH, Ho JSY, Leow AST, Li TY, Lee ECY, Chan MY, Kong WKF, Yeo TC, Chai P, Yip JW, Poh KK, Sharma VK, et al. Left Ventricular Ejection Fraction Association with Acute Ischemic Stroke Outcomes in Patients Undergoing Thrombolysis. J Cardiovasc Dev Dis. 2023; 10:231. https://doi.org/10.3390/jcdd10060231 [PubMed]
- 33. Gong P, Liu Y, Gong Y, Chen G, Zhang X, Wang S, Zhou F, Duan R, Chen W, Huang T, Wang M, Deng Q, Shi H, et al. The association of neutrophil to lymphocyte ratio, platelet to lymphocyte ratio, and lymphocyte to monocyte ratio with post-thrombolysis early neurological outcomes in patients with acute ischemic stroke. J Neuroinflammation. 2021; 18:51. https://doi.org/10.1186/s12974-021-02090-6 [PubMed]
- 34. Mori M, Naganuma M, Okada Y, Hasegawa Y, Shiokawa Y, Nakagawara J, Furui E, Kimura K, Yamagami H, Kario K, Okuda S, Koga M, Minematsu K, Toyoda K. Early neurological deterioration within 24 hours after intravenous rt-PA therapy for stroke patients: the Stroke Acute Management with Urgent Risk Factor Assessment and Improvement rt-PA Registry. Cerebrovasc Dis. 2012; 34:140–6. https://doi.org/10.1159/000339759 [PubMed]
- 35. Seners P, Turc G, Tisserand M, Legrand L, Labeyrie MA, Calvet D, Meder JF, Mas JL, Oppenheim C, Baron JC. Unexplained early neurological deterioration after intravenous thrombolysis: incidence, predictors, and associated factors. Stroke. 2014; 45:2004–9. https://doi.org/10.1161/STROKEAHA.114.005426 [PubMed]
- 36. Park TH, Lee JK, Park MS, Park SS, Hong KS, Ryu WS, Kim DE, Park MS, Choi KH, Kim JT, Kang J, Kim BJ, Han MK, et al. Neurologic deterioration in patients with acute ischemic stroke or transient ischemic attack. Neurology. 2020; 95:e2178–91. https://doi.org/10.1212/WNL.0000000000010603 [PubMed]
- 37. Battaglini D, Robba C, Lopes da Silva A, Dos Santos Samary C, Leme Silva P, Dal Pizzol F, Pelosi P, Rocco PR. Brain-heart interaction after acute ischemic stroke. Crit Care. 2020; 24:163. https://doi.org/10.1186/s13054-020-02885-8 [PubMed]
- 38. Doehner W, Ural D, Haeusler KG, Čelutkienė J, Bestetti R, Cavusoglu Y, Peña-Duque MA, Glavas D, Iacoviello M, Laufs U, Alvear RM, Mbakwem A, Piepoli MF, et al. Heart and brain interaction in patients with heart failure: overview and proposal for a taxonomy. A position paper from the Study Group on Heart and Brain Interaction of the Heart Failure Association. Eur J Heart Fail. 2018; 20:199–215. https://doi.org/10.1002/ejhf.1100 [PubMed]
- 39. Siedler G, Sommer K, Macha K, Marsch A, Breuer L, Stoll S, Engelhorn T, Dörfler A, Arnold M, Schwab S, Kallmünzer B. Heart Failure in Ischemic Stroke: Relevance for Acute Care and Outcome. Stroke. 2019; 50:3051–6. https://doi.org/10.1161/STROKEAHA.119.026139 [PubMed]
- 40. Silvani A, Calandra-Buonaura G, Dampney RA, Cortelli P. Brain-heart interactions: physiology and clinical implications. Philos Trans A Math Phys Eng Sci. 2016; 374:20150181. https://doi.org/10.1098/rsta.2015.0181 [PubMed]
- 41. Fassbender K, Schmidt R, Mössner R, Daffertshofer M, Hennerici M. Pattern of activation of the hypothalamic-pituitary-adrenal axis in acute stroke. Relation to acute confusional state, extent of brain damage, and clinical outcome. Stroke. 1994; 25:1105–8. https://doi.org/10.1161/01.str.25.6.1105 [PubMed]
- 42. Iadecola C, Anrather J. The immunology of stroke: from mechanisms to translation. Nat Med. 2011; 17:796–808. https://doi.org/10.1038/nm.2399 [PubMed]
- 43. Hilz MJ, Moeller S, Akhundova A, Marthol H, Pauli E, De Fina P, Schwab S. High NIHSS values predict impairment of cardiovascular autonomic control. Stroke. 2011; 42:1528–33. https://doi.org/10.1161/STROKEAHA.110.607721 [PubMed]
- 44. Desilles JP, Meseguer E, Labreuche J, Lapergue B, Sirimarco G, Gonzalez-Valcarcel J, Lavallée P, Cabrejo L, Guidoux C, Klein I, Amarenco P, Mazighi M. Diabetes mellitus, admission glucose, and outcomes after stroke thrombolysis: a registry and systematic review. Stroke. 2013; 44:1915–23. https://doi.org/10.1161/STROKEAHA.111.000813 [PubMed]
- 45. Dhillon PS, Butt W, Marei O, Podlasek A, McConachie N, Lenthall R, Nair S, Malik L, Bhogal P, Makalanda HL, Dineen RA, England TJ. Incidence and predictors of poor functional outcome despite complete recanalisation following endovascular thrombectomy for acute ischaemic stroke. J Stroke Cerebrovasc Dis. 2023; 32:107083. https://doi.org/10.1016/j.jstrokecerebrovasdis.2023.107083 [PubMed]
- 46. Liu SY, Cao WF, Wu LF, Xiang ZB, Liu SM, Liu HY, Pan Y, Nie F, Wu XM, Xie XF. Effect of glycated hemoglobin index and mean arterial pressure on acute ischemic stroke prognosis after intravenous thrombolysis with recombinant tissue plasminogen activator. Medicine (Baltimore). 2018; 97:e13216. https://doi.org/10.1097/MD.0000000000013216 [PubMed]
- 47. Thanvi B, Treadwell S, Robinson T. Early neurological deterioration in acute ischaemic stroke: predictors, mechanisms and management. Postgrad Med J. 2008; 84:412–7. https://doi.org/10.1136/pgmj.2007.066118 [PubMed]
- 48. Zhang Z, Qian M, Ge Z, Zhou P, Liu J, Chen J. Effects of blood glucose and glycosylated hemoglobin levels on intravenous thrombolysis in patients with acute cerebral infarction and type 2 diabetes mellitus. Pak J Med Sci. 2019; 35:862–7. https://doi.org/10.12669/pjms.35.3.8 [PubMed]
- 49. Cai Y, Zhang H, Li Q, Zhang P. Correlation Between Blood Glucose Variability and Early Therapeutic Effects After Intravenous Thrombolysis With Alteplase and Levels of Serum Inflammatory Factors in Patients With Acute Ischemic Stroke. Front Neurol. 2022; 13:806013. https://doi.org/10.3389/fneur.2022.806013 [PubMed]
- 50. Furie KL, Jayaraman MV. 2018 Guidelines for the Early Management of Patients With Acute Ischemic Stroke. Stroke. 2018; 49:509–10. https://doi.org/10.1161/STROKEAHA.118.020176 [PubMed]



