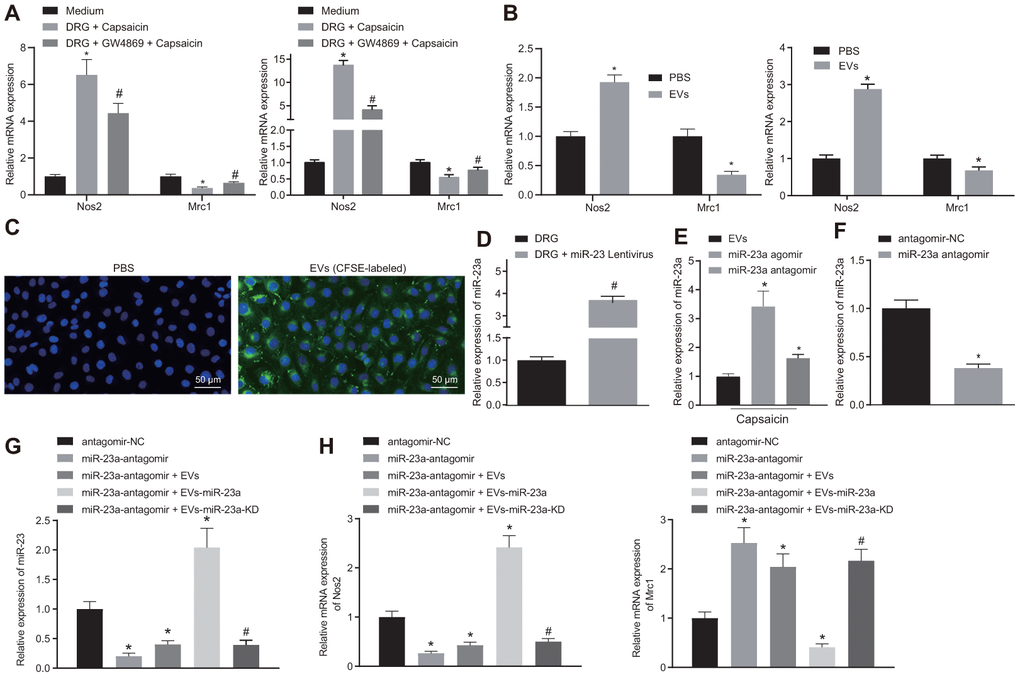
Figure 3.DRG sensory neurons transferred miR-23a to macrophages via EVs to enhance M1 polarization in vitro. (A) mRNA expression of Nos2 (M1) and Mrc1 (M2) in macrophages co-cultured with DRG neurons and treated with EV release inhibitor GW4869 (RT-qPCR, normalized to U6); (B) mRNA expression of Nos2 (M1) and Mrc1 (M2) in macrophages treated with DRG-secreted EVs induced by capsaicin determined using RT-qPCR, normalized to β-actin; (C) DRG neuron-derived EVs in macrophages observed using immunofluorescence staining (Scale bar = 50 μm); (D) miR-23a expression in DRG neurons treated with lentivirus-overexpressing miR-23a determined using RT-qPCR, normalized to U6; (E) miR-23a expression in DRG neurons treated with lentivirus-overexpressing miR-23a following capsaicin treatment determined using RT-qPCR, normalized to U6; (F) miR-23a expression in macrophages determined using RT-qPCR, normalized to U6; (G) miR-23a expression in macrophages with miR-23a knockdown and then treated with EVs determined using RT-qPCR, normalized to U6; (H) mRNA expression of Nos2 (M1) and Mrc1 (M2) in macrophages with miR-23a knockdown and then treated with EVs determined using RT-qPCR, normalized to β-actin. Values obtained from three independent experiments are expressed as mean ± SD and analyzed by unpaired t-test between two groups and by one-way ANOVA, followed by Bonferroni’s multiple comparison test among multiple groups. * p < 0.05 vs. apical chamber in transwell added with F-12 medium, DRG neurons, or DRG neurons treated with antagomir-NC plasmids; # p < 0.05 vs. DRG neurons, or DRG neurons treated with antagomir-NC + EVs-miR-23a. Cell experiment was independently repeated for three times.