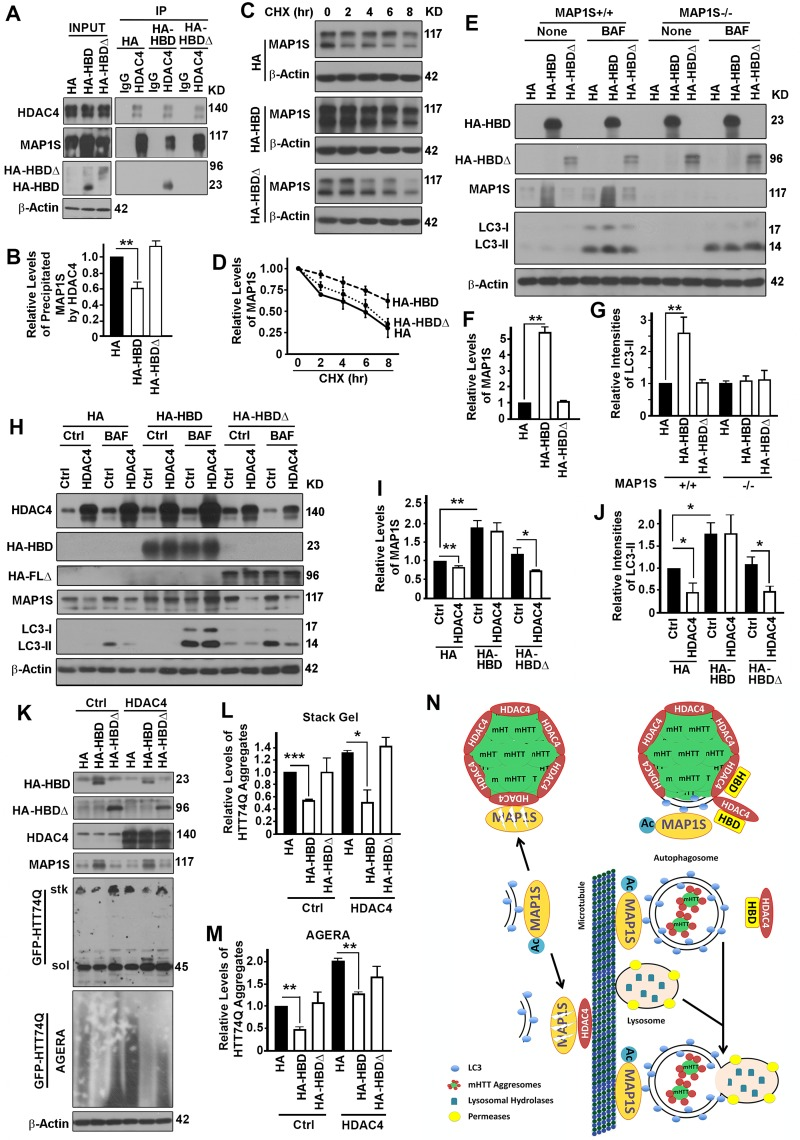
Figure 7.The interaction between HDAC4 and MAP1S is required for HDAC4 to exert suppressive roles on MAP1S-mediated autophagy turnover of mHTT aggregates(A, B) Overexpression of HBD but not HA-HBDΔ reduces HDAC4-bound endogenous full length MAP1S. Equal amounts of lysates collected from HeLa cells expressing control HA, HBD and HA-HBDΔ were subjected to immunoprecipitation with HDAC4-specific antibody. Representative immunoblots (A) and quantification of the precipitated MAP1S (B) were shown. (C, D) Overexpression of HBD but not HA-HBDΔ enhances stability of endogenous MAP1S protein in HeLa cells. Lysates from HeLa cells transiently transfected with HA vector and HA-HBD and HA-HBDΔ were collected at different times after cycloheximide treatment. Representative immunoblots (C) and quantification (D) are shown. (E-G) Overexpression of HBD but not HA-HBDΔ enhances autophagy flux in the presence of MAP1S. Lysates were collected from control (MAP1S+/+) or MAP1S knockout HeLa cells (MAP1S−/−) transiently transfected with HA vector and HA-HBD and HA-HBDΔ in the absence (Ctrl) or presence of BAF. Representative immunoblots (E) and quantification of the relative levels of MAP1S in control HeLa cells in the absence of BAF (F) or relative levels of LC3-II in the presence of BAF (G) are shown. (H-J) Overexpression of HBD but not HA-HBDΔ prevents the HDAC4-induced MAP1S destabilization and autophagy flux suppression. Lysates were collected from HeLa cells transiently co-transfected with HDAC4 and HA vector, HA-HBD or HA-HBDΔ in the absence (Ctrl) or presence of BAF. Representative immunoblots (H) and quantification of the relative levels of MAP1S in the absence of BAF (I) or LC3-II in the presence of BAF (J) are shown. (K-M) Overexpression of HBD but not HA-HBDΔ reduces levels of mHTT aggregates. N2a cells transiently expressing GFP-HTT74Q were simultaneously transfected with two additional plasmids with one to express control, HBD or HA-HBDΔ, and another to express control or HDAC4, respectively. Equal amounts of cell lysates are subjected to immunoblot. Representative blots of GFP-74Q resolved by stack gel or AGERA (K) and their quantification in stack gel (L) or AGERA (M) are shown. (N) A diagram showing the potential mechanism by which HDAC4 regulates MAP1S-mediated autophagy turnover of mHTT aggregates. Under normal condition, isolation membrane-associated acetylated-MAP1S is deacetylated and destabilized by microtubule or aggregate-associated HDAC4. In the presence of excessive HBD, HBD competes with acetylated MAP1S for interaction with HDAC4, which leads to the exposure of mHTT aggregates to be packaged by the MAP1S-associated isolation membrane. The resulted mHTT-containing autophagosomes are connected to microtubules by acetylated MAP1S and fuse with lysosome to become autolysosomes in which mHTT aggregates are degraded by lysosomal enzymes.