Introduction
Cardiovascular disease contributes to major cause of mortality. Obesity, as a global pandemic, has been one of the main independent risk factors of cardiovascular disease such as hypertension, coronary heart disease and heart failure [1, 2]. When the amount of fat exceeds the capacity of subcutaneous storage, obesity spontaneously occurs [3]. Adipose tissue was initially related to metabolism, where stem cells had been recently identified. These so-called ADSCs displayed the ability of differentiation to vascular lineages such as smooth muscle cells (SMCs), endothelial cells (ECs) and even cardiomyocytes. Therefore, the interaction between ADSCs and vascular remodeling becomes a research hotspot.
Since the discovery of bone marrow mesenchymal stem cells (MSCs), researchers keep trying to search for other stem cell pools. Perfect pools should possess similar therapeutic potential with relatively easy harvest in large quantities and minimally invasive procedure. Under such circumstances, ADSCs were first isolated from the processed lipoaspirate by Zuk and colleagues in 2001 [4]. It reported that adipose tissue contained 500 times more stem cells than the same amount of bone marrow [5, 6]. In addition, ADSCs are less ethically or morally constrained. In addition to subcutaneous region, which is the most common site for adipose-related experiments, ADSCs can also be acquired from visceral, perivascular and pericardial regions. ADSCs, similar to other MSCs such as bone marrow-derived MSCs, express common markers of MSCs and have self-renewal ability [7]. They can also differentiate toward a variety of cell lineages such as adipocytes, osteocytes, chondrocytes and myogenic cells in response to specific culture media. Furthermore, ADSCs could give rise to cardiomyocytes, ECs, SMCs, hepatocytes, epithelial cells, and neural lineage cells [8, 9]. Detailed differentiation capacities of ADSCs toward different cell lineages are listed in Table 1.
Table 1. Differentiation abilities of ADSCs into multilineage cells.
| Cell lineage | Author | Cell source | Surface marker | |
| positive | negative | |||
| Adipocytes/ Osteoblasts/ Chondrocytes | Li et al. [99] | Human subcutaneous adipose tissue | CD13, CD29, CD90, CD105, HLA-ABC | CD14, CD19, CD34, CD45, HLA-DR |
| Viero Nora et al. [100] | Murine inguinal adipose tissue | CD29, CD44, CD49e | CD11b, CD34, CD45, CD90.2, CD117 | |
| Saler et al. [101] | Human subcutaneous adipose tissue | CD13, CD73, CD90, CD105 | CD14, CD34, CD45 | |
| Griffin et al. [102] | Human abdominal subcutaneous adipose tissue | CD73, CD90, CD105 | CD14, CD19, CD34, CD45, HLA-DR | |
| Skeletal myocytes | J.-H. Lee and D.M. Kemp [103] | Human subcutaneous adipose tissue | CD13, CD44, CD73, CD90, HLA-ABC | CD34, CD45, CD56, CD184, HLA-DP, HLA-DQ, HLA-DR |
| Bayati et al. [104] | Rat gonadal adipose tissue | CD44, CD73, CD90 | CD45 | |
| Cardiomyocytes | Choi et al. [105] | Human subcutaneous adipose tissue | CD73, CD90, CD105 | CD34, CD45 |
| Chang et al. [106] | Rat subcutaneous adipose tissue | CD13, CD29, CD44, CD90 | CD31, CD34, CD45, CD117, CD140a, Flk-1 | |
| Kim et al. [107] | Human intra-abdominal adipose tissue | CD13, CD29, CD44, CD90, CD166, HLA-ABC | CD31, CD34, CD45, CD117, HLA-DR | |
| Smooth muscle myocytes | Parvizi et al. [61] | Human subcutaneous adipose tissue | CD29, CD44, CD90, CD105 | CD31, CD45 |
| Endothelial cells | Planat-Benard et al. [31] | Human subcutaneous adipose tissue | CD13, CD34, HLA-ABC | CD14, CD31, CD45, CD144 |
| Moon et al. [34] | Human subcutaneous adipose tissue | CD34, CD44, CD90 | CD31, CD45, Flk-1 | |
| Zhang et al. [37] | Human periumbilical subcutaneous adipose tissue | CD13, CD29, CD44, CD73, CD90, CD105 | CD31, CD45 | |
| Nerve cells | Kang et al. [108] | Rhesus subcutaneous adipose tissue | CD13, CD59, CD90, HLA-1 | CD3, CD4, CD8, CD34, CD45 |
| Krampera et al. [109] | Human abdominal subcutaneous adipose tissue | CD44, CD73, CD90, CD105 | CD11c, CD14, CD31, CD34, CD45, CD123 | |
| Ying et al. [110] | Rat subcutaneous adipose tissue | CD44, CD90 | CD34, CD45 | |
| Hepatocytes | Lue et al. [111] | Human subcutaneous adipose tissue | CD34, CD90, CD105, CD133 | CD13, CD45 |
| Banas et al. [112] | Human abdominal subcutaneous adipose tissue | CD10, CD13, CD29, CD34, CD44, CD49d, CD59, CD71, CD90, CD105, CD120a, CD124, CD166, SH3 | CD11b, CD45, CD48, CD135 | |
| Epithelial cells | Brzoska et al. [113] | Human subcutaneous adipose tissue | CD10, CD13, CD44, CD90, vimentin | CD31, CD34, CD45, vWF |
| Pancreatic islet-like cells | Dhanasekaran et al. [114] | Human omentum adipose tissue | CD73, CD90, CD105 | CD31, CD34, CD45, HLA-DR |
ADSCs are separated from stromal-vascular fraction (SVF). SVF is the main component in the adipose tissue apart from adipocytes. Currently, isolation of SVF from adipose tissue is mainly by collagenase digestion followed by centrifugation [10]. SVF is composed of heterogeneous cellular population including ECs, SMCs, fibroblasts, pericytes, immune cells, MSCs and other undefined cells [11]. Flow cytometry was applied in plenty of studies to illustrate the CD (cluster of differentiation) antigenic profile of both cultured and freshly-isolated ADSCs [12, 13]. There was common consensus that cultured ADSCs were positive for CD29 [14–17], CD44 [14, 17], CD73 [18, 19], CD90 [14–18] and CD105 [14–18], negative for CD45 [14, 16] and CD31 [14, 15, 18]. The expression of some cell surface proteins remained controversial. For instance, CD34 was expressed in the SVF cells and freshly isolated ADSCs while its expression disappeared after several passages [19, 20].
There are two types of adipose tissue in mammals, white and brown adipose tissue. White adipocyte functions as an energy storage pool, whereas brown adipocyte usually oxidizes fatty acids with specific expression of uncoupling protein-1 [21]. Most studies focus on ADSCs from white fat. Recently, two groups of scientists demonstrated a population of ADSCs from human brown adipose tissue [22, 23]. Silva et al found that ADSCs from adult mediastinal fat were able to differentiate toward both white and brown adipocytes. In addition, these cells expressed higher level of transmembrane protein 26 (TMEM26) and CD137 than white ADSCs [22]. On the contrary, human fetal brown ADSCs only differentiated into classical brown adipocytes with low expression of CD137. Subcutaneous fat and visceral fat are the two main white adipose tissues. They were structurally and functionally different due to distinct characteristics biologically and functionally [24]. In the aspect of yield, the frequency of SVF cells in omental adipose tissue was significantly higher than that in subcutaneous adipose tissue [25]. In addition, it was noted that ADSCs isolated from each gram of visceral fat had more colony forming units than those from subcutaneous fat, implying that the visceral adipose tissue contained more ADSCs [26]. These differences may attribute to different sources of fat tissue. Not only characteristics of ADSCs vary from region to region, but also in one adipose tissue ADSCs display distinct features from subpopulation to subpopulation. A recent single-cell RNA sequencing article identified three major mesenchymal cell populations in the adipose tissue, which were DPP4+, ICAM1+ and CD142+ ADSCs in visceral adipose tissue of obese mice. Each of them had unique biological properties compared to other two subpopulations [27]. Above data demonstrates that ADSCs from different origins or even different subpopulations present distinct characteristics.
In the present article, we will compare the profiles of ADSCs from multiple origins and further discuss their biological functions in vascular remodeling.
Subcutaneous adipose-derived stem cells
Subcutaneous ADSCs were one of the most commonly used cells in ADSCs research. Subcutaneous fat makes up about 80% of the whole body fat [28]. Subcutaneous ADSCs can differentiate into ECs in vitro [9, 29–31]. CD13+CD34+ cells from subcutaneous SVF cultured in semisolid medium expressed CD31 and vWF. Further experiment demonstrated that these cells could form vessel-like structure by applying matrigel-plug assay subcutaneously in mice [31]. Similar findings were reported in CD34+CD31- [30], Flk-1+ [29] and CD31-CD34-c-kit- [32] subcutaneous ADSCs. Subcutaneous ADSCs also undergo endothelial differentiation in vivo. In a rat ischemic hindlimb model, injection of subcutaneous SVF cells could recover vascular supply and regenerate numerous CD31+ cells lining vessels [31]. Such effect of subcutaneous ADSCs was also confirmed by Cao et al [33] and Moon et al [34]. However, Nakagami et al claimed that injection of subcutaneous ADSCs did not express CD31 and von Willebrand factor in ischemic tissue in their model [32]. Another report showed that promoters of CD31 and CD144 in ADSCs were still methylated in response to endothelial growth factors, suggesting that ADSCs possessed the limited differentiation ability toward ECs lineage [35, 36]. Therefore, whether subcutaneous ADSCs can form functional endothelial cells remains unclear.
To understand the mechanism of ECs differentiation, Zhang et al confirmed that the inhibition of PI3K pathway reduced expression of CD31 genetically as well as capillary density in subcutaneous ADSCs, while inhibition of MAPK did not have such effect [37]. Consistent results were also observed by Cao et al, indicating that PI3K was a key point for ADSCs differentiation toward ECs [33].
ADSCs could also differentiate into smooth muscle cells [38–40]. Rodriguez et al reported that processed lipoaspirate cells exhibited typical SMCs morphology and upregulated the expression of SMCs markers at both transcriptional and translation levels when cultured in SMCs induction media [39]. Specific vascular SMCs-like ion channels were also identified in ADSCs treated by TGF-β1 [41]. In vivo, human ADSCs expressed alpha-smooth muscle actin and could survive for several months in the lower urinary tract of immunodeficient mice [38]. These results indicated that ADSCs could give rise to SMCs both in vitro and in vivo. However, compared to the ECs, the studies of SMCs differentiation in subcutaneous ADSCs are still limited.
In terms of SMCs differentiation from ADSCs, various potential mechanisms behind cell differentiation were reported [42]. TGF-β signaling pathway was one of them. Many downstream proteins were involved including mitogen activated protein kinase (MEK)/extracellular signal regulated kinase (ERK), c-Jun N-terminal kinase (JNK) and Smad2/3 [42]. Sphingosylphosphorylcholine (SPC) and sphingosine-1-phosphate were also referred to induce the differentiation of ADSCs into SMCs-like cells through G-protein coupled receptor by activating MEK/ERK signalling cascades [43], ras homolog gene family member A (RhoA)/Rho kinase mechanism [44] and TGF-β1 pathway [43, 45]. Autocrine TGF-β1/Smad2 pathway was activated in ADSCs in response to angiotensin-II when differentiated into contractile smooth muscle-like cells [46]. The activation of Notch pathway was also identified [47]. Additionally, bone morphogenetic protein-4 [48] and Med23 [49] were strongly related with the initiation of SMCs differentiation. Deleting Med23 in ADSCs increased SMCs marker myosin regulatory light polypeptide 9 (Myl9). Activin A was indispensable in the early stages of ADSCs differentiation into vascular SMCs when cultured with ECs [50]. However, induction of activin A secretion after stimulating ADSCs towards SMCs diminished ADSCs’ vasculogenic activity [51]. The summary of mechanisms of ADSCs differentiation into SMCs is displayed in Figure 1.
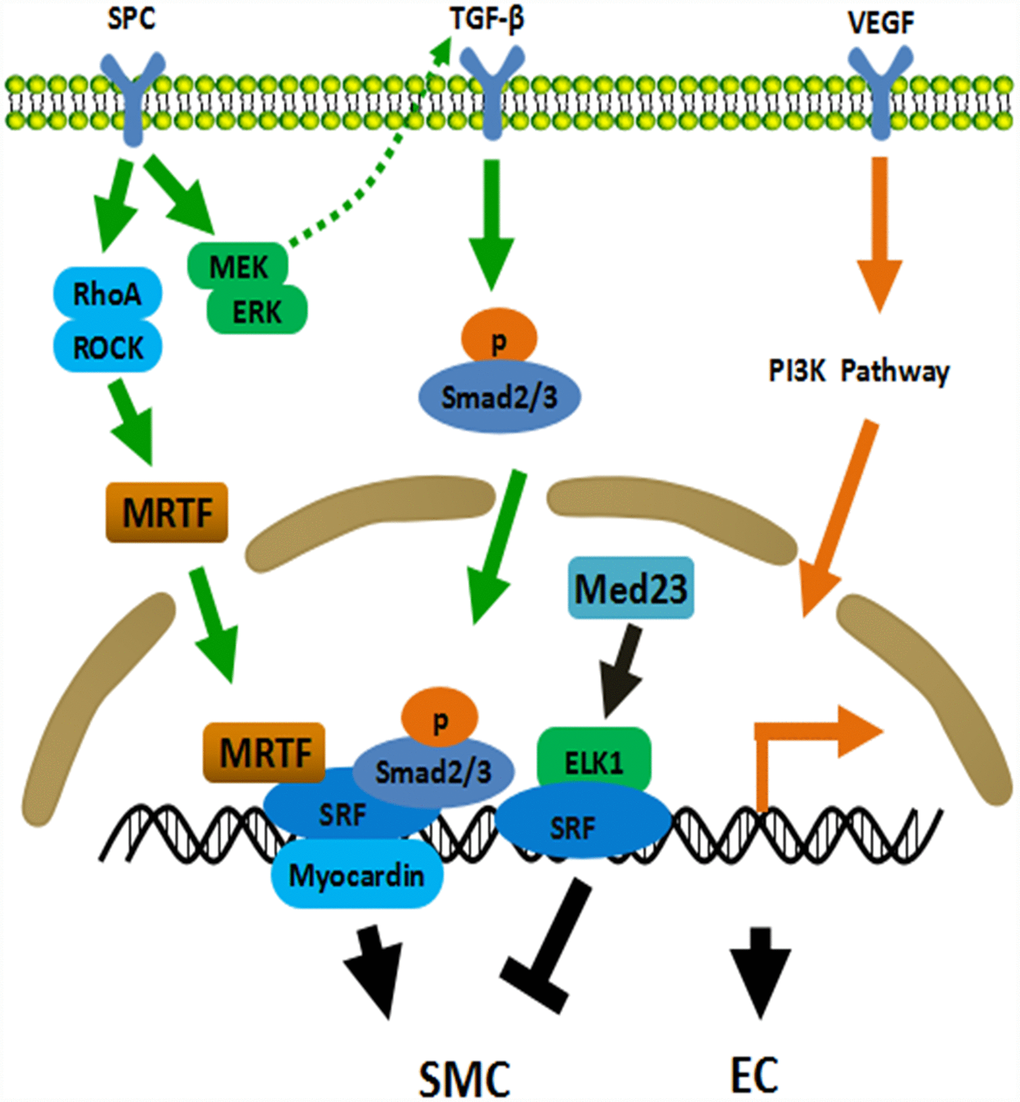
Figure 1. Mechanisms of ADSCs differentiation into SMC and EC. TGF-β pathway plays a central role in the differentiation of ADSCs into SMC. ADSCs secrete TGF-β through MEK/ERK-dependent mechanism when treated with SPC. Meanwhile, SPC activates the Rho/ROCK system and subsequently promotes the binding of MRTF and SRF. MRTF, SRF, Smad2/3 and Myocardin jointly initiate the expression of smooth muscle related genes. Med23 represses SMC differentiation via promoting ELK1-SRF to combine with the promoter. PI3K pathway involves in EC differentiation of ADSCs. EC: endothelial cell, SMC: smooth muscle cell, ROCK: Rho-associated protein kinase, SRF: serum response factor, MRTF: myocardin related transcription factor, ELK1: ETS Like-1 protein.
ADSCs can differentiate to cardiomyocytes as well. Planat-Bénard et al obtained functional cardiomyocyte-like cells from ADSCs [52], which were subsequently applied in models of chronic myocardial infarction in rats [53] and nonhuman primates [54], significantly improving heart function. In addition, ADSCs were believed to possess therapeutic effect in acute myocardial infarction [55] and chronic heart failure [56]. Angiogenesis was commonly observed in these disease models facilitated by a series of paracrine factors secreted from ADSCs [57]. miRNAs derived from ADSCs were found to be involved in the process of neovascularization [58].
ADSCs have a great potential in vascular tissue engineering based on their multiple differentiation abilities. A small artificial blood vessel was successfully manufactured by seeding SMCs-differentiated ADSCs on the scaffold [59]. After the application of pulsatile stimulation, the collagen content and biomechanical indicators of artificial vascular wall were significantly improved. Nevertheless, only the medial layer of blood vessels was produced. The feasibility and safety of these vascular grafts also required further investigation. Another research group constructed a two-layered small-diameter blood vessel by applying SMCs and ECs both differentiated from ADSCs [60]. Mechanical stimulation enhanced SMCs generation from ADSCs, which provided a novel method in vascular tissue engineering [61]. ADSCs from different donors varied from on the differentiation and migration of SMCs in tissue engineered blood vessel construction. Diabetic donors showed impaired differentiation abilities of ADSCs while different gender or body-mass index had no such effect [62]. ADSCs from the obese diminished the pro-angiogenic potential owing to reduced expression of miR-126 through inhibition of ERK1/2 MAPK pathway [63]. Another article reported that diabetic ADSCs led to increased inflammation in adipose tissue [64], which may impair vascular remodeling. Besides, ADSCs from the aged failed to facilitate both SMCs differentiation and migration [62]. Cryopreservation of ADSCs also attenuated their differentiation towards SMCs-like cells [65]. Above findings provide new ideas for the application of adipose-derived stem cells in vascular engineering.
ADSCs also have therapeutic effect on abdominal aortic aneurysm treatment via their immunoregulatory capacity, trophic factor production and extracellular matrix synthesis [66]. Xie et al performed tail-vein injection of ADSCs in mice with abdominal aortic aneurysm and found increased aortic FoxP3+ regulatory T cells and M2 macrophages, but decreased neutrophils, CD28− T cells and circulating monocytes, suggesting that ADSCs had anti-inflammatory capability. Moreover, they confirmed that the immunosuppression was mainly mediated through paracrine factors secreted by ADSCs [67]. Despite limited application in aneurysm, ADSCs will be promising alternatives from the perspective of vascular tissue engineering [68].
Abdominal (visceral) adipose-derived stem cells
Subcutaneous and visceral ADSCs displayed inherently different potentials in proliferation and differentiation, even though both of them were able to generate specific cells under corresponding culture conditions [69, 70]. For instance, subcutaneous ADSCs were better and more easily differentiated to mature adipocytes than visceral ADSCs in vitro [71, 72]. However, no agreement was reached with respect to the trends for proliferation and differentiation since the results between different study groups seemed contradictory [25, 73, 74]. This may result from inconsistent methods they adopted, such as culture conditions, passage number and donor populations. One article pointed out that visceral ADSCs was a more appropriate cell model in vitro for the investigation of molecular mechanisms of metabolic disorders such as obesity because visceral adipose tissue was closely associated with metabolic process [69]. Cells from the superficial layer (subcutaneous) had a greater proliferative rate and induced more outgrowth of neurite-like processes than those from the deep layer [75]. Wee Kiat Ong et al comprehensively screened the cell-surface markers of subcutaneous and visceral ADSCs. The expression of two cell-surface markers, CD10 and CD200, were pointed out to be associated with adipogenic capacity [76]. They discovered that CD10 was specifically expressed in subcutaneous ADSCs and CD200 was predominantly expressed in visceral ADSCs, which could be the specific markers for those ADSCs from different locations [76]. Above researches demonstrated that visceral ADSCs and subcutaneous ADSCs shared a lot of similarities but also had many differences.
Madonna et al described that murine visceral ADSCs in methylcellulose medium could spontaneously undergo neovascularization differentiation, forming CD31+CD34+ tube-like structures [77]. Another group focused on the adipose tissue-derived myogenic cells. They identified a subpopulation of myogenic cells from the rat visceral ADSCs, and discovered coexpressing telomerase and myocardin A with enhanced abilities of proliferation and differentiation. Further experiments confirmed that myocardin A helped maintain the myogenic stemness of visceral ADSCs through the upregulation of telomerase activation and enhancing myogenic gene expression [78]. The differences of subcutaneous, preperitoneal and visceral ADSCs from morbidly obese women were investigated. Visceral ADSCs secreted the highest levels of IL (interleukin)-6 and MCP (monocyte chemoattractant protein)-1, indicating that they had the most pro-inflammatory effect. On the contrary, preperitoneal ADSCs showed less pro-inflammatory features although they were from internal adipose depot [79].
Due to the promising conclusion from in vitro experiments, visceral ADSCs were exploited therapeutically. KDR+CD34−CD31− cells isolated from human visceral adipose tissue and ADSCs from murine adipose tissue were intravenously and intramuscularly respectively injected to the surgical mice with femoral artery ligation [33, 80]. Both groups concluded the enhanced capillary density and Doppler tissue perfusion scores. Others applied visceral ADSCs in acute and chronic animal models of myocardial infarction [81]. They transplanted rat visceral ADSCs intramuscularly into the rat with left anterior descending coronary artery ligation. Left ventricular end-diastolic volume, left ventricular ejection fraction and cardiac output were improved in rats received ADSCs therapy [53, 82]. Although many in vivo experiments have been done, the mechanism whereby ADSCs improved cardiac or vascular functions remained poorly understood. Additionally, whether to use raw or purified ADSCs such as c-kit+CD34+ was still under debate. At last, data from large animals such as the dogs and the primates were limited. Metabolic dysfunction leads to many diseases such as atherosclerosis and diabetes mellitus. Silvana Baglioni et al believed that the metabolic dysfunctions were related to ADSCs. They assessed the abilities of proliferation and differentiation of subcutaneous ADSCs and visceral ADSCs from the perspective of electrophysiological properties and functional activities. Visceral ADSCs showed less membrane potential, capacitance and K+-current parameters, as well as less adiponectin secretion and susceptibility to lipolysis. Such differences may contribute to the development of many metabolic-related diseases [24].
Other adipose-derived stem cells
Most of the literatures paid heavily attention on the identification and function of subcutaneous and visceral ADSCs for their easy access. Only little effort has been carried out on ADSCs from perivascular, cardiac and other regions. Theoretically, adipose tissue could be adjacent to all vessels and organs except for central nerve system. Such adipose tissues anatomically contact the adventitial side of the arteries and organs, which may play a more important role in vascular remodeling. Mihaela Crisan et al firstly confirmed a perivascular origin for mesenchymal stem cells in multiple human organs, such as skeletal muscle, pancreas, adipose tissue, and placenta, with the absence of expression of hematopoietic, endothelial, and myogenic cell markers. They also identified the multilineage potentials including osteogenic, chondrogenic, and adipogenic potentials but did not further study the relationship between these perivascular MSCs and vascular remodeling [83]. G. Lin et al also succeeded to identify ADSCs from perivascular location where they seemed to express both CD34 and smooth muscle actin. This research demonstrated a more precise location of ADSCs within in human adipose tissue by employing immunofluorescence of SSEA (stage-specific embryonic antigen) 1, STRO-1 and OCT (octamer-binding transcription factor)-4. Based on the results gained above, they proposed that ADSCs were either subsets of pericytes or vascular progenitors surrounding around the vessels [84]. Meanwhile, a population of CD34+ ADSCs, which expressed pericyte and MSCs markers, was discovered in periendothelial location, participating endothelial stabilization. These CD31-CD144-CD34+ cells localized in pericytic position and the functional analysis revealed that these cells were associated with vascular structures. The effect of vascular stabilization by CD31-CD144-CD34+ cells was achieved by bidirectional paracrine interaction with endothelial cells, such as VEGF, IL-6, IL-8 and MCP-1. In addition, a substantial proliferative response was detected in these cells when treated with FGF, EGF and PDGF-BB which were all produced by endothelial cells, suggesting a potential interaction with endothelial cells in vascular remodeling [85]. Another study revealed the expression of STRO-1, 3G5 and CD146 in MSCs around the perivascular regions of blood vessels in human adipose tissue sections. However, further relationship between MSCs and vascular function was not discussed [86]. Investigation into adipocyte progenitors revealed that preadipocytes localized to pericytes and endothelial cells of the blood vessels within adipose tissue which shed light on a developmental relationship between these cells [87]. Single-cell transcriptional analysis identified four mesenchymal stem-like cells populations locating in the adipose tissue of the perivascular niche. However, they did not investigate the effect of vascular remodeling for each subpopulation [88]. Overall, the existence of perivascular adipose tissue-derived stem cells has been confirmed but their functions and properties remain unknown where many scientists are currently paying more and more attention.
Cardiac adipose-derived stem cells, another novel type of stem cells separated from adipose tissue surrounding the heart, have been uncovered to be conducive to cardiovascular remodeling over recent years. In accordance to two different adipose depots, cardiac adipose-derived stem cells can be divided into epicardial and pericardial ADSCs. In comparison with adipose stem cells from other sources, cardiac ADSCs were prone to differentiate prominently into cardiovascular cells [89]. What is more, it was known that epicardial ADSCs embraced higher cardiomyogenic potential than pericardial ADSCs [90]. Of note, electrical and mechanical stimulation could strengthen the expression of several specific genes that were worthwhile for cardiodifferentiation [91, 92]. Up to now, it has been continuously reported that cardiac ADSCs exerted advantageous effect on cardiac function improvement and angiogenesis in experimental animal models of myocardial infarction. There was a research showing that perivascular ADSCs provided more potent cardiac reparative activity than subcutaneous ADSCs in view of their intrinsic properties toward myogenic differentiation and vasculogenesis [93]. Furthermore, injury-induced perivascular ADSCs might promote this process as a result of extra HGF production [94]. Angiogenesis is essential for cardiac repair in ischemic heart disease. Epicardial ADSCs isolated from samples within cardiovascular risk factors such as obese, hyperlipidemic and type-2 diabetic obviously impeded vessel formation [95], reminding us that selection of suitable tissue was vital for cell therapies. Differentiation and maturation of the transplanted cells collaborated with more relevant paracrine effect were considered to account for angiogenesis [96]. Agreed with this mechanistic basis, Wang et al speculated that newly formed cardiomyocytes might be partially derived from self-replicating cardiac cells albeit the instructive signals between ADSCs and cardiomyocytes remained unclear [97]. Taken together, cardiac ADSCs represent promising candidates for future use in cardiovascular regeneration therapies even though these cells are not easily accessible [98].
Adipose-derived stem cells around other regions, despite being rarely discussed, are of great value, which calls for extensive and in-depth studies to explore their physiological and pathological mechanisms.
Summary
Stem cell therapy presents a promising future in the field of regenerative medicine. Generating a large number of cardiovascular cell lineages is a key step in cell therapy of cardiovascular disease. ADSCs are among one of the most promising cell types for translational medicine and provide unprecedented opportunities for their easier isolation and less ethical aspects. In addition to differentiation into osteoblasts, cartilage and fat cells, ADSCs have been shown to differentiate into endothelial cells, smooth muscle cells and cardiomyocytes. The latter three are closely related to cardiovascular remodeling and disease progression. Previous studies demonstrated that different sources of ADSCs have different proliferative and differentiation abilities. In this review, we roughly divided ADSCs into subcutaneous, visceral and other ADSCs based on their origins. The properties of three different ADSCs were carefully compared to each other. This is also the first review of the relationship between cardiovascular remodeling and ADSCs from different origins. Although there are still many obstacles to overcome, we hope that clinical application of ADSCs will be widely used in the near future.
Conflicts of Interest
The authors declare that they have no competing interests.
Funding
This research was supported by the grant from The National Natural Science Foundation of China (NSFC) [grant number: 81800427(Yao Xie), 81870203 (Meixiang Xiang)] and The Medical and Health Research Fund of Zhejiang Province [grant number: 2015KYB177 (Yiming Zhao)].
References
- 1. Afshin A, Forouzanfar MH, Reitsma MB, Sur P, Estep K, Lee A, Marczak L, Mokdad AH, Moradi-Lakeh M, Naghavi M, Salama JS, Vos T, Abate KH, et al, and GBD 2015 Obesity Collaborators. Health effects of overweight and obesity in 195 countries over 25 years. N Engl J Med. 2017; 377:13–27. https://doi.org/10.1056/NEJMoa1614362 [PubMed]
- 2. Elagizi A, Kachur S, Lavie CJ, Carbone S, Pandey A, Ortega FB, Milani RV. An overview and update on obesity and the obesity paradox in cardiovascular diseases. Prog Cardiovasc Dis. 2018; 61:142–50. https://doi.org/10.1016/j.pcad.2018.07.003 [PubMed]
- 3. Bozzetto L, Costabile G, Della Pepa G, Ciciola P, Vetrani C, Vitale M, Rivellese AA, Annuzzi G. Dietary fibre as a unifying remedy for the whole spectrum of obesity-associated cardiovascular risk. Nutrients. 2018; 10:943. https://doi.org/10.3390/nu10070943 [PubMed]
- 4. Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001; 7:211–28. https://doi.org/10.1089/107632701300062859 [PubMed]
- 5. Hass R, Kasper C, Böhm S, Jacobs R. Different populations and sources of human mesenchymal stem cells (MSC): A comparison of adult and neonatal tissue-derived MSC. Cell Commun Signal. 2011; 9:12. https://doi.org/10.1186/1478-811X-9-12 [PubMed]
- 6. Fraser JK, Wulur I, Alfonso Z, Hedrick MH. Fat tissue: an underappreciated source of stem cells for biotechnology. Trends Biotechnol. 2006; 24:150–54. https://doi.org/10.1016/j.tibtech.2006.01.010 [PubMed]
- 7. Cho KA, Park M, Kim YH, Woo SY, Ryu KH. RNA sequencing reveals a transcriptomic portrait of human mesenchymal stem cells from bone marrow, adipose tissue, and palatine tonsils. Sci Rep. 2017; 7:17114. https://doi.org/10.1038/s41598-017-16788-2 [PubMed]
- 8. Mizuno H, Tobita M, Uysal AC. Concise review: adipose-derived stem cells as a novel tool for future regenerative medicine. Stem Cells. 2012; 30:804–10. https://doi.org/10.1002/stem.1076 [PubMed]
- 9. Gimble JM, Katz AJ, Bunnell BA. Adipose-derived stem cells for regenerative medicine. Circ Res. 2007; 100:1249–60. https://doi.org/10.1161/01.RES.0000265074.83288.09 [PubMed]
- 10. Bora P, Majumdar AS. Adipose tissue-derived stromal vascular fraction in regenerative medicine: a brief review on biology and translation. Stem Cell Res Ther. 2017; 8:145. https://doi.org/10.1186/s13287-017-0598-y [PubMed]
- 11. Sun Y, Chen S, Zhang X, Pei M. Significance of Cellular Cross-Talk in Stromal Vascular Fraction of Adipose Tissue in Neovascularization. Arterioscler Thromb Vasc Biol. 2019; 39:1034–44. https://doi.org/10.1161/ATVBAHA.119.312425 [PubMed]
- 12. Mitchell JB, McIntosh K, Zvonic S, Garrett S, Floyd ZE, Kloster A, Di Halvorsen Y, Storms RW, Goh B, Kilroy G, Wu X, Gimble JM. Immunophenotype of human adipose-derived cells: temporal changes in stromal-associated and stem cell-associated markers. Stem Cells. 2006; 24:376–85. https://doi.org/10.1634/stemcells.2005-0234 [PubMed]
- 13. Prins HJ, Schulten EA, Ten Bruggenkate CM, Klein-Nulend J, Helder MN. Bone Regeneration Using the Freshly Isolated Autologous Stromal Vascular Fraction of Adipose Tissue in Combination With Calcium Phosphate Ceramics. Stem Cells Transl Med. 2016; 5:1362–74. https://doi.org/10.5966/sctm.2015-0369 [PubMed]
- 14. Salehi H, Amirpour N, Niapour A, Razavi S. An overview of neural differentiation potential of human adipose derived stem cells. Stem Cell Rev Rep. 2016; 12:26–41. https://doi.org/10.1007/s12015-015-9631-7 [PubMed]
- 15. Kim JH, Park Y, Jung Y, Kim SH, Kim SH. Combinatorial therapy with three-dimensionally cultured adipose-derived stromal cells and self-assembling peptides to enhance angiogenesis and preserve cardiac function in infarcted hearts. J Tissue Eng Regen Med. 2017; 11:2816–27. https://doi.org/10.1002/term.2181 [PubMed]
- 16. Vilahur G, Oñate B, Cubedo J, Béjar MT, Arderiu G, Peña E, Casaní L, Gutiérrez M, Capdevila A, Pons-Lladó G, Carreras F, Hidalgo A, Badimon L. Allogenic adipose-derived stem cell therapy overcomes ischemia-induced microvessel rarefaction in the myocardium: systems biology study. Stem Cell Res Ther. 2017; 8:52. https://doi.org/10.1186/s13287-017-0509-2 [PubMed]
- 17. Luo Q, Guo D, Liu G, Chen G, Hang M, Jin M. Exosomes from MiR-126-overexpressing Adscs are therapeutic in relieving acute myocardial ischaemic injury. Cell Physiol Biochem. 2017; 44:2105–16. https://doi.org/10.1159/000485949 [PubMed]
- 18. Chen C, Tang Q, Zhang Y, Dai M, Jiang Y, Wang H, Yu M, Jing W, Tian W. Metabolic reprogramming by HIF-1 activation enhances survivability of human adipose-derived stem cells in ischaemic microenvironments. Cell Prolif. 2017; 50:e12363. https://doi.org/10.1111/cpr.12363 [PubMed]
- 19. Dubey NK, Mishra VK, Dubey R, Deng YH, Tsai FC, Deng WP. Revisiting the Advances in Isolation, Characterization and Secretome of Adipose-Derived Stromal/Stem Cells. Int J Mol Sci. 2018; 19:E2200. https://doi.org/10.3390/ijms19082200 [PubMed]
- 20. Dykstra JA, Facile T, Patrick RJ, Francis KR, Milanovich S, Weimer JM, Kota DJ. Concise Review: Fat and Furious: Harnessing the Full Potential of Adipose-Derived Stromal Vascular Fraction. Stem Cells Transl Med. 2017; 6:1096–108. https://doi.org/10.1002/sctm.16-0337 [PubMed]
- 21. Lazar MA. Developmental biology. How now, brown fat? Science. 2008; 321:1048–49. https://doi.org/10.1126/science.1164094 [PubMed]
- 22. Silva FJ, Holt DJ, Vargas V, Yockman J, Boudina S, Atkinson D, Grainger DW, Revelo MP, Sherman W, Bull DA, Patel AN. Metabolically active human brown adipose tissue derived stem cells. Stem Cells. 2014; 32:572–81. https://doi.org/10.1002/stem.1595 [PubMed]
- 23. Di Franco A, Guasti D, Squecco R, Mazzanti B, Rossi F, Idrizaj E, Gallego-Escuredo JM, Villarroya F, Bani D, Forti G, Vannelli GB, Luconi M. Searching for Classical Brown Fat in Humans: Development of a Novel Human Fetal Brown Stem Cell Model. Stem Cells. 2016; 34:1679–91. https://doi.org/10.1002/stem.2336 [PubMed]
- 24. Baglioni S, Cantini G, Poli G, Francalanci M, Squecco R, Di Franco A, Borgogni E, Frontera S, Nesi G, Liotta F, Lucchese M, Perigli G, Francini F, et al. Functional differences in visceral and subcutaneous fat pads originate from differences in the adipose stem cell. PLoS One. 2012; 7:e36569. https://doi.org/10.1371/journal.pone.0036569 [PubMed]
- 25. Russo V, Yu C, Belliveau P, Hamilton A, Flynn LE. Comparison of human adipose-derived stem cells isolated from subcutaneous, omental, and intrathoracic adipose tissue depots for regenerative applications. Stem Cells Transl Med. 2014; 3:206–17. https://doi.org/10.5966/sctm.2013-0125 [PubMed]
- 26. Chi C, Wang F, Xiang B, Deng J, Liu S, Lin HY, Natarajan K, Li G, Wang L, Wang J, Lin F, Freed DH, Arora RC, et al. Adipose-derived stem cells from both visceral and subcutaneous fat deposits significantly improve contractile function of infarcted rat hearts. Cell Transplant. 2015; 24:2337–51. https://doi.org/10.3727/096368914X685780 [PubMed]
- 27. Merrick D, Sakers A, Irgebay Z, Okada C, Calvert C, Morley MP, Percec I, Seale P. Identification of a mesenchymal progenitor cell hierarchy in adipose tissue. Science. 2019; 364:eaav2501. https://doi.org/10.1126/science.aav2501 [PubMed]
- 28. Ibrahim MM. Subcutaneous and visceral adipose tissue: structural and functional differences. Obes Rev. 2010; 11:11–18. https://doi.org/10.1111/j.1467-789X.2009.00623.x [PubMed]
- 29. Martínez-Estrada OM, Muñoz-Santos Y, Julve J, Reina M, Vilaró S. Human adipose tissue as a source of Flk-1+ cells: new method of differentiation and expansion. Cardiovasc Res. 2005; 65:328–33. https://doi.org/10.1016/j.cardiores.2004.11.015 [PubMed]
- 30. Miranville A, Heeschen C, Sengenès C, Curat CA, Busse R, Bouloumié A. Improvement of postnatal neovascularization by human adipose tissue-derived stem cells. Circulation. 2004; 110:349–55. https://doi.org/10.1161/01.CIR.0000135466.16823.D0 [PubMed]
- 31. Planat-Benard V, Silvestre JS, Cousin B, André M, Nibbelink M, Tamarat R, Clergue M, Manneville C, Saillan-Barreau C, Duriez M, Tedgui A, Levy B, Pénicaud L, Casteilla L. Plasticity of human adipose lineage cells toward endothelial cells: physiological and therapeutic perspectives. Circulation. 2004; 109:656–63. https://doi.org/10.1161/01.CIR.0000114522.38265.61 [PubMed]
- 32. Nakagami H, Maeda K, Morishita R, Iguchi S, Nishikawa T, Takami Y, Kikuchi Y, Saito Y, Tamai K, Ogihara T, Kaneda Y. Novel autologous cell therapy in ischemic limb disease through growth factor secretion by cultured adipose tissue-derived stromal cells. Arterioscler Thromb Vasc Biol. 2005; 25:2542–47. https://doi.org/10.1161/01.ATV.0000190701.92007.6d [PubMed]
- 33. Cao Y, Sun Z, Liao L, Meng Y, Han Q, Zhao RC. Human adipose tissue-derived stem cells differentiate into endothelial cells in vitro and improve postnatal neovascularization in vivo. Biochem Biophys Res Commun. 2005; 332:370–79. https://doi.org/10.1016/j.bbrc.2005.04.135 [PubMed]
- 34. Moon MH, Kim SY, Kim YJ, Kim SJ, Lee JB, Bae YC, Sung SM, Jung JS. Human adipose tissue-derived mesenchymal stem cells improve postnatal neovascularization in a mouse model of hindlimb ischemia. Cell Physiol Biochem. 2006; 17:279–90. https://doi.org/10.1159/000094140 [PubMed]
- 35. Boquest AC, Noer A, Sørensen AL, Vekterud K, Collas P. CpG methylation profiles of endothelial cell-specific gene promoter regions in adipose tissue stem cells suggest limited differentiation potential toward the endothelial cell lineage. Stem Cells. 2007; 25:852–61. https://doi.org/10.1634/stemcells.2006-0428 [PubMed]
- 36. Bajpai VK, Andreadis ST. Stem cell sources for vascular tissue engineering and regeneration. Tissue Eng Part B Rev. 2012; 18:405–25. https://doi.org/10.1089/ten.teb.2011.0264 [PubMed]
- 37. Zhang P, Moudgill N, Hager E, Tarola N, Dimatteo C, McIlhenny S, Tulenko T, DiMuzio PJ. Endothelial differentiation of adipose-derived stem cells from elderly patients with cardiovascular disease. Stem Cells Dev. 2011; 20:977–88. https://doi.org/10.1089/scd.2010.0152 [PubMed]
- 38. Jack GS, Almeida FG, Zhang R, Alfonso ZC, Zuk PA, Rodríguez LV. Processed lipoaspirate cells for tissue engineering of the lower urinary tract: implications for the treatment of stress urinary incontinence and bladder reconstruction. J Urol. 2005; 174:2041–45. https://doi.org/10.1097/01.ju.0000176489.96993.84 [PubMed]
- 39. Rodríguez LV, Alfonso Z, Zhang R, Leung J, Wu B, Ignarro LJ. Clonogenic multipotent stem cells in human adipose tissue differentiate into functional smooth muscle cells. Proc Natl Acad Sci USA. 2006; 103:12167–72. https://doi.org/10.1073/pnas.0604850103 [PubMed]
- 40. Jack GS, Zhang R, Lee M, Xu Y, Wu BM, Rodríguez LV. Urinary bladder smooth muscle engineered from adipose stem cells and a three dimensional synthetic composite. Biomaterials. 2009; 30:3259–70. https://doi.org/10.1016/j.biomaterials.2009.02.035 [PubMed]
- 41. Park WS, Heo SC, Jeon ES, Hong DH, Son YK, Ko JH, Kim HK, Lee SY, Kim JH, Han J. Functional expression of smooth muscle-specific ion channels in TGF-β(1)-treated human adipose-derived mesenchymal stem cells. Am J Physiol Cell Physiol. 2013; 305:C377–91. https://doi.org/10.1152/ajpcell.00404.2012 [PubMed]
- 42. Zhang X, Bendeck MP, Simmons CA, Santerre JP. Deriving vascular smooth muscle cells from mesenchymal stromal cells: evolving differentiation strategies and current understanding of their mechanisms. Biomaterials. 2017; 145:9–22. https://doi.org/10.1016/j.biomaterials.2017.08.028 [PubMed]
- 43. Jeon ES, Moon HJ, Lee MJ, Song HY, Kim YM, Bae YC, Jung JS, Kim JH. Sphingosylphosphorylcholine induces differentiation of human mesenchymal stem cells into smooth-muscle-like cells through a TGF-beta-dependent mechanism. J Cell Sci. 2006; 119:4994–5005. https://doi.org/10.1242/jcs.03281 [PubMed]
- 44. Jeon ES, Park WS, Lee MJ, Kim YM, Han J, Kim JH. A Rho kinase/myocardin-related transcription factor-A-dependent mechanism underlies the sphingosylphosphorylcholine-induced differentiation of mesenchymal stem cells into contractile smooth muscle cells. Circ Res. 2008; 103:635–42. https://doi.org/10.1161/CIRCRESAHA.108.180885 [PubMed]
- 45. Nincheri P, Luciani P, Squecco R, Donati C, Bernacchioni C, Borgognoni L, Luciani G, Benvenuti S, Francini F, Bruni P. Sphingosine 1-phosphate induces differentiation of adipose tissue-derived mesenchymal stem cells towards smooth muscle cells. Cell Mol Life Sci. 2009; 66:1741–54. https://doi.org/10.1007/s00018-009-9181-8 [PubMed]
- 46. Kim YM, Jeon ES, Kim MR, Jho SK, Ryu SW, Kim JH. Angiotensin II-induced differentiation of adipose tissue-derived mesenchymal stem cells to smooth muscle-like cells. Int J Biochem Cell Biol. 2008; 40:2482–91. https://doi.org/10.1016/j.biocel.2008.04.016 [PubMed]
- 47. Kurpinski K, Lam H, Chu J, Wang A, Kim A, Tsay E, Agrawal S, Schaffer DV, Li S. Transforming growth factor-β and notch signaling mediate stem cell differentiation into smooth muscle cells. Stem Cells. 2010; 28:734–42. https://doi.org/10.1002/stem.319 [PubMed]
- 48. Wang C, Yin S, Cen L, Liu Q, Liu W, Cao Y, Cui L. Differentiation of adipose-derived stem cells into contractile smooth muscle cells induced by transforming growth factor-beta1 and bone morphogenetic protein-4. Tissue Eng Part A. 2010; 16:1201–13. https://doi.org/10.1089/ten.tea.2009.0303 [PubMed]
- 49. Yin JW, Liang Y, Park JY, Chen D, Yao X, Xiao Q, Liu Z, Jiang B, Fu Y, Bao M, Huang Y, Liu Y, Yan J, et al. Mediator MED23 plays opposing roles in directing smooth muscle cell and adipocyte differentiation. Genes Dev. 2012; 26:2192–205. https://doi.org/10.1101/gad.192666.112 [PubMed]
- 50. Merfeld-Clauss S, Lupov IP, Lu H, Feng D, Compton-Craig P, March KL, Traktuev DO. Adipose stromal cells differentiate along a smooth muscle lineage pathway upon endothelial cell contact via induction of activin A. Circ Res. 2014; 115:800–09. https://doi.org/10.1161/CIRCRESAHA.115.304026 [PubMed]
- 51. Merfeld-Clauss S, Lease BR, Lu H, March KL, Traktuev DO. Adipose stromal cells differentiation toward smooth muscle cell phenotype diminishes their vasculogenic activity due to induction of activin A secretion. J Tissue Eng Regen Med. 2017; 11:3145–56. https://doi.org/10.1002/term.2223 [PubMed]
- 52. Planat-Bénard V, Menard C, André M, Puceat M, Perez A, Garcia-Verdugo JM, Pénicaud L, Casteilla L. Spontaneous cardiomyocyte differentiation from adipose tissue stroma cells. Circ Res. 2004; 94:223–29. https://doi.org/10.1161/01.RES.0000109792.43271.47 [PubMed]
- 53. Mazo M, Planat-Bénard V, Abizanda G, Pelacho B, Léobon B, Gavira JJ, Peñuelas I, Cemborain A, Pénicaud L, Laharrague P, Joffre C, Boisson M, Ecay M, et al. Transplantation of adipose derived stromal cells is associated with functional improvement in a rat model of chronic myocardial infarction. Eur J Heart Fail. 2008; 10:454–62. https://doi.org/10.1016/j.ejheart.2008.03.017 [PubMed]
- 54. Bel A, Planat-Bernard V, Saito A, Bonnevie L, Bellamy V, Sabbah L, Bellabas L, Brinon B, Vanneaux V, Pradeau P, Peyrard S, Larghero J, Pouly J, et al. Composite cell sheets: a further step toward safe and effective myocardial regeneration by cardiac progenitors derived from embryonic stem cells. Circulation. 2010 (11 Suppl); 122:S118–23. https://doi.org/10.1161/CIRCULATIONAHA.109.927293 [PubMed]
- 55. Cai L, Johnstone BH, Cook TG, Tan J, Fishbein MC, Chen PS, March KL. IFATS collection: human adipose tissue-derived stem cells induce angiogenesis and nerve sprouting following myocardial infarction, in conjunction with potent preservation of cardiac function. Stem Cells. 2009; 27:230–37. https://doi.org/10.1634/stemcells.2008-0273 [PubMed]
- 56. Ishida O, Hagino I, Nagaya N, Shimizu T, Okano T, Sawa Y, Mori H, Yagihara T. Adipose-derived stem cell sheet transplantation therapy in a porcine model of chronic heart failure. Transl Res. 2015; 165:631–39. https://doi.org/10.1016/j.trsl.2014.12.005 [PubMed]
- 57. Zhao L, Johnson T, Liu D. Therapeutic angiogenesis of adipose-derived stem cells for ischemic diseases. Stem Cell Res Ther. 2017; 8:125. https://doi.org/10.1186/s13287-017-0578-2 [PubMed]
- 58. Ma T, Sun J, Zhao Z, Lei W, Chen Y, Wang X, Yang J, Shen Z. A brief review: adipose-derived stem cells and their therapeutic potential in cardiovascular diseases. Stem Cell Res Ther. 2017; 8:124. https://doi.org/10.1186/s13287-017-0585-3 [PubMed]
- 59. Wang C, Cen L, Yin S, Liu Q, Liu W, Cao Y, Cui L. A small diameter elastic blood vessel wall prepared under pulsatile conditions from polyglycolic acid mesh and smooth muscle cells differentiated from adipose-derived stem cells. Biomaterials. 2010; 31:621–30. https://doi.org/10.1016/j.biomaterials.2009.09.086 [PubMed]
- 60. Zhou R, Zhu L, Fu S, Qian Y, Wang D, Wang C. Small diameter blood vessels bioengineered from human adipose-derived stem cells. Sci Rep. 2016; 6:35422. https://doi.org/10.1038/srep35422 [PubMed]
- 61. Parvizi M, Bolhuis-Versteeg LA, Poot AA, Harmsen MC. Efficient generation of smooth muscle cells from adipose-derived stromal cells by 3D mechanical stimulation can substitute the use of growth factors in vascular tissue engineering. Biotechnol J. 2016; 11:932–44. https://doi.org/10.1002/biot.201500519 [PubMed]
- 62. Krawiec JT, Weinbaum JS, St Croix CM, Phillippi JA, Watkins SC, Rubin JP, Vorp DA. A cautionary tale for autologous vascular tissue engineering: impact of human demographics on the ability of adipose-derived mesenchymal stem cells to recruit and differentiate into smooth muscle cells. Tissue Eng Part A. 2015; 21:426–37. https://doi.org/10.1089/ten.tea.2014.0208 [PubMed]
- 63. Togliatto G, Dentelli P, Gili M, Gallo S, Deregibus C, Biglieri E, Iavello A, Santini E, Rossi C, Solini A, Camussi G, Brizzi MF. Obesity reduces the pro-angiogenic potential of adipose tissue stem cell-derived extracellular vesicles (EVs) by impairing miR-126 content: impact on clinical applications. Int J Obes. 2016; 40:102–11. https://doi.org/10.1038/ijo.2015.123 [PubMed]
- 64. Stafeev I, Podkuychenko N, Michurina S, Sklyanik I, Panevina A, Shestakova E, Yah’yaev K, Fedenko V, Ratner E, Vorotnikov A, Menshikov M, Yashkov Y, Parfyonova Y, Shestakova M. Low proliferative potential of adipose-derived stromal cells associates with hypertrophy and inflammation in subcutaneous and omental adipose tissue of patients with type 2 diabetes mellitus. J Diabetes Complications. 2019; 33:148–59. https://doi.org/10.1016/j.jdiacomp.2018.10.011 [PubMed]
- 65. Zhang X, Simmons CA, Santerre JP. Alterations of MEK1/2-ERK1/2, IFNγ and Smad2/3 associated Signalling pathways during cryopreservation of ASCs affect their differentiation towards VSMC-like cells. Stem Cell Res. 2018; 32:115–25. https://doi.org/10.1016/j.scr.2018.09.005 [PubMed]
- 66. Parvizi M, Harmsen MC. Therapeutic Prospect of Adipose-Derived Stromal Cells for the Treatment of Abdominal Aortic Aneurysm. Stem Cells Dev. 2015; 24:1493–505. https://doi.org/10.1089/scd.2014.0517 [PubMed]
- 67. Xie J, Jones TJ, Feng D, Cook TG, Jester AA, Yi R, Jawed YT, Babbey C, March KL, Murphy MP. Human Adipose-Derived Stem Cells Suppress Elastase-Induced Murine Abdominal Aortic Inflammation and Aneurysm Expansion Through Paracrine Factors. Cell Transplant. 2017; 26:173–89. https://doi.org/10.3727/096368916X692212 [PubMed]
- 68. Wang Y, Yin P, Bian GL, Huang HY, Shen H, Yang JJ, Yang ZY, Shen ZY. The combination of stem cells and tissue engineering: an advanced strategy for blood vessels regeneration and vascular disease treatment. Stem Cell Res Ther. 2017; 8:194. https://doi.org/10.1186/s13287-017-0642-y [PubMed]
- 69. Baglioni S, Francalanci M, Squecco R, Lombardi A, Cantini G, Angeli R, Gelmini S, Guasti D, Benvenuti S, Annunziato F, Bani D, Liotta F, Francini F, et al. Characterization of human adult stem-cell populations isolated from visceral and subcutaneous adipose tissue. FASEB J. 2009; 23:3494–505. https://doi.org/10.1096/fj.08-126946 [PubMed]
- 70. Macotela Y, Emanuelli B, Mori MA, Gesta S, Schulz TJ, Tseng YH, Kahn CR. Intrinsic differences in adipocyte precursor cells from different white fat depots. Diabetes. 2012; 61:1691–99. https://doi.org/10.2337/db11-1753 [PubMed]
- 71. Tchkonia T, Tchoukalova YD, Giorgadze N, Pirtskhalava T, Karagiannides I, Forse RA, Koo A, Stevenson M, Chinnappan D, Cartwright A, Jensen MD, Kirkland JL. Abundance of two human preadipocyte subtypes with distinct capacities for replication, adipogenesis, and apoptosis varies among fat depots. Am J Physiol Endocrinol Metab. 2005; 288:E267–77. https://doi.org/10.1152/ajpendo.00265.2004 [PubMed]
- 72. Tang Y, Pan ZY, Zou Y, He Y, Yang PY, Tang QQ, Yin F. A comparative assessment of adipose-derived stem cells from subcutaneous and visceral fat as a potential cell source for knee osteoarthritis treatment. J Cell Mol Med. 2017; 21:2153–62. https://doi.org/10.1111/jcmm.13138 [PubMed]
- 73. Shahparaki A, Grunder L, Sorisky A. Comparison of human abdominal subcutaneous versus omental preadipocyte differentiation in primary culture. Metabolism. 2002; 51:1211–15. https://doi.org/10.1053/meta.2002.34037 [PubMed]
- 74. Van Harmelen V, Röhrig K, Hauner H. Comparison of proliferation and differentiation capacity of human adipocyte precursor cells from the omental and subcutaneous adipose tissue depot of obese subjects. Metabolism. 2004; 53:632–37. https://doi.org/10.1016/j.metabol.2003.11.012 [PubMed]
- 75. Kalbermatten DF, Schaakxs D, Kingham PJ, Wiberg M. Neurotrophic activity of human adipose stem cells isolated from deep and superficial layers of abdominal fat. Cell Tissue Res. 2011; 344:251–60. https://doi.org/10.1007/s00441-011-1142-5 [PubMed]
- 76. Ong WK, Tan CS, Chan KL, Goesantoso GG, Chan XH, Chan E, Yin J, Yeo CR, Khoo CM, So JB, Shabbir A, Toh SA, Han W, Sugii S. Identification of specific cell-surface markers of adipose-derived stem cells from subcutaneous and visceral fat depots. Stem Cell Reports. 2014; 2:171–79. https://doi.org/10.1016/j.stemcr.2014.01.002 [PubMed]
- 77. Madonna R, De Caterina R. In vitro neovasculogenic potential of resident adipose tissue precursors. Am J Physiol Cell Physiol. 2008; 295:C1271–80. https://doi.org/10.1152/ajpcell.00186.2008 [PubMed]
- 78. Madonna R, Willerson JT, Geng YJ. Myocardin a enhances telomerase activities in adipose tissue mesenchymal cells and embryonic stem cells undergoing cardiovascular myogenic differentiation. Stem Cells. 2008; 26:202–11. https://doi.org/10.1634/stemcells.2007-0490 [PubMed]
- 79. Silva KR, Côrtes I, Liechocki S, Carneiro JR, Souza AA, Borojevic R, Maya-Monteiro CM, Baptista LS. Characterization of stromal vascular fraction and adipose stem cells from subcutaneous, preperitoneal and visceral morbidly obese human adipose tissue depots. PLoS One. 2017; 12:e0174115. https://doi.org/10.1371/journal.pone.0174115 [PubMed]
- 80. Sumi M, Sata M, Toya N, Yanaga K, Ohki T, Nagai R. Transplantation of adipose stromal cells, but not mature adipocytes, augments ischemia-induced angiogenesis. Life Sci. 2007; 80:559–65. https://doi.org/10.1016/j.lfs.2006.10.020 [PubMed]
- 81. Schenke-Layland K, Strem BM, Jordan MC, Deemedio MT, Hedrick MH, Roos KP, Fraser JK, Maclellan WR. Adipose tissue-derived cells improve cardiac function following myocardial infarction. J Surg Res. 2009; 153:217–23. https://doi.org/10.1016/j.jss.2008.03.019 [PubMed]
- 82. Li B, Zeng Q, Wang H, Shao S, Mao X, Zhang F, Li S, Guo Z. Adipose tissue stromal cells transplantation in rats of acute myocardial infarction. Coron Artery Dis. 2007; 18:221–27. https://doi.org/10.1097/MCA.0b013e32801235da [PubMed]
- 83. Crisan M, Yap S, Casteilla L, Chen CW, Corselli M, Park TS, Andriolo G, Sun B, Zheng B, Zhang L, Norotte C, Teng PN, Traas J, et al. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell. 2008; 3:301–13. https://doi.org/10.1016/j.stem.2008.07.003 [PubMed]
- 84. Lin G, Garcia M, Ning H, Banie L, Guo YL, Lue TF, Lin CS. Defining stem and progenitor cells within adipose tissue. Stem Cells Dev. 2008; 17:1053–63. https://doi.org/10.1089/scd.2008.0117 [PubMed]
- 85. Traktuev DO, Merfeld-Clauss S, Li J, Kolonin M, Arap W, Pasqualini R, Johnstone BH, March KL. A population of multipotent CD34-positive adipose stromal cells share pericyte and mesenchymal surface markers, reside in a periendothelial location, and stabilize endothelial networks. Circ Res. 2008; 102:77–85. https://doi.org/10.1161/CIRCRESAHA.107.159475 [PubMed]
- 86. Zannettino AC, Paton S, Arthur A, Khor F, Itescu S, Gimble JM, Gronthos S. Multipotential human adipose-derived stromal stem cells exhibit a perivascular phenotype in vitro and in vivo. J Cell Physiol. 2008; 214:413–21. https://doi.org/10.1002/jcp.21210 [PubMed]
- 87. Gupta RK, Mepani RJ, Kleiner S, Lo JC, Khandekar MJ, Cohen P, Frontini A, Bhowmick DC, Ye L, Cinti S, Spiegelman BM. Zfp423 expression identifies committed preadipocytes and localizes to adipose endothelial and perivascular cells. Cell Metab. 2012; 15:230–39. https://doi.org/10.1016/j.cmet.2012.01.010 [PubMed]
- 88. Hardy WR, Moldovan NI, Moldovan L, Livak KJ, Datta K, Goswami C, Corselli M, Traktuev DO, Murray IR, Péault B, March K. Transcriptional Networks in Single Perivascular Cells Sorted from Human Adipose Tissue Reveal a Hierarchy of Mesenchymal Stem Cells. Stem Cells. 2017; 35:1273–89. https://doi.org/10.1002/stem.2599 [PubMed]
- 89. Nagata H, Ii M, Kohbayashi E, Hoshiga M, Hanafusa T, Asahi M. Cardiac adipose-derived stem cells exhibit high differentiation potential to cardiovascular cells in C57BL/6 mice. Stem Cells Transl Med. 2016; 5:141–51. https://doi.org/10.5966/sctm.2015-0083 [PubMed]
- 90. Wystrychowski W, Patlolla B, Zhuge Y, Neofytou E, Robbins RC, Beygui RE. Multipotency and cardiomyogenic potential of human adipose-derived stem cells from epicardium, pericardium, and omentum. Stem Cell Res Ther. 2016; 7:84. https://doi.org/10.1186/s13287-016-0343-y [PubMed]
- 91. Llucià-Valldeperas A, Sanchez B, Soler-Botija C, Gálvez-Montón C, Prat-Vidal C, Roura S, Rosell-Ferrer J, Bragos R, Bayes-Genis A. Electrical stimulation of cardiac adipose tissue-derived progenitor cells modulates cell phenotype and genetic machinery. J Tissue Eng Regen Med. 2015; 9:E76–83. https://doi.org/10.1002/term.1710 [PubMed]
- 92. Llucià-Valldeperas A, Bragós R, Soler-Botija C, Roura S, Gálvez-Montón C, Prat-Vidal C, Perea-Gil I, Bayes-Genis A. Unravelling the effects of mechanical physiological conditioning on cardiac adipose tissue-derived progenitor cells in vitro and in silico. Sci Rep. 2018; 8:499. https://doi.org/10.1038/s41598-017-18799-5 [PubMed]
- 93. Wang X, Zhang H, Nie L, Xu L, Chen M, Ding Z. Myogenic differentiation and reparative activity of stromal cells derived from pericardial adipose in comparison to subcutaneous origin. Stem Cell Res Ther. 2014; 5:92. https://doi.org/10.1186/scrt481 [PubMed]
- 94. Tang J, Wang X, Tan K, Zhu H, Zhang Y, Ouyang W, Liu X, Ding Z. Injury-induced fetal reprogramming imparts multipotency and reparative properties to pericardial adipose stem cells. Stem Cell Res Ther. 2018; 9:218. https://doi.org/10.1186/s13287-018-0959-1 [PubMed]
- 95. Bejar MT, Ferrer-Lorente R, Peña E, Badimon L. Inhibition of Notch rescues the angiogenic potential impaired by cardiovascular risk factors in epicardial adipose stem cells. FASEB J. 2016; 30:2849–59. https://doi.org/10.1096/fj.201600204R [PubMed]
- 96. Bayes-Genis A, Soler-Botija C, Farré J, Sepúlveda P, Raya A, Roura S, Prat-Vidal C, Gálvez-Montón C, Montero JA, Büscher D, Izpisúa Belmonte JC. Human progenitor cells derived from cardiac adipose tissue ameliorate myocardial infarction in rodents. J Mol Cell Cardiol. 2010; 49:771–80. https://doi.org/10.1016/j.yjmcc.2010.08.010 [PubMed]
- 97. Wang X, Liu X, Zhang H, Nie L, Chen M, Ding Z. Reconstitute the damaged heart via the dual reparative roles of pericardial adipose-derived flk-1+ stem cells. Int J Cardiol. 2016; 202:256–64. https://doi.org/10.1016/j.ijcard.2015.09.002 [PubMed]
- 98. Bayes-Genis A, Gálvez-Montón C, Prat-Vidal C, Soler-Botija C. Cardiac adipose tissue: a new frontier for cardiac regeneration? Int J Cardiol. 2013; 167:22–25. https://doi.org/10.1016/j.ijcard.2012.05.082 [PubMed]
- 99. Li CY, Wu XY, Tong JB, Yang XX, Zhao JL, Zheng QF, Zhao GB, Ma ZJ. Comparative analysis of human mesenchymal stem cells from bone marrow and adipose tissue under xeno-free conditions for cell therapy. Stem Cell Res Ther. 2015; 6:55. https://doi.org/10.1186/s13287-015-0066-5 [PubMed]
- 100. Nora CC, Camassola M, Bellagamba B, Ikuta N, Christoff AP, Meirelles LS, Ayres R, Margis R, Nardi NB. Molecular analysis of the differentiation potential of murine mesenchymal stem cells from tissues of endodermal or mesodermal origin. Stem Cells Dev. 2012; 21:1761–68. https://doi.org/10.1089/scd.2011.0030 [PubMed]
- 101. Saler M, Caliogna L, Botta L, Benazzo F, Riva F, Gastaldi G. hASC and DFAT, Multipotent Stem Cells for Regenerative Medicine: A Comparison of Their Potential Differentiation In Vitro. Int J Mol Sci. 2017; 18:2699. https://doi.org/10.3390/ijms18122699 [PubMed]
- 102. Griffin M, Ryan CM, Pathan O, Abraham D, Denton CP, Butler PE. Characteristics of human adipose derived stem cells in scleroderma in comparison to sex and age matched normal controls: implications for regenerative medicine. Stem Cell Res Ther. 2017; 8:23. https://doi.org/10.1186/s13287-016-0444-7 [PubMed]
- 103. Lee JH, Kemp DM. Human adipose-derived stem cells display myogenic potential and perturbed function in hypoxic conditions. Biochem Biophys Res Commun. 2006; 341:882–88. https://doi.org/10.1016/j.bbrc.2006.01.038 [PubMed]
- 104. Bayati V, Hashemitabar M, Gazor R, Nejatbakhsh R, Bijannejad D. Expression of surface markers and myogenic potential of rat bone marrow- and adipose-derived stem cells: a comparative study. Anat Cell Biol. 2013; 46:113–21. https://doi.org/10.5115/acb.2013.46.2.113 [PubMed]
- 105. Choi YS, Dusting GJ, Stubbs S, Arunothayaraj S, Han XL, Collas P, Morrison WA, Dilley RJ. Differentiation of human adipose-derived stem cells into beating cardiomyocytes. J Cell Mol Med. 2010; 14:878–89. https://doi.org/10.1111/j.1582-4934.2010.01009.x [PubMed]
- 106. Chang JC, Lee PC, Lin YC, Lee KW, Hsu SH. Primary adipose-derived stem cells enriched by growth factor treatment improves cell adaptability toward cardiovascular differentiation in a rodent model of acute myocardial infarction. J Stem Cells. 2011; 6:21–37. [PubMed]
- 107. Kim U, Shin DG, Park JS, Kim YJ, Park SI, Moon YM, Jeong KS. Homing of adipose-derived stem cells to radiofrequency catheter ablated canine atrium and differentiation into cardiomyocyte-like cells. Int J Cardiol. 2011; 146:371–78. https://doi.org/10.1016/j.ijcard.2009.07.016 [PubMed]
- 108. Kang SK, Putnam LA, Ylostalo J, Popescu IR, Dufour J, Belousov A, Bunnell BA. Neurogenesis of Rhesus adipose stromal cells. J Cell Sci. 2004; 117:4289–99. https://doi.org/10.1242/jcs.01264 [PubMed]
- 109. Krampera M, Marconi S, Pasini A, Galiè M, Rigotti G, Mosna F, Tinelli M, Lovato L, Anghileri E, Andreini A, Pizzolo G, Sbarbati A, Bonetti B. Induction of neural-like differentiation in human mesenchymal stem cells derived from bone marrow, fat, spleen and thymus. Bone. 2007; 40:382–90. https://doi.org/10.1016/j.bone.2006.09.006 [PubMed]
- 110. Ying C, Hu W, Cheng B, Zheng X, Li S. Neural differentiation of rat adipose-derived stem cells in vitro. Cell Mol Neurobiol. 2012; 32:1255–63. https://doi.org/10.1007/s10571-012-9850-2 [PubMed]
- 111. Lue J, Lin G, Ning H, Xiong A, Lin CS, Glenn JS. Transdifferentiation of adipose-derived stem cells into hepatocytes: a new approach. Liver Int. 2010; 30:913–22. https://doi.org/10.1111/j.1478-3231.2010.02231.x [PubMed]
- 112. Banas A, Teratani T, Yamamoto Y, Tokuhara M, Takeshita F, Quinn G, Okochi H, Ochiya T. Adipose tissue-derived mesenchymal stem cells as a source of human hepatocytes. Hepatology. 2007; 46:219–28. https://doi.org/10.1002/hep.21704 [PubMed]
- 113. Brzoska M, Geiger H, Gauer S, Baer P. Epithelial differentiation of human adipose tissue-derived adult stem cells. Biochem Biophys Res Commun. 2005; 330:142–50. https://doi.org/10.1016/j.bbrc.2005.02.141 [PubMed]
- 114. Dhanasekaran M, Indumathi S, Harikrishnan R, Mishra R, Lissa RP, Rajkumar JS, Sudarsanam D. Human omentum fat-derived mesenchymal stem cells transdifferentiates into pancreatic islet-like cluster. Cell Biochem Funct. 2013; 31:612–19. https://doi.org/10.1002/cbf.2948 [PubMed]



