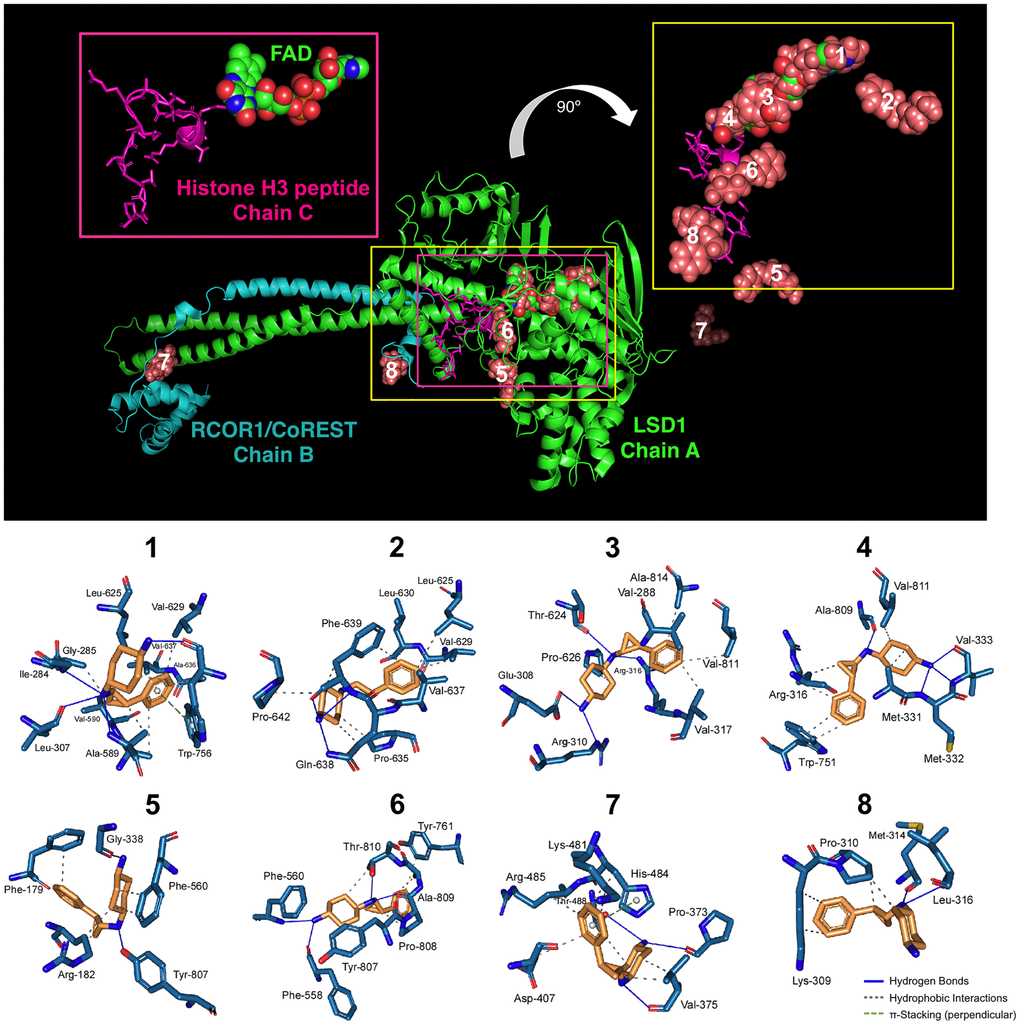
Figure 1.Binding mode of iadademstat to LSD1.Top. The backbone of LSD1 (chain A)/REST corepressor 1 (chain B)/histone H3 peptide (chain C) heterotrimeric complex is shown. For each cluster of the docked iadademstat (salmon color), only the molecule (spheres) with better binding energy is shown. The molecular docking was performed using the A and B chains in the absence of FAD and histone H3 peptide; however, the clusters of docked iadademstat are shown superimposed on the position that would occupy both the FAD and the histone H3 peptide. The cluster number is also indicated. The insert on the left shows the peptide histone H3 (chain C, backbone as cartoon and side chains as sticks) and the FAD (represented as spheres and with the green carbons). The insert on the right shows only the best pose of iadademstat docked in each cluster and the situation of the histone H3 peptide. The clusters #1, #3 and #4 of iadademstat would occupy the same position of the FAD and are shown superimposed. Bottom. The detailed map of the molecular interactions of iadademstat in each cluster is detailed (see also Table 1). Each inset shows the detailed interactions of each compound docked to the LSD1 heterodimer, indicating the participating amino acids involved in the interaction and the type of interaction (hydrogen bonds, hydrophilic interactions, salt bridges, Π-stacking, etc).