Introduction
Epilepsy is a nervous system disorder resulting from the abnormal neuronal excitation [1–3]. Status epilepticus can cause persistent and acute damage to the nervous system and hippocampal neurons [4–6]. Hypoxia, ischemia, inflammation, and edema may occur within the hippocampus after status epilepticus [7–9]. These stimuli can induce release of excitatory amino acids from the cells, leading to Ca2+ and Na+ influx, which can in turn damage the neurons, leading to neuronal apoptosis, fiber sprouting, glial cell proliferation, loss of hippocampal neurons, and hippocampal sclerosis, among others [10–12]. Temporal lobe epilepsy (TLE) is one of the most prevalent form of the disease and often leads to hippocampal sclerosis [13, 14]. Despite some progress, the mechanism underlying TLE remains unknown.
Evidence suggests that increased expression or activity of matrix metalloproteinases (MMPs) after an insult can contribute to epileptogenesis [15–18]. It has therefore been suggested that MMP inhibition could be an effective therapy for epilepsy; however, available MMP inhibitors lack specificity and have serious side effects. The molecules upstream of MMPs may be more suitable for targeted treatment than MMPs themselves. However, the causes of the MMP upregulation in these cases is unclear.
Long noncoding RNAs (lncRNAs) are a subgroup of noncoding RNAs that are greater than 200 nucleotides in length [19–22]. Multiple studies have demonstrated that lncRNAs play key roles in the progression and pathogenesis of a variety of diseases, including cardiovascular disease, intervertebral disc degeneration, neurodegenerative diseases, and cancer [23–27]. LncRNAs were also found to be involved in numerous biological processes, including cell development, apoptosis, stem cell differentiation, migration, and inflammation [28–31]. For example, dysregulated expression of a newly identified lncRNA, ILF3-AS1, has been detected in several cancers, including colon cancer, osteosarcoma, cervical tumor, and melanoma [32–36]. In addition, two lncRNAs, H19 and RNA-UCA1, were recently shown to be involved in the development of epilepsy [37, 38]. The aim of the present study, therefore, was to investigate whether ILF3-AS1 expression is altered in TLE patients and to assess the relationships between ILF3-AS1 and MMPs. The results showed that both hippocampal and serum levels of ILF3-AS1 are higher in TLE than in the control group. Moreover, ectopic expression of ILF3-AS1 induced expression of inflammatory cytokines, MMP3 and MMP9 through targeting of miR-212.
Results
Quantitative analysis of the subjects
In this study, 23 patients met the inclusion criteria for the TLE group and 18 patients met those for the control group. These 41 patients were included in the final analysis. Age and gender were well matched between the two groups.
ILF3-AS1 is overexpressed in the hippocampus of TLE patients
We first analyzed ILF3-AS1 expression in the hippocampus of TLE and control patients. As indicated in Figure 1A, hippocampal ILF3-AS1 expression was higher in the TLE group than in the control group. Additionally, our results showed that serum ILF3-AS1 levels were also higher in TLE than control group (Figure 1B).
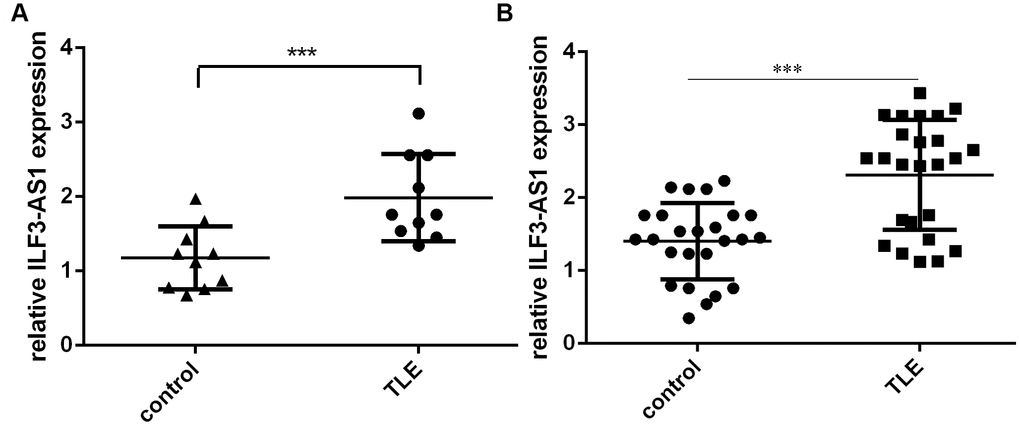
Figure 1. qRT-PCR analysis showing ILF3-AS1 is upregulated in the hippocampus and serum of TLE patients. Shown are levels of ILF3-AS1 expression in the hippocampus (A) and serum (B) of TLE patients and their matched controls. Data are shown as mean ± standard deviation (SD). ***p<0.001.
Relations between ILF3-AS1 and inflammatory cytokine expression
To study the effects of inflammatory cytokines on ILF3-AS1 expression in TLE, astrocytes were treated with IL-1β or TNF-α. Our data revealed that IL-1β and TNF-α were each able to induce ILF3-AS1 expression in the astrocytes (Figure 2A and 2B).
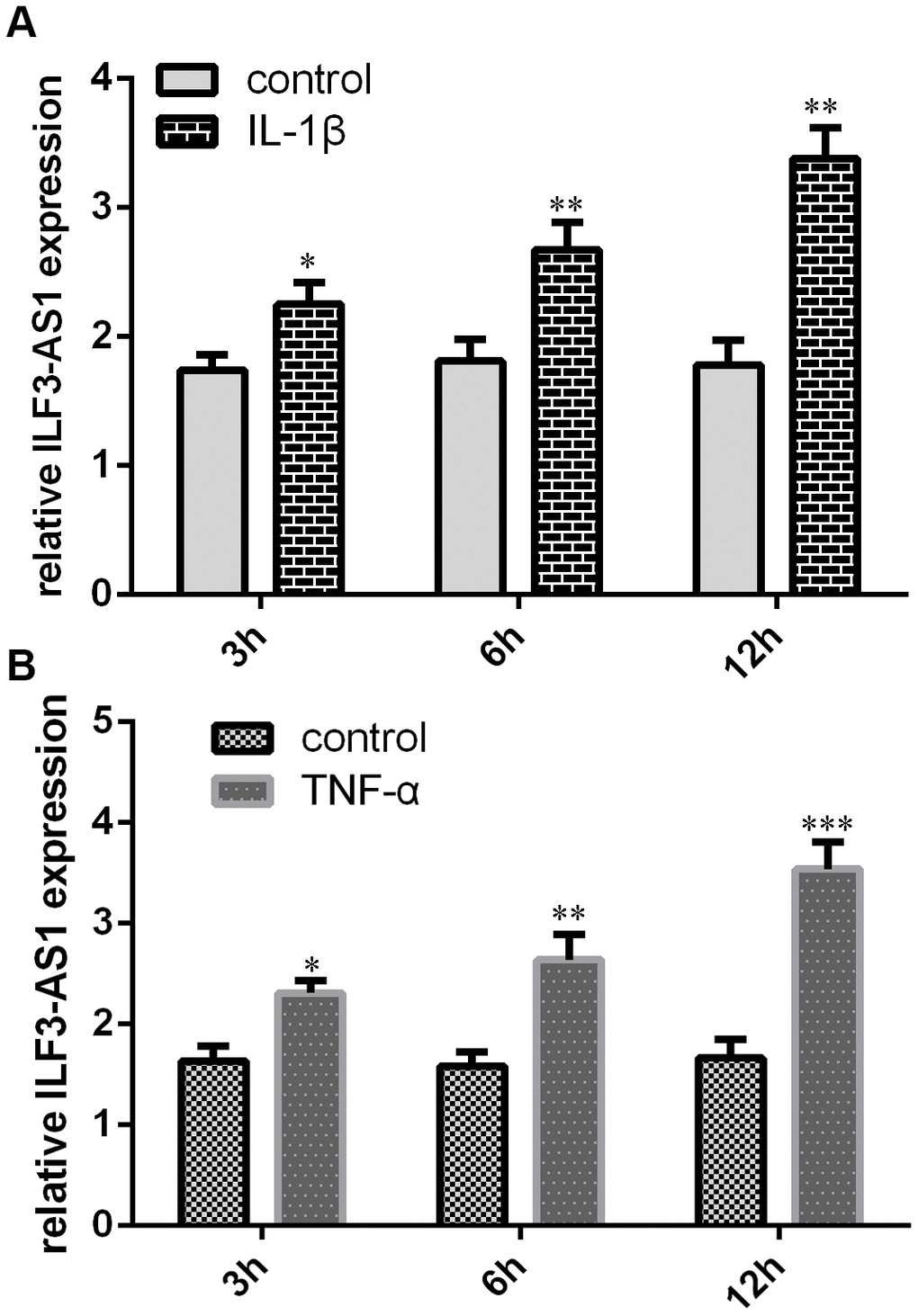
Figure 2. qRT-PCR analysis showing IL-1β and TNF-α induce ILF3-AS1 expression. Shown are levels of ILF3-AS1 expression induced by IL-1β (A) and TNF-α (B) in astrocytes. *p<0.05, **p<0.01, and ***p<0.001.
To further explore the relation between ILF3-AS1 and inflammatory cytokines in TLE, astrocytes were transfected with the pcDNA-ILF3-AS1 plasmid, which led to overexpression of ILF3-AS1 in the transfectants (Figure 3A). Notably, this ectopic ILF3-AS1 expression also promoted expression of IL-1β (Figure 3B), IL-6 (Figure 3C) and TNF-α in the astrocytes (Figure 3D).
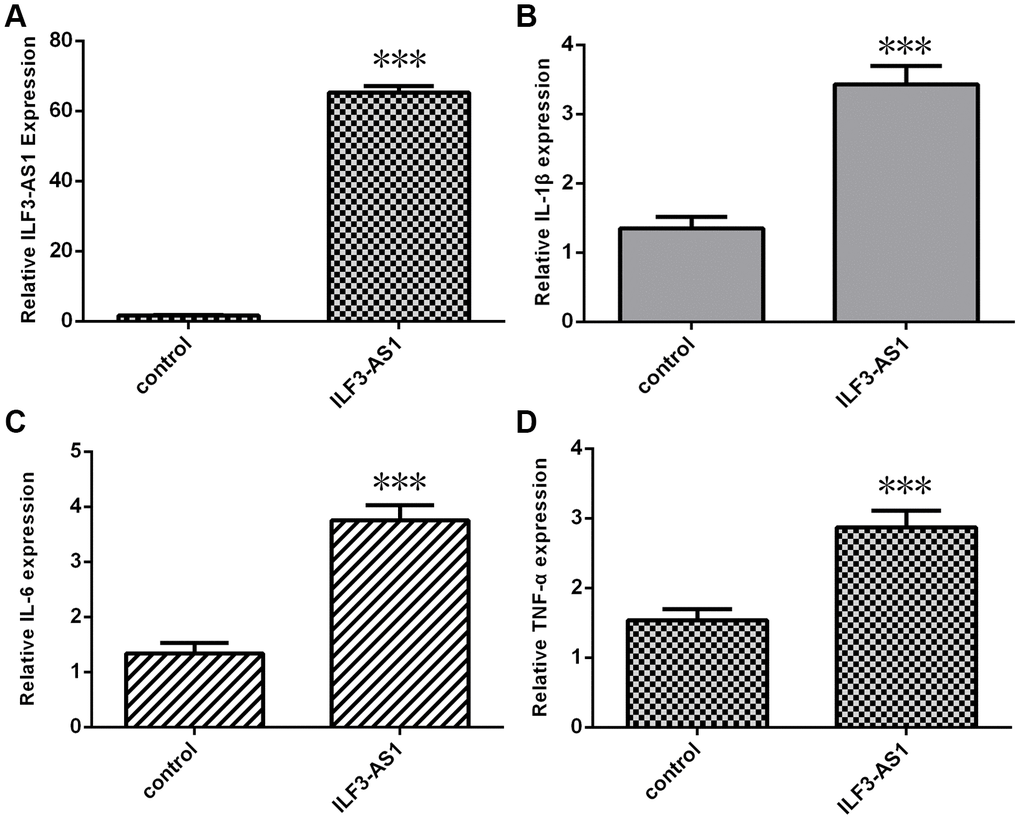
Figure 3. qRT-PCR analysis showing ILF3-AS1 induces inflammatory cytokine expression. (A) ILF3-AS1 expression in astrocytes transfected with pcDNA-ILF3-AS1 plasmid. Ectopic expression of ILF3-AS1 led to enhanced expression IL-1β (B), IL-6 (C), and TNF-α (D).***p<0.001.
Overexpression of ILF3-AS1 promotes MMP2, MMP3, MMP9, and MMP14 expression
Because MMP2, MMP3, MMP9, and MMP14 expression is reportedly enhanced in TLE after inflammatory cytokine stimulation [15–18], we studied whether the expression of these genes is regulated by ILF3-AS1. We found that levels of MMP2 (Figure 4A), MMP3 (Figure 4B), MMP9 (Figure 4C) and MMP14 (Figure 4D) expression were all upregulated in astrocytes ectopically overexpressing ILF3-AS1.
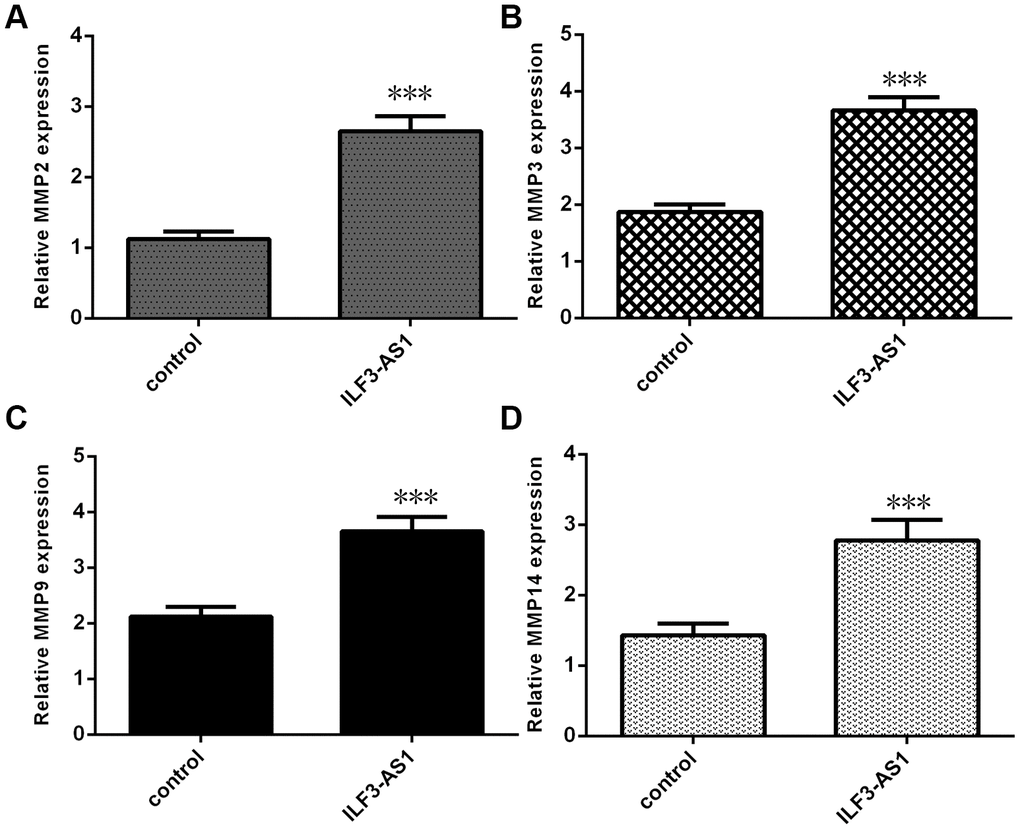
Figure 4. qRT-PCR analysis showing ILF3-AS1 promotes MMP2, MMP3, MMP9, and MMP14 expression. Ectopic expression of ILF3-AS1 in astrocytes induced expression of MMP2 (A), MMP3 (B), MMP9 (C), and MMP14 (D).***p<0.001.
ILF3-AS1 targets miR-212 in cultured astrocytes and the hippocampus of TLE patients
To investigate the molecular mechanism by which ILF3-AS1 may contribute to TLE progression, we analyzed its interactome using Starbase (http://starbase.sysu.edu.cn/index.php), which predicted that ILF3-AS1 likely binds miR-212 (Figure 5A). We also showed that miR-212 expression was increased in astrocytes treated with miR-212 mimic (Figure 5B). Using luciferase reporter assays, we observed that miR-212 overexpression decreased luciferase activity in astrocytes transfected with a wild-type ILF3-AS1 reporter plasmid (Figure 5C). Conversely, forced ILF3-AS1 expression suppressed miR-212 expression in the cells (Figure 5D). Consistent with those findings, both hippocampal and serum miR-212 levels were lower in TLE patients than the control group (Figure 6A and 6B).
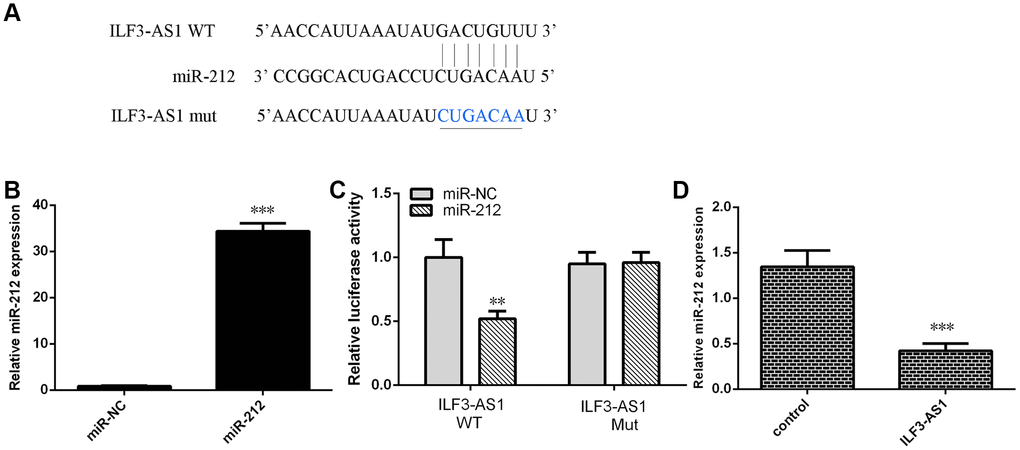
Figure 5. Ectopically expressed ILF3-AS1 targets miR-212. (A) Starbase (http://starbase.sysu.edu.cn/index.php) analysis predicting a binding site between ILF3-AS1 and miR-212. (B) qRT-PCR analysis of miR-212 expression. (C) ILF3-AS1-luciferase reporter assay showing miR-212 overexpression decreases ILF3-AS1 expression in astrocytes. (D) Forced ILF3-AS1 expression suppresses miR-212 expression in astrocytes. **p<0.01.
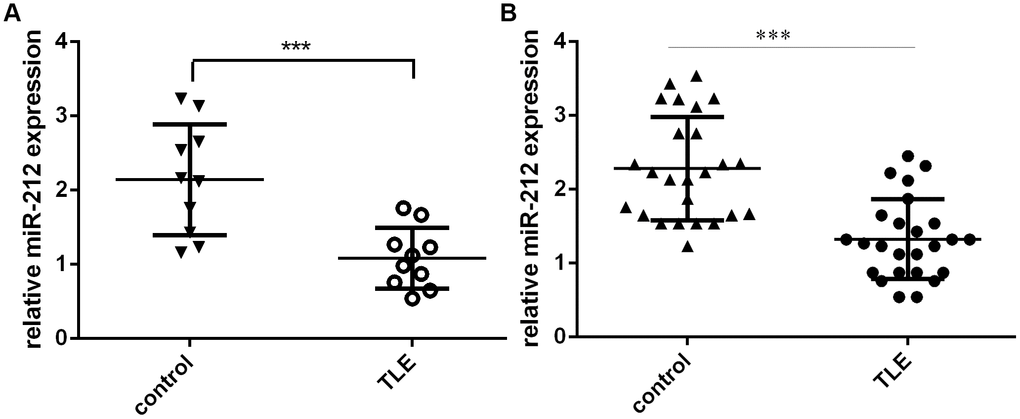
Figure 6. miR-212 expression is downregulated in TLE patients. Shown are levels of hippocampal (A) and serum (B) miR-212 expression in TLE patients and their matched controls. ***p<0.001.
ILF3-AS1 induces expression of inflammatory cytokines, MMP3 and MMP9 by targeting miR-212
To determine whether ILF3-AS1 promotes expression of inflammatory cytokines and MMPs by suppressing miR-212, a series of rescue experiments were performed. Forced miR-212 expression reduced expression of IL-1β (Figure 7A), IL-6 (Figure 7B), and TNF-α (Figure 7C) in astrocytes overexpressing ILF3-AS1. Likewise, elevated miR-212 expression decreased expression of MMP3 (Figure 7D) and MMP9 (Figure 7E) in the ILF3-AS1-overexpressing cells.
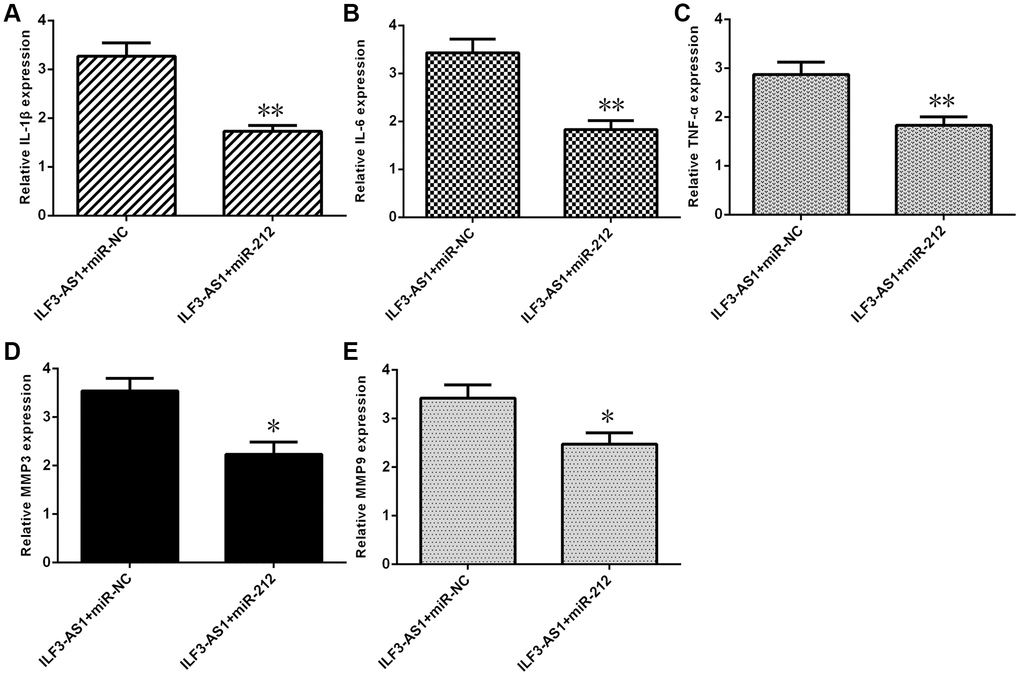
Figure 7. ILF3-AS1 induces expression of inflammatory cytokines, MMP3, and MMP9 by targeting miR-212. qRT-PCR analysis showing that forced miR-212 expression decreases expression of IL-1β (A), IL-6 (B), TNF-α (C), MMP3 (D), and MMP9 (E) in astrocytes ectopically expressing ILF3-AS1.*p<0.05, **p<0.01.
Discussion
TLE is the most common form of chronic drug-resistant epilepsy, and several studies have shown that patients with chronic TLE are more likely to develop cognitive impairment and premature aging [39, 40]. Although ILF3-AS1 has been found to play roles in colon cancer, osteosarcoma, cervical cancer, and melanoma [32–36], its potential function in TLE has remained unexplored. In this paper, we found that both hippocampal and serum ILF3-AS1 levels are higher in TLE patients than matched control individuals. In addition, we also found that overexpression of ILF3-AS1 promoted expression of MMP2, MMP3, MMP9, and MMP14. Our findings are consistent with those of Rempe et al. [16], who reported that MMP2 and MMP9 activities are elevated in a TLE rat model, and with those of Korotkov et al. [18], who observed increased MMP3 expression in the hippocampus of TLE-HS patients and in the rat TLE model. Based on those results it was suggested that MMP inhibitors may be an effective for anti-epilepsy therapy [15–18]. Moreover, our findings suggest ILF3-AS1 is situated upstream of MMPs, making it a potentially useful target for epilepsy treatment.
Evidence suggests inflammation is both a cause and a consequence of TLE [41, 42]. Numerous inflammatory mediators have been identified in brain samples from patients with refractory epilepsy [43]. Korotkov et al. [18] demonstrated that miR-155 inhibition attenuates MMP3 overexpression in astrocytes after IL-1β induction, and that miR-155 and MMP3 are overexpressed in the hippocampus of TLE model and TLE patients. In the present study, we showed that the inflammatory cytokines IL-1β and TNF-α each enhanced ILF3-AS1 expression in astrocytes, and that ectopic expression of ILF3-AS1 induced expression of IL-6 and TNF-α.
Previous studies demonstrated that lncRNAs act as ceRNAs against microRNAs, serving as sponges to inhibit miRNA expression. For example, PVT1 was shown to act via the Wnt pathway to suppress astrocyte activation and induce BDNF expression in hippocampal tissues from epilepsy cases [44]. In a TLE rat model, the lncRNA H19 was found to mediate apoptosis in hippocampal neurons by downregulating let-7b expression [45]. In addition, UCA1 reportedly reduced epilepsy and seizure-caused brain injury by modulating miR-495 expression [46]. In addition, Hu et al. [36] showed that ILF3-AS1 knockdown suppressed osteosarcoma cell growth, invasion, and migration and induced apoptosis through inhibition of miR-212 expression. This prompted us to perform a luciferase reporter analysis, which showed that miR-212 overexpression leads to a decrease in ILF3-AS1-luciferase activity in astrocytes. Moreover, forced ILF3-AS1 expression suppressed miR-212 expression in the astrocytes, and hippocampal and serum miR-212 expression was lower in TLE patients than in their control group. Taken together these results suggest ILF3-AS1 targets miR-212 to induce inflammatory cytokine and MMP3 and MMP9 expression.
In summary, our findings indicate that ILF3-AS1 levels are elevated in the hippocampus and serum of TLE patients, and that ILF3-AS1 induces expression of inflammatory cytokines and MMP3 and MMP9 by targeting miR-212. This is noteworthy and suggests a therapeutic strategy targeting the ILF3-AS1/miR-212/MMP3/9 axis may be an effective approach to treating TLE.
Materials and Methods
Tissues
Peripheral blood specimens were obtained from TLE patients and age- and sex-matched controls. All TLE cases met the diagnostic criteria for TLE from the International League Against Epilepsy (ILAE) [47]. Temporal cortex samples were collected from TLE cases undergoing lesionectomy; the control tissues were from patients with hypertension who needed an emergency operation to resolve an intracranial hematoma. Written consent was provided by all participants, and this research was approved by the Ethics Committee of the Sixth Affiliated Hospital of Sun Yat-Sen University.
Cell cultures and transfections
The astrocytes were collected from the Cell Center of the Chinese Academy of Medical Sciences (Beijing, China). These cells were cultured in DMEM supplemented with 20% fetal bovine serum (FBS) and L-glutamine (Sigma, St. Louis, MO). miR-NC, miR-212 mimic, pcDNA-control, and pcDNA-ILF3-AS1 were purchased from FitGene (Guangzhou, China). A Lipofectamine 2000 kit was used to transfect astrocytes with the mimics and plasmids.
RNA isolation and qRT-PCR
A Trizol kit was used to isolate RNA from cells and specimens. qRT-PCR assays were applied to determine the levels of ILF3-AS1, miR-212 and mRNA expression on an iQ5 qRT-PCR System (Bio-Rad, Hercules, CA) using SYBR Green (Takara Biotechnology, Dalian, China). U6 or GAPDH was utilized as the normal control. Expression level was determined using the 2−ΔΔCt method. The primers utilized in this research were as follows: for ILF3-AS1, 5′-TAAACCCCACTGTCTTCC-3′ (forward) and 5′-TTCCTTGCTCTTCTTGCTC-3′ (reverse); for miR-212, 5′-TGGTGTAACAGTCTCCAGTCA-3′ (forward) and 5′-CGATGACCTATGAATTGACAGACG-3′ (reverse); and for GAPDH, 5′- CACCGTAGCCT TCCGAGTA-3′ (forward) and 5′-GCCCTTGATG AGCTGTTGA-3′ (reverse).
Dual-luciferase reporter assays
Astrocytes were co-transfected with a reporter construct, pGL-3 control plasmid, miR-NC, or miR-212 mimic. Cells were harvested 24 h later and subjected to dual-luciferase analysis (Promega, Madison, WI). Firefly luciferase activity was normalized that of Renilla and expressed as the firefly/Renilla ratio.
Statistical analysis
Statistical analyses were conducted using SPSS 18.0 (SPSS Inc., Chicago, IL, USA). Results are presented as the mean ± standard deviation (SD). Student’s t-test was used to evaluate the statistical significance of differences between means. Values of P<0.05 were considered significant.
Acknowledgments
We thank Dr. Kang Kang from the Department of Neurosurgery, Guangdong 999 Brain Hospital, for their contribution to the research reported here. We also thank the patients for their support in this research.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
This study was funded by grants from the National Natural Science Foundation of China (Grant number 81501117 and 81301088), and Guangdong Basic and Applied Basic Research Foundation (Grant number 201709070039).
References
- 1. Puri I, Dash D, Padma MV, Tripathi M. Quality of Life and Its Determinants in Adult Drug Refractory Epilepsy Patients Who Were Not Candidates for Epilepsy Surgery: A Correlational Study. J Epilepsy Res. 2018; 8:81–86. https://doi.org/10.14581/jer.18013 [PubMed]
- 2. Kim SY, Kim WJ, Kim H, Choi SA, Lim BC, Chae JH, Kim KJ. Clinical Experience with Perampanel in Intractable Focal Epilepsy Over 12 Months of Follow-Up. J Epilepsy Res. 2018; 8:61–65. https://doi.org/10.14581/jer.18010 [PubMed]
- 3. Fu J, Liu Y, Yang K, Long H, Wang K, Qi S. Effect of accumbens nucleus shell lesioning on bitemporal lobe epilepsy in rat model. Folia Neuropathol. 2018; 56:346–53. https://doi.org/10.5114/fn.2018.80868 [PubMed]
- 4. Mehvari-Habibabadi J, Basiratnia R, Moein H, Zare M, Barakatain M, Aghakhani Y, Tabrizi N. Prognostic value of ictal onset patterns in postsurgical outcome of temporal lobe epilepsy. Iran J Neurol. 2017; 16:185–91. [PubMed]
- 5. McGinnity CJ, Smith AB, Yaakub SN, Weidenbach Gerbase S, Gammerman A, Tyson AL, Bell TK, Elmasri M, Barker GJ, Richardson MP, Pal DK. Decreased functional connectivity within a language subnetwork in benign epilepsy with centrotemporal spikes. Epilepsia Open. 2017; 2:214–25. https://doi.org/10.1002/epi4.12051 [PubMed]
- 6. Viguera AC, Fan Y, Thompson NR, Lapin B, Chaitoff A, Griffith SD, Miller DM, Jehi L, Katzan IL. Prevalence and Predictors of Depression Among Patients With Epilepsy, Stroke, and Multiple Sclerosis Using the Cleveland Clinic Knowledge Program Within the Neurological Institute. Psychosomatics. 2018; 59:369–78. https://doi.org/10.1016/j.psym.2017.12.003 [PubMed]
- 7. Rodriguez AS, Engel T, Palfi A, Farrar GJ, Henshall DC, Jimenez-Mateos EM. Tubby-like protein 1 (Tulp1) is a target of microRNA-134 and is down-regulated in experimental epilepsy. Int J Physiol Pathophysiol Pharmacol. 2017; 9:178–87. [PubMed]
- 8. Kim JH. Grey and White Matter Alterations in Juvenile Myoclonic Epilepsy: A Comprehensive Review. J Epilepsy Res. 2017; 7:77–88. https://doi.org/10.14581/jer.17013 [PubMed]
- 9. Zhang C, Yang H, Qin W, Liu C, Qi Z, Chen N, Li K. Characteristics of Resting-State Functional Connectivity in Intractable Unilateral Temporal Lobe Epilepsy Patients with Impaired Executive Control Function. Front Hum Neurosci. 2017; 11:609. https://doi.org/10.3389/fnhum.2017.00609 [PubMed]
- 10. Twele F, Schidlitzki A, Töllner K, Löscher W. The intrahippocampal kainate mouse model of mesial temporal lobe epilepsy: lack of electrographic seizure-like events in sham controls. Epilepsia Open. 2017; 2:180–87. https://doi.org/10.1002/epi4.12044 [PubMed]
- 11. Veersema TJ, Ferrier CH, van Eijsden P, Gosselaar PH, Aronica E, Visser F, Zwanenburg JM, de Kort GA, Hendrikse J, Luijten PR, Braun KP. Seven tesla MRI improves detection of focal cortical dysplasia in patients with refractory focal epilepsy. Epilepsia Open. 2017; 2:162–71. https://doi.org/10.1002/epi4.12041 [PubMed]
- 12. Kakooza-Mwesige A, Ndyomugyenyi D, Pariyo G, Peterson SS, Waiswa PM, Galiwango E, Chengo E, Odhiambo R, Ssewanyana D, Bottomley C, Ngugi AK, Newton CR, and SEEDS Writing Group. Adverse perinatal events, treatment gap, and positive family history linked to the high burden of active convulsive epilepsy in Uganda: A population-based study. Epilepsia Open. 2017; 2:188–98. https://doi.org/10.1002/epi4.12048 [PubMed]
- 13. Colnaghi S, Beltrami G, Poloni G, Pichiecchio A, Bastianello S, Galimberti CA, Versino M. Parahippocampal Involvement in Mesial Temporal Lobe Epilepsy with Hippocampal Sclerosis: A Proof of Concept from Memory-Guided Saccades. Front Neurol. 2017; 8:595. https://doi.org/10.3389/fneur.2017.00595 [PubMed]
- 14. Tao JX, Wu S, Lacy M, Rose S, Issa NP, Yang CW, Dorociak KE, Bruzzone M, Kim J, Daif A, Choi J, Towle VL, Warnke PC. Stereotactic EEG-guided laser interstitial thermal therapy for mesial temporal lobe epilepsy. J Neurol Neurosurg Psychiatry. 2018; 89:542–48. https://doi.org/10.1136/jnnp-2017-316833 [PubMed]
- 15. Dubey D, McRae PA, Rankin-Gee EK, Baranov E, Wandrey L, Rogers S, Porter BE. Increased metalloproteinase activity in the hippocampus following status epilepticus. Epilepsy Res. 2017; 132:50–58. https://doi.org/10.1016/j.eplepsyres.2017.02.021 [PubMed]
- 16. Rempe RG, Hartz AM, Soldner EL, Sokola BS, Alluri SR, Abner EL, Kryscio RJ, Pekcec A, Schlichtiger J, Bauer B. Matrix Metalloproteinase-Mediated Blood-Brain Barrier Dysfunction in Epilepsy. J Neurosci. 2018; 38:4301–15. https://doi.org/10.1523/JNEUROSCI.2751-17.2018 [PubMed]
- 17. Wang R, Zeng GQ, Liu X, Tong RZ, Zhou D, Hong Z. Evaluation of serum matrix metalloproteinase-3 as a biomarker for diagnosis of epilepsy. J Neurol Sci. 2016; 367:291–97. https://doi.org/10.1016/j.jns.2016.06.031 [PubMed]
- 18. Korotkov A, Broekaart DW, van Scheppingen J, Anink JJ, Baayen JC, Idema S, Gorter JA, Aronica E, van Vliet EA. Increased expression of matrix metalloproteinase 3 can be attenuated by inhibition of microRNA-155 in cultured human astrocytes. J Neuroinflammation. 2018; 15:211. https://doi.org/10.1186/s12974-018-1245-y [PubMed]
- 19. Sun L, Sun P, Zhou QY, Gao X, Han Q. Long noncoding RNA MALAT1 promotes uveal melanoma cell growth and invasion by silencing of miR-140. Am J Transl Res. 2016; 8:3939–46. [PubMed]
- 20. Wang Z, Yuan J, Li L, Yang Y, Xu X, Wang Y. Long non-coding RNA XIST exerts oncogenic functions in human glioma by targeting miR-137. Am J Transl Res. 2017; 9:1845–55. [PubMed]
- 21. Wu Z, He Y, Li D, Fang X, Shang T, Zhang H, Zheng X. Long noncoding RNA MEG3 suppressed endothelial cell proliferation and migration through regulating miR-21. Am J Transl Res. 2017; 9:3326–35. [PubMed]
- 22. Li J, Li Z, Zheng W, Li X, Wang Z, Cui Y, Jiang X. LncRNA-ATB: an indispensable cancer-related long noncoding RNA. Cell Prolif. 2017; 50:e12381. https://doi.org/10.1111/cpr.12381 [PubMed]
- 23. Aguilo F, Di Cecilia S, Walsh MJ. Long Non-coding RNA ANRIL and Polycomb in Human Cancers and Cardiovascular Disease. Curr Top Microbiol Immunol. 2016; 394:29–39. https://doi.org/10.1007/82_2015_455 [PubMed]
- 24. Xi Y, Jiang T, Wang W, Yu J, Wang Y, Wu X, He Y. Long non-coding HCG18 promotes intervertebral disc degeneration by sponging miR-146a-5p and regulating TRAF6 expression. Sci Rep. 2017; 7:13234. https://doi.org/10.1038/s41598-017-13364-6 [PubMed]
- 25. Wu P, Zuo X, Deng H, Liu X, Liu L, Ji A. Roles of long noncoding RNAs in brain development, functional diversification and neurodegenerative diseases. Brain Res Bull. 2013; 97:69–80. https://doi.org/10.1016/j.brainresbull.2013.06.001 [PubMed]
- 26. Chen X, Dong H, Liu S, Yu L, Yan D, Yao X, Sun W, Han D, Gao G. Long noncoding RNA MHENCR promotes melanoma progression via regulating miR-425/489-mediated PI3K-Akt pathway. Am J Transl Res. 2017; 9:90–102. [PubMed]
- 27. Liu J, Song Z, Feng C, Lu Y, Zhou Y, Lin Y, Dong C. The long non-coding RNA SUMO1P3 facilitates breast cancer progression by negatively regulating miR-320a. Am J Transl Res. 2017; 9:5594–602. [PubMed]
- 28. Yu Y, Yang J, Li Q, Xu B, Lian Y, Miao L. LINC00152: A pivotal oncogenic long non-coding RNA in human cancers. Cell Prolif. 2017; 50:e12349. https://doi.org/10.1111/cpr.12349 [PubMed]
- 29. Chen J, Zhang F, Wang J, Hu L, Chen J, Xu G, Wang Y. LncRNA LINC01512 Promotes the Progression and Enhances Oncogenic Ability of Lung Adenocarcinoma. J Cell Biochem. 2017; 118:3102–10. https://doi.org/10.1002/jcb.26178 [PubMed]
- 30. Ye K, Wang S, Zhang H, Han H, Ma B, Nan W. Long Noncoding RNA GAS5 Suppresses Cell Growth and Epithelial-Mesenchymal Transition in Osteosarcoma by Regulating the miR-221/ARHI Pathway. J Cell Biochem. 2017; 118:4772–81. https://doi.org/10.1002/jcb.26145 [PubMed]
- 31. Yang C, Wu K, Wang S, Wei G. Long non-coding RNA XIST promotes osteosarcoma progression by targeting YAP via miR-195-5p. J Cell Biochem. 2018; 119:5646–56. https://doi.org/10.1002/jcb.26743 [PubMed]
- 32. Chen X, Liu S, Zhao X, Ma X, Gao G, Yu L, Yan D, Dong H, Sun W. Long noncoding RNA ILF3-AS1 promotes cell proliferation, migration, and invasion via negatively regulating miR-200b/a/429 in melanoma. Biosci Rep. 2017; 37:BSR20171031. https://doi.org/10.1042/BSR20171031 [PubMed]
- 33. Mao X, Qin X, Li L, Zhou J, Zhou M, Li X, Xu Y, Yuan L, Liu QN, Xing H. A 15-long non-coding RNA signature to improve prognosis prediction of cervical squamous cell carcinoma. Gynecol Oncol. 2018; 149:181–87. https://doi.org/10.1016/j.ygyno.2017.12.011 [PubMed]
- 34. Zhou M, Hu L, Zhang Z, Wu N, Sun J, Su J. Recurrence-Associated Long Non-coding RNA Signature for Determining the Risk of Recurrence in Patients with Colon Cancer. Mol Ther Nucleic Acids. 2018; 12:518–29. https://doi.org/10.1016/j.omtn.2018.06.007 [PubMed]
- 35. Gao G, Li W, Liu S, Han D, Yao X, Jin J, Han D, Sun W, Chen X. The positive feedback loop between ILF3 and lncRNA ILF3-AS1 promotes melanoma proliferation, migration, and invasion. Cancer Manag Res. 2018; 10:6791–802. https://doi.org/10.2147/CMAR.S186777 [PubMed]
- 36. Hu XH, Dai J, Shang HL, Zhao ZX, Hao YD. SP1-mediated upregulation of lncRNA ILF3-AS1 functions a ceRNA for miR-212 to contribute to osteosarcoma progression via modulation of SOX5. Biochem Biophys Res Commun. 2019; 511:510–17. https://doi.org/10.1016/j.bbrc.2019.02.110 [PubMed]
- 37. Han CL, Ge M, Liu YP, Zhao XM, Wang KL, Chen N, Meng WJ, Hu W, Zhang JG, Li L, Meng FG. LncRNA H19 contributes to hippocampal glial cell activation via JAK/STAT signaling in a rat model of temporal lobe epilepsy. J Neuroinflammation. 2018; 15:103. https://doi.org/10.1186/s12974-018-1139-z [PubMed]
- 38. Wang HK, Yan H, Wang K, Wang J. Dynamic regulation effect of long non-coding RNA-UCA1 on NF-kB in hippocampus of epilepsy rats. Eur Rev Med Pharmacol Sci. 2017; 21:3113–19. [PubMed]
- 39. Beghi E, Giussani G. Aging and the Epidemiology of Epilepsy. Neuroepidemiology. 2018; 51:216–23. https://doi.org/10.1159/000493484 [PubMed]
- 40. Sarkis RA, McGinnis S, Rushia SN, Park S, Ansari EE, Willment KC. Growing older with drug-resistant epilepsy: cognitive and psychosocial outcomes. J Neurol. 2018; 265:1059–64. https://doi.org/10.1007/s00415-018-8805-z [PubMed]
- 41. van Vliet EA, Aronica E, Vezzani A, Ravizza T. Review: Neuroinflammatory pathways as treatment targets and biomarker candidates in epilepsy: emerging evidence from preclinical and clinical studies. Neuropathol Appl Neurobiol. 2018; 44:91–111. https://doi.org/10.1111/nan.12444 [PubMed]
- 42. de Lemos L, Junyent F, Camins A, Castro-Torres RD, Folch J, Olloquequi J, Beas-Zarate C, Verdaguer E, Auladell C. Neuroprotective Effects of the Absence of JNK1 or JNK3 Isoforms on Kainic Acid-Induced Temporal Lobe Epilepsy-Like Symptoms. Mol Neurobiol. 2018; 55:4437–52. https://doi.org/10.1007/s12035-017-0669-1 [PubMed]
- 43. Weidner LD, Kannan P, Mitsios N, Kang SJ, Hall MD, Theodore WH, Innis RB, Mulder J. The expression of inflammatory markers and their potential influence on efflux transporters in drug-resistant mesial temporal lobe epilepsy tissue. Epilepsia. 2018; 59:1507–17. https://doi.org/10.1111/epi.14505 [PubMed]
- 44. Zhao T, Ding Y, Li M, Zhou C, Lin W. Silencing lncRNA PVT1 inhibits activation of astrocytes and increases BDNF expression in hippocampus tissues of rats with epilepsy by downregulating the Wnt signaling pathway. J Cell Physiol. 2019; 234:16054–67. https://doi.org/10.1002/jcp.28264 [PubMed]
- 45. Han CL, Ge M, Liu YP, Zhao XM, Wang KL, Chen N, Hu W, Zhang JG, Li L, Meng FG. Long non-coding RNA H19 contributes to apoptosis of hippocampal neurons by inhibiting let-7b in a rat model of temporal lobe epilepsy. Cell Death Dis. 2018; 9:617. https://doi.org/10.1038/s41419-018-0496-y [PubMed]
- 46. Geng JF, Liu X, Zhao HB, Fan WF, Geng JJ, Liu XZ. LncRNA UCA1 inhibits epilepsy and seizure-induced brain injury by regulating miR-495/Nrf2-ARE signal pathway. Int J Biochem Cell Biol. 2018; 99:133–39. https://doi.org/10.1016/j.biocel.2018.03.021 [PubMed]
- 47. Fisher RS. The New Classification of Seizures by the International League Against Epilepsy 2017. Curr Neurol Neurosci Rep. 2017; 17:48. https://doi.org/10.1007/s11910-017-0758-6 [PubMed]


