Introduction
Esophageal cancer (EC) is one of the most frequent cancers globally [1], and it carries a poor prognosis with roughly 25% of patients presenting with metastatic disease [2]. There are two main types of EC that occur in the esophagus: esophageal squamous cell carcinoma (ESCC) and esophageal adenocarcinoma (EAC). Rates of esophageal adenocarcinoma have been rising over the past four decades. The reasons for this remarkable increasing are unknown [3, 4]. The mortality rate of EAC is higher worldwide. However, effective strategies to decrease the incidence and mortality of EAC remain lacking.
Accumulating evidence has indicated that long non-coding RNAs (lncRNAs) play important roles in cancer biology [5–9]. Recently, long noncoding RNA (lncRNA) has attracted researchers’ attentions [10]. LncRNAs are involved in various malignant tumors, such as those of the brain [11, 12], breast [13], lung [14, 15], liver [16] and pancreas [17, 18]. LncRNAs, which are defined as being longer than 200 nucleotides without or with limit protein coding ability [19–21], emerge as essential regulators in almost all aspects of biology via regulation at chromatin organization, transcriptional and post-transcriptional levels [22, 23]. Additionally, a number of studies extend our knowledge lncRNAs play important roles in carcinogenesis and cancer metastasis [24–27]. Mounting evidence has shown that lncRNAs are capable of influencing various cellular processes such as cell proliferation, cell cycle regulation, tumor growth and apoptosis [28–31]. Also, despite the vast number of recent lncRNA studies [32, 33], the exact function of lncRNA in esophageal carcinoma tumor genesis is still unknown.
As we know, research on the effects of lncRNA on EAC is still in the preliminary stage, and the related reports are rare [34]. Yang et al [35] reported that dysregulation of HNF1A-AS1 participated in esophageal tumorigenesis, knockdown of HNF1A-AS1 inhibited the proliferation and invasion of esophageal adenocarcinoma cells. Wu et al [36] showed that long non-coding RNA AFAP1-AS1 was reduced in BE and EAC, and its expression inhibited cancer-related biologic functions of EAC cells. Although, there has been a heavy focus on the ways that lncRNAs contribute to cancers development, but their aberrant expression and functional role in EAC development is still not well documented.
LINC00857 was reported to play oncologic roles in several cancers, for example, lung adenocarcinoma [37], bladder [38], gastric [39, 40] and liver cancer [41], and it has already been suggested that LINC00857 acts as a cell cycle regulator in lung adenocarcinoma by our previous work [37]. However, the functional role and underlying mechanism of LINC00857 in EAC remains unclear. Here we investigated the role of LINC00857 in EAC. We found that knockdown of LINC00857 decreased cell proliferation, invasion and migration, as well as increased apoptosis in EAC cell lines. The oncogenic role of LINC00857 may be through multiple oncogenes.
Results
LINC00857 expression was increased in primary EAC and cell lines
LINC00857 expression was increased in many types of cancer [37], but there is no report of this lncRNA expression status in esophagus cancer. By analyzed our esophagus cancer Affymetrix array data (unpublished data), we found LINC00857 was increased in EAC as compared to normal esophagus tissues (Figure 1A). Next, we performed RT-PCR for LINC00857 expression using another set of tissues including 8 EAC and 8 normal esophagus controls. We confirmed that LINC00857 expression was higher in EAC (Figure 1B). We also evaluated the LINC00857 expression in a larger RNA-Seq data [42] including 26 esophagus cancer cell lines. We found 19/26 cell lines have higher LINC00857 expression level more than 1 FPKM value, and OE33, an adenocarcinoma, was the highest one (Figure 1C).
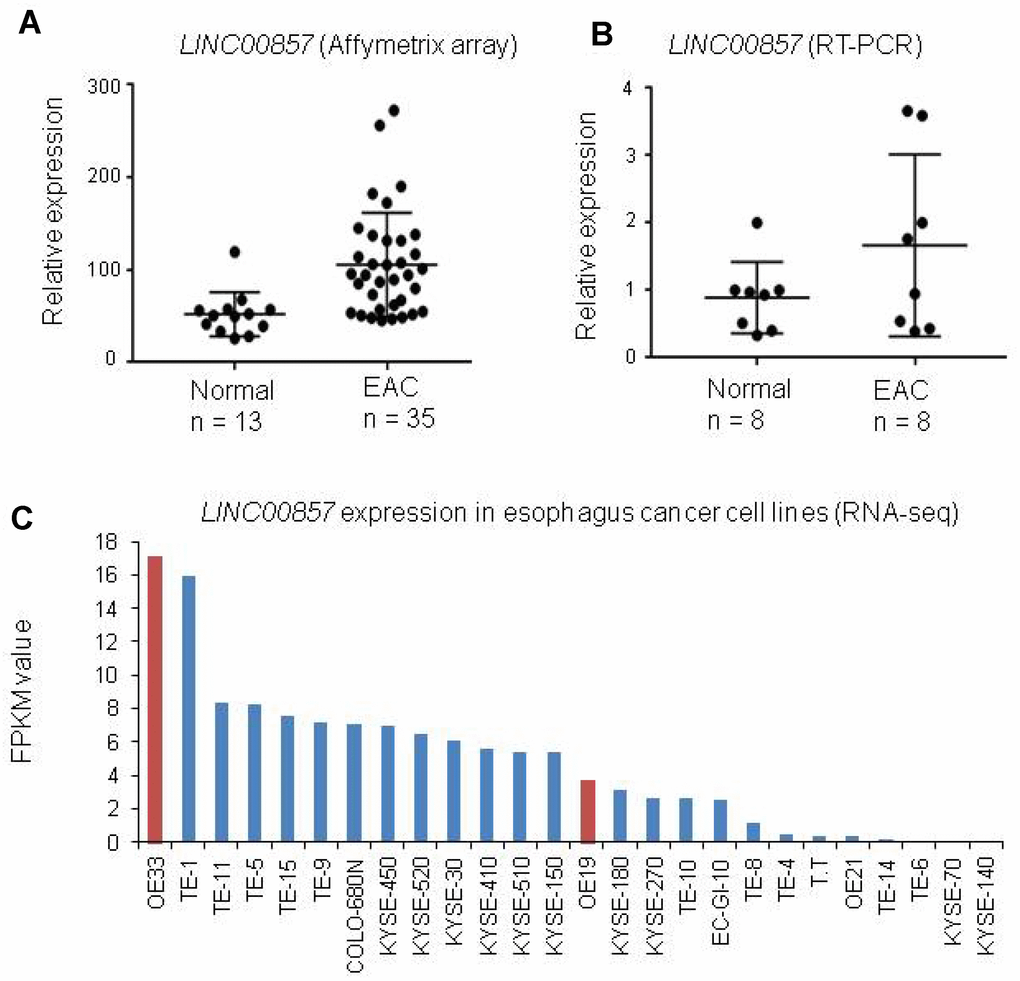
Figure 1. LINC00857 expression was increased in primary EAC and cell lines. (A) LINC00857 was increased in EAC as compared to normal esophagus tissues (The original data is coming from our unpublished Affymetrix Human Gene ST2.1 including 35 EAC and 13 paired non-tumor esophageal tissues). (B) LINC00857 expression was higher in EAC by RT-PCR using another set of tissues including 8 EAC and 8 normal esophagus controls. (C) LINC00857 expression in a larger RNA-Seq data [42] including 26 esophagus cancer cell lines.
The cell proliferation and colony formation were decreased after knockdown of LINC00857 in EAC
LINC00857 was reported to play an oncogenic role in lung cancer. To test if LINC00857 was functionally involved in EAC, we measured cell proliferation and colony formation followed by LINC00857 knockdown with siRNAs in 3 EAC cell lines, OE19, OE33 and FLO1. QRT-PCR assays revealed that LINC00857 expression was significantly reduced after transfection with LINC00857 siRNAs (**p < 0.01, Figure 2A). The cell proliferation was performed using WST-1 assay and the result showed that the cell proliferation was decreased by more than 35% after LINC00857 knockdown at 120 h in OE19, OE33 and FLO1 cell lines (*p < 0.05, Figure 2B). Similarly, the colony-formation was significantly decreased following inhibition of LINC00857 in OE33 and FLO1 cell lines (**p < 0.01, Figures 2C and 1D). Flow cytometry analysis indicated that the cell cycle was arrested at G1 phase after LINC00857 knockdown in OE33 cells (Figure 2E). These findings suggested that LINC00857 was involved in the regulation of cell proliferation/cell cycle in EAC cells.
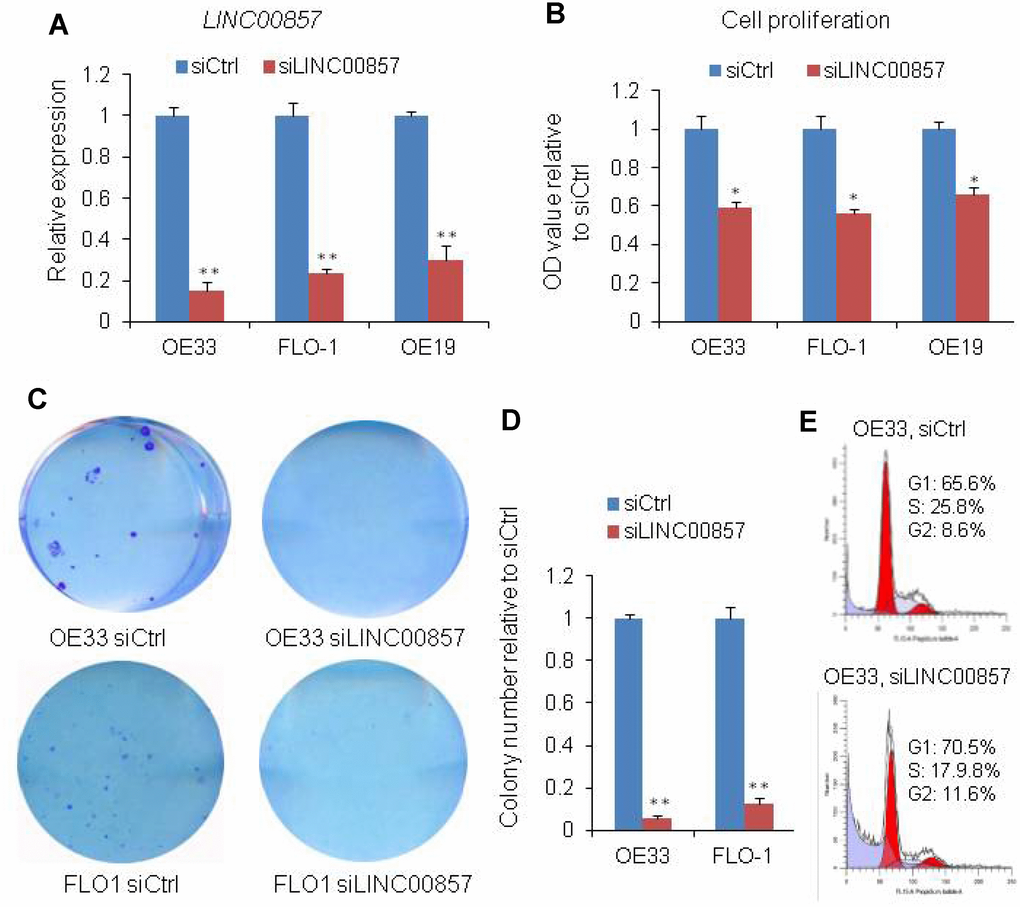
Figure 2. SiRNA-mediated knockdown of LINC00857 inhibits EAC cell proliferation. (A) The LINC00857 expression level was determined by qPCR when OE19, OE33 and FLO1 cells transfected with siLINC00857. (B) WST-1 assays were used to determine the cell viability for siLINC00857-transfected OE33, FLO1 and OE19. (C) Colony-forming assays were conducted to determine the colony formation of siLINC00857-transfected OE33 and FLO1 cells. (D) The bar chart represented the count number of cloning. Values represented the mean ± s.d. from three independent experiments. *p < 0.05, **p < 0.01. (E) Flow cytometry analysis showed that the cell cycle was arrested at G1 phase after LINC00857 knockdown in OE33 cells.
Cell migration and invasion were decreased after LINC00857 knockdown in EAC cells
Cell migration and invasion are significant aspects of cancer progression, which involves the dissolution of extracellular matrix proteins and the migration of tumor cells into contiguous tissues. In order to investigate the potential role of LINC00857 on cell migration and invasion in EAC cells, we performed transwell assays. We found that the cell migration was inhibited by 90% after LINC00857 knockdown in OE33 and FLO-1 cells (**p < 0.01, Figure 3A and 3B). The cell invasion was down-regulated by 90% in OE33 and FLO-1 followed LINC00857 knockdown (**p < 0.01, Figure 3C and 3D). These results implied that LINC00857 may be involved in mechanisms relevant to the metastatic potential of EAC.
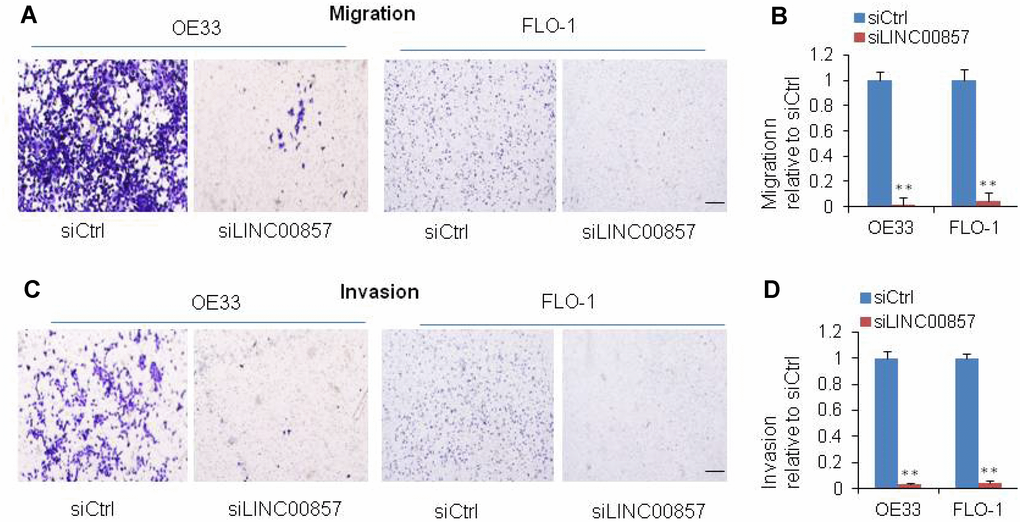
Figure 3. SiRNA-mediated knockdown of LINC00857 inhibits EAC cell migration and invasion. (A) Migration was decreased after LINC00857 siRNA transfection of OE33 and FLO1 cells. (B) The bar chart represented the count number of migration cells. (C) Invasion was decreased after LINC00857 siRNA transfection of OE33 and FLO1 cells. (D) The bar chart represented the count number of invasion cells. Values represented the mean ± s.d. from three independent experiments. *p < 0.05, **p < 0.01.
Apoptosis was induced after LINC00857 knockdown in EAC cells
To probe potential role of LINC00857 in cell death regulation, we assessed apoptosis assay by measuring cleavedPARP protein in OE33 and FLO1 cells. Western blot indicated that the cleavedPARP bands (Figure 4A) occurred only after LINC00857 siRNA treatment, suggesting that inhibition of LINC00857 could induce apoptosis in these cell lines. Moreover, LINC00857 knockdown led to an increased expression of p53, suggesting this apoptosis may be p53 mediated.
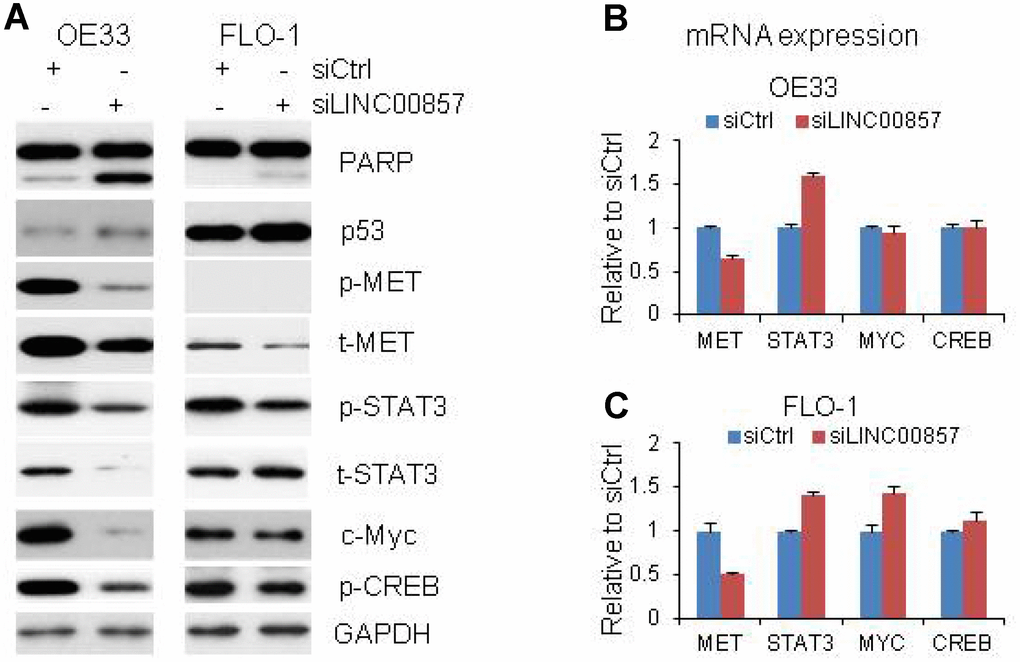
Figure 4. Proteins and mRNA regulated by LINC00857. (A) Protein levels of p53, Cleavage of PARP, MET, STAT3, c-Myc and CREB were regulated by LINC00857 siRNA in OE33 and FLO cells, GAPDH was used as a protein loading control. (B and C) QRT-PCR showing the mRNA expression of MET, STAT3, c-Myc and CREB in OE33 and FLO1 cells. GAPDH was used as control.
Multiple oncogenic proteins including MET, STAT3, c-Myc and CREB were decreased upon LINC00857 knockdown
In a further attempt to dissect potential molecular signaling regulated by LINC00857, we applied Western blot to identify proteins whose expression altered after LINC00857 knockdown in OE33 and FLO1 cells. We found that several oncogenic proteins including t-MET, p-MET, p-STAT3, c-Myc and p-CREB were decreased after LINC00857 knockdown with siRNA treated cells at 72 hrs in OE33 and FLO1 cells (Figure 4A). The t-STAT3 was not decrease in FLO1 cells. The results indicated that these proteins may take an important role in regulating cell proliferation and apoptosis in EAC cells in LINC00857 network. We performed the mRNA expression of these genes by RT-PCR, we found the MET mRNA was down regulated by 40–50% in both OE33 and FLO1 cells, indicating the MET gene was regulated at the transcriptional level upon LINC00857 knockdown (Figure 4B and 4C). While, the mRNA levels of STAT3, c-Myc and CREB were either increased or not changed after LINC00857 knockdown, suggesting these proteins were regulated at post-transcriptional level. We didn’t find that ERK, AKT, FAK, p27, and Bax proteins were changed after LINC00857 siRNA treatment at 72 hours in EAC.
OE33 is known as MET driven cell, in order to evaluate the role of MET in EAC cells, we performed MET siRNA knockdown on OE33 and FLO1cells. After MET knockdown, the cell proliferation measured by WST-1 assay was decreased by more than 33%–49% relative to control scramble siRNA (Figure 5A). We also found that the cell proliferation was decreased upon STAT3 knockdown (Figure 5B). This suggested that MET and STAT3 are important oncogenes in LINC00857 regulating cancer progression in EAC cells. Taken together, LINC00857 affecting tumor cell proliferation, colony formation, apoptosis, as well as migration and invasion may be via MET, STAT3, c-Myc and CREB oncoproteins (Figure 5C).
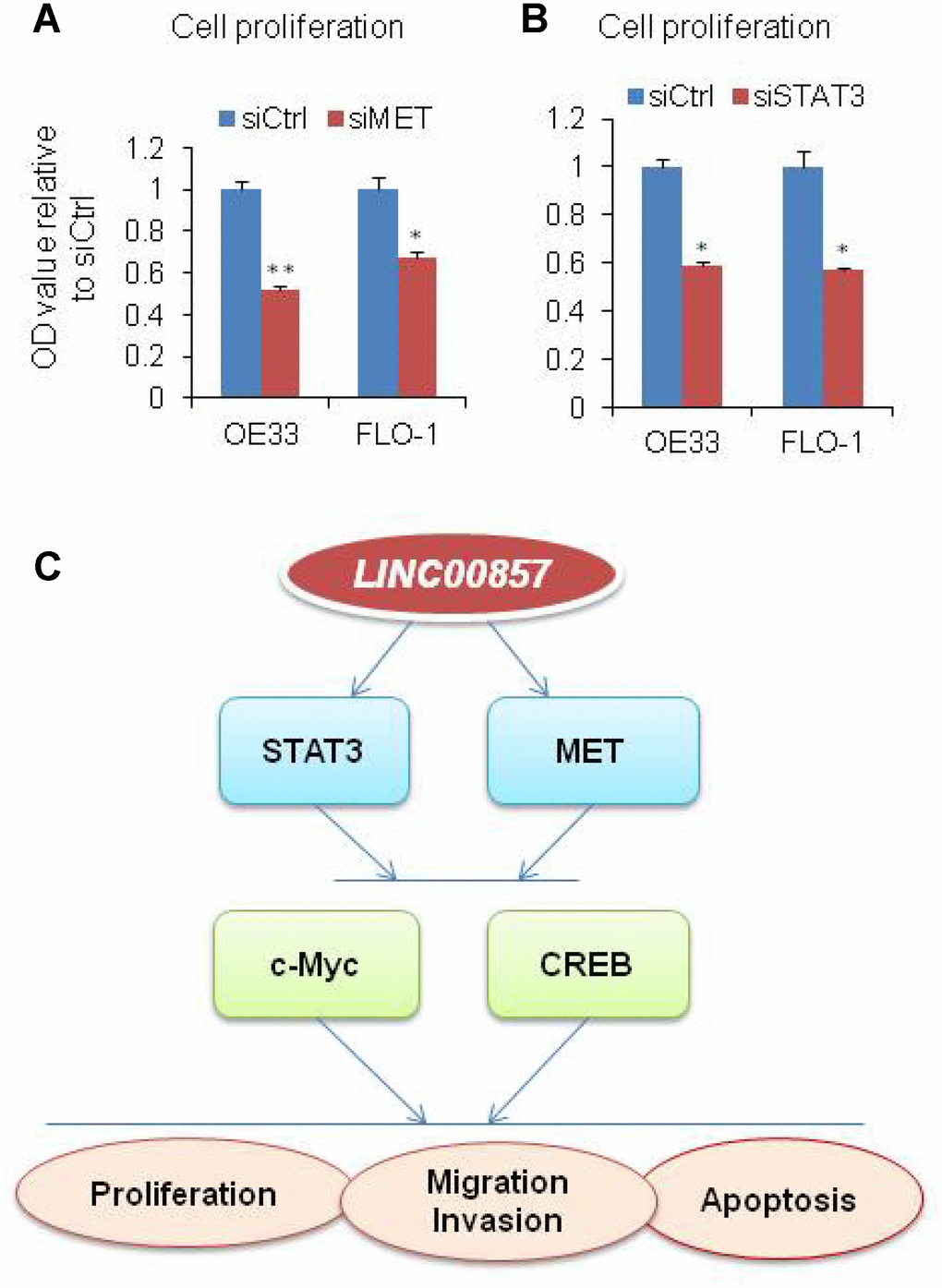
Figure 5. Model of LINC00857 in EAC. (A and B) WST-1 assays were used to determine the cell viability for MET and STAT3 siRNA transfecting OE33 and FLO1 cells. Values represented the mean ± s.d. from three independent experiments. *p < 0.05, **p < 0.01. (C) Model of LINC00857 modulating the cells proliferation, migration, invasion and induced apoptosis via MET, STAT3, c-Myc and CREB proteins in EAC.
Discussion
LINC00857 is a novel lncRNA transcribed from chromosome 11q22.3, and played an oncogenic role in lung cancer [37]. LINC00857 was also reported to be detectable in patient plasma with gastric cancer [40]. The oncogenic role of this lncRNA in EAC has not been reported. Our study showing that the LINC00857 was overexpressed in EAC tissues and EAC cell lines. We found that the cell proliferation and colony formation were decreased after LINC00857 knockdown with siRNA in EAC cells. The cell apoptosis was also induced upon LINC00857 knockdown, indicating LINC00857 may be involved in EAC tumor growth. Metastasis is another important malignant behavior of cancer and is the most troublesome problem in tumor prognosis and therapy. LncRNAs have been reported to involve in the regulation of tumor metastasis such as MALAT-1 [43] and HOX antisense intergenic RNA (HOTAIR) [44]. We found that the cell migration and invasion ability of EAC cells were significantly decreased after the knockdown of LINC00857, suggesting LINC00857 may be involved in the regulator of metastasis in EAC.
LINC00857 induces tumorigenesis through complicated mechanisms, involving activation of signaling pathways that regulate cell survival and proliferation. Previous studies have shown that downregulation of LINC00857 was able to restrain HCC cell migration and invasion capacity via enhancing epithelial-mesenchymal transition (EMT) process [41]. LINC00857 knockdown decreased protein expression of cyclin D1 and cyclin E1 in GC cells [39]. Wang [37] shows that LINC00857 mediated tumor progression via cell cycle regulation in lung cancer. In this study, mechanistically, we found that multiple oncogenic proteins such as MET, STAT3, c-Myc and CREB were decreased upon LINC00857 knockdown in EAC cells. Knockdown MET or STAT3, the cell proliferation were also decreased indicating these two proteins may be involved in LINC00857 signaling in regulation of EAC cell proliferation. According to recently reports, Stat3 /c-Myc signaling pathways induced tumor development in gastric cancer [45]. c-Met strongly increased the level of transgenic c-Myc that was expressed via a constitutive CMV promoter in large-cell medulloblastoma [46]. These results are supporting evidence that LINC00857 abrogation induced apoptosis and decreased migration and invasion ability maybe through the inhibition of MET or STAT3 signaling.
In summary, LINC00857 influenced tumor cell proliferation, colony formation, apoptosis, as well as migration and invasion which may be via affecting MET/STAT3/c-Myc/CREB oncoproteins (Figure 5C). These findings suggested that LINC00857 is a functional lncRNA in human EAC cells and plays an important role in EAC progression.
Materials and Methods
Cell culture
EAC cell lines OE19, OE33 and FLO1 were purchased from Sigma Chemical (St Louis, Missouri, USA), and the European Collection of Cell Culture (Porton Down, UK), respectively. All media were supplemented with 10% fetal bovine serum (Invitrogen, San Diego, California, USA), unless otherwise stated.
Esophageal cancer specimen
Esophageal adenocarcinoma tissues and paired non-tumor tissues were obtained from patients undergoing cancer surgery during the period from 1995 to 2013 at the University of Michigan Health System. None of the patients included in this study received any preoperative radiation or chemotherapy. All the patients provided informed consent, and all experimental protocols were approved by the University of Michigan Institutional Review Board and Ethics Committee. Resected specimens were frozen in liquid nitrogen and then stored at -80°C until use. Frozen tissues for regions containing a minimum of 70% tumor cellularity defined by cryostat sectioning were utilized for RNA isolation.
Cell Proliferation Assays
Cells were plated at a density of 1000 cells per well onto 96-well plates. LINC00857 siRNA and control siRNA were added at 24 hrs and 48 hrs. Cell proliferation was measured at 96 hrs-120 hrs after siRNA transfection using WST-1 reagent (Roche, Mannheim, Germany) according to manufacturer’s instructions. All experiments were performed in triplicate.
RNA isolation and qRT-PCR
Total RNA was isolated from tissues or cultured cells using miRNeasy Mini kit (Qiagen) according to the manufacturers' instructions. One microgram of total RNA was used for the reverse transcription reaction in a final volume of 20 μL with random primers under standard conditions using High Capacity cDNA Reverse Transcription kit (Thermo Fisher Scientific). 1 μL of the corresponding cDNA was used for subsequent qRT-PCR reactions using Power SYBR Green master Mix (Thermo Fisher Scientific) according to the manufacturer’s instructions. The expression of GAPDH was used to normalize the results. The PCR amplification was performed for 40 cycles of 95°C for 15 sec and 60°C for 60 sec on StepOne Real-Time PCR System (Thermo Fisher Scientific). All reactions were run in triplicate and data were analyzed using the comparative cycle threshold (CT) method. The primer sequences are summarized in Table 1.
Table 1. Primer sequences used in this study.
| Gene name | Primer ID | Primer Sequence (5'->3') | Product size |
| LINC00857 | Lnc1 F | CCCCTGCTTCATTGTTTCCC | 131 |
| LINC00857 | Lnc1 R | AGCTTGTCCTTCTTGGGTACT | |
| MYC | MYC F | CAGCGACTCTGAGGAGGAAC | 131 |
| MYC | MYC R | TGTGAGGAGGTTTGCTGTGG | |
| STAT3 | STAT3_F | TGGCCCAATGGAATCAGCTAC | 200 |
| STAT3 | STAT3_R | CTGCTGGTCAATCTCTCCCA | |
| MET | MET F | CAACCCGAATACTGCCCAGA | 99 |
| MET | MET R | CCGGGACACCAGTTCAGAAA | |
| CREB | CREB F | GCACTATTGCCCCTGGAGTT | 127 |
| CREB | CREB R | CTACGACACTCTCGAGCTGC | |
| GAPDH | GAPDH gcF | GTCAAGGCTGAGAACGGGAA | 158 |
| GAPDH | GAPDH gcR | AAATGAGCCCCAGCCTTCTC |
Cell migration and invasion assay
Migration and invasion capabilities of the esophageal cells were measured in transwell chamber system. Sixty microliters of diluted extracellular matrix (ECM) gel solution was added into the upper chambers (Costar Inc., USA). For migration assay, the same method was used without ECM membrane on the upper chamber. The chamber was incubated at 37 °C for 4 h. Next, a density of 1×105 per well cells was seeded in 100-μl medium with 1% FBS. The lower level chambers of the transwells were filled with 500 μl RPMI and DMEM containing 10% FBS. To allow cell migration, the transwells were then incubated at 37 °C with 5 % CO2 for 24-48 h. After incubation, a cotton swab was used to carefully remove the cells in the upper chamber. Cells at the bottom of the ECM gel-coated membrane were fixed and stained with paraformaldehyde and crystal violet. The crystal violet was dissolved in 200 μl glacial acetic acid and incubated at room temperature for 30 min.
Colony formation assay
Esophageal cells were cultured with siRNA mock and LINC00857 siRNA and then seeded in a six-well plate at a density of 200 cells / well. After a 2-week period of incubation at 37 °C, 0.1 % crystal violet (Sigma-Aldrich) and 20 % methanol were used as dye solution to fix and stain the colonies. The number of colonies was counted in each well. Clones containing more than 50 cells were counted using a grid. Three independent experiments were performed. The formula for the colony formation ratio was as follows: Ratio = Numbers of Colony/Initiative Cells × 100%.
Western blotting
Cells were harvested 72 h after siRNA transfection. Lysis, electrophoresis and target protein visualisation were performed as described previously [37]. Total cell lysates were prepared with sample buffer and boiled at 95 °C for 5 min. The samples were transferred to SDS–PAGE at 80 V for 3 h and then transferred to PVDF membranes for another 3 h. After incubation with specific antibodies for STAT3, FAK, PARP, c-Myc, CREB, MET, AKT, ERK1/2 and GAPDH at 4 °C overnight, the membranes then were washed by 1% TBST for three times, incubated with secondary antibodies for 1 h, and the membranes were developed using ECL and exposed to X-ray film.
Statistical analysis
Data were analyzed using GraphPad Prism 7 (GraphPad software) and R software. All data are continuous variables and follow a normal distribution. The other data such as proliferation were evaluated by unpaired Student’s t-test. All values were expressed as mean±SD. Statistical significance was noted at p<0.05. Three independent triplicated experiments were performed for cell biological assays, unless otherwise stated.
Conflicts of Interest
No potential conflicts of interest were disclosed.
Funding
This work was supported in part by National Natural Science Foundation of China (NSFC) (81702270 to W.S.; 81871883 to Z.Y.; 81803564 to L.W.); Affiliated Hospital of Guangdong Medical University Doctoral Foundation (Grant No. 2018052638) to W.S.; Guangxi Natural Science Foundation (Grant No. 2015GXNSFBA139117) to L.W.; China Postdoctoral Science Foundation (Grant No. 2018M633619XB) to L.W.; Medical Discipline Reserve Talents of Yunnan Province (Grant No. H-2017037).
References
- 1. Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015; 136:E359–86. https://doi.org/10.1002/ijc.29210 [PubMed]
- 2. Reid BJ, Li X, Galipeau PC, Vaughan TL. Barrett’s oesophagus and oesophageal adenocarcinoma: time for a new synthesis. Nat Rev Cancer. 2010; 10:87–101. https://doi.org/10.1038/nrc2773 [PubMed]
- 3. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018; 68:7–30. https://doi.org/10.3322/caac.21442 [PubMed]
- 4. Simard EP, Ward EM, Siegel R, Jemal A. Cancers with increasing incidence trends in the United States: 1999 through 2008. CA Cancer J Clin. 2012; 62:118–28. https://doi.org/10.3322/caac.20141 [PubMed]
- 5. Tang J, Zhuo H, Zhang X, Jiang R, Ji J, Deng L, Qian X, Zhang F, Sun B. A novel biomarker Linc00974 interacting with KRT19 promotes proliferation and metastasis in hepatocellular carcinoma. Cell Death Dis. 2014; 5:e1549. https://doi.org/10.1038/cddis.2014.518 [PubMed]
- 6. Ma MZ, Chu BF, Zhang Y, Weng MZ, Qin YY, Gong W, Quan ZW. Long non-coding RNA CCAT1 promotes gallbladder cancer development via negative modulation of miRNA-218-5p. Cell Death Dis. 2015; 6:e1583. https://doi.org/10.1038/cddis.2014.541 [PubMed]
- 7. Wang Y, He L, Du Y, Zhu P, Huang G, Luo J, Yan X, Ye B, Li C, Xia P, Zhang G, Tian Y, Chen R, Fan Z. The long noncoding RNA lncTCF7 promotes self-renewal of human liver cancer stem cells through activation of Wnt signaling. Cell Stem Cell. 2015; 16:413–25. https://doi.org/10.1016/j.stem.2015.03.003 [PubMed]
- 8. Guo G, Kang Q, Zhu X, Chen Q, Wang X, Chen Y, Ouyang J, Zhang L, Tan H, Chen R, Huang S, Chen JL. A long noncoding RNA critically regulates Bcr-Abl-mediated cellular transformation by acting as a competitive endogenous RNA. Oncogene. 2015; 34:1768–79. https://doi.org/10.1038/onc.2014.131 [PubMed]
- 9. Xue Y, Ma G, Zhang Z, Hua Q, Chu H, Tong N, Yuan L, Qin C, Yin C, Zhang Z, Wang M. A novel antisense long noncoding RNA regulates the expression of MDC1 in bladder cancer. Oncotarget. 2015; 6:484–93. https://doi.org/10.18632/oncotarget.2861 [PubMed]
- 10. Hao Y, Wu W, Shi F, Dalmolin RJ, Yan M, Tian F, Chen X, Chen G, Cao W. Prediction of long noncoding RNA functions with co-expression network in esophageal squamous cell carcinoma. BMC Cancer. 2015; 15:168. https://doi.org/10.1186/s12885-015-1179-z [PubMed]
- 11. Zhang X, Sun S, Pu JK, Tsang AC, Lee D, Man VO, Lui WM, Wong ST, Leung GK. Long non-coding RNA expression profiles predict clinical phenotypes in glioma. Neurobiol Dis. 2012; 48:1–8. https://doi.org/10.1016/j.nbd.2012.06.004 [PubMed]
- 12. Aloyz R, Xu ZY, Bello V, Bergeron J, Han FY, Yan Y, Malapetsa A, Alaoui-Jamali MA, Duncan AM, Panasci L. Regulation of cisplatin resistance and homologous recombinational repair by the TFIIH subunit XPD. Cancer Res. 2002; 62:5457–62. [PubMed]
- 13. Xing Z, Lin A, Li C, Liang K, Wang S, Liu Y, Park PK, Qin L, Wei Y, Hawke DH, Hung MC, Lin C, Yang L. lncRNA directs cooperative epigenetic regulation downstream of chemokine signals. Cell. 2014; 159:1110–25. https://doi.org/10.1016/j.cell.2014.10.013 [PubMed]
- 14. Yang F, Huo XS, Yuan SX, Zhang L, Zhou WP, Wang F, Sun SH. Repression of the long noncoding RNA-LET by histone deacetylase 3 contributes to hypoxia-mediated metastasis. Mol Cell. 2013; 49:1083–96. https://doi.org/10.1016/j.molcel.2013.01.010 [PubMed]
- 15. Ji P, Diederichs S, Wang W, Böing S, Metzger R, Schneider PM, Tidow N, Brandt B, Buerger H, Bulk E, Thomas M, Berdel WE, Serve H, Müller-Tidow C. MALAT-1, a novel noncoding RNA, and thymosin beta4 predict metastasis and survival in early-stage non-small cell lung cancer. Oncogene. 2003; 22:8031–41. https://doi.org/10.1038/sj.onc.1206928 [PubMed]
- 16. Askarian-Amiri ME, Crawford J, French JD, Smart CE, Smith MA, Clark MB, Ru K, Mercer TR, Thompson ER, Lakhani SR, Vargas AC, Campbell IG, Brown MA, et al. SNORD-host RNA Zfas1 is a regulator of mammary development and a potential marker for breast cancer. RNA. 2011; 17:878–91. https://doi.org/10.1261/rna.2528811 [PubMed]
- 17. Yang F, Yi F, Zheng Z, Ling Z, Ding J, Guo J, Mao W, Wang X, Wang X, Ding X, Liang Z, Du Q. Characterization of a carcinogenesis-associated long non-coding RNA. RNA Biol. 2012; 9:110–16. https://doi.org/10.4161/rna.9.1.18332 [PubMed]
- 18. Brunner AL, Beck AH, Edris B, Sweeney RT, Zhu SX, Li R, Montgomery K, Varma S, Gilks T, Guo X, Foley JW, Witten DM, Giacomini CP, et al. Transcriptional profiling of long non-coding RNAs and novel transcribed regions across a diverse panel of archived human cancers. Genome Biol. 2012; 13:R75. https://doi.org/10.1186/gb-2012-13-8-r75 [PubMed]
- 19. Mercer TR, Dinger ME, Mattick JS. Long non-coding RNAs: insights into functions. Nat Rev Genet. 2009; 10:155–59. https://doi.org/10.1038/nrg2521 [PubMed]
- 20. Derrien T, Johnson R, Bussotti G, Tanzer A, Djebali S, Tilgner H, Guernec G, Martin D, Merkel A, Knowles DG, Lagarde J, Veeravalli L, Ruan X, et al. The GENCODE v7 catalog of human long noncoding RNAs: analysis of their gene structure, evolution, and expression. Genome Res. 2012; 22:1775–89. https://doi.org/10.1101/gr.132159.111 [PubMed]
- 21. ENCODE Project Consortium. An integrated encyclopedia of DNA elements in the human genome. Nature. 2012; 489:57–74. https://doi.org/10.1038/nature11247 [PubMed]
- 22. Nagano T, Fraser P. No-nonsense functions for long noncoding RNAs. Cell. 2011; 145:178–81. https://doi.org/10.1016/j.cell.2011.03.014 [PubMed]
- 23. Ulitsky I, Bartel DP. lincRNAs: genomics, evolution, and mechanisms. Cell. 2013; 154:26–46. https://doi.org/10.1016/j.cell.2013.06.020 [PubMed]
- 24. Han L, Zhang EB, Yin DD, Kong R, Xu TP, Chen WM, Xia R, Shu YQ, De W. Low expression of long noncoding RNA PANDAR predicts a poor prognosis of non-small cell lung cancer and affects cell apoptosis by regulating Bcl-2. Cell Death Dis. 2015; 6:e1665. https://doi.org/10.1038/cddis.2015.30 [PubMed]
- 25. Zhang EB, Kong R, Yin DD, You LH, Sun M, Han L, Xu TP, Xia R, Yang JS, De W, Chen J. Long noncoding RNA ANRIL indicates a poor prognosis of gastric cancer and promotes tumor growth by epigenetically silencing of miR-99a/miR-449a. Oncotarget. 2014; 5:2276–92. https://doi.org/10.18632/oncotarget.1902 [PubMed]
- 26. Zhang EB, Yin DD, Sun M, Kong R, Liu XH, You LH, Han L, Xia R, Wang KM, Yang JS, De W, Shu YQ, Wang ZX. P53-regulated long non-coding RNA TUG1 affects cell proliferation in human non-small cell lung cancer, partly through epigenetically regulating HOXB7 expression. Cell Death Dis. 2014; 5:e1243. https://doi.org/10.1038/cddis.2014.201 [PubMed]
- 27. Kong R, Zhang EB, Yin DD, You LH, Xu TP, Chen WM, Xia R, Wan L, Sun M, Wang ZX, De W, Zhang ZH. Long noncoding RNA PVT1 indicates a poor prognosis of gastric cancer and promotes cell proliferation through epigenetically regulating p15 and p16. Mol Cancer. 2015; 14:82. https://doi.org/10.1186/s12943-015-0355-8 [PubMed]
- 28. Ginger MR, Shore AN, Contreras A, Rijnkels M, Miller J, Gonzalez-Rimbau MF, Rosen JM. A noncoding RNA is a potential marker of cell fate during mammary gland development. Proc Natl Acad Sci USA. 2006; 103:5781–86. https://doi.org/10.1073/pnas.0600745103 [PubMed]
- 29. Gutschner T, Diederichs S. The hallmarks of cancer: a long non-coding RNA point of view. RNA Biol. 2012; 9:703–19. https://doi.org/10.4161/rna.20481 [PubMed]
- 30. Mourtada-Maarabouni M, Hedge VL, Kirkham L, Farzaneh F, Williams GT. Growth arrest in human T-cells is controlled by the non-coding RNA growth-arrest-specific transcript 5 (GAS5). J Cell Sci. 2008; 121:939–46. https://doi.org/10.1242/jcs.024646 [PubMed]
- 31. Clark MB, Mattick JS. Long noncoding RNAs in cell biology. Semin Cell Dev Biol. 2011; 22:366–76. https://doi.org/10.1016/j.semcdb.2011.01.001 [PubMed]
- 32. Lv XB, Lian GY, Wang HR, Song E, Yao H, Wang MH. Long noncoding RNA HOTAIR is a prognostic marker for esophageal squamous cell carcinoma progression and survival. PLoS One. 2013; 8:e63516. https://doi.org/10.1371/journal.pone.0063516 [PubMed]
- 33. Ge XS, Ma HJ, Zheng XH, Ruan HL, Liao XY, Xue WQ, Chen YB, Zhang Y, Jia WH. HOTAIR, a prognostic factor in esophageal squamous cell carcinoma, inhibits WIF-1 expression and activates Wnt pathway. Cancer Sci. 2013; 104:1675–82. https://doi.org/10.1111/cas.12296 [PubMed]
- 34. Zang W, Wang T, Huang J, Li M, Wang Y, Du Y, Chen X, Zhao G. Long noncoding RNA PEG10 regulates proliferation and invasion of esophageal cancer cells. Cancer Gene Ther. 2015; 22:138–44. https://doi.org/10.1038/cgt.2014.77 [PubMed]
- 35. Yang X, Song JH, Cheng Y, Wu W, Bhagat T, Yu Y, Abraham JM, Ibrahim S, Ravich W, Roland BC, Khashab M, Singh VK, Shin EJ, et al. Long non-coding RNA HNF1A-AS1 regulates proliferation and migration in oesophageal adenocarcinoma cells. Gut. 2014; 63:881–90. https://doi.org/10.1136/gutjnl-2013-305266 [PubMed]
- 36. Wu W, Bhagat TD, Yang X, Song JH, Cheng Y, Agarwal R, Abraham JM, Ibrahim S, Bartenstein M, Hussain Z, Suzuki M, Yu Y, Chen W, et al. Hypomethylation of noncoding DNA regions and overexpression of the long noncoding RNA, AFAP1-AS1, in Barrett's esophagus and esophageal adenocarcinoma. Gastroenterology. 2013; 144:956–966.e4. https://doi.org/10.1053/j.gastro.2013.01.019 [PubMed]
- 37. Wang L, He Y, Liu W, Bai S, Xiao L, Zhang J, Dhanasekaran SM, Wang Z, Kalyana-Sundaram S, Balbin OA, Shukla S, Lu Y, Lin J, et al. Non-coding RNA LINC00857 is predictive of poor patient survival and promotes tumor progression via cell cycle regulation in lung cancer. Oncotarget. 2016; 7:11487–99. https://doi.org/10.18632/oncotarget.7203 [PubMed]
- 38. Dudek AM, van Kampen JG, Witjes JA, Kiemeney LA, Verhaegh GW. LINC00857 expression predicts and mediates the response to platinum-based chemotherapy in muscle-invasive bladder cancer. Cancer Med. 2018; 7:3342–50. https://doi.org/10.1002/cam4.1570 [PubMed]
- 39. Pang K, Ran MJ, Zou FW, Yang TW, He F. Long non-coding RNA LINC00857 promotes gastric cancer cell proliferation and predicts poor patient survival. Oncol Lett. 2018; 16:2119–24. https://doi.org/10.3892/ol.2018.8883 [PubMed]
- 40. Zhang K, Shi H, Xi H, Wu X, Cui J, Gao Y, Liang W, Hu C, Liu Y, Li J, Wang N, Wei B, Chen L. Genome-Wide lncRNA Microarray Profiling Identifies Novel Circulating lncRNAs for Detection of Gastric Cancer. Theranostics. 2017; 7:213–27. https://doi.org/10.7150/thno.16044 [PubMed]
- 41. Xia C, Zhang XY, Liu W, Ju M, Ju Y, Bu YZ, Wang W, Shao H. LINC00857 contributes to hepatocellular carcinoma malignancy via enhancing epithelial-mesenchymal transition. J Cell Biochem. 2018 Dec 3. https://doi.org/10.1002/jcb.28074 [PubMed] [Epub ahead of print]. .
- 42. Barretina J, Caponigro G, Stransky N, Venkatesan K, Margolin AA, Kim S, Wilson CJ, Lehár J, Kryukov GV, Sonkin D, Reddy A, Liu M, Murray L, et al. The Cancer Cell Line Encyclopedia enables predictive modelling of anticancer drug sensitivity. Nature. 2012; 483:603–07. https://doi.org/10.1038/nature11003 [PubMed]
- 43. Gutschner T, Hämmerle M, Eissmann M, Hsu J, Kim Y, Hung G, Revenko A, Arun G, Stentrup M, Gross M, Zörnig M, MacLeod AR, Spector DL, Diederichs S. The noncoding RNA MALAT1 is a critical regulator of the metastasis phenotype of lung cancer cells. Cancer Res. 2013; 73:1180–89. https://doi.org/10.1158/0008-5472.CAN-12-2850 [PubMed]
- 44. Gupta RA, Shah N, Wang KC, Kim J, Horlings HM, Wong DJ, Tsai MC, Hung T, Argani P, Rinn JL, Wang Y, Brzoska P, Kong B, et al. Long non-coding RNA HOTAIR reprograms chromatin state to promote cancer metastasis. Nature. 2010; 464:1071–76. https://doi.org/10.1038/nature08975 [PubMed]
- 45. Gao S, Chen M, Wei W, Zhang X, Zhang M, Yao Y, Lv Y, Ling T, Wang L, Zou X. Crosstalk of mTOR/PKM2 and STAT3/c-Myc signaling pathways regulate the energy metabolism and acidic microenvironment of gastric cancer. J Cell Biochem. 2018 Nov 11. https://doi.org/10.1002/jcb.26915 [PubMed] [Epub ahead of print]. .
- 46. Li Y, Guessous F, Johnson EB, Eberhart CG, Li XN, Shu Q, Fan S, Lal B, Laterra J, Schiff D, Abounader R. Functional and molecular interactions between the HGF/c-Met pathway and c-Myc in large-cell medulloblastoma. Lab Invest. 2008; 88:98–111. https://doi.org/10.1038/labinvest.3700702 [PubMed]


