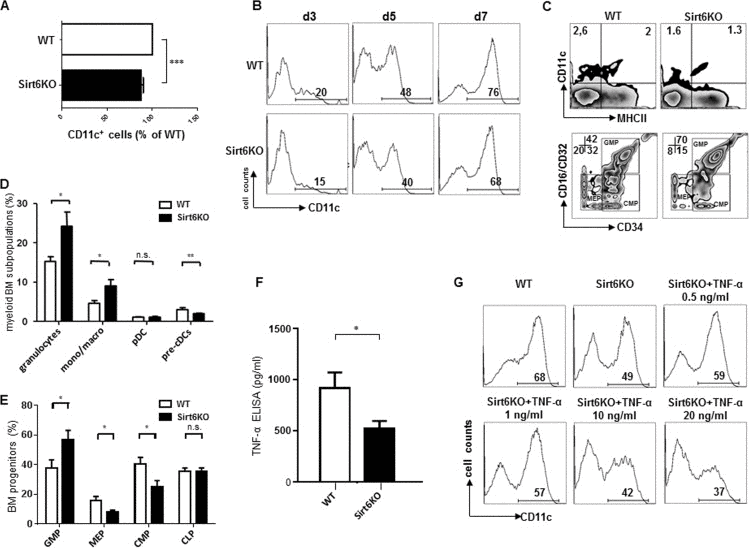
Figure 1.Sirt6 regulates the generation of cDCs in vivo and in vitro(A, B) WT or Sirt6KO BM cells were cultured with GM-CSF and their ability to generate CD11c+ cells was analyzed at day 3, 5, 7 by flow cytometry. (A) The percentage of CD11c+ cells within cultures of Sirt6KO BMs (day 7) was normalized to that of WT CD11c+ cells. Results are presented are means ± SEM of 6 separate experiments, n=13 for each genotype; ***: p<0.001. (B) one representative experiment out of two is presented. (C-E) BM cells from WT and Sirt6KO mice were analyzed by flow cytometry for the frequency of cDC precursors (pre-cDCs, CD11c+MHCII−), pDCs, monocyte/macrophage subsets, mature granulocytes and of BM progenitors of different lineages. (C) one representative experiment out of six is presented; (D, E) results are presented are means ± SEM of fifteen and six separate experiments, respectively, n=6-15 for each genotype; *: p<0.05; **: p<0.01; n.s.: not significant. (F) TNF-α concentration in the supernatants of WT and Sirt6KO BMDCs (harvested at day 6) were determined by ELISA. Results are means ± SEM of three separate experiments, n=10 for each genotype; *: p<0.05. (G) WT and Sirt6KO BM cells were cultured with GM-SCF with or without addition of the indicated concentrations of TNF-α. CD11c+ cells were quantified at day 6 by flow cytometry. One representative experiment out of six is presented, n=6-9 for each genotype.