Introduction
Aging, including cardiac aging, is a natural and multifactorial process characterized by a series of mechanisms, including deregulated autophagy, oxidative stress, systemic inflammation and mitochondrial dysfunction [1–9]. Recent studies have emphasized the importance of NAD+-dependent deacetylase sirtuins, mainly cytosolic- or nucleus-localized Sirt1 and mitochondrial-localized Sirt3, in the aging process [10]. Decreased sirtuin expression has been found in aged humans and animals [11,12]. Additionally, recent reports have suggested a significant reduction in the cellular NAD+ concentration and sirtuin activity in aged animals [11]. Furthermore, AMPK, a key regulator of cellular metabolism that significantly contributes to sirtuin activity, e.g., via increasing NAD+ synthesis [13], showed declined activity associated with aging [14].
The decline of Sirt1 and Sirt3 expression or activity may result in the following two detrimental consequences: first, it may impair mitochondrial biogenesis [15–17] and function [18,19], and second, it may lead to excessive inflammatory response, particularly due to the reduced anti-inflammatory actions of Sirt1 [20,21]. Several reports suggest that Sirt3 ameliorates mitochondrial stress by upregulating mitophagy and anti-oxidant machinery proteins, including manganese superoxide dismutase (SOD2) and catalase [22,23].
Both mitochondrial dysfunction and chronic inflammation are well-known hallmarks of aging [1,24,25]. Furthermore, in both cases, elevated ROS formation, another aging hallmark [26], is to be expected. Though the age-associated reduction in sirtuin expression and activity as well as its contribution to mitochondrial dysfunction and pro-inflammatory shift are well-documented, the underlying cellular mechanisms are far from understood.
Recent studies applying in vivo and in vitro models have suggested a role for the female sex hormone estradiol (E2) in the expression and activity of AMPK and sirtuins [27,28]. Apart from being the downstream target of AMPK, Sirt1 deacetylates the upstream activator of AMPK, LKB1 kinase, thus providing the positive feedback loop between AMPK and Sirt1 [29,30]. Of note, E2 blood concentrations are reduced in aged women, i.e., after menopause. In fact, postmenopausal may women have lower E2 concentrations in their blood compared with age-matched men [31]. The loss of E2 is accompanied by the release of pro-inflammatory cytokines, leading to activation of inflammatory pathways in aging [32]. Furthermore, loss of ovarian hormones due to reproductive aging leads to the decline in anti-oxidative defense, mitochondrial biogenesis and function in females [33,34].
Though several reports have demonstrated sex differences in the expression of AMPK and sirtuins in mouse brain and kidney [35], alterations in the human heart in aging remain unknown. Therefore, in the present study, we aimed to investigate the age-related alterations in Sirt1, AMPK and Sirt3 signaling in cardiac tissue from men and women along with markers for mitochondrial biogenesis, anti-oxidative defense and the inflammatory state. We found a female sex-specific downregulation of Sirt1 and Sirt3 expression in aged hearts, which is accompanied by the downregulation of SOD2, a key mitochondrial anti-oxidative enzyme, and by increased expression of inflammatory mediators.
Results
Discussion
The aim of the study was to examine age-related changes in Sirt1, AMPK and Sirt3 expression in women and men in relation to mitochondrial biogenesis, anti-oxidative defense and inflammation in human hearts. The main findings are as follows: (i) Aging leads to a significant downregulation in Sirt1 expression and the corresponding elevated acetylation of nuclear proteins in female but not male hearts; (ii) The higher expression of some mitochondrial and anti-oxidative proteins in young females is lost with aging either due to their downregulation in aged females, e.g., Sirt3 and SOD2, or due to upregulation in aged males (TOM40, SOD2 and catalase); and (iii) Aging leads to a significant pro-inflammatory shift in female but not male hearts.
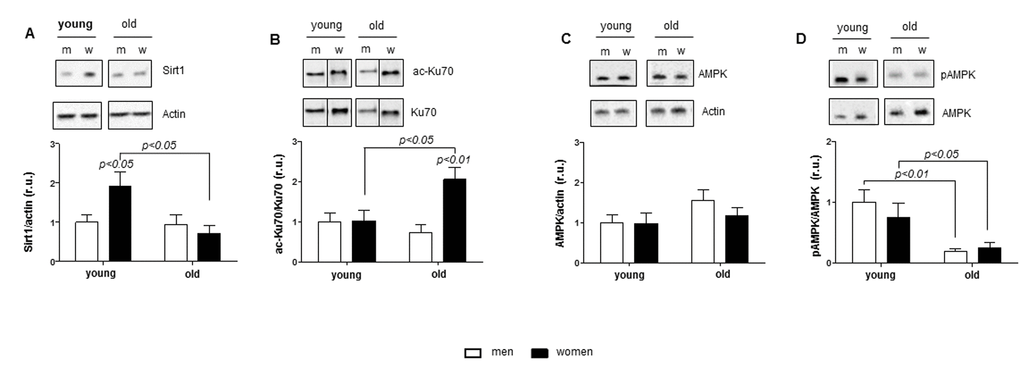
AMPK and Sirt1 are partner proteins orchestrating a wide variety of intracellular processes including cellular resistance to oxidative stress, general metabolism, inflammation, and mitochondrial biogenesis and function [42]. Aging is accompanied by the downregulation of AMPK and Sirt1 activity in animals and humans [43]. Whether these alterations are sex-dependent remains unknown. We found a sex-independent downregulation of the AMPK phosphorylation rate, a widely used indicator of AMPK activity. Furthermore, the female-specific downregulation of Sirt1 expression was observed in aged hearts. Consistently, acetylation of Ku70, a nuclear protein and a direct Sirt1 client, was significantly increased only in old female but not male hearts, demonstrating reduced Sirt1 activity. Although we did not investigate whether the nuclear or cytosolic Sirt1 content is specifically reduced with aging in female hearts, previous reports demonstrated that Sirt1 physically interacts with and deacetylates nuclear Ku70 [44].
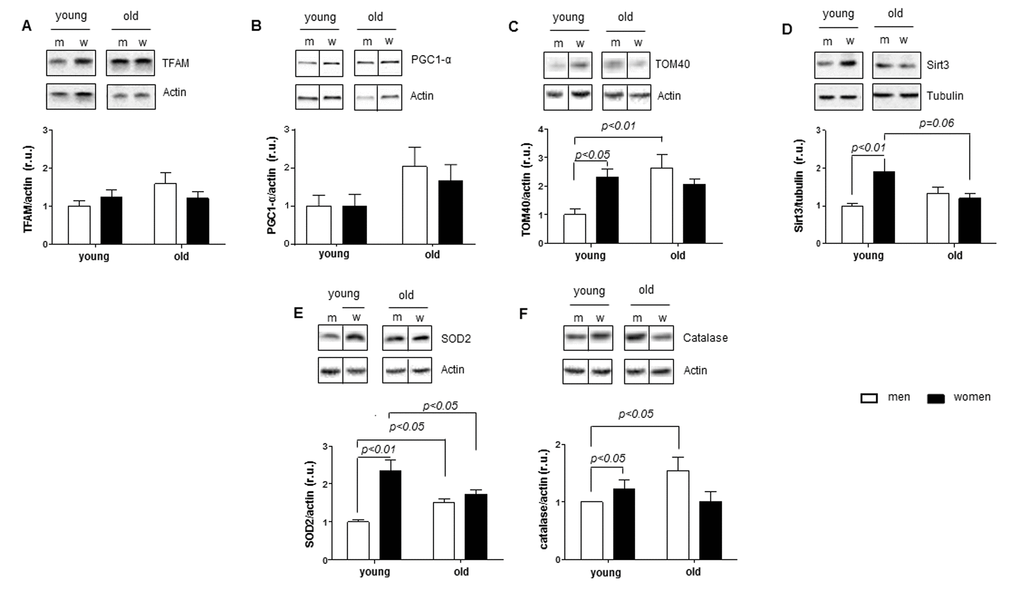
Mitochondrial dysfunction is a characteristic fingerprint of aging [45]. Notably, downregulation of Sirt1 activity has been previously attributed to disturbances in mitochondrial biogenesis [19]. To examine the age-related effects on mitochondrial biogenesis in male and female hearts, expression analysis of key transcription factors (PGC1alpha and TFAM) as well as several nuclear- and mitochondria-encoded mitochondrial proteins was performed by western blot and PCR. Surprisingly, we found no sex- or age-related differences in the expression of mitochondrial genes or PGC1alpha and TFAM expression, which excludes the sex-dependent regulation of mitochondrial biogenesis in aging human hearts, even though such changes were reported in lower organisms or in the liver and kidneys from 22-month-old mice. Species and organ differences may account for these discrepancies [35,46].
Previous reports argue for an activating effect of Sirt1-dependent deacetylation on PGC1alpha [47,48]. It is tempting to speculate that female-specific Sirt1 downregulation and enhanced acetylation of nuclear proteins (e.g., Ku70) may also be accompanied by PGC1alpha hyperacetylation and inactivation. Indeed, considering that PGC1alpha directly interacts with the SOD2 promoter regions and regulates its expression [49], the observed downregulation of SOD2 in aged female but not male hearts argues for a potential reduction in PGC1alpha activity.
Several studies have demonstrated favorable expression of anti-oxidative enzymes in female vs. male hearts, which is lost in postmenopausal women [50]. In agreement with these findings, the expression of two key anti-oxidative enzymes, i.e., SOD2 and catalase, in our study was significantly higher in young female vs. male hearts, whereas this sex difference is lost in aged hearts. Interestingly, this age-related alteration in the sex difference in the anti-oxidative enzymes is due to their opposite regulation in female and male hearts. Indeed, SOD2 expression in aged female hearts was downregulated, whereas expression of both enzymes was upregulated in aged male hearts. This finding leads to an intriguing idea that there are mechanisms activated in aged male hearts leading to improved anti-oxidative defense. Though the underlying cellular mechanisms upregulating the anti-oxidative enzymes SOD2 and catalase in aged male hearts still have to be elucidated, previous reports demonstrated an elevation in blood estrogen concentrations in males with age [31]. Given that estrogen upregulates anti-oxidative enzymes including SOD2 [51,52], one may assume that the known age-related changes in the estrogen concentration in blood from males (increase) and females (decrease) may contribute to the sex difference in SOD2 and catalase expression observed in our study.
Similar to SOD2, a sex-dependent alteration in the expression of Sirt3, a major regulator of the mitochondrial acetylome, was observed in our study. Global analysis of the mitochondrial protein acetylome performed by Herbert et al. [53] revealed a several-fold increase in the acetylation of multiple lysine residues in the liver of Sirt3-deficient mice. The net contribution of Sirt3 activity comprises the regulation of mitochondrial dynamics [54] and function [55], i.e., OXPHOS activity, ATP synthesis and fatty acid oxidation. Therefore, downregulation of Sirt3 expression in aged female hearts may lead to increased mitochondrial protein acetylation of i.e. SOD2 and disturbed mitochondrial functions, which may further exacerbate the reduced anti-oxidative defense in the mitochondria, i.e., reduced SOD2 expression.
Aside from the transcriptional regulation of Sirt3 expression, a recent study by Kwon et al. [12] suggested an unexpected mechanism for the post-translational regulation of Sirt3 activity and stability via Sirt1-mediated deacetylation. Indeed, the authors found the presence of Sirt1 in the mitochondria and its interaction with Sirt3. The Sirt1-driven deacetylation of Sirt3 significantly increases its stability and enzymatic activity. Of note, the authors observed a hyperacetylation, and therefore instability of Sirt3 in aged mice. Based on our finding, i.e., reduced Sirt1 and Sirt3 expression in aged female hearts, one may suppose a female-specific downregulation of this novel Sirt1-Sirt3 axis. Altogether, the current study provides convincing evidence of female sex-specific downregulation of Sirt1 and Sirt3 accompanied by disturbed expression in some mitochondrial proteins and anti-oxidative defense.
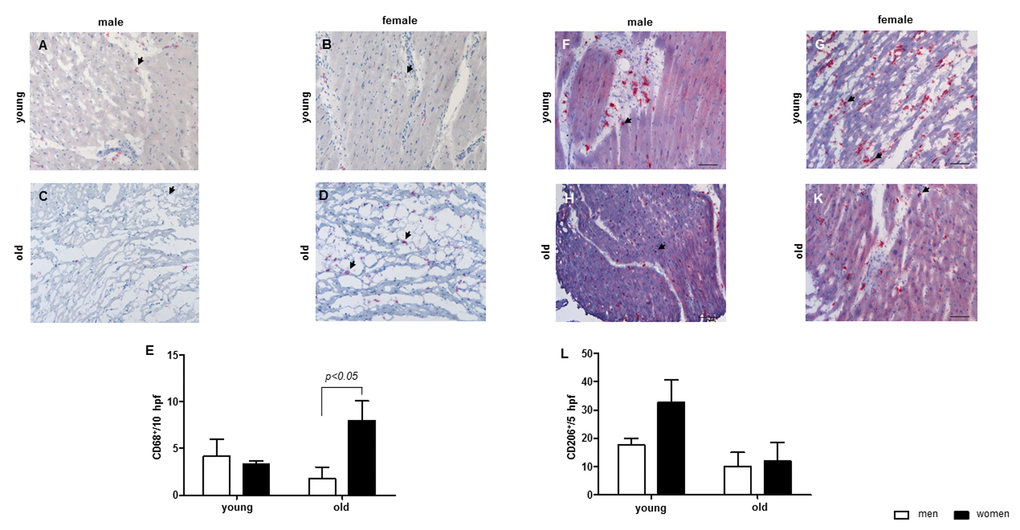
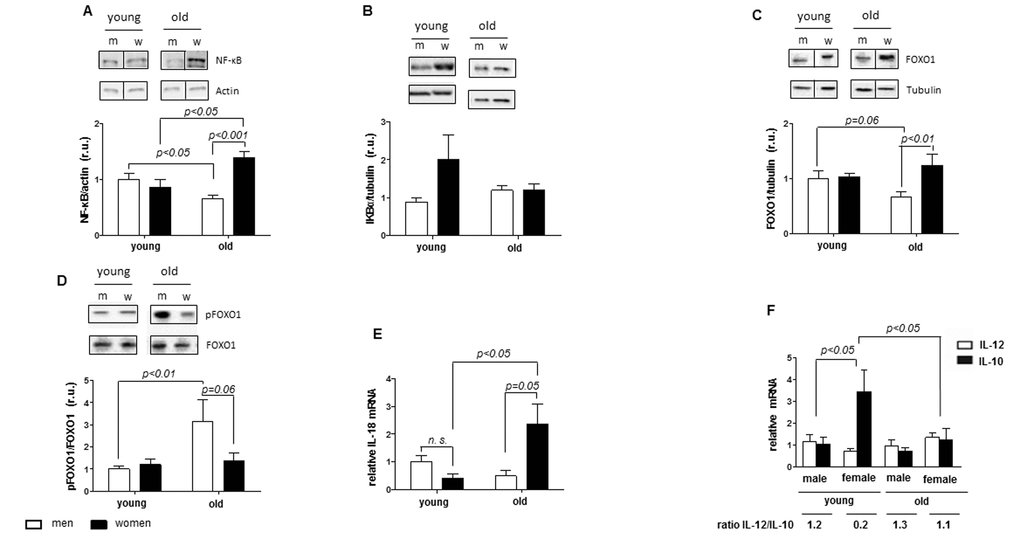
In addition to regulating mitochondrial function and biogenesis, emerging data also suggest the role of Sirt1 in acute and chronic inflammatory response, e.g., via inhibition of transcription factor NF-kB [20,21]. Since, chronic inflammation is a characteristic feature of aging [40,56], we were wondering whether female sex-specific Sirt1 downregulation may be accompanied by enhanced inflammation in aging female hearts. Indeed, the present study revealed a significant pro-inflammatory shift in aged female but not male hearts demonstrated by an increase in the cardiac macrophage content as well as upregulation of NF-kB, IL-12 and IL-18 expression, specifically in hearts from old women. In contrast, the anti-inflammatory protection was significantly decreased in women with age, which was confirmed by the elevation of the IL-12/IL-10 ratio. Similar increase in the IL-12/IL-10 ratio in the brain of old female mice has been found by Zhang et al. [57]. Interestingly, it seems that NF-κB is only involved in the inflammatory response in female but not involved in male hearts based on the data of NF- κB, IKBα and the pro-inflammatory cytokines.
In agreement with pro-inflammatory shift in heart of aged women, an increased number of CD68-positive macrophages along with increased pro-inflammatory cytokine expression was found. However, the macrophages found in the cardiac tissue from young women seem to be anti-inflammatory macrophages, since these macrophages were CD206 positive. A shift in the IL-12/IL-10 ratio to the site of IL-10 might indicate that the cardiac macrophages in the cardiac tissue of young women are regulatory macrophages, since regulatory macrophages produce high amounts of IL-10 and low amounts of IL-12 []64]. This shift is lost in aged women. Altogether, the findings from this study argue for a female sex-specific pro-inflammatory shift in aged hearts. Although the underlying mechanisms of this shift remain unclear, the female sex-specific downregulation of Sirt1 and SOD2 expression suggest a potential contribution.
Sexual hormones modulate the immune system via hormone receptors and regulate inflammation [58]. Although both the pro- and anti-inflammatory actions of estrogens have been described [59], the majority of studies argue for the anti-inflammatory effects of estrogen receptor activation. Particularly, E2 exhibits anti-inflammatory actions on endothelial and immune cells in vitro [60,61]. E2 loss leads to the expression of pro-inflammatory cytokines in humans [32]. In human activated peripheral blood mononuclear cells (PBMCs), E2 inhibits the expression of pro-inflammatory cytokines [62] and decreases NF-κB activity [63].
One limitation of the study is that our cohorts are relatively small and the human material archived high heterogeneity. Due to the small sample number, the statistical significance might not accurately show age or sex differences.
In conclusion, the present study revealed a female sex-specific downregulation of Sirt1 and Sirt3 in aged human hearts accompanied by a decline in the mitochondrial anti-oxidative defense and a pro-inflammatory shift.
Materials and Methods
Human left ventricular samples
Human non-diseased whole lateral left ventricular (LV) wall tissue was collected from organ donors (men= 16 and women= 15). The whole tissue was frozen immediately after collection in liquid nitrogen and stored at -80°C. The donors were between 17 and 68 years of age. We divided the LV samples into 4 groups: young (17-40 years; male: n= 7 and female: n= 7) and old (50-68 years; male: n= 9 and female: n=8) individuals.
We obtained the informed consent from all donors or their legal guardians. Sample collection and the experimental protocols were approved by the Scientific Board at the Hungarian Ministry of Health (ETT-TUKEB: 4991-0/2010-1018EKU). All research was performed in accordance with the German and Hungarian guidelines.
RNA extraction and quantitative real-time PCR
Total RNA isolation from cardiac human tissue as well as quantitative real-time PCR were performed as previously described [36]. The mRNA contents of target genes were normalized to the expression of ribosomal protein large P0 (RPLP0) or hypoxanthine phosphoribosyl transferase (HPRT). The purity of the isolated RNA was analyzed with the Bioanalyzer “caliper LabChip” (Agilent Technologies, Rattingen, Germany).
Protein extraction and immunoblotting
LV samples were homogenized in RIPA buffer (50 mmol/l Tris-HCl, pH 7.4, 150 mmol/l NaCl, 1 mmol/l EDTA, 1% NP-40, and 0.25% Na-deoxycholate) supplemented with protease inhibitor cocktail (Roche, Mannheim, Germany), and the phosphatase inhibitors sodium orthovanadate (1 mmol/l Na3VO4) and sodium fluoride (1 mmol/l NaF). Proteins were quantified using the BCA Assay (Thermo Scientific Pierce Protein Biology, Schwerte, Germany). Equal amounts of total proteins were separated on SDS-polyacrylamide gels and transferred to a nitrocellulose membrane. The membranes were immunoblotted overnight with the following primary antibodies: Sirt3 (1:1000, Cell Signaling, USA), Ku70 (1:200, Santa Cruz, USA), ac-Ku70 (1:2000, Lys 331, Abcam, UK), TFAM (1:200, Santa Cruz, USA), SOD2 (1:1000, Santa Cruz, USA), catalase (1:1000, Cell Signaling, USA), Sirt1 (1:1000, Cell Signaling, USA), AMPK (1:2000, Cell Signaling, USA), p-AMPK (1:2000, Thr172, Cell Signaling, USA), FOXO1 (1:1000, Cell Signaling, USA), p-FOXO1 (1:1000, Ser 256, Cell Signaling (USA), TOM40 (1:1000, Abcam, UK) and NFκB p50 (1:1000, Santa Cruz, USA). Equal sample loading was confirmed by analysis of actin (1:1000, Santa Cruz, USA) and tubulin (1:50.000, Sigma-Aldrich, USA). Immunoreactive proteins were detected using ECL Plus (GE Healthcare, Buckinghamshire, UK) and quantified with ImageLab (version 5.2.1 build 11, Bio-Rad Laboratories (USA)).
Immunohistochemistry
For immunohistochemistry, 5 µm cryo-sections of human left ventricle were fixed in formalin for 1 hour at room temperature and subjected to a heat-induced epitope retrieval step prior to incubation with anti-CD68 antibody (clone PGM-1, Agilent Technologies, Santa Clara, CA, USA). The detection was performed by the LSAB method applying the Dako REAL™ Detection System (Agilent Technologies, Santa Clara, CA, USA). Nuclei were counterstained with hematoxylin and mounted on slides with glycerol gelatin (both Merck KGaA, Darmstadt, Germany). Negative controls were performed by omitting the primary antibody. Images were acquired using an AxioImager Z1 microscope (Carl Zeiss MicroImaging, Inc.). Positive cells were quantified in 5 high power fields (hpf) (field of vision in x400 original magnification). All evaluations were performed in a blinded manner.
Statistical analysis
The data are given as the mean ± SEM. The GraphPad Prism 5 (GraphPad Software, 2003, San Diego, USA) was used for the statistical analysis. The data were evaluated using the non-parametric test (Mann-Whitney test, for two independent groups) or two-way analysis of variance (to test two independent variables). The Bonferroni post-test was used as a post hoc test. Statistical significance was accepted when p < 0.05.
Supplementary Materials
Author Contributions
M.B.d.A. conceived the project, analyzed the data, prepared the figures and wrote the main manuscript text. S.P. analyzed the data and wrote the main manuscript text. A.K. performed and analyzed the immunohistochemical experiments and revised the manuscript, I.B. provided the human tissue and revised the manuscript, Y.L. analyzed the data and wrote the main part of the manuscript and V.R.Z. generated research funds and coordinated the project. All authors commented on the manuscript.
Acknowledgements
We thank Jenny Jansen, Natalie Haritonow, Arne Kühne and Pavelas Karkacas for their technical assistance.
Conflicts of Interest
The authors declare no competing interests.
Funding
This work was funded by the DZHK (German Centre for Cardiovascular Research; 81Z2100201) (including salaries and consumables). Non-diseased cardiac tissue collection and management was supported by the Hungarian National Research, Development and Innovation Office (GINOP-2.3.2-15-2016-00040) and Ministry of Human Capacities (20391-3/2018/FEKUSTRAT) to IB.
References
- 1. López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013; 153:1194–217. https://doi.org/10.1016/j.cell.2013.05.039 [PubMed]
- 2. Zahn JM, Poosala S, Owen AB, Ingram DK, Lustig A, Carter A, Weeraratna AT, Taub DD, Gorospe M, Mazan-Mamczarz K, Lakatta EG, Boheler KR, Xu X, et al. AGEMAP: a gene expression database for aging in mice. PLoS Genet. 2007; 3:e201. https://doi.org/10.1371/journal.pgen.0030201 [PubMed]
- 3. Zahn JM, Sonu R, Vogel H, Crane E, Mazan-Mamczarz K, Rabkin R, Davis RW, Becker KG, Owen AB, Kim SK. Transcriptional profiling of aging in human muscle reveals a common aging signature. PLoS Genet. 2006; 2:e115. https://doi.org/10.1371/journal.pgen.0020115 [PubMed]
- 4. Jiang P, Du W, Mancuso A, Wellen KE, Yang X. Reciprocal regulation of p53 and malic enzymes modulates metabolism and senescence. Nature. 2013; 493:689–93. https://doi.org/10.1038/nature11776 [PubMed]
- 5. Keller KM, Howlett SE. Sex Differences in the Biology and Pathology of the Aging Heart. Can J Cardiol. 2016; 32:1065–73. https://doi.org/10.1016/j.cjca.2016.03.017 [PubMed]
- 6. Lee S, Jeong SY, Lim WC, Kim S, Park YY, Sun X, Youle RJ, Cho H. Mitochondrial fission and fusion mediators, hFis1 and OPA1, modulate cellular senescence. J Biol Chem. 2007; 282:22977–83. https://doi.org/10.1074/jbc.M700679200 [PubMed]
- 7. Salminen A, Kaarniranta K, Hiltunen M, Kauppinen A. Krebs cycle dysfunction shapes epigenetic landscape of chromatin: novel insights into mitochondrial regulation of aging process. Cell Signal. 2014; 26:1598–603. https://doi.org/10.1016/j.cellsig.2014.03.030 [PubMed]
- 8. Wiel C, Lallet-Daher H, Gitenay D, Gras B, Le Calvé B, Augert A, Ferrand M, Prevarskaya N, Simonnet H, Vindrieux D, Bernard D. Endoplasmic reticulum calcium release through ITPR2 channels leads to mitochondrial calcium accumulation and senescence. Nat Commun. 2014; 5:3792. https://doi.org/10.1038/ncomms4792 [PubMed]
- 9. Zhong Z, Umemura A, Sanchez-Lopez E, Liang S, Shalapour S, Wong J, He F, Boassa D, Perkins G, Ali SR, McGeough MD, Ellisman MH, Seki E, et al. NF-κB Restricts Inflammasome Activation via Elimination of Damaged Mitochondria. Cell. 2016; 164:896–910. https://doi.org/10.1016/j.cell.2015.12.057 [PubMed]
- 10. Matsushima S, Sadoshima J. The role of sirtuins in cardiac disease. Am J Physiol Heart Circ Physiol. 2015; 309:H1375–89. https://doi.org/10.1152/ajpheart.00053.2015 [PubMed]
- 11. Braidy N, Guillemin GJ, Mansour H, Chan-Ling T, Poljak A, Grant R. Age related changes in NAD+ metabolism oxidative stress and Sirt1 activity in wistar rats. PLoS One. 2011; 6:e19194. https://doi.org/10.1371/journal.pone.0019194 [PubMed]
- 12. Kwon S, Seok S, Yau P, Li X, Kemper B, Kemper JK. Obesity and aging diminish sirtuin 1 (SIRT1)-mediated deacetylation of SIRT3, leading to hyperacetylation and decreased activity and stability of SIRT3. J Biol Chem. 2017; 292:17312–23. https://doi.org/10.1074/jbc.M117.778720 [PubMed]
- 13. Cantó C, Gerhart-Hines Z, Feige JN, Lagouge M, Noriega L, Milne JC, Elliott PJ, Puigserver P, Auwerx J. AMPK regulates energy expenditure by modulating NAD+ metabolism and SIRT1 activity. Nature. 2009; 458:1056–60. https://doi.org/10.1038/nature07813 [PubMed]
- 14. Qiang W, Weiqiang K, Qing Z, Pengju Z, Yi L. Aging impairs insulin-stimulated glucose uptake in rat skeletal muscle via suppressing AMPKalpha. Exp Mol Med. 2007; 39:535–43. https://doi.org/10.1038/emm.2007.59 [PubMed]
- 15. Dumke CL, Mark Davis J, Angela Murphy E, Nieman DC, Carmichael MD, Quindry JC, Travis Triplett N, Utter AC, Gross Gowin SJ, Henson DA, McAnulty SR, McAnulty LS. Successive bouts of cycling stimulates genes associated with mitochondrial biogenesis. Eur J Appl Physiol. 2009; 107:419–27. https://doi.org/10.1007/s00421-009-1143-1 [PubMed]
- 16. Kong X, Wang R, Xue Y, Liu X, Zhang H, Chen Y, Fang F, Chang Y. Sirtuin 3, a new target of PGC-1alpha, plays an important role in the suppression of ROS and mitochondrial biogenesis. PLoS One. 2010; 5:e11707. https://doi.org/10.1371/journal.pone.0011707 [PubMed]
- 17. Ranhotra HS. Up-regulation of orphan nuclear estrogen-related receptor alpha expression during long-term caloric restriction in mice. Mol Cell Biochem. 2009; 332:59–65. https://doi.org/10.1007/s11010-009-0174-6 [PubMed]
- 18. Ahn BH, Kim HS, Song S, Lee IH, Liu J, Vassilopoulos A, Deng CX, Finkel T. A role for the mitochondrial deacetylase Sirt3 in regulating energy homeostasis. Proc Natl Acad Sci USA. 2008; 105:14447–52. https://doi.org/10.1073/pnas.0803790105 [PubMed]
- 19. Price NL, Gomes AP, Ling AJ, Duarte FV, Martin-Montalvo A, North BJ, Agarwal B, Ye L, Ramadori G, Teodoro JS, Hubbard BP, Varela AT, Davis JG, et al. SIRT1 is required for AMPK activation and the beneficial effects of resveratrol on mitochondrial function. Cell Metab. 2012; 15:675–90. https://doi.org/10.1016/j.cmet.2012.04.003 [PubMed]
- 20. Yang XD, Tajkhorshid E, Chen LF. Functional interplay between acetylation and methylation of the RelA subunit of NF-kappaB. Mol Cell Biol. 2010; 30:2170–80. https://doi.org/10.1128/MCB.01343-09 [PubMed]
- 21. Yeung F, Hoberg JE, Ramsey CS, Keller MD, Jones DR, Frye RA, Mayo MW. Modulation of NF-kappaB-dependent transcription and cell survival by the SIRT1 deacetylase. EMBO J. 2004; 23:2369–80. https://doi.org/10.1038/sj.emboj.7600244 [PubMed]
- 22. Papa L, Germain D. SirT3 regulates the mitochondrial unfolded protein response. Mol Cell Biol. 2014; 34:699–710. https://doi.org/10.1128/MCB.01337-13 [PubMed]
- 23. Tseng AH, Wu LH, Shieh SS, Wang DL. SIRT3 interactions with FOXO3 acetylation, phosphorylation and ubiquitinylation mediate endothelial cell responses to hypoxia. Biochem J. 2014; 464:157–68. https://doi.org/10.1042/BJ20140213 [PubMed]
- 24. Howcroft TK, Campisi J, Louis GB, Smith MT, Wise B, Wyss-Coray T, Augustine AD, McElhaney JE, Kohanski R, Sierra F. The role of inflammation in age-related disease. Aging (Albany NY). 2013; 5:84–93. https://doi.org/10.18632/aging.100531 [PubMed]
- 25. Salminen A, Kauppinen A, Suuronen T, Kaarniranta K. SIRT1 longevity factor suppresses NF-kappaB -driven immune responses: regulation of aging via NF-kappaB acetylation? BioEssays. 2008; 30:939–42. https://doi.org/10.1002/bies.20799 [PubMed]
- 26. Stefanatos R, Sanz A. The role of mitochondrial ROS in the aging brain. FEBS Lett. 2018; 592:743–50. https://doi.org/10.1002/1873-3468.12902 [PubMed]
- 27. Alenazi FS, Ibrahim BA, Al-Hamami H, Shakiya M, Briski KP. Role of estradiol in intrinsic hindbrain AMPK regulation of hypothalamic AMPK, metabolic neuropeptide, and norepinephrine activity and food intake in the female rat. Neuroscience. 2016; 314:35–46. https://doi.org/10.1016/j.neuroscience.2015.11.048 [PubMed]
- 28. Guo JM, Shu H, Wang L, Xu JJ, Niu XC, Zhang L. SIRT1-dependent AMPK pathway in the protection of estrogen against ischemic brain injury. CNS Neurosci Ther. 2017; 23:360–69. https://doi.org/10.1111/cns.12686 [PubMed]
- 29. Lan F, Cacicedo JM, Ruderman N, Ido Y. SIRT1 modulation of the acetylation status, cytosolic localization, and activity of LKB1. Possible role in AMP-activated protein kinase activation. J Biol Chem. 2008; 283:27628–35. https://doi.org/10.1074/jbc.M805711200 [PubMed]
- 30. Salminen A, Kaarniranta K. AMP-activated protein kinase (AMPK) controls the aging process via an integrated signaling network. Ageing Res Rev. 2012; 11:230–41. https://doi.org/10.1016/j.arr.2011.12.005 [PubMed]
- 31. Sharma AN, Aoun P, Wigham JR, Weist SM, Veldhuis JD. Estradiol, but not testosterone, heightens cortisol-mediated negative feedback on pulsatile ACTH secretion and ACTH approximate entropy in unstressed older men and women. Am J Physiol Regul Integr Comp Physiol. 2014; 306:R627–35. https://doi.org/10.1152/ajpregu.00551.2013 [PubMed]
- 32. Pfeilschifter J, Köditz R, Pfohl M, Schatz H. Changes in proinflammatory cytokine activity after menopause. Endocr Rev. 2002; 23:90–119. https://doi.org/10.1210/edrv.23.1.0456 [PubMed]
- 33. Guo J, Duckles SP, Weiss JH, Li X, Krause DN. 17β-Estradiol prevents cell death and mitochondrial dysfunction by an estrogen receptor-dependent mechanism in astrocytes after oxygen-glucose deprivation/reperfusion. Free Radic Biol Med. 2012; 52:2151–60. https://doi.org/10.1016/j.freeradbiomed.2012.03.005 [PubMed]
- 34. Klinge CM. Estrogenic control of mitochondrial function and biogenesis. J Cell Biochem. 2008; 105:1342–51. https://doi.org/10.1002/jcb.21936 [PubMed]
- 35. Zawada I, Masternak MM, List EO, Stout MB, Berryman DE, Lewinski A, Kopchick JJ, Bartke A, Karbownik-Lewinska M, Gesing A. Gene expression of key regulators of mitochondrial biogenesis is sex dependent in mice with growth hormone receptor deletion in liver. Aging (Albany NY). 2015; 7:195–204. https://doi.org/10.18632/aging.100733 [PubMed]
- 36. Dworatzek E, Baczko I, Kararigas G. Effects of aging on cardiac extracellular matrix in men and women. Proteomics Clin Appl. 2016; 10:84–91. https://doi.org/10.1002/prca.201500031 [PubMed]
- 37. Cantó C, Auwerx J. AMP-activated protein kinase and its downstream transcriptional pathways. Cell Mol Life Sci. 2010; 67:3407–23. https://doi.org/10.1007/s00018-010-0454-z [PubMed]
- 38. Ma S, Feng J, Zhang R, Chen J, Han D, Li X, Yang B, Li X, Fan M, Li C, Tian Z, Wang Y, Cao F. SIRT1 Activation by Resveratrol Alleviates Cardiac Dysfunction via Mitochondrial Regulation in Diabetic Cardiomyopathy Mice. Oxid Med Cell Longev. 2017; 2017:4602715. https://doi.org/10.1155/2017/4602715 [PubMed]
- 39. Yuan Y, Cruzat VF, Newsholme P, Cheng J, Chen Y, Lu Y. Regulation of SIRT1 in aging: roles in mitochondrial function and biogenesis. Mech Ageing Dev. 2016; 155:10–21. https://doi.org/10.1016/j.mad.2016.02.003 [PubMed]
- 40. Toba H, de Castro Brás LE, Baicu CF, Zile MR, Lindsey ML, Bradshaw AD. Secreted protein acidic and rich in cysteine facilitates age-related cardiac inflammation and macrophage M1 polarization. Am J Physiol Cell Physiol. 2015; 308:C972–82. https://doi.org/10.1152/ajpcell.00402.2014 [PubMed]
- 41. Kim YR, Lee EK, Kim DH, Kim KM, Lee B, An HJ, Park JW, Moon KM, Park MH, Chung KW, Park JY, Kim SJ, Yun HY, et al. PPARα activation by MHY908 attenuates age-related renal inflammation through modulation of the ROS/Akt/FoxO1 pathway. Exp Gerontol. 2017; 92:87–95. https://doi.org/10.1016/j.exger.2017.03.015 [PubMed]
- 42. Burkewitz K, Zhang Y, Mair WB. AMPK at the nexus of energetics and aging. Cell Metab. 2014; 20:10–25. https://doi.org/10.1016/j.cmet.2014.03.002 [PubMed]
- 43. Costantino S, Paneni F, Cosentino F. Ageing, metabolism and cardiovascular disease. J Physiol. 2016; 594:2061–73. https://doi.org/10.1113/JP270538 [PubMed]
- 44. Zhang W, Wu H, Yang M, Ye S, Li L, Zhang H, Hu J, Wang X, Xu J, Liang A. SIRT1 inhibition impairs non-homologous end joining DNA damage repair by increasing Ku70 acetylation in chronic myeloid leukemia cells. Oncotarget. 2016; 7:13538–50. https://doi.org/10.18632/oncotarget.6455 [PubMed]
- 45. Judge S, Jang YM, Smith A, Hagen T, Leeuwenburgh C. Age-associated increases in oxidative stress and antioxidant enzyme activities in cardiac interfibrillar mitochondria: implications for the mitochondrial theory of aging. FASEB J. 2005; 19:419–21. https://doi.org/10.1096/fj.04-2622fje [PubMed]
- 46. Palikaras K, Lionaki E, Tavernarakis N. Coordination of mitophagy and mitochondrial biogenesis during ageing in C. elegans. Nature. 2015; 521:525–28. https://doi.org/10.1038/nature14300 [PubMed]
- 47. Lagouge M, Argmann C, Gerhart-Hines Z, Meziane H, Lerin C, Daussin F, Messadeq N, Milne J, Lambert P, Elliott P, Geny B, Laakso M, Puigserver P, Auwerx J. Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1alpha. Cell. 2006; 127:1109–22. https://doi.org/10.1016/j.cell.2006.11.013 [PubMed]
- 48. Nemoto S, Fergusson MM, Finkel T. SIRT1 functionally interacts with the metabolic regulator and transcriptional coactivator PGC-1alpha. J Biol Chem. 2005; 280:16456–60. https://doi.org/10.1074/jbc.M501485200 [PubMed]
- 49. Valle I, Alvarez-Barrientos A, Arza E, Lamas S, Monsalve M. PGC-1alpha regulates the mitochondrial antioxidant defense system in vascular endothelial cells. Cardiovasc Res. 2005; 66:562–73. https://doi.org/10.1016/j.cardiores.2005.01.026 [PubMed]
- 50. Diedrich M, Tadic J, Mao L, Wacker MA, Nebrich G, Hetzer R, Regitz-Zagrosek V, Klose J. Heart protein expression related to age and sex in mice and humans. Int J Mol Med. 2007; 20:865–74. [PubMed]
- 51. Liu Z, Gou Y, Zhang H, Zuo H, Zhang H, Liu Z, Yao D. Estradiol improves cardiovascular function through up-regulation of SOD2 on vascular wall. Redox Biol. 2014; 3:88–99. https://doi.org/10.1016/j.redox.2014.11.001 [PubMed]
- 52. White RE, Gerrity R, Barman SA, Han G. Estrogen and oxidative stress: A novel mechanism that may increase the risk for cardiovascular disease in women. Steroids. 2010; 75:788–93. https://doi.org/10.1016/j.steroids.2009.12.007 [PubMed]
- 53. Hebert AS, Dittenhafer-Reed KE, Yu W, Bailey DJ, Selen ES, Boersma MD, Carson JJ, Tonelli M, Balloon AJ, Higbee AJ, Westphall MS, Pagliarini DJ, Prolla TA, et al. Calorie restriction and SIRT3 trigger global reprogramming of the mitochondrial protein acetylome. Mol Cell. 2013; 49:186–99. https://doi.org/10.1016/j.molcel.2012.10.024 [PubMed]
- 54. Morigi M, Perico L, Rota C, Longaretti L, Conti S, Rottoli D, Novelli R, Remuzzi G, Benigni A. Sirtuin 3-dependent mitochondrial dynamic improvements protect against acute kidney injury. J Clin Invest. 2015; 125:715–26. https://doi.org/10.1172/JCI77632 [PubMed]
- 55. Parihar P, Solanki I, Mansuri ML, Parihar MS. Mitochondrial sirtuins: emerging roles in metabolic regulations, energy homeostasis and diseases. Exp Gerontol. 2015; 61:130–41. https://doi.org/10.1016/j.exger.2014.12.004 [PubMed]
- 56. Kinn PM, Holdren GO, Westermeyer BA, Abuissa M, Fischer CL, Fairley JA, Brogden KA, Brogden NK. Age-dependent variation in cytokines, chemokines, and biologic analytes rinsed from the surface of healthy human skin. Sci Rep. 2015; 5:10472. https://doi.org/10.1038/srep10472 [PubMed]
- 57. Zhang B, Bailey WM, Braun KJ, Gensel JC. Age decreases macrophage IL-10 expression: implications for functional recovery and tissue repair in spinal cord injury. Exp Neurol. 2015; 273:83–91. https://doi.org/10.1016/j.expneurol.2015.08.001 [PubMed]
- 58. Wilder RL. Neuroendocrine-immune system interactions and autoimmunity. Annu Rev Immunol. 1995; 13:307–38. https://doi.org/10.1146/annurev.iy.13.040195.001515 [PubMed]
- 59. Cutolo M, Capellino S, Sulli A, Serioli B, Secchi ME, Villaggio B, Straub RH. Estrogens and autoimmune diseases. Ann N Y Acad Sci. 2006; 1089:538–47. https://doi.org/10.1196/annals.1386.043 [PubMed]
- 60. Straub RH. The complex role of estrogens in inflammation. Endocr Rev. 2007; 28:521–74. https://doi.org/10.1210/er.2007-0001 [PubMed]
- 61. Kublickiene K, Luksha L. Gender and the endothelium. Pharmacol Rep. 2008; 60:49–60. [PubMed]
- 62. Asai K, Hiki N, Mimura Y, Ogawa T, Unou K, Kaminishi M. Gender differences in cytokine secretion by human peripheral blood mononuclear cells: role of estrogen in modulating LPS-induced cytokine secretion in an ex vivo septic model. Shock. 2001; 16:340–43. https://doi.org/10.1097/00024382-200116050-00003 [PubMed]
- 63. Deshpande R, Khalili H, Pergolizzi RG, Michael SD, Chang MD. Estradiol down-regulates LPS-induced cytokine production and NFkB activation in murine macrophages. Am J Reprod Immunol. 1997; 38:46–54. https://doi.org/10.1111/j.1600-0897.1997.tb00275.x [PubMed]
- 64. Mosser DM, Edwards JP. Exploring the full spectrum of macrophage activation. Nat Rev Immunol. 2008; 8:958–69. https://doi.org/10.1038/nri2448 [PubMed]