Introduction
Late-onset hypogonadism (LOH) is defined as a clinical and biochemical syndrome associated with advancing age and is characterized by symptoms along with deficiency in serum testosterone levels [1]. Testosterone deficiency can adversely affect multiple organ functions and cause both sexual and nonsexual symptoms. The sexual symptoms of LOH include impotence or erectile dysfunction, low libido, and diminished frequency of morning erections, while the nonsexual symptoms include fatigue, depression, and decreased sense of vitality, among others. Because LOH adversely affects the quality of life in aging males with high prevalence (2%~40%) [2, 3]. its clinical significance is becoming increasingly recognized as many countries are facing an aging society. Specifically in Asia, which accounts for more than 60% of the world population, there are more than 8 hundred million aging men (>=40 years old) and the number is increasing (http://populationpyramid.net/).
Although LOH has been increasingly recognized as an age-related disorder, most men with LOH remain undiagnosed [1]. LOH diagnostic criteria have not been established for Asian men because there is no sufficient data to base on. For Caucasian men, a previous study suggested the presence of three sexual symptoms associated with a total testosterone (TT) level of less than 11 nmol/L and a calculated free testosterone (cFT) level of less than 220 pmol/L as the minimum criteria to identify LOH [2]. However, the serum concentration of TT, which shows age-associated decline in Caucasian men and is used as the primary biochemical parameter used to define testosterone deficiency for LOH identification [2, 4], do not decline or even increase with aging in Asian men [5–8]. Thus it is inappropriate to directly apply the criterion developed based on Caucasian population. LOH identification and its epidemiology are likely different among different ethnic groups. In fact, for many diseases, substantial racial or ethnic differences exist in many aspects, including the diagnostic criteria and prevalence [9, 10].
Based on the definition and pathophysiology of LOH [1]. LOH belongs to the organic and primary hypogonadism resulted from the failure of testis to produce physiological concentrations of testosterone [11, 12]. It is hypothesized that the ability of testicular testosterone secretion should be impaired resulting in testosterone deficiency in some, if not all, aging males, which in turn may cause LOH symptoms. The present study has carried out a nationwide, multi-center study in China aimed at establishing an evidence-based and comprehensive criteria for LOH diagnosis with the revelation of pathogenesis of LOH.
Results
Participants
Totally 6296 men aged 40 to 79 years old (Supplementary Figure 1) from six representative areas of China attended this study. Totally 316 participants aged 40 to 79 years old were excluded from the analysis, with 217 because of incomplete response to questionnaires, and 99 because of known diseases of hypothalamus-pituitary-testis axis, or conditions may induce testicular impairment, or current use of medications may affect testosterone level. These exclusions left total 5980 men aged 40 to 79 for analyses. Men aged 22-39 (n=598) were recruited to obtain the baseline of hormones and response to questionnaires. All men (n=598) aged 22-39 completed the survey and were included in the analyses.
Age trends of serum total testosterone
Age trends of hormones of all participants were analyzed with three categories of BMI according to the standards for Chinese population: less than 24 kg/m2 (normal), 24 or greater to less than 28 kg/m2 (overweight), and ≥28 kg/m2 (obese) [13]. Serum TT showed significant difference among the three BMI categories (Supplementary Table 1) and a little fluctuation with aging, whereas no age trend was observed (Figure 1A). Considering some factors may influence TT and conceal the age trend of TT level. Potential factors associated with TT levels were further screened using stepwise multiple linear regression. BMI, residence, smoking, alcohol-drinking, and marital status, were found to be correlated with TT levels, whereas age was not after removing effects of these listed factors (Supplementary Table 2) in our population. We further analyzed the occurrence of several proposed abnormal TT levels (below 8, 11, and 12nmol/L, respectively) with aging [1, 2], and no age trend was observed (Figure 2).
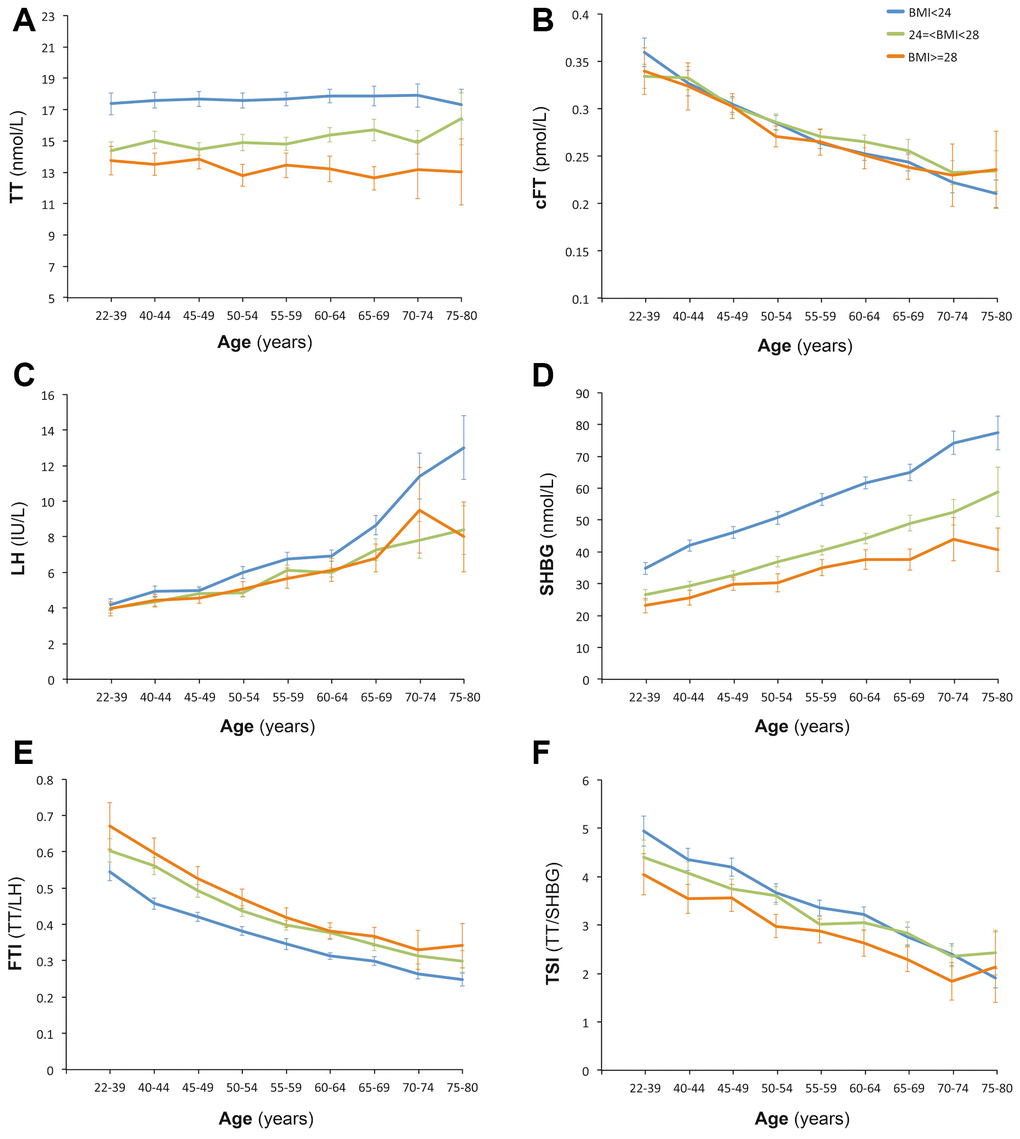
Figure 1. Age trend of hormones. Participants of each age was stratified according to the BMI standard for Chinese population: less than 24 kg/m2 (normal), 24 or greater to less than 28 kg/m2 (overweight), and ≥28 kg/m2 (obese). Pearson Correlation Analysis indicated that age was positively correlated with TT (A) (r=0.05, P<0.01), SHBG (D) (r=0.400, P<0.01) and LH (C) (r=0.360, P<0.01), and was negatively correlated with cFT (B) (r=-0.327, P<0.01), FTI (E) (r=-0.421, P<0.01) and TSI (F) (r=-0.295, P<0.01).
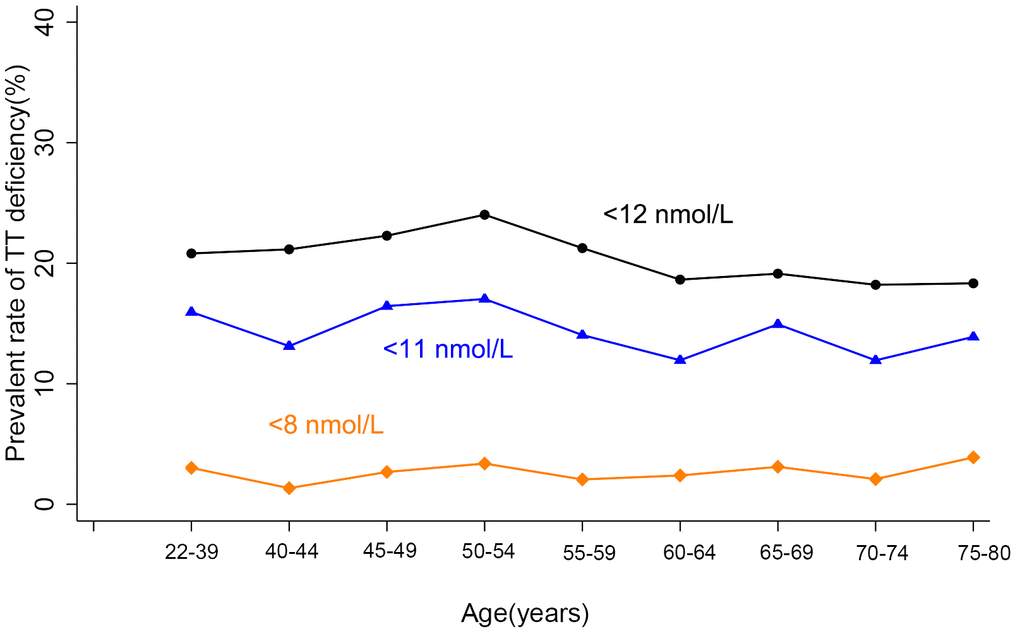
Figure 2. Age trend of TT deficiency. TT deficiency was defined with three kinds of standards: TT<8 nmol/L is the standard for TT supplement; <11 nmol/L and <12 nmol/L is the standard for diagnosis of LOH in EMAS study and other studies, respectively. No age trend of the occurrence of TT deficiency defined by any standard was observed.
Thresholds of cFT and TSI for LOH identification
The cAMS score of 17 was used to classify aging men in the training set into symptomatic and asymptomatic subjects. The trend of the probability of symptom along with the change of cFT, FTI, TSI, TT was further analyzed by Lowess smoothing. Increases of the probability of symptom were observed with decreased cFT, FTI, and TSI, but not with TT (Figure 3). In the training set, thresholds for the significant increase of the probability of symptom were approximately 210 pmol/L for cFT, 0.26 for FTI, and 1.9 for TSI, while no threshold was found for TT. In the validation set, similar results were obtained, with thresholds of 220 pmol/L for cFT, and 1.8 for TSI. The thresholds of cFT and FTI were further identified by piecewise repression in both the training set and the validation set. Consistent results were obtained for cFT<210 pmol/L and TSI<1.8 in these two sets (Table 2).
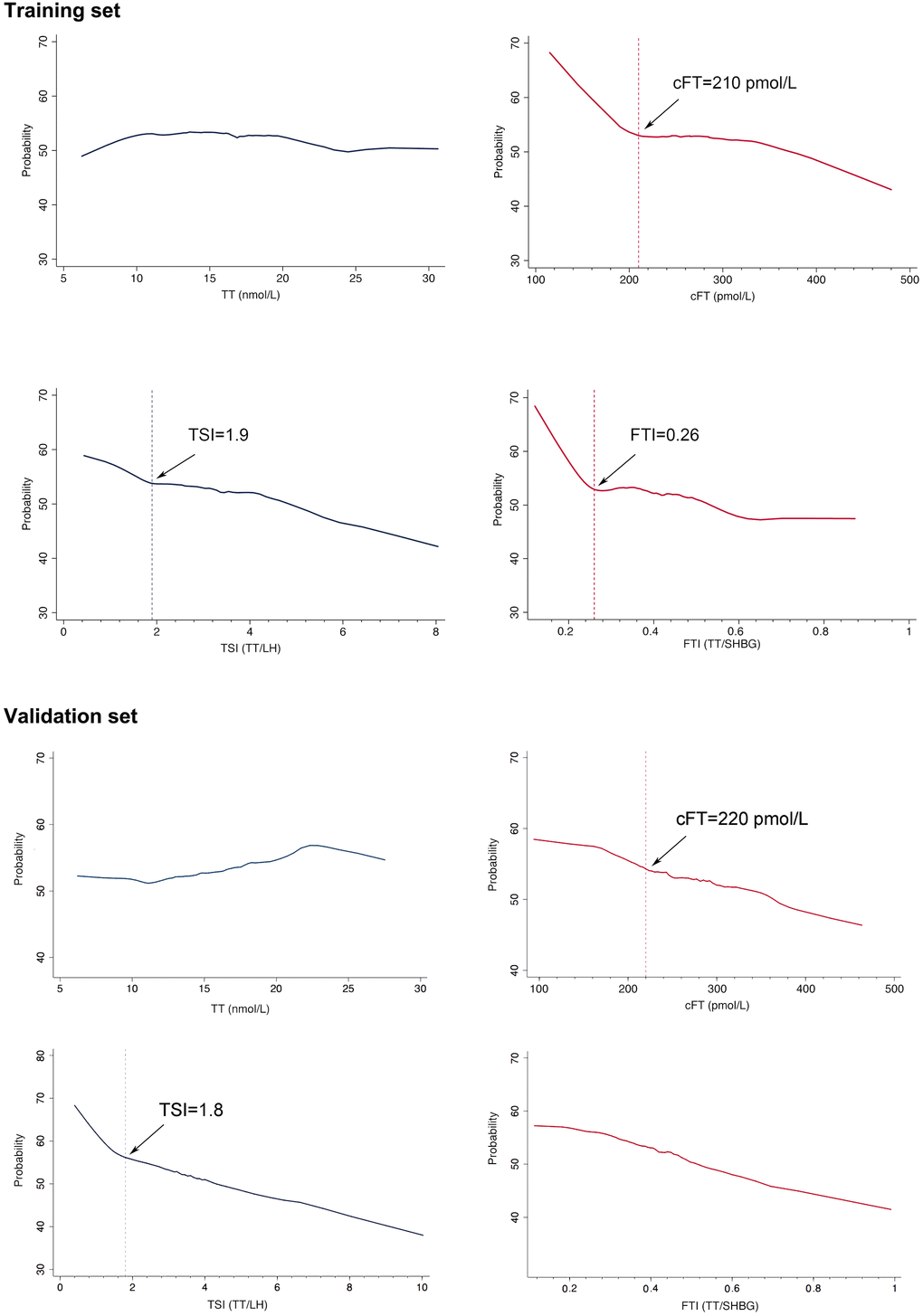
Figure 3. Probability of symptom (cAMS>=17) on the basis of levels of bioavailable testosterone (cFT) and testosterone secreting index (TSI). Symptom was defined by cAMS score (>=17), which contained 10 items related with cFT. Obvious increases of the trend of probability of symptom were observed with decreased cFT, and TSI. Thresholds for the significant changes of the probability of symptom with hormones were denoted by vertical lines.
Table 2. Identification of cFT and TSI thresholds.
| Symptoms | Linear splines | OR (95% CI) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Training set | Validation set | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| cAMS>=17 | cFT<210 pmol/L | 42.10 (2.29, 765.10) | 172.43 (5.75, 5218.68) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| cFT>=210 pmol/L | 9.39 (6.41*10-7, 1.38*108) | 6.36 (1.44*10-6, 2.81*107) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| cFT<220 pmol/L | 5.64 (3.05*10-6, 1.03*107) | 105.64 (2.92, 3827.63) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| cFT>=220 pmol/L | 44.26 (2.20, 880.07) | 6.17 (1.11*10-5, 3.47*106) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TSI<1.8 | 1.24*105 (1.04*105, 1.45*105) | 1.21*105 (1.01*105, 1.41*105) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TSI>=1.8 | 1.62 (0.75, 2.72) | 3.71 (1.03, 13.33) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TSI<1.9 | 3.78 (0.88, 1.32) | 1.28 (1.12, 1.49) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TSI>=1.9 | 1.35 (0.38, 4.76) | 3.90 (1.26, 11.94) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OR (95% CI) was calculated using piecewise regression. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Prevalence of LOH
We thus used the criteria for the LOH identification include the combination of LOH symptom screening by the cAMS score (>=17), with lower bioavailable testosterone (cFT<210pmol/L) and decreased testosterone secretion (TSI<1.8). The overall prevalence of LOH in the study population was 5.32%, with gradual increase from 0.4% in men aged 40-44 years to 16.1% in those 75-79 years. The prevalence of LOH was also found to be increased in men with poor education, never or quit alcohol-drinking, smoking cessation, comorbidities, and living in the country area (Table 3).
Table 3. Prevalence and characterization of LOH.
| Variable | Categories | n | LOH | Prevalence (%) | P value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years) | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 40-44 | 746 | 3 | 0.40 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 45-49 | 1047 | 16 | 1.53 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 50-54 | 916 | 14 | 1.53 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 55-59 | 1067 | 57 | 5.34 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 60-64 | 1046 | 66 | 6.31 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 65-69 | 643 | 75 | 11.66 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 70-74 | 335 | 58 | 17.31 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 75-79 | 180 | 29 | 16.11 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Residence | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| City | 758 | 27 | 3.56 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Country | 5222 | 291 | 5.57 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Education | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Illiteracy | 392 | 45 | 11.48 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Elementary school | 1823 | 145 | 7.95 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Middle school to secondary technical school | 3261 | 115 | 3.53 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Junior college and above | 493 | 13 | 2.64 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | >0.05 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Never | 1887 | 97 | 5.14 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 3371 | 178 | 5.28 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cessation | 691 | 40 | 5.79 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alcohol-drinking | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Never | 1649 | 111 | 6.73 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Social drinker | 2170 | 85 | 3.92 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Often | 1805 | 93 | 5.15 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Quit | 315 | 26 | 8.25 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Comorbidities | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0 | 3341 | 130 | 3.89 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1 | 1957 | 132 | 6.75 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2 | 543 | 44 | 8.10 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| >=3 | 139 | 12 | 8.63 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| P values were calculated by chi square (SPSS). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Discussion
Having confirmed no age-associated decline of TT in 5980 Chinese men aged 40-79 years, this study has screened these men and identified 10 symptoms that are significantly correlated with decreased levels of bioavailable testosterone (cFT) and low indices of testosterone secretion ability (TSI) using the AMS scale. We have then defined the cut-off scores for the 10 symptoms, the thresholds for cFT and TSI, respectively, for LOH diagnosis. Thus, this study is recommending the identification for LOH include the origin of testosterone decline (TSI), the presence of testosterone deficiency (cFT), and the multiple symptoms attributable to the testosterone deficiency (cAMS scores). According to the definition of LOH as well as the physiology and regulation of testosterone, the above recommendation appears to be reasonable. Our results also emphasize that, along with aging, testosterone secretion is diminished leading to the deficiency of bioavailable testosterone, which in turn causes LOH, in spite of normal serum TT levels in aging men.
According the definition of LOH, decreased testosterone secretion and testosterone deficiency form the basis of the pathogenesis for LOH. TT/LH ratio (TSI) and cFT are decreased significantly with aging, demonstrating declines of testicular testosterone secreting function and bioavailable testosterone with advancing age, which is consistent with the basic pathogenesis of LOH. Testosterone is mainly secreted from testicular Leydig cells. In aged males, the total number of Leydig cells are decreased dramatically (about 50%) resulting in impaired functions, which is the major cause of declined testicular function during aging [17, 18]. At the same time, LH level is increased with aging, indicating a compensatory response to testicular function decline. Considering the coexistence of testicular function decline and the compensatory response in aging males, it is reasonable to recommend TSI to be included in the LOH diagnosis. A lower TSI can denote a declined but not sufficiently compensated testicular function and the hypothalamic- pituitary failure, which is considered to be the basic pathogenesis of LOH [1, 3, 12]. In the present study, most (8 of 10) items correlated with cFT also have significant relationships with TSI.
Although TT is usually used in the diagnosis of LOH, its change in aging men is slight and inconsistent [2, 4–8]. Except the compensative effect of LH, the increasing SHBG levels along with aging may bind to and store more testosterone, thus attenuate the decline of TT. TT can not accurately reflect androgen status under this situation [19]. Moreover, it appears that ethnic and lifestyle factors may contribute to the inconsistent change of TT. In most studies from western countries, slight age-associated decline in TT has been observed [2, 4], however, no age-associated declines in TT have been found in studies using large sample size in Asian men [5–8]. Weight gain and smoking cessation are consistently found to be correlated with age-associated declines in TT in longitudinal studies from two independent studies [4, 20]. Body weight and weight change vary among different ethnic groups [21, 22]. In Caucasian men, >25% of men have BMI≧30 (obese) [4, 20], while in the present study, only 4.9% of men have BMI≧30 and 13.0% of men have BMI≧28 (obese in Chinese standard). Smoking cessation rate is much lower (11.6%) in the present study than that in a Caucasian population (48.8%) [2]. Similar difference in smoking cessation rates has been reported previously [23, 24]. On the other hand, only 2 items (in psychological subscale) from the AMS scale and 2 items from the SF-36 scale has been found to be correlated with TT, contrary to 10 items from the AMS and 9 items from the SF-36, respectively, correlating with cFT. Moreover, no sexual symptoms was found to be correlated with TT, but instead, 4 of them are found to be correlated with cFT. These results suggest that cFT, a form of bioavailable testosterone, may better serve as an indicator for testosterone deficiency in aging men than TT. A report also demonstrated that low cFT was associated with hypogonadal symptoms in men with normal TT levels [25].
Among the scales for screening LOH symptoms, AMS remains the most widely used [15]. The present study has identified symptoms correlated with cFT to form a concise version of AMS (cAMS). The cut-off score of the cAMS (≧17) is defined as the threshold because aging males with a score above 17 have significantly lower cFT and TSI. Interestingly, this score is close to the suggested score of AMS scale for LOH screening (score 27 for 17 items) [15]. Moreover, these 10 items show uniform distribution in the three subscales, of which 3 items from psychological subscale, 3 items from somatic subscale, and 4 items from sexual subscale, respectively [15]. Importantly, among these 10 items, 10 of 10 are correlated with cFT and 8 of 10 are correlated with TSI and therefore, they systematically reflect the symptoms caused by the testosterone deficiency and are reasonable for the LOH screening according to the definition and pathogenesis of LOH.
Using the cAMS scores in this study, the participants were classified into symptomatic and asymptomatic categories. The probability of the symptoms is increased with decreased levels of cFT, FTI, and TSI, respectively, confirming the negative correlations between the symptoms and the deficiency of bioavailable testosterone as well as the decompensated testicular functional decline. Further analysis has identified the thresholds of cFT, FTI, and TSI, respectively, below which the probability of the symptoms becomes increasingly prevalent in the training set. Similar thresholds of cFT and TSI have been identified in the validation set. The thresholds of cFT, which represents the bioavailable testosterone, and TSI, which implies the impaired testosterone secretion, and in combination with the cAMS, which includes symptoms correlated with testosterone deficiency, are thus recommended to constitute the comprehensive criteria proposed for the diagnosis of LOH.
Taken together, the present study recommended the identification for LOH include impaired testosterone secretion ability (TSI<1.8), deficiency of bioavailable testosterone (cFT<210 pmol/L) and correlated multiple hypogonadal symptoms (cAMS≧17), respectively. This criteria could be adopted or modified for LOH identification in Asians and the population without obvious TT decline during aging. On the other hand, sales of commercial testosterone products have increased substantially among older men during the past decade [26]. Many men initiate testosterone replacement therapy without testosterone testing [27, 28]. However, the testosterone replacement treatment in LOH men is under extensive debate [11, 12, 29, 30]. Our results suggest that the diminished testosterone secretion and the deficiency of bioavailable testosterone, other than the total testosterone, may better explain the cause and pathogenesis of LOH. And the criteria recommended here for LOH identification were developed based on evidence and according to the definition of LOH, with emphasize on decreased testosterone secretion and bioavailable testosterone levels. Thus our results proposed here may provide a new direction in LOH treatment emphasizing the improvement of testicular secretion of testosterone and the elevation of serum levels of bioavailable testosterone.
Materials and Methods
Study design and participants
From June 1, 2013 to August 31, 2016, a multiple-center cross-sectional survey including six representative areas (provinces) of China was conducted. Multistage random, cluster sampling was performed in this study. Purposive sampling was used at the first stage: east (Jiangsu), southwest (Guizhou), northeast (Shanxi), north (Hebei), south (Guangdong), and the center (Hubei) of China were selected based on the status of economy, life style, and population distribution (Supplementary Figure 1); Stratified cluster sampling was used at the second stage: communities were stratified by urban, suburban, and rural status, communities within the three strata were random sampled with proportional allocation from each locate (province). At the third stage, all men between 22 to 79 years in the selected communities (villages) were informed to attend this study. This study was approved by the Ethical Committee Review Board of Tongji Medical College (NO. 2013S073). Written informed consent was obtained from all participants.
Questionnaires
The participants were invited to fill interviewer-assisted questionnaires including basic information, history of disease, the simplified Chinese version of the AMS scale [15, 16], the Medical Outcomes Study 36-Item Short-Form Healthy Survey (SF-36), and the Beck Depression Inventory. All questionnaires were in simplified Chinese, and local interviewers were trained for the assistance to old participants in some areas with dialect.
Measurements
Height, weight, circumference of abdomen and chest, and blood pressure were measured. A fasting venous blood sample for the hormone measurement was obtained from each participant in the morning between 7:00 and 11:00. Serum was processed, stored at -70° C, and assayed later for measurement of TT, LH), and SHBG by chemiluminescent immunoassays on a Beckman Access Immunoassay system (Beckman coulter, Inc., USA). Between-day imprecision (coefficient of variation) was 8.10 and 6.26% at 0.35 and 12.88 ng/ml, 5.4 and 5.2% at 6.3 and 171 nmol/L, and 6.4 and 5.4% at 4.01 and 55.04 IU/L for TT, SHBG and LH, respectively. cFT was calculated from testosterone and SHBG using mass action equations as previously described [14]. TSI and FTI was calculated using TT/LH equation and TT/SHBG equation, respectively.
Statistical analysis
Questionnaire and hormone data were proofread and entered in EPIdata version 3.02 (Odense, Denmark). Data analyses were performed with SPSS version 18.0 (Armonk, NY, USA), and some graphics were produced using both SPSS and Stata 13.0 (College Station, TX, USA). A P-value <0.05 was considered statistically significant. In all subjects, age trend of hormones was analyzed using multivariate multiple regression; and the differences of hormone levels among BMI categories were compared by Games-Howell. Subjects were then randomly subdivided into a training set and a validation set. In the training set, symptoms associated with testosterone levels and secretion were screened with proportional ordinal logistic regression, and were further confirmed by Mann-Whitney U test. The confirmed symptoms were used to form the scale of LOH symptoms. In both the training set and validation set, the trend of the probability of LOH symptom along with the change of hormone levels was analyzed by Lowess smoothing, and the thresholds of cFT and FTI were identified with piecewise repression.
Author Contributions
Chengliang Xiong, Yiqun Gu, Xuejun Shang, Yuanzhong Zhou, Honggang Li concepted and designed the study. Chengliang Xiong, Yiqun Gu, Xuejun Shang, and Yuanzhong Zhou provided administrative, technical, or material support, and led the research teams at each study center. All authors performed experiments and attended the acquisition, analysis, or interprepation of data. Honggang Li and Yuanzhong Zhou drafted the manuscript. All authors have read and approved the final submitted manuscript.
Conflicts of Interest
All the authors declare that they have no conflicts of interest.
Funding
This study was funded by the 12th Five-Year Plan of National Science and Technology of China (No. 2012BAI32B03) and Training Program of the Major Research Plan of National Natural Science Foundation of China (No. 91649111).
References
- 1. Lunenfeld B, Mskhalaya G, Zitzmann M, Arver S, Kalinchenko S, Tishova Y, Morgentaler A. Recommendations on the diagnosis, treatment and monitoring of hypogonadism in men. Aging Male. 2015; 18:5–15. https://doi.org/10.3109/13685538.2015.1004049 [PubMed]
- 2. Wu FC, Tajar A, Beynon JM, Pye SR, Silman AJ, Finn JD, O’Neill TW, Bartfai G, Casanueva FF, Forti G, Giwercman A, Han TS, Kula K, et al, and EMAS Group. Identification of late-onset hypogonadism in middle-aged and elderly men. N Engl J Med. 2010; 363:123–35. https://doi.org/10.1056/NEJMoa0911101 [PubMed]
- 3. Corona G, Rastrelli G, Maggi M. Diagnosis and treatment of late-onset hypogonadism: systematic review and meta-analysis of TRT outcomes. Best Pract Res Clin Endocrinol Metab. 2013; 27:557–79. https://doi.org/10.1016/j.beem.2013.05.002 [PubMed]
- 4. Shi Z, Araujo AB, Martin S, O’Loughlin P, Wittert GA. Longitudinal changes in testosterone over five years in community-dwelling men. J Clin Endocrinol Metab. 2013; 98:3289–97. https://doi.org/10.1210/jc.2012-3842 [PubMed]
- 5. Li JY, Li XY, Li M, Zhang GK, Ma FL, Liu ZM, Zhang NY, Meng P. Decline of serum levels of free testosterone in aging healthy Chinese men. Aging Male. 2005; 8:203–06. https://doi.org/10.1080/13685530500356010 [PubMed]
- 6. Liu Z, Liu J, Shi X, Wang L, Yang Y, Tao M. Dynamic alteration of serum testosterone with aging: a cross-sectional study from Shanghai, China. Reprod Biol Endocrinol. 2015; 13:111. https://doi.org/10.1186/s12958-015-0107-z [PubMed]
- 7. Iwamoto T, Yanase T, Koh E, Horie H, Baba K, Namiki M, Nawata H. [Reference ranges of total serum and free testosterone in Japanese male adults]. Nihon Hinyokika Gakkai Zasshi. 2004; 95:751–60. https://doi.org/10.5980/jpnjurol1989.95.751 [PubMed]
- 8. Xia F, Wang N, Han B, Li Q, Chen Y, Zhu C, Chen Y, Pu X, Cang Z, Zhu C, Lu M, Meng Y, Guo H, et al. Hypothalamic-Pituitary-Gonadal Axis in Aging Men and Women: Increasing Total Testosterone in Aging Men. Neuroendocrinology. 2017; 104:291–301. https://doi.org/10.1159/000446656 [PubMed]
- 9. Saunders E, Ofili E. Epidemiology of atherothrombotic disease and the effectiveness and risks of antiplatelet therapy: race and ethnicity considerations. Cardiol Rev. 2008; 16:82–88. https://doi.org/10.1097/CRD.0b013e31815685fa [PubMed]
- 10. Wong WL, Su X, Li X, Cheung CM, Klein R, Cheng CY, Wong TY. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob Health. 2014; 2:e106–16. https://doi.org/10.1016/S2214-109X(13)70145-1 [PubMed]
- 11. Bhasin S, Brito JP, Cunningham GR, Hayes FJ, Hodis HN, Matsumoto AM, Snyder PJ, Swerdloff RS, Wu FC, Yialamas MA. Testosterone therapy in men with hypogonadism: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2018; 103:1715–44. https://doi.org/10.1210/jc.2018-00229 [PubMed]
- 12. Salonia A, Rastrelli G, Hackett G, Seminara SB, Huhtaniemi IT, Rey RA, Hellstrom WJ, Palmert MR, Corona G, Dohle GR, Khera M, Chan YM, Maggi M. Paediatric and adult-onset male hypogonadism. Nat Rev Dis Primers. 2019; 5:38. https://doi.org/10.1038/s41572-019-0087-y [PubMed]
- 13. Zhou BF, and Cooperative Meta-Analysis Group of the Working Group on Obesity in China. Predictive values of body mass index and waist circumference for risk factors of certain related diseases in Chinese adults —study on optimal cut-off points of body mass index and waist circumference in Chinese adults. Biomed Environ Sci. 2002; 15:83−96.
- 14. Vermeulen A, Verdonck L, Kaufman JM. A critical evaluation of simple methods for the estimation of free testosterone in serum. J Clin Endocrinol Metab. 1999; 84:3666–72. https://doi.org/10.1210/jcem.84.10.6079 [PubMed]
- 15. Heinemann LA, Zimmermann T, Vermeulen A, Thiel C, Hummel W. A new “aging males” symptoms’ rating scale. The Aging Male. 1999; 2:105–14.
- 16. Kong XB, Guan HT, Li HG, Zhou Y, Xiong CL. The ageing males’ symptoms scale for Chinese men: reliability, validation and applicability of the Chinese version. Andrology. 2014; 2:856–61. https://doi.org/10.1111/j.2047-2927.2013.00145.x [PubMed]
- 17. Huhtaniemi I, Forti G. Male late-onset hypogonadism: pathogenesis, diagnosis and treatment. Nat Rev Urol. 2011; 8:335–44. https://doi.org/10.1038/nrurol.2011.47 [PubMed]
- 18. Baburski AZ, Sokanovic SJ, Bjelic MM, Radovic SM, Andric SA, Kostic TS. Circadian rhythm of the leydig cells endocrine function is attenuated during aging. Exp Gerontol. 2016; 73:5–13. https://doi.org/10.1016/j.exger.2015.11.002 [PubMed]
- 19. Rastrelli G, O’Neill TW, Ahern T, Bártfai G, Casanueva FF, Forti G, Keevil B, Giwercman A, Han TS, Slowikowska-Hilczer J, Lean ME, Pendleton N, Punab M, et al, and EMAS study group. Symptomatic androgen deficiency develops only when both total and free testosterone decline in obese men who may have incident biochemical secondary hypogonadism: prospective results from the EMAS. Clin Endocrinol (Oxf). 2018; 89:459–69. https://doi.org/10.1111/cen.13756 [PubMed]
- 20. Camacho EM, Huhtaniemi IT, O’Neill TW, Finn JD, Pye SR, Lee DM, Tajar A, Bartfai G, Boonen S, Casanueva FF, Forti G, Giwercman A, Han TS, et al, and EMAS Group. Age-associated changes in hypothalamic-pituitary-testicular function in middle-aged and older men are modified by weight change and lifestyle factors: longitudinal results from the european male ageing study. Eur J Endocrinol. 2013; 168:445–55. https://doi.org/10.1530/EJE-12-0890 [PubMed]
- 21. Deurenberg P, Deurenberg-Yap M, Guricci S. Asians are different from caucasians and from each other in their body mass index/body fat per cent relationship. Obes Rev. 2002; 3:141–46. https://doi.org/10.1046/j.1467-789x.2002.00065.x [PubMed]
- 22. Ouyang Y, Wang H, Su C, Wang Z, Song Y, Xiao Y, Du W, Zhang B. Use of quantile regression to investigate changes in the body mass index distribution of Chinese adults aged 18-60 years: a longitudinal study. BMC Public Health. 2015; 15:278. https://doi.org/10.1186/s12889-015-1606-8 [PubMed]
- 23. Giovino GA, Mirza SA, Samet JM, Gupta PC, Jarvis MJ, Bhala N, Peto R, Zatonski W, Hsia J, Morton J, Palipudi KM, Asma S, and GATS Collaborative Group. Tobacco use in 3 billion individuals from 16 countries: an analysis of nationally representative cross-sectional household surveys. Lancet. 2012; 380:668–79. https://doi.org/10.1016/S0140-6736(12)61085-X [PubMed]
- 24. Chen Z, Peto R, Zhou M, Iona A, Smith M, Yang L, Guo Y, Chen Y, Bian Z, Lancaster G, Sherliker P, Pang S, Wang H, et al, and GATS Collaborative Group. Contrasting male and female trends in tobacco-attributed mortality in China: evidence from successive nationwide prospective cohort studies. Lancet. 2015; 386:1447–56. https://doi.org/10.1016/S0140-6736(15)00340-2
- 25. Antonio L, Wu FC, O’Neill TW, Pye SR, Ahern TB, Laurent MR, Huhtaniemi IT, Lean ME, Keevil BG, Rastrelli G, Forti G, Bartfai G, Casanueva FF, et al, and European Male Ageing Study Study Group. Low free testosterone is associated with hypogonadal signs and symptoms in men with normal total testosterone. J Clin Endocrinol Metab. 2016; 101:2647–57. https://doi.org/10.1210/jc.2015-4106 [PubMed]
- 26. Baillargeon J, Kuo YF, Westra JR, Urban RJ, Goodwin JS. Testosterone prescribing in the United States, 2002-2016. JAMA. 2018; 320:200–02. https://doi.org/10.1001/jama.2018.7999 [PubMed]
- 27. Layton JB, Li D, Meier CR, Sharpless JL, Stürmer T, Jick SS, Brookhart MA. Testosterone lab testing and initiation in the United Kingdom and the United States, 2000 to 2011. J Clin Endocrinol Metab. 2014; 99:835–42. https://doi.org/10.1210/jc.2013-3570 [PubMed]
- 28. Muram D, Zhang X, Cui Z, Matsumoto AM. Use of Hormone Testing for the Diagnosis and Evaluation of Male Hypogonadism and Monitoring of Testosterone Therapy: Application of Hormone Testing Guideline Recommendations in Clinical Practice. J Sex Med. 2015; 12:1886–94. https://doi.org/10.1111/jsm.12968 [PubMed]
- 29. Nguyen CP, Hirsch MS, Moeny D, Kaul S, Mohamoud M, Joffe HV. Testosterone and ”age-related hypogonadism“—FDA concerns. N Engl J Med. 2015; 373:689–91. https://doi.org/10.1056/NEJMp1506632 [PubMed]
- 30. Yeap BB, Page ST, Grossmann M. Testosterone treatment in older men: clinical implications and unresolved questions from the testosterone trials. Lancet Diabetes Endocrinol. 2018; 6:659–72. https://doi.org/10.1016/S2213-8587(17)30416-3 [PubMed]



