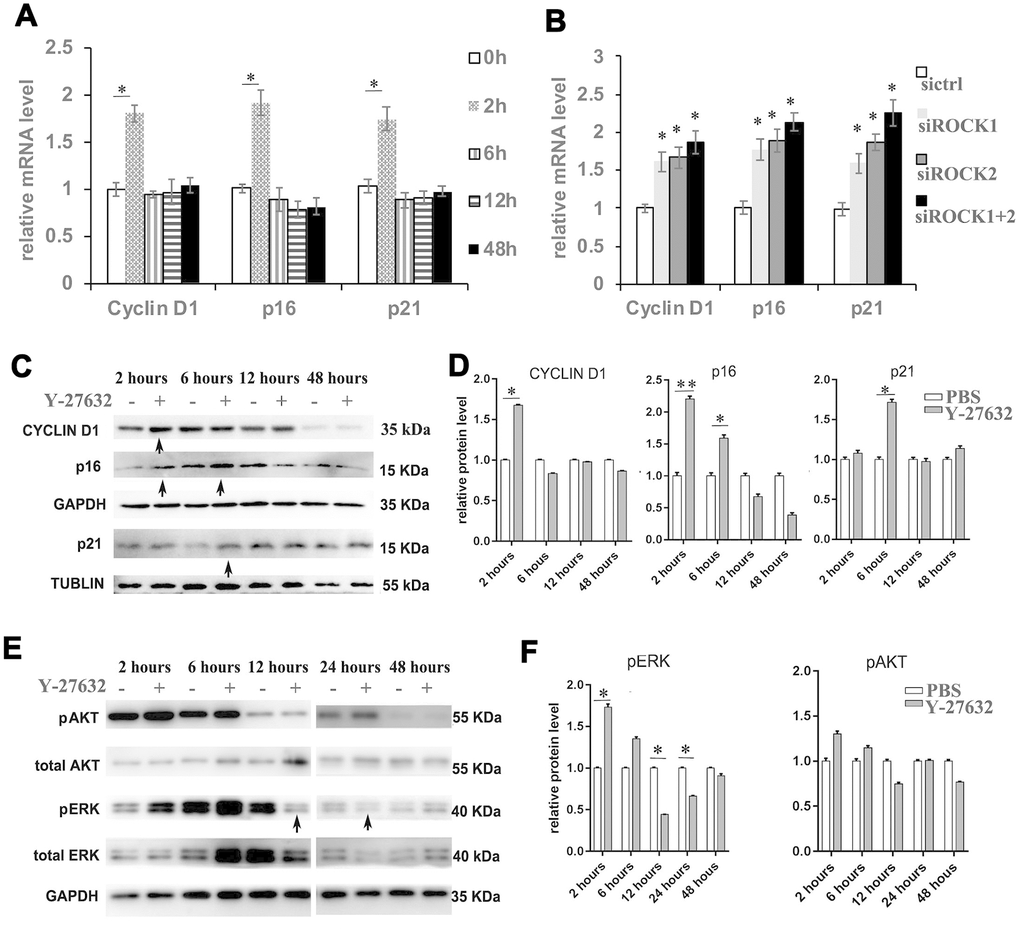
Figure 4.Inhibition of ROCK signaling increases the expression of proliferation and senescence markers. (A) HDFs were cultured in growth medium in the presence of PBS or 10 μM Y-27632, then were collected at the indicated times for RT-qPCR analysis of cyclin D1, p16 and p21. The expression level of those 3 genes was adjusted with the house-keeping gene 36B4, the mRNA level in the Y-27632 treated HDFs relative to the control HDFs (expression level as 1) is shown. (B) HDFs were transfected with siRNAs targeting ROCK1 or and ROCK2, the control HDFs were transfected with a scrambled siRNA (sictrl). HDFs were collected at 48 h after the transfection and subjected to RT-qPCR analysis to detect mRNA expression levels of cyclin D1, p16 and p21. The relative mRNA level of 3 genes in ROCK knockdown HDFs is shown. (C, D) HDFs were treated with PBS (-) or 10 μM Y-27632 (+), then were collected at the indicated times and cellular lysates were analyzed for protein expression levels of cyclin D1, p16 and p21 by western blot. GAPDH or β-tubulin protein levels were used as loading controls. Arrows inidcated increased expression of genes in the Y-27632 treated cells (C). The fold change of those proteins in the Y-27632 treated HDFs relative to the control HDFs (expression level as 1) is shown on the right (D). (E, F) Western blot analysis of pERK and pAKT in HDFs at the indicated times; GAPDH protein levels were detected as a loading control. The fold change of these 3 proteins in the Y-27632 treated HDFs relative to the control HDFs (expression level as 1) is shown on the right (F). Densitometry measurements for pERK and pAKT were normalized to the amounts of total ERK and AKT, respectively. Arrows inidcated decreased expression of phorylation form of ERK in the Y-27632 treated cells (E). All experiments were repeated at least 3 times, *P<0.05, **P<0.01 when two groups were compared as indicated, or were compared to the corresponding controls.