Introduction
Bladder urothelial carcinoma (BCa) has been identified as the ninth most common malignant neoplasm all over the world [1, 2]. More than 199,000 people died of it and over 549,000 cases were newly diagnosed in 2018 [1, 2]. In the past twenty years, the number of BCa incident cases is growing globally and the BCa burden may ascend in the future due to aging of population and environmental pollution [3, 4]. Although various methods including transurethral resection, radical cystectomy, radiotherapy and chemotherapy are performed in treatment of BCa patients, BCa is aggressive and has a high risk of recurrence, progression, metastasis and poor prognosis [5–7]. The risk of recurrence within 5 years after initial treatment ranges from 50% to 90% in non-muscle-invasive bladder cancer (NMIBC) [2]. The invasion and metastasis of muscle-invasive bladder cancer (MIBC) are vital causes of recurrence and poor prognosis [8, 9]. Hence, it is critical that we should explore and develop reliable prognostic biomarkers to provide prognostic predictors and treatment targets for BCa, which could improve the prognosis of BCa patients.
Recently, lots of evidence shows that disorders of immune system process and immune response play a critical role in tumor microenvironment [10]. Through perturbing the molecular signal and activating the immune response, immune cells could suppress tumor recurrence, progression and metastasis [11, 12]. However, some tumor cells could avoid detection by the immune system, suppress immune response and escape from immune elimination to induce tumor invasion and metastasis [13, 14]. It was reported that dysregulation of immune status induced by tumors might be associated with glioblastoma progression [15, 16]. In the meantime, the immune microenvironment of pancreatic cancer is highly suppressed by immunosuppressive macrophages and myeloid-derived suppressive cells [14]. As to BCa, it is clear from the evidence considered that immune system is highly active in the microenvironment of BCa. Nevertheless, some of these activities are greatly counter-productive and pro-tumorigenic [17].
Long non-coding RNAs (lncRNAs) are a series of RNAs without protein-coding capacity and their lengths are over 200 nucleotides (bp) [18]. Abundant evidence exists to suggest that lncRNAs contribute to tumor development and metastasis through activating immune system process and immune response including antigen release, antigen presentation, immune cell differentiation, immune cells migration, T cells infiltration and recognition and killing of cancer cells [10, 19]. LncRNA CECR7 (cat eye syndrome chromosome region, candidate 7) activates the expression of CTLA4 (cytotoxic T-lymphocyte-associated protein 4) by targeting miR-429 in diabetic pancreatic cancer, which suggested that lncRNAs are involved in immune cell differentiation [20]. Another research revealed that, lnc-sox5 is pivotal in immune cells infiltration in colorectal cancer (CRC). Lnc-sox5 knock-down can directly increase the activity of regulatory T cells and their cytotoxicity is also dramatically enhanced in CRC [21]. In addition, bioinformatics analyses based on The Cancer Genome Atlas (TCGA) database demonstrated that lncRNAs are strongly implicated in the carcinogenesis of BCa through immune-associated pathways [22].
Therefore, aberrantly expressed lncRNAs may be potential prognostic biomarkers for BCa patients and may be served as potential therapeutic targets. As a consequence, based on the gene expression profile of high-throughput sequencing data obtained from TCGA, we carried out this present study in order to explore lncRNAs targeting immune-related genes and further construct an immune-related prognostic signature for MIBC patients.
Results
The flowchart of the present study was summarized in Figure 1.
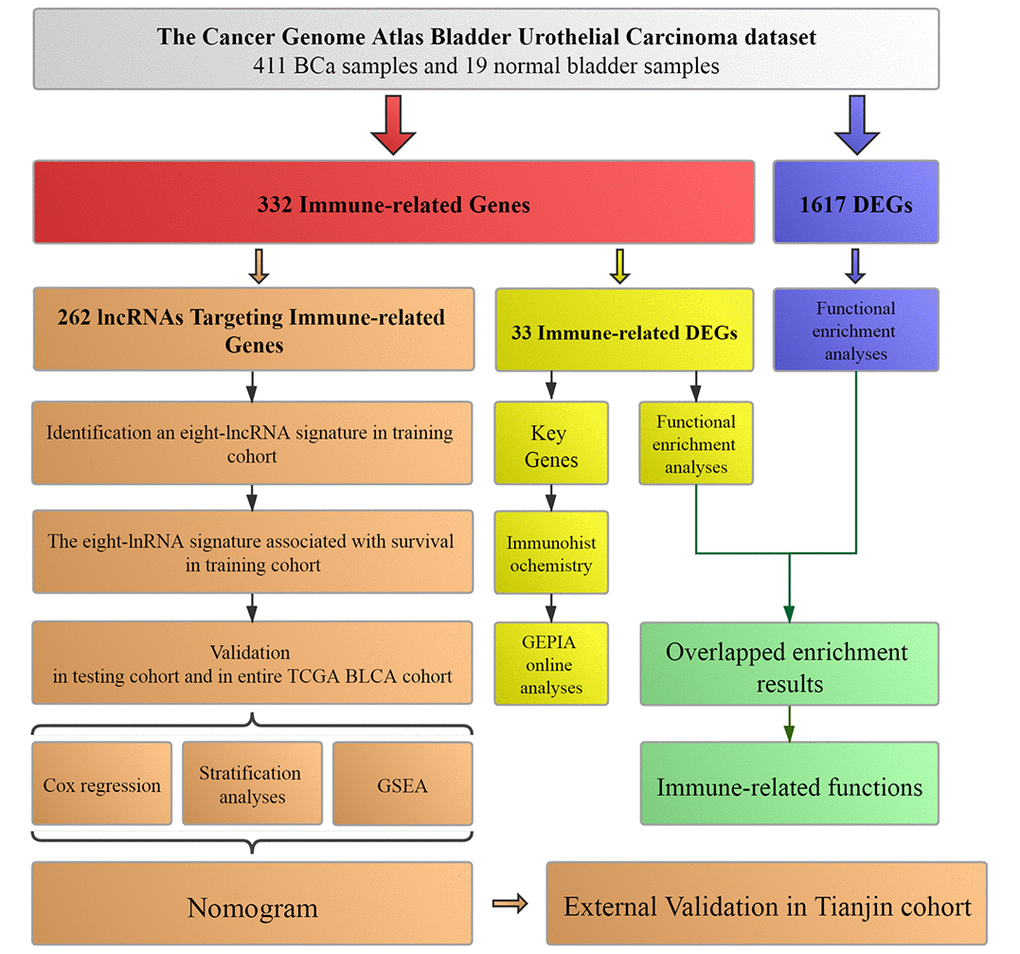
Figure 1. Workflow of this study. The study was carried out in TCGA (The Cancer Genome Atlas) BLCA (Bladder Urothelial Carcinoma) dataset. Immune-related genes were extracted from Molecular Signatures Database v4.0. LncRNAs targeting immune-related genes were identified according to Pearson correlation. DEGs (differentially expressed genes) were calculated between BCa (bladder urothelial carcinoma) samples and normal bladder samples in TCGA BLCA dataset. The training cohort was used to identify the lncRNAs targeting immune-related genes and establish a prognostic signature based on the prognostic lncRNAs. The prognosis analysis was validated in the testing cohort, entire TCGA BLCA cohort and Tianjin validation cohort, respectively. Nomogram was constructed by including the immune-related signature and other prognosis-related clinical features in training cohort. Immunohistochemistry from THPA (The Human Protein Atlas) and online analyses from GEPIA (Gene Expression Profiling Interactive Analysis) were used to validate four key immune-related genes (CTSG, CXCL12, LIG1 and TBX1). Functional enrichment analyses were utilized to explore immune-related functions.
Functional enrichment analysis
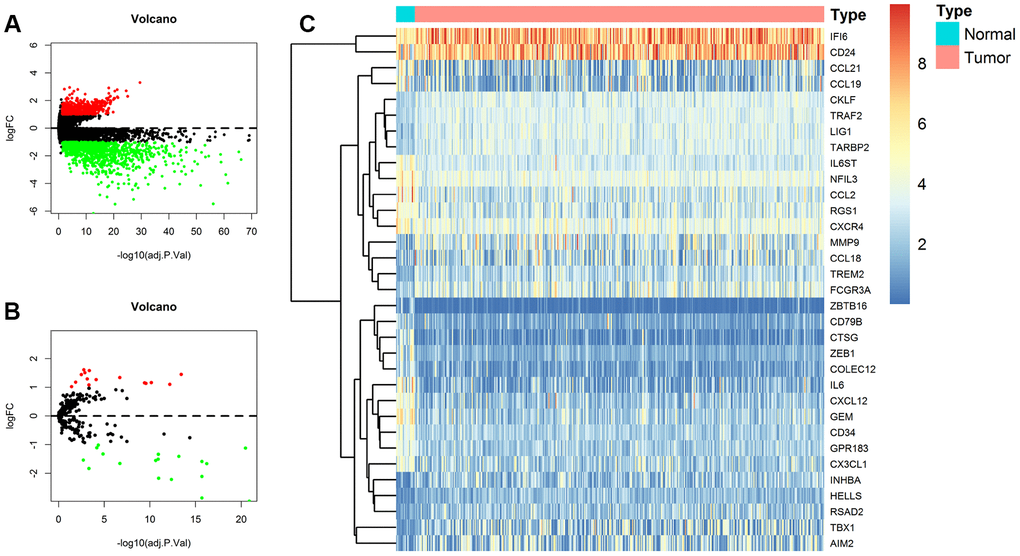
We performed Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analyses to determine the biological function and pathways involved in the 1617 DEGs in TCGA BLCA dataset. The top 10 enrichment items of biological process (BP) and KEGG pathway analyses were illustrated in Figure 3A, 3B, separately.
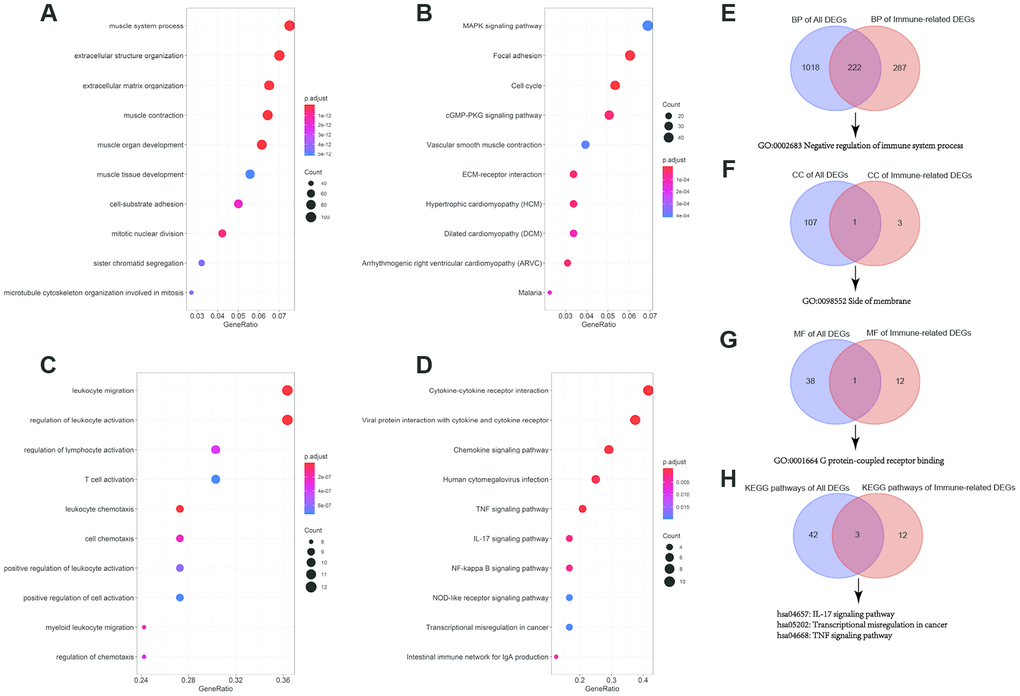
Figure 3. GO (Gene Ontology) functional and KEGG (Kyoto Encyclopedia of Genes and Genomes) pathway enrichment analyses of differentially expressed genes (DEGs) and immune-related DEGs. (A) Top 10 BP (biological process) terms of all DEGs; (B) Top 10 KEGG pathways of all DEGs; (C) Top 10 BP terms of immune-related DEGs; (D) Top 10 KEGG pathways of immune-related DEGs; (E) Venn diagram for overlapped BP terms; (F) Venn diagram for overlapped CC (cell component) terms; (G) Venn diagram for overlapped MF (molecular function) terms; (H) Venn diagram for overlapped MF (molecular function) KEGG pathways.
At the same time, we also analyzed the 33 immune-related DEGs with GO and KEGG pathway enrichment analyses (Figure 3C, 3D).
Next, to validate and confirm that BCa is closely immune-related, we adopted Venn diagram to obtain overlapped enrichment results between all DEGs enrichment analysis and immune-related DEGs enrichment analysis. We found that they both had enrichments in negative regulation of immune system process (GO:0002683), G protein-coupled receptor binding (GO:0001664) and transcriptional misregulation in cancer (hsa05202), which indicated that the development of BCa was associated with immune system and immune response (Figure 3E).
Clinical characteristics of TCGA BLCA patients
A total of 389 BCa patients were involved in construction and validation of the lncRNA signature. They were randomly assigned to a training cohort (n = 195) for construction and a testing cohort (n = 194) for validation. Clinical characteristics of 389 BCa patients were displayed in Table 1.
Table 1. Clinical characteristics of 389 bladder urothelial carcinoma patients involved in identification and validation of the 8-lncRNA prognostic signature.
| Characteristics | Entire TCGA BLCA cohort (N=389) | Detailed data | P-value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Training cohort (N=195) | Testing cohort (N=194) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age at diagnosis (years) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| <65 | 147 (37.8%) | 64 (32.8%) | 83 (42.8%) | 0.055 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≥65 | 242 (62.2%) | 131 (67.2%) | 111 (57.2%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male | 287 (73.8%) | 140 (71.8%) | 147 (75.8%) | 0.372 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female | 102 (26.2%) | 55 (28.2%) | 47 (24.2%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Histological grade | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Low grade | 25 (6.4%) | 8 (4.1%) | 17 (8.8%) | 0.061 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| High grade | 364 (93.6%) | 187 (95.9%) | 177 (91.2%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TNM (UICC) stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stage I | 2 (0.5%) | 2 (1.0%) | 0 (0.0%) | 0.42 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stage II | 128 (32.9%) | 68 (34.9%) | 60 (30.9%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stage III | 136 (35.0%) | 66 (33.8%) | 70 (36.1%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stage IV | 123 (31.6%) | 59 (30.3%) | 64 (33.0%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| a Chi-square test. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Identification of the 8-lncRNA prognostic signature in training cohort
Pearson correlation analysis identified that 262 lncRNAs were correlated with the 18 up-regulated immune-related DEGs or 15 down-regulated immune-related DEGs in the TCGA BLCA dataset (|R| > 0.5 and P < 0.01). And the 262 lncRNAs were lncRNAs targeting immune-related genes. Next, we used univariate Cox regression analysis method to identify prognosis-related lncRNAs from the 262 lncRNAs in the training cohort and univariate Cox regression analysis identified 21 prognosis-related lncRNAs (P < 0.01). Then, we used stepwise selection with Akaike information criteria (AIC) from the 21 prognosis-related lncRNAs to select the optimal model. Ultimately, we selected 8 lncRNAs with the smallest AIC value to construct the predictive model002E
Details of the 8 lncRNAs with their gene symbols, Ensembel IDs, descriptions, coefficients and results of univariate Cox regression analysis were summarized in Table 2. Among these eight lncRNAs, two are deleterious lncRNAs with univariate Cox hazard ratio (HR) > 1 (WNT5A-AS1 and AL136084.3), which indicated that patients with high expression of the two lncRNAs might have a poor survival time. The other six are protective lncRNAs with univariate Cox HR < 1 (MIF-AS1, AC008735.2, AL357033.4, LINC00649, AC099343.2 and USP30-AS1), which indicated that high expression of the six lncRNAs might result in a better survival time.
Table 2. The eight lncRNAs identified from Cox regression analysis.
| Gene symbol | Ensembel ID | Description | Coefficient | Univariate Cox regression analysis | ||
| HR | 95%CI | P-value | ||||
| WNT5A-AS1 | ENSG00000244586 | WNT5A Antisense RNA 1 | 0.231 | 1.335 | 1.113-1.601 | 0.002 |
| AL136084.3 | ENSG00000270412 | - | 0.420 | 1.686 | 1.194-2.380 | 0.003 |
| MIF-AS1 | ENSG00000218537 | MIF Antisense RNA 1 | -0.564 | 0.460 | 0.258-0.820 | 0.009 |
| AC008735.2 | ENSG00000267523 | - | -0.388 | 0.672 | 0.510-0.885 | 0.005 |
| AL357033.4 | ENSG00000277496 | - | -0.456 | 0.576 | 0.403-0.823 | 0.002 |
| LINC00649 | ENSG00000237945 | Long Intergenic Non-Protein Coding RNA 649 | -0.661 | 0.486 | 0.290-0.817 | 0.006 |
| AC099343.2 | ENSG00000270426 | - | -0.633 | 0.280 | 0.143-0.548 | <0.001 |
| USP30-AS1 | ENSG00000256262 | USP30 Antisense RNA 1 | -0.263 | 0.697 | 0.536-0.905 | 0.007 |
Based on the expression of these eight lncRNAs for overall survival (OS) prediction, we established a risk score of the 8-lncRNA signature with the following formula:
Risk score = (0.231 × ExpressionWNT5A-AS1) + (0.420 × ExpressionAL136084.3) + (-0.564 × ExpressionMIF-AS1) + (-0.388 × ExpressionAC008735.2) + (-0.456 × ExpressionAL357033.4) + (-0.661 × ExpressionLINC00649) + (-0.633 × ExpressionAC099343.2) + (-0.263 × ExpressionUSP30-AS1).
Evaluating the predictive power of the 8-lncRNA prognostic signature in training cohort
195 BCa patients in training cohort were divided into high-risk (n = 98) and low-risk (n = 97) groups determined by the median value of 8-lncRNA signature risk score. The risk score, survival time and survival status of each patient in training cohort were illustrated in Figure 4A, 4B. The heat map of the expression of the eight lncRNAs was displayed in Figure 4C.
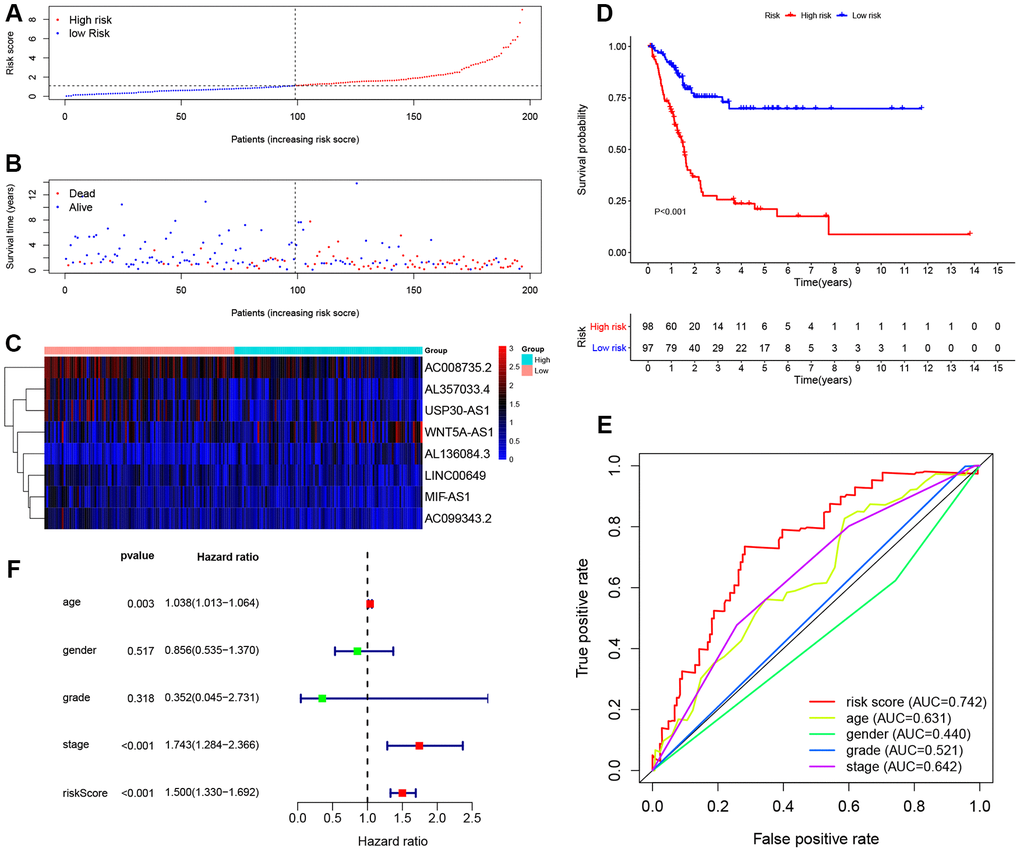
Figure 4. Evaluating the predictive power of the 8-lncRNA immune-related signature in the training cohort. (A–C) Distribution of risk score, survival status, and lncRNA expression of patients in the training cohort; (D) Kaplan-Meier survival curve of the high-risk and low-risk groups in the training cohort; (E) Time-dependent ROC (receiver operating characteristic) curves and AUC (area under curve) based on the training cohort for 5-year overall survival; (F) Forest plot for multivariate Cox regression analysis.
The Kaplan-Meier (KM) survival curve of low-risk and high-risk groups in training cohort was shown in Figure 4D. The survival time of the low-risk group was significantly higher than that of the high-risk group (P < 0.001). In addition, time-dependent receiver operating characteristic (ROC) curves of 8-lncRNA prognostic signature and other clinical characteristics were plotted. The area under curve (AUC) of 8-lncRNA prognostic signature risk score was 0.742, which was better than other clinical characteristics in Figure 4E.
Furthermore, multivariate Cox regression analyses of age, gender, Union for International Cancer Control (UICC) stage, histological grade and 8-lncRNA prognostic signature were evaluated in Figure 4F. Risk score of the 8-lncRNA signature (HR = 1.500, 95%CI = 1.330-1.692, P < 0.001) might be considered as an independent prognostic indicator of OS (Table 3).
Table 3. Univariate and multivariate Cox regression analyses of the training, testing, and entire TCGA BLCA cohorts.
| Variables | Univariate analysis | Multivariate analysis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HR | 95%CI | P-value | HR | 95%CI | P-value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Training cohort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 8-lncRNA Risk score (continuous variables) | 1.514 | 1.356-1.691 | <0.001 | 1.500 | 1.330-1.692 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age at diagnosis (≥65 years vs. <65 years) | 1.042 | 1.018-1.067 | 0.001 | 1.038 | 1.013-1.064 | 0.003 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender (Male vs. Female) | 0.761 | 0.480-1.207 | 0.246 | 0.856 | 0.535-1.370 | 0.517 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Histological grade (High grade vs. Low grade) | 1.708 | 0.235-12.389 | 0.597 | 0.352 | 0.045-2.731 | 0.318 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TNM (UICC) stage (Stage IV+Stage III vs. Stage II+Stage I) | 1.865 | 1.412-2.464 | <0.001 | 1.743 | 1.284-2.366 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Testing cohort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 8-lncRNA Risk score (continuous variables) | 1.141 | 1.100-1.184 | <0.001 | 1.137 | 1.090-1.186 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age at diagnosis (≥65 years vs. <65 years) | 1.029 | 1.003-1.055 | 0.027 | 1.015 | 0.989-1.042 | 0.250 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender (Male vs. Female) | 1.011 | 0.584-1.750 | 0.968 | 0.814 | 0.463-1.432 | 0.476 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Histological grade (High grade vs. Low grade) | 1.340 | 0.322-5.578 | 0.687 | 0.588 | 0.130-2.665 | 0.491 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TNM (UICC) stage (Stage IV+Stage III vs. Stage II+Stage I) | 1.623 | 1.185-2.223 | 0.003 | 1.749 | 1.238-2.471 | 0.002 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Entire TCGA BLCA cohort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 8-lncRNA Risk score (continuous variables) | 1.042 | 1.037-1.047 | <0.001 | 1.047 | 1.041-1.053 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age at diagnosis (≥65 years vs. <65 years) | 1.037 | 1.020-1.055 | <0.001 | 1.043 | 1.024-1.062 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender (Male vs. Female) | 0.859 | 0.604-1.220 | 0.395 | 0.636 | 0.441-0.915 | 0.015 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Histological grade (High grade vs. Low grade) | 2.090 | 0.977-4.470 | 0.057 | 1.026 | 0.441-2.385 | 0.952 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TNM (UICC) stage (Stage IV+Stage III vs. Stage II+Stage I) | 1.848 | 1.496-2.282 | <0.001 | 1.935 | 1.500-2.496 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HR hazard ratio, CI confidence interval. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Validation of the 8-lncRNA prognostic signature in testing cohort and entire TCGA BLCA cohort
In order to further confirm the predictive power and stability of the 8-lncRNA signature in predicting the OS of BCa patients, we validated it in testing cohort (n = 194) and entire TCGA BLCA cohort (n = 389) (Figures 5 and 6). Risk scores of the 8-lncRNA signature were also calculated with the above mentioned formula.
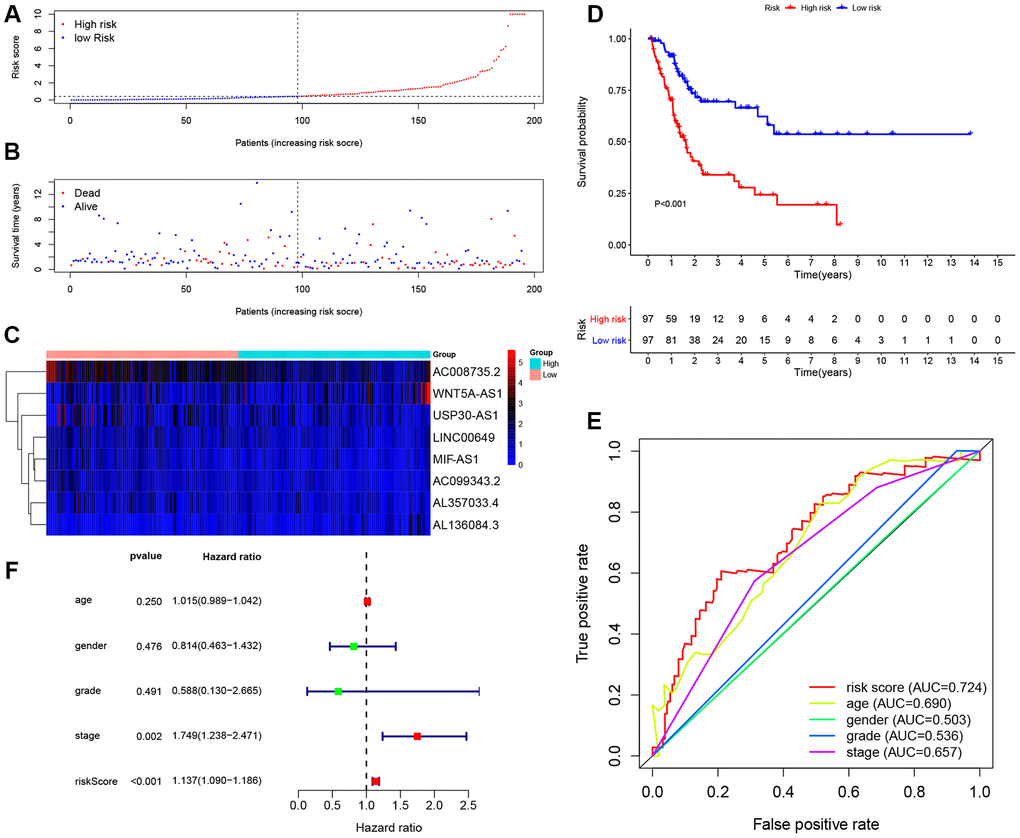
Figure 5. Evaluating the predictive power of the 8-lncRNA immune-related signature in the testing cohort. (A–C) Distribution of risk score, survival status, and lncRNA expression of patients in the testing cohort; (D) Kaplan-Meier survival curve of the high-risk and low-risk groups in the testing cohort; (E) Time-dependent ROC (receiver operating characteristic) curves and AUC (area under curve) based on the testing cohort for 5-year overall survival; (F) Forest plot for multivariate Cox regression analysis.
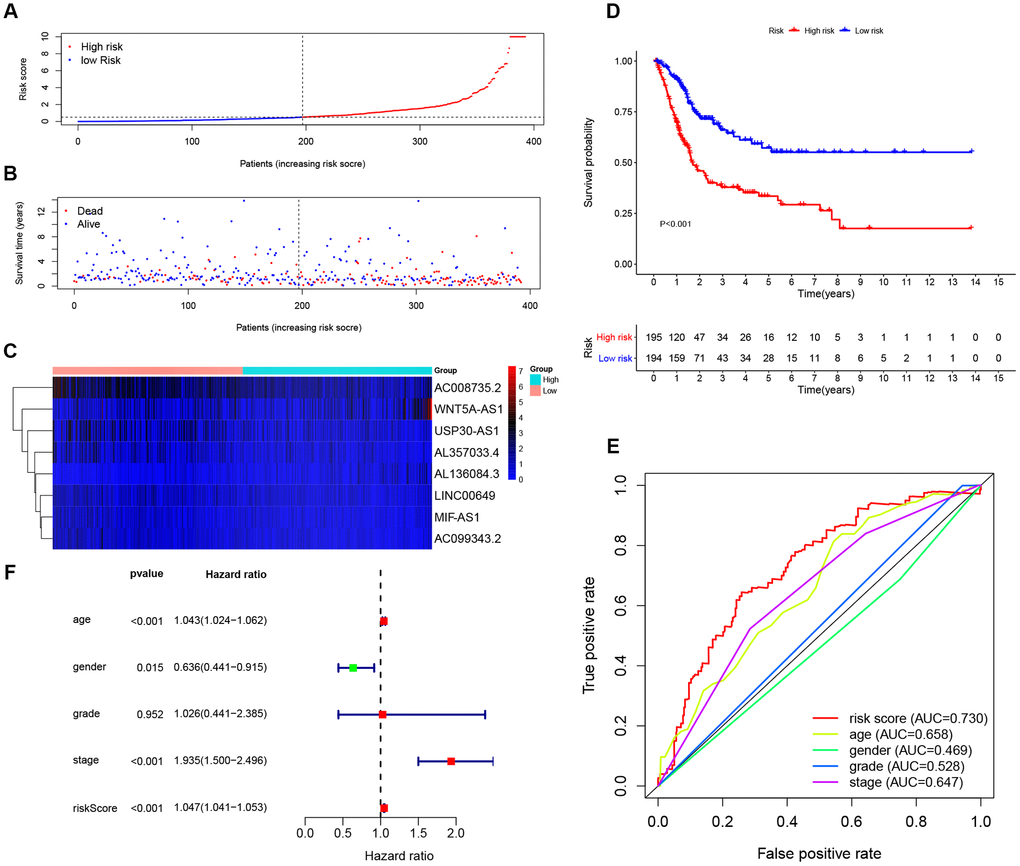
Figure 6. Evaluating the predictive power of the 8-lncRNA immune-related signature in the entire TCGA (The Cancer Genome Atlas) BLCA (Bladder Urothelial Carcinoma) cohort. (A–C) Distribution of risk score, survival status, and lncRNA expression of patients in the entire TCGA BLCA cohort; (D) Kaplan-Meier survival curve of the high-risk and low-risk groups in the entire TCGA BLCA cohort; (E) Time-dependent ROC (receiver operating characteristic) curves and AUC (area under curve) based on the entire TCGA BLCA cohort for 5-year overall survival; (F) Forest plot for multivariate Cox regression analysis.
The KM survival curve of low-risk and high-risk groups in testing cohort was shown in Figure 5D. The survival time of the low-risk group was significantly higher than that of the high-risk group (P < 0.001). In addition, time-dependent ROC curves of 8-lncRNA prognostic signature and other clinical characteristics were plotted. Consistent with the finding in training cohort, the AUC of 8-lncRNA prognostic signature risk score was 0.724, which was better than other clinical characteristics in Figure 5E. Multivariate Cox regression analysis revealed that risk score of the 8-lncRNA signature (HR = 1.137, 95%CI = 1.090-1.186, P < 0.001) might be an independent prognostic indicator in Figure 5F and Table 3. Similar results of validation were obtained in entire TCGA BLCA cohort.
Survival and Clinical characteristics with 8-lncRNA prognostic signature in entire TCGA BLCA cohort
To further validate the prognostic value and explore the wide applicability of the immune-related signature, we performed survival analyses through stratification analysis based on entire TCGA BLCA cohort (Figure 7A–7G). Samples in the high-risk group had poor survival compared with those in the low-risk group among stage III (P < 0.001) and stage IV (P = 0.022) patients. Similar significant results were revealed in different age groups and different gender groups.
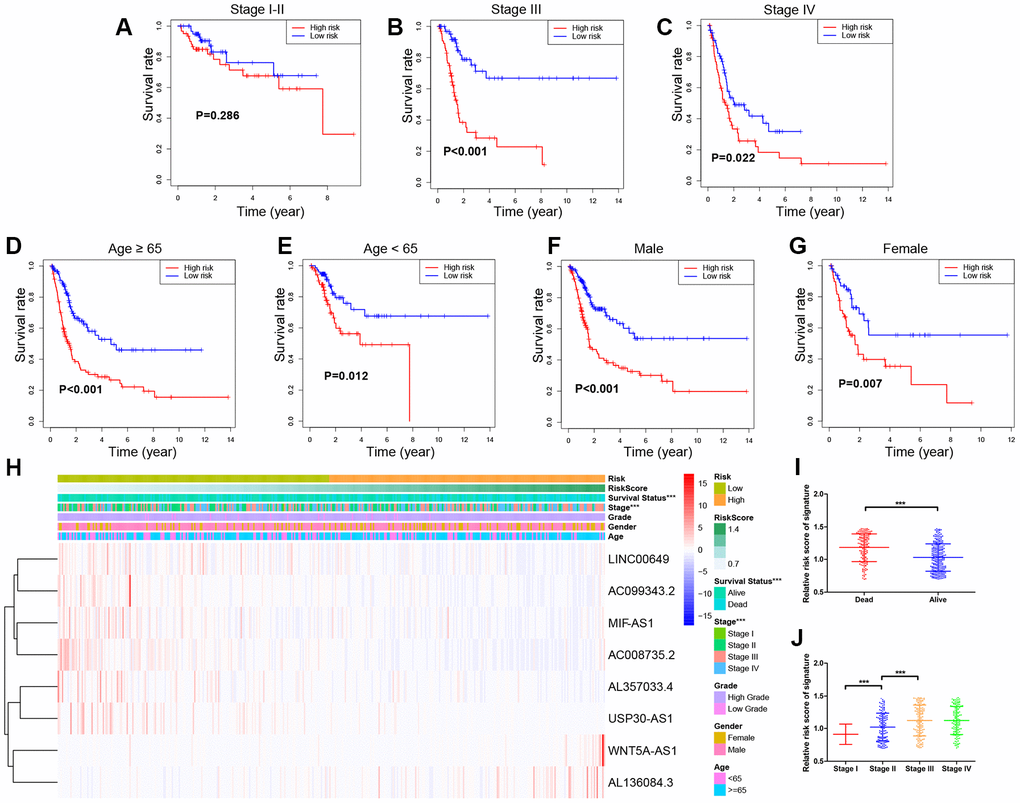
Figure 7. Stratified survival analyses and Clinical characteristics with 8-lncRNA prognostic signature in the entire TCGA (The Cancer Genome Atlas) BLCA (Bladder Urothelial Carcinoma) cohort. (A–G) Kaplan-Meier survival curves in subgroups stratified by different clinical characteristics; (H) Distribution of clinicopathologic features, and lncRNA expression in low-risk and high-risk groups; (I) Risk score comparison between alive and dead patients; (J) Risk score comparison between different tumor stages. *** P-value < 0.005.
Clinical characteristics exhibited distributed patterns corresponding to the risk score of 8-lncRNA prognostic signature on the entire TCGA BLCA cohort (Figure 7H). We found that dead samples more likely gathered in the high-risk group, indicating that samples with high risk score might have worse survival (P < 0.005) (Figure 7I). In addition, samples with higher UICC stages (stage III and stage IV) were more likely to have higher risk scores than those with lower stages (stage I and stage II) (P < 0.005) (Figure 7J), which confirmed that higher scores of the 8-lncRNA prognostic signature might be significantly associated with the progression of BCa.
Principal component analysis (PCA) and Gene set enrichment analysis (GSEA)
PCA was performed to explore the different distribution patterns between low-risk and high-risk groups according to the eight lncRNAs targeting immune-related genes and the whole gene set. Based on the eight lncRNAs, low-risk and high-risk groups were significantly distributed in two different directions. That is to say, the eight lncRNAs were used to separate BCa patients into two sections, indicating that the BCa patients in the low-risk group was quite distinguished from those in the high-risk group (Figure 8A, 8B). However, these two groups did not show significant distinctions when PCA was performed based on the whole gene set (Figure 8C).
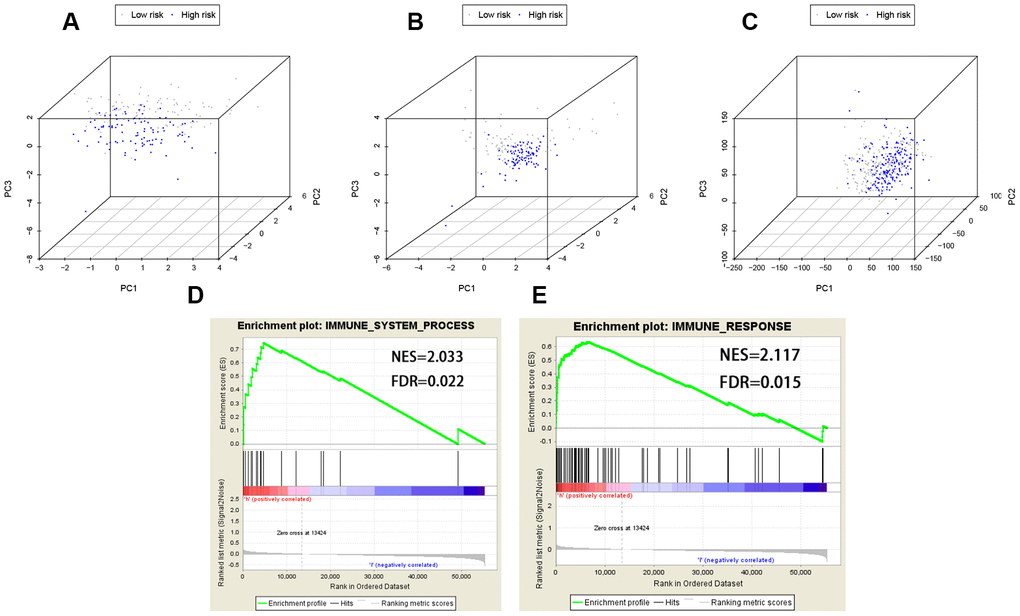
Figure 8. PCA (Principal components analysis) and GSEA (Gene set enrichment analysis). PCA based on the eight lncRNAs indicated low-risk and high-risk groups were generally distributed in two different directions in (A) the training cohort and (B) the testing cohort, respectively; (C) PCA based on the whole gene set indicated these two groups did not show significant distinctions; (D, E) GSEA indicated significant enrichment of immune-related phenotype in the high-risk group patients. FDR false discovery rate; NES normalized enrichment score.
GSEA was further utilized to validate the functional annotation between high-risk and low-risk groups in entire TCGA cohort. The DEGs between the two groups were enriched in immune system process pathway (FDR [False discovery rate] = 0.022, NES [normalized enrichment score] = 2.033) and immune response pathway (FDR = 0.015, NES = 2.117) (Figure 8D, 8E). In summary, the immune-related prognostic signature based on the eight lncRNAs targeting immune-related genes was significantly associated with the immune status of BCa, and high risk score of the signature was more likely to activate immune-related pathways in BCa patients.
Building and validating a predictive nomogram
Nomogram was constructed by combining the details of age, UICC stage, histological grade, and the 8-lncRNA prognostic signature in training cohort (Figure 10A). By using bootstrap method, calibration plots showed no significant deviation from the ideal for 1-year, 3-year and 5-year survival (Figure 10B). The training cohort (N = 195) was divided into two high-nomogram-score (N = 98) and low-nomogram-score (N = 97) groups by the median value of nomogram score. The KM curve showed that BCa patients in high-nomogram-score group had worse prognosis compared with low-nomogram-score group (P < 0.001) (Figure 10C). The AUCs for 1-year, 3-year and 5-year of their time-dependent ROC curves were 0.746, 0.829, and 0.825, respectively (Figure 10F). Combining the immune-related prognostic signature with other prognosis-related clinical factors increased the AUC for predicting OS of BCa patients.
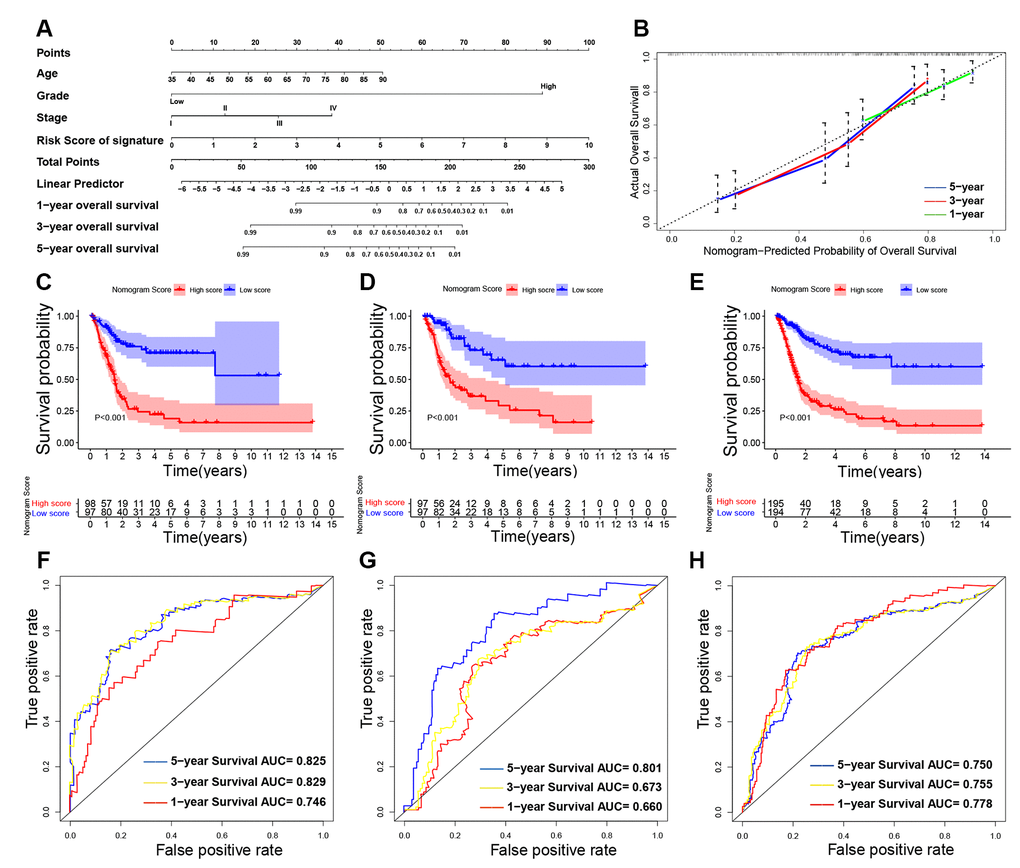
Figure 10. Building and validating the nomogram to predict prognosis in TCGA (The Cancer Genome Atlas) BLCA (Bladder Urothelial Carcinoma) dataset. (A) The nomogram was constructed based on age, UICC (Union for International Cancer Control) stage, histological grade and the immune-related signature in the training cohort; (B) The calibration plot for internal validation of the nomogram; (C–E) Kaplan-Meier survival curves between high-nomogram-score and low-nomogram-score groups in the training cohort, testing cohort and entire TCGA BLCA cohort, respectively; (F–H) Time-dependent ROC (receiver operating characteristic) curves and AUC (area under curve) for 1-year, 3-year, and 5-year overall survival based on the training cohort, testing cohort and entire TCGA BLCA cohort, respectively.
In addition, we also validated the nomogram in testing cohort (n = 194) and in entire TCGA BLCA cohort (n = 389). KM curves and their ROC curves were displayed in Figure 10D, 10E, 10G, 10H.
Table 4 showed the concordance index (c-index) of the immune-related signature and nomogram. Table 5 showed the AUC in time-dependent ROC curves. Both the c-index and AUC are indicators for evaluating the predictive value of prognostic model. The c-index of nomogram exceeded the c-index of immune-related signature in training cohort, testing cohort and entire TCGA BLCA cohort (P < 0.05), which indicated the predictive value of nomogram was better than the immune-related signature. In addition, the AUC of nomogram exceeded the AUC of immune-related signature, especially in predicting 5-year OS (P < 0.05), which indicated the nomogram had better predictive power in long-term follow-up. Therefore, the nomogram combined the immune-related prognostic signature with other prognosis-related clinical factors and increased the predictive power of OS, which might help to improve clinical management of BCa patients.
Table 4. Concordance index (C-index).
| C-index (95%CI) | Z-test | P-value | ||
| 8-lncRNA Risk score | Nomogram | |||
| Entire TCGA BLCA cohort | 0.678 (0.626-0.715) | 0.727 (0.697-0.748) | 2.303 | 0.021 |
| Training cohort | 0.694 (0.659-0.728) | 0.734 (0.691-0.756) | 1.975 | 0.048 |
| Testing cohort | 0.642 (0.598-0.671) | 0.716 (0.662-0.742) | 3.800 | <0.001 |
Discussion
Recently, the roles of lncRNAs in the development and prognosis of various tumors have been well investigated in many studies. Aberrantly expressed lncRNAs in cancer can be utilized as biomarkers for diagnosis, prognosis and target therapy [22]. Shen et al. performed a study based on TCGA BLCA dataset identified a panel of 20 key lncRNAs that were most implicated in MIBC prognosis after differential expression analysis and prognostic correlations [22]. Among the 20 lncRNAs, lnc-BOD1-1:7, -1:8, and -1:9, as well as lnc-GCH1-2:1, -2:2, and -2:3 were associated with both immune-related pathways and cancer-associated pathways. High expression level of lnc-CGRRF1-3:1 was mainly correlated with increased immune activity and decreased apoptosis in MIBC. In addition, Cao et al. carried out another study based on TCGA BLCA dataset to explore the tumor immune microenvironment of BCa [23]. They found the abundance ratio of four immune cells including T cell CD4 memory activated, T cell CD4 memory resting, T cell CD8 and natural killer (NK) cell resting was related to BCa survival. T cell CD4 memory resting, T cell CD8, NK cell resting decreased as the increase of UICC stage and lymph node metastasis, which indicated that the four cells are vital in immune infiltration as well as BCa immunotherapy.
As immunotherapy has attracted increasing attention in the field of cancer treatment, the gene expression profile of high-throughput sequencing data have been used in order to explore valuable immune-related biomarkers, identify potential prognostic targets and analyze underlying mechanisms [24, 25]. In the present study, we adopted TCGA BLCA dataset to explore lncRNAs targeting immune-related genes and construct an immune-related signature, which may serve as promising prognostic indicators for BCa and can be applied as promising immune therapeutic targets.
Previous studies have identified that the immune system is significantly associated with the tumor microenvironment, especially tumor immune escape [23]. Immune components in the tumor microenvironment have essential effects on gene expression by tumor tissues and the clinical outcomes [26, 27]. In this study, a total of 332 immune-related genes were identified from TCGA BLCA dataset. According to differential expression analysis, we found 1617 DEGs and 33 immune-related DEGs between BCa samples and normal bladder samples. Negative regulation of immune system process (GO:0002683) is the overlapped GO item based on the results of enrichment analyses of both DEGs and immune-related DEGs, which indicated that the development of BCa was associated with immune system and immune response (Figure 3E).
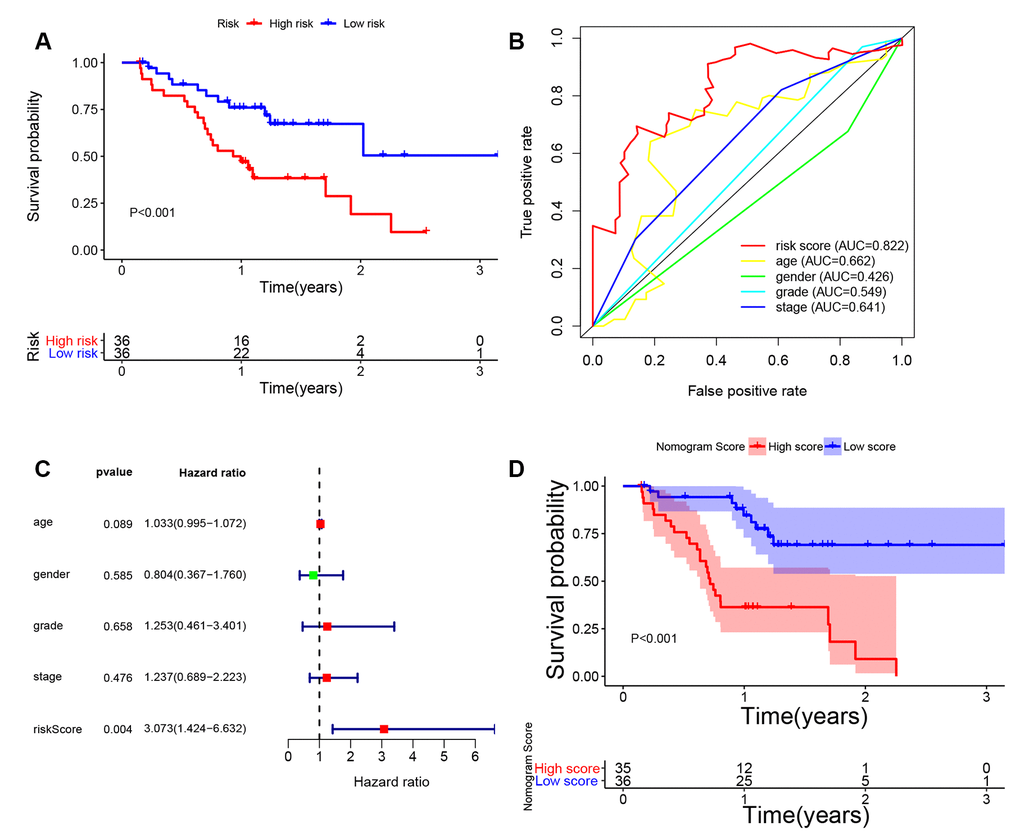
Next, we established an immune-related prognostic signature based on eight lncRNAs targeting immune-related genes (WNT5A-AS1, AL136084.3, MIF-AS1, AC008735.2, AL357033.4, LINC00649, AC099343.2 and USP30-AS1) in the training cohort. BCa patients were divided into low-risk and high-risk groups according to the median risk score of 8-lncRNA signature and we further found the immune-related signature was able to distinguish samples with a better prognosis or a worse prognosis. Low-risk group had better OS than high-risk group (Figures 4D, 5D and 6D). Results of multivariate Cox regression analyses revealed that higher risk score was an independent poor prognostic indicator of OS (HR > 1) (Table 3, Figures 4F, 5F and 6F). ROC curves showed that the AUCs of the immune-related signature were higher than those of UICC stage and histological grade, indicating that the 8-lncRNA signature may be superior to other clinicopathologic features and serve as better prognostic biomarkers (Figures 4E, 5E and 6E). For external validation of the immune-related signature for MIBC, we recruited 72 MIBC patients and validated these results in Tianjin validation cohort. The KM survival curve, time-dependent ROC curves and multivariate Cox regression analysis were consistent with the results of TCGA dataset, which confirmed the predictive power of the prognostic signature (Figure 11).
In order to explore the wide applicability of the 8-lncRNA signature, we performed survival analyses in different subgroups stratified by UICC stage, age and gender. We observed that the 8-lncRNA signature was able to assess the risk score in various subgroups by accurately dividing the patients into high-risk group with poor survival and low-risk group with good survival (Figure 7B–7G), except for patients with stage I and stage II BCa (Figure 7A). One explanation for this phenomenon is that most of the patients with low stage BCa had favourable prognosis with good survival and the heterogeneity of prognosis among them is small [4]. In addition, we also compared the risk scores between different subgroups and we identified that patients with advanced BCa (stage IV and stage III) had a higher risk score than those low stage BCa (stage II and stage I) (Figure 7J). The above results indicated that the 8-lncRNA prognostic signature could help to distinguish prognosis of patients with different variables and may also predict the tumor progression.
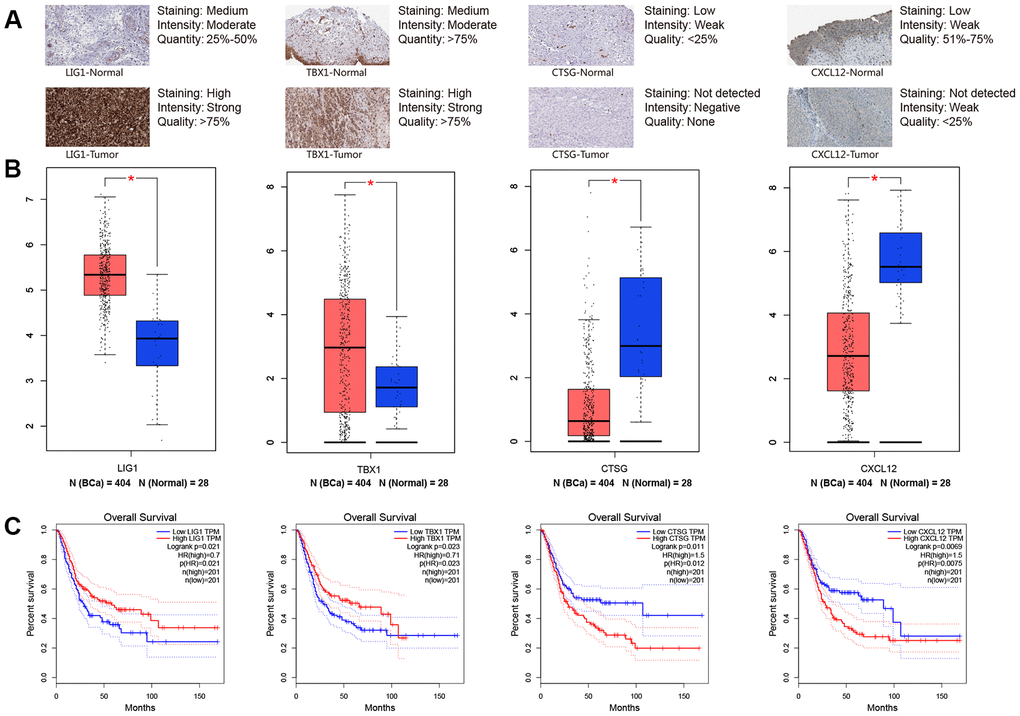
To further validate the 8-lncRNA prognostic signature was tightly associated with immune system, GSEA was carried out between high-risk and low-risk groups. We identified that up-regulation of the immune-related signature was enriched in immune system process pathway (FDR = 0.022, Figure 8D) and immune response pathway (FDR = 0.015, Figure 8E). We also selected four immune-related DEGs including CTSG, CXCL12, LIG1 and TBX1 to explore their functions in BCa tumorigenesis and prognosis. CTSG is reported to activate neutrophil effector functions through release of formyl peptide receptor agonists in inflammatory and immune responses [28, 29]. The CXCL12-CXCR4/CXCR7 chemokine axis activates immune cell migration and inhibits immune resistance in gastrointestinal malignancies [30]. Biallelic et al. found mutations in LIG1 are identified to underlie a spectrum of immune deficiencies by in vitro studies and TBX1 mutation is responsible for most of the congenital immune defect seen in the mouse models and in patients [31, 32]. Immunohistochemistry from THPA and online analyses from GEPIA demonstrated that all the 4 immune-related DEGs differentially expressed between BCa and normal bladder tissues and they were associated with OS of BCa patients (P < 0.05, Figure 9).
In addition, our predictive nomogram combining our immune-related signature suggested that AUCs of 1-year, 3-year and 5-year survival in the entire TCGA BLCA cohort are all greater than 0.700, indicating our nomogram and immune-related signature have wide applicability for both long-term and short-term follow-up patients (Figure 10F–10H). In addition, we used c-index and AUC to compare the predictive value of the signature and nomogram. Both c-index and AUC of nomogram exceeded the immune-related signature, especially in predicting 5-year OS (Tables 4 and 5), which indicated the nomogram had better predictive power in long-term follow-up. Therefore, through combination of the 8-lncRNA signature and other prognosis-related clinical factors, the nomogram further increased the predictive power of survival.
Table 5. Area under curve (AUC) from time-dependent receiver operating characteristic (ROC) curves.
| AUC (95%CI) | Z-test | P-value | ||
| 8-lncRNA Risk score | Nomogram | |||
| Training cohort | ||||
| 1-Year | 0.764 (0.721-0.807) | 0.746 (0.700-0.792) | -0.564 | 0.573 |
| 3-Year | 0.756 (0.720-0.792) | 0.829 (0.793-0.865) | 2.798 | 0.005 |
| 5-Year | 0.742 (0.704-0.780) | 0.825 (0.781-0.869) | 2.777 | 0.006 |
| Testing cohort | ||||
| 1-Year | 0.797 (0.748-0.846) | 0.660 (0.581-0.739) | -2.900 | 0.004 |
| 3-Year | 0.739 (0.678-0.799) | 0.673 (0.611-0.735) | -1.491 | 0.136 |
| 5-Year | 0.724 (0.651-0.797) | 0.801 (0.759-0.843) | 2.302 | 0.021 |
| Entire TCGA BLCA cohort | ||||
| 1-Year | 0.742 (0.710-0.774) | 0.778 (0.748-0.808) | 1.608 | 0.108 |
| 3-Year | 0.769 (0.732-0.806) | 0.755 (0.720-0.790) | -0.537 | 0.591 |
| 5-Year | 0.730 (0.685-0.775) | 0.750 (0.704-0.796) | 0.612 | 0.541 |
In recent years, immunotherapy for BCa attracted more and more attention owing to the availability of targeted immunotherapies and checkpoint inhibitors [33–35]. Among these eight lncRNAs, WNT5A-AS1 and AL136084.3 are risk lncRNAs, while MIF-AS1, AC008735.2, AL357033.4, LINC00649, AC099343.2 and USP30-AS1 are protective lncRNAs. MIF-AS1 acted as a competing endogenous RNA by activating miR-1249-3p/HOXB8 axis in breast cancer [36]. LINC00649 might participate in intracellular receptor signaling pathways in prostate cancer patients [37]. LINC00649 was reported to be involved in vacuolar transport and histone modification functions as well as G protein-coupled receptor and Rho GTPases signaling pathways and is significantly related to the development and prognosis of CRC [38]. Although most of the identified lncRNAs have not been reported to be directly associated with immune-related function, the eight lncRNAs might participate in immune system through indirect pathways for the reason that enrichment results were immune-related. Hence, the eight lncRNAs targeting immune-related genes will have attractive applications to provide therapeutic targets for BCa, which could improve the prognosis of BCa patients.
Nowadays, many studies used high-throughput sequencing data to identify gene signatures and construct clinical predictive models. Gao et al. [39] identified a 6-lncRNA signature to predict the prognosis of BCa and their AUC for 5-year survival is 0.751, which is similar to 0.742 by our signature. However, they didn’t report the AUC value for 1-year survival and thus the predictive power for short-term follow-up is uncertain. In addition, we validated its application in the external validation cohort, which further confirmed the predictive value of our signature. We also constructed a nomogram by enrolling the immune-related signature and other prognosis-related clinical factors to broaden the applicability and increase the clinical significance. In the present study, we validated our findings in other online databases and discussed the relationship between immune system and BCa by immunohistochemistry.
To our knowledge, our study focused on lncRNAs targeting immune-related genes and further construct a signature and a nomogram for BCa. However, several limitations still existed. Firstly, most enrolled patients from TCGA BLCA dataset are MIBC and only 2 patients are NMIBC (stage I). Hence, the 8-lncRNA signature and nomogram might be more suitable for MIBC patients and further cohorts based on NMIBC patients are required to verify its application in NMIBC management in the future. Secondly, although external validation has been performed to confirm the predictive power of the signature and nomogram, the exact molecular mechanisms of eight lncRNAs have not been investigated in the present study. Therefore, further in vitro and in vivo studies based on functional studies are warranted to verify these hypotheses and to make these results more convincible for clinical application in the future.
In summary, we identified an immune-related signature and nomogram based on eight lncRNAs targeting immune-related genes in MIBC. The 8-lncRNA signature and nomogram were confirmed to be independent prognostic indicators for MIBC and could act as a good classifier in different subgroups of MIBC patients. External validation was utilized to verify the predictive value and immunohistochemistry as well as GSEA validated the association between the signature and immune-related functions. These eight lncRNAs will have attractive applications to provide new diagnostic methods and treatment targets for MIBC, which could improve the prognosis of MIBC patients, if validated by further experiments.
Materials and Methods
TCGA BLCA dataset and functional enrichment analysis
TCGA BLCA dataset contained BCa samples (n = 411) and normal bladder samples (n = 19). The RNA-sequencing data and clinical data were downloaded from TCGA (http://tcga-data.nci.nih.gov/tcga/) database.
To identify DEGs between BCa samples and normal bladder samples in TCGA BLCA dataset, we utilized limma R package [40]. The cut-off criteria of adjusted P-value (adj. P-value) was set as 0.05 and the criterion of Fold change was set as |logFC| ≥ 1. We also generated a volcano plot for these DEGs.
In order to reveal the potential functions of DEGs, the clusterProfiler R package is used to perform GO enrichment analysis and KEGG pathway enrichment analysis. In addition, adj. P-value < 0.05 was set as the cutoff criteria.
Predictive power of the lncRNA prognostic signature in training cohort and validation
We performed univariate and multivariate Cox regression analysis to evaluate the predictive power of lncRNA signature and other clinical characteristics (age, gender, UICC stage, and histological grade) in training cohort (n = 195). C-index and time-dependent ROC curve with AUC value were performed to further assess the predictive value of lncRNA signature and these clinical characteristics by survivalROC, timeROC and rms R packages. The training cohort was divided into two low-risk and high-risk groups by the median value of risk score. Then, KM survival curve was plotted to compare the differences of OS in the two groups. P-value < 0.05 was considered as statistically significant difference.
At the same time, stability and reliability of the lncRNA signature was validated in testing cohort (n = 194) and in entire TCGA BLCA cohort (n = 389). They were also divided into two low-risk and high-risk groups by the median value of risk score. Furthermore, univariate and multivariate Cox regression analysis, time-dependent ROC curve, KM survival curve and c-index were analyzed for the validation as mentioned earlier.
Bioinformatics analysis
PCA was carried out to profile expression patterns of grouped samples by using scatterplot3d R package. GSEA (http://www.broadinstitute.org/gsea/index.jsp) was carried out to find different functional phenotypes between the two groups [48]. Gene set permutations were performed 1000 times for each analysis. FDR < 25% and nominal P-value < 0.05 were regarded as the cut-off criteria of sorting GO and KEGG pathway enrichments in GSEA.
Identification and validation of a predictive nomogram
Nomogram [51] was constructed by including age, UICC stage, histological grade, and the immune-related prognostic signature in training cohort. The training cohort was divided into two low-nomogram-score and high-nomogram-score groups by the median value of nomogram score. KM survival curve, time-independent ROC curve and c-index were conducted to evaluate the predictive power of nomogram. We also performed calibration plot to explore the calibration and the discrimination of the nomogram by a bootstrap method with 1000 resamples.
In the meantime, stability and reliability of the nomogram was validated in testing cohort. KM survival curve, ROC curve and c-index were analyzed for the validation in testing cohort (n = 194) and in entire TCGA BLCA cohort (n = 389) as mentioned earlier.
Quantitative real-time polymerase chain reaction and External validation in Tianjin cohort
A total of 72 patients who were pathologically and clinically diagnosed with MIBC were enrolled from Tianjin Medical University General Hospital. MIBC tissues were obtained at the time of first surgery as Tianjin cohort. The study was approved by the Ethics Committee of Tianjin Medical University General Hospital. All recruited participants signed informed consent before being enrolled in our study.
Total RNA from MIBC samples were extracted using RNeasy kit (Qiagen, Valencia, CA). The first chain of cDNA was synthesized by reverse transcription with TaqMan® Reverse Transcription Reagents (Applied Biosystems, Grand Island, NY). GAPDH was used as internal control. The sequences of the primers were displayed in Table 7. qRT-PCR was performed using the CFX96 Touch PCR system (Bio-Rad). The relative lncRNA expression levels of WNT5A-AS1, AL136084.3, MIF-AS1, AC008735.2, AL357033.4, LINC00649, AC099343.2 and USP30-AS1 were calculated by 2-ΔΔCt method. Risk score was calculated with the previous following formula of signature. In addition, univariate and multivariate Cox regression analysis, time-dependent ROC curve and KM survival curve were analyzed for the validation in Tianjin cohort (n = 72) as mentioned earlier.
Table 7. Primer sequences used to amplify target lncRNAs by quantitative real-time polymerase chain reaction (qRT-PCR).
| lncRNA name | Primer sequences | |
| WNT5A-AS1 | Forward | 5'-AAAACGCACAAGTCGCCATC-3' |
| Reverse | 5'-CCGCACAGCAATAAGTTCCG-3' | |
| AL136084.3 | Forward | 5'-GCTGCCTTATGTAACCTGCG-3' |
| Reverse | 5'-AAGAGTGCTTTCTTGCGGGT-3' | |
| MIF-AS1 | Forward | 5'-CACTGTGGTCCCGCCTTTTG-3' |
| Reverse | 5'-CTAGCCGCCAAGTGGAGAAC-3' | |
| AC008735.2 | Forward | 5'-CAAATATGAAACTGCCACAGAGAGG-3' |
| Reverse | 5'-TTACTATTGACTTCTACACCCCCAC-3' | |
| AL357033.4 | Forward | 5'-AATGATGTCTGGTCCGCGTT-3' |
| Reverse | 5'-CTGCAATGTCCTGTTCCCCT-3' | |
| LINC00649 | Forward | 5'-GTTATTGTCAACGCCAGCCC-3' |
| Reverse | 5'-GGTTGTCTCGGACCTCATGG-3' | |
| AC099343.2 | Forward | 5'-TAGACCAGGCGGTGGATAGT-3' |
| Reverse | 5'-GAATCCTGAATCTGCGTGCG-3' | |
| USP30-AS1 | Forward | 5'-ATACGACGGTTCCCGAGACA-3' |
| Reverse | 5'-GACGTGGTCCGTCAGCTATT-3' | |
| GAPDH | Forward | 5'-AGAAGGCTGGGGCTCATTTG-3' |
| Reverse | 5'-AGGGGCCATCCACAGTCTTC-3' |
Statistical analysis
Statistical analyses were carried out using R software (v3.5.3: http://www.r-project.org) and SPSS v23.0. The RNA-sequencing data of gene and lncRNA were log2-transformed. Univariate and multivariate Cox regression analyses, KM method and log-rank test were used to compare the influence of the lncRNA prognostic signature on survival along with other clinical characteristics. Chi-square test and Student’s t test were used to evaluate qualitative variables and quantitative variables, respectively. Time-dependent ROC curve and c-index was utilized to assess the prognostic value based on the lncRNA signature and nomogram. Delong’s Z-test was utilized to compare the AUC and c-index between the signature and nomogram [52]. The two-sided P-value < 0.05 was regarded as statistically significant.
Author Contributions
Research design: SYX, JDH and CJY; Data extraction and statistical analysis: SYX, JDH, LZW and CGY; Drafting of the manuscript and modification: SYX, LXQ and YYJ.
Acknowledgments
All authors would like to thank TCGA BLCA dataset (http://tcga-data.nci.nih.gov/tcga/) for the availability of the data.
Conflicts of Interest
No conflicts of interest existed in the submission.
Funding
Tianjin Natural Science Foundation of China (17JCQNJC11900, 16JCZDJC34600); Zhao Yi-Cheng Medical Science Foundation (ZYYFY2018031).
References
- 1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018; 68:394–424. https://doi.org/10.3322/caac.21492 [PubMed]
- 2. Antoni S, Ferlay J, Soerjomataram I, Znaor A, Jemal A, Bray F. Bladder cancer incidence and mortality: a global overview and recent trends. Eur Urol. 2017; 71:96–108. https://doi.org/10.1016/j.eururo.2016.06.010 [PubMed]
- 3. Yang Y, Cheng Z, Jia X, Shi N, Xia Z, Zhang W, Shi X. Mortality trends of bladder cancer in China from 1991 to 2015: an age-period-cohort analysis. Cancer Manag Res. 2019; 11:3043–51. https://doi.org/10.2147/CMAR.S189220 [PubMed]
- 4. Ebrahimi H, Amini E, Pishgar F, Moghaddam SS, Nabavizadeh B, Rostamabadi Y, Aminorroaya A, Fitzmaurice C, Farzadfar F, Nowroozi MR, Black PC, Daneshmand S. Global, regional and national burden of bladder cancer, 1990 to 2016: results from the GBD study 2016. J Urol. 2019; 201:893–901. https://doi.org/10.1097/JU.0000000000000025 [PubMed]
- 5. Kirkali Z, Chan T, Manoharan M, Algaba F, Busch C, Cheng L, Kiemeney L, Kriegmair M, Montironi R, Murphy WM, Sesterhenn IA, Tachibana M, Weider J. Bladder cancer: epidemiology, staging and grading, and diagnosis. Urology. 2005; 66:4–34. https://doi.org/10.1016/j.urology.2005.07.062 [PubMed]
- 6. Humphrey PA, Moch H, Cubilla AL, Ulbright TM, Reuter VE. The 2016 WHO classification of tumours of the urinary system and male genital organs-part B: prostate and bladder tumours. Eur Urol. 2016; 70:106–19. https://doi.org/10.1016/j.eururo.2016.02.028 [PubMed]
- 7. Goebell PJ, Legal W, Weiss C, Fietkau R, Wullich B, Krause S. [Multimodal therapy for bladder sparing with high grade bladder tumors]. Urologe A. 2008; 47:838. https://doi.org/10.1007/s00120-008-1715-4 [PubMed]
- 8. Lightfoot AJ, Breyer BN, Rosevear HM, Erickson BA, Konety BR, O’Donnell MA. Multi-institutional analysis of sequential intravesical gemcitabine and mitomycin C chemotherapy for non-muscle invasive bladder cancer. Urol Oncol. 2014; 32:35.e15–19. https://doi.org/10.1016/j.urolonc.2013.01.009 [PubMed]
- 9. Choueiri TK, Raghavan D. Chemotherapy for muscle-invasive bladder cancer treated with definitive radiotherapy: persisting uncertainties. Nat Clin Pract Oncol. 2008; 5:444–54. https://doi.org/10.1038/ncponc1159 [PubMed]
- 10. Yu WD, Wang H, He QF, Xu Y, Wang XC. Long noncoding RNAs in cancer-immunity cycle. J Cell Physiol. 2018; 233:6518–23. https://doi.org/10.1002/jcp.26568 [PubMed]
- 11. Yoshihara K, Shahmoradgoli M, Martínez E, Vegesna R, Kim H, Torres-Garcia W, Treviño V, Shen H, Laird PW, Levine DA, Carter SL, Getz G, Stemke-Hale K, et al. Inferring tumour purity and stromal and immune cell admixture from expression data. Nat Commun. 2013; 4:2612. https://doi.org/10.1038/ncomms3612 [PubMed]
- 12. Pagès F, Berger A, Camus M, Sanchez-Cabo F, Costes A, Molidor R, Mlecnik B, Kirilovsky A, Nilsson M, Damotte D, Meatchi T, Bruneval P, Cugnenc PH, et al. Effector memory T cells, early metastasis, and survival in colorectal cancer. N Engl J Med. 2005; 353:2654–66. https://doi.org/10.1056/NEJMoa051424 [PubMed]
- 13. Sideras K, Braat H, Kwekkeboom J, van Eijck CH, Peppelenbosch MP, Sleijfer S, Bruno M. Role of the immune system in pancreatic cancer progression and immune modulating treatment strategies. Cancer Treat Rev. 2014; 40:513–22. https://doi.org/10.1016/j.ctrv.2013.11.005 [PubMed]
- 14. Martinez-Bosch N, Vinaixa J, Navarro P. Immune evasion in pancreatic cancer: from mechanisms to therapy. Cancers (Basel). 2018; 10:6. https://doi.org/10.3390/cancers10010006 [PubMed]
- 15. Kim R, Emi M, Tanabe K. Cancer immunoediting from immune surveillance to immune escape. Immunology. 2007; 121:1–14. https://doi.org/10.1111/j.1365-2567.2007.02587.x [PubMed]
- 16. Cheng W, Ren X, Zhang C, Cai J, Liu Y, Han S, Wu A. Bioinformatic profiling identifies an immune-related risk signature for glioblastoma. Neurology. 2016; 86:2226–34. https://doi.org/10.1212/WNL.0000000000002770 [PubMed]
- 17. Joseph M, Enting D. Immune responses in bladder cancer-role of immune cell populations, prognostic factors and therapeutic implications. Front Oncol. 2019; 9:1270. https://doi.org/10.3389/fonc.2019.01270 [PubMed]
- 18. Ulitsky I, Bartel DP. lincRNAs: genomics, evolution, and mechanisms. Cell. 2013; 154:26–46. https://doi.org/10.1016/j.cell.2013.06.020 [PubMed]
- 19. Fatica A, Bozzoni I. Long non-coding RNAs: new players in cell differentiation and development. Nat Rev Genet. 2014; 15:7–21. https://doi.org/10.1038/nrg3606 [PubMed]
- 20. Yao K, Wang Q, Jia J, Zhao H. A competing endogenous RNA network identifies novel mRNA, miRNA and lncRNA markers for the prognosis of diabetic pancreatic cancer. Tumour Biol. 2017; 39:1010428317707882. https://doi.org/10.1177/1010428317707882 [PubMed]
- 21. Wu K, Zhao Z, Liu K, Zhang J, Li G, Wang L. Long noncoding RNA lnc-sox5 modulates CRC tumorigenesis by unbalancing tumor microenvironment. Cell Cycle. 2017; 16:1295–301. https://doi.org/10.1080/15384101.2017.1317416 [PubMed]
- 22. Shen H, Wong LM, Li WT, Chu M, High RA, Chang EY, Wang-Rodriguez J, Ongkeko WM. The landscape of long non-coding RNA dysregulation and clinical relevance in muscle invasive bladder urothelial carcinoma. Cancers (Basel). 2019; 11:1919. https://doi.org/10.3390/cancers11121919 [PubMed]
- 23. Cao J, Yang X, Li J, Wu H, Li P, Yao Z, Dong Z, Tian J. Screening and identifying immune-related cells and genes in the tumor microenvironment of bladder urothelial carcinoma: based on TCGA database and bioinformatics. Front Oncol. 2020; 9:1533. https://doi.org/10.3389/fonc.2019.01533 [PubMed]
- 24. Wei C, Liang Q, Li X, Li H, Liu Y, Huang X, Chen X, Guo Y, Li J. Bioinformatics profiling utilized a nine immune-related long noncoding RNA signature as a prognostic target for pancreatic cancer. J Cell Biochem. 2019; 120:14916–27. https://doi.org/10.1002/jcb.28754 [PubMed]
- 25. Wang W, Zhao Z, Yang F, Wang H, Wu F, Liang T, Yan X, Li J, Lan Q, Wang J, Zhao J. An immune-related lncRNA signature for patients with anaplastic gliomas. J Neurooncol. 2018; 136:263–71. https://doi.org/10.1007/s11060-017-2667-6 [PubMed]
- 26. Şenbabaoğlu Y, Gejman RS, Winer AG, Liu M, Van Allen EM, de Velasco G, Miao D, Ostrovnaya I, Drill E, Luna A, Weinhold N, Lee W, Manley BJ, et al. Tumor immune microenvironment characterization in clear cell renal cell carcinoma identifies prognostic and immunotherapeutically relevant messenger RNA signatures. Genome Biol. 2016; 17:231. https://doi.org/10.1186/s13059-016-1092-z [PubMed]
- 27. Cooper LA, Gutman DA, Chisolm C, Appin C, Kong J, Rong Y, Kurc T, Van Meir EG, Saltz JH, Moreno CS, Brat DJ. The tumor microenvironment strongly impacts master transcriptional regulators and gene expression class of glioblastoma. Am J Pathol. 2012; 180:2108–19. https://doi.org/10.1016/j.ajpath.2012.01.040 [PubMed]
- 28. Woloszynek JC, Hu Y, Pham CT. Cathepsin g-regulated release of formyl peptide receptor agonists modulate neutrophil effector functions. J Biol Chem. 2012; 287:34101–09. https://doi.org/10.1074/jbc.M112.394452 [PubMed]
- 29. Sun R, Iribarren P, Zhang N, Zhou Y, Gong W, Cho EH, Lockett S, Chertov O, Bednar F, Rogers TJ, Oppenheim JJ, Wang JM. Identification of neutrophil granule protein cathepsin G as a novel chemotactic agonist for the G protein-coupled formyl peptide receptor. J Immunol. 2004; 173:428–36. https://doi.org/10.4049/jimmunol.173.1.428 [PubMed]
- 30. Daniel SK, Seo YD, Pillarisetty VG. The CXCL12-CXCR4/CXCR7 axis as a mechanism of immune resistance in gastrointestinal malignancies. Semin Cancer Biol. 2019. [Epub ahead of print]. https://doi.org/10.1016/j.semcancer.2019.12.007 [PubMed]
- 31. Paylor R, Lindsay E. Mouse models of 22q11 deletion syndrome. Biol Psychiatry. 2006; 59:1172–79. https://doi.org/10.1016/j.biopsych.2006.01.018 [PubMed]
- 32. Maffucci P, Chavez J, Jurkiw TJ, O’Brien PJ, Abbott JK, Reynolds PR, Worth A, Notarangelo LD, Felgentreff K, Cortes P, Boisson B, Radigan L, Cobat A, et al. Biallelic mutations in DNA ligase 1 underlie a spectrum of immune deficiencies. J Clin Invest. 2018; 128:5489–504. https://doi.org/10.1172/JCI99629 [PubMed]
- 33. Pettenati C, Ingersoll MA. Mechanisms of BCG immunotherapy and its outlook for bladder cancer. Nat Rev Urol. 2018; 15:615–25. https://doi.org/10.1038/s41585-018-0055-4 [PubMed]
- 34. Kamat AM, Bellmunt J, Galsky MD, Konety BR, Lamm DL, Langham D, Lee CT, Milowsky MI, O’Donnell MA, O'Donnell PH, Petrylak DP, Sharma P, Skinner EC, et al. Society for immunotherapy of cancer consensus statement on immunotherapy for the treatment of bladder carcinoma. J Immunother Cancer. 2017; 5:68. https://doi.org/10.1186/s40425-017-0271-0 [PubMed]
- 35. Boegemann M, Aydin AM, Bagrodia A, Krabbe LM. Prospects and progress of immunotherapy for bladder cancer. Expert Opin Biol Ther. 2017; 17:1417–31. https://doi.org/10.1080/14712598.2017.1366445 [PubMed]
- 36. Ding J, Wu W, Yang J, Wu M. Long non-coding RNA MIF-AS1 promotes breast cancer cell proliferation, migration and EMT process through regulating miR-1249-3p/HOXB8 axis. Pathol Res Pract. 2019; 215:152376. https://doi.org/10.1016/j.prp.2019.03.005 [PubMed]
- 37. Ye G, Guo L, Xing Y, Sun W, Yuan M. Identification of prognostic biomarkers of prostate cancer with long non-coding RNA-mediated competitive endogenous RNA network. Exp Ther Med. 2019; 17:3035–40. https://doi.org/10.3892/etm.2019.7277 [PubMed]
- 38. He M, Lin Y, Xu Y. Identification of prognostic biomarkers in colorectal cancer using a long non-coding RNA-mediated competitive endogenous RNA network. Oncol Lett. 2019; 17:2687–94. https://doi.org/10.3892/ol.2019.9936 [PubMed]
- 39. Gao X, Zhang S, Chen Y, Wen X, Chen M, Wang S, Zhang Y. Development of a novel six-long noncoding RNA signature predicting survival of patients with bladder urothelial carcinoma. J Cell Biochem. 2019; 120:19796–809. https://doi.org/10.1002/jcb.29285 [PubMed]
- 40. Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W, Smyth GK. Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015; 43:e47. https://doi.org/10.1093/nar/gkv007 [PubMed]
- 41. Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL, Golub TR, Lander ES, Mesirov JP. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA. 2005; 102:15545–50. https://doi.org/10.1073/pnas.0506580102 [PubMed]
- 42. Akaike, H. (1973). Information theory and an extension of the maximum likelihood principle, Second International Symposium on Information Theory, pp. 267–281.
- 43. Zhang CB, Zhu P, Yang P, Cai JQ, Wang ZL, Li QB, Bao ZS, Zhang W, Jiang T. Identification of high risk anaplastic gliomas by a diagnostic and prognostic signature derived from mRNA expression profiling. Oncotarget. 2015; 6:36643–51. https://doi.org/10.18632/oncotarget.5421 [PubMed]
- 44. Cai J, Zhang W, Yang P, Wang Y, Li M, Zhang C, Wang Z, Hu H, Liu Y, Li Q, Wen J, Sun B, Wang X, et al. Identification of a 6-cytokine prognostic signature in patients with primary glioblastoma harboring M2 microglia/macrophage phenotype relevance. PLoS One. 2015; 10:e0126022. https://doi.org/10.1371/journal.pone.0126022 [PubMed]
- 45. Bao ZS, Li MY, Wang JY, Zhang CB, Wang HJ, Yan W, Liu YW, Zhang W, Chen L, Jiang T. Prognostic value of a nine-gene signature in glioma patients based on mRNA expression profiling. CNS Neurosci Ther. 2014; 20:112–18. https://doi.org/10.1111/cns.12171 [PubMed]
- 46. Zhang W, Zhang J, Yan W, You G, Bao Z, Li S, Kang C, Jiang C, You Y, Zhang Y, Chen CC, Song SW, Jiang T. Whole-genome microRNA expression profiling identifies a 5-microRNA signature as a prognostic biomarker in chinese patients with primary glioblastoma multiforme. Cancer. 2013; 119:814–24. https://doi.org/10.1002/cncr.27826 [PubMed]
- 47. Lossos IS, Czerwinski DK, Alizadeh AA, Wechser MA, Tibshirani R, Botstein D, Levy R. Prediction of survival in diffuse large-B-cell lymphoma based on the expression of six genes. N Engl J Med. 2004; 350:1828–37. https://doi.org/10.1056/NEJMoa032520 [PubMed]
- 48. Subramanian A, Kuehn H, Gould J, Tamayo P, Mesirov JP. GSEA-P: a desktop application for gene set enrichment analysis. Bioinformatics. 2007; 23:3251–53. https://doi.org/10.1093/bioinformatics/btm369 [PubMed]
- 49. Uhlen M, Oksvold P, Fagerberg L, Lundberg E, Jonasson K, Forsberg M, Zwahlen M, Kampf C, Wester K, Hober S, Wernerus H, Björling L, Ponten F. Towards a knowledge-based human protein atlas. Nat Biotechnol. 2010; 28:1248–50. https://doi.org/10.1038/nbt1210-1248 [PubMed]
- 50. Tang Z, Li C, Zhang K, Yang M, Hu X. GE-mini: a mobile APP for large-scale gene expression visualization. Bioinformatics. 2017; 33:941–43. https://doi.org/10.1093/bioinformatics/btw775 [PubMed]
- 51. Iasonos A, Schrag D, Raj GV, Panageas KS. How to build and interpret a nomogram for cancer prognosis. J Clin Oncol. 2008; 26:1364–70. https://doi.org/10.1200/JCO.2007.12.9791 [PubMed]
- 52. DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988; 44:837–45. [PubMed]