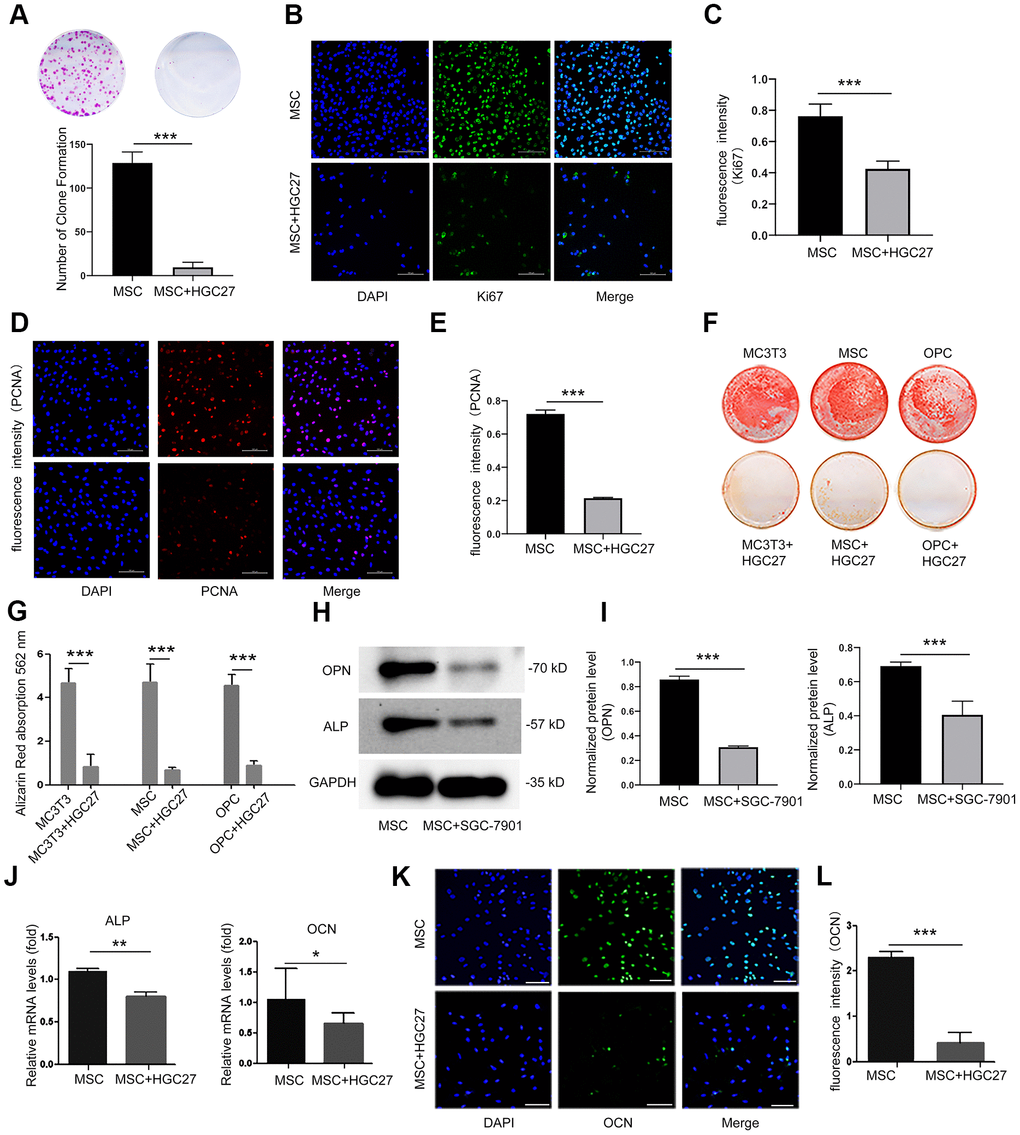
Figure 3.Co-culturing of osteoblasts with HGC27 or SGC-7901 cells in vitro confirms that gastric cancer cells inhibit the proliferation and differentiation of osteoblasts. (A) Colony formation assay showing that the proliferation ability of MSC cells was impaired upon co-culture with HGC27 cells. (B) Immunofluorescence staining of MSCs at day 3 of OS medium induction with or without co-cultured HGC27. Shown is Ki67 expression (green). Nuclei were stained with DAPI (blue). Scale bar, 100 μm. (C) Quantitative analysis of the fluorescence intensity in (B). (D) Immunofluorescence staining of PCNA (red) in MSCs at day 3 of OS induction with or without co-cultured HGC27 and Nuclei were stained with DAPI. (blue) states. Scale bar, 100 μm. (E) Quantitative analysis of the fluorescence intensity in (D). (F) Alizarin Red staining analysis of MC3T3, MSC, and OPC cells at days 16 of OS induction with or without HGC27 cells co-cultured. (G) Quantitative mineralization level based on (F). (H) Western blot analysis of OPN and ALP expression in MSCs with or without co-cultured SGC-7901. (I) Quantitative analysis of OPN and ALP protein levels from the immunoblots in (H). OPN and ALP protein levels were normalized to GAPDH (n=3). (J) qRT-PCR results showing ALP and OCN genes transcription levels in MSCs at day 3 of OS induction with or without co-culturing of HGC27. Expression levels of ALP and OCN were normalized to GAPDH expression. (K) Immunofluorescence staining of MSCs at day 3 of OS induction with or without co-cultured HGC27 to test OCN (green). Nuclei were stained with DAPI (blue) states. Scale bar, 100 μm. (L) Quantitative analysis of the fluorescence intensity in (K). Data are shown as mean±SEM. Statistical differences were obtained using Student's t-test, *, p<0.05, **, p<0.01, ***, p<0.001. n=3 per-group. OPC, oligodendrocyte progenitor cells; OPN, osteopontin; ALP, alkaline phosphatase; MSC, mesenchymal stem cells.