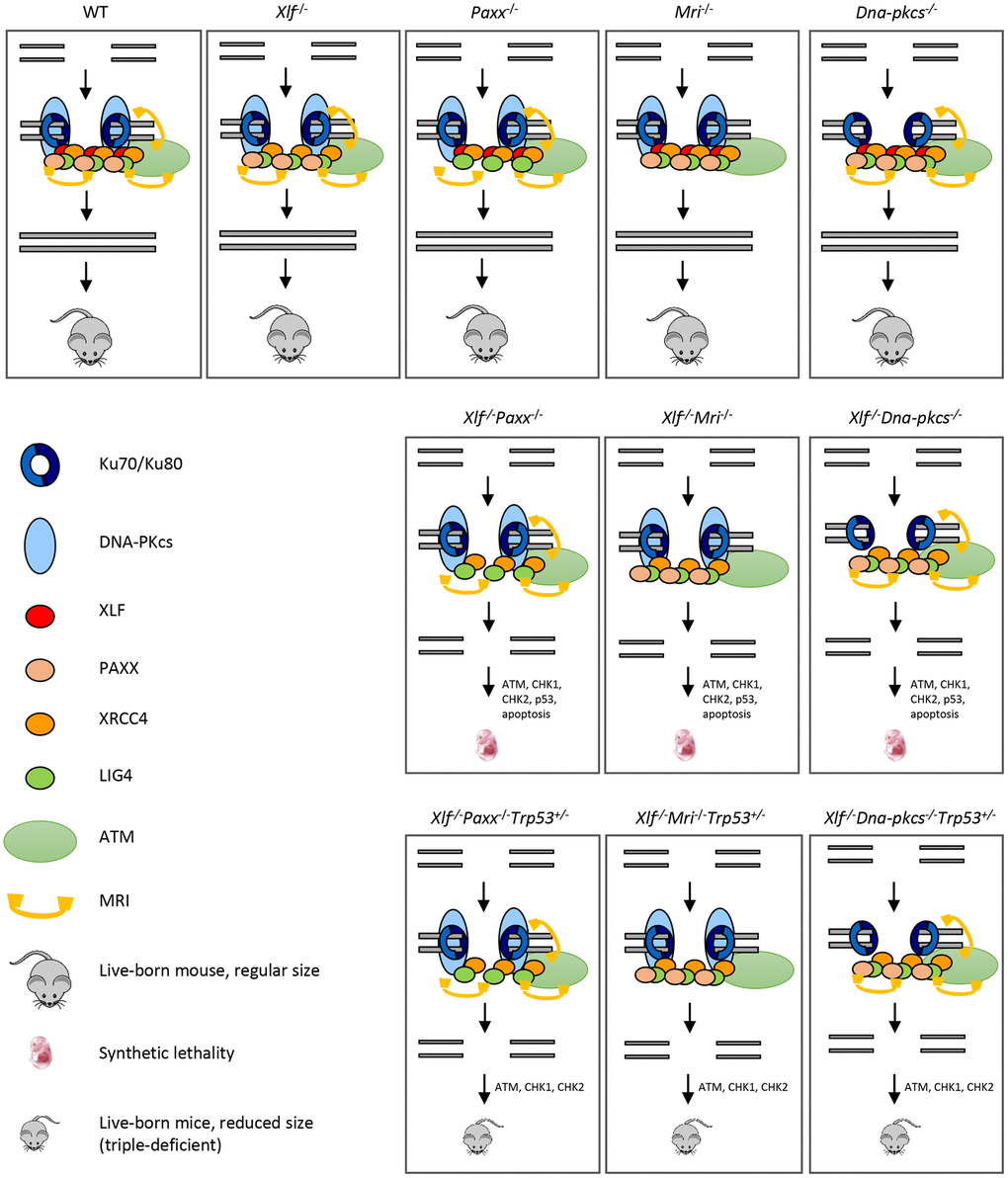
Figure 5.Mutations in NHEJ genes result in distinct phenotypes. Suggested models. Inactivation of Paxx or Mri results in live-born mice with nearly no DNA repair defects. Inactivation of Xlf or Dna-pkcs results in live-born mice with increased levels of genomic instability due to reduced NHEJ activity. Combined inactivation of Xlf/Paxx, Xlf/Mri or Xlf/Dna-pkcs leads to embryonic lethality in mice that correlate with high levels of genomic instability and nearly no NHEJ. Accumulated DSBs activate the ATM-dependent DNA damage response (DDR) pathway; ATM phosphorylates CHK checkpoint proteins that further trigger cell cycle arrest and apoptosis. Alternative end-joining is blocked by presence of Ku70/Ku80. Inactivation of one or two alleles of Trp53 rescues embryonic lethality of Xlf/Paxx, Xlf/Mri and Xlf/Dna-pkcs mice. While in these mice the levels of DSBs are increased and ATM-dependent DDR response is activated, lack of p53 prevents massive apoptosis and thus results in alive mice. Sizes of the triple-deficient mice are reduced, as one option, due to DNA damage-dependent cell cycle arrest in multiple cells of the body. The embryonic lethality in mice lacking Xlf/Paxx and Xlf/Mri is likely to be rescued by inactivation of Ku70 or Ku80.