Introduction
With a rapidly increasing prevalence worldwide, abdominal obesity has become a significant public health problem [1–5]. Compared to subcutaneous fat, abdominal fat is more pro-inflammatory; it was shown for instance that the synthesis of components of the complement system, a key mediator of inflammatory responses and innate immunity, is exacerbated in abdominal fat depots of obese individuals [6, 7]. In turn, excessive accumulation of abdominal fat is linked to the development of metabolic syndrome and a range of chronic health conditions including diabetes, cardiovascular disease, and cancer. Not surprisingly, studies have shown that obesity is associated with reduced life expectancy [8, 9]. As a hallmark of human aging, the accrual of abdominal fat is likely to have, also in non-obese people, a significant impact on longevity. Periumbilical fat is measured by noninvasive ultrasound and represents a useful tool in determining abdominal obesity. Whereas limited data on the association between periumbilical fat and antibody and complement systems are currently available from studies carried out in the United States and European countries, studies addressing these aspects in Chinese oldest-old population are still lacking [10, 11]. Asians are predisposed to have a higher ratio of abdominal obesity to overall obesity than Westerners, suggesting that the adverse impact of abdominal obesity on longevity may be more pronounced in the Asian population [12, 13]. So far, no studies have analyzed the potential association between periumbilical fat and longevity in relation to antibody and complement systems.
Cardiac diseases are the leading cause of premature mortality worldwide [14], with abdominal obesity being a fundamental cause of abnormal cardiac structure and function [15]. Indeed, a 2010 survey conducted by China’s Ministry of Health suggested that abdominal obesity contributes to increased mortality rate and reduced life expectancy by impairing cardiac structure and function [16]. However, since racial differences in body build, fat distribution, and genetic background may determine distinct effects of periumbilical fat on cardiac structure and function, aging and longevity research conducted in Western and Asian populations may conceivably yield different results [13].
Centenarians have been suggested to have delayed or escaped onset and interaction of age-related abnormalities, and are thus considered a prototype of successful aging [17, 18]. Therefore, studies analyzing associations between genetic, physical, and metabolic factors influencing centenarians will provide valuable information to formulate strategies promoting successful aging and longevity [10, 19]. To our knowledge, few studies have specifically discussed these associations in Chinese oldest-old population, including centenarians. The China Hainan Centenarian Cohort Study (CHCCS) was launched to establish a multidimensional database of longevity-related information. Based on demographic data from Hainan, the area with the highest population density of centenarians in China, the CHCCS constitutes a significant population-based sample of Chinese centenarians. Based on CHCCS data, the current study was designed to test the hypothetical association between periumbilical fat and longevity mediated by the antibody-complement systems and cardiac structure and function in a Chinese oldest-old population (Figure 1A).
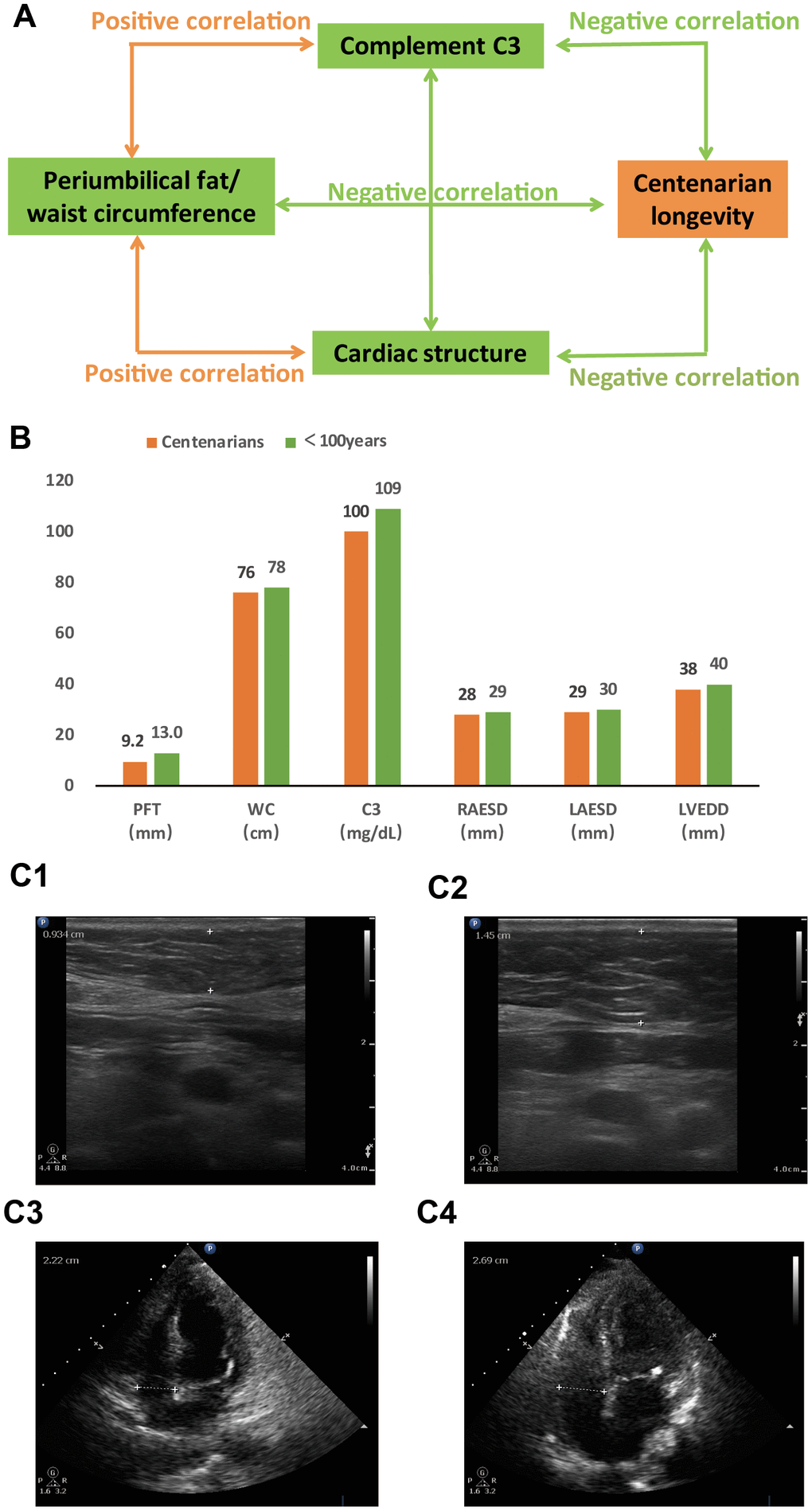
Figure 1. Comparisons between centenarians and non-centenarians in Chinese oldest-old population. (A) Association network between periumbilical fat and longevity mediated by antibody and complement systems and cardiac structure and function (P<0.05 for all variables). (B) Graphic summary of periumbilical fat, complement C3, and cardiac structure in centenarians and non-centenarian oldest-old population (P<0.05 for all comparisons). (C) Representative ultrasound images of periumbilical fat (top) and cardiac structure (bottom) from a centenarian (C1 and C3) and a non-centenarian oldest-old individual (C2 and C4). Abbreviations: PFT: periumbilical fat thickness; WC: waist circumference; C3: complement C3; RAESD: right atrium end-systolic diameter; LAESD: left atrium end-systolic diameter; LVEDD: left ventricular end-diastolic diameter.
Results
Comparisons between centenarian and non-centenarian oldest-old populations
The median age of all participants was 92 years (84 years for non-centenarians, 102 years for centenarians; range: 80-116 years of age). The characteristics of the study’s 419 centenarians and 491 non-centenarians are shown in Table 1. Centenarians had lower periumbilical fat thickness (PFT), waist circumference (WC), diastolic blood pressure (DBP), total cholesterol (TC) level, low-density lipoprotein cholesterol (LDL-C) level, complement C3 level, complement C4 level, immunoglobulin M (IgM) level, right atrium end-systolic diameter (RAESD), pulmonary artery diameter (PAD), left atrium end-systolic diameter (LAESD), left ventricular end-diastolic diameter (LVEDD), interventricular septum thickness (IVST), and aorta diameter (AOD) than non-centenarians (Figure 1B, 1C; P<0.05 for all comparisons). Centenarians had higher SBP, fasting blood glucose (FBG) level, immunoglobulin G (IgG) level, immunoglobulin E (IgE) level, immunoglobulin kappa level, and right ventricle end-diastolic diameter (RVEDD) than non-centenarians (P<0.05 for all comparisons). The proportion of males was lower in the centenarians than non-centenarians (P<0.05). There was no difference in TG level, HDL-C level, immunoglobulin A (IgA) level, immunoglobulin lambda level, and left ventricular ejection fraction (LVEF) between the two groups (P>0.05 for all comparisons).
Table 1. Characteristics of all oldest old participants.
| Characteristic | Total (n=910) | Centenarians (n=419) | <100 years (n=491) | P | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years)* | 92(84, 102) | 102(101, 104) | 84(82, 88) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Males (%) | 286(31.4) | 90(21.5) | 196(39.9) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Periumbilical fat thickness (cm)* | 1.15(0.76, 1.60) | 0.92(0.60, 1.40) | 1.30(0.90, 1.70) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Waist circumference (cm)* | 76(71, 83) | 76(70, 81) | 78(72, 86) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Systolic blood pressure (mmHg)* | 149(132, 169) | 151(136, 173) | 146(130, 165) | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diastolic blood pressure (mmHg)* | 77(69, 86) | 76(67, 83) | 79(71, 89) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fasting blood glucose (mmol/L)* | 4.59(3.97, 5.48) | 4.89(4.25, 5.72) | 4.31(3.74, 5.20) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Triglyceride (mmol/L)* | 1.07(0.81, 1.48) | 1.06(0.81, 1.44) | 1.07(0.81, 1.51) | 0.584 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total cholesterol (mmol/L)* | 4.75(4.21, 5.51) | 4.59(4.10, 5.29) | 4.92(4.34, 5.74) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Low-density lipoprotein cholesterol (mmol/L)* | 2.83(2.36, 3.46) | 2.76(2.32, 3.29) | 2.94(2.43, 3.60) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| High-density lipoprotein cholesterol (mmol/L)* | 1.39(1.15, 1.69) | 1.41(1.20, 1.67) | 1.37(1.13, 1.70) | 0.186 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Complement C3 (mg/dL)* | 105(91, 120) | 100(88, 112) | 109(95, 127) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Complement C4 (mg/dL)* | 25(20, 31) | 23(18, 28) | 27(21, 33) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin A (g/dL)* | 0.330(0.253, 0.418) | 0.336(0.253, 0.426) | 0.326(0.252, 0.410) | 0.580 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin G (g/dL)* | 1.540(1.350, 1.760) | 1.570(1.360, 1.820) | 1.500(1.330, 1.690) | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin M (g/dL)* | 0.106(0.075, 0.142) | 0.100(0.072, 0.140) | 0.109(0.081, 0.146) | 0.012 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin E (IU/dL)* | 0.272(0.091, 0.796) | 0.364(0.110, 0.932) | 0.222(0.082, 0.644) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin kappa (g/dL)* | 0.400(0.339, 0.462) | 0.418(0.359, 0.484) | 0.379(0.325, 0.443) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin lambda (g/dL)* | 0.205(0.178, 0.240) | 0.206(0.178, 0.249) | 0.205(0.178, 0.236) | 0.242 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Right atrium end-systolic diameter (mm)* | 29(27, 32) | 28(26, 31) | 29(27, 32) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Right ventricle end-diastolic diameter (mm)* | 28(26, 31) | 28(26, 31) | 28(26, 30) | 0.026 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pulmonary artery diameter (mm) * | 20(18, 21) | 19(18, 21) | 20(18, 21) | 0.004 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Left atrium end-systolic diameter (mm)* | 30(27, 32) | 29(26, 32) | 30(27, 33) | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Left ventricular end-diastolic diameter (mm)* | 39(36, 42) | 38(35, 41) | 40(37, 43) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Interventricular septum thickness (mm)* | 10(9, 10) | 9(8, 10) | 10(9, 11) | 0.003 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Aorta diameter (mm)* | 30(28, 32) | 30(27, 32) | 30(28, 33) | 0.013 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Left ventricular ejection fraction (%)* | 60(58, 61) | 60(58, 60) | 60(58, 62) | 0.062 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| *Median (interquartile range). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Associations of abdominal fat and obesity, antibody and complement systems, and cardiac structure and function with centenarians
As shown in Table 2, PFT, WC, complement C3 level, complement C4 level, IgM level, RAESD, LAESD, and LVEDD were all inversely associated with centenarians (P<0.05 for all variables). A positive association with centenarians was instead detected for IgG level, immunoglobulin kappa level, and RVEDD (P<0.05 for all variables). In turn, no association with centenarians was found for IgA level, IgE level, immunoglobulin lambda level, PAD, IVST, AOD, and LVEF (P>0.05 for all variables).
Table 2. Characteristics associated with centenarians in logistic regression analysis.
| Characteristic | OR(95% CI) | P | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Periumbilical fat thickness (cm) | 0.430(0.330-0.560) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Waist circumference (cm) | 0.965(0.948, 0.982) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Complement C3 (mg/dL) | 0.982(0.974, 0.989) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Complement C4 (mg/dL) | 0.962(0.945, 0.980) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin A (mg/dL) | 1.000(0.999, 1.001) | 0.467 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin G (mg/dL) | 1.001(1.000, 1.001) | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin M (mg/dL) | 0.997(0.995, 1.000) | 0.032 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin E (IU/dL) | 1.000(1.000, 1.000) | 0.129 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin kappa (mg/dL) | 1.004(1.003, 1.006) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin lambda (mg/dL) | 1.002(1.000, 1.005) | 0.082 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Right atrium end-systolic diameter (mm) | 0.941(0.906, 0.977) | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Right ventricle end-diastolic diameter (mm) | 1.058(1.015, 1.102) | 0.008 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pulmonary artery diameter (mm) | 0.957(0.902, 1.015) | 0.140 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Left atrium end-systolic diameter (mm) | 0.960(0.930, 0.990) | 0.010 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Left ventricular end-diastolic diameter (mm) | 0.893(0.864, 0.923) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Interventricular septum thickness (mm) | 0.908(0.821, 1.005) | 0.061 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Aorta diameter (mm) | 0.973(0.937, 1.011) | 0.160 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Left ventricular ejection fraction (%) | 0.980(0.942, 1.019) | 0.314 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: OR: odds ratio; 95%CI: 95% confidence interval. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Notes: With adjustment of age, sex, systolic blood pressure, diastolic blood pressure, fasting blood glucose, total cholesterol, triglyceride, high-density lipoprotein cholesterol, and low-density lipoprotein cholesterol. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Associations of abdominal fat and obesity with antibody and complement systems and cardiac structure and function
As shown in Table 3, complement C3 level, RVEDD, PAD, LAESD, LVEDD, IVST, and AOD were all positively associated with both PFT and WC (P<0.05 for all variables). IgM and RAESD were inversely and positively associated, respectively, with WC (P<0.05) but not PFT (P>0.05). In contrast, no association with either PFT or WC was detected for complement C4 level, IgA level, IgG level, IgE level, immunoglobulin kappa level, immunoglobulin lambda level, and LVEF (P>0.05 for all variables).
Table 3. Associations of abdominal fat and obesity with antibody and complement systems and cardiac structure and function in linear regression analyses.
| Characteristic | Periumbilical fat thickness | Waist circumference | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standardized coefficient | P | Standardized coefficient | P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Complement C3 (mg/dL) | 0.067 | 0.041 | 0.151 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Complement C4 (mg/dL) | 0.022 | 0.512 | 0.048 | 0.153 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin A (mg/dL) | -0.047 | 0.187 | -0.029 | 0.424 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin G (mg/dL) | -0.052 | 0.146 | -0.023 | 0.511 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin M (mg/dL) | -0.062 | 0.084 | -0.074 | 0.037 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin E (IU/dL) | -0.021 | 0.550 | -0.026 | 0.455 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin kappa (mg/dL) | -0.056 | 0.106 | -0.036 | 0.302 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin lambda (mg/dL) | -0.032 | 0.368 | 0.031 | 0.391 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Right atrium end-systolic diameter (mm) | 0.059 | 0.089 | 0.082 | 0.017 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Right ventricle end-diastolic diameter (mm) | 0.156 | <0.001 | 0.137 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pulmonary artery diameter (mm) | 0.140 | <0.001 | 0.096 | 0.007 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Left atrium end-systolic diameter (mm) | 0.181 | <0.001 | 0.271 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Left ventricular end-diastolic diameter (mm) | 0.116 | <0.001 | 0.164 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Interventricular septum thickness (mm) | 0.186 | <0.001 | 0.140 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Aorta diameter (mm) | 0.145 | <0.001 | 0.138 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Left ventricular ejection fraction (%) | 0.056 | 0.120 | -0.019 | 0.592 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Notes: With adjustment of age, sex, systolic blood pressure, diastolic blood pressure, fasting blood glucose, total cholesterol, triglyceride, high-density lipoprotein cholesterol, and low-density lipoprotein cholesterol. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Associations between antibody and complement systems and cardiac structure and function
As shown in Table 4, complement C3 level was inversely associated with RAESD, PAD, and IVST (P<0.05 for all variables). Complement C4 level was inversely associated with PAD (P<0.05). Immunoglobulin kappa level was positively associated with RAESD, LAESD, and LVEF (P<0.05 for all variables). Immunoglobulin lambda level was positively associated with RAESD and LAESD (P<0.05 for both variables). No association with cardiac structure and function was found for IgA, IgG, IgM, and IgE levels (P>0.05 for all variables).
Table 4. Associations of antibody and complement systems with cardiac structure and function in linear regression analyses.
| Characteristic | Right atrium end-systolic diameter (mm) | Right ventricle end-diastolic diameter (mm) | Pulmonary artery diameter (mm) | Left atrium end-systolic diameter (mm) | Left ventricular end-diastolic diameter (mm) | Interventricular septum thickness (mm) | Aorta diameter (mm) | Left ventricular ejection fraction (%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standardized coefficient | P | Standardized coefficient | P | Standardized coefficient | P | Standardized coefficient | P | Standardized coefficient | P | Standardized coefficient | P | Standardized coefficient | P | Standardized coefficients | P value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Complement C3 (mg/dL) | -0.087 | 0.016 | -0.032 | 0.376 | -0.081 | 0.028 | -0.059 | 0.098 | -0.032 | 0.353 | -0.073 | 0.045 | -0.038 | 0.305 | -0.065 | 0.086 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Complement C4 (mg/dL) | -0.040 | 0.237 | 0.000 | 0.981 | -0.077 | 0.028 | -0.052 | 0.119 | -0.059 | 0.069 | -0.032 | 0.361 | -0.061 | 0.078 | -0.006 | 0.859 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin A (mg/dL) | 0.055 | 0.090 | -0.008 | 0.794 | 0.013 | 0.687 | 0.045 | 0.155 | 0.000 | 0.996 | -0.042 | 0.196 | 0.016 | 0.638 | -0.057 | 0.091 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin G (mg/dL) | 0.020 | 0.548 | 0.023 | 0.487 | -0.012 | 0.729 | 0.040 | 0.215 | -0.022 | 0.472 | 0.003 | 0.924 | 0.005 | 0.877 | -0.056 | 0.101 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin M (mg/dL) | 0.025 | 0.422 | 0.026 | 0.431 | 0.003 | 0.929 | 0.052 | 0.100 | 0.020 | 0.519 | 0.000 | 0.997 | -0.019 | 0.570 | -0.030 | 0.377 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin E (IU/dL) | 0.005 | 0.879 | 0.012 | 0.715 | -0.008 | 0.797 | 0.038 | 0.225 | 0.031 | 0.306 | -0.007 | 0.839 | 0.006 | 0.856 | 0.021 | 0.527 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin kappa (mg/dL) | 0.077 | 0.020 | 0.060 | 0.072 | 0.005 | 0.872 | 0.094 | 0.004 | -0.005 | 0.875 | 0.038 | 0.261 | 0.026 | 0.451 | -0.110 | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunoglobulin lambda (mg/dL) | 0.065 | 0.043 | 0.027 | 0.412 | -0.029 | 0.376 | 0.075 | 0.018 | -0.001 | 0.961 | 0.008 | 0.819 | -0.034 | 0.297 | -0.050 | 0.133 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Notes: With adjustment of age, sex, waist circumference, systolic blood pressure, diastolic blood pressure, fasting blood glucose, total cholesterol, triglyceride, high-density lipoprotein cholesterol, and low-density lipoprotein cholesterol. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Discussion
We have witnessed over the last decades an alarming increase in abdominal obesity in virtually every country, irrespective of economic status and cultural background [1–4]. Our previous study indicated that abdominal obesity is associated with both arterial stiffness and impaired hemodynamics in Chinese adults [20]. Since abdominal obesity triggers chronic inflammation of adipose tissue and compromises immune function, we speculated that periumbilical fat may adversely influence longevity by altering the expression or the activity of the antibody-complement system. Although a few studies indicated an association between periumbilical fat and changes in antibody and complement profiles in Western populations [10, 11], it has not yet been determined whether such association impacts longevity, in either Western or Asian populations. The current study demonstrated that Chinese centenarians had less periumbilical fat and a weaker complement system compared with non-centenarian oldest-old population. Moreover, periumbilical fat was negatively associated with longevity mediated by complement C3 in the study’s total oldest-old population. These data indicate that abdominal obesity is an obstacle to successful aging and longevity in China.
Excess periumbilical fat can trigger innate immunity by producing cytokines that stimulate hepatic production of complement C3, an acute phase protein [21, 22]. Complement C3 is also synthesized by activated adipocytes and macrophages and functions as both a cytokine and an adipokine [23]. Complement C3 and other adipokines may promote insulin resistance (IR) by increasing phosphorylation and proteosomal degradation of insulin receptor substrates or by disturbing insulin receptor-substrate interaction [24]. As the main degradation product and active fragment of C3, acylation stimulating protein (ASP, C3a desArg) has insulin-like properties and favors lipid synthesis in adipocytes [25]. In the same way that IR determines increased insulin secretion, an increase in ASP precursor (C3) levels may be triggered by ASP resistance [26]. Since ASP is a potent stimulator of lipogenesis in adipocytes, increased complement C3 levels have the potential to disturb lipid metabolism and aggravate abdominal obesity [27]. Therefore, periumbilical fat may be partly responsible for increased mortality rate and reduced life expectancy mediated by complement C3 [28].
Substantial evidence indicates that metabolically abnormal obesity is associated with increased cardiovascular morbidity and mortality rates. Specifically, it has been proposed that abdominal obesity rather than overall obesity is linked to abnormal cardiac structure and function [29, 30]. Accordingly, a number of studies have confirmed the deleterious impact of obesity on life expectancy and longevity [9]. Still, scarce studies have been dedicated to analyzing the association between periumbilical fat and cardiac structure, and it remains unclear whether cardiac structure could mediate the association of periumbilical fat and longevity in either Western or Chinese oldest-old populations [13]. The current study showed that Chinese centenarians had less periumbilical fat and smaller cardiac structure compared with non-centenarian oldest-old population. In turn, periumbilical fat was negatively associated with longevity mediated by cardiac structure in the study’s total oldest-old population. There are several reasons why periumbilical fat might affect cardiac structure and reduce life expectancy. They include cardiac effects related to IR, which can be partly driven by periumbilical fat via increased fatty acid secretion, decreased adiponectin secretion, and promoted hyperinsulinemia development [31]. In turn, cardiac effects of hyperinsulinemia, including myocardial hypertrophy, can result among other factors from disturbed electrolyte balance, oxidative stress, enhanced sympathetic tone, and dysfunction of the renin-angiotensin-aldosterone system [32]. Excess periumbilical fat may also contribute to the development of an abnormal cardiac structure by releasing proinflammatory cytokines. These effects may be further compounded by genetic and epigenetic associations between periumbilical fat and cardiac structure [33].
The effects of immune activation on cardiac structure are time-dependent. After an acute injury event, like myocardial infarction, the innate immune system is activated as a prerequisite for adequate healing [34]. However, long-term innate immune activation is detrimental, resulting in adverse cardiac remodeling and structural and functional compromise of the organ [35]. C3 seems to be not only a good indicator of overall complement activation but may also be of pathophysiological relevance in the cardiovascular system. Experimental evidence from animal studies suggests that complement activation is mechanistically involved in adverse cardiac healing and remodeling [36]. For instance, it was shown that elevated C3 levels lead to abnormal left ventricular structure and contractile failure in isolated guinea pig hearts [37]. C3 is an effector protein that can efficiently attract inflammatory cells and directly destroy cells by the complement membrane attack complex. To our knowledge, this is the first study detecting an inverse association between complement C3 and cardiac structure in Chinese oldest-old population, including centenarians.
Conclusions
Based on CHCCS data, the current study demonstrated a complex association network between periumbilical fat and longevity mediated by complement C3 and cardiac structure in Chinese oldest-old population, including centenarians. By highlighting the associations of both complement C3 and cardiac structure with centenarian status, our study suggests that preventing abdominal obesity represents a key strategy to promote successful aging. Nevertheless, molecular studies are required to explore the specific mechanisms underlying the associations identified in the current study.
Materials and Methods
Study population
The population-based CHCCS was carried out in 18 cities and counties of Hainan Province, China. It started in July 2014 and ended in December 2016, and its cohort profile has been described previously [38]. There were 910 oldest-old participants (at least 80 years of age) identified from the National Civil Registry from Hainan Civil Affairs Bureau. Age was ascertained from national identification cards. The current study was carried out after approval of the Ethics Committee of Hainan Hospital of Chinese People’s Liberation Army General Hospital (Sanya, Hainan; Number: 301hn11201601). All participants provided written informed consent prior to the start of the study.
Standard procedures
Based on a standardized protocol, face interviews, physical examinations, and blood analyses were administered by a well-trained research team from the Chinese People’s Liberation Army General Hospital using a home-visiting model. This interdisciplinary research team included internists, geriatricians, cardiologists, endocrinologists, nephrologists, and nurses. Based on the recommendations of the World Health Organization, waist circumference (WC) was measured in standing subjects using a flexible tape, midway between the lowest rib and the iliac crest. Blood pressure was measured on the participants’ right arms with a calibrated desktop sphygmomanometer (Yuwell medical equipment and supply Co., Ltd., Jiangsu, China). Appropriate cuff sizes were determined based on arm circumference. Participants sat in a chair for five minutes with their feet on the floor and the right arm supported at heart level. Systolic blood pressure (SBP) and diastolic blood pressure (DBP) were recorded at the first and fifth Korotkoff sounds, respectively, and reported as the average of two separate measurements at an interval of more than one minute.
Samples of venous blood were routinely drawn by venipuncture, stored at 4° C, and delivered within four hours to the central laboratory at the Department of Biochemistry, Hainan Hospital of Chinese People’s Liberation Army General Hospital. Serum levels of immunoglobulins and complement components were measured with a fully automatic protein analyzer (BNII; Siemens AG, Munich, Germany). Serum levels of triglyceride (TG), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), and fasting blood glucose (FBG) were measured by enzymatic assays (Roche Products Ltd, Basel, Switzerland) using a biochemical autoanalyzer (Cobas c702; Roche Products Ltd), with low-density lipoprotein cholesterol (LDL-C) calculated by the Friedewald equation. Qualified technicians who performed laboratory analyses were blinded to clinical data.
Ultrasound examinations
A portable ultrasound machine (Philips CX50, Philips Medical Systems, Andover, MA, USA) was used to measure periumbilical fat thickness (PFT) and parameters of cardiac structure and function by experienced radiologists who were unaware of clinical and laboratory data. All participants were placed in a supine position and the abdomen was scanned using a 3-12 MHz linear array ultrasound transducer (L12-3; Phillips). Cardiac ultrasound was performed using a 1-5 MHz cardiac transducer (S5-1; Phillips).
Statistical analyses
Continuous variables were described using means with standard deviations for data with normal distribution, and medians with interquartile ranges for data with skewed distribution. Categorical variables were described as numbers and percentages. Comparisons between groups were made using Student’s t-test for continuous variables with normal distribution, Mann–Whitney U test for continuous variables with skewed distribution, and χ2 test for categorical variables. Logistic regression was performed with centenarians as dependent variable, adjusting for age, sex, SBP, DBP, FBG, TC, TG, HDL-C, and LDL-C. PFT, WC, and parameters of cardiac structure were logarithmically transformed to meet the multinormality assumption. Linear regression was performed with PFT, WC, and parameters of cardiac structure as dependent variables, adjusting for age, sex, SBP, DBP, FBG, TC, TG, HDL-C, and LDL-C. Age and sex are demographic characteristics, whereas SBP, DBP, FBG, TC, TG, HDL-C, and LDL-C are generally believed to be related to PFT, WC, and risk factors for cardiac structure. Statistical analyses were made with SPSS version 17 (IBM Corporation, Armonk, NY, USA) at the significance level of P<0.05 for two-sided tests.
Author Contributions
SF, YY and YZ participated in the study design, data analysis and manuscript draft. SW, JD and FL helped with data acquirement and interpretation. JD, FL and YZ helped with manuscript revision. All authors read and approved the final manuscript.
Acknowledgments
We are grateful to all the staff of the CHCCS for their continued cooperation and field work contributions.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Funding
This work was supported by grants from the National Natural Science Foundation of China (81900357, 82001476, 81903392, 81941021), the Military Medical Science and Technology Youth Incubation Program (20QNPY110), the National Key R&D Program of China (2016YFC0105006, 2018YFC2000400), the National S&T Resource Sharing service platform Project of China (YCZYPT[2018]07), the General Hospital of PLA Medical Big Data R&D Project (MBD2018030), the China Postdoctoral Science Foundation funded project (2019M650359), the National Geriatric Disease Clinical Medicine Research Center Project (NCRCG-PLAGH-2017-014), the Central Health Care Scientific Research Project (W2017BJ12), the Hainan Medical and Health Research Project (16A200057), the Sanya Medical and Health Science and Technology Innovation Project (2016YW21, 2017YW22, 2018YW11), the Military Medicine Youth Program of Chinese PLA General Hospital (QNF19069) and the Clinical Scientific Research Supporting Fund of Chinese PLA General Hospital (2017FC-CXYY-3009). The sponsors had no roles in the design, conduct, interpretation, review, approval or control of this article.
References
- 1. Ward ZJ, Bleich SN, Cradock AL, Barrett JL, Giles CM, Flax C, Long MW, Gortmaker SL. Projected U.S. State-level prevalence of adult obesity and severe obesity. N Engl J Med. 2019; 381:2440–50. https://doi.org/10.1056/NEJMsa1909301 [PubMed]
- 2. Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of childhood and adult obesity in the United States, 2011-2012. JAMA. 2014; 311:806–14. https://doi.org/10.1001/jama.2014.732 [PubMed]
- 3. Swinburn BA, Kraak VI, Allender S, Atkins VJ, Baker PI, Bogard JR, Brinsden H, Calvillo A, De Schutter O, Devarajan R, Ezzati M, Friel S, Goenka S, et al. The global syndemic of obesity, undernutrition, and climate change: the lancet commission report. Lancet. 2019; 393:791–846. https://doi.org/10.1016/S0140-6736(18)32822-8 [PubMed]
- 4. Afshin A, Forouzanfar MH, Reitsma MB, Sur P, Estep K, Lee A, Marczak L, Mokdad AH, Moradi-Lakeh M, Naghavi M, Salama JS, Vos T, Abate KH, et al, and GBD 2015 Obesity Collaborators. Health effects of overweight and obesity in 195 countries over 25 years. N Engl J Med. 2017; 377:13–27. https://doi.org/10.1056/NEJMoa1614362 [PubMed]
- 5. Fu S, Luo L, Ye P, Liu Y, Zhu B, Bai Y, Bai J. The abilities of new anthropometric indices in identifying cardiometabolic abnormalities, and influence of residence area and lifestyle on these anthropometric indices in a Chinese community-dwelling population. Clin Interv Aging. 2014; 9:179–89. https://doi.org/10.2147/CIA.S54240 [PubMed]
- 6. Gabrielsson BG, Johansson JM, Lönn M, Jernås M, Olbers T, Peltonen M, Larsson I, Lönn L, Sjöström L, Carlsson B, Carlsson LM. High expression of complement components in omental adipose tissue in obese men. Obes Res. 2003; 11:699–708. https://doi.org/10.1038/oby.2003.100 [PubMed]
- 7. Hernández-Mijares A, Jarabo-Bueno MM, López-Ruiz A, Solá-Izquierdo E, Morillas-Ariño C, Martínez-Triguero ML. Levels of C3 in patients with severe, morbid and extreme obesity: its relationship to insulin resistance and different cardiovascular risk factors. Int J Obes (Lond). 2007; 31:927–32. https://doi.org/10.1038/sj.ijo.0803543 [PubMed]
- 8. Drenick EJ, Bale GS, Seltzer F, Johnson DG. Excessive mortality and causes of death in morbidly obese men. JAMA. 1980; 243:443–45. [PubMed]
- 9. Grover SA, Kaouache M, Rempel P, Joseph L, Dawes M, Lau DC, Lowensteyn I. Years of life lost and healthy life-years lost from diabetes and cardiovascular disease in overweight and obese people: a modelling study. Lancet Diabetes Endocrinol. 2015; 3:114–22. https://doi.org/10.1016/S2213-8587(14)70229-3 [PubMed]
- 10. Nilsson B, Hamad OA, Ahlström H, Kullberg J, Johansson L, Lindhagen L, Haenni A, Ekdahl KN, Lind L. C3 and C4 are strongly related to adipose tissue variables and cardiovascular risk factors. Eur J Clin Invest. 2014; 44:587–96. https://doi.org/10.1111/eci.12275 [PubMed]
- 11. Karkhaneh M, Qorbani M, Mohajeri-Tehrani MR, Hoseini S. Association of serum complement C3 with metabolic syndrome components in normal weight obese women. J Diabetes Metab Disord. 2017; 16:49. https://doi.org/10.1186/s40200-017-0330-6 [PubMed]
- 12. Kagawa M, Kerr D, Uchida H, Binns CW. Differences in the relationship between BMI and percentage body fat between Japanese and Australian-caucasian young men. Br J Nutr. 2006; 95:1002–07. https://doi.org/10.1079/bjn20061745 [PubMed]
- 13. Deurenberg-Yap M, Schmidt G, van Staveren WA, Deurenberg P. The paradox of low body mass index and high body fat percentage among Chinese, Malays and Indians in Singapore. Int J Obes Relat Metab Disord. 2000; 24:1011–17. https://doi.org/10.1038/sj.ijo.0801353 [PubMed]
- 14. Hu Y, Zong G, Liu G, Wang M, Rosner B, Pan A, Willett WC, Manson JE, Hu FB, Sun Q. Smoking cessation, weight change, type 2 diabetes, and mortality. N Engl J Med. 2018; 379:623–32. https://doi.org/10.1056/NEJMoa1803626 [PubMed]
- 15. Fox CS, Massaro JM, Hoffmann U, Pou KM, Maurovich-Horvat P, Liu CY, Vasan RS, Murabito JM, Meigs JB, Cupples LA, D’Agostino RB
Sr , O’Donnell CJ. Abdominal visceral and subcutaneous adipose tissue compartments: association with metabolic risk factors in the framingham heart study. Circulation. 2007; 116:39–48. https://doi.org/10.1161/CIRCULATIONAHA.106.675355 [PubMed] - 16. Ministry of Health of the People’s Republic of China. China Health Statistical Yearbook 2010. Beijing: Peking Union Medical College Press; 2010. Chinese.
- 17. Ismail K, Nussbaum L, Sebastiani P, Andersen S, Perls T, Barzilai N, Milman S. Compression of morbidity is observed across cohorts with exceptional longevity. J Am Geriatr Soc. 2016; 64:1583–91. https://doi.org/10.1111/jgs.14222 [PubMed]
- 18. Jopp DS, Park MK, Lehrfeld J, Paggi ME. Physical, cognitive, social and mental health in near-centenarians and centenarians living in New York city: findings from the fordham centenarian study. BMC Geriatr. 2016; 16:1. https://doi.org/10.1186/s12877-015-0167-0 [PubMed]
- 19. Forouhi NG, Sattar N. CVD risk factors and ethnicity—a homogeneous relationship? Atheroscler Suppl. 2006; 7:11–19. https://doi.org/10.1016/j.atherosclerosissup.2006.01.003 [PubMed]
- 20. Fu S, Luo L, Ye P, Liu Y, Zhu B, Zheng J, Bai Y, Bai J. Overall and abdominal obesity indicators had different association with central arterial stiffness and hemodynamics independent of age, sex, blood pressure, glucose, and lipids in Chinese community-dwelling adults. Clin Interv Aging. 2013; 8:1579–84. https://doi.org/10.2147/CIA.S54352 [PubMed]
- 21. Hotamisligil GS. Inflammation and metabolic disorders. Nature. 2006; 444:860–67. https://doi.org/10.1038/nature05485 [PubMed]
- 22. Muscari A, Antonelli S, Bianchi G, Cavrini G, Dapporto S, Ligabue A, Ludovico C, Magalotti D, Poggiopollini G, Zoli M, and Pianoro Study Group. Serum C3 is a stronger inflammatory marker of insulin resistance than c-reactive protein, leukocyte count, and erythrocyte sedimentation rate: comparison study in an elderly population. Diabetes Care. 2007; 30:2362–68. https://doi.org/10.2337/dc07-0637 [PubMed]
- 23. Baumann H, Gauldie J. The acute phase response. Immunol Today. 1994; 15:74–80. https://doi.org/10.1016/0167-5699(94)90137-6 [PubMed]
- 24. Marette A. Mediators of cytokine-induced insulin resistance in obesity and other inflammatory settings. Curr Opin Clin Nutr Metab Care. 2002; 5:377–83. https://doi.org/10.1097/00075197-200207000-00005 [PubMed]
- 25. Germinario R, Sniderman AD, Manuel S, Lefebvre SP, Baldo A, Cianflone K. Coordinate regulation of triacylglycerol synthesis and glucose transport by acylation-stimulating protein. Metabolism. 1993; 42:574–80. https://doi.org/10.1016/0026-0495(93)90215-a [PubMed]
- 26. Cianflone KM, Maslowska MH, Sniderman AD. Impaired response of fibroblasts from patients with hyperapobetalipoproteinemia to acylation-stimulating protein. J Clin Invest. 1990; 85:722–30. https://doi.org/10.1172/JCI114497 [PubMed]
- 27. Phieler J, Garcia-Martin R, Lambris JD, Chavakis T. The role of the complement system in metabolic organs and metabolic diseases. Semin Immunol. 2013; 25:47–53. https://doi.org/10.1016/j.smim.2013.04.003 [PubMed]
- 28. Muscari A, Massarelli G, Bastagli L, Poggiopollini G, Tomassetti V, Drago G, Martignani C, Pacilli P, Boni P, Puddu P. Relationship of serum C3 to fasting insulin, risk factors and previous ischaemic events in middle-aged men. Eur Heart J. 2000; 21:1081–90. https://doi.org/10.1053/euhj.1999.2013 [PubMed]
- 29. Bastien M, Poirier P, Lemieux I, Després JP. Overview of epidemiology and contribution of obesity to cardiovascular disease. Prog Cardiovasc Dis. 2014; 56:369–81. https://doi.org/10.1016/j.pcad.2013.10.016 [PubMed]
- 30. Peters SA, Bots SH, Woodward M. Sex differences in the association between measures of general and central adiposity and the risk of myocardial infarction: results from the UK biobank. J Am Heart Assoc. 2018; 7:e008507. https://doi.org/10.1161/JAHA.117.008507 [PubMed]
- 31. Wahrenberg H, Hertel K, Leijonhufvud BM, Persson LG, Toft E, Arner P. Use of waist circumference to predict insulin resistance: retrospective study. BMJ. 2005; 330:1363–64. https://doi.org/10.1136/bmj.38429.473310.AE [PubMed]
- 32. ter Maaten JC, Bakker SJ, Serné EH, ter Wee PM, Donker AJ, Gans RO. Insulin’s acute effects on glomerular filtration rate correlate with insulin sensitivity whereas insulin’s acute effects on proximal tubular sodium reabsorption correlation with salt sensitivity in normal subjects. Nephrol Dial Transplant. 1999; 14:2357–63. https://doi.org/10.1093/ndt/14.10.2357 [PubMed]
- 33. Holmes MV, Pulit SL, Lindgren CM. Genetic and epigenetic studies of adiposity and cardiometabolic disease. Genome Med. 2017; 9:82. https://doi.org/10.1186/s13073-017-0474-5 [PubMed]
- 34. Hofmann U, Beyersdorf N, Weirather J, Podolskaya A, Bauersachs J, Ertl G, Kerkau T, Frantz S. Activation of CD4+ T lymphocytes improves wound healing and survival after experimental myocardial infarction in mice. Circulation. 2012; 125:1652–63. https://doi.org/10.1161/CIRCULATIONAHA.111.044164 [PubMed]
- 35. Frantz S, Hofmann U. Monocytes on the scar’s edge. J Am Coll Cardiol. 2012; 59:164–65. https://doi.org/10.1016/j.jacc.2011.09.047 [PubMed]
- 36. Singh MV, Kapoun A, Higgins L, Kutschke W, Thurman JM, Zhang R, Singh M, Yang J, Guan X, Lowe JS, Weiss RM, Zimmermann K, Yull FE, et al. Ca2+/calmodulin-dependent kinase II triggers cell membrane injury by inducing complement factor B gene expression in the mouse heart. J Clin Invest. 2009; 119:986–96. https://doi.org/10.1172/JCI35814 [PubMed]
- 37. del Balzo UH, Levi R, Polley MJ. Cardiac dysfunction caused by purified human C3a anaphylatoxin. Proc Natl Acad Sci USA. 1985; 82:886–90. https://doi.org/10.1073/pnas.82.3.886 [PubMed]
- 38. Fu S, Yao Y, Zhao Y, Luan F. Relationships of hyperhomocysteinemia and hyperuricemia with metabolic syndrome and renal function in Chinese centenarians. Front Endocrinol (Lausanne). 2018; 9:502. https://doi.org/10.3389/fendo.2018.00502 [PubMed]


