Introduction
The Hydra is a simple multicellular organism consisting of an endoderm and ectoderm, the latter composed mostly of epithelial cells with some interstitial cells [1]. Hydra contain continuously cycling stem cells that result in the doubling of tissue mass every three to four days [1, 2]. However, tissue homeostasis keeps organismal body mass in equilibrium; excess cells are utilized for asexual reproduction or are lost from Hydra tentacles post-differentiation [1, 2]. Thus, Hydra are characterized by proliferating epithelium, and it has been shown that the epithelium and interstitial stem cells of Hydra are characterized by continuous self-renewal and differentiation [1, 2]. Continuous cell cycling in Hydra typically prevents the accumulation of aged or damaged somatic cells by replacement with progeny from self-renewing stem cells [1, 2].
Thus, Hydra vulgaris (henceforth, “Hydra” unless other species are otherwise noted) exhibits negligible senescence under laboratory conditions, with no detectable increase in age-specific mortality over extended observation periods [1, 2] This suppression of senescence should not be equated with absolute “immortality” (complete lack of mortality), as extrinsic factors (e.g., predation, starvation) can still cause death [1, 2].
Hydra oligactis exhibits different lifecycle potentialities than Hydra vulgaris and is a powerful model for inducible aging. In asexual phases at stable warmer temperatures (18° C), Hydra oligactis exhibits negligible senescence similar to Hydra vulgaris, maintaining long-term viability through budding. However, cold temperatures (10° C) trigger sexual reproduction in many strains, inducing a rapid aging phenotype characterized by massive loss of interstitial stem cells, impaired regeneration, reduced motility and food capture, and high mortality (e.g., most individuals die within 3–4 months post-sexual maturity) [3, 4]. Transcriptomic studies of this induced aging pattern reveal upregulation of genes involved in gametogenesis, cellular senescence, apoptosis, and DNA repair, alongside downregulation of stem cell maintenance genes (including FoxO orthologs) [4]. Strains vary in this activity; thus, cold-sensitive (CS) lines undergo full aging and ~100% mortality after sexual reproduction, while cold-resistant (CR) lines can revert to asexual non-aging states [5]. This inducible switch makes Hydra oligactis a key model for studying stem cell loss, Wnt/β-catenin and FoxO pathway roles in longevity, and trade-offs between reproduction and aging resistance [4, 6]. The focus of the current hypothesis remains on Hydra vulgaris, but Hydra oligactis highlights the context-dependence of negligible senescence and potential costs of changing life history modes.
When considering life history, it is important to note that high cell turnover does not automatically eliminate all aging-related processes even in Hydra (e.g., Hydra vulgaris or asexual H. oligactis) exhibiting negligible senescence. Thus, stem cells can accumulate damage over time, and continuous proliferation carries inherent oncogenic risk, as evidenced by naturally occurring Hydra (Hydra oligactis and Pelmatohydra robusta) tumors [7]. These tumors demonstrate that Hydra can manifest dysregulation of homeostatic mechanisms resulting in neoplastic overgrowth. This fact underscores the need to monitor such trade-offs in any attempt to recapitulate the Hydra-like pattern in aging animal models.
Maintenance of the all-important stem cell compartment of Hydra is a function of the transcription factor Forkhead box O (FoxO), as FoxO knockdown in Hydra results in reduced growth and enhanced differentiation, while FoxO overexpression upregulates expression of “stemness” genes and increases proliferation of stem and progenitor cells [1]. Indeed, FoxO overexpression in Hydra upregulates expression of stemness genes (e.g., piwi-like and nanos-like markers) even in terminally differentiated cells such as nematocytes [1]. This suggests partial reprogramming or reactivation of stemness signatures in differentiated cells, rather than full cellular immortalization. True cellular immortalization would require demonstration of indefinite replicative capacity, bypass of senescence-associated checkpoints (if present), and stable long-term proliferation in culture or in vivo. These criteria are not met by the Hydra nematocyte data, which instead show transient stemness gene induction without evidence of sustained cell division or escape from a post-mitotic fate. Nevertheless, this finding is relevant to our hypothesis, as it indicates that Hydra-like gene expression programs could enhance resilience or functional maintenance in post-mitotic rotifer cells without necessitating cell proliferation. Further, in addition, and also of relevance to the current manuscript, different FoxO genotypes have been correlated to human longevity [8, 9].
As noted above, FoxO knockdown in Hydra results in reduced organismal growth and enhanced terminal differentiation, particularly of epithelial cells and nematocytes, shifting the balance toward post-mitotic states [1]. This phenotype is not functionally equivalent to mammalian replicative senescence. Instead, the mammalian pattern involves permanent cell cycle exit mediated by the p53/p21 and/or p16/Rb pathways, accompanied by a senescence-associated secretory phenotype (SASP), increased heterochromatin foci, and resistance to mitogenic stimuli. Hydra lacks true orthologs of some of these canonical senescence effectors and does not exhibit a true senescent state characterized by irreversible growth arrest or pro-inflammatory signaling. Instead, FoxO acts primarily as a gatekeeper of stem cell multipotency and continuous proliferation, enabling tissue self-renewal and negligible senescence without the diversified roles (e.g., tumor suppression, metabolic regulation, or stress response integration) observed in bilaterians. Thus, FoxO in Hydra functions more as a regulator of inhibited senescence through maintenance of stemness than as a theoretical "immortality switch" (a hypothetical molecular mechanism or genetic factor that could be activated or manipulated to confer indefinite replicative potential, and/or permanent survival without aging) in the mammalian sense.
Importantly, single-cell RNA sequencing has been conducted in Hydra, on cell types ranging from stem cells to terminally differentiated cells [10]. These data can provide useful targets for genetic modification, with the aim of elucidating gene expression programs contributing to Hydra’s continual self-renewal and suppressed senescence. These gene expression programs can be used to manipulate other model organisms to influence aging-related phenotypes.
The Hydra model is contrasted to that of rotifers, which, although they mostly proliferate asexually (like the typical Hydra pattern), are eutelic; that is, they have a fixed number of somatic cells upon adulthood and do not exhibit somatic cell proliferation after hatching [11]. Thus, Hydra and rotifers represent extremes on the spectrum of asexual invertebrate lifestyles and it is this profound difference that makes rotifers ideal models for evaluating the hypothesis outlined in this manuscript. The rotifer's eutelic nature (fixed somatic cell number post-hatching, limited proliferation) can act as a "stringent test" for the Hydra-like interventions described as an objective of this paper, given rotifers’ significant deviation from the Hydra-like life plan.
Indeed, rotifers have been effective invertebrate models for studying aging [11–15]. For example, the rotifer Brachionus manjavacas is particularly well suited for such studies, exhibiting a short lifespan (median of 10 days) and well-defined life stages including a highly structured and reproducible aging phenotype [11]. The gene expression pattern associated with aging in Brachionus manjavacas has been studied in detail [11] and serves as a counterpoint to the molecular data obtained from Hydra. One finding from such studies is that altered gene expression associated with aging seems to begin at relatively early (young) age points [11]. These data suggest that genetic suppression of senescence in the rotifer would be optimally achieved via early intervention. In general, changes in gene expression related to metabolism, cell signaling, mitochondrial function, and proteostasis were observed in rotifers [11], all relevant to human aging.
Canonical mammalian senescence pathways are incompletely conserved in Hydra and rotifers (Table 1). In mammals, the p53/p21 axis drives DNA damage-induced cell cycle arrest and SASP, while the p16INK4a/Rb pathway enforces permanent G1 arrest in response to oncogenic or replicative stress. Hydra possesses a p53-like ortholog that participates in DNA damage response and apoptosis [1] but lacks a clear p21 ortholog and shows no evidence of p53-dependent permanent arrest or SASP-like secretory activity. No definitive p16INK4a ortholog has been identified, and Rb-like proteins exist but are not linked to senescence-like phenotypes. Rotifers similarly exhibit limited orthologs of these pathways, with no strong p53/p21 or p16/Rb signaling documented [11]. Although rotifers retain a more complete set of vertebrate-shared genes than C. elegans or Drosophila (due to less extensive genome reduction), their senescence appears driven more by eutelic constraints and gradual functional decline than by canonical arrest mechanisms [13]. These differences explain why Hydra achieves negligible senescence through continuous stem cell renewal rather than replicative arrest, and why rotifers represent a stringent test model for introducing Hydra-like self-renewal patterns.
Table 1. Comparative overview of key senescence and stemness pathways across species.
| Pathway/Gene Family | Mammals (mouse/human) | Hydra | Rotifers | C. elegans | Drosophila | Notes on conservation and/or function | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| p53/p21 axis | Present, fundamental for DNA damage arrest, SASP | p53 ortholog present (DNA damage/apoptosis) no strong p21 ortholog | Limited orthologs; no clear p21 | daf-16 (FOXO) dominant; no p53/p21 | foxo dominant; no p53/p21 | Poorly conserved in invertebrates; Hydra uses stem renewal instead of arrest | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| p16/Rb axis | Present; p16 induces Rb-mediated G1 arrest | No clear p16; Rb-like present but not senescence-linked | No clear p16; Rb-like possible | No p16/Rb orthologs | No p16/Rb orthologs | Absent or divergent in invertebrates | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FOXO/FoxO family | FOXO1/3/4; longevity variants, tumor suppression, metabolism | High FoxO in stem cells; maintains multipotency/self-renewal | FoxO ortholog; aging-related but limited stemness role | daf-16; major longevity regulator | foxo; stress resistance, lifespan | Highly conserved; central to Hydra immortality | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stemness markers | Present in germline/stem cells | High in stem lineages; continuous renewal | Limited somatic stem cells | Present in germline | Present in germline | Strong in Hydra; weak somatic in rotifers | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Senescence phenotype | Replicative arrest + SASP | Absent; negligible senescence | Present -eutelic aging | Present -replicative | Present -replicative | Hydra lacks machinery for arrest | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Retained vertebrate-shared genes absent in C. elegans/Drosophila | N/A (vertebrate baseline) | Moderate retention of some vertebrate-associated genes | High retention of genes shared with vertebrates (absent/lost in C. elegans/Drosophila), including stress responses, xenobiotic resistance, metabolic regulation [11] | Significant genome reduction; many vertebrate-associated genes absent | Significant genome reduction; many vertebrate-associated genes absent | Rotifers retain a more complete set of vertebrate-like genes, making them a stronger bridge model for testing aging interventions | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Presence and functional role of key pathways in senescence and stemness. Hydra relies on stem cell renewal rather than canonical arrest pathways, while rotifers share more vertebrate-like genes than nematodes or flies but lack robust senescence effectors. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Despite these differences with mammals, it is important to note that rotifers represent a more “human-like” invertebrate aging model compared to C. elegans and Drosophila and [11]. This advantage derives from the fact that rotifers, unlike those other organisms, are not characterized by significant genome reduction [11]. Thus, rotifers have been found to share important genes with vertebrates that are not present in other invertebrate model systems such as C. elegans or Drosophila [11]. Some of these retained genes are involved in aging-relevant processes (Table 1), including stress responses, xenobiotic resistance, and metabolic regulation [11]. This greater conservation of vertebrate-associated gene content underscores the utility of rotifers as a particularly informative model system for evaluating the effects of introducing Hydra-like patterns of gene expression. By using an organism that already possesses a more complete set of aging-related orthologs, the proposed experiments can better test the transferability and functional impact of Hydra's self-renewal programs in an aging context that is relatively more similar to vertebrates.
Therefore, rotifers represent a relevant invertebrate model for human aging. This, combined with the other rotifer traits previously mentioned (e.g., rapidly aging, eutelic), make it particularly useful for evaluating the potential of delayed senescence through partial recapitulation of the Hydra genetic program. On the one hand, rotifers being at the opposite end of the aging/self-renewal spectrum from Hydra, make anti-aging findings generated from this model especially powerful, as any positive results would be obtained despite the rotifer starting from a life history point opposite of the desired endpoint. On the other hand, being a simple invertebrate organism makes the possibility of success with rotifers greater than immediately attempting these approaches in a vertebrate model (e.g., mice). Mouse experiments would instead be a long-range future possibility depending on the rotifer (and subsequent organisms testing) data.
Daphnia are another invertebrate model of aging, which have been previously utilized to evaluate the theory of antagonistic pleiotropy [16]. Therefore, Daphnia represent another organism that can be utilized alongside rotifers to test the hypothesis presented here.
Also, of relevance to the hypothesis of the current paper is the MRL mouse strain, which exhibits enhanced healing properties, allowing for efficient wound healing in multiple tissue types without formation of fibrotic scars [17–19]. Careful studies of these mice have identified changes in gene expression that may explain these properties, including modified expression of stem cell markers that is likely linked to altered numbers of certain progenitor cell types [17]. One hypothesis proposed was that these mice may exhibit an extended lifespan as an extension of their enhanced healing [18]. However, the opposite was shown to be the case, with these mice exhibiting shorter lifespans than other strains [19]; one possibility is that enhanced IGF-1 levels in the MRL strain may mediate the inverse relationship between enhanced healing and decreased longevity [17]. Like the rotifer, the MRL mouse is an attractive model for evaluating whether Hydra-like patterns of gene expression could promote delayed senescence at the organismal level.
Hypothesis
Hydra and rotifers can therefore be considered as representatives of the opposite ends of the invertebrate lifecycle and aging spectrum, with the negligibly senescent, continuous replicating Hydra at one end and the short-lived, rapidly aging, and eutelic rotifers at the other end. Both Hydra and rotifers are tractable for genetic manipulation via exogenous gene expression including overexpression and genetic knockdown [1, 9–11, 13–15, 20]. Therefore, these two organisms represent an opportunity to test the hypothesis that Hydra-like patterns of gene expression, including but not limited to levels of FoxO, can progressively repress senescence in aging animal models.
We hypothesize that delayed senescence at the organismal level is possible through recapitulation of Hydra-like patterns of gene expression in rotifers, and that data obtained may help generate hypotheses for somatic interventions and prioritize pathways for mammalian validation in future studies.
Hydra-like gene expression patterns refer to the molecular signature that supports continuous stem cell self-renewal, multipotency, and negligible senescence in Hydra vulgaris [1, 10]. This includes: (a) sustained high activity of the transcription factor FoxO, which maintains stem cell proliferation and suppresses terminal differentiation; (b) upregulation of stemness genes (e.g., orthologs of piwi, nanos, vasa-like, and other interstitial/stem cell markers) in stem and progenitor populations; (c) transcriptomic profiles enriched in pathways for DNA repair, proteostasis, mitochondrial function, and stress resistance, with minimal expression of aging-associated genes (e.g., those involved in proteotoxic stress or inflammation). These patterns could be assessed quantitatively via RNA-seq, comparing pre- and post-perturbation rotifer transcriptomes to Hydra stem/progenitor cell clusters [10]. “More Hydra-like” could be defined operationally as achieving ≥0.7 Pearson correlation with Hydra stem cell states or significant upregulation of conserved stemness markers without major fitness costs.
The rotifer's eutelic adult soma (fixed post-hatching cell number with minimal somatic proliferation) fundamentally limits the potential for stem cell compartment expansion or progenitor replenishment [13, 20]. Therefore, the primary mechanism by which Hydra-like gene expression is expected to delay rotifer senescence is through enhanced resilience, proteostasis, stress resistance, and damage repair in existing post-mitotic somatic cells, rather than renewed tissue turnover. Candidate proliferative populations in rotifers are limited to germline-associated cells or rare, tissue-specific progenitors (if any); somatic stem cell activity is not a feature of the adult. Accordingly, success will be measured by functional readouts such as extended post-mitotic cell lifespan, improved mitochondrial function, reduced proteotoxic stress, and delayed aging phenotypes (e.g., reproductive decline, locomotion impairment), rather than increased cell numbers or proliferation markers. Attempts to override eutely (e.g., via forced expression of proliferation regulators) would likely disrupt developmental constraints, leading to malformations, reduced fitness, or tumor-like overgrowth, and will be avoided or tightly controlled in initial experiments. Thus, if increased cell numbers or proliferation markers do occur, this would likely be associated with the aforementioned negative outcomes that will be subject to careful monitoring.
The hypothesis presented here builds on existing data from single-cell RNA-seq in Hydra [10] and aging transcriptomics in Brachionus manjavacas [11]. Recapitulating such patterns for lifespan extension has already been conceptually discussed in reviews [1, 21, 22]. However, the bidirectional approach (Hydra to rotifer overexpression/knockdown) and the reverse (imposing rotifer aging signatures on Hydra) is presented here as a novel integration for iterative refinement. It is not merely a logical extension of prior FoxO-focused Hydra work or of general longevity pathway reviews. The current proposal systematically tests causality in both directions, moving from the extreme of negligible senescence and continuous stem cell renewal (Hydra) to the opposite extreme of short lifespan, eutelic soma, and rapid aging (rotifer), and then reverses the flow (rotifer to Hydra). Thus, this strategy allows for a more rigorous evaluation of which Hydra-like expression patterns are truly causal for delaying senescence vs. which are context-specific or are “bystander” or “passenger” features.
No previous proposal has systematically combined these opposite lifecycle endpoints in an iterative, bidirectional framework to refine targets and validate mechanisms. This dual-direction testing adds a layer of causal inference that single-direction studies (e.g., Hydra-to-other models only) or purely correlative reviews do not provide. It allows for clearer discrimination between necessary vs. sufficient elements of the Hydra genetic program and can in theory reduce the risk of transferring non-causal or maladaptive features into aging-prone organisms. The bidirectional design is therefore a distinctive strength of the current hypothesis; it outlines a structured strategy for progressive iterative refinement that has not been explicitly discussed in previous work on Hydra-inspired longevity pathways.
Evaluating the hypothesis
The basic experimental approach is to recapitulate elements of relevant Hydra gene expression programs in rotifers. Genetic manipulation, in an iterative fashion, can be utilized to progressively modify Brachionus manjavacas. Subsequently, more complex organisms (Daphnia to mice, including the MRL mouse) would be utilized, with the aim of repressing aging at the mammalian level. Data obtained from these studies can be used to evaluate geroprotective approaches to repress aging in humans.
Thus, the primary strategy (Figure 1) is to modulate endogenous rotifer orthologs (e.g., FoxO and stemness-related genes) to mimic Hydra expression levels and dynamics via CRISPR-based overexpression or knockdown. Limited heterologous expression of select Hydra genes (e.g., FoxO or piwi orthologs) may be tested in parallel where ortholog function is uncertain. Cross-species differences in cell types and developmental programs will be addressed by focusing on conserved functional modules (e.g., FoxO-mediated stemness) rather than direct cell-type matching. Given the rotifer's eutelic adult soma, success is expected to manifest primarily as enhanced resilience, proteostasis, and stress resistance in existing cells rather than new stem cell compartment expansion. Any attempt to override eutely will be monitored for developmental defects or fitness loss.
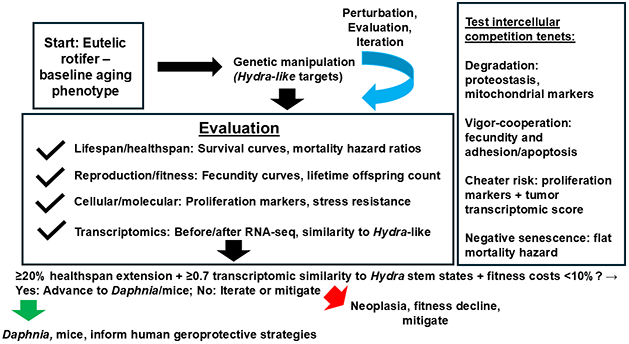
Figure 1. Detailed flowchart of the iterative experimental strategy to delay senescence by introducing Hydra-like gene expression in rotifers (Brachionus manjavacas). Key manipulations target rotifer orthologs (primarily FoxO and stemness markers) via CRISPR. Evaluation endpoints include lifespan/mortality hazard (survival curves), fertility (fecundity curves), locomotion (swimming assays), stress resistance, and transcriptomics (pre/post similarity to Hydra stem/progenitor states; ≥0.7 correlation threshold). Neoplasia monitoring uses proliferation markers (BrdU/phospho-histone H3) and deviation from Hydra tumor profiles. Iteration decisions are guided by predefined criteria (healthspan extension, transcriptomic shift, fitness constraints). Controls: wild-type and mock-treated rotifers. The framework also tests intercellular competition and multicellular aging through integrated measurements of cellular degradation, vigor-cooperation correlation, cheater emergence (neoplasia proxies), and negative senescence potential (mortality hazard). “Hydra-like” is defined in the text.
Transgenic rotifers exhibiting more Hydra-like patterns of gene expression (e.g., enhanced FoxO activity and stemness marker upregulation in existing post-mitotic cells) will be evaluated for changes in age-related mortality/senescence, health, and function, with emphasis on post-mitotic cell maintenance (e.g., proteostasis, mitochondrial integrity, stress resistance assays) rather than proliferation. Proliferation markers (e.g., BrdU incorporation, phospho-histone H3) will be monitored primarily to detect unintended overgrowth or neoplastic risk rather than to confirm stem cell expansion. Given eutely, any measurable delay in senescence is expected to arise from improved damage resistance/repair in the fixed somatic cell population, with germline effects potentially contributing to transgenerational healthspan benefits.
More specifically, the testing design could be as follows. The proposed experimental approach is iterative and begins with Brachionus manjavacas as the primary target. Rotifers will be subjected to CRISPR-mediated overexpression or knockdown of rotifer orthologs of key Hydra genes (primarily FoxO and 3–5 stemness markers such as piwi/nanos orthologs), with limited heterologous expression of Hydra genes where ortholog function is uncertain. Controls will include wild-type rotifers, mock-transfected animals, and negative-target manipulations (e.g., non-conserved genes). Primary endpoints for delayed senescence include: (a) lifespan and mortality hazard (survival curves, Kaplan-Meier analysis, hazard ratios); (b) fertility and reproduction (fecundity curves, lifetime offspring count, reproductive lifespan); (c) locomotion and functional decline (swimming speed, escape response assays over age); (d) stress resistance (oxidative, thermal, UV challenge survival), and (e) molecular markers (proteostasis via proteasome activity, mitochondrial function via ATP/respiration, transcriptomic aging signatures).
Neoplasia-like changes in microscopic eutelic rotifers will be detected via (a) morphological abnormalities (microscopy for tissue overgrowth, asymmetry, or cysts); (b) proliferation markers (BrdU incorporation, phospho-histone H3 immunostaining, etc.) to identify unintended cell division; and (c) transcriptomic deviation toward Hydra tumor profiles, with RNA-seq comparison to previous work [7], so that high similarity score indicates risk.
Iteration will be guided by predefined decision criteria: (a) advance to next target (e.g., from rotifer to Daphnia) if: ≥20% healthspan extension (median lifespan increase); (b) significant transcriptomic shift toward Hydra stem/progenitor states (e.g., Pearson correlation ≥0.7 with Hydra scRNA-seq clusters) [10]; (c) and no major fitness cost (fecundity decline <10%, no developmental defects).
Iterate or mitigate if: (a) no healthspan benefit, high neoplasia signal (proliferation markers >2-fold baseline), or (b) fitness costs (fecundity <80% control, locomotion decline >30%).
Additional trade-offs that can be monitored include metabolic costs (oxygen consumption, ATP levels), stress sensitivity, and developmental defects (morphological scoring at hatching).
Note that genetic information from the Hydra oligactis inducible aging phenotype [3–6] can be used in a secondary extension of evaluating the main hypothesis. This can include, e.g., genetic manipulation to “swap” aging phenotypes between Hydra vulgaris and Hydra oligactis, ascertain effects of Hydra oligactis gene expression patterns in rotifers and other organisms, as well as to explore whether the potentiality for inducible aging makes Hydra oligactis more susceptible to neoplasia. That could be follow-up work succeeding the results of the main Hydra vulgaris-based experiments outlined here.
The expected outcome of the main experimental protocols (involving Hydra vulgaris) would be decreased age-related mortality/senescence of Brachionus manjavacas, resulting in an increased lifespan. Further, organism function (e.g., locomotion, reproduction) is expected to demonstrate a prolonged “youthful” period (increased healthspan). At the cellular level, these changes would be reflected in increased post-mitotic cell maintenance as described above. At the molecular level, we expect to observe gene expression patterns consistent with earlier, more “vigorous,” stages of the rotifer lifecycle being maintained for longer periods of time. It is possible we may observe a particular emphasis on maintenance of gene expression patterns of stem cell self-renewal, coupled to continuing replenishment of progenitor cells, which may cause neoplastic overgrowth and/or developmental defects.
While not a practical objective, or an expected outcome, from a speculative standpoint the experiments could provide a definitive answer to (a) whether full suppression of senescence in Brachionus manjavacas is at all possible, and (b) if possible whether it can be achieved without radically altering the phenotype of the organism and its normal lifecycle. The answer to both questions is almost certainly “no” and this can be empirically demonstrated.
Success with this foundational model organism can be utilized to identify gene expression profiles to be used to progressively repress senescence and enhance healthspan in ever more complex organisms, starting with the genetically tractable Daphnia, another invertebrate model of aging [16, 23]. The studies could then further extend to mouse models, including the more rapidly aging MRL mouse [17–19], with the ultimate objective of repressing aging at the mammalian level. Various other mouse models of aging [19] would no doubt be useful targets once the experimental strategy reaches the mammalian level of genetic modification. We would expect similar age-repressing effects in these model organisms as with the rotifer.
Of course, the opposite strategy can also be employed; gene expression patterns reflecting aging of Brachionus manjavacas can be recapitulated into Hydra to ascertain whether age-related increased mortality/senescence is thus induced. Findings from these reverse experiments can identify more genetic targets for modification required for the main approach for suppression of senescence. This iterative bidirectional process can in theory be utilized as the experimental strategy moves up through increasing organism complexity. Epigenetic alterations of interest [24] can also be analyzed.
Considering evolutionary trade-offs
Considering evolutionary trade-offs, the model of Nelson and Masel [25] posits that multicellular aging is inevitable due to three key tenets: (1) inevitable cellular degradation over time, (2) long-term uncoupling of organismal vigor from developmental outcomes (e.g., body size), and (3) lack of selective coordination between cellular vigor and cooperation, leading to intercellular competition where “cheater' cells” that evade cooperation gain selective advantage and drive neoplasia. In Hydra, constant stem cell renewal appears to maintain positive correlation between vigor and cooperation, enabling negative senescence [26]. However, even in Hydra (Hydra oligactis and Pelmatohydra robusta), spontaneous tumors arise when this coordination is disrupted [7]. Differences in Hydra species as regards, e.g., inducible aging of Hydra oligactis [3–6] could be useful for interpreting evolutionary trade-offs involving senescence vs. neoplasia.
However, Hydra vulgaris is multicellular and seemingly lacking in senescence. One “out” to explain this phenotype in terms of intercellular competition is that a positive correlation between cellular vigor and cellular cooperation is possible if there is a continuous supply of “nondegraded” cells (i.e., those derived from stem cells) that can compete against both nonfunctional and neoplastic cells, concomitantly selecting for function and cooperation [26]. It is believed that this is an unrealistic scenario since stem cells are expected to also exhibit degradation and decreased function over time [26].
However, since Hydra vulgaris apparently can evade this fate, the importance of this organism for senescence research is underscored. This brings up the question of neoplasia as a trade-off for suppressed senescence: does Hydra exhibit sufficient positive correlation between cellular vigor and cell cooperation to evade neoplasia, or does neoplasia in this organism exist with implications for a strategy of enforced genetic suppression of senescence? As has been noted, certain Hydra species can exhibit neoplasia and develop transplantable tumors that exhibit altered gene expression, similar to vertebrate neoplasms [7]. These Hydra tumors reduce the fitness of the animals but typically do not kill nor completely prevent reproduction [7]. Further, the tumors seem to derive from differentiation-arrested female gametes, and while they do not exhibit increased proliferative potential, they do exhibit resistance to apoptosis, enhanced migration, and autonomy from the cellular microenvironment [7]. Thus, Hydra neoplasia displays some similarities to that of vertebrates. On the other hand, Hydra tumors are not universal, nor do they necessarily lead to mortality. Thus, consequences for an suppression of senescence program as outlined in Figure 1 would be the possibility of neoplasia, which would have to be carefully monitored as the anti-senescence program progresses, particularly with higher organisms (Figure 2).
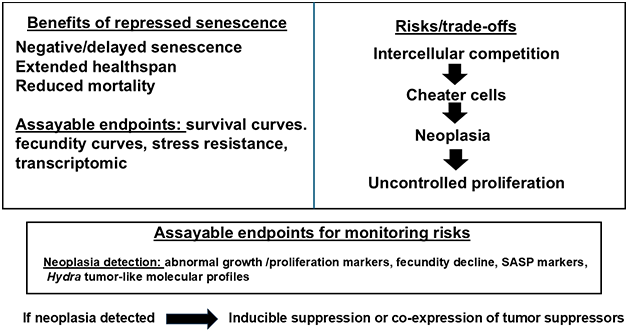
Figure 2. Schematic of trade-offs in repressed aging. Left: Benefits of continuous stem renewal enabling negative senescence (measurable via survival/fecundity curves, stress assays). Right: Risks including neoplasia from cheater cells or over proliferation. Bottom: Assayable endpoints for monitoring (e.g., proliferation markers for neoplasia, fitness metrics) and mitigation strategies. In this scheme, senescence has dual roles: early tumor suppression via cell arrest (protective) and late-life promotion of inflammation/dysfunction via SASP (detrimental). The strategy prioritizes renewal-based suppression of aging while monitoring both neoplasia risk and SASP-like inflammatory markers.
It should be noted that senescence in mammals plays a dual role: it acts as a tumor-suppressive barrier early in life by arresting damaged cells and preventing neoplastic progression, but in later life the accumulation of senescent cells promotes chronic inflammation through the SASP and contributes to age-related tissue dysfunction, immune dysregulation, and metabolic decline. The proposed Hydra-like renewal strategy aims to bypass this arrest-based senescence altogether, potentially avoiding SASP-driven late-life deterioration while still requiring careful monitoring for dysregulation-induced neoplasia.
To test the Nelson and Masel tenets in the rotifer system, we propose the following measurements: (a) cellular degradation to assess proteostasis (proteasome activity, aggregate clearance), mitochondrial function (ATP/respiration), and oxidative damage markers over time in wild-type vs. manipulated rotifers; (b) vigor-cooperation correlation in which the experimenter will monitor fitness proxies (fecundity, locomotion) alongside cellular cooperation markers (e.g., cell-cell adhesion gene expression, apoptosis rates in damaged cells); (c) intercellular competition and cheater emergence, for which the approach would be to detect neoplasia-like overgrowth via proliferation markers (BrdU incorporation, phospho-histone H3) and transcriptomic deviation toward Hydra tumor profiles, keeping in mind Hydra species differences [7].
Cheater dynamics will be inferred from disproportionate expansion of any proliferating subpopulations (if induced), or fitness decline despite molecular renewal signatures. Further, there would also be an examination of (d) negative senescence potential, in which the experimenter would quantify whether manipulated rotifers show flat or declining mortality hazard (survival curves) compared to controls, alongside reduced senescence hallmarks (e.g., lower inflammatory gene expression or SASP-like signatures). These readouts will be integrated into the iterative experimental framework (Figure 1), with iteration decisions guided by whether the manipulations shift the balance toward vigor-cooperation correlation (e.g., extended healthspan without increased neoplasia) or toward competition-driven dysfunction (e.g., tumor-like overgrowth or fitness cost). This approach allows direct testing of the model's tenets in the rotifer, a simple, eutelic metazoan.
Sustained FoxO activity or stemness promotion in a eutelic organism like the rotifer could promote aberrant proliferation rather than the desired result of benign coordinated renewal, potentially resulting in neoplasia-like changes analogous to those observed in Hydra [7]. In mammals, forced maintenance of stem cell compartments frequently elevates tumorigenesis risk, particularly when p53 function is compromised and FoxO-p53 crosstalk is disrupted [27]. Studies in model systems have provided quantitative measures of the relevant trade-offs. Thus, FoxO activation in Drosophila extends lifespan by ~20–30% but increases cancer-like phenotypes in intestinal stem cells [28]. In mice, inducible FOXO3a overexpression extends median and maximum lifespan by ~30% through enhanced DNA repair and radiation resistance [29], whereas skeletal muscle-specific FOXO1 overexpression yields no lifespan extension and is associated with atrophy and proteostatic dysregulation [30]. These mixed results underscore that potential delayed senescence and/or healthspan gains are context-dependent and often accompanied by risks. Further, the associated risks can include therapy resistance or metastasis promotion in cancer models in which high FOXO expression offsets its suppressive effects [31]. For our proposed bidirectional experiments (e.g., enforcing rotifer-like aging gene expression patterns on Hydra), we predict outcomes such as a 20–50% increase in age-related mortality based on FoxO knockdown data [1]. To mitigate the aforementioned risks, we can, e.g., propose reasonable safeguards including inducible expression systems (e.g., Tet-On for reversible activation) and co-expression of tumor suppressors (e.g., p53 orthologs). Thus, if neoplasia is identified, genetic modulation to suppress neoplasia and enhance intercellular cooperation will be attempted.
Data obtained from other invertebrate model organisms, such as C. elegans and Drosophila, can be useful, but most promising are more complex animal models that demonstrate both reduced senescence as well as resistance to neoplasia, such as the naked mole rat [32–34]. Such organisms can assist in the search for strategies to repress neoplasia that may be selected for and promoted via suppression of senescence, particularly in more complex animal models. Using naked mole rat genetics in mouse models as an approach to enhance murine resistance to aging and cancer, as has been proposed [33], and demonstrated with considerable success [34], is an example of this strategy.
Full “organismal physiological immortality,” defined as a lack of senescence and no increase in age-related mortality rates, in bilaterians is not possible due to factors like tissue complexity and evolutionary trade-offs, including resource allocation [35] and neoplasia. Thus, while Hydra achieves negligible senescence via continuous stem cell renewal, full recapitulation in complex bilaterians is biologically implausible. Instead, we can more reasonably expect delayed senescence and/or extended healthspan rather than true non-senescence. Ultimately, in complex animal models, the optimal practical outcome would manifest as relatively negligible senescence (as seen in naked mole-rats) rather than absolute non-senescence [36].
Technical challenges and limitations
While rotifers such as Brachionus manjavacas are tractable for genetic manipulation [20], the proposed iterative manipulation of Hydra-like gene patterns faces substantial biological and technical hurdles.
A primary limitation stems from the eutelic nature of rotifers: adult somatic cell number is fixed post-hatching, with minimal or no post-developmental somatic proliferation [13]. These characteristics contrast sharply with the continuously cycling stem cell compartments of Hydra and therefore restrict the potential for expansion of stem cell-like activity or progenitor cell replenishment as initially envisioned in an earlier version of the experimental plan. Forced promotion of stemness programs (e.g., via FoxO overexpression) may thus manifest primarily as enhanced resilience, proteostasis, or stress resistance in existing post-mitotic cells rather than renewed tissue turnover. Moreover, attempts to significantly alter the eutelic lifecycle, whether through ectopic activation of proliferation pathways or forced stem-like behavior, could disrupt body plan integrity, reproduction, or locomotion. That could result in developmental malformations, reduced fitness, or even lethality in extreme cases. In practice, therefore, the most realistic positive outcome of introducing Hydra-like patterns would be improved functional maintenance of the fixed somatic cell population rather than creation of a renewable stem cell pool. However, in the event that a renewable stem cell population unexpectedly develops, the expected negative consequences of neoplasia and/or developmental defects will be carefully monitored and mitigated (Figure 2).
Stable, heritable, multi-gene manipulations pose further technical challenges. Although CRISPR delivery via microinjection or electroporation is feasible [20], achieving inducible, tissue-specific, or precisely dosage-controlled expression across generations remains technically demanding. Risks include off-target effects, epigenetic silencing of transgenes, variable penetrance, and dosage toxicity from overexpression. These issues are common across genetic manipulation studies in many model organisms but are particularly problematic in a microscopic, eutelic species with limited cell numbers and a rapid life cycle.
Pleiotropic consequences are likely and may complicate desired phenotypic outcomes. In Hydra, FoxO regulates not only stemness but also antimicrobial peptide production and microbiome composition [1, 37]; analogous effects in rotifers could alter microbial interactions that are critical for health and aging, offsetting any gains in cellular maintenance. In parthenogenetic Daphnia, tissue complexity increases the likelihood of off-target effects [23]. Extending these objectives to more complex animal models could amplify these issues. In mice (including MRL strains), viral or transgenic delivery faces immune rejection, epigenetic instability, and heightened potential for neoplasia arising from sustained stemness promotion. Prior FoxO manipulations in mammals and flies demonstrate predominantly mixed outcomes: overexpression can yield no lifespan extension, accelerated aging phenotypes, increased thermal stress sensitivity, or proteostatic deregulation in a dose-dependent manner [30, 38, 39]. Some conditional mouse models show modest healthspan benefits (e.g., ~30% lifespan extension via inducible FOXO3a) [29].
Without pilot data or highly constrained initial targets, the proposed approach remains speculative. Inducible systems and careful monitoring of reproduction, behavior, neoplasia proxies, and other fitness parameters will be essential to (a) mitigate these limitations and (b) refine the strategy iteratively. Note that the iterative nature of the experimental plan is a fundamental aspect of the proposal.
It must also be noted that the hypothesis and the proposed experiments, although highly speculative, are introduced in part for their epistemological value: to provoke discussion and debate within the scientific community on these fundamental issues, perhaps stimulating more optimal or alternative approaches. In this sense, the development of the hypothesis and its testing can itself be approached in an iterative fashion by the broader research field.
Discussion
Data from successful rotifer experiments may help identify conserved longevity-regulatory modules (e.g., FoxO-mediated stemness and renewal pathways) that might inform human geroprotective strategies. Rather than unfeasible direct human genome manipulation, these insights could generate hypotheses for somatic interventions, small-molecule modulators of conserved targets, or epigenetic approaches aimed at delaying aging hallmarks. Intermediate validation in mammalian models (e.g., mice, including strains with altered healing or aging phenotypes) would be required to prioritize pathways for further development. Additional animal models, such as the naked mole rat [32–34], can serve as both targets for (a) enhanced aging suppression via Hydra-like gene expression patterns and (b) being sources of gene expression profiles to be reproduced in other organisms to facilitate delayed senescence. While translation to humans remains highly speculative at this early stage, the rotifer-Hydra model provides a proof-of-principle framework for discovering targets potentially more relevant to mammalian aging than those from other invertebrate systems.
In summary, despite the possible advantages of incorporating these other animal models in the latter stages of the suppression of senescence scheme, the fundamental approach is to utilize Hydra and rotifers as “proof of principle” to demonstrate the feasibility of suppression of senescence at the organism level. The Hydra is a demonstrated model of multicellular suppressed senescence, with defined gene expression profiles [1, 10]. Hydra also reflects possible negative consequences of repressed senescence, such as neoplasia, also with a defined neoplastic gene expression profile [7]. At the other end of the lifecycle spectrum, the rotifer Brachionus manjavacas (as one example) represents an optimal target of the suppression of senescence program, as it is a simple invertebrate, short-lived, rapidly aging, eutelic, with life stages reflecting defined gene expression programs [11]. If Brachionus manjavacas can, at least to some extent, exhibit more negligible senescence via transfer of relevant Hydra-like gene expression patterns, this would constitute the required proof-of-principle for the overall concept. It is a long leap from rotifers to geroprotective strategies for humans, but without the initial step from Hydra to rotifer, nothing else would likely be possible. Therefore, we posit that Hydra to rotifer, considered long term, is of relevance to the question of aging, senescence, and geroprotective strategies for humans.
Author Contributions
This manuscript is entirely the product of MB.
Conflicts of Interest
The author declares that he has no conflicts of interest.
Funding
The work was supported by the Geisinger College of Health Sciences.
References
- 1. Boehm AM, Khalturin K, Anton-Erxleben F, Hemmrich G, Klostermeier UC, Lopez-Quintero JA, Oberg HH, Puchert M, Rosenstiel P, Wittlieb J, Bosch TC. FoxO is a critical regulator of stem cell maintenance in immortal Hydra. Proc Natl Acad Sci USA. 2012; 109:19697–702. https://doi.org/10.1073/pnas.1209714109 [PubMed]
- 2. Martínez DE. Mortality patterns suggest lack of senescence in hydra. Exp Gerontol. 1998; 33:217–25. https://doi.org/10.1016/s0531-5565(97)00113-7 [PubMed]
- 3. Yoshida K, Fujisawa T, Hwang JS, Ikeo K, Gojobori T. Degeneration after sexual differentiation in hydra and its relevance to the evolution of aging. Gene. 2006; 385:64–70. https://doi.org/10.1016/j.gene.2006.06.031 [PubMed]
- 4. Sun S, White RR, Fischer KE, Zhang Z, Austad SN, Vijg J. Inducible aging in Hydra oligactis implicates sexual reproduction, loss of stem cells, and genome maintenance as major pathways. Geroscience. 2020; 42:1119–32. https://doi.org/10.1007/s11357-020-00214-z [PubMed]
- 5. Tomczyk S, Suknovic N, Schenkelaars Q, Wenger Y, Ekundayo K, Buzgariu W, Bauer C, Fischer K, Austad S, Galliot B. Deficient autophagy in epithelial stem cells drives aging in the freshwater cnidarian Hydra. Development. 2020; 147:dev177840. https://doi.org/10.1242/dev.177840 [PubMed]
- 6. Tökölyi J, Kumagai Y, Szilágyi RK, Nagy NA, Szakál V, Daryono RNH. Canonical Wnt/β-catenin signalling regulates inducible aging and regeneration loss in hydra. 2025;04.01.646560. https://doi.org/10.1101/2025.04.01.646560
- 7. Domazet-Lošo T, Klimovich A, Anokhin B, Anton-Erxleben F, Hamm MJ, Lange C, Bosch TC. Naturally occurring tumours in the basal metazoan Hydra. Nat Commun. 2014; 5:4222. https://doi.org/10.1038/ncomms5222 [PubMed]
- 8. Martins R, Lithgow GJ, Link W. Long live FOXO: unraveling the role of FOXO proteins in aging and longevity. Aging Cell. 2016; 15:196–207. https://doi.org/10.1111/acel.12427 [PubMed]
- 9. Zeng Y, Cheng L, Chen H, Cao H, Hauser ER, Liu Y, Xiao Z, Tan Q, Tian XL, Vaupel JW. Effects of FOXO genotypes on longevity: a biodemographic analysis. J Gerontol A Biol Sci Med Sci. 2010; 65:1285–99. https://doi.org/10.1093/gerona/glq156 [PubMed]
- 10. Siebert S, Farrell JA, Cazet JF, Abeykoon Y, Primack AS, Schnitzler CE, Juliano CE. Stem cell differentiation trajectories in Hydra resolved at single-cell resolution. Science. 2019; 365:eaav9314. https://doi.org/10.1126/science.aav9314 [PubMed]
- 11. Gribble KE, Mark Welch DB. Genome-wide transcriptomics of aging in the rotifer Brachionus manjavacas, an emerging model system. BMC Genomics. 2017; 18:217. https://doi.org/10.1186/s12864-017-3540-x [PubMed]
- 12. Gribble KE, Welch DB. Life-span extension by caloric restriction is determined by type and level of food reduction and by reproductive mode in Brachionus manjavacas (Rotifera). J Gerontol A Biol Sci Med Sci. 2013; 68:349–58. https://doi.org/10.1093/gerona/gls170 [PubMed]
- 13. Snell TW. Rotifers as models for the biology of aging. Int Rev Hydrobiol. 2014; 99:84–95. https://doi.org/10.1002/iroh.201301707 [PubMed]
- 14. Snell TW, Johnston RK, Matthews AB, Zhou H, Gao M, Skolnick J. Repurposed FDA-approved drugs targeting genes influencing aging can extend lifespan and healthspan in rotifers. Biogerontology. 2018; 19:145–57. https://doi.org/10.1007/s10522-018-9745-9 [PubMed]
- 15. Bock MJ, Jarvis GC, Corey EL, Stone EE, Gribble KE. Maternal age alters offspring lifespan, fitness, and lifespan extension under caloric restriction. Sci Rep. 2019; 9:3138. https://doi.org/10.1038/s41598-019-40011-z [PubMed]
- 16. Iampolskiĭ LI, Galimov Ia. Evolutionary genetics of aging in Daphnia. Zh Obshch Biol. 2005; 66:416–24. [PubMed]
- 17. Heydemann A. The super super-healing MRL mouse strain. Front Biol (Beijing). 2012; 7:522–38. https://doi.org/10.1007/s11515-012-1192-4 [PubMed]
- 18. Heber-Katz E, Leferovich J, Bedelbaeva K, Gourevitch D, Clark L. Conjecture: Can continuous regeneration lead to immortality? Studies in the MRL mouse. Rejuvenation Res. 2006; 9:3–9. https://doi.org/10.1089/rej.2006.9.3 [PubMed]
- 19. Ackert-Bicknell CL, Anderson LC, Sheehan S, Hill WG, Chang B, Churchill GA, Chesler EJ, Korstanje R, Peters LL. Aging Research Using Mouse Models. Curr Protoc Mouse Biol. 2015; 5:95–133. https://doi.org/10.1002/9780470942390.mo140195 [PubMed]
- 20. Feng H, Bavister G, Gribble KE, Mark Welch DB. Highly efficient CRISPR-mediated gene editing in a rotifer. PLoS Biol. 2023; 21:e3001888. https://doi.org/10.1371/journal.pbio.3001888 [PubMed]
- 21. Schaible R, Sussman M. FOXO in aging: did evolutionary diversification of FOXO function distract it from prolonging life? Bioessays. 2013; 35:1101–10. https://doi.org/10.1002/bies.201300078 [PubMed]
- 22. Schaum N, Lehallier B, Hahn O, Pálovics R, Hosseinzadeh S, Lee SE, Sit R, Lee DP, Losada PM, Zardeneta ME, Fehlmann T, Webber JT, McGeever A, et al., and Tabula Muris Consortium. Ageing hallmarks exhibit organ-specific temporal signatures. Nature. 2020; 583:596–602. https://doi.org/10.1038/s41586-020-2499-y [PubMed]
- 23. Kato Y, Matsuura T, Watanabe H. Genomic integration and germline transmission of plasmid injected into crustacean Daphnia magna eggs. PLoS One. 2012; 7:e45318. https://doi.org/10.1371/journal.pone.0045318 [PubMed]
- 24. Pillai A, Gungi A, Reddy PC, Galande S. Epigenetic Regulation in Hydra: Conserved and Divergent Roles. Front Cell Dev Biol. 2021; 9:663208. https://doi.org/10.3389/fcell.2021.663208 [PubMed]
- 25. Nelson P, Masel J. Intercellular competition and the inevitability of multicellular aging. Proc Natl Acad Sci USA. 2017; 114:12982–7. https://doi.org/10.1073/pnas.1618854114 [PubMed]
- 26. Vaupel JW, Baudisch A, Dölling M, Roach DA, Gampe J. The case for negative senescence. Theor Popul Biol. 2004; 65:339–51. https://doi.org/10.1016/j.tpb.2003.12.003 [PubMed]
- 27. Eijkelenboom A, Burgering BM. FOXOs: signalling integrators for homeostasis maintenance. Nat Rev Mol Cell Biol. 2013; 14:83–97. https://doi.org/10.1038/nrm3507 [PubMed]
- 28. Biteau B, Karpac J, Supoyo S, Degennaro M, Lehmann R, Jasper H. Lifespan extension by preserving proliferative homeostasis in Drosophila. PLoS Genet. 2010; 6:e1001159. https://doi.org/10.1371/journal.pgen.1001159 [PubMed]
- 29. Inci G, Warkad MS, Kang BG, Kim SO, Lee NK, Heo JI, Park SH, Kim JH, Kang HJ, Kim CH, Kim BH, Park SJ, Suh JG, et al. FOXO3a upregulates DNA repair activities by transcriptional activation of target genes and provides the resistance to gamma radiation and the extension of lifespan in mouse. Biogerontology. 2025; 26:196. https://doi.org/10.1007/s10522-025-10341-9 [PubMed]
- 30. Chiba T, Kamei Y, Shimizu T, Shirasawa T, Katsumata A, Shiraishi L, Sugita S, Ogawa Y, Miura S, Ezaki O. Overexpression of FOXO1 in skeletal muscle does not alter longevity in mice. Mech Ageing Dev. 2009; 130:420–8. https://doi.org/10.1016/j.mad.2009.04.004 [PubMed]
- 31. Bullock M. FOXO factors and breast cancer: outfoxing endocrine resistance. Endocr Relat Cancer. 2016; 23:R113–30. https://doi.org/10.1530/ERC-15-0461 [PubMed]
- 32. Finch CE. Update on slow aging and negligible senescence—a mini-review. Gerontology. 2009; 55:307–13. https://doi.org/10.1159/000215589 [PubMed]
- 33. Oka K, Yamakawa M, Kawamura Y, Kutsukake N, Miura K. The Naked Mole-Rat as a Model for Healthy Aging. Annu Rev Anim Biosci. 2023; 11:207–26. https://doi.org/10.1146/annurev-animal-050322-074744 [PubMed]
- 34. Zhang Z, Tian X, Lu JY, Boit K, Ablaeva J, Zakusilo FT, Emmrich S, Firsanov D, Rydkina E, Biashad SA, Lu Q, Tyshkovskiy A, Gladyshev VN, et al. Increased hyaluronan by naked mole-rat Has2 improves healthspan in mice. Nature. 2023; 621:196–205. https://doi.org/10.1038/s41586-023-06463-0 [PubMed]
- 35. Hamilton WD. The moulding of senescence by natural selection. J Theor Biol. 1966; 12:12–45. https://doi.org/10.1016/0022-5193(66)90184-6 [PubMed]
- 36. Buffenstein R. Negligible senescence in the longest living rodent, the naked mole-rat: insights from a successfully aging species. J Comp Physiol B. 2008; 178:439–45. https://doi.org/10.1007/s00360-007-0237-5 [PubMed]
- 37. He J, Bosch TC. Hydra's Lasting Partnership with Microbes: The Key for Escaping Senescence? Microorganisms. 2022; 10:774. https://doi.org/10.3390/microorganisms10040774 [PubMed]
- 38. Paik JH, Kollipara R, Chu G, Ji H, Xiao Y, Ding Z, Miao L, Tothova Z, Horner JW, Carrasco DR, Jiang S, Gilliland DG, Chin L, et al. FoxOs are lineage-restricted redundant tumor suppressors and regulate endothelial cell homeostasis. Cell. 2007; 128:309–23. https://doi.org/10.1016/j.cell.2006.12.029 [PubMed]
- 39. Manola MS, Gumeni S, Trougakos IP. Differential Dose- and Tissue-Dependent Effects of foxo on Aging, Metabolic and Proteostatic Pathways. Cells. 2021; 10:3577. https://doi.org/10.3390/cells10123577 [PubMed]



