Introduction
The germ-line breast cancer 1 interacting protein 1 (BRIP1) comes to light as a crucial protein for BRCA1-dependent DNA damage repair functions [1-3]. The human BRIP1 gene (also named FANCJ or BACH1) is located on chromosome 17q22, comprising of 19 introns and 20 exons, and encodes BRCA1-associated C-terminal helicase 1 [1,4] And its mutations that affect helicase activity have been identified in patients suffering early-stage breast cancer. Missense mutations in BRIP1 may increase breast cancer risk [5]. Therefore, it is considered as a moderate-penetrance susceptibility gene for breast cancer. However, previous studies declared that BRIP1 mutation not only has effect on breast cancer, but also in other various cancers including cervical cancer [5-7], ovarian cancer [4,8] and prostate cancer [9].
It is observed that the genetic polymorphisms in BRIP1 influence the cancer susceptibility by altering their natural function. And many single-nucleotide polymorphisms (SNPs) in BRIP1 have been recognized. SNPs may alter the expression, processing, and transcription of genes, and thus contribute to cancer development. Numerous epidemiological studies have demonstrated that some SNPs located within genes can alter their expression and/or maturation and are associated with cancer susceptibility and progression.
Recently, numerous molecular epidemiology studies explored the relationship between BRIP1 polymorphisms and cancer susceptibility. Nevertheless, their results were inconclusive. Polymorphisms of BRIP1 is regarded as an important susceptibility factor in cervical cancer, but not in breast cancer [5-7]. For example, Due to the inconsistencies among these previous studies, we conducted this meta-analysis covering all eligible molecular epidemiology studies to validate the correlation of four most common BRIP1 polymorphisms (rs2048718, rs4988344, rs4986764, and rs6504074) and cancer risk.
Results
Study characteristics
According to our inclusion criteria, 18 studies from 15 articles containing 13,716 cancer patients and 15,590 cancer-free controls were finally included. The detail screening process was exhibited in Figure 1. It contained four separate studies in Song’s articles focusing on breast cancer and ovarian cancer.
Figure 1. The flow diagram of the meta-analysis. CNKI: China National Knowledge Infrastructure.
Among the 18 eligible studies, 11 were conducted in Caucasian populations from the UK, Italy, Germany, USA, Canada, Denmark, Cyprus, and Finland. Five studies were on Asian background and all samples were Chinese. And one study was performed on mixed population. All the studies were case-control design, studying on various cancer types including breast, ovarian, cervical, gastric and prostate cancer. Table 1 listed the main characteristics of all included studies.
Table 1. Characteristics of the studies included in the meta-analysis.
| Study | Year | Country | Ethnicity | Cancer type | Genotyping medthod | Source of control | Case/Control | SNP No. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carrera-Lasfuentes [20] | 2017 | Spain | Mixed | GC | QIAamp | Population | 598/601 | 3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Zhou [21] | 2014 | China | Asian | CC | MassARRAY | Population | 309/315 | 1, 3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ren [14] | 2013 | China | Asian | BC | Mass ARRAY | Hospital | 319/306 | 1,2,3,4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ma 1 [6] | 2013 | China | Asian | CC | Mass ARRAY | Population | 454/562 | 1,2,3,4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ma 2 [5] | 2013 | China | Asian | CC | Mass ARRAY | Hospital | 298/286 | 1,2,3,4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Silvestri [22] | 2011 | Italy | Caucasian | BC | PCR-SSCP | Hospital | 97/203 | 3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Loizidou [23] | 2010 | Cyprus | Caucasian | BC | MassArray | Population | 1108/1170 | 3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Huo [24] | 2009 | China | Asian | BC | PCR-PIRA | Population | 568/624 | 3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kote-Jarai [9] | 2009 | UK | Caucasian | PC | TaqMan | Population | 1841/1880 | 4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Guénard [25] | 2008 | Canada | Caucasian | BC | PCR-RFLP | Hospital | 96/70 | 1,3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Song 1 [4] | 2007 | UK | Caucasian | BC | TaqMan | Population | 2270/2280 | 1,2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Song 2 [4] | 2007 | UK | Caucasian | OC | TaqMan | Population | 730/855 | 1,2,4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Song 3 [4] | 2007 | USA | Caucasian | OC | TaqMan | Population | 327/429 | 1,2,4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Song 4 [4] | 2007 | Denmark | Caucasian | OC | TaqMan | Population | 456/1231 | 1,2,4 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Frank [26] | 2007 | Germany | Caucasian | BC | TaqMan | Population | 571/712 | 1,3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Garcia-Closas [20] | 2006 | Mixed | Caucasian | BC | qPCR | Population | 1596/1254 | 3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vahteristo [27] | 2006 | Finland | Caucasian | BC | TaqMan | Population | 866/731 | 3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Seal [28] | 2006 | UK | Caucasian | BC | Pyrosequencing | Population | 1212/2081 | 3 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations BC: breast cancer; CC: cervical cancer; PC: Prostate cancer; OC: ovarian cancer; PCR: polymerase chain reaction; RFLP: restriction fragment length polymorphism. PIRA: PCR-primer introduced restriction analysis; DHPLC, denaturing high -performance liquid chromatography; SSCP: single strand conformation polymorphism; SNP: single-nucleotide polymorphisms; SNP No.1: rs2048718; 2: rs4988344; 3: rs4986764; 4.rs6504074 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Quantitative synthesis of the four polymorphisms
As Table 2 showed, the frequencies of the minor allele (MAF) in the cancer-free controls varied widely across included studies, ranging from 0.23 to 0.62 for rs2048718, 0.14 to 0.62 for rs4988344, 0.26 to 0.60 for rs4986764, and 0.24 to 0.28 for rs6504074. The average frequencies of the minor allele for the four abovementioned polymorphisms were 0.39, 0.35, 0.40, and 0.26, respectively.
Table 2. BRIP1 polymorphisms Genotype Distribution and Allele Frequency in this meta-analysis.
| Study | Genotype (N) | Allele frequency (N) | MAF | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Case | Control | Case | Control | HWE | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| total | AA | AB | BB | total | AA | AB | BB | A | B | A | B | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2048718 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Zhou 2014 | 309 | 196 | 94 | 19 | 314 | 183 | 118 | 13 | 486 | 132 | 484 | 144 | 0.23 | 0.26 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ren 2013 | 319 | 201 | 98 | 20 | 305 | 177 | 115 | 13 | 500 | 138 | 469 | 141 | 0.23 | 0.29 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ma 1 2013 | 454 | 285 | 141 | 28 | 560 | 326 | 208 | 26 | 711 | 197 | 860 | 260 | 0.25 | 0.32 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ma 2 2013 | 298 | 188 | 92 | 18 | 285 | 166 | 106 | 13 | 468 | 128 | 438 | 132 | 0.23 | 0.45 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Guénard 2008 | 96 | 38 | 41 | 17 | 70 | 23 | 32 | 15 | 117 | 75 | 78 | 62 | 0.41 | 0.54 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Song 1 2007 | 2170 | 655 | 1063 | 452 | 2264 | 655 | 1151 | 458 | 2373 | 1967 | 2461 | 2067 | 0.47 | 0.24 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Song 2 2007 | 722 | 236 | 341 | 145 | 847 | 246 | 425 | 176 | 813 | 631 | 917 | 777 | 0.50 | 0.76 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Song 3 2007 | 322 | 91 | 157 | 74 | 421 | 131 | 195 | 95 | 339 | 305 | 457 | 385 | 0.51 | 0.17 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Song 4 2007 | 429 | 118 | 215 | 96 | 1209 | 352 | 632 | 225 | 451 | 407 | 1336 | 1082 | 0.62 | 0.05 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Frank 2007 | 571 | 181 | 283 | 107 | 712 | 228 | 340 | 144 | 645 | 497 | 796 | 628 | 0.49 | 0.40 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs4988344 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ren 2013 | 319 | 65 | 145 | 109 | 306 | 43 | 148 | 115 | 275 | 363 | 234 | 378 | 0.62 | 0.67 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ma 1 2013 | 454 | 92 | 207 | 155 | 562 | 84 | 270 | 208 | 391 | 517 | 438 | 686 | 0.61 | 0.81 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ma 2 2013 | 297 | 60 | 136 | 101 | 286 | 42 | 138 | 106 | 256 | 338 | 222 | 350 | 0.61 | 0.79 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Song 1 2007 | 2189 | 1552 | 585 | 52 | 2278 | 1609 | 616 | 53 | 3689 | 689 | 3834 | 722 | 0.16 | 0.51 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Song 2 2007 | 729 | 498 | 203 | 28 | 848 | 589 | 239 | 20 | 1199 | 259 | 1417 | 279 | 0.16 | 0.46 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Song 3 2007 | 323 | 228 | 86 | 9 | 427 | 309 | 103 | 15 | 542 | 104 | 721 | 133 | 0.16 | 0.09 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Song 4 2007 | 278 | 188 | 82 | 8 | 712 | 526 | 174 | 12 | 458 | 98 | 1226 | 198 | 0.14 | 0.58 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs4986764 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carrera-Lasfuentes 2017 | 598 | 232 | 297 | 69 | 601 | 224 | 270 | 107 | 761 | 435 | 718 | 484 | 0.40 | 0.11 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Zhou 2014 | 309 | 164 | 116 | 29 | 315 | 136 | 146 | 33 | 444 | 174 | 418 | 212 | 0.34 | 0.50 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ren 2013 | 319 | 168 | 120 | 31 | 306 | 132 | 141 | 33 | 456 | 182 | 405 | 207 | 0.34 | 0.61 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ma 1 2013 | 454 | 247 | 165 | 42 | 562 | 240 | 258 | 64 | 659 | 249 | 738 | 386 | 0.34 | 0.67 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ma 2 2013 | 298 | 160 | 110 | 28 | 286 | 122 | 132 | 32 | 430 | 166 | 376 | 196 | 0.34 | 0.68 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Silvestri 2011 | 97 | 37 | 49 | 11 | 203 | 82 | 95 | 26 | 123 | 71 | 259 | 147 | 0.36 | 0.85 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Loizidou 2010 | 1108 | 465 | 502 | 141 | 1170 | 475 | 534 | 161 | 1432 | 784 | 1484 | 856 | 0.37 | 0.58 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Huo 2009 | 568 | 308 | 227 | 33 | 624 | 345 | 232 | 47 | 843 | 293 | 922 | 326 | 0.26 | 0.36 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Guénard 2008 | 96 | 18 | 48 | 30 | 70 | 12 | 32 | 26 | 84 | 108 | 56 | 84 | 0.60 | 0.69 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Frank 2007 | 571 | 181 | 295 | 95 | 712 | 226 | 365 | 121 | 657 | 485 | 817 | 607 | 0.43 | 0.20 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Garcia-Closas 2006 | 1596 | 529 | 761 | 306 | 1254 | 406 | 612 | 236 | 1819 | 1373 | 1424 | 1084 | 0.43 | 0.84 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vahteristo 2006 | 866 | 184 | 428 | 254 | 731 | 148 | 382 | 201 | 796 | 936 | 678 | 784 | 0.54 | 0.17 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Seal 2006 | 1212 | 462 | 549 | 201 | 2081 | 783 | 970 | 328 | 1473 | 951 | 2536 | 1626 | 0.39 | 0.34 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs6504074 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ren 2013 | 319 | 203 | 95 | 21 | 304 | 176 | 110 | 18 | 501 | 137 | 462 | 146 | 0.24 | 0.88 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ma 1 2013 | 454 | 288 | 137 | 29 | 558 | 317 | 205 | 36 | 713 | 195 | 839 | 277 | 0.25 | 0.71 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ma 2 2013 | 298 | 188 | 92 | 18 | 284 | 162 | 104 | 18 | 468 | 128 | 428 | 140 | 0.25 | 0.81 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kote-Jarai 2009 | 1841 | 963 | 727 | 151 | 1880 | 1031 | 727 | 122 | 2653 | 1029 | 2789 | 971 | 0.26 | 0.68 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Song 2 2007 | 725 | 417 | 270 | 38 | 847 | 457 | 325 | 65 | 1104 | 346 | 1239 | 455 | 0.27 | 0.50 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Song 3 2007 | 324 | 170 | 117 | 37 | 421 | 225 | 156 | 40 | 457 | 191 | 606 | 236 | 0.28 | 0.09 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Song 4 2007 | 260 | 137 | 104 | 19 | 650 | 340 | 258 | 52 | 378 | 142 | 938 | 362 | 0.28 | 0.76 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations A: the major allele, B: the minor allele. MAF: minor allele frequencies. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 3 listed the results of this meta-analysis. There were 10 studies with 5,690 cancer patients and 6,087 healthy individuals for rs2048718. As displayed in Table 3 and Figure 2, rs2048718 polymorphism had a decrease risk of overall cancer based on the heterozygous and dominant models (AB vs. AA: OR = 0.90, 95% CI = 0.83–0.97, P = 0.008; AB+BB vs. AA: OR = 0.92, 95%CI = 0.86–0.99, P = 0.037). In the stratified analysis by ethnicity, the results showed significant associations between rs2048718 and cancer risk in the Asian population by heterozygous and dominant comparison (AB vs. AA: OR = 0.76, 95% CI = 0.65–0.89, P = 0.001; AB+BB vs. AA: OR = 0.82, 95%CI = 0.70–0.95, P = 0.008), while no association was found among Caucasians. However, in the stratified analysis by cancer types, we found a decrease risk between rs2048718 and cervical cancer under heterozygous and dominant model (AB vs. AA: OR = 0.76, 95% CI = 0.64–0.91, P = 0.003; AB+BB vs. AA: OR = 0.82, 95%CI = 0.69–0.97, P = 0.021), and the results showed no significant difference between rs2048718 polymorphism and gynecologic (breast and ovarian) cancer susceptibility.
Table 3. Meta-analysis results.
| Comparisons | B vs A | BB vs AA | AB vs AA | BB vs AA+AB | AB+BB vs AA | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OR (95%CI) | P | OR (95%CI) | P | OR (95%CI) | P | OR (95%CI) | P | OR (95%CI) | P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2048718 | 0.98(0.93–1.03) | 0.408 | 1.02(0.92–1.14) | 0.704 | 0.90(0.83–0.97) | 0.008 | 1.05 (0.96–1.16) | 0.269 | 0.92(0.86–0.99) | 0.037 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Caucasian | 0.99(0.89–1.12) | 0.921 | 0.99(0.89–1.12) | 0.921 | 0.95(0.86–1.04) | 0.274 | 1.03 (0.93–1.14) | 0.595 | 0.96(0.88–1.05) | 0.381 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Asian | 0.91(0.81–1.04) | 0.160 | 1.28(0.92–1.80) | 0.147 | 0.76(0.65–0.89) | 0.001 | 1.42 (1.02–1.97) | 0.040 | 0.82(0.70–0.95) | 0.008 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CC | 0.91(0.79–1.05) | 0.209 | 1.26(0.86-1.85) | 0.228 | 0.76(0.64-0.91) | 0.003 | 1.39(0.96–2.03) | 0.084 | 0.82(0.69-0.97) | 0.021 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BC | 0.98(0.91–1.05) | 0.479 | 0.98(0.85–1.13) | 0.771 | 0.92(0.82-1.03) | 0.151 | 1.02 (0.90–1.15) | 0.820 | 0.94(0.84–1.04) | 0.21 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OC | 1.01(0.92-1.11) | 0.772 | 1.04(0.86–1.26) | 0.665 | 0.96(0.82-1.11) | 0.563 | 1.07(0.91-1.26) | 0.410 | 0.98(0.85-1.13) | 0.775 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs4988344 | 0.97(0.87–1.09) | 0.620 | 0.89(0.67–1.18) | 0.418 | 0.94(0.80–1.10) | 0.441 | 0.95(0.82-1.10) | 0.468 | 0.93(0.79–1.11) | 0.430 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Caucasian | 1.03(0.93-1.14) | 0.533 | 1.18(0.89–1.57) | 0.260 | 1.03(0.93–1.14) | 0.533 | 1.17(0.88–1.55) | 0.282 | 1.44 (0.99–2.08) | 0.38 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Asian | 0.68(0.54-0.86) | 0.001 | 0.66(0.52–0.85) | 0.001 | 0.68 (0.54–0.86) | 0.001 | 0.88 (0.74–1.04) | 0.132 | 0.78 (0.62–0.97) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BC | 0.92(0.77-1.11) | 0.395 | 0.81(0.51-1.30) | 0.391 | 0.85(0.57-1.25) | 0.405 | 0.93(0.72-1.19) | 0.542 | 0.87(0.56-1.34) | 0.519 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OC | 1.14(0.99-1.30) | 0.063 | 1.41(0.92-2.14) | 0.114 | 1.11(0.94-1.30) | 0.211 | 1.37(0.90-2.09) | 0.139 | 1.63(0.98-2.72) | 0.059 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs4986764 | 0.94(0.90-0.98) | 0.001 | 0.90(0.82–0.99) | 0.024 | 0.89 (0.80–0.99) | 0.025 | 0.95 (0.87–1.03 | 0.203 | 0.88(0.80–0.97) | 0.009 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Caucasian | 0.99(0.94–1.04) | 0.700 | 0.98(0.88–1.10) | 0.779 | 0.96 (0.89–1.05) | 0.361 | 1.01 (0.92–1.12) | 0.781 | 0.97 (0.90–1.05) | 0.426 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chinese | 0.81(0.73–0.89) | 0.004 | 0.71 (0.56-0.88) | 0.002 | 0.73 (0.57–0.93) | 0.011 | 0.82(0.66–1.02) | 0.072 | 0.72(0,59-0.90) | 0.004 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CC | 0.74(0.65-0.84) | <0.001 | 0.67(0.50-0.89) | 0.006 | 0.64 (0.53-076) | <0.001 | 0.83(0.63-1.09) | 0.175 | 0.64 (0.54–0.77) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BC | 0.98(0.93–1.03) | 0.406 | 0.96(0.87–1.07) | 0.481 | 0.96 (0.89–1.03) | 0.253 | 1.00 (0.91–1.09) | 0.958 | 0.96(0.89–1.03) | 0.244 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs6504074 | 0.96(0.85–1.09) | 0.556 | 1.04(0.89–1.23) | 0.614 | 0.94 (0.87–1.03) | 0.197 | 1.06 (0.91–1.25) | 0.457 | 0.92(0.79–1.08) | 0.298 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Caucasian | 1.09 (1.00-1.21) | 0.059 | 1.01(0.70–1.44) | 0.978 | 1.02 (0.92–1.12) | 0.766 | 1.01 (0.73–1.40) | 0.953 | 1.08(0.97–1.20) | 0.171 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chinese | 0.84(0.73–0.97) | 0.016 | 0.91(0.64–1.30) | 0.610 | 0.75(0.62–0.89) | 0.002 | 1.01(0.71-1.43) | 0.939 | 0.77(0.65–0.91) | 0.003 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OC | 1.02(0.87–1.20) | 0.787 | 0.86(0.65–1.13) | 0.269 | 0.95 (0.82–1.11) | 0.513 | 0.87(0.67–1.14) | 0.325 | 1.01 (0.83–1.24) | 0.911 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GC | 0.90(0.82-0.98) | 0.015 | 0.88(0.71-1.09) | 0.236 | 0.86(0.77-0.97) | 0.011 | 0.92(0.75-1.14) | 0.463 | 0.86(0.77-0.96) | 0.008 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations A: the major allele; B: the minor allele; CI: confidence interval; OR: odds ratio; GC: gynecologic cancer; BC: breast cancer;OC: ovarian cancer. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
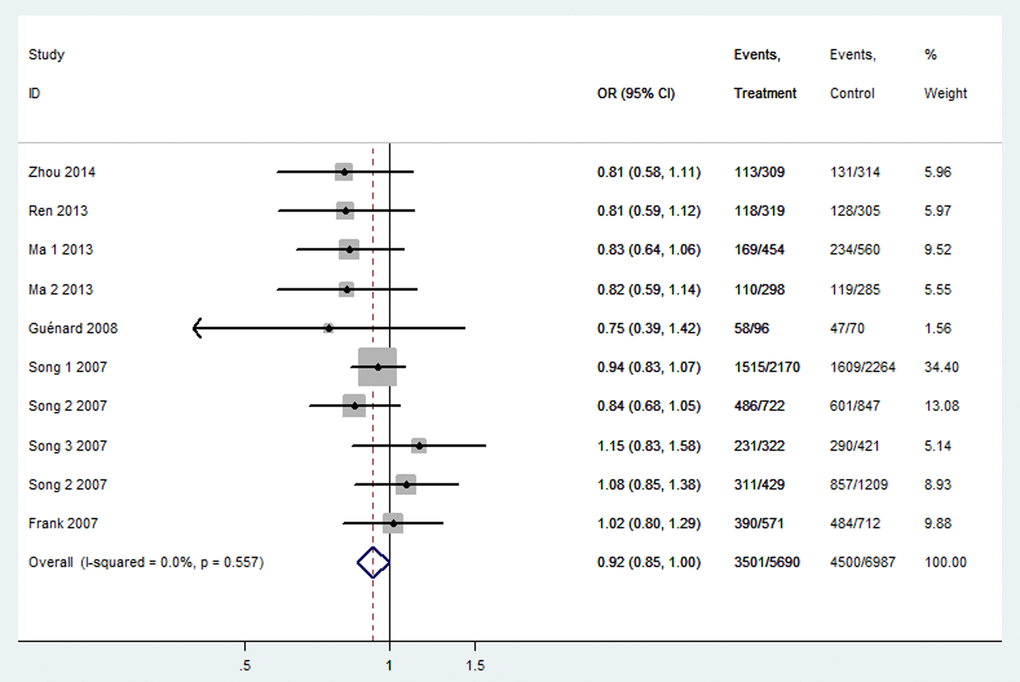
Figure 2. Forest plot of OR with 95%CI for the BRIP1 polymorphisms with cancer risk under dominant model rs2048718. CI: confidence interval, OR: odds ratio.
There were 7 studies containing 4,589 cancer cases and 5,419 cancer-free controls for rs4988344. As shown in Table 3, The pooled analysis displayed no association of any genetic models and overall cancer risk (All P > 0.05). In subgroup analysis by ethnicity, we detected a significant association among Chinese people in four genetic models (B vs. A: OR = 0.68, 95% CI = 0.54–0.86, P = 0.001; BB vs. AA: OR = 0.66, 95% CI = 0.54–0.86, P = 0.001; AB vs. AA: OR = 0.88, 95% CI = 0.54–0.86, P = 0.001; AB+BB vs. AA: OR = 0.78, 95% CI = 0.63–0.97, P < 0.001).
Thirteen studies covering 8,092 cases and 8,915 controls were pooled to evaluate the correlation of rs4986764 and cancer risk. Showing in Table 3 and Figure 3, rs4986764 was associated to reduce cancer risk among the overall population by allele comparison (OR = 0.94, 95% CI = 0.90–0.98, P = 0.001), heterozygous comparison (OR = 0.89, 95% CI = 0.80–0.99, P = 0.025), and dominant comparison (OR = 0.88, 95% CI = 0.80–0.97, P = 0.009). Stratified analysis by ethnicity also displayed significant differences in Chinese population (B vs. A: OR = 0.81, 95% CI = 0.73–0.89, P = 0.004; BB vs. AA: OR = 0.71, 95% CI = 0.56–0.88, P = 0.005; AB vs. AA: OR = 0.77, 95% CI = 0.57–0.93, P = 0.011; AB+BB vs. AA: OR = 0.72, 95% CI = 0.59–0.90, P = 0.004). However, there was no significant correlation found in Caucasians for all genetic models (all P > 0.05). In the stratified analysis by cancer types, all genetic models failed to detect significant correlations in breast cancer. However, a statistical significance suggested that rs4986764 polymorphism may decrease cervical cancer risk (B vs. A: OR = 0.74, 95% CI = 0.65–0.84, P < 0.001; BB vs. AA: OR = 0.67, 95% CI = 0.50–0.89, P = 0.006; AB vs. AA: OR = 0.64, 95% CI = 0.53–0.76, P < 0.001; AB+BB vs. AA: OR = 0.64, 95% CI = 0.54–0.77, P = 0.0001).
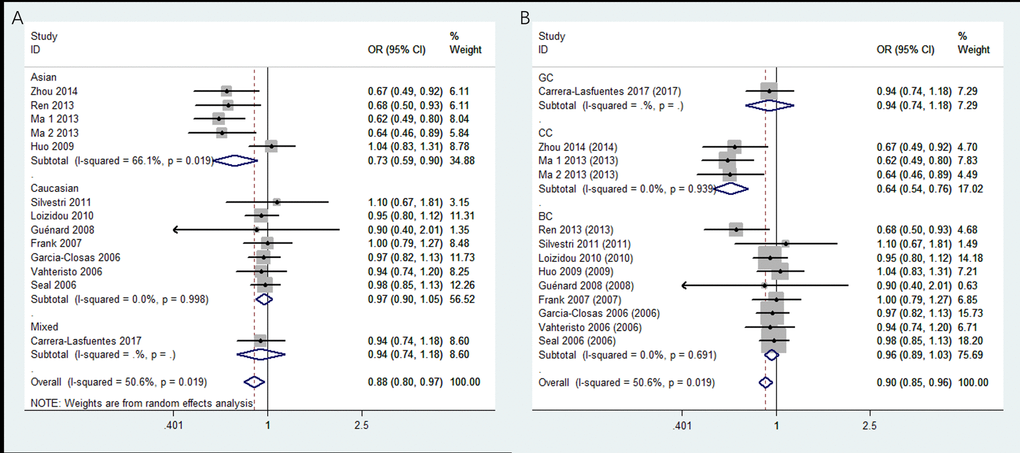
Figure 3. Stratified analysis based on ethnicity for the association between BRIP1 rs4986764 polymorphism and cancer risk using dominant model. (A) based on ethnicity; (B) based on cancer type. CI: confidence interval, OR: odds ratio.
A total of 7 studies for rs6504074 included 4,221 cases and 4,944 controls. As shown in Table 3, the overall analysis showed no association between rs6504074 between cancer risk in any genetic models (All P﹥0.05). But, further stratification analysis by ethnicity indicated that rs6504074 was significantly associated with a lower cancer risk among Chinese population by three models (B vs. A: OR = 0.84, 95% CI = 0.73–0.97, P = 0.016; AB vs. AA: OR = 0.75, 95% CI = 0.62–0.89, P = 0.002; BB + AB vs. AA: OR = 0.77, 95% CI = 0.65–0.91, P = 0.003). The subgroup analysis by cancer type of rs6504074 also showed a reduced risk for gynecologic cancers in allele model (OR = 0.90, 95% CI = 0.82–0.98, P = 0.015), heterozygous model (OR = 0.88, 95% CI = 0.77–0.97, P = 0.011), and dominant model (OR = 0.86, 95% CI = 0.77–0.96, P = 0.008).
Heterogeneity analysis and publication bias
The results of the heterogeneity test are displayed in Table 4. When the P value of the heterogeneity tests was less than 0.1 (P < 0.1), a random effects model was selected. Otherwise a fixed-effect model was applied.
Table 4. Heterogeneity-analysis results.
| Comparisons | B vs A | BB vs AA | AB vs AA | BB vs AA+AB | AB+BB vs AA | ||||||||||
| I2 | P | EM | I2 | P | EM | I2 | P | EM | I2 | P | EM | I2 | P | EM | |
| rs2048718 | 0.0% | 0.729 | F | 0.0% | 0.679 | F | 4.6% | 0.399 | F | 0.0% | 0.629 | F | 0.0% | 0.557 | F |
| Caucasian | 0.0% | 0.448 | F | 0.0% | 0.470 | F | 0.0% | 0.998 | F | 0.0% | 0.59 | F | 0.0% | 0.493 | F |
| Chinese | 0.0% | 1.000 | F | 0.0% | 0.990 | F | 0.0% | 0.572 | F | 0.0% | 0.991 | F | 0.0% | 0.999 | F |
| CC | 0.0% | 0.998 | F | 0.0% | 0.972 | F | 0% | 0.982 | F | 0.0% | 0.963 | F | 0.0% | 0.993 | F |
| BC | 0.0% | 0.805 | F | 0.0% | 0.682 | F | 0% | 0.443 | F | 0.0% | 0.525 | F | 0.0% | 0.641 | F |
| OC | 45.0% | 0.162 | F | 42.0% | 0.179 | F | 27% | 0.253 | F | 11.5% | 0.323 | F | 42.1% | 0.178 | F |
| rs4988344 | 55.5% | 0.036 | R | 52.9% | 0.047 | R | 55.0% | 0.038 | R | 5.6% | 0.384 | F | 63.3% | 0.430 | R |
| Caucasian | 27.7% | 0.246 | F | 15.8% | 0.312 | F | 6.7% | 0.359 | F | 11.7% | 0.334 | F | 89.9% | 0.000 | R |
| Chinese | 0.0% | 0.975 | F | 0.0% | 0.963 | F | 0.0% | 0.964 | F | 0.0% | 0.994 | F | 0.0% | 0.428 | F |
| BC | 55.6% | 0.134 | R | 59.0% | 0.118 | R | 67.5% | 0.080 | R | 0.0% | 0.512 | F | 74.8% | 0.046 | R |
| OC | 0.0% | 0.404 | F | 12.6% | 0.319 | F | 0.0% | 0.374 | F | 14.0% | 0.312 | F | 89.0% | 0.000 | R |
| rs4986764 | 45.9% | 0.036 | F | 6.8% | 0.378 | F | 52.8% | 0.013 | R | 12.2% | 0.323 | F | 50.6% | 0.019 | R |
| Caucasian | 0.0% | 0.980 | F | 0.0% | 0.980 | F | 0.0% | 0.991 | F | 0.0% | 0.887 | F | 0.0% | 0.998 | F |
| Chinese | 37.5% | 0.171 | F | 0.0% | 0.974 | F | 70.5% | 0.009 | R | 0.0% | 0.989 | F | 66.1% | 0.019 | R |
| BC | 0.0% | 0.787 | F | 0.0% | 0.939 | F | 0.0% | 0.589 | F | 0.0% | 0.857 | F | 0.0% | 0.691 | F |
| CC | 0.0% | 0.912 | F | 0.0% | 0.932 | F | 0.0% | 0.964 | F | 0.0% | 0.949 | F | 0.0% | 0.939 | F |
| rs6504074 | 52.4% | 0.062 | R | 39.2% | 0.130 | F | 41.2% | 0.116 | F | 26.2% | 0.229 | F | 55.4% | 0.047 | R |
| Caucasian | 0.0% | 0.967 | F | 66.7% | 0.029 | R | 0.0% | 0.645 | F | 62.0% | 0.048 | R | 0.0% | 0.740 | F |
| Chinese | 0.0% | 0.567 | F | 0.0% | 0.936 | F | 0.0% | 0.987 | F | 0.0% | 0.935 | F | 0.0% | 0.984 | F |
| OC | 0.0% | 0.551 | F | 48.8% | 0.142 | F | 0.0% | 0.840 | F | 45.6% | 0.159 | F | 0.0% | 0.794 | F |
| GC | 0.0% | 0.527 | F | 0.0% | 0.534 | F | 0.0% | 0.486 | F | 0.0% | 0.516 | F | 0.0% | 0.535 | F |
In this meta-analysis, we used funnel plots and Egger’s test to estimate publication bias. The funnel plots (Figure 4) failed to discover any prominent asymmetry for the four polymorphisms, which was consistent with the results of Egger’s test (P > 0.05). Therefore, we considered that the publication bias in this meta-analysis was not significant.
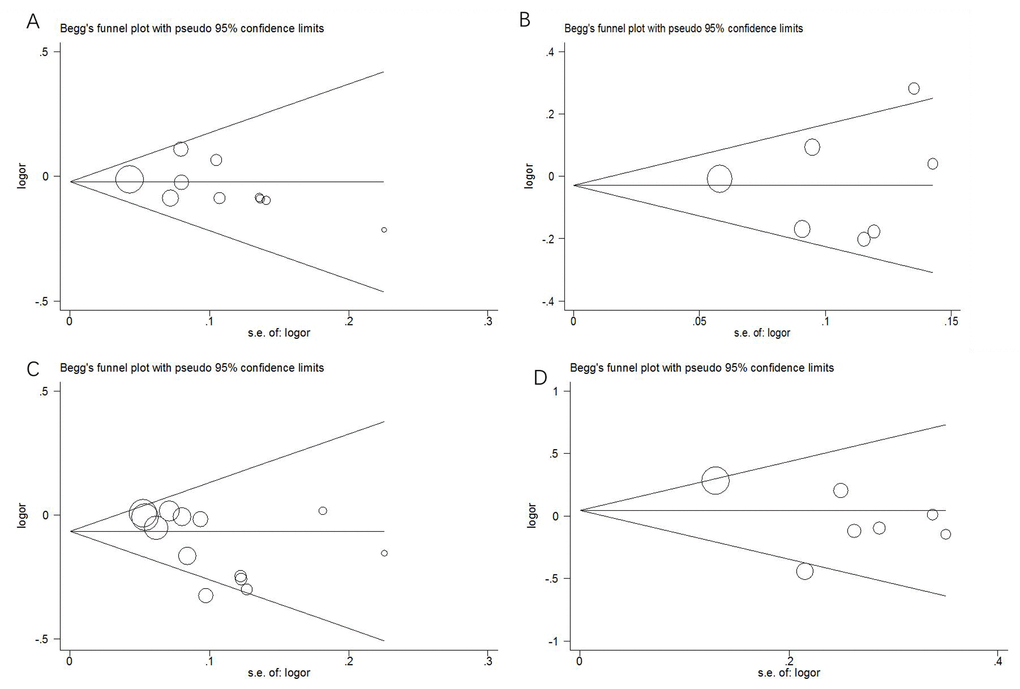
Figure 4. Funnel plots of publication bias. (A) rs2048718; (B) rs4988344; (C) rs4986764; (D) rs6504074.
Sensitivity analysis
The sensitivity analysis was used in rs4986764 showed no individual research could alter the pooled ORs significantly (Figure 5), which proved the reliability and credibility of the outcomes.
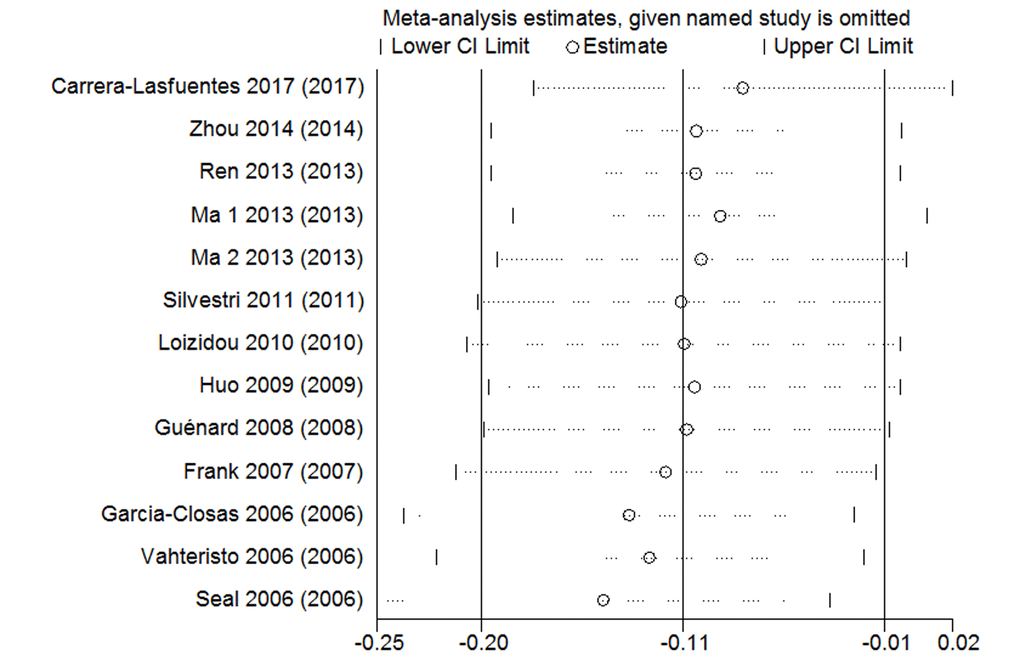
Figure 5. Sensitivity analysis of the associations between rs4986764 polymorphisms and cancer risk.
Discussion
As a DNA helicase interacts with BRCA1 directly, BRIP1 regulates DNA normal double-strand break repair function [1,4]. Germline BRIP1 mutations, which negatively affect DNA repair and genomic stability, and thus increase the likelihood of cancer development, have been reported to be connected with breast, colon, prostate and ovarian cancer [4,8,12,13]. Recent studies showed that SNPs in genes had an influence on multiple types of cancer [4-6,14]. Numerous previous studies have suggested that BRIP1 polymorphisms were potentially related to susceptibility of human cancers, especially breast, cervical, and ovarian cancer. However, these published studies reported inconsistent results [7], probably due to the limitations, such as small sample size, mixed ethnic groups, and cancer types. To our knowledge, there are few researches have simultaneously estimated the relationship between common variants in BRIP1 and the risks of common cancers. With a sample size of 13,716, our meta-analysis showed the overall associations between common polymorphisms of BRIP1 (rs2048718, rs4988344, rs4986764, and rs6504074) and cancer risk. The pooled analysis found significant association with rs2048718 and rs4986764. And, we found all these 4 SNPs predicted a decreased risk of cancer in Chinese. Moreover, rs6504074 showed an association with gynecologic cancer. And, rs2048718 and rs4986764 polymorphisms showed a decreased risk of cervical cancer.
Compared to other published meta-analyses, we found both consistent and conflicting results. Based on the results of 2 studies in USA and Poland using mouthwash samples, rs4986764 (P919S) had no association with breast cancer [15]. In another meta-analysis, Shi et al reported that rs4986764 may reduce the breast cancer risk for the Caucasian population, especially postmenopausal females who has a family history of breast cancer excluding mutations of BRCA1/2 [16]. However, no association was found in Pabalan’s study [17]. Unfortunately, their meta-analysis failed to analyze the data according to ethnic subgroup. Previous studies failed to find a relation between rs2048718 and rs4988344 polymorphisms and the susceptibility of cervical cancer [5,6,14]. Our study showed that rs4988344 polymorphism has no significant influence on cancer risk among overall population, while subgroup analysis by ethnicity showed a significant association in the Chinese population (Table 3). The most important polymorphisms of BRIP1 (rs2048718, rs4988344, rs4986764, and rs6504074) were associated with cancer risk among Chinese; however, no significant relationship was observed in Caucasians. We presumed that this discrepancy may have resulted from variations in the genomes of different ethnic groups. These results suggest that ethnic differences and the patient’s environment may play a role in malignancy. On account of the findings of these studies, we estimated the possible association between the 4 SNPs of BRIP1 and cancer risk. However, in vivo, BRIP1 might be regulated by multiple mechanisms [1,18,19]. We speculated that discrepancy for BRIP1 in different ethnic groups stems from a diversity regulating mechanism as well.
As to the limitations of this meta-analysis, absence of detailed information and adjusted outcomes should be acknowledged. We failed to considered detailed information like age, sex, lifestyle, and environmental exposures. Besides, we found a decreased risk of rs2048718 and rs4986764 polymorphisms to cervical cancer based on three studies, which needs further well-design multicenter studies including more study subjects to confirm.
Conclusions
Overall, this meta-analysis showed that rs2048718 and rs4986764 were associated with a lower cancer risk among overall population. According to the stratified analysis by ethnicity, the rs2048718, rs4988344, rs4986764 and rs6504074 polymorphisms of BRIP1 were strongly related to cancer susceptibility among Chinese population. And rs6504074 was significant associated with gynecologic cancer. These may made SNPs of BRIP1 (rs2048718, rs4988344, rs4986764, and rs6504074) be a potential tool for cancer screening and improve early cancer diagnosis.
Materials and Methods
Search strategy
A profound literature search from PubMed, Web of Science, WanFang, VIP and Chinese National Knowledge Infrastructure (CNKI) databases was conducted up until December 31, 2017, applying the search terms: cancer/tumor/carcinoma/neoplasm, BRIP1, and polymorphism/genotype /SNP. The reference of literature review and eligible articles were also screened for additional relevant publication. Studies conformed to the following criteria were selected: (1) case-control design estimating the relationship of BRIPI SNPs and cancer risk; (2) full-text study; (3) all cancer cases confirmed by histopathology, and all cancer-free controls without history of malignant diseases; (4) published in English or Chinese; (5) detailed genotyping data offered. Review papers, not case-control design or studies lack of detailed gene data were excluded. If overlapping cases or controls appeared in two or more different studies, the paper with larger sample size was finally chosen.
Data extraction
Two reviewers (Liu Di and Wang Meng) reviewed included articles independently. The following information was collected from each included publication: first author, publication year, country or origin, ethnicity, source of control, total number of cases and controls, genotyping methods, genetic distribution of cases and controls group, and P value of Hardy–Weinberg equilibrium (HWE) for controls. Ethnic groups were categorized as Caucasian, Asian, African, and “mixed.” All case and control groups were well controlled. Data with discrepancies were discussed with a senior author until consensus reached.
Statistical analysis
To measure the associations between BRIP1 polymorphisms and cancer risk, odds ratio (OR) with 95% confidence interval (CI) was calculated according to the genotypes in cases and controls. The significance of the pooled OR was determined by the Z test. All P values in this study were two-sided, and a statistic significance was considered if P < 0.05. All statistical analyses in our investigation were performed by the software STATA (Version 12.0, Stata Corp, College Station, TX).
The meta-analysis assessed the associations using 5 different genetic models [10,11]: homozygous model (BB vs. AA), heterozygous model (AB vs. AA), dominant model (BB+ AB vs. AA), recessive model (BB vs. AA+ AB), and allele model (B vs. A). “A”, “B” represents the major, minor allele, respectively. Statistical heterogeneity among included studies was evaluated by the Q and I2 statistics. Publication bias was accessed with funnel plots and Egger’s test. Sensitivity analysis was conducted to access the statistic stability of polymorphisms including more than 10 studies, by sequentially excluding every individual research and re-checked whether the pooled ORs were changed.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Funding
Our study was supported by National Natural Science Foundation of China (No. 81471670); the Science and Technology Project of Shaanxi Province(No. 2017SF-172); the Key research and development plan, Shaanxi Province, People’s Republic of China (2017ZDXM-SF-066); the International Cooperative Project of Shaanxi province, China (No.2016KW-008); the Fundamental Research Funds for the Central Universities, China(No.2014gjhz11); BIKANG Funding (2017BIKANGJIJIN-020); the Funds of the Second Affiliated Hospital of Xi’an Jiaotong University for Young Scientists, China [YJ(QN)201305].
References
- 1. Cantor SB, Guillemette S. Hereditary breast cancer and the BRCA1-associated FANCJ/BACH1/BRIP1. Future Oncol. 2011; 7:253–61. https://doi.org/10.2217/fon.10.191 [PubMed]
- 2. Levran O, Attwooll C, Henry RT, Milton KL, Neveling K, Rio P, Batish SD, Kalb R, Velleuer E, Barral S, Ott J, Petrini J, Schindler D, et al. The BRCA1-interacting helicase BRIP1 is deficient in Fanconi anemia. Nat Genet. 2005; 37:931–33. https://doi.org/10.1038/ng1624 [PubMed]
- 3. Walsh T, King MC. Ten genes for inherited breast cancer. Cancer Cell. 2007; 11:103–05. https://doi.org/10.1016/j.ccr.2007.01.010 [PubMed]
- 4. Song H, Ramus SJ, Kjaer SK, Hogdall E, Dicioccio RA, Whittemore AS, McGuire V, Hogdall C, Jacobs IJ, Easton DF, Ponder BA, Dunning AM, Gayther SA, Pharoah PD. Tagging single nucleotide polymorphisms in the BRIP1 gene and susceptibility to breast and ovarian cancer. PLoS One. 2007; 2:e268. https://doi.org/10.1371/journal.pone.0000268 [PubMed]
- 5. Ma XD, Cai GQ, Zou W, Huang YH, Zhang JR, Wang DT, Chen BL. First evidence for the contribution of the genetic variations of BRCA1-interacting protein 1 (BRIP1) to the genetic susceptibility of cervical cancer. Gene. 2013; 524:208–13. https://doi.org/10.1016/j.gene.2013.04.025 [PubMed]
- 6. Ma XD, Cai GQ, Zou W, Huang YH, Zhang JR, Wang DT, Chen BL. BRIP1 variations analysis reveals their relative importance as genetic susceptibility factor for cervical cancer. Biochem Biophys Res Commun. 2013; 433:232–36. https://doi.org/10.1016/j.bbrc.2013.02.089 [PubMed]
- 7. Cao AY, Huang J, Hu Z, Li WF, Ma ZL, Tang LL, Zhang B, Su FX, Zhou J, Di GH, Shen KW, Wu J, Lu JS, et al. Mutation analysis of BRIP1/BACH1 in BRCA1/BRCA2 negative Chinese women with early onset breast cancer or affected relatives. Breast Cancer Res Treat. 2009; 115:51–55. https://doi.org/10.1007/s10549-008-0052-z [PubMed]
- 8. Rafnar T, Gudbjartsson DF, Sulem P, Jonasdottir A, Sigurdsson A, Jonasdottir A, Besenbacher S, Lundin P, Stacey SN, Gudmundsson J, Magnusson OT, le Roux L, Orlygsdottir G, et al. Mutations in BRIP1 confer high risk of ovarian cancer. Nat Genet. 2011; 43:1104–07. https://doi.org/10.1038/ng.955 [PubMed]
- 9. Kote-Jarai Z, Jugurnauth S, Mulholland S, Leongamornlert DA, Guy M, Edwards S, Tymrakiewitcz M, O’Brien L, Hall A, Wilkinson R, Al Olama AA, Morrison J, Muir K, et al, and UKGPCS Collaborators, and British Association of Urological Surgeons’ Section of Oncology. A recurrent truncating germline mutation in the BRIP1/FANCJ gene and susceptibility to prostate cancer. Br J Cancer. 2009; 100:426–30. https://doi.org/10.1038/sj.bjc.6604847 [PubMed]
- 10. Li SX, Dai QS, Chen SX, Zhang SD, Liao XY, Deng X, Chi HB, Li FJ, Zhu JH, Jiang YY. Xeroderma pigmentosum complementation group D (XPD) gene polymorphisms contribute to bladder cancer risk: a meta-analysis. Tumour Biol. 2014; 35:3905–15. https://doi.org/10.1007/s13277-013-1519-z [PubMed]
- 11. Gao J, Kang AJ, Lin S, Dai ZJ, Zhang SQ, Liu D, Zhao Y, Yang PT, Wang M, Wang XJ. Association between MDM2 rs 2279744 polymorphism and breast cancer susceptibility: a meta-analysis based on 9,788 cases and 11,195 controls. Ther Clin Risk Manag. 2014; 10:269–77. https://doi.org/10.2147/TCRM.S60680 [PubMed]
- 12. Levitus M, Waisfisz Q, Godthelp BC, de Vries Y, Hussain S, Wiegant WW, Elghalbzouri-Maghrani E, Steltenpool J, Rooimans MA, Pals G, Arwert F, Mathew CG, Zdzienicka MZ, et al. The DNA helicase BRIP1 is defective in Fanconi anemia complementation group J. Nat Genet. 2005; 37:934–35. https://doi.org/10.1038/ng1625 [PubMed]
- 13. Narayan G, Arias-Pulido H, Nandula SV, Basso K, Sugirtharaj DD, Vargas H, Mansukhani M, Villella J, Meyer L, Schneider A, Gissmann L, Dürst M, Pothuri B, Murty VV. Promoter hypermethylation of FANCF: disruption of Fanconi Anemia-BRCA pathway in cervical cancer. Cancer Res. 2004; 64:2994–97. https://doi.org/10.1158/0008-5472.CAN-04-0245 [PubMed]
- 14. Ren LP, Xian YS, Diao DM, Chen Y, Guo Q, Dang CX. Further evidence for the contribution of the BRCA1-interacting protein-terminal helicase 1 (BRIP1) gene in breast cancer susceptibility. Genet Mol Res. 2013; 12:5793–801. https://doi.org/10.4238/2013.November.22.6 [PubMed]
- 15. García-Closas M, Egan KM, Newcomb PA, Brinton LA, Titus-Ernstoff L, Chanock S, Welch R, Lissowska J, Peplonska B, Szeszenia-Dabrowska N, Zatonski W, Bardin-Mikolajczak A, Struewing JP. Polymorphisms in DNA double-strand break repair genes and risk of breast cancer: two population-based studies in USA and Poland, and meta-analyses. Hum Genet. 2006; 119:376–88. https://doi.org/10.1007/s00439-006-0135-z [PubMed]
- 16. Shi J, Tong J, Cai S, Qu X, Liu Y. Correlation of the BACH1 Pro919Ser polymorphism with breast cancer risk: A literature-based meta-analysis and meta-regression analysis. Exp Ther Med. 2013; 6:435–44. https://doi.org/10.3892/etm.2013.1148 [PubMed]
- 17. Pabalan N, Jarjanazi H, Ozcelik H. Association between BRIP1 (BACH1) polymorphisms and breast cancer risk: a meta-analysis. Breast Cancer Res Treat. 2013; 137:553–58. https://doi.org/10.1007/s10549-012-2364-2 [PubMed]
- 18. Cantor SB, Bell DW, Ganesan S, Kass EM, Drapkin R, Grossman S, Wahrer DC, Sgroi DC, Lane WS, Haber DA, Livingston DM. BACH1, a novel helicase-like protein, interacts directly with BRCA1 and contributes to its DNA repair function. Cell. 2001; 105:149–60. https://doi.org/10.1016/S0092-8674(01)00304-X [PubMed]
- 19. Litman R, Peng M, Jin Z, Zhang F, Zhang J, Powell S, Andreassen PR, Cantor SB. BACH1 is critical for homologous recombination and appears to be the Fanconi anemia gene product FANCJ. Cancer Cell. 2005; 8:255–65. https://doi.org/10.1016/j.ccr.2005.08.004 [PubMed]
- 20. Carrera-Lasfuentes P, Lanas A, Bujanda L, Strunk M, Quintero E, Santolaria S, Benito R, Sopeña F, Piazuelo E, Thomson C, Pérez-Aisa A, Nicolás-Pérez D, Hijona E, et al. Relevance of DNA repair gene polymorphisms to gastric cancer risk and phenotype. Oncotarget. 2017; 8:35848–62. https://doi.org/10.18632/oncotarget.16261 [PubMed]
- 21. Zhou S, Chen L, Zhou H. Association of the single nucleotide polymorphism in functional region of BRCA1 -interacting protein 1 (BRIP1) with cervical cancer. Zhejiang JITCWM. 2014; 24:960–63.
- 22. Silvestri V, Rizzolo P, Falchetti M, Zanna I, Masala G, Bianchi S, Palli D, Ottini L. Mutation analysis of BRIP1 in male breast cancer cases: a population-based study in Central Italy. Breast Cancer Res Treat. 2011; 126:539–43. https://doi.org/10.1007/s10549-010-1289-x [PubMed]
- 23. Loizidou MA, Cariolou MA, Neuhausen SL, Newbold RF, Bashiardes E, Marcou Y, Michael T, Daniel M, Kakouri E, Papadopoulos P, Malas S, Hadjisavvas A, Kyriacou K. Genetic variation in genes interacting with BRCA1/2 and risk of breast cancer in the Cypriot population. Breast Cancer Res Treat. 2010; 121:147–56. https://doi.org/10.1007/s10549-009-0518-7 [PubMed]
- 24. Huo X, Lu C, Huang X, Hu Z, Jin G, Ma H, Wang X, Qin J, Wang X, Shen H, Tang J. Polymorphisms in BRCA1, BRCA1-interacting genes and susceptibility of breast cancer in Chinese women. J Cancer Res Clin Oncol. 2009; 135:1569–75. https://doi.org/10.1007/s00432-009-0604-6 [PubMed]
- 25. Guénard F, Labrie Y, Ouellette G, Joly Beauparlant C, Simard J, Durocher F, and INHERIT BRCAs. Mutational analysis of the breast cancer susceptibility gene BRIP1 /BACH1/FANCJ in high-risk non-BRCA1/BRCA2 breast cancer families. J Hum Genet. 2008; 53:579–91. https://doi.org/10.1007/s10038-008-0285-z [PubMed]
- 26. Frank B, Hemminki K, Meindl A, Wappenschmidt B, Sutter C, Kiechle M, Bugert P, Schmutzler RK, Bartram CR, Burwinkel B. BRIP1 (BACH1) variants and familial breast cancer risk: a case-control study. BMC Cancer. 2007; 7:83. https://doi.org/10.1186/1471-2407-7-83 [PubMed]
- 27. Vahteristo P, Yliannala K, Tamminen A, Eerola H, Blomqvist C, Nevanlinna H. BACH1 Ser919Pro variant and breast cancer risk. BMC Cancer. 2006; 6:19. https://doi.org/10.1186/1471-2407-6-19 [PubMed]
- 28. Seal S, Thompson D, Renwick A, Elliott A, Kelly P, Barfoot R, Chagtai T, Jayatilake H, Ahmed M, Spanova K, North B, McGuffog L, Evans DG, et al, and Breast Cancer Susceptibility Collaboration (UK). Truncating mutations in the Fanconi anemia J gene BRIP1 are low-penetrance breast cancer susceptibility alleles. Nat Genet. 2006; 38:1239–41. https://doi.org/10.1038/ng1902 [PubMed]



