Introduction
Percutaneous coronary intervention (PCI) has become an effective and safe method for revascularization in patients with coronary artery disease (CAD), especially in an elective setting [1]. However, the high incidence of periprocedural myocardial injury (PMI) is of great concern as emerging evidence suggests a link between PMI and mortality [2–4]. PMI is frequently attributed to myocardial damage via distal embolization, side-branch occlusion, and other unexpected procedural complications. As such, PMI is most likely to occur in patients with advanced lesions, complex anatomy, and difficult procedures [5].
Angiopoietins (Ang) are a family of growth factors that consist of four members: Ang-1, Ang-2, Ang-3, and Ang-4 [6]. Whereas Ang-1 maintains endothelial quiescence and integrity through binding the ligand of Tie2 receptor, Ang-2 acts as a context-dependent Tie2 antagonist, which inhibits the Ang-1-induced Tie2 phosphorylation, thus impairing endothelial integrity and promoting vascular leakage [7]. Ang-2 can be rapidly released from Weibel-Palade bodies of endothelial cells upon stimulation, such as hypoxia and inflammation [8, 9]. As an important proangiogenic factor, Ang-2 has been recently implicated in mediating inflammation [10–12] and atherosclerosis [13, 14]. Ang-2 is abundantly expressed in advanced human atherosclerotic lesions, and is associated with microvessel density [15, 16]. In patients with CAD, especially acute myocardial infarction (AMI), the circulating Ang-2 levels are significantly increased [17].
Previous studies have indicated an association between lesion-related risk factors and PMI [18]. The morphological characteristics of a vulnerable plaque phenotype, determined by intravascular imaging modalities, can predict the PMI occurrence [19–21]. Given the close relationship between Ang-2 levels, inflammation, and atherosclerosis, it is important to determine whether the circulating Ang-2 levels can predict the occurrence of PMI. Furthermore, previous studies have shown a significant association between mortality and circulating Ang-2 levels measured over time in multiple clinical scenarios [22–24]. We hypothesize that an increase in the circulating Ang-2 levels during hospitalization might be partly related to prominent myocardial injury, which contributes to poorer outcomes. Thus, this study aimed to evaluate the predictive value of circulating Ang-2 levels for PMI occurrence, and to investigate whether the changes in Ang-2 levels after PCI are influenced by PMI.
Results
PCI patients’ characteristics
From 145 patients who underwent elective PCI, PMI occurred in 40 patients (28%). The patients’ baseline clinical characteristics are detailed in Table 1. Table 2 summarizes the angiographic and procedural characteristics. The mean age of the patients was 61 ± 10 years; 83% were men. Patients with PMI were older, and had higher serum levels of N-terminal pro-brain natriuretic peptide (NT-proBNP) compared to non-PMI patients (Table 1). There was no difference in demography, medical history, medications, and baseline laboratory data between the two groups (Table 1). Figure 1 shows the post-procedural levels of high-sensitivity troponin T (hsTnT) in PMI and non-PMI patients. After PCI, hsTnT was higher in PMI patients than in non-PMI patients, while there was no significant difference in serum creatinine (Table 1). Of note, regarding to the procedural characteristics, patients with PMI had higher SYNTAX (Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery) scores, and more frequently received longer stent placements compared with non-PMI patients (Table 2).
Table 1. Patients’ characteristics.
| All patients (n=145) | PMI group (n=40) | Non-PMI group (n=105) | P-value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Demography and medical history | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 61 ± 10 | 64 ± 10 | 60 ± 9 | 0.008 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI | 24.6 ± 3.6 | 24.0 ± 4.2 | 24.9 ± 3.4 | 0.170 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male gender | 120 (83%) | 34 (85%) | 86 (82%) | 0.659 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | 71 (49%) | 18 (45%) | 53 (50%) | 0.555 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension | 103 (71%) | 30 (75%) | 73 (70%) | 0.516 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes | 52 (36%) | 17 (43%) | 35 (33%) | 0.304 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Renal dysfunction | 17 (12%) | 6 (15%) | 11 (11%) | 0.640 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Previous PCI | 51 (35%) | 12 (30%) | 39 (37%) | 0.421 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chronic heart failure | 36 (25%) | 13 (33%) | 23 (22%) | 0.187 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cerebrovascular disease | 28 (19%) | 8 (20%) | 20 (19%) | 0.897 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Medications | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Statin | 142 (98%) | 39 (98%) | 103 (98%) | 1.00 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Aspirin | 140 (97%) | 38 (95%) | 102 (97%) | 0.902 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Clopidogrel | 98 (68%) | 27 (68%) | 71 (68%) | 0.989 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ticagrelor | 44 (30%) | 13 (33%) | 31 (30%) | 0.728 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Glycoprotein IIb/IIIa inhibitor use | 23 (16%) | 5 (13%) | 18 (17%) | 0.494 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Echocardiographic result | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LVEF (%) | 66 (61–71) | 66 (57–70) | 66 (62–71) | 0.438 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Laboratory tests at admission | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HbA1c (%) | 6.2 (5.8–6.8) | 6.2 (5.8–6.8) | 6.2 (5.8–6.8) | 0.712 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total cholesterol (mmol/L) | 4.3 ± 1.0 | 4.3 ± 0.8 | 4.2 ± 1.1 | 0.642 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDL cholesterol (mmol/L) | 2.3 ± 0.9 | 2.4 ± 0.8 | 2.3 ± 1.0 | 0.626 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WBC (×109/L) | 6.9 ± 1.8 | 6.7 ± 1.6 | 7.0 ± 1.9 | 0.401 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NLR | 2.4 ± 1.1 | 2.3 ± 1.1 | 2.4 ± 1.2 | 0.645 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NT-proBNP (pg/ml) | 121 (50–423) | 245 (69–673) | 88 (50–271) | 0.011 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creatinine (umol/L) | 87 ± 21 | 88 ± 23 | 86 ± 20 | 0.691 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| hsTnT (ng/L) | 9 (6–12) | 10 (6–12) | 9 (6–11) | 0.285 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ang-2 (Log) at the baseline | 3.18 ± 0.28 | 3.20 ± 0.35 | 3.17 ± 0.25 | 0.554 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Laboratory tests post-PCI | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Creatinine (umol/L) | 92 ± 24 | 92 ± 25 | 91 ± 24 | 0.855 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| hsTnT (ng/L) | 35 (19–78) | 135 (89–269) | 24 (18–37) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ang-2 (Log) on postoperative day 1 | 3.14 ± 0.26 | 3.17 ± 0.26 | 3.12 ± 0.26 | 0.296 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ang-2 (Log) on postoperative day 3 | 3.11 ± 0.28 | 3.19 ± 0.26 | 3.09 ± 0.28 | 0.051 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Data are presented as mean ± SD, or median (IQR, interquartile range), or number (percentage). Abbreviations: Ang-2=angiopoietin-2; BMI=body mass index; BP=blood pressure; HbA1c=hemoglobin A1c; hsTnT=high sensitive troponin-T; LDL=low-density lipoprotein; LVEF=left ventricular ejection fraction; NLR=neutrophil-to-lymphocyte ratio; NT-proBNP=N-terminal pro-brain natriuretic peptide; PCI=percutaneous coronary intervention; WBC=white blood cell. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 2. Procedural Characteristics.
| All patients (n=145) | PMI group (n=40) | Non-PMI group (n=105) | P-value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SYNTAX score | 16 ± 10 | 19 ± 10 | 15 ± 9 | 0.019 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Description of the target lesions | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Multiple vessels (two or more) | 24 (17%) | 7 (18%) | 17 (16%) | 0.850 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LM | 8 (6%) | 2 (5%) | 6 (6%) | 1.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LAD | 74 (51%) | 22 (55%) | 52 (50%) | 0.555 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LCx | 36 (25%) | 14 (35%) | 22 (21%) | 0.080 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| RCA | 55 (38%) | 11 (28%) | 44 (42%) | 0.110 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CTO | 14 (10%) | 5 (13%) | 9 (9%) | 0.688 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PTCA without stenting | 8 (6%) | 1 (3%) | 7 (7%) | 0.565 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Number of stents | 1.4 ± 0.6 | 1.6 ± 0.6 | 1.3 ± 0.6 | 0.004 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total stent length, mm | 35 ± 21 | 44 ± 21 | 32 ± 20 | 0.003 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pre-dilation | 139 (96%) | 39 (98%) | 100 (95%) | 0.885 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Post-dilation | 127 (88%) | 37 (93%) | 90 (86%) | 0.409 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Max balloon pressure (atm) | 17.5 ± 3.8 | 17.7 ± 3.7 | 17.4 ± 3.9 | 0.625 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Post-PCI TIMI-3 flow | 144 (99%) | 39 (98%) | 105 (100%) | 0.276 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: CTO=chronic total occlusion; LAD=left anterior descending coronary artery; LM=left main coronary artery; LCx=left circumflex coronary artery; PTCA=percutaneous transluminal coronary angioplasty; RCA=right coronary artery; SYNTAX=Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
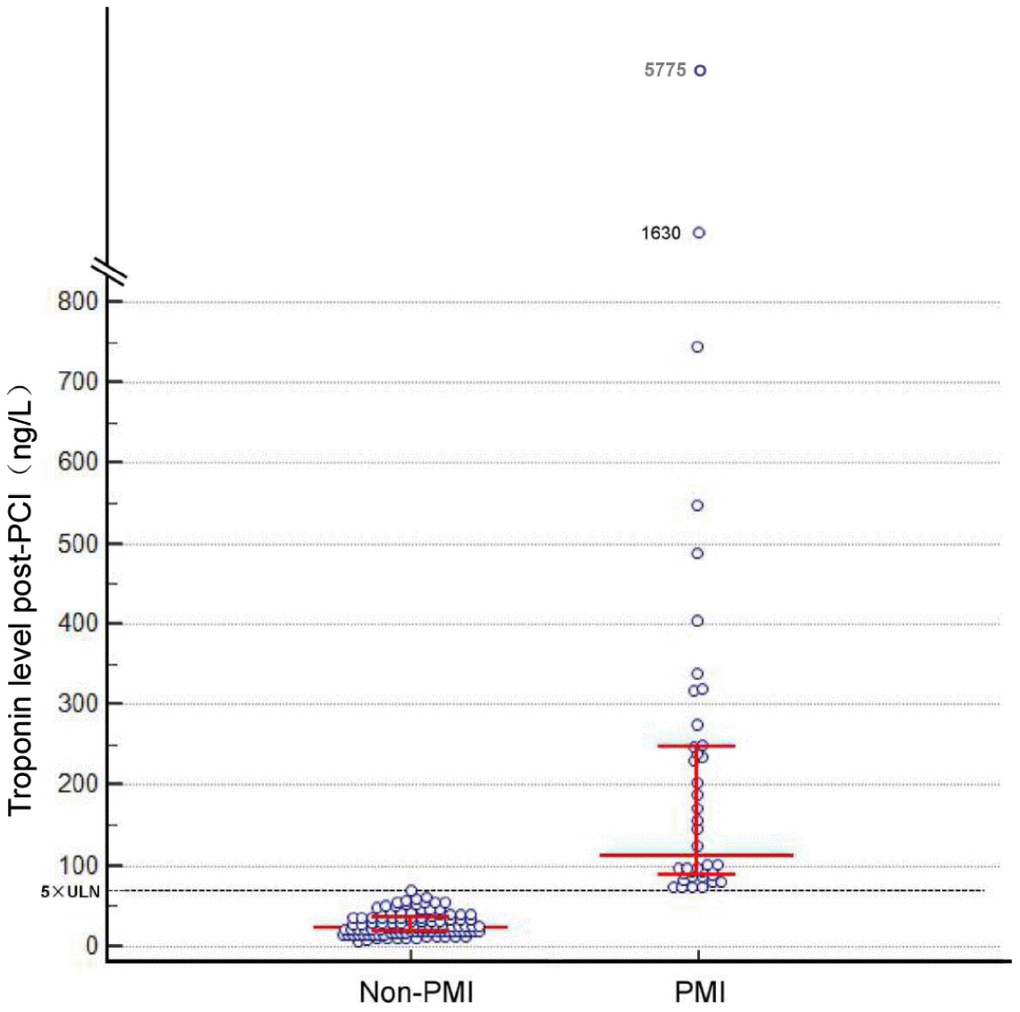
Figure 1. Post-percutaneous coronary intervention troponin levels. Bars indicate median levels with interquartile range. The dotted line indicates the limit of 70 ng/L. ULN, upper limit of the normal.
Association between circulating Ang-2 levels and PMI
At baseline, there was no difference in Ang-2 levels between PMI and non-PMI patients (Log: 3.20 ± 0.35 vs. 3.17 ± 0.25, P=0.554; Figure 2A). However, a significant interaction effect between PMI occurrence and time on Ang-2 levels was observed (interaction P=0.036; Figure 2B). Serum Ang-2 levels in non-PMI patients gradually decreased after PCI (Log: 3.17 ± 0.25 vs. 3.12 ± 0.26 vs. 3.09 ± 0.28, P<0.001), whereas there was no change in Ang-2 levels after PCI in PMI patients (Log: 3.20 ± 0.35 vs. 3.17 ± 0.26 vs. 3.19 ± 0.26, P=0.442; Figure 2B).
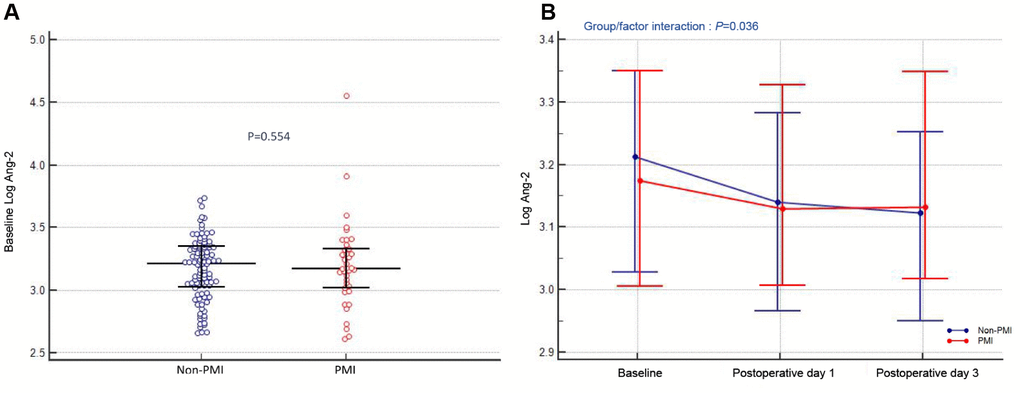
Figure 2. Association between circulating Ang-2 levels and PMI. (A) Preoperative angiopoietin-2 levels (Log). (B) Angiopoietin-2 levels (Log) at different time-points (at admission, postoperative day 1 and day 3) in patients stratified by PMI. The dots indicate median levels, and bars represent interquartile range.
Predictive factors analysis of PMI
As illustrated in Figure 3, receiver operating characteristic (ROC) curve analysis showed that NT-proBNP predicted PMI with 53% sensitivity and 74% specificity, using an optimal cutoff of 213 pg/ml (area under curve [AUC], 0.64; 95% confidence interval [CI], 0.55-0.72; P=0.011). In multivariable logistic regression analysis, age (per 10 years, odds ratio [OR]: 2.08; 95% CI: 1.25–3.48; P=0.005), total stent length (per 10 mm, OR: 1.36; 95% CI: 1.09–1.70; P=0.007), and NT-proBNP >213 pg/ml (OR: 2.63; 95% CI: 1.08–6.38; P=0.032) were independent predictors of PMI (Table 3). Combination of these 3 characteristics showed a high predictive value (AUC, 0.73; 95% CI, 0.65-0.80; P<0.001; Figure 3).
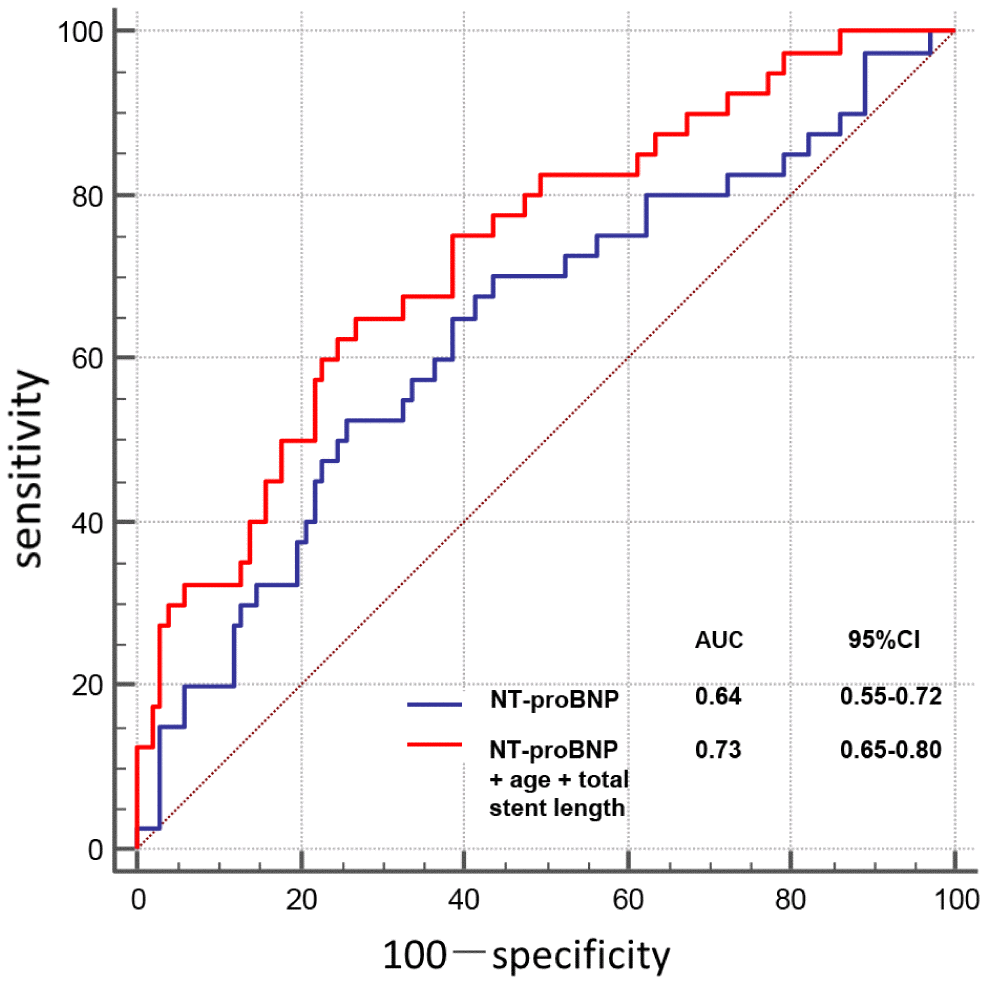
Figure 3. Receiver operating characteristic curve for relevant variables predicting PMI.
Table 3. Binary logistic regression model in univariate and multivariate analysis for predicting PMI.
| Univariable | Multivariable | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OR (95% CI) | P value | OR (95% CI) | P value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age, per 10 years | 1.75 (1.15–2.68) | 0.010 | 2.08 (1.25–3.48) | 0.005 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SYNTAX score, per 10 points | 1.55 (1.07–2.26) | 0.022 | 1.12 (0.69–1.83) | 0.636 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total stent length, per 10 mm | 1.31 (1.10–1.58) | 0.003 | 1.36 (1.09–1.70) | 0.007 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NT-proBNP (>213 pg/ml) | 3.19 (1.49–6.85) | 0.003 | 2.63 (1.08–6.38) | 0.032 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Notes: The multivariate model included age, gender, hypertension, diabetes, renal dysfunction, SYNTAX score, multivessel lesion, left main artery lesion, left anterior descending artery lesion, total stent length, and NT-proBNP. CI=confidence interval; NT-proBNP=N-terminal pro-brain natriuretic peptide; OR=odds ratio; PMI=periprocedural myocardial injury; SYNTAX=Synergy Between Percutaneous Coronary Intervention With Taxus and Cardiac Surgery. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Discussion
This study is the first to describe the relationship between serum Ang-2 levels and PMI occurrence in patients undergoing elective PCI. The major findings are: (1) There is no association between preoperative Ang-2 levels and PMI occurrence; (2) after PCI, the change of Ang-2 levels is closely related to PMI; and (3) NT-proBNP is an independent predictor of PMI.
In previous studies, PMI has been reported to be patient-, lesion-, and procedure-related [18]. An increase in cardiac biomarkers post procedure is very common for patients undergoing PCI [25], and the incidence of PMI can vary remarkably from 5 to 33%, depending on the cardiac biomarker, marker assay, and the criteria of cut-off value [26, 27]. Notably, probably because of the use of high-sensitivity troponin assays, and the lack of effective preventive strategies [28, 29], we observed a high PMI rate of 28%. Since our study did not observe fatal or severe complications, such as acute stent thrombosis, bleeding, perforation, or stroke during the procedure, we assume that a large proportion of PMI may be attributed to the distal embolization of plaque debris.
Plaque composition, verified by multiple intravascular imaging modalities, such as optical coherence tomography [19], intravascular ultrasound [20], or near infrared spectroscopy [21], is a predominant contributor to cardiac enzyme elevation post-PCI. Several inflammatory biomarkers have been shown to predict PMI occurrence, though the underlying mechanisms are not understood [30, 31]. We assume that the predictive value of these biomarkers for PMI may not be solely based on a direct influence of the systemic inflammatory state but reflect a parallel activation of related factors and the plaque status. Our study indicates that the pre-procedural serum Ang-2 levels do not affect the occurrence of PMI. One possible explanation is that even though Ang-2 plays a pivotal role in the inflammatory response, the circulating Ang-2 levels may not be a good biomarker reflecting the plaque status. Ang-2 expression is low in quiescent endothelium, but is upregulated following endothelial activation by stress, hypoxia, and pro-inflammatory factors [8–10]. Previous studies have demonstrated that Ang-2 is abundantly expressed locally in advanced lesions [15, 16]. However, it remains unclear whether the circulating Ang-2 levels are related to unstable plaque features, such as plaque volume, lipid core, or fibrous cap thickness. Lee et al. reported no difference in plasma Ang-2 levels between patients with unstable and stable angina [17]. Charytan et al. recently found that, in non-AMI patients, there was no significant association between serum Ang-2 levels and the total burden of coronary plaque [32]. Similar results were also described by Mitsuma et al., who found that serum Ang-2 levels were not significantly different between mild and severe CAD groups (classified by maximal percent stenosis) [33]. Given the fact that our study subjects were in a non-acute condition and scheduled for elective procedure, the relatively low circulating Ang-2 levels, which were insensitive for the discrimination of vulnerable coronary plaque, might have contributed to the negative results in this study. Since the circulating Ang-2 levels are influenced by many factors, including cardiac function [34], renal function [35], and various inflammatory conditions [11], the predictive efficiency of Ang-2 for PMI might be compromised. In addition, in this study, we used high-sensitivity troponin assays that allowed detection of a minuscule damage to myocardial tissue. Given the use of 5-fold hsTnT criteria in this study, the different cut-off values and other cardiac biomarkers, such as creatine kinase-MB, should be investigated in the future.
Importantly, our study indicates that the level of NT-proBNP, a marker reflecting pressure and volume load on the heart, is an independent predictor of PMI. This finding is consistent with a previous study which showed that the NT-pro-BNP level is a useful biomarker for predicting PMI in diabetic patients without cardiac dysfunction [36]. Although NT-proBNP is not a direct indicator of necrosis, NT-proBNP characterizes the functional state of the myocardium. Previous studies have shown that the high levels of NT-proBNP are associated with myocardial ischemia [37], and the magnitude of NT-proBNP increase after PCI correlates with the extent of myocardial injury [38]. However, we failed to provide an explanation regarding the association of preoperative NT-proBNP levels with PMI occurrence. Given the multiple clinical application values of NT-proBNP in different scenarios beyond heart failure [34, 39, 40], further large studies need to be conducted to explore and verify its precise cut-off point in predicting PMI.
Alleviation of myocardial ischemia and the effects of drugs, such as statins [41], may explain the decrease in Ang-2 levels in day 1 samples compared to the admission samples. This trend even remains in patients with PMI, although it did not reach a statistical difference. Of note, the fall in Ang-2 levels was followed by a significant increase in day 3 in PMI patients, whereas in non-PMI patients, the falling trend continued. Increased Ang-2 levels measured over time have been established as an important prognostic factor in various diseases [22–24]. We have recently found that Ang-2 levels post-PCI, but not pre-PCI, can predict the occurrence of adverse cardiovascular events [42]. However, it remained unknown whether circulating Ang-2 levels after PCI are influenced by PMI. Therefore, this study focused on a well-characterized study population with negative troponin at admission to assess the association between Ang-2 and PMI. Intriguingly, the results are in line with our hypothesis that PMI can increase circulating Ang-2 levels. We assume that this phenomenon could be explained by microembolization. It has been previously suggested that the extent of troponin elevation after PCI correlates with a new, irreversible myocardial necrosis [43], which can induce local myocardial inflammation. Fam et al. [44] showed that Ang-2 is one of the most upregulated genes in biopsies of ischemic myocardium in patients with acute coronary syndrome. In a mouse infarct model, there was a significant gradual increase of Ang-2 expression by endothelial cells occurring at the infarct border zone, and this increase was maintained until day 7. Likewise, in a model of ischemia/reperfusion injury, Ang-2 expression began rising on day 2, but waned beginning on day 3 [45]. Therefore, the myocardial damage by microembolization due to plaque debris may increase the expression of Ang-2 in endothelial cells residing at the damaged border zone and influence its circulating levels. Thus, our data indicate that myocardial injury, reflected by increased troponin levels, is an important contributor to the elevation of circulating Ang-2 levels. This marked association can partly explain the recent evidence regarding the Ang-2 prognostic values.
An excessive and uncontrolled inflammation can exacerbate myocardial damage following PCI. Ang-2 can prime the inflammatory response, sensitize endothelial cells toward tumor necrosis factor-α, and increase expression of adhesion molecules, resulting in increased leukocyte recruitment [9]. Furthermore, Ang-2 can act as a paracrine chemoattractant promoting migration of innate immune cells by binding Tie2 receptors or in an integrin-dependent manner [12, 46, 47]. Whereas Ang-2 overexpression can facilitate the responsiveness of acute inflammation [12], Ang-2 downregulation can ameliorate post-ischemic cardiovascular remodeling after myocardial infarction, and attenuate myocardial death after ischemia/reperfusion injury [45]. Thus, the increased Ang-2 release from the heart after PCI can further aggravate the myocardial injury by amplifying cardiac hypoxia and inflammation.
Several limitations should be considered when interpreting our findings. First, this study was a single-center observational study with a relatively small sample size; thus, it is reasonable to assume that a certain amount of selection bias may exist in our study. Second, the post-procedural evidence of new myocardial ischemia, which is indispensable for the diagnosis of “type 4a myocardial infarction” [48], was not considered in this study. Third, since we collected the fasting blood samples in the morning on postoperative days 1 and 3, the extraction time was not strictly controlled. More data and large-scale studies are needed in the future.
Collectively, our findings demonstrate that the pre-procedural Ang-2 levels do not impact the PMI occurrence after elective PCI. However, the changes in Ang-2 levels after the procedure are closely related to PMI. Targeting the Ang-2 pathway may serve as a potential novel strategy for reducing the occurrence of PMI.
Materials and Methods
Study population
The study enrolled 145 consecutive patients who underwent elective PCI with a negative hsTnT level at the time of admission, between September 2018 and September 2019. Patients were referred for numerous reasons, including atypical chest pain, stable or unstable angina, previous diagnosis of CAD in a setting of scheduled operation, or other indications for diagnostic coronary angiography. The following exclusion criteria were applied: AMI, acute heart failure, elevated hsTnT levels at admission (>99th percentile upper reference limit [URL]), unavailable measurements of serial (preprocedural and postprocedural) hsTnT, tachyarrhythmia, valvular or congenital heart disease, concomitant inflammatory conditions, end-stage renal disease, malignancies, and recent surgery or trauma (within 3 months). Medications at admission and perioperative anticoagulant therapy were chosen based on the accepted guidelines and standard methods. Dual antiplatelet treatment, including aspirin, and a P2Y12 inhibitor (chosen by the interventional cardiologist) were initiated at admission, during PCI, or immediately after PCI. Unfractionated heparin was used during the operation, and the periprocedural use of glycoprotein IIb/IIIa inhibitors was left to the operator’s discretion. Drug-eluting stents were used for patients who needed stent placement. Angiographic characteristics were recorded by experienced angiographers. The study protocol was reviewed and approved by the Human Research Ethics Committee of the First Affiliated Hospital of Guangxi Medical University, China. All patients provided written informed consent.
Laboratory measurements
In our hospital, screening for PMI and contrast-induced nephropathy was routinely performed after the PCI. In brief, hsTnT levels were measured at admission, postoperative day 1, and when clinically indicated. Furthermore, repeated measurement of serum creatinine was conducted on postoperative day 3. Fasting blood samples for Ang-2 analysis were collected under standardized conditions at admission and on postoperative days 1 and 3. HsTnT was measured by electrochemiluminescence immunoassay (Roche Diagnostics, Risch-Rotkreuz, Switzerland) (99th percentile URL: 14 ng/L). Serum Ang-2 levels were measured using commercially available enzyme-linked immunosorbent assay kits (RayBiotech, Inc, Norcross, GA, USA). The limit of detection for Ang-2 was 10 pg/ml, with the intra-assay variability <10 % and the inter-assay variability <12 %.
Statistical analysis
Continuous variables are presented as mean ± SD or median (IQR, interquartile range), and were compared using Student t test or Mann-Whitney U test, as appropriate. Categorical variables are presented as percentage of patients; they were compared by chi-square or Fisher exact test. As the distribution of Ang-2 was skewed, a base-10 logarithmic transformation was applied. A 2-factor mixed ANOVA was used to test the interaction effect between PMI and time on Ang-2 levels after the procedure. One-way repeated-measures ANOVA test was used to analyze the Ang-2 levels over time in each group. ROC curve analysis was performed to evaluate the diagnostic value of relevant variables for PMI. Multivariable logistic regression model was constructed to assess the independent predictors of PMI, including the variables that were selected based on clinical judgement, or the known risk factors based on literature review [18], or P value < 0.05 in the univariate analysis. Ultimately, age, gender, hypertension, diabetes, renal dysfunction, SYNTAX score, multivessel lesion, left main artery lesion, left anterior descending artery lesion, total stent length, and NT-proBNP were included in the multivariate model. A 2-tailed P value < 0.05 was considered significant. Statistical analysis was performed using SPSS, version 22 and MedCalc, version 19.0.4.
Author Contributions
Chun Gui designed the research; Wen Jian, Jia-Hui Guan, Wen-Bo Zheng, Chang-Hua Mo, Yu-Tao Xu, Qi-Li Huang, Zhi-Jie Yang, and Guo-Liang Yang carried out the research; Wen Jian, Jia-Hui Guan, Chun-Mei Wei, and Can Wang analyzed the data; Wen Jian wrote the paper.
Conflicts of Interest
All authors have declared that there are no conflicts of interest related to the contents of this article.
Funding
This study was supported by the National Natural Science Foundation of China (No. 81460063), Guangxi Natural Science Foundation (No. 2014GXNSFDA118024), and the Health Technology Research and Development Project of Guangxi (No. S2019094).
References
- 1. Montalescot G, Sechtem U, Achenbach S, Andreotti F, Arden C, Budaj A, Bugiardini R, Crea F, Cuisset T, Di Mario C, Ferreira JR, Gersh BJ, Gitt AK, et al, and Document Reviewers. 2013 ESC guidelines on the management of stable coronary artery disease: the Task Force on the management of stable coronary artery disease of the European Society of Cardiology. Eur Heart J. 2013; 34:2949–3003. https://doi.org/10.1093/eurheartj/eht296 [PubMed]
- 2. Zeitouni M, Silvain J, Guedeney P, Kerneis M, Yan Y, Overtchouk P, Barthelemy O, Hauguel-Moreau M, Choussat R, Helft G, Le Feuvre C, Collet JP, Montalescot G, and ACTION Study Group. Periprocedural myocardial infarction and injury in elective coronary stenting. Eur Heart J. 2018; 39:1100–09. https://doi.org/10.1093/eurheartj/ehx799 [PubMed]
- 3. Thygesen K, Jaffe AS. The prognostic impact of periprocedural myocardial infarction and injury. Eur Heart J. 2018; 39:1110–12. https://doi.org/10.1093/eurheartj/ehy089 [PubMed]
- 4. Koskinas KC, Ndrepepa G, Räber L, Karagiannis A, Kufner S, Zanchin T, Hieber J, Hunziker L, Mayer K, Byrne RA, Heg D, Windecker S, Kastrati A. Prognostic Impact of Periprocedural Myocardial Infarction in Patients Undergoing Elective Percutaneous Coronary Interventions. Circ Cardiovasc Interv. 2018; 11:e006752. https://doi.org/10.1161/CIRCINTERVENTIONS.118.006752 [PubMed]
- 5. Cuculi F, Lim CC, Banning AP. Periprocedural myocardial injury during elective percutaneous coronary intervention: is it important and how can it be prevented? Heart. 2010; 96:736–40. https://doi.org/10.1136/hrt.2009.186189 [PubMed]
- 6. Fagiani E, Christofori G. Angiopoietins in angiogenesis. Cancer Lett. 2013; 328:18–26. https://doi.org/10.1016/j.canlet.2012.08.018 [PubMed]
- 7. Saharinen P, Eklund L, Miettinen J, Wirkkala R, Anisimov A, Winderlich M, Nottebaum A, Vestweber D, Deutsch U, Koh GY, Olsen BR, Alitalo K. Angiopoietins assemble distinct Tie2 signalling complexes in endothelial cell-cell and cell-matrix contacts. Nat Cell Biol. 2008; 10:527–37. https://doi.org/10.1038/ncb1715 [PubMed]
- 8. Fiedler U, Scharpfenecker M, Koidl S, Hegen A, Grunow V, Schmidt JM, Kriz W, Thurston G, Augustin HG. The Tie-2 ligand angiopoietin-2 is stored in and rapidly released upon stimulation from endothelial cell Weibel-Palade bodies. Blood. 2004; 103:4150–56. https://doi.org/10.1182/blood-2003-10-3685 [PubMed]
- 9. Fiedler U, Reiss Y, Scharpfenecker M, Grunow V, Koidl S, Thurston G, Gale NW, Witzenrath M, Rosseau S, Suttorp N, Sobke A, Herrmann M, Preissner KT, et al. Angiopoietin-2 sensitizes endothelial cells to TNF-alpha and has a crucial role in the induction of inflammation. Nat Med. 2006; 12:235–39. https://doi.org/10.1038/nm1351 [PubMed]
- 10. Fiedler U, Augustin HG. Angiopoietins: a link between angiogenesis and inflammation. Trends Immunol. 2006; 27:552–58. https://doi.org/10.1016/j.it.2006.10.004 [PubMed]
- 11. Scholz A, Plate KH, Reiss Y. Angiopoietin-2: a multifaceted cytokine that functions in both angiogenesis and inflammation. Ann N Y Acad Sci. 2015; 1347:45–51. https://doi.org/10.1111/nyas.12726 [PubMed]
- 12. Scholz A, Lang V, Henschler R, Czabanka M, Vajkoczy P, Chavakis E, Drynski J, Harter PN, Mittelbronn M, Dumont DJ, Plate KH, Reiss Y. Angiopoietin-2 promotes myeloid cell infiltration in a β₂-integrin-dependent manner. Blood. 2011; 118:5050–59. https://doi.org/10.1182/blood-2011-03-343293 [PubMed]
- 13. Trollope AF, Golledge J. Angiopoietins, abdominal aortic aneurysm and atherosclerosis. Atherosclerosis. 2011; 214:237–43. https://doi.org/10.1016/j.atherosclerosis.2010.08.051 [PubMed]
- 14. Theelen TL, Lappalainen JP, Sluimer JC, Gurzeler E, Cleutjens JP, Gijbels MJ, Biessen EA, Daemen MJ, Alitalo K, Ylä-Herttuala S. Angiopoietin-2 blocking antibodies reduce early atherosclerotic plaque development in mice. Atherosclerosis. 2015; 241:297–304. https://doi.org/10.1016/j.atherosclerosis.2015.05.018 [PubMed]
- 15. Calvi C, Dentelli P, Pagano M, Rosso A, Pegoraro M, Giunti S, Garbarino G, Camussi G, Pegoraro L, Brizzi MF. Angiopoietin 2 induces cell cycle arrest in endothelial cells: a possible mechanism involved in advanced plaque neovascularization. Arterioscler Thromb Vasc Biol. 2004; 24:511–18. https://doi.org/10.1161/01.ATV.0000116864.86607.35 [PubMed]
- 16. Post S, Peeters W, Busser E, Lamers D, Sluijter JP, Goumans MJ, de Weger RA, Moll FL, Doevendans PA, Pasterkamp G, Vink A. Balance between angiopoietin-1 and angiopoietin-2 is in favor of angiopoietin-2 in atherosclerotic plaques with high microvessel density. J Vasc Res. 2008; 45:244–50. https://doi.org/10.1159/000112939 [PubMed]
- 17. Lee KW, Lip GY, Blann AD. Plasma angiopoietin-1, angiopoietin-2, angiopoietin receptor tie-2, and vascular endothelial growth factor levels in acute coronary syndromes. Circulation. 2004; 110:2355–60. https://doi.org/10.1161/01.CIR.0000138112.90641.7F [PubMed]
- 18. Park DW, Kim YH, Yun SC, Ahn JM, Lee JY, Kim WJ, Kang SJ, Lee SW, Lee CW, Park SW, Park SJ. Frequency, causes, predictors, and clinical significance of peri-procedural myocardial infarction following percutaneous coronary intervention. Eur Heart J. 2013; 34:1662–69. https://doi.org/10.1093/eurheartj/eht048 [PubMed]
- 19. Kini AS, Motoyama S, Vengrenyuk Y, Feig JE, Pena J, Baber U, Bhat AM, Moreno P, Kovacic JC, Narula J, Sharma SK. Multimodality Intravascular Imaging to Predict Periprocedural Myocardial Infarction During Percutaneous Coronary Intervention. JACC Cardiovasc Interv. 2015; 8:937–45. https://doi.org/10.1016/j.jcin.2015.03.016 [PubMed]
- 20. Jang JS, Jin HY, Seo JS, Yang TH, Kim DK, Park YA, Cho KI, Park YH, Kim DS. Meta-analysis of plaque composition by intravascular ultrasound and its relation to distal embolization after percutaneous coronary intervention. Am J Cardiol. 2013; 111:968–72. https://doi.org/10.1016/j.amjcard.2012.12.016 [PubMed]
- 21. Goldstein JA, Maini B, Dixon SR, Brilakis ES, Grines CL, Rizik DG, Powers ER, Steinberg DH, Shunk KA, Weisz G, Moreno PR, Kini A, Sharma SK, et al. Detection of lipid-core plaques by intracoronary near-infrared spectroscopy identifies high risk of periprocedural myocardial infarction. Circ Cardiovasc Interv. 2011; 4:429–37. https://doi.org/10.1161/CIRCINTERVENTIONS.111.963264 [PubMed]
- 22. Ricciuto DR, dos Santos CC, Hawkes M, Toltl LJ, Conroy AL, Rajwans N, Lafferty EI, Cook DJ, Fox-Robichaud A, Kahnamoui K, Kain KC, Liaw PC, Liles WC. Angiopoietin-1 and angiopoietin-2 as clinically informative prognostic biomarkers of morbidity and mortality in severe sepsis. Crit Care Med. 2011; 39:702–10. https://doi.org/10.1097/CCM.0b013e318206d285 [PubMed]
- 23. Link A, Pöss J, Rbah R, Barth C, Feth L, Selejan S, Böhm M. Circulating angiopoietins and cardiovascular mortality in cardiogenic shock. Eur Heart J. 2013; 34:1651–62. https://doi.org/10.1093/eurheartj/ehs487 [PubMed]
- 24. Pöss J, Ukena C, Kindermann I, Ehrlich P, Fuernau G, Ewen S, Mahfoud F, Kriechbaum S, Böhm M, Link A. Angiopoietin-2 and outcome in patients with acute decompensated heart failure. Clin Res Cardiol. 2015; 104:380–87. https://doi.org/10.1007/s00392-014-0787-y [PubMed]
- 25. Herrmann J. Peri-procedural myocardial injury: 2005 update. Eur Heart J. 2005; 26:2493–519. https://doi.org/10.1093/eurheartj/ehi455 [PubMed]
- 26. Babu GG, Walker JM, Yellon DM, Hausenloy DJ. Peri-procedural myocardial injury during percutaneous coronary intervention: an important target for cardioprotection. Eur Heart J. 2011; 32:23–31. https://doi.org/10.1093/eurheartj/ehq393 [PubMed]
- 27. Prasad A, Herrmann J. Myocardial infarction due to percutaneous coronary intervention. N Engl J Med. 2011; 364:453–64. https://doi.org/10.1056/NEJMra0912134 [PubMed]
- 28. Kawakita N, Ejiri K, Miyoshi T, Kohno K, Nakahama M, Doi M, Munemasa M, Murakami M, Nakamura K, Ito H, and RINC investigators. Protective effect of nicorandil on myocardial injury following percutaneous coronary intervention in older patients with stable coronary artery disease: secondary analysis of a randomized, controlled trial (RINC). PLoS One. 2018; 13:e0194623. https://doi.org/10.1371/journal.pone.0194623 [PubMed]
- 29. Schüpke S, Hein-Rothweiler R, Mayer K, Janisch M, Sibbing D, Ndrepepa G, Hilz R, Laugwitz KL, Bernlochner I, Gschwendtner S, Kupka D, Gori T, Zeiher AM, et al, and ISAR-PLASTER-Trial Investigators. Revacept, a Novel Inhibitor of Platelet Adhesion, in Patients Undergoing Elective PCI-Design and Rationale of the Randomized ISAR-PLASTER Trial. Thromb Haemost. 2019; 119:1539–45. https://doi.org/10.1055/s-0039-1692423 [PubMed]
- 30. Zeng RX, Li JJ, Liao PD, Zhang MZ. Relationship of non-cardiac biomarkers with periprocedural myocardial injury in patients undergoing percutaneous coronary intervention. Int J Cardiol. 2016; 221:726–33. https://doi.org/10.1016/j.ijcard.2016.07.131 [PubMed]
- 31. Jin P, Zhou Q, Song S, Xu J, Zhang M, Zhu M, Kang M, Shi X, Shi J, Lu D, Li J. Elevated preoperative HMGB1 as predictor of myocardial injury post-percutaneous coronary intervention. Medicine (Baltimore). 2016; 95:e5149. https://doi.org/10.1097/MD.0000000000005149 [PubMed]
- 32. Charytan DM, Cinelli A, Zeisberg EM. Association of circulating angiogenesis inhibitors and asymmetric dimethyl arginine with coronary plaque burden. Fibrogenesis Tissue Repair. 2015; 8:13. https://doi.org/10.1186/s13069-015-0029-6 [PubMed]
- 33. Mitsuma W, Kodama M, Hirono S, Ito M, Ramadan MM, Tanaka K, Hoyano M, Saigawa T, Kashimura T, Fuse K, Okura Y, Aizawa Y. Angiopoietin-1, angiopoietin-2 and tie-2 in the coronary circulation of patients with and without coronary collateral vessels. Circ J. 2007; 71:343–47. https://doi.org/10.1253/circj.71.343 [PubMed]
- 34. Jian W, Mo CH, Yang GL, Li L, Gui C. Angiopoietin-2 provides no incremental predictive value for the presence of obstructive coronary artery disease over N-terminal pro-brain natriuretic peptide. J Clin Lab Anal. 2019; 33:e22972. https://doi.org/10.1002/jcla.22972 [PubMed]
- 35. David S, Kümpers P, Lukasz A, Fliser D, Martens-Lobenhoffer J, Bode-Böger SM, Kliem V, Haller H, Kielstein JT. Circulating angiopoietin-2 levels increase with progress of chronic kidney disease. Nephrol Dial Transplant. 2010; 25:2571–76. https://doi.org/10.1093/ndt/gfq060 [PubMed]
- 36. Zeng RX, Li XL, Zhang MZ, Wang XW, Guo YL, Zhu CG, Ren Y, Li S, Zhang Y, Liu G, Xu RX, Dong Q, Li JJ. Preprocedural N-terminal pro-B-type natriuretic peptide as a useful marker for predicting periprocedural myocardial injury following percutaneous coronary intervention in diabetic patients without cardiac dysfunction. Scand J Clin Lab Invest. 2015; 75:568–77. https://doi.org/10.3109/00365513.2015.1060518 [PubMed]
- 37. Sabatine MS, Morrow DA, de Lemos JA, Omland T, Desai MY, Tanasijevic M, Hall C, McCabe CH, Braunwald E. Acute changes in circulating natriuretic peptide levels in relation to myocardial ischemia. J Am Coll Cardiol. 2004; 44:1988–95. https://doi.org/10.1016/j.jacc.2004.07.057 [PubMed]
- 38. Bonaca MP, Wiviott SD, Sabatine MS, Buros J, Murphy SA, Scirica BM, Rifai N, Antman EM, Morrow DA. Hemodynamic significance of periprocedural myocardial injury assessed with N-terminal pro-B-type natriuretic peptide after percutaneous coronary intervention in patients with stable and unstable coronary artery disease (from the JUMBO-TIMI 26 trial). Am J Cardiol. 2007; 99:344–48. https://doi.org/10.1016/j.amjcard.2006.08.035 [PubMed]
- 39. du Fay de Lavallaz J, Badertscher P, Nestelberger T, Zimmermann T, Miró Ò, Salgado E, Christ M, Geigy N, Cullen L, Than M, Martin-Sanchez FJ, Di Somma S, Peacock WF, et al. B-Type Natriuretic Peptides and Cardiac Troponins for Diagnosis and Risk-Stratification of Syncope. Circulation. 2019. [Epub ahead of print]. https://doi.org/10.1161/CIRCULATIONAHA.118.038358 [PubMed]
- 40. Zhang C, Jiang L, Xu L, Tian J, Liu J, Zhao X, Feng X, Wang D, Zhang Y, Sun K, Xu B, Zhao W, Hui R, et al. Implications of N-terminal pro-B-type natriuretic peptide in patients with three-vessel disease. Eur Heart J. 2019; 40:3397–405. https://doi.org/10.1093/eurheartj/ehz394 [PubMed]
- 41. Hilbert T, Poth J, Frede S, Klaschik S, Hoeft A, Baumgarten G, Knuefermann P. Anti-atherogenic effects of statins: impact on angiopoietin-2 release from endothelial cells. Biochem Pharmacol. 2013; 86:1452–60. https://doi.org/10.1016/j.bcp.2013.09.004 [PubMed]
- 42. Jian W, Li L, Wei XM, Wu CQ, Gui C. Prognostic value of angiopoietin-2 for patients with coronary heart disease after elective PCI. Medicine (Baltimore). 2019; 98:e14216. https://doi.org/10.1097/MD.0000000000014216 [PubMed]
- 43. Selvanayagam JB, Porto I, Channon K, Petersen SE, Francis JM, Neubauer S, Banning AP. Troponin elevation after percutaneous coronary intervention directly represents the extent of irreversible myocardial injury: insights from cardiovascular magnetic resonance imaging. Circulation. 2005; 111:1027–32. https://doi.org/10.1161/01.CIR.0000156328.28485.AD [PubMed]
- 44. Fam NP, Arab S, Billia F, Han R, Proteau G, Latter D, Errett L, Bonneau D, Dunne R, Liu PP, Stewart DJ. Increased myocardial expression of angiopoietin-2 in patients undergoing urgent surgical revascularization for acute coronary syndromes. Can J Cardiol. 2010; 26:365–70. https://doi.org/10.1016/S0828-282X(10)70417-7 [PubMed]
- 45. Lee SJ, Lee CK, Kang S, Park I, Kim YH, Kim SK, Hong SP, Bae H, He Y, Kubota Y, Koh GY. Angiopoietin-2 exacerbates cardiac hypoxia and inflammation after myocardial infarction. J Clin Invest. 2018; 128:5018–33. https://doi.org/10.1172/JCI99659 [PubMed]
- 46. Venneri MA, De Palma M, Ponzoni M, Pucci F, Scielzo C, Zonari E, Mazzieri R, Doglioni C, Naldini L. Identification of proangiogenic TIE2-expressing monocytes (TEMs) in human peripheral blood and cancer. Blood. 2007; 109:5276–85. https://doi.org/10.1182/blood-2006-10-053504 [PubMed]
- 47. Lemieux C, Maliba R, Favier J, Théorêt JF, Merhi Y, Sirois MG. Angiopoietins can directly activate endothelial cells and neutrophils to promote proinflammatory responses. Blood. 2005; 105:1523–30. https://doi.org/10.1182/blood-2004-09-3531 [PubMed]
- 48. Thygesen K, Alpert JS, Jaffe AS, Chaitman BR, Bax JJ, Morrow DA, White HD, and Executive Group on behalf of the Joint European Society of Cardiology (ESC)/American College of Cardiology (ACC)/American Heart Association (AHA)/World Heart Federation (WHF) Task Force for the Universal Definition of Myocardial Infarction. Fourth Universal Definition of Myocardial Infarction (2018). J Am Coll Cardiol. 2018; 72:2231–64. https://doi.org/10.1016/j.jacc.2018.08.1038 [PubMed]
- 49. American Diabetes Association. 2. Classification and Diagnosis of Diabetes. Diabetes Care. 2017 (Suppl 1); 40:S11–24. https://doi.org/10.2337/dc17-S005 [PubMed]
- 50. Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF
3rd , Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J, and CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration). A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009; 150:604–12. https://doi.org/10.7326/0003-4819-150-9-200905050-00006 [PubMed] - 51. Sianos G, Morel MA, Kappetein AP, Morice MC, Colombo A, Dawkins K, van den Brand M, Van Dyck N, Russell ME, Mohr FW, Serruys PW. The SYNTAX Score: an angiographic tool grading the complexity of coronary artery disease. EuroIntervention. 2005; 1:219–27. [PubMed]



