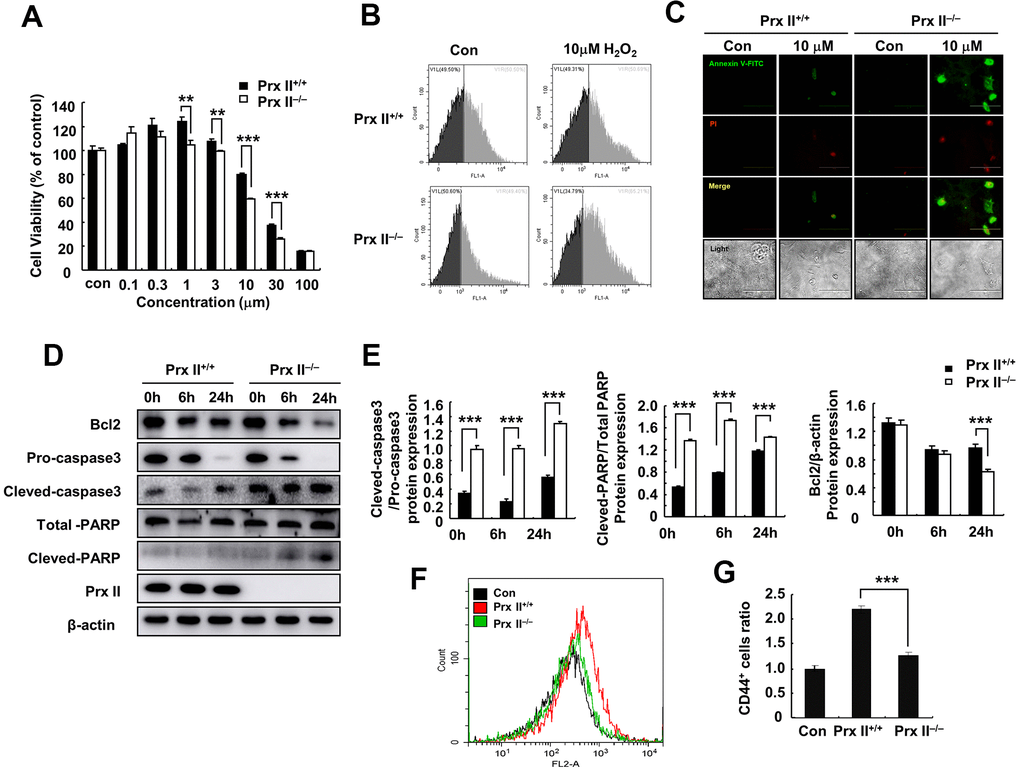
Figure 4.Deletion of Prx II promoted DMSC apoptosis under H2O2-induced oxidative stress. (A) Cell viabilities of Prx II+/+ DMSCs and Prx II−/− DMSCs after treatment with increasing concentrations of H2O2. **p < 0.01, ***p < 0.001, when compared with the control group. (B) Cell death was detected by flow cytometry after treatment for 24 h with 10 μM H2O2. (C) Annexin V and PI staining were performed to visualize apoptosis after treatment for 24 h with 10 μM H2O2. (D, E) Western blotting of Prx II+/+ DMSC and Prx II−/− DMSC extracts, and data quantification, in order to investigate the effect of 10 μM H2O2 on the expression of Prx II and apoptosis-related proteins, such as Bcl2, pro-caspase 3, and cleaved-caspase 3, total PARP, and cleaved PARP after 6 and 24 h. (F, G) Flow cytometry was used to detect the number of CD44-positive cells in the wound site after treatment with Prx II+/+ DMSCs and Prx II−/− DMSCs treatment, and to quantify the data.