Introduction
Cardiovascular disease (CVD), principally due to atherosclerosis, remains the largest cause of death and therefore influences longevity in the US [1,2]. This also applies to other middle/high-income countries. Two large meta-analyses have concluded that short leukocyte telomere length (LTL) is associated with CVD [3,4], while other studies [5–8], including a meta-analysis [9], have shown that short LTL predicts diminished longevity. Thus, the association between short LTL and diminished longevity might relate in part to the association of short LTL with CVD. Moreover, genetic analyses have revealed that alleles associated with short LTL are also associated with CVD [10–12], largely excluding the possibility of reverse causality, i.e., CVD causing LTL shortening. Coupled with observations that LTL, which reflects telomere length in somatic tissues [13], is highly heritable [14,15] and is largely determined at birth [16], these findings suggest that telomere length might play an active role in CVD and longevity. Such a conclusion is relevant to the debate about the existence of a natural lifespan limit for humans [17–21]. Here we show that in some individuals LTL becomes critically short at an age younger than that of the current life expectancy. Moreover, we show that with predicted upward trajectories of life expectancy, the proportion of these individuals will only increase in the general population.
Results
We examined the potential role of telomere length in human longevity in two settings: contemporary life expectancy, and life expectancy of 100 years (LE-100) i.e., assuming survival until the age of 100 years [17]. To this end, we first defined the LTL threshold below which the probability of survival substantially declines. We refer to this threshold as the ‘telomeric brink’ (TB).
Figure 1 is a composite that illustrates the relation between LTL, measured in the same laboratory, and age for whites of European ancestry residing in Denmark (n=1,727), France (n=185), Italy (n=548), the UK (n=3,514), the USA (n=5,726), and Jews in Israel (n=620). Characteristics of these subjects are displayed in Table 1S. The scatter plots (Figure 1, top panels) show that LTL progressively becomes shorter with age and that it varies widely between individuals of the same age.
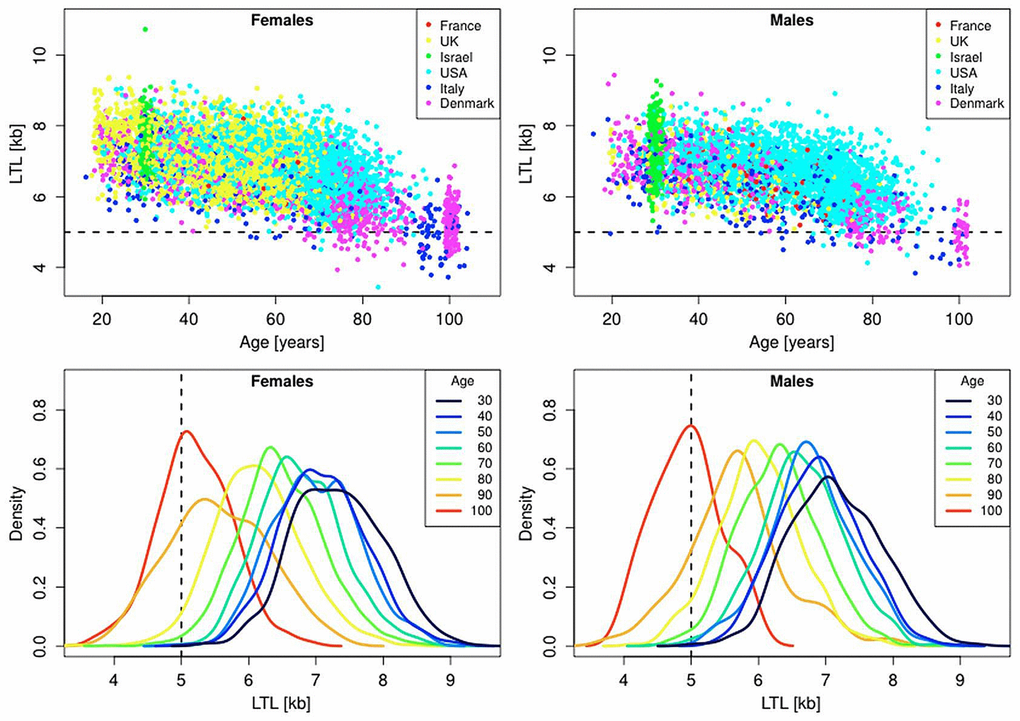
Figure 1. Scatter plots and density plots of LTL as a function of age for males and females residing in different countries. Measurements of LTL were performed in the same laboratory on DNA donated by participants in different studies in different countries (Supplemental Table 1). The horizontal dashed lines in the top panels and vertical dashed lines in the bottom panels indicate LTL values of 5 kb. The bottom plots are smoothed histograms obtained by kernel density estimation.
Table 1. Probability of reaching the LTL brink of 5.0 kb at ages 35, 50, 65 and 80 years for period life table mortality and for a life expectancy of 100 years by quintiles of LTL ranking, assuming that yearly LTL attrition (in bp) is independent and gamma distributed with shape parameter 0.4 and scale parameter 75.
| Age (yrs) | 35 | 50 | 65 | 80 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Life Expectancy (yrs) | Period | LE-100 | Period | LE-100 | Period | LE-100 | Period | LE-100 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All (%) | 14 | 39 | 8 | 27 | 6 | 20 | 10 | 24 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Prob Q1 (%) | 50 | 93 | 34 | 83 | 25 | 73 | 45 | 88 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Prob Q2 (%) | 16 | 62 | 6 | 37 | 3 | 21 | 4 | 28 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Prob Q3 (%) | 5 | 30 | 1 | 12 | 0 | 5 | 0 | 5 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Prob Q4 (%) | 1 | 10 | 0 | 2 | 0 | 1 | 0 | 1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Prob Q5 (%) | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LTL= leukocyte telomere length; Period = mortality based on period life tables; LE-100 = expectancy of 100 years for all; Prob = probability of reaching the telomeric brink; Q1 = 0-19% of the LTL distribution, Q2 = 20-39% of the LTL distribution, Q3 = 40-59% of the LTL distribution, Q4 = 60-79% of the LTL distribution, Q5 = 80-99% of the LTL distribution. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
To establish a reference for a critically short LTL that engenders a considerable risk of imminent death, i.e., the TB, we measured LTL (in the same laboratory that generated the LTL data displayed in Figure 1) in individuals suffering from dyskeratosis congenita (DC) and their unaffected relatives. DC, the outcome of rare germline mutations, is expressed in extremely short telomere length, which is the main cause of the patients’ premature demise [22]. In patients with DC the mean LTL was 4.99 kb, while in relatives it was 6.49 kb (p<0.0001) (Table 2S).
Although DC and related telomere disorders [23] unfold under different circumstances from those experienced by the individuals with short telomeres in late life, for the following reason LTL of 5 kb is a reasonable cutoff point to define a critically short LTL: Only 0.78%, of live subjects younger than 90 years displayed LTL ≤ 5 kb (Figure 1 top panel). In the oldest old, i.e., between the ages of 95-105 years, where mortality rate is very high, 37% (95% CI: 30-44%) of females showed an LTL ≤ 5 kb, compared with 58% (95% CI: 43-72%) of males (p=0.009 by Fisher's exact test). These cross-sectional data provide no direct information about selective survival with respect to LTL. A variance-ratio test suggests, however, that this might be the case (Figure 1, bottom panels), as, for instance, individuals aged 100 (95-105) years displayed less LTL variance than those aged 50 (45-55) years (variance ratios of 1.60 for males, p = 0.048; variance ratio of 1.36 for females, p = 0.011).
Figure 1 does not offer information that foretells if a person whose LTL is, for instance, 6.4 kb at the age of 35 years could reach his/her life expectancy before crossing the TB. However, with information from life tables [24], combined with the known rate of telomere shortening with age [25], we estimated the proportion of individuals reaching the TB during their predicted life expectancy. We also calculated the proportion of individuals reaching the TB for LE-100. This estimated proportion indicates to what extent living to 100 years may be constrained by LTL.
We computed the proportion of our composite study sample that would reach the TB based on the baseline LTL for four age groups, 35 (30-40), 50 (45-55), 65 (60-70) and 80 (75-85) years, and for different LTL attrition rates (Materials and Methods). Our initial computations were strictly based on the empirical data displayed in Figure 1 and the assumption of a constant rate of age-dependent LTL attrition for a given individual. For individuals who had already crossed the TB at baseline, we let the TB age be equal to their age at blood collection. We then found the individual’s probability of reaching the TB through reference to the corresponding period life table [24], based on the nationality, sex and age of the individual and the date of blood collection. By taking the mean of the individual probabilities of reaching an LTL= 5 kb, we determined the overall probability of reaching the TB for a given LTL attrition rate. We used the same approach to quantify the probability of reaching the TB before the LE-100, except that instead of using period life table mortality, we used a lifespan of exactly 100 years. Thus, an individual is judged to have reached the TB age if his/her LTL becomes shorter than 5 kb before the age of 100 years.
Younger subjects (ages 35 and 50 years) in the samples displayed in Figure 1, whose LTL attrition is > 30 bp/year, already run some risk of reaching the TB based on period mortality (Figure 2). This risk escalates for LE-100, such that even individuals whose LTL attrition rates are as low as 20 bp/year could be at risk. Given that a longer life expectancy entails a greater chance of reaching the TB, the increased probability of reaching the TB based on LE-100 is expected, but what is relevant here is that this effect is of quantitative importance.
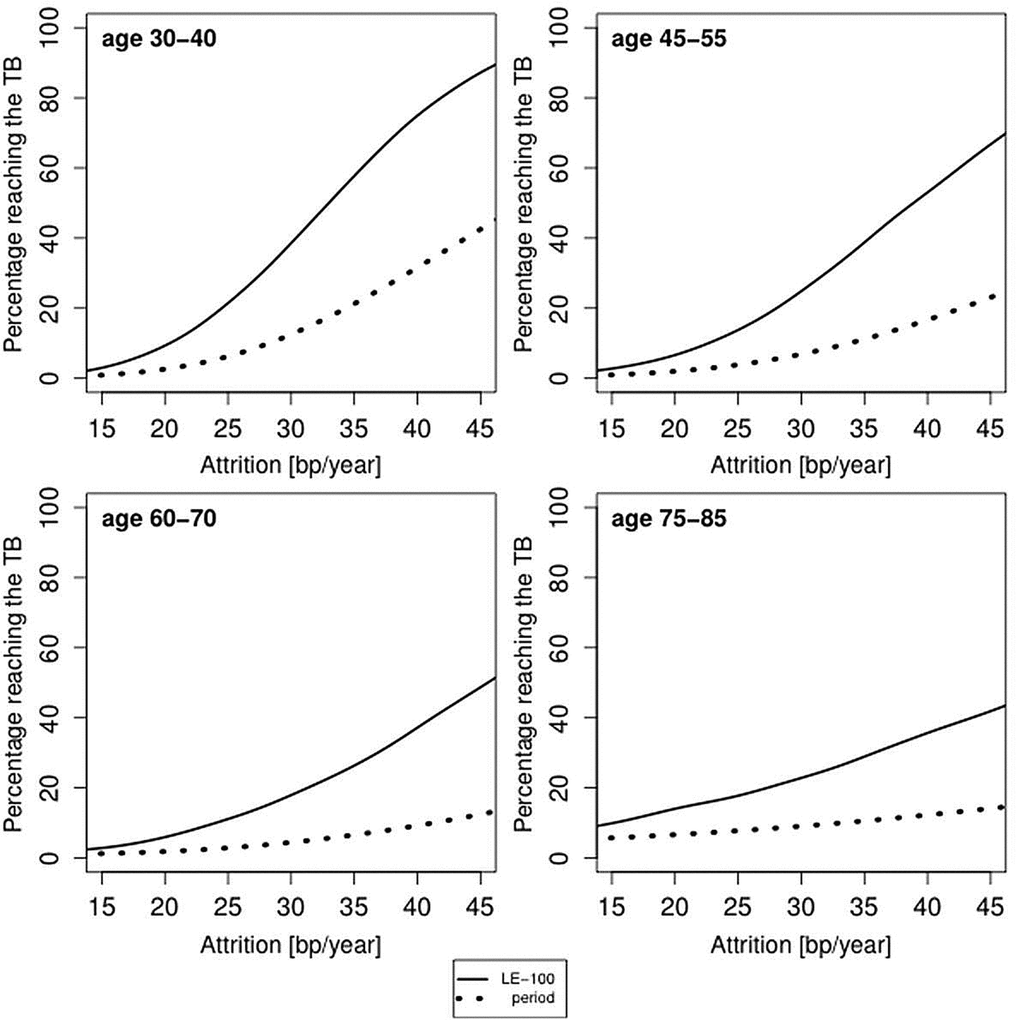
Figure 2. Predicted proportion of the composite study population reaching the telomere brink (TB; 5 kb) based on period life table mortality (period), life expectancy of 100 years (LE-100) and LTL attrition. The panels display findings for four age groups: 35 years (range 30-40 years); 50 years (range 45-55 years); 60 years (range 55-65 years); 80 years (range 75-85 years), based on different LTL attrition rates (15-45 bp/year). For period mortality, the proportion (in %) of individuals reaching an LTL of 5 kb before their life expectancy is based on the aggregate mortality data for a given country and sex at the time of blood collection.
Females typically have a longer LTL than males [26]. Females of modern societies also outlive males by approximately 5 years [24,27]. The Human Mortality Database for 2010 [24] indicates, for instance, that on the average, a Danish female dies at 81.3 years, while Danish male dies at 77.1 years; a French female dies at 84.7 years, while a French male dies at 78.0 years. Typically, females display an age-adjusted LTL that is approximately 150 bp longer than that of males (data presented in Table 1S are not adjusted for age). We examined, therefore, the impact of sex on the probability of reaching the TB (Figure 3). For period life table mortality, the probabilities for males and females of reaching the TB are almost identical. However, for the fixed LE-100, the probability of reaching the TB is higher in males than in females.
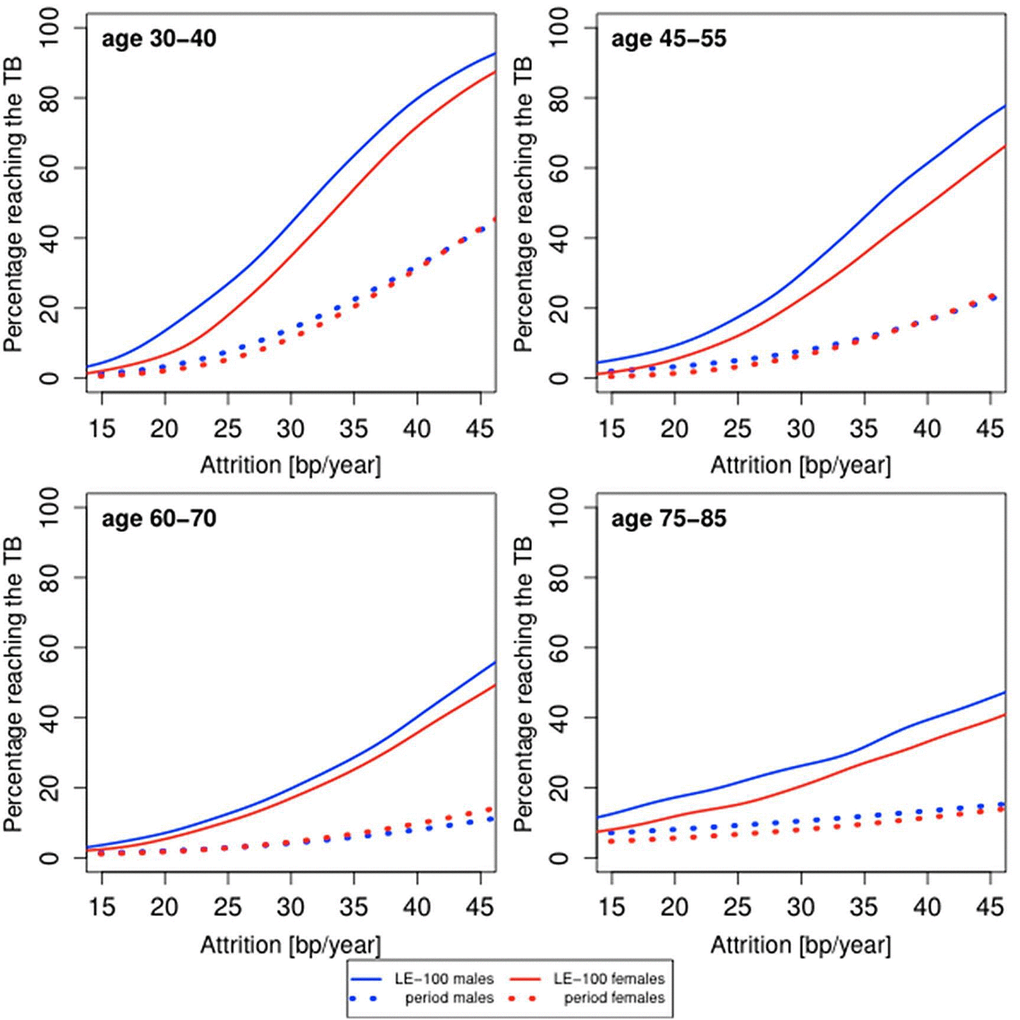
Figure 3. Predicted proportion of the composite study population of males and females reaching the telomere brink (TB; 5 kb) based on period life table mortality (period), life expectancy of 100 years (LE-100), LTL ranking and LTL attrition. The panels display findings for four age groups: 35 years (range 30-40 years); 50 years (range 45-55 years); 60 years (range 55-65 years); 80 years (range 75-85 years), based on different LTL attrition rates (15-45 bp/year).
Thus far, we presented the population average probability of reaching the TB, but this probability is likely to be heterogeneous, depending on the initial LTL. Adults typically display strong tracking of LTL, such that compared with peers, the individual’s LTL is virtually anchored to a given LTL rank at least over the course of up to 13 years of follow-up [28]. We attributed this phenomenon to the outsized influence of the inter-individual variation in LTL at birth and to a lesser extent to the inter-individual variation in LTL attrition during growth on the individual’s LTL throughout the life course [16]. We therefore examined the impact of LTL ranking by quintiles (1st quintile, shortest LTL; 5th quintile, longest LTL) on the probability of the individual reaching an LTL of 5 kb during his/her future life course. We observed that having an LTL ranked in the lower two quintiles of the LTL distribution significantly increased the probability of reaching the TB as compared to the higher quintiles, based on period mortality and more so based on LE-100 (Figure 4).
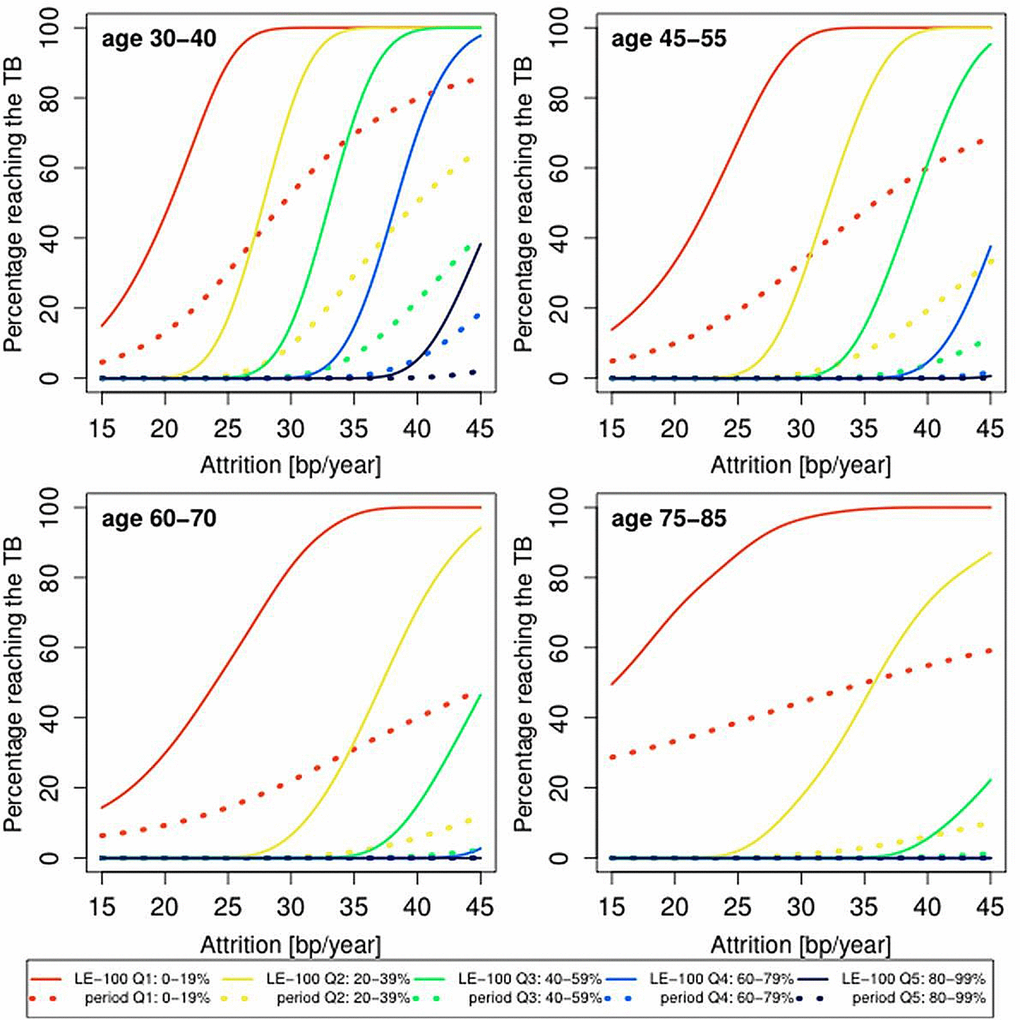
Figure 4. Predicted proportion of the composite study population reaching the telomere brink (TB; 5 kb) based on period life table mortality (period), life expectancy of 100 years (LE-100), LTL ranking and LTL attrition. Individuals were ranked by quintiles, where the shortest (1st) LTL quintile is 0-19% and the longest (5th) LTL quintile is 80-99%. The panels display findings for four age groups: 35 years (range 30-40 years); 50 years (range 45-55 years); 60 years (range 55-65 years); 80 years (range 75-85 years), based on different LTL attrition rates (15-45 bp/year).
The probabilities displayed in Figures 2-4 underscore that a subset of the general population might reach the TB at present life expectancies and more so at LE-100. However, they are based on constant rates of LTL attrition throughout the adult life course. In reality, the individual’s rate of LTL attrition probably fluctuates. Accordingly, we have also modeled the probability of reaching the TB with a theoretical distribution for the yearly LTL attrition based on randomized, variable LTL attrition rates over a 10-year LTL shortening of 300 bp ± 150 bp (SD) [25].
Table 1 displays the aggregate findings for the probability of reaching the TB inferred for the general population and the five LTL ranking quintiles. In this setting, for instance, a 35 year-old individual has a 14% and 39% probability of reaching the TB for period mortality and LE-100, respectively. An individual ranked in the lowest (1st) quintile of the LTL distribution has a 50% and a 93% probability of reaching the TB for period mortality and LE-100, respectively. We note that the dataset is a composite of studies comprising individuals of different age groups and nations (Figure 1). Thus, some of the small variations in the probability of reaching the TB for “All” and specific quintiles in Table 1 might reflect different mean ages of mortality in different nations. The relevant comparisons of interest are between period mortality and LE-100 and between individuals ranked in the upper versus lower quintiles.
Discussion
Our analysis suggests that the individual’s LTL, as reflected in his/her ranking, may be a major determinant of that individual’s natural lifespan limit from the standpoint of telomere biology. Clearly, the link between LTL and human longevity is more complex, since telomere biology might also contribute to human aging and longevity by mechanisms additional to a probability of having critically short telomeres that impact cell viability and the individual’s survival. For instance, age-dependent telomere shortening might alter gene expression in sub-telomeric regions [29], thus influencing aging and longevity. Moreover, double stranded DNA breaks in telomeres are irreparable and can bring about cell senescence and affect cellular viability when TL is not critically short [30]. That said, the TB concept underscores the fact that critically short telomeres can be reached in a subset of the general population for current life expectancy and in a substantially larger proportion for the LE-100.
Demonstrating that for period life table mortality the probability of reaching the TB is similar in males and females suggests that the female life expectancy advantage is approximately equivalent in terms of telomeric attrition years to the LTL sex gap. For instance, assuming tha females live 5 years longer than males, an LTL that is longer by 150 bp in females amounts to 5 years of telomeric equivalence for an average of 30 bp/year LTL attrition rate during adulthood. Further support for this thesis is provided by the trajectories based on the fixed LE-100. As females have a longer LTL than males, their risk of reaching the TB is smaller than that of males within a 100-year lifespan. Empirical data displayed in Figure 1 support this thesis; between the ages of 95-105 years, the fraction of males who have reached the TB of 5 kb is considerably higher than that of females. Collectively, these findings point to an association between LTL and the sex difference in lifespan.
Observing a nontrivial proportion with LTL ≤ 5 kb in the oldest old but not in the general population might stem from selection on two levels. First, in the context of current life expectancy, the oldest old are naturally a highly selected group. Their TB might be situated at lower LTLs. Second, epidemiological studies in the general population rarely recruit moribund, very sick individuals, who might display LTL ≤ 5 kb. However, studies of the oldest old rarely exclude participants in poor health. Therefore, if an LTL of 5 kb denotes a high probability of death or even imminent death, it is unlikely to be found in the general adult population but more likely to be found in the oldest old.
In conclusion, at present, most individuals are not reaching the LTL brink during their life course, but our findings suggest that further extension in human longevity will be increasingly constrained by telomere length. This inference requires an assumption that a possible increase in telomere length at birth and a decrease in the average rate of telomere length attrition after birth in future generations will not offset this prediction. Notably, however, potential interventions to forestall the telomeric brink may have adverse consequences. While short LTL [3,4] and alleles associated with a shorter LTL [10–12] increase CVD risk, recent studies show that long LTL [31–34] and alleles associated with long LTL [12,35–38] increase risk of major cancers. Such findings beg the (evolutionary) question: Why is human telomere length as long as it is? Emerging data suggests that evolution has been fine-tuning our telomere length to balance cancer against degenerative diseases [39–41]. In contemporary humans, this balance has ostensibly influenced longevity beyond the reproductive years.
Materials and Methods
Subjects
The LTL dataset was based on LTL measurements performed for cohorts participating in several studies [42–49]. Patients with DC and their unaffected relatives were participants in the National Cancer Institute’s Longitudinal Inherited Bone Marrow Failure Syndromes Study (NCI Protocol 02-C-0052, ClinicalTrials.gov Identifier NCT-00027274). DC patients included in this analysis had to have a germline mutation in one of the known causative DC genes and/or at least two features of the diagnostic triad (oral leukoplakia, reticular skin pigmentation, and/or nail dysplasia) along with other clinical findings consistent with DC. All relatives are mutation-negative except for 6/46 (13%); the probands in these 6 families are negative for known genes but have the diagnostic triad and other features consistent with DC.
All subjects whose LTL data are summarized in Tables S1 and S2 and those displayed in Figure 1 provided written informed consent approved by various institutional review boards and equivalent committees in the US, Europe and Israel.
Measurements of LTL by Southern blots of the terminal restriction fragments
These measurements were performed as previously described using the restriction enzymes Hinf I/Rsa I [50].
Modeling
For a chosen rate of attrition, we find each individual's TB age by:
where age is the age when the blood was collected. In case an individual has LTL < 5 kb, we let the TB age be the age at blood collection. The probability of survival until the TB age is determined through lookup in period life tables [24] or by assuming survival until the age of 100 years. We aggregate the individual probabilities in order to find the overall probability of reaching the TB age. To construct Figures 2-4, we have used attrition rates of 15-45 bp/year in steps of 1 bp/year. The smooth curves in Figures 2-4 were obtained by kernel smoothing of these point-estimates (normal kernel and a bandwidth of 5 bp/year).
For illustration, consider a 50 year-old French woman who had blood drawn in 1998 for measurements of LTL. If this individual’s LTL was 6.1 kb and her constant attrition is assumed to be 30 bp/year, she would reach the TB of 5 kb after 37 years (her TB age is 87 years). The Human Mortality Database for French females in 1998 shows that 46% of women, aged 50 years, live to be 87 or more. Thus, the probability for this 50 year-old French woman of reaching the brink is 46%.
We also modeled the probability of reaching the TB using a theoretical distribution of variable yearly LTL attrition. In order to find the aggregate probability of reaching the TB, we modeled the LTL attrition based on observations made in longitudinal studies with at least 10 years of follow-up, because measurement errors of LTL in longitudinal studies of shorter duration might produce an artifact in the form of LTL elongation [25]. We chose to model changes to LTL as the sum of yearly independent attrition, where the yearly attrition (in bp) is drawn from a gamma distribution with shape parameter 0.4 and scale parameter 75. The shape and scale parameters were chosen such that the total LTL attrition after 10 years has a mean of 300 bp and an SD of 150 bp . The gamma distribution was chosen (Figure S1) because it gives a rather good fit to observed data from a comprehensive longitudinal study of approximately 12 years LTL attrition, where LTL was measured by Southern blots.
This model enabled finding the theoretical LTL attrition distribution for any number of years after blood collection. When combined with mortality data, this approach allows us to effectively aggregate the curves in Figures 2 and 3 into single values for the probability of reaching the TB of 5 kb (Table 1) for each age group for the general population and for LTL ranking by quintiles.
Supplementary Materials
Acknowledgements
We thank James W. Vaupel for his valuable comments.
Conflicts of Interest
The authors have no conflict of interests to declare.
Funding
The Center of Human Development and Aging. AA present research support includes: NIH grants R01HD071180, R01HL116446, R01HL13840.
TwinsUK. The study was funded by the Wellcome Trust; European Community’s Seventh Framework Programme (FP7/2007-2013). The study also receives support from the National Institute for Health Research (NIHR) BioResource Clinical Research Facility and Biomedical Research Centre based at Guy's and St Thomas' NHS Foundation Trust and King's College London. Tim Spector is holder of an ERC Advanced Principal Investigator award. SNP Genotyping was performed by The Wellcome Trust Sanger Institute and National Eye Institute via NIH/CIDR.
The Bogalusa Heart Study was supported by grants 5R01ES021724 from National Institute of Environmental Health Science, and 2R01AG016592 from the National Institute on Aging.
The Framingham Heart Study. Supported by NIH contract N01-HC-25195. This project was supported in part by intramural funding from the National Heart, Lung, and Blood Institute and the Center for Population Studies of the NHLBI.
CHS. This CHS research was supported by NHLBI grant 1 R01 HL80698-01 and contracts N01-HC-85239, N01-HC-85079 through N01-HC-85086; N01-HC-35129, N01 HC-15103, N01 HC-55222, N01-HC-75150, N01-HC-45133, HHSN268201200036C and NHLBI grants HL080295, HL087652, HL105756 with additional contribution from NINDS. Additional support was provided through AG-023629, AG-15928, AG-20098, and AG-027058 from the NIA. See also http://www.chs-nhlbi.org/pi.htm. DNA handling and genotyping was supported in part by National Center of Advancing Translational Technologies CTSI grant UL1TR000124 and National Institute of Diabetes and Digestive and Kidney Diseases grant DK063491 to the Southern California Diabetes Endocrinology Research Center and Cedars-Sinai Board of Governors' Chair in Medical Genetics (JIR).
The Jerusalem LRC Longitudinal Study. The study was funded by the US-Israel Binational Science Foundation and the Israel Science Foundation.
The Danish Twin Registry was supported by the Danish Council for Independent Research - Medical Sciences; the Danish Aging Research Center is supported by the Velux Foundation; the INTERREG 4 A - Program Southern Denmark-Schleswig-K.E.R.N., supported by the European Regional Development Fund; and the A.P. Møller Foundation for the Advancement of Medical Science.
ADELAHYDE - Nancy study and ERA- France study. The study received support from the French Fondation pour la Recherche Médicale (FRM DCV2007-0409250) and the Plan Pluriformation (PPF815 PSVT-2005). Special thanks to Ms Cynthia Thiriot (INSERM U961, Nancy France) for her contribution to the genotyping of French cohorts.
HyperGEN. The study was supported by cooperative agreements HL54471, HL54472, HL54473, HL54495, HL54496, HL54509, HL54515 and grant HL055673. HyperGEN investigators and institutions can be found at http://www.biostat.wustl.edu/hypergen/hypergen.shtml.
The work of S.A.S. and S.M.G. was supported by the intramural research program of the Division of Cancer Epidemiology and Genetics, National Cancer Institute, National Institutes of Health. We thank the patients and their families for their valuable contributions.
References
- 1. Driver JA, Djoussé L, Logroscino G, Gaziano JM, Kurth T. Incidence of cardiovascular disease and cancer in advanced age: prospective cohort study. BMJ. 2008; 337:a2467. https://doi.org/10.1136/bmj.a2467 [PubMed]
- 2. Kochanek KD, Xu J, Murphy SL, Miniño AM, Kung HC. Deaths: preliminary data for 2009. Natl Vital Stat Rep. 2011; 59:1–51. [PubMed]
- 3. D’Mello MJ, Ross SA, Briel M, Anand SS, Gerstein H, Paré G. Association between shortened leukocyte telomere length and cardiometabolic outcomes: systematic review and meta-analysis. Circ Cardiovasc Genet. 2015; 8:82–90. https://doi.org/10.1161/CIRCGENETICS.113.000485 [PubMed]
- 4. Haycock PC, Heydon EE, Kaptoge S, Butterworth AS, Thompson A, Willeit P. Leucocyte telomere length and risk of cardiovascular disease: systematic review and meta-analysis. BMJ. 2014; 349:g4227. https://doi.org/10.1136/bmj.g4227 [PubMed]
- 5. Bakaysa SL, Mucci LA, Slagboom PE, Boomsma DI, McClearn GE, Johansson B, Pedersen NL. Telomere length predicts survival independent of genetic influences. Aging Cell. 2007; 6:769–74. https://doi.org/10.1111/j.1474-9726.2007.00340.x [PubMed]
- 6. Deelen J, Beekman M, Codd V, Trompet S, Broer L, Hägg S, Fischer K, Thijssen PE, Suchiman HE, Postmus I, Uitterlinden AG, Hofman A, de Craen AJ, et al. Leukocyte telomere length associates with prospective mortality independent of immune-related parameters and known genetic markers. Int J Epidemiol. 2014; 43:878–86. https://doi.org/10.1093/ije/dyt267 [PubMed]
- 7. Kimura M, Hjelmborg JV, Gardner JP, Bathum L, Brimacombe M, Lu X, Christiansen L, Vaupel JW, Aviv A, Christensen K. Telomere length and mortality: a study of leukocytes in elderly Danish twins. Am J Epidemiol. 2008; 167:799–806. https://doi.org/10.1093/aje/kwm380 [PubMed]
- 8. Fitzpatrick AL, Kronmal RA, Kimura M, Gardner JP, Psaty BM, Jenny NS, Tracy RP, Hardikar S, Aviv A. Leukocyte telomere length and mortality in the Cardiovascular Health Study. J Gerontol A Biol Sci Med Sci. 2011; 66:421–29. https://doi.org/10.1093/gerona/glq224 [PubMed]
- 9. Boonekamp JJ, Simons MJ, Hemerik L, Verhulst S. Telomere length behaves as biomarker of somatic redundancy rather than biological age. Aging Cell. 2013; 12:330–32. https://doi.org/10.1111/acel.12050 [PubMed]
- 10. Codd V, Nelson CP, Albrecht E, Mangino M, Deelen J, Buxton JL, Hottenga JJ, Fischer K, Esko T, Surakka I, Broer L, Nyholt DR, Mateo Leach I, et al, and CARDIoGRAM consortium. Identification of seven loci affecting mean telomere length and their association with disease. Nat Genet. 2013; 45:422–27, e1–2. https://doi.org/10.1038/ng.2528 [PubMed]
- 11. Scheller Madrid A, Rode L, Nordestgaard BG, Bojesen SE. Short telomere length and ischemic heart disease: observational and genetic studies in 290 022 Individuals. Clin Chem. 2016; 62:1140–49. https://doi.org/10.1373/clinchem.2016.258566 [PubMed]
- 12. Haycock P, et al. The association between genetically elevated telomere length and risk of cancer and non-neoplastic diseases. JAMA Oncol. 2017. Epub ahead of print. https://doi.org/10.1001/jamaoncol.2016.5945
- 13. Daniali L, Benetos A, Susser E, Kark JD, Labat C, Kimura M, Desai K, Granick M, Aviv A. Telomeres shorten at equivalent rates in somatic tissues of adults. Nat Commun. 2013; 4:1597. https://doi.org/10.1038/ncomms2602 [PubMed]
- 14. Hjelmborg JB, Dalgård C, Möller S, Steenstrup T, Kimura M, Christensen K, Kyvik KO, Aviv A. The heritability of leucocyte telomere length dynamics. J Med Genet. 2015; 52:297–302. https://doi.org/10.1136/jmedgenet-2014-102736 [PubMed]
- 15. Slagboom PE, Droog S, Boomsma DI. Genetic determination of telomere size in humans: a twin study of three age groups. Am J Hum Genet. 1994; 55:876–82. [PubMed]
- 16. Factor-Litvak P, Susser E, Kezios K, McKeague I, Kark JD, Hoffman M, Kimura M, Wapner R, Aviv A. Leukocyte telomere length in newborns: implications for the role of telomeres in human disease. Pediatrics. 2016; 137:e20153927. https://doi.org/10.1542/peds.2015-3927 [PubMed]
- 17. Christensen K, Doblhammer G, Rau R, Vaupel JW. Ageing populations: the challenges ahead. Lancet. 2009; 374:1196–208. https://doi.org/10.1016/S0140-6736(09)61460-4 [PubMed]
- 18. Dong X, Milholland B, Vijg J. Evidence for a limit to human lifespan. Nature. 2016; 538:257–59. https://doi.org/10.1038/nature19793 [PubMed]
- 19. Oeppen J, Vaupel JW. Demography. Broken limits to life expectancy. Science. 2002; 296:1029–31. https://doi.org/10.1126/science.1069675 [PubMed]
- 20. Olshansky SJ, Passaro DJ, Hershow RC, Layden J, Carnes BA, Brody J, Hayflick L, Butler RN, Allison DB, Ludwig DS. A potential decline in life expectancy in the United States in the 21st century. N Engl J Med. 2005; 352:1138–45. https://doi.org/10.1056/NEJMsr043743 [PubMed]
- 21. Olshansky SJ, Carnes BA, Désesquelles A. Demography. Prospects for human longevity. Science. 2001; 291:1491–92. https://doi.org/10.1126/science.291.5508.1491 [PubMed]
- 22. Ballew BJ, Savage SA. Updates on the biology and management of dyskeratosis congenita and related telomere biology disorders. Expert Rev Hematol. 2013; 6:327–37. https://doi.org/10.1586/ehm.13.23 [PubMed]
- 23. Nelson ND, Bertuch AA. Dyskeratosis congenita as a disorder of telomere maintenance. Mutat Res. 2012; 730:43–51. https://doi.org/10.1016/j.mrfmmm.2011.06.008 [PubMed]
- 24. Human Mortality Database. University of California, Berkeley (USA), and Max Planck Institute for Demographic Research (Germany).Available at www.mortality.org or www.humanmortality.de.
- 25. Steenstrup T, Hjelmborg JV, Kark JD, Christensen K, Aviv A. The telomere lengthening conundrum--artifact or biology? Nucleic Acids Res. 2013; 41:e131. https://doi.org/10.1093/nar/gkt370 [PubMed]
- 26. Gardner M, Bann D, Wiley L, Cooper R, Hardy R, Nitsch D, Martin-Ruiz C, Shiels P, Sayer AA, Barbieri M, Bekaert S, Bischoff C, Brooks-Wilson A, et al, and Halcyon study team. Gender and telomere length: systematic review and meta-analysis. Exp Gerontol. 2014; 51:15–27. https://doi.org/10.1016/j.exger.2013.12.004 [PubMed]
- 27. Colchero F, Rau R, Jones OR, Barthold JA, Conde DA, Lenart A, Nemeth L, Scheuerlein A, Schoeley J, Torres C, Zarulli V, Altmann J, Brockman DK, et al. The emergence of longevous populations. Proc Natl Acad Sci USA. 2016; 113:E7681–90. https://doi.org/10.1073/pnas.1612191113 [PubMed]
- 28. Benetos A, Kark JD, Susser E, Kimura M, Sinnreich R, Chen W, Steenstrup T, Christensen K, Herbig U, von Bornemann Hjelmborg J, Srinivasan SR, Berenson GS, Labat C, Aviv A. Tracking and fixed ranking of leukocyte telomere length across the adult life course. Aging Cell. 2013; 12:615–21. https://doi.org/10.1111/acel.12086 [PubMed]
- 29. Ottaviani A, Gilson E, Magdinier F. Telomeric position effect: from the yeast paradigm to human pathologies? Biochimie. 2008; 90:93–107. https://doi.org/10.1016/j.biochi.2007.07.022 [PubMed]
- 30. Fumagalli M, Rossiello F, Clerici M, Barozzi S, Cittaro D, Kaplunov JM, Bucci G, Dobreva M, Matti V, Beausejour CM, Herbig U, Longhese MP, d’Adda di Fagagna F. Telomeric DNA damage is irreparable and causes persistent DNA-damage-response activation. Nat Cell Biol. 2012; 14:355–65. https://doi.org/10.1038/ncb2466 [PubMed]
- 31. Anic GM, Sondak VK, Messina JL, Fenske NA, Zager JS, Cherpelis BS, Lee JH, Fulp WJ, Epling-Burnette PK, Park JY, Rollison DE. Telomere length and risk of melanoma, squamous cell carcinoma, and basal cell carcinoma. Cancer Epidemiol. 2013; 37:434–39. https://doi.org/10.1016/j.canep.2013.02.010 [PubMed]
- 32. Seow WJ, Cawthon RM, Purdue MP, Hu W, Gao YT, Huang WY, Weinstein SJ, Ji BT, Virtamo J, Hosgood HD
3rd , Bassig BA, Shu XO, Cai Q, et al. Telomere length in white blood cell DNA and lung cancer: a pooled analysis of three prospective cohorts. Cancer Res. 2014; 74:4090–98. https://doi.org/10.1158/0008-5472.CAN-14-0459 [PubMed] - 33. Sanchez-Espiridion B, Chen M, Chang JY, Lu C, Chang DW, Roth JA, Wu X, Gu J. Telomere length in peripheral blood leukocytes and lung cancer risk: a large case-control study in Caucasians. Cancer Res. 2014; 74:2476–86. https://doi.org/10.1158/0008-5472.CAN-13-2968 [PubMed]
- 34. Julin B, Shui I, Heaphy CM, Joshu CE, Meeker AK, Giovannucci E, De Vivo I, Platz EA. Circulating leukocyte telomere length and risk of overall and aggressive prostate cancer. Br J Cancer. 2015; 112:769–76. https://doi.org/10.1038/bjc.2014.640 [PubMed]
- 35. Iles MM, Bishop DT, Taylor JC, Hayward NK, Brossard M, Cust AE, Dunning AM, Lee JE, Moses EK, Akslen LA, Andresen PA, Avril MF, Azizi E, et al, and AMFS Investigators, and IBD investigators, and QMEGA and QTWIN Investigators, and SDH Study Group, and GenoMEL Consortium. The effect on melanoma risk of genes previously associated with telomere length. J Natl Cancer Inst. 2014; 106:dju267. https://doi.org/10.1093/jnci/dju267 [PubMed]
- 36. Ojha J, Codd V, Nelson CP, Samani NJ, Smirnov IV, Madsen NR, Hansen HM, de Smith AJ, Bracci PM, Wiencke JK, Wrensch MR, Wiemels JL, Walsh KM, and ENGAGE Consortium Telomere Group. Genetic Variation Associated with Longer Telomere Length Increases Risk of Chronic Lymphocytic Leukemia. Cancer Epidemiol Biomarkers Prev. 2016; 25:1043–49. https://doi.org/10.1158/1055-9965.EPI-15-1329 [PubMed]
- 37. Walsh KM, Wiencke JK, Lachance DH, Wiemels JL, Molinaro AM, Eckel-Passow JE, Jenkins RB, Wrensch MR. Telomere maintenance and the etiology of adult glioma. Neuro-oncol. 2015; 17:1445–52. https://doi.org/10.1093/neuonc/nov082 [PubMed]
- 38. Zhang C, Doherty JA, Burgess S, Hung RJ, Lindström S, Kraft P, Gong J, Amos CI, Sellers TA, Monteiro AN, Chenevix-Trench G, Bickeböller H, Risch A, et al, and GECCO and GAME-ON Network: CORECT, DRIVE, ELLIPSE, FOCI, and TRICL. Genetic determinants of telomere length and risk of common cancers: a Mendelian randomization study. Hum Mol Genet. 2015; 24:5356–66. https://doi.org/10.1093/hmg/ddv252 [PubMed]
- 39. Hansen ME, Hunt SC, Stone RC, Horvath K, Herbig U, Ranciaro A, Hirbo J, Beggs W, Reiner AP, Wilson JG, Kimura M, De Vivo I, Chen MM, et al. Shorter telomere length in Europeans than in Africans due to polygenetic adaptation. Hum Mol Genet. 2016; 25:2324–30. https://doi.org/10.1093/hmg/ddw070 [PubMed]
- 40. Mangino M, Christiansen L, Stone R, Hunt SC, Horvath K, Eisenberg DT, Kimura M, Petersen I, Kark JD, Herbig U, Reiner AP, Benetos A, Codd V, et al. DCAF4, a novel gene associated with leucocyte telomere length. J Med Genet. 2015; 52:157–62. https://doi.org/10.1136/jmedgenet-2014-102681 [PubMed]
- 41. Stone RC, Horvath K, Kark JD, Susser E, Tishkoff SA, Aviv A. Telomere length and the cancer-atherosclerosis trade-off. PLoS Genet. 2016; 12:e1006144. https://doi.org/10.1371/journal.pgen.1006144 [PubMed]
- 42. Fitzpatrick AL, Kronmal RA, Gardner JP, Psaty BM, Jenny NS, Tracy RP, Walston J, Kimura M, Aviv A. Leukocyte telomere length and cardiovascular disease in the cardiovascular health study. Am J Epidemiol. 2007; 165:14–21. https://doi.org/10.1093/aje/kwj346 [PubMed]
- 43. Benetos A, Okuda K, Lajemi M, Kimura M, Thomas F, Skurnick J, Labat C, Bean K, Aviv A. Telomere length as an indicator of biological aging: the gender effect and relation with pulse pressure and pulse wave velocity. Hypertension. 2001; 37:381–85. https://doi.org/10.1161/01.HYP.37.2.381 [PubMed]
- 44. Barbieri M, Paolisso G, Kimura M, Gardner JP, Boccardi V, Papa M, Hjelmborg JV, Christensen K, Brimacombe M, Nawrot TS, Staessen JA, Pollak MN, Aviv A. Higher circulating levels of IGF-1 are associated with longer leukocyte telomere length in healthy subjects. Mech Ageing Dev. 2009; 130:771–76. https://doi.org/10.1016/j.mad.2009.10.002 [PubMed]
- 45. Chen W, Kimura M, Kim S, Cao X, Srinivasan SR, Berenson GS, Kark JD, Aviv A. Longitudinal versus cross-sectional evaluations of leukocyte telomere length dynamics: age-dependent telomere shortening is the rule. J Gerontol A Biol Sci Med Sci. 2011; 66:312–19. https://doi.org/10.1093/gerona/glq223 [PubMed]
- 46. Hunt SC, Chen W, Gardner JP, Kimura M, Srinivasan SR, Eckfeldt JH, Berenson GS, Aviv A. Leukocyte telomeres are longer in African Americans than in whites: the National Heart, Lung, and Blood Institute Family Heart Study and the Bogalusa Heart Study. Aging Cell. 2008; 7:451–58. https://doi.org/10.1111/j.1474-9726.2008.00397.x [PubMed]
- 47. Kark JD, Goldberger N, Kimura M, Sinnreich R, Aviv A. Energy intake and leukocyte telomere length in young adults. Am J Clin Nutr. 2012; 95:479–87. https://doi.org/10.3945/ajcn.111.024521 [PubMed]
- 48. Mangino M, Hwang SJ, Spector TD, Hunt SC, Kimura M, Fitzpatrick AL, Christiansen L, Petersen I, Elbers CC, Harris T, Chen W, Srinivasan SR, Kark JD, et al. Genome-wide meta-analysis points to CTC1 and ZNF676 as genes regulating telomere homeostasis in humans. Hum Mol Genet. 2012; 21:5385–94. https://doi.org/10.1093/hmg/dds382 [PubMed]
- 49. O’Donnell CJ, Demissie S, Kimura M, Levy D, Gardner JP, White C, D’Agostino RB, Wolf PA, Polak J, Cupples LA, Aviv A. Leukocyte telomere length and carotid artery intimal medial thickness: the Framingham Heart Study. Arterioscler Thromb Vasc Biol. 2008; 28:1165–71. https://doi.org/10.1161/ATVBAHA.107.154849 [PubMed]
- 50. Kimura M, Stone RC, Hunt SC, Skurnick J, Lu X, Cao X, Harley CB, Aviv A. Measurement of telomere length by the Southern blot analysis of terminal restriction fragment lengths. Nat Protoc. 2010; 5:1596–607. https://doi.org/10.1038/nprot.2010.124 [PubMed]


