Introduction
Asthma is one of the most common chronic diseases in children and adults. As the main cause of emergency department visits and hospitalizations, asthma severely affects patients’ quality of life [1]. Over the past few decades, the prevalence of asthma has continuously increased, which may be mainly due to environmental pollution caused by industrialization and urbanization [2, 3]. It has been confirmed that unfavorable meteorological factors, including high temperature, increased concentrations of greenhouse gases, and fine particulate pollution, affect patients with existing respiratory diseases and increase the incidence and prevalence of respiratory diseases and epidemic trends [4–6].
Among meteorological factors, air pollution is the most closely related to respiratory health. Particulate matter (PM) is one of the main components of air pollutants, which is mainly composed of coal combustion, automobile exhaust, dust from construction sites, and smoke from combustions [7]. Because of its physicochemical characteristics, PM can adsorb harmful substances on the surface and persist in the air for a long time [8]. With the process of gas exchange, PM enters the peripheral respiratory system or even alveoli and causes direct cell damage, which leads to multiple diseases, such as asthma, tumors, and acute coronary events [9, 10].
Previous epidemiological studies have reported that PM likely aggravates the respiratory symptoms and increases the morbidity and emergency department visits of asthma patients [11, 12]. It has been confirmed that multiple prolonged exposures to PM can exacerbate airway inflammation in the house dust mite (HDM)-induced asthma model and the pathological mechanisms might be related to the transforming growth factor-beta (TGF-β), PD-L1, TH17 cells, and Treg cells [13–15].
Due to the differences in the natural environment and economic status in regions, air pollution and disease occurrence also differ between regions. Moreover, few studies have focused on the effects of short-term PM exposure on airway inflammation in asthma. Therefore, it is urgent to understand the relationship between PM concentration and asthma in a particular region and the impact on airway inflammation posed by short-term exposure to PM.
Our study is the first to analyze the correlation between the asthma visits of children’s hospitals affiliated with Zhejiang University Medical College and the PM concentration in Hangzhou in the past 5 years. It was found that PM concentration in the atmosphere was positively correlated to the emergency department visits of children with asthma. In a further study in mice, we verified that short-term exposure to PM could aggravate allergic airway inflammation, in which IL-33 might be a promising target.
Results
PM concentration and pediatric asthma visits have correlating seasonal fluctuations
According to the data from the national air pollution prevention and control monitoring station, we described the monthly average PM concentration, including PM2.5 and PM10. It was found that the monthly average PM concentration in Hangzhou shows a strong correlation to seasons. Fine particulate pollution, including PM2.5 and PM10, aggravated in winter and moderated in summer. From January 2013 to December 2017, the monthly average PM2.5 concentration fluctuated between 22.8 and 146.5 μg/m3, while PM10 varies between 38.2 and 197.2 μg/m3 (Figure 1A, 1B).
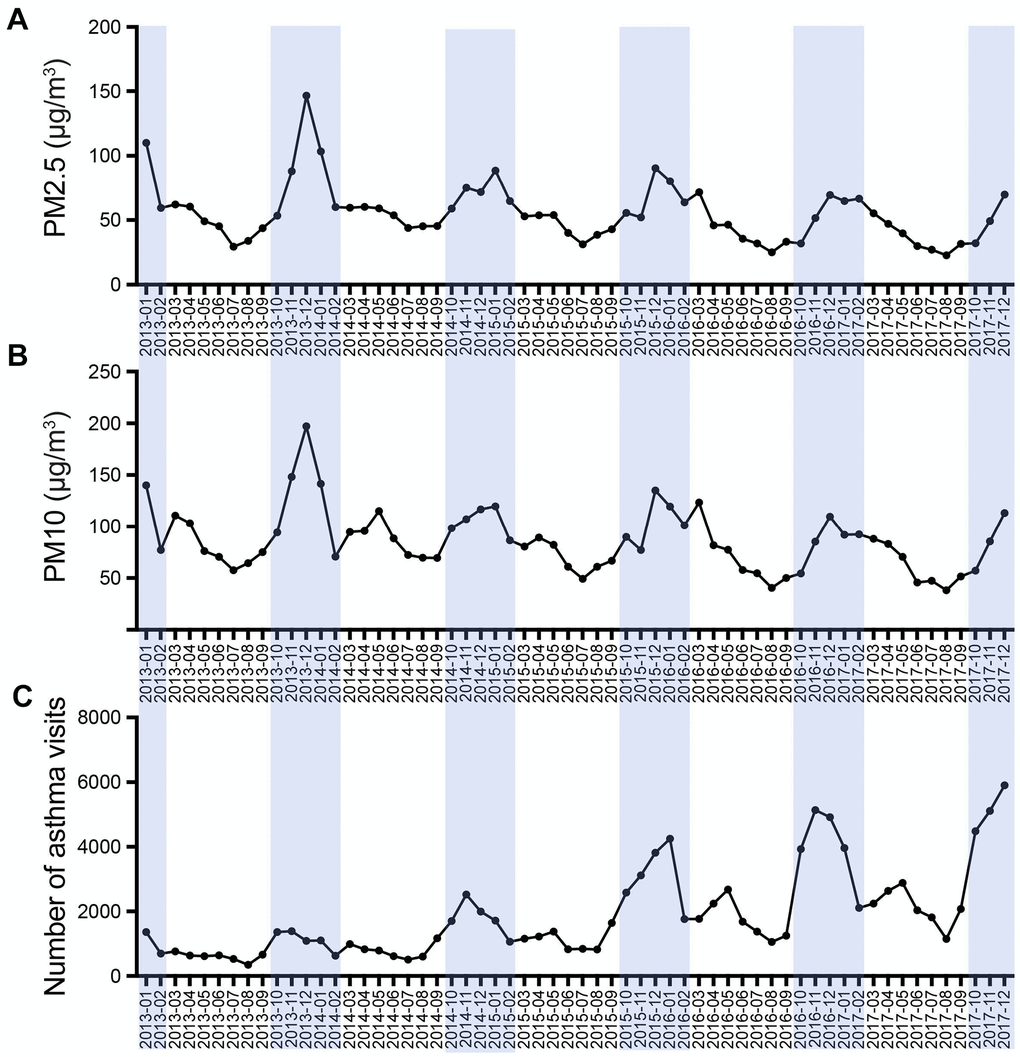
Figure 1. Temporal pattern of monthly average PM concentration (A, B) and asthma visits (C) during January 2013 to December 2017.
In the 5-year study period, there were 16,779,739 visits involving children aged 0–14 in the outpatient and emergency department in the Children’s Hospital of Zhejiang University. We counted the number of children with asthma and found that the visits showed clear seasonal fluctuations (Figure 1C). From January 2013 to December 2017, the attendance of children with asthma (median) was 1377.50 per month, and in outpatient and emergency departments, the monthly ratio (median) of children with asthma in the total patients was 0.55% (Table 1).
Table 1. Average monthly outpatient and emergency visits in 5 years.
| Variables | <6y | ≥6y | All ages | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Outpatient visits | 161,227.00 (138,575.25-190,498.50) | 56,584.50 (47,212.75-67,357.00) | 219,485.00 (185,633.75-258,372.50) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Outpatient visits for asthma | 1,005.00 (586.00-1,919.75) | 313.50 (232.50-457.00) | 1,360.50 (819.75-2,342.50) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Emergency visits | 40,221.50 (32,030.00-53,310.00) | 9,854.50 (8,763.00-13,654.50) | 50,890.50 (40,371.25-66,558.75) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Emergency visits for asthma | 5.00 (3.00-9.00) | 5.50 (3.00-9.00) | 9.50 (6.25-15.75) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Asthma visits | 1,017.00 (592.00-1,955.25) | 317.00 (239.00-464.75) | 1,377.50 (827.50-2,453.25) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total outpatient and emergency visits | 202,946.00 (169,656.00-242,557.50) | 68,469.00 (56,088.50-81,791.00) | 266,244.00 (228,224.25-326,361.25) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ratio | 0.56% (0.37%-0.86%) | 0.47% (0.34%-0.68%) | 0.55% (0.37%-0.80%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Expressed as Median (quartile). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Table 1: The table shows the monthly average visits of patients (medians and interquartile) in the Children’s Hospital of Zhejiang University during 2013–2017. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Note: Ratio, the proportion of asthmatic patients in the outpatient and emergency visits. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The visits of asthma were associated with of PM2.5 and PM10 concentrations
We analyzed the monthly visits of asthmatic children and the concentration of PM in the atmosphere. The concentrations of both PM2.5 and PM10 showed moderately positive correlation to emergency visits of asthma (r = 0.554, p < 0.001 and r = 0.552, p < 0.001) (Figures 2A, 3A and Tables 2, 3). Meanwhile, the concentrations of PM2.5 and PM10 were correlated to the ratio of asthmatic patients in total outpatient and emergency department visits (r = 0.309, p = 0.016 and r = 0.302, p = 0.019) (Figures 2D, 3D and Tables 2, 3). After grouping the children according to age, the emergency visits, and the ratio of asthmatic children in total patients under 6 years old were moderately correlated with the concentrations of PM2.5 (r = 0.611, p < 0.001 and r = 0.323, p = 0.012) (Figure 2B, 2E and Table 2) and PM10 (r = 0.615, p < 0.001 and r = 0.296, p = 0.022) (Figure 3B, 3E and Table 3), respectively. However, in the age group of 6 years and older, PM2.5 and PM10 concentrations generally correlated to the emergency visits (r = 0.400, p = 0.002 and r = 0.410, p = 0.001) (Figures 2C, 3C and Tables 2, 3), and there was no correlation between PM and the ratio of asthmatic visits in total outpatient and emergency department visits (r = 0.169, p = 0.196 and r = 0.228, p = 0.080) (Figures 2F, 3F and Tables 2, 3).
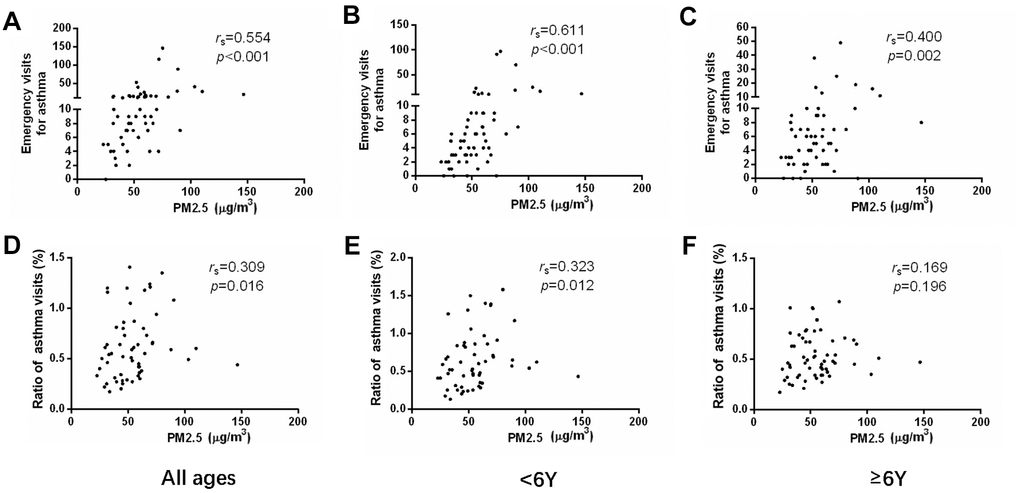
Figure 2. The correlations between PM2.5 concentration and emergency department visits (A–C) or ratio of asthmatic children to total patients in outpatient and emergency departments (D–F) in different age groups.
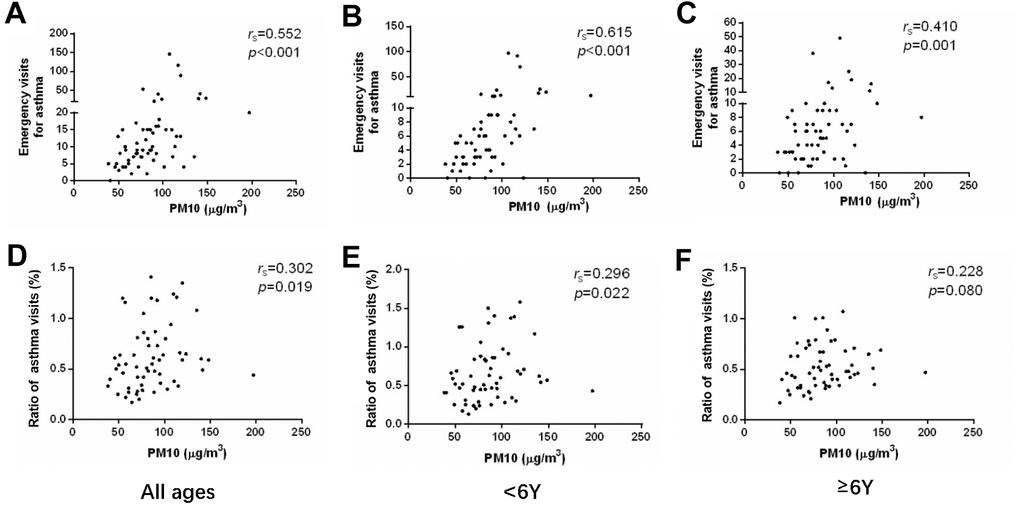
Figure 3. The correlations between PM10 concentration and emergency department visits (A–C) or ratio of asthmatic children to total patients in outpatient and emergency departments (D–F) in different age groups.
Table 2. The correlation between visits and PM2.5 concentration.
| Variables | <6y | ≥6y | All ages | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs | p | rs | p | rs | p | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Outpatient visits | 0.096 | 0.463 | -0.450 | <0.001 | 0.227 | 0.081 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Outpatient visits for asthma | 0.184 | 0.159 | -0.060 | 0.649 | 0.154 | 0.240 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Emergency visits | 0.110 | 0.401 | -0.003 | 0.983 | 0.089 | 0.497 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Emergency visits for asthma | 0.611 | <0.001 | 0.400 | 0.002 | 0.554 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Asthma visits | 0.192 | 0.143 | -0.032 | 0.806 | 0.167 | 0.202 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total outpatient and emergency visits | 0.106 | 0.419 | -0.398 | 0.002 | 0.197 | 0.132 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ratio | 0.323 | 0.012 | 0.169 | 0.196 | 0.309 | 0.016 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Table 2: The table shows the correlation between visits and PM2.5 concentration in different age groups. 0.00 ≤ rs < 0.30 means feeble correlation; 0.30 ≤ rs <0.80 means moderate correlation; 0.80 ≤ rs < 1.00 means strong correlation. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Note: Ratio, the proportion of asthmatic patients in the outpatient and emergency visits. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 3. The correlation between visits and PM10 concentration.
| Variables | <6y | ≥6y | All ages | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs | p | rs | p | rs | p | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Outpatient visits | -0.041 | 0.758 | -0.415 | 0.001 | -0.179 | 0.170 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Outpatient visits for asthma | 0.193 | 0.139 | 0.005 | 0.967 | 0.168 | 0.199 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Emergency visits | -0.054 | 0.680 | 0.065 | 0.620 | -0.032 | 0.806 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Emergency visits for asthma | 0.615 | <0.001 | 0.410 | 0.001 | 0.552 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Asthma visits | 0.202 | 0.122 | 0.035 | 0.791 | 0.183 | 0.161 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total outpatient and emergency visits | -0.051 | 0.696 | -0.357 | 0.005 | -0.143 | 0.275 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ratio | 0.296 | 0.022 | 0.228 | 0.080 | 0.302 | 0.019 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Table 3: The table shows the correlation between visits and PM10 concentration in different age groups. 0.00 ≤ rs < 0.30 means feeble correlation; 0.30 ≤ rs < 0.80 means moderate correlation; 0.80 ≤ rs < 1.00 means strong correlation. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Note: Ratio, the proportion of asthmatic patients in the outpatient and emergency visits. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
PM aggravated the HDM-induced airway inflammation
We established an HDM-induced allergic airway inflammation model with short-term PM exposure. Compared to the HDM group, PM intervention led to a significantly increasing tendency in total cell number and eosinophil (absolute and differential counts) in bronchoalveolar lavage fluid (BALF) (p < 0.05, p < 0.05, p < 0.01) (Figure 4A–4C). Additionally, lungs were homogenized to measure the concentration of cytokines by enzyme-linked immunosorbent assay (ELISA). The result showed that PM exposure resulted in the pulmonary accumulation of IL-33 and IL-25 in the HDM-induced airway inflammation (p < 0.001, p < 0.01) (Figure 4D, 4E). Hematoxylin and eosin (H&E) staining and Periodic Acid-Schiff (PAS) staining of pathological sections showed that infiltration of inflammatory cells around the airway and secretion of mucus in the airway in the HDM-induced airway inflammation were increased remarkably with PM intervention (p < 0.05, p < 0.01) (Figure 4F–4I).
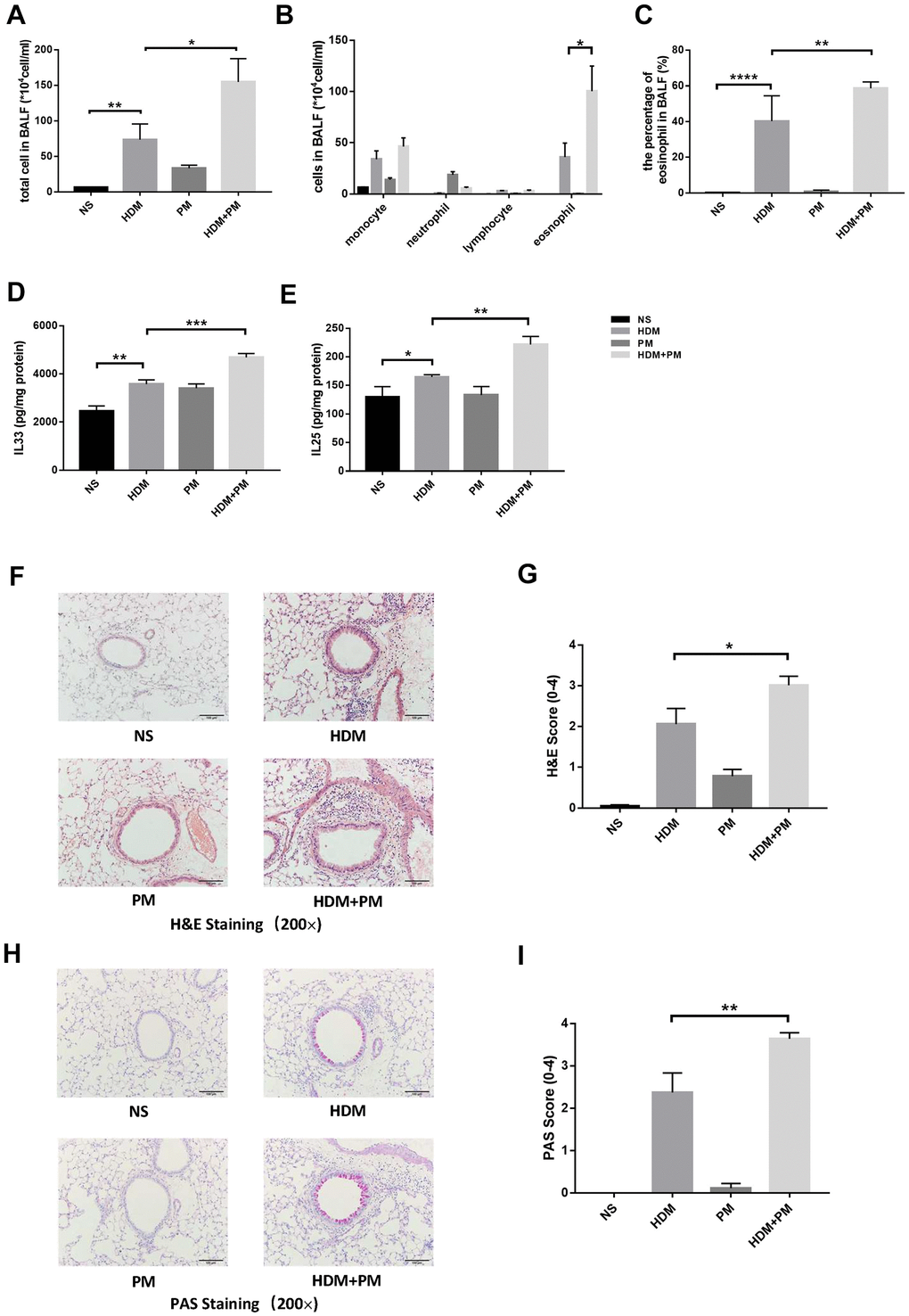
Figure 4. PM2.5 aggravated the HDM-induced airway inflammation. Male C57BL/6 mice (N = 8 for each group) were instilled intratracheally with NS, HDM, PM, or HDM+PM. Total inflammatory cells, cell differentiation of inflammatory cells, and the percentage of eosinophils in BALF were assessed (A–C). Protein levels of IL-33 (D) and IL-25 (E) in the lung tissues were measured using ELISA. Representative images of lung tissues stained with H&E (F) and PAS (H) were showed under the microscope (10 × 20 magnification). Inflammatory score (G) and mucus production (I) with semi-quantification (score: 0–4) was analyzed (N = 10 images for each group). Data were presented as Means ± SEM (*, P < 0.05; **, P < 0.01; ***, P < 0.001; ****P < 0.0001).
IL-33 neutralizing antibody exerted a protective role on PM-aggravated HDM-induced airway inflammation
To further investigate the effect of IL-33 on the PM exposure-aggravated airway inflammation, we administered an IL-33 neutralizing antibody before PM exposure in HDM-induced asthma models (Figure 5A). We evaluated the number of total cells, and cell differential counts in the BALF. Compared to the control group, the allergic mice exposed to PM had a prominent decrease in total cell number and eosinophil (absolute and differential counts) in BALF with the administration of IL-33 antibody (p < 0.01, p < 0.01, p < 0.001) (Figure 5B–5D). The protein level of IL-33 and IL-25 in the lung tissues of asthmatic mice were increased markedly after PM intervention (p < 0.05, p < 0.05) and decreased dramatically with the IL-33 antibody administration (p < 0.0001, p < 0.05) (Figure 5E, 5F). Moreover, histological analyses revealed the infiltration of inflammatory cells and the secretion of mucus of the PM-exposed asthmatic mice with IL-33 antibody were significantly alleviated versus the control group (p < 0.05, p < 0.05) (Figure 5G–5J).
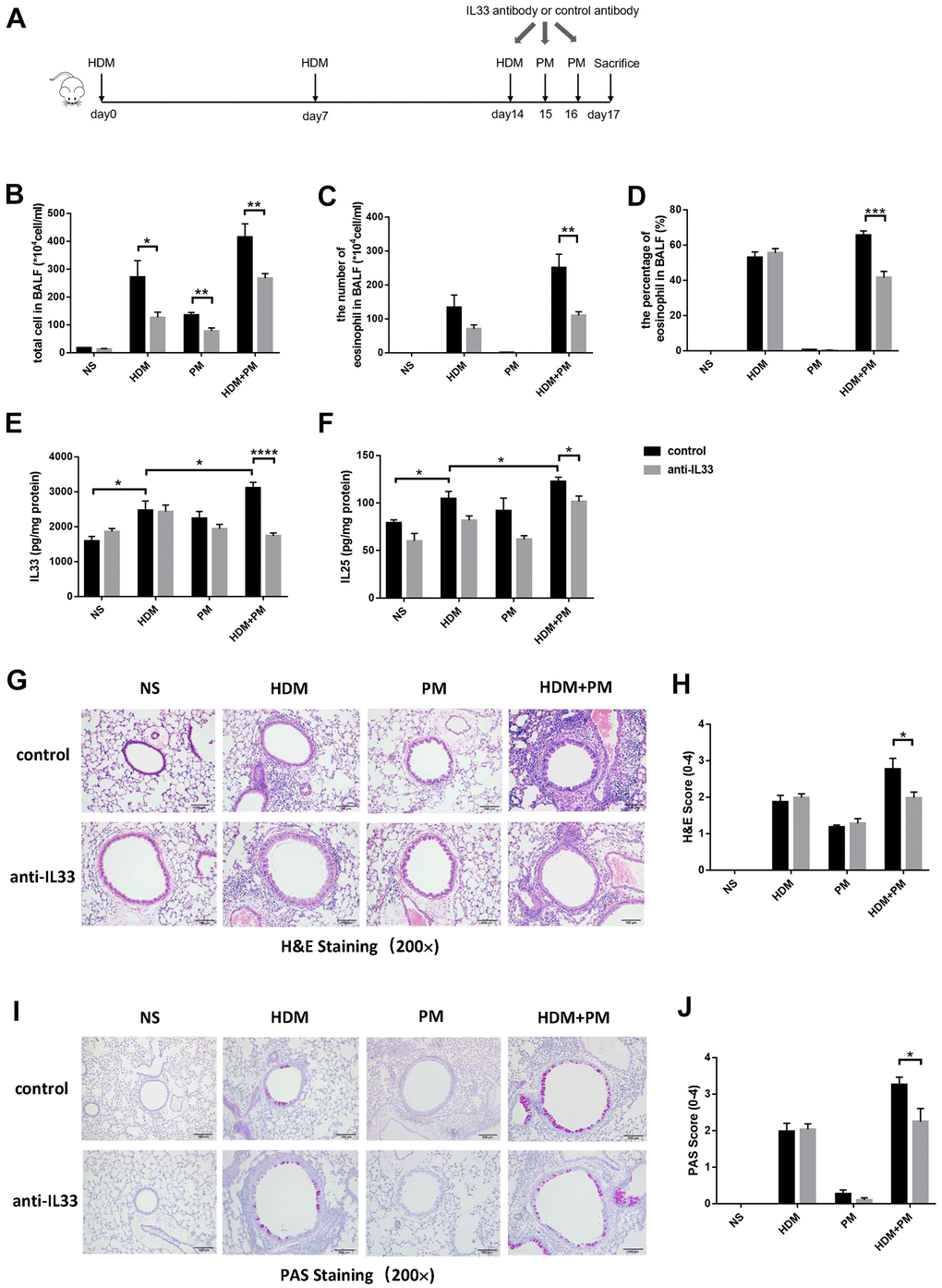
Figure 5. IL-33 antibody alleviated the aggravation of airway inflammation caused by PM exposure in the HDM-induced allergic mice. Male mice in the control and experimental groups were injected intraperitoneally with IL-33 antibody or control antibody, respectively (N = 8 for each group). Protocol for the IL-33 antibody or control antibody intervention (A). Total inflammatory cells, eosinophil proportion, and eosinophil number were analyzed (B–D). Protein levels of IL-33 (E) and IL-25 (F) in the lung tissues were measured using ELISA. Representative images of lung tissues stained with H&E (G) and PAS (I) were showed under the microscope (10 × 20 magnification). Inflammatory score (H) and mucus production (J) with semi-quantification (score: 0–4) were analyzed under the microscope (10 × 20 magnification). Data were presented as means ± SEM (*, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, P < 0.0001).
Discussion
PM is one of the most harmful constituents of air pollution to human health. Exposure to PM, especially PM2.5, can increase hospitalizations and mortality of respiratory diseases, including asthma, impaired lung function, and even lung cancer [16, 17]. The epidemiological investigation found that the emergency department visits of asthmatic patients increased along with the higher levels of PM2.5 concentration, and children were more susceptible to increased PM2.5 than adults [18]. In addition, the severity of the symptoms of childhood asthma and the incidence of severe asthma is believed to be related to the concentration of PM2.5 [19]. Since the compositions of PM and the incidence of asthma were different between regions, we analyzed the correlation between the visits of asthmatic children and PM concentration in the Hangzhou area. We verified that PM 2.5 and PM10 concentrations positively correlated to both the number of asthmatic children in the emergency department and the ratio of asthmatic patients in total visits of outpatient and emergency departments.
Several studies have shown that different populations respond differently to atmospheric PM. In children and people over 65 years old, PM2.5 has a greater impact on the respiratory system than other populations [20]. Therefore, we further divided the asthmatic children into two groups (children < 6 years old and ≥ 6 years old). The results confirmed that in children younger than 6 years old, the correlation between PM concentration and asthma aggravation was more obvious than the older children. Consequently, young children with asthma are more sensitive to the influence of PM, which might be due to the incomplete development of the children’s respiratory and immune systems. This also suggests that more attention is needed to the impact of PM on children, especially young children when formulating policies on air pollution.
It has been reported that long-term repeated exposure to PM can lead to increased airway inflammation in the ovalbumin (OVA)-induced asthma model [21, 22]. However, there are few studies on the short-term exposure to PM in the HDM-induced airway inflammation model. We established the HDM-induced airway inflammation model and substantiated that short-term PM intervention aggravated the airway inflammatory response in mice. Histological analysis further confirmed that PM exacerbated the infiltration of inflammatory cells around the airway and secretion of mucus in the airway. These results were essentially consistent with the effect of long-term PM exposure on airway inflammation in the asthma model [21, 22]. This indicates that short-term intervention of PM induced the increase of eosinophils in the airway and aggravated the airway inflammation of asthma.
Previous studies have suggested that the activation of inflammatory-associated cells might be involved in the molecular mechanism of PM-induced airway inflammation [23, 24]. In vitro, PM was associated with increased release of cytokines, such as IL-33, IL-1, IL-6, and IL-8 [25–27]. But in vivo, the relationship between PM and the release of associated cytokines remains uncertain [28]. Our study revealed that the airway inflammatory response aggravated by PM intervention was accompanied by elevated expression of IL-33. IL-33 is expressed in the epithelial cells of the lungs and plays an important role in type-2 innate immunity by activating eosinophils, which are associated with allergic inflammation [29, 30]. Antibodies to IL-33 are evaluated in ongoing asthma-related clinical studies [31]. However, few studies have explored the role of IL-33 in HDM-induced asthma models under PM exposure. We confirmed that IL-33 neutralizing antibody could alleviate the aggravated airway inflammation induced by PM exposure in asthmatic mice. This verifies that the IL-33 antibody exerted a protective role on PM-aggravated HDM-induced airway inflammation. Nevertheless, there is no obvious alleviation of airway inflammation in the HDM-induced asthma mice treated with IL-33 antibody, which might because the airway inflammation induced by HDM had basically formed when the IL-33 antibody was administrated.
As stated, our study findings suggest that there is a positive correlation between the concentration of PM and the pediatric asthma visits, and the increase in risk was strongest in emergency visits, especially in children under 6 years old. Additionally, short-term exposure to PM aggravated the allergic airway inflammation, in which IL-33 might play an important role. Thus, these data provide insights into novel preventive and/or protective approaches for PM-induced airway disorders.
Materials and Methods
City-level estimates of environmental variables
In this research, we analyze the meteorological data in Hangzhou. The monthly average PM2.5 and PM10 concentrations from January 2013 to December 2017 were derived from 11 National Air Pollution Monitoring Stations located in Hangzhou. These monitoring points are distributed throughout the city, including urban and suburban areas, and can objectively and accurately reflect the concentration of atmospheric particles in this region.
Health and population outcome data
The study subjects were children suffering from asthma. Data on outpatient and emergency visits of asthma patients were obtained from the Children’s Hospital of Zhejiang University. The children’s hospital of Zhejiang University is the largest children’s hospital in Zhejiang province, with monthly outpatient and emergency visits reaching 20,000 or more. We collected the average monthly outpatient and emergency visits of asthmatic patients in children’s hospital and compared them with the total hospital visits. This data can reflect the incidence of asthma in the total patient population and asthma attacks. The study was approved by the ethics committee of the Children’s Hospital, Zhejiang University School of Medicine.
Animals
C57BL/6 mice (wild-type, aged 6–8 weeks) were purchased from the Slac Laboratory Animal Co. Ltd. (Shanghai, China). All mice were housed in the Laboratory Animal Center of Zhejiang University, Hangzhou, China. All experiments involving animals were maintained in a specific-pathogen-free facility and were given a standard diet with normal drinking water. The room temperature was precisely maintained at 23 ± 2° C, and the humidity was kept at 50% ± 10%. The study was in accordance with the stipulations and protocols approved by Ethics Committee for Animal Studies at Zhejiang University.
HDM-induced allergic airway inflammation in the mouse model
Freeze-dried HDM extract was purchased from Greer’s laboratory (10.52 EU/mg endotoxin) and dissolved in normal saline to a concentration of 2 μg/μL. The HDM-induced asthma model was established in mice at 6 weeks old. Mice in the HDM group were anesthetized and challenged with 50 μL of HDM (100 μg per mouse) via intratracheal instillation on days 0, 7, and 14. The control mice in the NS group were given NS. Mice were euthanized using a sodium pentobarbitone overdose intraperitoneally (i.p) at 72 h after the final allergen challenge on day 17.
PM intervention
PM was purchased from the National Institute of Standards and Technology, and the main components of PM were PAHs, nitro-substituted PAHs, polychlorinated biphenyl congeners, chlorinated pesticides, and inorganic constituents. Allergic airway inflammation was established in the HDM+PM group as previously described. After the last HDM administration on day 14, mice were given 50 μL of PM (100 μg per mouse) per day by intratracheal instillation on days 15 and 16. The control mice in the PM group were administered 50 μL of NS on days 0, 7, and 14, and PM (100 μg per mouse) on days 15 and 16 via intratracheal instillation. Mice were euthanized using a sodium pentobarbitone overdose intraperitoneally, and parameters were analyzed on day 17.
IL-33 neutralizing antibody intervention
Mice were divided into IL-33 antibody and control groups. In each group, NS, HDM, PM, and HDM+PM were treated according to the protocol described above, respectively. Anti-mouse IL-33 and the isotype control antibodies (R&D Systems) were resuspended in sterile PBS and administered three consecutive times from day 14 to 16 with 2 μg/mouse per day via intraperitoneal injection (i.p).
BALF collection and differential cell count
A cannula was inserted in the trachea, and then the left lungs were administered with 0.4 mL PBS three times for 1 mL BALF. Cells in BALF were counted under a microscope with a double-blind method. After cell count, BALF was centrifuged, spun onto glass slides, and stained with Wright-Giemsa. The completed slides were placed under a microscope to count the number of differential cells, and at least 200 cells were observed.
Histologic analysis
The left lung tissues were fixed with formaldehyde for 24 h and then embedded in paraffin. The sections were stained with H&E and PAS. H&E staining sections were assessed for the airway inflammatory situation and quantitated as previously described [32]. PAS staining sections were assessed for mucus production of goblet cells according to the previous studies [33]. All slides were examined in a randomly blinded fashion by two independent investigators.
Enzyme-linked immunosorbent assay (ELISA)
The lungs of each group were cut into small pieces, homogenized with RIPA lysis buffer, and centrifuged at 12,000 g for 15 min. Then, the supernatant was collected. IL-33 (Biolegend, CA, USA) and IL-25 (MultiScience, Germany) expression were quantified by ELISA kit according to the manufacturer’s protocol.
Statistical analyses
Statistical Package for the Social Sciences (SPSS) version 23.0 (SPSS, Chicago, IL, USA) was used for the clinical data analysis. PM2.5 and patient numbers as nonparametrically distributed data were expressed as medians and interquartile range. Accordingly, correlations among the selected variables were examined by Spearman rank correlation analysis.
Statistical analyses were performed with one-way ANOVA for the comparison of different groups in animal studies. Data were expressed as the means ± SEM. The analyses and graphs were performed with GraphPad Prism 7.0 software (GraphPad Software Inc., CA, USA). P-values < 0.05 were considered statistically significant.
Author Contributions
Zhimin Chen and Songmin Ying proposed this study and designed the experiments. Xin Yang, Yuanyuan Zhang, Xueqin Zhan and Xuchen Xu performed the experiments. Xin Yang, Shuxian Li, Xuefeng Xu analyzed and interpreted the data. Xin Yang, Xuchen Xu and Xueqin Zhan completed the manuscript. Zhimin Chen, Songmin Ying and Yuanyuan Zhang revised this manuscript.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This work was supported by the National Natural Science Foundation of China (No.81870023 and No.82000030).
References
- 1. Khaltaev N. GARD, a new way to battle with chronic respiratory diseases, from disease oriented programmes to global partnership. J Thorac Dis. 2017; 9:4676–89. https://doi.org/10.21037/jtd.2017.11.91 [PubMed]
- 2. Mukherjee A, Agrawal M. A Global Perspective of Fine Particulate Matter Pollution and Its Health Effects. Rev Environ Contam Toxicol. 2018; 244:5–51. https://doi.org/10.1007/398_2017_3 [PubMed]
- 3. Hao Y, Balluz L, Strosnider H, Wen XJ, Li C, Qualters JR. Ozone, Fine Particulate Matter, and Chronic Lower Respiratory Disease Mortality in the United States. Am J Respir Crit Care Med. 2015; 192:337–41. https://doi.org/10.1164/rccm.201410-1852OC [PubMed]
- 4. Stafoggia M, Forastiere F, Agostini D, Caranci N, de’Donato F, Demaria M, Michelozzi P, Miglio R, Rognoni M, Russo A, Perucci CA. Factors affecting in-hospital heat-related mortality: a multi-city case-crossover analysis. J Epidemiol Community Health. 2008; 62:209–15. https://doi.org/10.1136/jech.2007.060715 [PubMed]
- 5. D’Amato G, Holgate ST, Pawankar R, Ledford DK, Cecchi L, Al-Ahmad M, Al-Enezi F, Al-Muhsen S, Ansotegui I, Baena-Cagnani CE, Baker DJ, Bayram H, Bergmann KC, et al. Meteorological conditions, climate change, new emerging factors, and asthma and related allergic disorders. A statement of the World Allergy Organization. World Allergy Organ J. 2015; 8:25. https://doi.org/10.1186/s40413-015-0073-0 [PubMed]
- 6. Ayres JG, Forsberg B, Annesi-Maesano I, Dey R, Ebi KL, Helms PJ, Medina-Ramón M, Windt M, Forastiere F, and Environment and Health Committee of the European Respiratory Society. Climate change and respiratory disease: European Respiratory Society position statement. Eur Respir J. 2009; 34:295–302. https://doi.org/10.1183/09031936.00003409 [PubMed]
- 7. Cho CC, Hsieh WY, Tsai CH, Chen CY, Chang HF, Lin CS. In Vitro and In Vivo Experimental Studies of PM2.5 on Disease Progression. Int J Environ Res Public Health. 2018; 15:1380. https://doi.org/10.3390/ijerph15071380 [PubMed]
- 8. Kim Y, Seo J, Kim JY, Lee JY, Kim H, Kim BM. Characterization of PM2.5 and identification of transported secondary and biomass burning contribution in Seoul, Korea. Environ Sci Pollut Res Int. 2018; 25:4330–43. https://doi.org/10.1007/s11356-017-0772-x [PubMed]
- 9. Baulig A, Garlatti M, Bonvallot V, Marchand A, Barouki R, Marano F, Baeza-Squiban A. Involvement of reactive oxygen species in the metabolic pathways triggered by diesel exhaust particles in human airway epithelial cells. Am J Physiol Lung Cell Mol Physiol. 2003; 285:L671–79. https://doi.org/10.1152/ajplung.00419.2002 [PubMed]
- 10. Pun VC, Kazemiparkouhi F, Manjourides J, Suh HH. Long-Term PM2.5 Exposure and Respiratory, Cancer, and Cardiovascular Mortality in Older US Adults. Am J Epidemiol. 2017; 186:961–69. https://doi.org/10.1093/aje/kwx166 [PubMed]
- 11. Jacquemin B, Siroux V, Sanchez M, Carsin AE, Schikowski T, Adam M, Bellisario V, Buschka A, Bono R, Brunekreef B, Cai Y, Cirach M, Clavel-Chapelon F, et al. Ambient air pollution and adult asthma incidence in six European cohorts (ESCAPE). Environ Health Perspect. 2015; 123:613–21. https://doi.org/10.1289/ehp.1408206 [PubMed]
- 12. Kim H, Kim H, Park YH, Lee JT. Assessment of temporal variation for the risk of particulate matters on asthma hospitalization. Environ Res. 2017; 156:542–50. https://doi.org/10.1016/j.envres.2017.04.012 [PubMed]
- 13. Zhang J, Fulgar CC, Mar T, Young DE, Zhang Q, Bein KJ, Cui L, Castañeda A, Vogel CF, Sun X, Li W, Smiley-Jewell S, Zhang Z, Pinkerton KE. TH17-Induced Neutrophils Enhance the Pulmonary Allergic Response Following BALB/c Exposure to House Dust Mite Allergen and Fine Particulate Matter From California and China. Toxicol Sci. 2018; 164:627–43. https://doi.org/10.1093/toxsci/kfy127 [PubMed]
- 14. Yan LI, Gong C, Ying L, Fu W, Liu S, Dai J, Fu Z. PM2.5 affects establishment of immune tolerance in newborn mice by reducing PD-L1 expression. J Biosci. 2019; 44:41. [PubMed]
- 15. Timmerman T, de Brito JM, de Almeida NM, de Almeida FM, Arantes-Costa FM, Guimaraes ET, Lichtenfels AJ, Rivero DH, de Oliveira RC, de Lacerda JP, Moraes JM, Pimental DA, Saraiva-Romanholo BM, et al. Inflammatory and functional responses after (bio)diesel exhaust exposure in allergic sensitized mice. A comparison between diesel and biodiesel. Environ Pollut. 2019; 253:667–79. https://doi.org/10.1016/j.envpol.2019.06.085 [PubMed]
- 16. Atkinson RW, Kang S, Anderson HR, Mills IC, Walton HA. Epidemiological time series studies of PM2.5 and daily mortality and hospital admissions: a systematic review and meta-analysis. Thorax. 2014; 69:660–65. https://doi.org/10.1136/thoraxjnl-2013-204492 [PubMed]
- 17. Lu F, Xu D, Cheng Y, Dong S, Guo C, Jiang X, Zheng X. Systematic review and meta-analysis of the adverse health effects of ambient PM2.5 and PM10 pollution in the Chinese population. Environ Res. 2015; 136:196–204. https://doi.org/10.1016/j.envres.2014.06.029 [PubMed]
- 18. Fan J, Li S, Fan C, Bai Z, Yang K. The impact of PM2.5 on asthma emergency department visits: a systematic review and meta-analysis. Environ Sci Pollut Res Int. 2016; 23:843–50. https://doi.org/10.1007/s11356-015-5321-x [PubMed]
- 19. Slaughter JC, Lumley T, Sheppard L, Koenig JQ, Shapiro GG. Effects of ambient air pollution on symptom severity and medication use in children with asthma. Ann Allergy Asthma Immunol. 2003; 91:346–53. https://doi.org/10.1016/S1081-1206(10)61681-X [PubMed]
- 20. Kim YK, Kim SH, Tak YJ, Jee YK, Lee BJ, Kim SH, Park HW, Jung JW, Bahn JW, Chang YS, Choi DC, Chang SI, Min KU, et al. High prevalence of current asthma and active smoking effect among the elderly. Clin Exp Allergy. 2002; 32:1706–12. https://doi.org/10.1046/j.1365-2222.2002.01524.x [PubMed]
- 21. He M, Ichinose T, Yoshida Y, Arashidani K, Yoshida S, Takano H, Sun G, Shibamoto T. Urban PM2.5 exacerbates allergic inflammation in the murine lung via a TLR2/TLR4/MyD88-signaling pathway. Sci Rep. 2017; 7:11027. https://doi.org/10.1038/s41598-017-11471-y [PubMed]
- 22. Liu H, Fan X, Wang N, Zhang Y, Yu J. Exacerbating effects of PM2.5 in OVA-sensitized and challenged mice and the expression of TRPA1 and TRPV1 proteins in lungs. J Asthma. 2017; 54:807–17. https://doi.org/10.1080/02770903.2016.1266495 [PubMed]
- 23. Li R, Kou X, Xie L, Cheng F, Geng H. Effects of ambient PM2.5 on pathological injury, inflammation, oxidative stress, metabolic enzyme activity, and expression of c-fos and c-jun in lungs of rats. Environ Sci Pollut Res Int. 2015; 22:20167–76. https://doi.org/10.1007/s11356-015-5222-z [PubMed]
- 24. Falcon-Rodriguez CI, Osornio-Vargas AR, Sada-Ovalle I, Segura-Medina P. Aeroparticles, Composition, and Lung Diseases. Front Immunol. 2016; 7:3. https://doi.org/10.3389/fimmu.2016.00003 [PubMed]
- 25. Nakayama Wong LS, Aung HH, Lamé MW, Wegesser TC, Wilson DW. Fine particulate matter from urban ambient and wildfire sources from California’s San Joaquin Valley initiate differential inflammatory, oxidative stress, and xenobiotic responses in human bronchial epithelial cells. Toxicol In Vitro. 2011; 25:1895–905. https://doi.org/10.1016/j.tiv.2011.06.001 [PubMed]
- 26. Wang J, Huang J, Wang L, Chen C, Yang D, Jin M, Bai C, Song Y. Urban particulate matter triggers lung inflammation via the ROS-MAPK-NF-κB signaling pathway. J Thorac Dis. 2017; 9:4398–412. https://doi.org/10.21037/jtd.2017.09.135 [PubMed]
- 27. Kortam MA, Ali BM, Fathy N. The deleterious effect of stress-induced depression on rat liver: Protective role of resveratrol and dimethyl fumarate via inhibiting the MAPK/ERK/JNK pathway. J Biochem Mol Toxicol. 2021; 35:e22627. https://doi.org/10.1002/jbt.22627 [PubMed]
- 28. Zuo H, Qin J, Zhang W, Bashir MA, Yu Q, Zhao W, Wu G, Zhong F. Hemin-Catalyzed Oxidative Phenol-Hydrazone [3+3] Cycloaddition Enables Rapid Construction of 1,3,4-Oxadiazines. Org Lett. 2020; 22:6911–16. https://doi.org/10.1021/acs.orglett.0c02442 [PubMed]
- 29. Moussion C, Ortega N, Girard JP. The IL-1-like cytokine IL-33 is constitutively expressed in the nucleus of endothelial cells and epithelial cells in vivo: a novel ‘alarmin’? PLoS One. 2008; 3:e3331. https://doi.org/10.1371/journal.pone.0003331 [PubMed]
- 30. Chan BCL, Lam CWK, Tam LS, Wong CK. IL33: Roles in Allergic Inflammation and Therapeutic Perspectives. Front Immunol. 2019; 10:364. https://doi.org/10.3389/fimmu.2019.00364 [PubMed]
- 31. Corren J. New Targeted Therapies for Uncontrolled Asthma. J Allergy Clin Immunol Pract. 2019; 7:1394–403. https://doi.org/10.1016/j.jaip.2019.03.022 [PubMed]
- 32. Lee KS, Lee HK, Hayflick JS, Lee YC, Puri KD. Inhibition of phosphoinositide 3-kinase delta attenuates allergic airway inflammation and hyperresponsiveness in murine asthma model. FASEB J. 2006; 20:455–65. https://doi.org/10.1096/fj.05-5045com [PubMed]
- 33. Mikhak Z, Fukui M, Farsidjani A, Medoff BD, Tager AM, Luster AD. Contribution of CCR4 and CCR8 to antigen-specific T(H)2 cell trafficking in allergic pulmonary inflammation. J Allergy Clin Immunol. 2009; 123:67–73.e3. https://doi.org/10.1016/j.jaci.2008.09.049 [PubMed]



