Introduction
Liver cancer is among the most deadly tumors and the second leading cause of cancer-related mortality worldwide [1]. Hepatocellular carcinoma (HCC), the most common type of liver cancer, is often diagnosed at relatively advanced stages, such that patients miss the optimal time window for surgery [2]. Even after surgical resection and standardized treatment, the incidence of recurrence and distant metastasis in HCC remain high [3]. At advanced stages, there are few treatment options and the prognosis is poor. Clarifying the mechanism of HCC progression can identify novel diagnostic and prognostic biomarkers for improved clinical outcomes.
The regulation of epithelial–mesenchymal transition (EMT), a hallmark of cancer, is an important mechanism underlying HCC metastasis [4–6]. Tumor epithelial cells lose contact with each other and gain motility as a result of the dissolution of cell–cell junctions formed by E-cadherin, and spread to adjacent and distant tissues [7]. Transforming growth factor (TGF)-β is involved in both fibrogenesis and carcinogenesis through the regulation of EMT [7]. EMT markers such as E-cadherin, β-catenin, and mucin (MUC)15 related to vascular invasion and poor differentiation are aberrantly expressed in HCC patients [8] and are promising markers for HCC diagnosis and treatment.
Abnormal expression of long noncoding (lnc)RNAs has been observed in multiple types of cancer. Some lncRNAs participate in EMT through interaction with transcriptional regulators. For example, the lncRNA homeobox antisense intergenic RNA (HOTAIR) modulates the interaction between SNAIL and enhancer of zeste homolog (EZH)2 via a SNAIL/HOTAIR/EZH2 repressor complex that promotes EMT [9], while the lncRNA metastasis-associated lung adenocarcinoma transcript (MALAT)1 acts as an oncogene by interacting with EZH2 or functioning as a competing endogenous (ce)RNA for several members of the miR-200 micro (mi)RNA family that negatively regulate EMT-related factors [10]. Additionally, the lncRNA H19 regulates multiple genes involved in EMT progression by acting as a ceRNA [11].
EMT-associated gene models have been developed to predict HCC prognosis [4, 12]. However, the functions and prognostic value of EMT-related lncRNAs have not been systematically investigated. In this study, we developed an EMT-related lncRNA signature for predicting HCC prognosis using data from The Cancer Genome Atlas (TCGA) database. We validated the prognostic value of the signature and investigated its relationship with immune cell infiltration- and immune checkpoint-related factors. Finally, we explored the role of 5 hub lncRNAs in HCC and the function of one of these (LINC01116) in vitro and in vivo. Our results demonstrate for the first time that LINC01116 acts as an immune-related oncogene in HCC and has clinical utility as a prognostic biomarker.
Materials and Methods
Data collection and processing
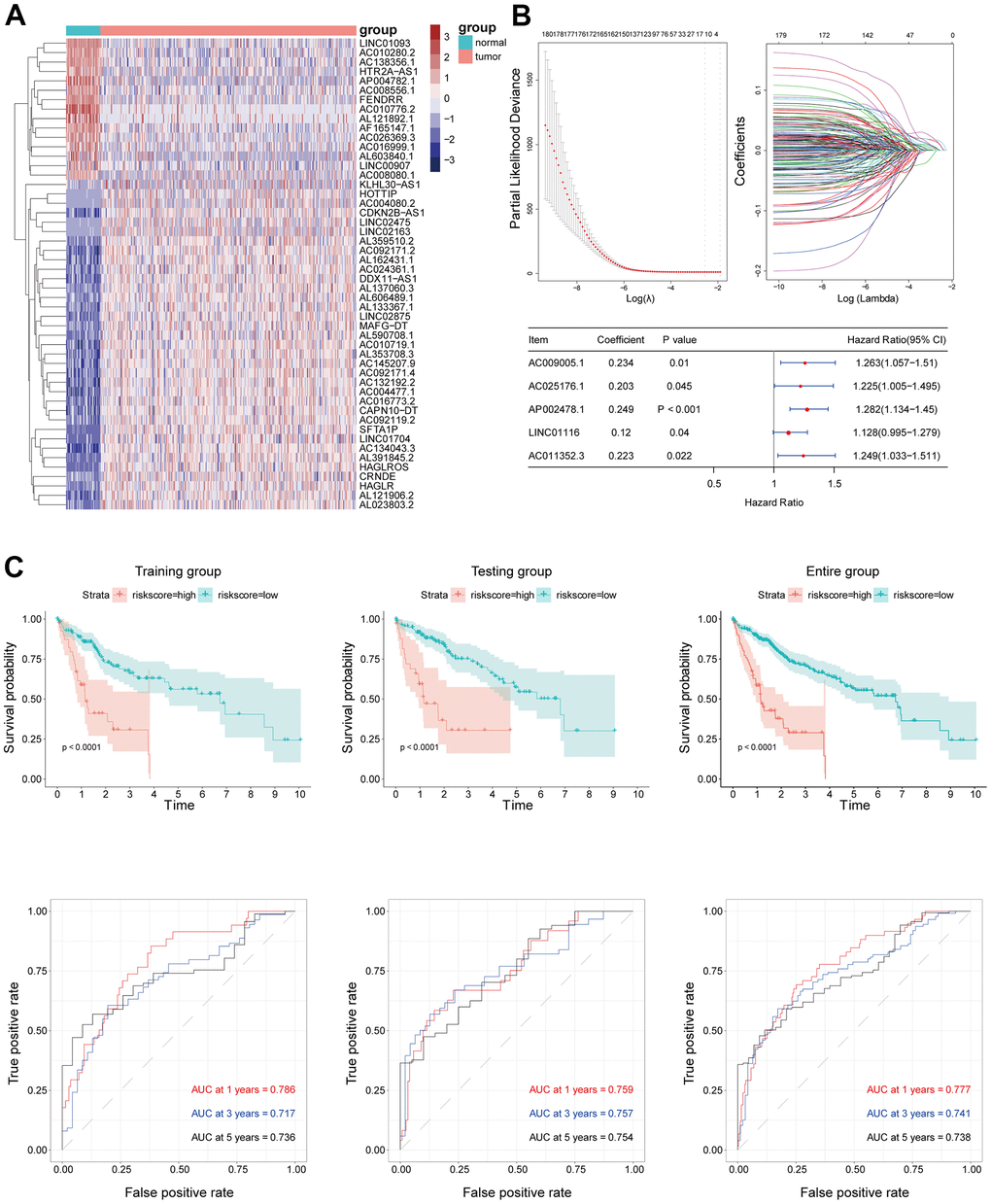
Gene expression data of 424 samples including 374 tumor and 50 normal samples and clinical information of 374 HCC patients were downloaded from TCGA database. LncRNAs and mRNAs were identified from RNA sequence data based on annotations in the human GENCODE database. The “edgeR” package in R software was used to identify differentially expressed lncRNAs with the threshold P value <0.05 and |log(fold change)|>2. Additionally, the data of 368 HCC patients with complete clinical information were randomly divided into training and validation sets at a 1:1 ratio. The clinical characteristics of the 2 groups were similar.
Analysis of the relationship between the prognostic signature, immune cell infiltration, and immune checkpoint targets
The CIBERSORT algorithm was used to analyze the abundance of 22 types of tumor-infiltrating immune cell (TIIC) and Tumor Immune Estimation Resource (TIMER) was used to calculate the abundance of 6 different types of TIIC. The association between the lncRNA signature and the different types of TIIC was analyzed to determine whether the signature can predict immune cell infiltration in HCC.
The success of programmed death (PD)-1/programmed death ligand (PD-L)1 and cytotoxic T-lymphocyte antigen (CTLA)-4 blockade for HCC treatment has intensified research efforts to identify novel immune checkpoint targets. In this study, we examined established targets as well as those with therapeutic potential such as T-cell immunoglobulin and mucin domain-containing (TIM)-3, indoleamine 2,3-dioxygenase (IDO)1, glucocorticoid-induced tumor necrosis factor receptor-related (GITR), histone deacetylase (HDAC)2, B7-histone (H)3, and V-domain Ig suppressor of T cell activation (VISTA) [13–15]. In order to evaluate the role of the lncRNA signature in the treatment response to immune checkpoint blockade (ICB), we performed a correlation analysis between the signature and the expression level of 10 immune checkpoint targets in HCC.
Tissue specimens and cell culture
We collected 50 paired HCC tissue and adjacent nontumor tissue samples from patients who underwent hepatectomy between March 2016 and October 2017 at the Hepatic Surgery Center of Tongji Hospital. OS information was collected through electronic medical records or telephone follow-up. Informed consent forms to donate their tissue samples for biomedical research, which were approved by the ethics committee of Tongji Hospital, were signed by all patients. Huh7 and SK-Hep1 HCC cell lines were purchased from the Liver Cancer Institute of Fudan University (Shanghai, China). The cells were cultured in Dulbecco’s Modified Eagle’s Medium supplemented with 10% fetal bovine serum at 37° C and 5% CO2.
Immunofluorescence analysis
HCC cells were cultured on coverslips in 24-well plates for 24 h. After 3 washes with phosphate-buffered saline, the cells were fixed with 4% paraformaldehyde for 15 min and permeabilized with 0.1% Triton X-100 for 10 min. After culturing in medium containing 1% bovine serum albumin for 2 h, the cells were incubated overnight at 4° C with rabbit anti–E-cadherin and mouse anti-vimentin antibodies and then labeled with Alexa Fluor 568- or 488-conjugated secondary antibody (from different species) for 2 h. Cell nuclei were stained with 4′, 6-diamidino-2-phenylindole for 10 min. The coverslips were mounted and observed with a confocal microscope.
Fluorescence in situ hybridization (FISH)
Cells were cultured on coverslips in 24-well plates. LINC01116 was detected in HCC cells by FISH using a probe and commercial kit from RiboBio (Guangzhou, China) according to the manufacturer’s instructions. The labeled cells were observed with a confocal microscope.
Xenograft tumor model
Male BALB/c nude mice (4 weeks old, weighing 15 g) were maintained under specific pathogen-free conditions. Huh7 cells transfected with empty vector or LINC01116 overexpression plasmid (3×106) were subcutaneously injected into the back of the mice on the right and left sides, respectively. Tumor growth was observed every 3 days and tumors were harvested after 3 weeks.
Immunohistochemistry
Tumor tissues from mice were fixed in 4% paraformaldehyde solution and then dehydrated in ethanol solutions, embedded in paraffin, and cut into sections that were incubated overnight at 4° C with primary antibodies against Ki67, vimentin, and E-cadherin followed by horseradish peroxidase-conjugated secondary antibody for 1 h at room temperature. Images of representative fields were acquired with a C1 Digital ECLIPSE microscope.
Cell proliferation and migration assays and cell cycle analysis
The cell proliferation (Cell Counting Kit [CCK]-8), 5-ethynyl-2′-deoxyuridine (EdU), transwell migration, and wound healing assays and cell cycle analysis were carried out as previously described [16].
Statistics analysis
Data analysis were conducted using R software (version 3.6.3) and Prism 7.0 software. Data are presented as mean ± SD. Qualitative variables were analyzed by Student's t test, one-way ANOVA and Pearson correlation test were used to analyze qualitative variables. P<0.05 was considered to be statistically significant.
Data availability statement
Publicly available datasets were analyzed in this study. The data are accessible at the following repositories: https://portal.gdc.cancer.gov/repository.
Ethics statement
This research protocol was approved by the ethical committee of Tongji Hospital and a consent form was signed by each participating patient.
Results
Validation of the prognostic lncRNA signature
The mortality rate in all groups increased with risk score; meanwhile, the expression levels of the 5 hub lncRNAs in the EMT-related signature also increased (Figure 3A). A multivariate Cox regression analysis that also included other clinical characteristics of HCC patients showed that risk score was an independent prognostic factor in HCC, with a hazard ratio of 1.690 (1.204−2.372) in the training set, 1.503 (1.159−1.948) in the validation set, and 1.446 (1.229−1.702) in the whole patient population (Figure 3B). The risk score had a higher AUC than the other clinical features in each group, confirming its value as an accurate prognostic factor (Figure 3B).
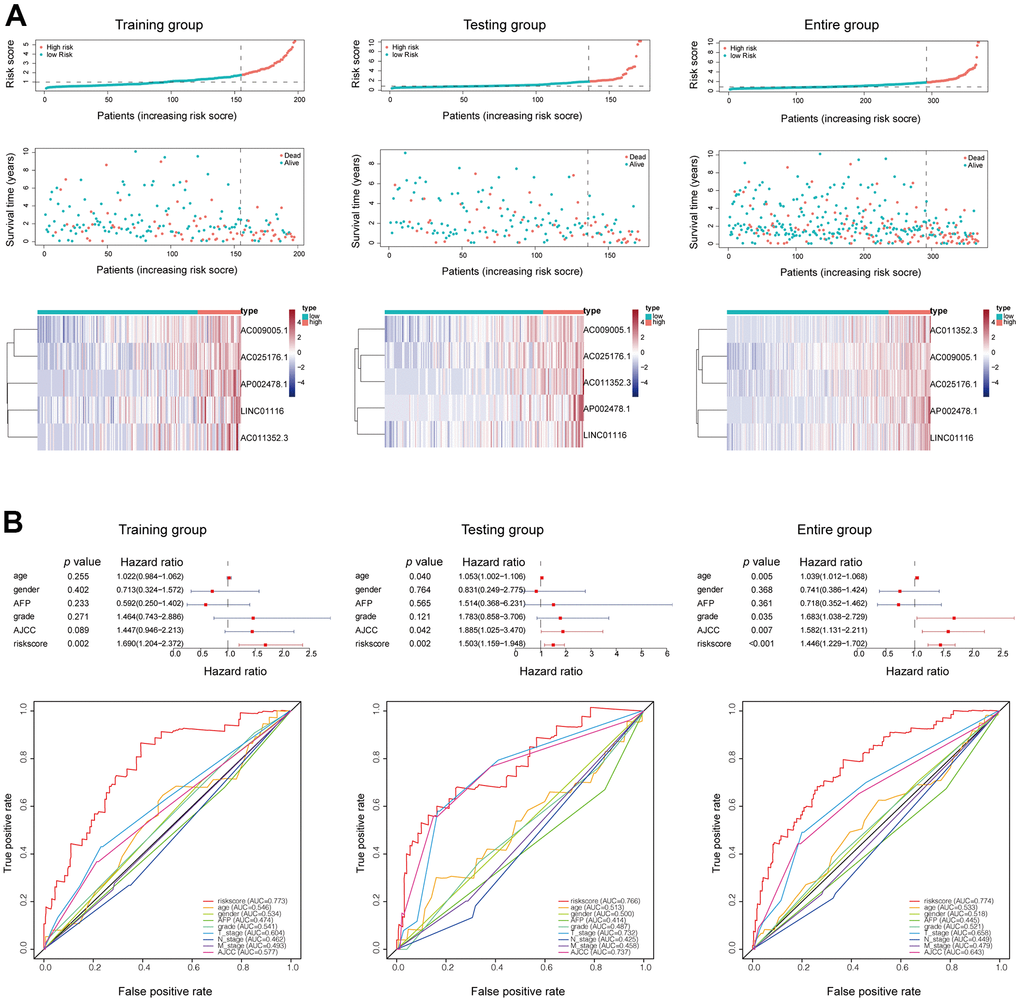
Figure 3. (A) Mortality status and lncRNAs expression in each patient were plotted according to the ordered risk score. (B) Multivariate and ROC curves confirmed the EMT-related lncRNA signature as an independent prognostic factor for HCC.
To investigate the robustness of the prognostic lncRNA signature, we evaluated its predictive accuracy in the Tongji cohort including 50 HCC patients. Consistent with the results from the TCGA cohort, that HCC patients with a high risk score had a shorter OS than those with a low risk score (Figure 4A). In addition, the prognostic lncRNA signature had an AUC of 0.754 at 1 year, 0.723 at 3 years, and 0.703 at 5 years in the Tongji cohort (Figure 4B), indicating an excellent predictive accuracy. Then, we analyzed the relationships between the EMT-related lncRNA signature and clinicopathologic characteristics of HCC patients. The risk score was significantly higher in patients with vascular invasion and those who died (Figure 4C, 4D). We also observed that the risk score tended to increase with tumor grade and stage (Figure 4E, 4F), implying that the signature reflects the high malignancy of HCC.
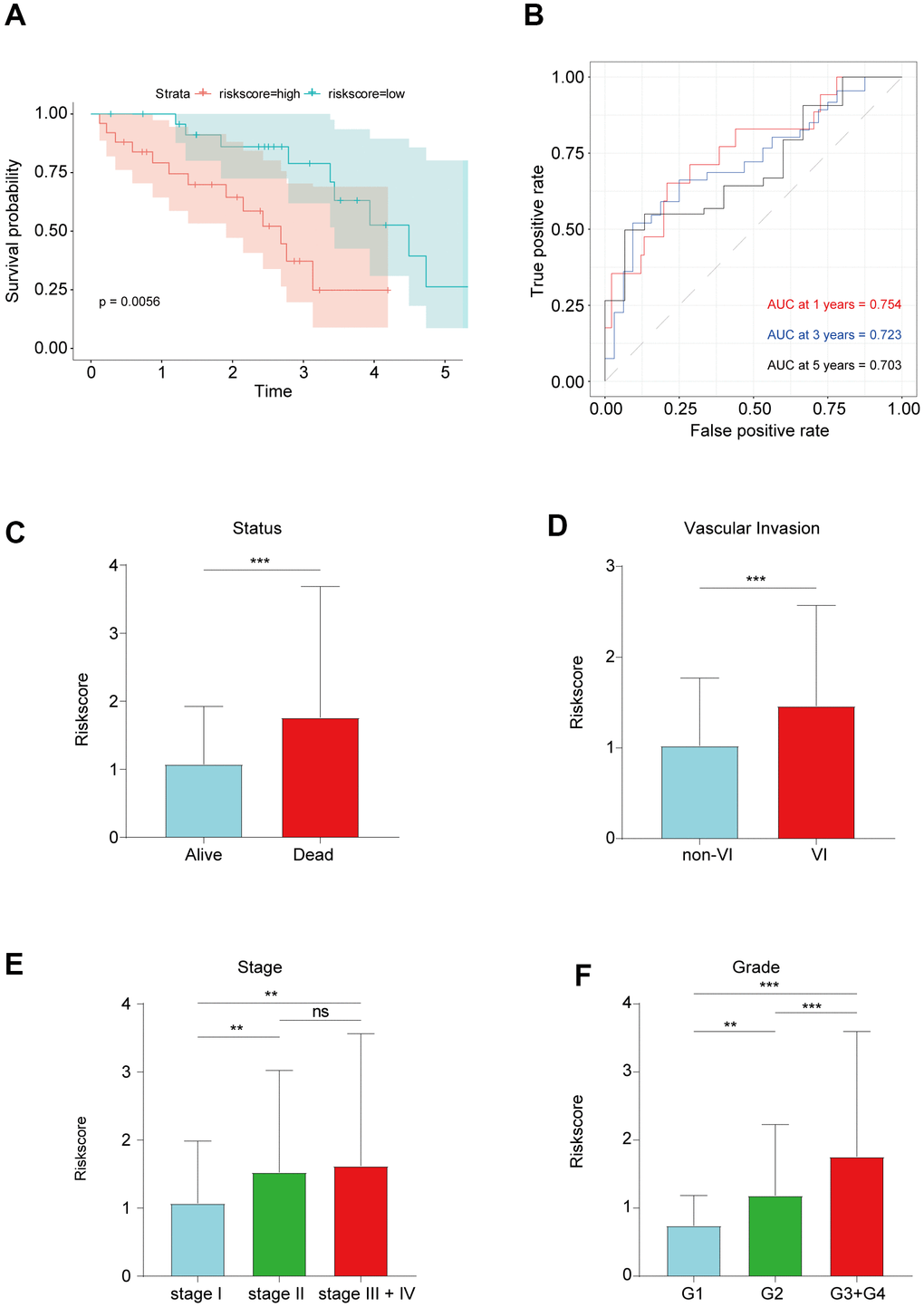
Figure 4. (A) Kaplan–Meier survival analysis in Tongji cohort. (B) ROC curve showed good accuracy of this signature in Tongji cohort. (C) The association between risk score and living status. (D) The association between risk score and vascular invasion. (E) The association between risk score and tumor grade. (F) The association between risk score and tumor stage. **P < 0.01 and ***P < 0.001.
Functional analysis of the prognostic signature
We performed a principal component analysis to compare low and high-risk patients in the training and validation sets and the whole patient population who were classified based on the EMT-related lncRNA signature. The low- and high-risk groups formed distinct clusters, confirming the prognostic accuracy of the signature (Figure 5A). We also explored the function of lncRNAs in the signature by performing a correlation analysis with mRNAs that were differentially expressed between high- and low-risk groups, whose functions were predicted by Gene Ontology and Kyoto Encyclopedia of Genes and Genomes pathway analyses. It showed that function annotations of this signature was enriched in terms related to signal release, intrinsic component of synaptic membrane, hormone activity and neuroactive ligand−receptor interaction (Figure 5B, 5C).
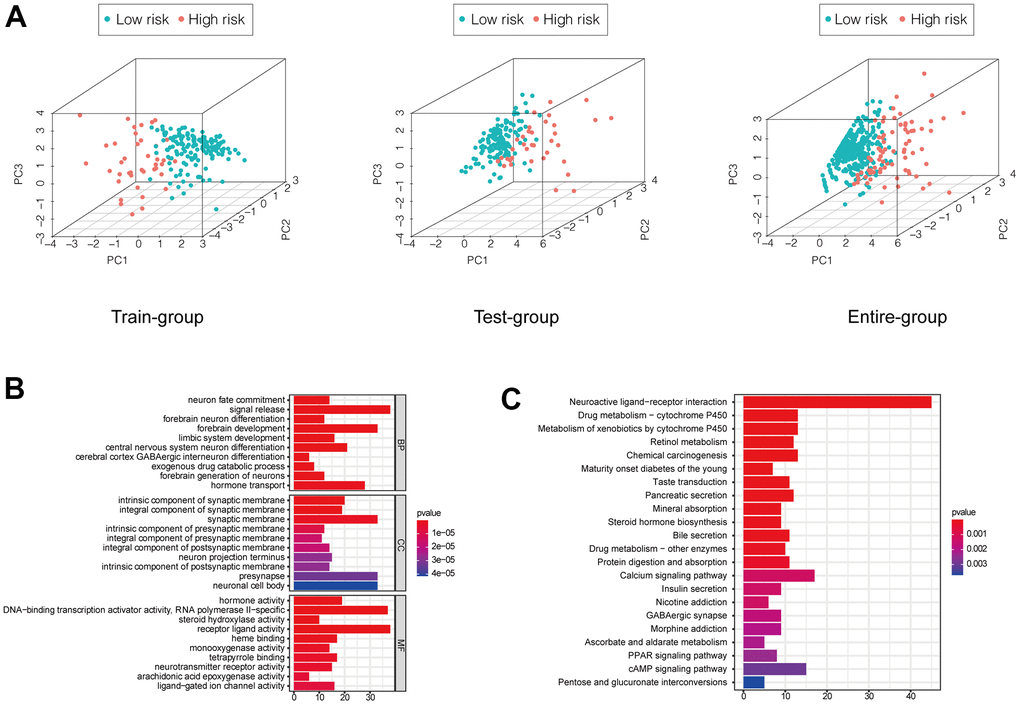
Figure 5. (A) PCA showed a scattered distribution of patients in each group. (B, C) GO and KEGG analysis of DEGs between high and low-risk groups of the entire group.
Correlation between the prognostic signature, immune cell infiltration, and immune checkpoint targets
Using the CIBERSORT algorithm, we investigated whether the EMT-related lncRNA signature was related to immune cell infiltration in HCC. The results of the correlation analysis revealed a significant correlation with infiltrating resting memory cluster of differentiation (CD)4+ T cells (r=−0.154; p=3.08e−03), regulatory T cells (r=0.197; p=1.46e−04), monocytes (r=-0.149; p=4.27e−03), macrophages M0 (r=0.265; p=2.53e−07), resting mast cells (r=−0.163; p=1.69e−03), and eosinophils (r=0.171; p=9.73e−04; Figure 6A). Additionally, all lncRNAs in the signature showed a significant correlation with the infiltration of multiple immune cell types.
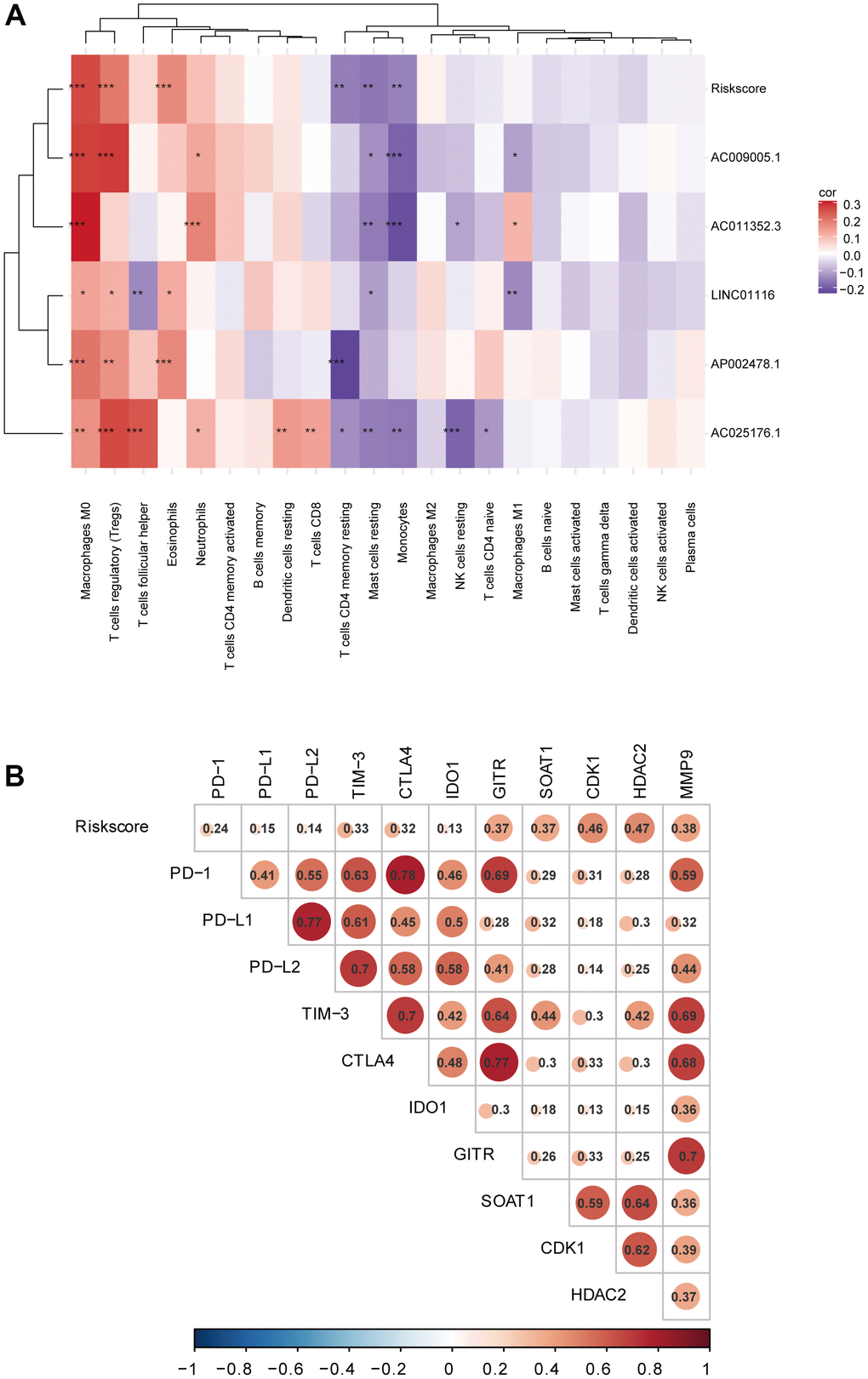
Figure 6. (A) Correlation analyses of the EMT-related lncRNA signature with immune cell infiltration. (B) Correlation analyses of the EMT-related lncRNA signature with immune checkpoint targets.
To investigate the potential role of the EMT-related lncRNA signature in ICB therapy for HCC, we performed a correlation analysis between risk score and 10 immune checkpoint targets (CTLA4, TIM-3, PD-L1, PD-L2, PD-1, IDO1, GITR, HDAC2, B7-H3, and VISTA) and found close correlations (Figure 6B), suggesting that the signature can predict the outcome of ICB therapy in HCC patients.
Hub lncRNAs in HCC
Using TCGA data, the expression levels of the 5 hub lncRNA s in this signature were compared in the HCC tissues and normal liver tissues. It showed that all the 5 hub lncRNAs expression levels were higher in HCC tissues than these in the normal liver tissues. Besides, the Kaplan–Meier analysis indicated that the increased the 5 hub lncRNAs were significantly correlated with OS of HCC patients (Supplementary Figure 1). The increased levels of the 5 hub lncRNAs in HCC was confirmed in HCC specimens from the Tongji Hospital cohort, and the Kaplan–Meier analysis with the log-rank test indicated a significant correlation with OS (Figure 7A). Additionally, 3 of the lncRNAs (AC025176.1, LINC01116, and AC011352.3) were highly expressed in 4 HCC cell lines compared to a normal liver cell line (Figure 7B). Of these, LINC01116 was selected for further analysis based on the fact that it had the highest relative expression in clinical HCC specimens and has not been previously characterized in HCC.
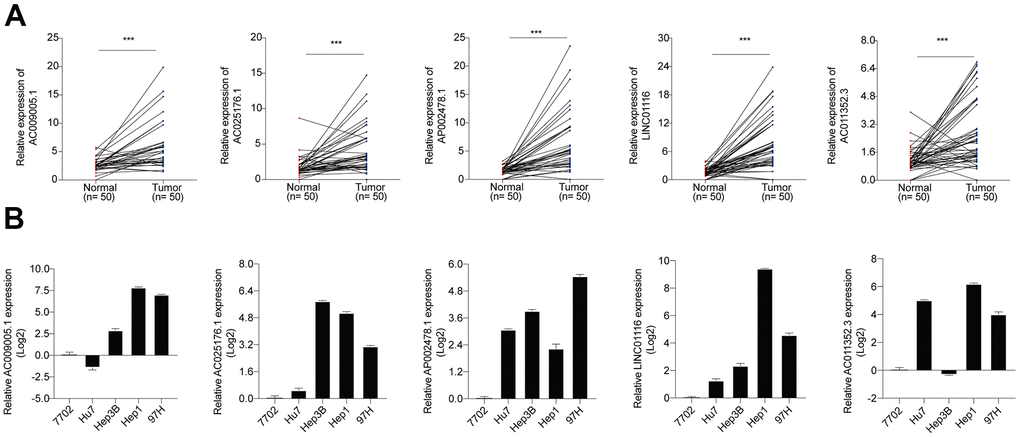
Figure 7. (A) The expression levels of 5 hub lncRNAs in HCC specimens of Tongji hospital. (B) The expression levels of 5 hub lncRNAs in normal liver cell line and 4 HCC cell lines.
LINC01116 enhances HCC cell proliferation and regulates the cell cycle
To determine the subcellular localization of LINC01116 in HCC cells, we performed cytoplasmic/nuclear fractionation and FISH in SK-Hep1 and Huh7 cells. LINC01116 was detected at a higher level in the cytoplasm than in the nucleus, implying that it mainly acts on cytoplasmic factors (Figure 8A, 8B).
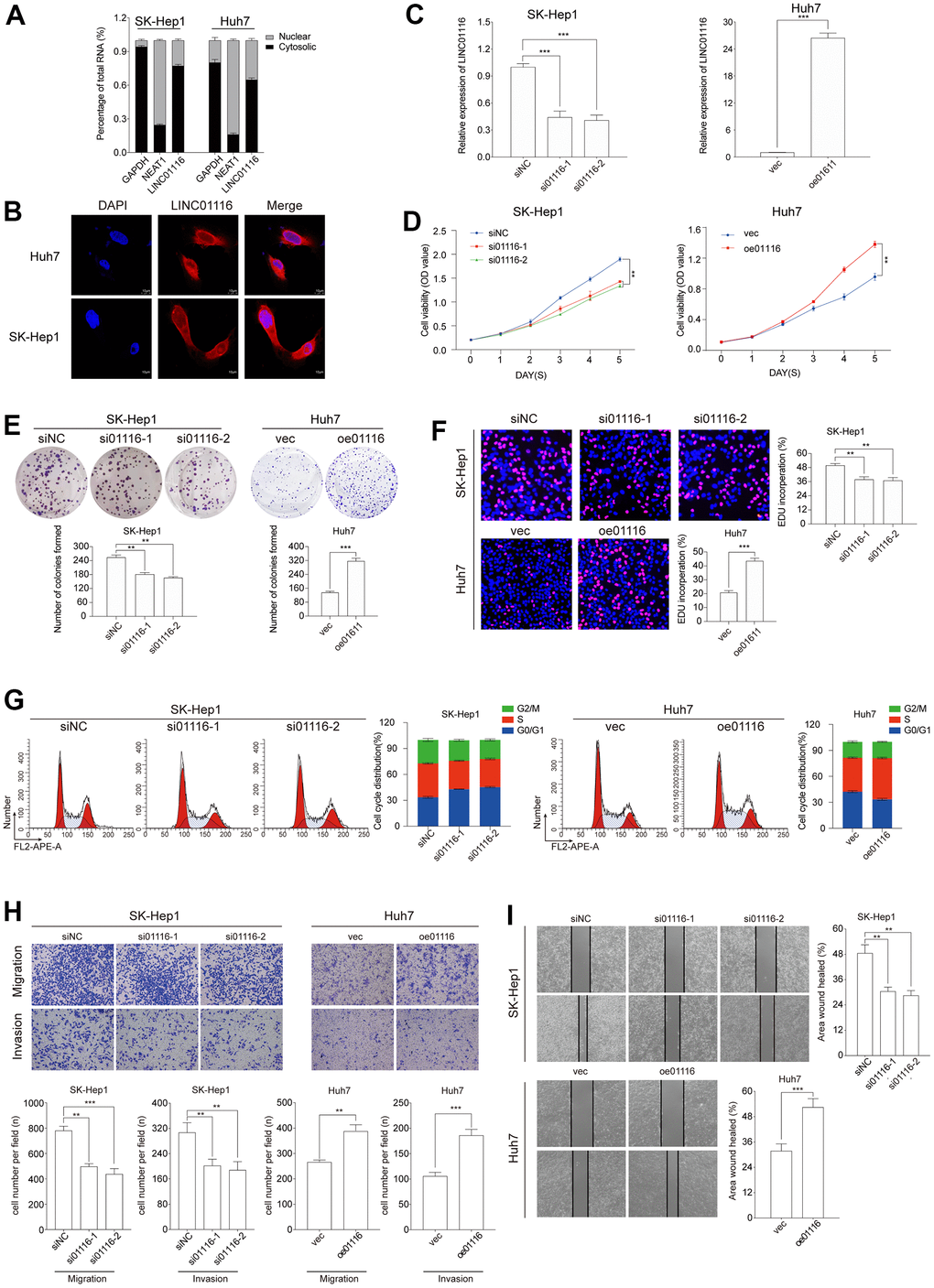
Figure 8. (A) The expression level of LINC01116 in the subcellular fractions of HCC cells was detected by qRT-PCR. (B) FISH assay analysis for the location of LINC01116 in HCC cells. (C) Transfection efficiency was verified by qRT-PCR. (D–F) Cell viability was evaluated with CCK-8, EdU and colony formation assays in HCC cells. (G) Cell cycle was examined by the flow cytometry. (H) Transwell assays were used to detect HCC cells invasion and migration. (I) Migration ability was evaluated by wound healing assay. **P < 0.01 and ***P < 0.001.
To investigate the function of LINC01116 in HCC, we used 2 different small interfering (si)RNAs to silence its expression in SK-Hep1 cells. Conversely, LINC01116 expression was markedly increased in Huh7 cells transfected with LINC01116 overexpression plasmid (Figure 8C).
The effect of LINC01116 loss or gain of function on HCC cell proliferation and viability was evaluated with the CCK-8, EdU, and colony formation assays. The results showed that LINC01116 knockdown suppressed the proliferation of SK-Hep1 cells while LINC01116 overexpression enhanced Huh7 cell proliferation (Figure 8D–8F). We also examined cell cycle distribution under the 2 conditions by flow cytometry. LINC01116 silencing increased the proportion of SK-Hep1 cells arrested in G0/G1 phase whereas LINC01116 overexpression induced cell cycle progression in Huh7 cells (Figure 8G). In contrast, HCC cell apoptosis was unaffected by LINC01116 depletion and overexpression (Supplementary Figure 2).
LINC01116 promotes EMT and metastasis in HCC
To investigate the role of LINC01116 in the migration and invasion of HCC cells, we performed transwell migration and wound healing assays. LINC01116 knockdown suppressed migration and invasion in SK-Hep1 cells, while its overexpression had the opposite effect in Huh7 cells (Figure 8H). In the wound-healing assay, migration distance was decreased in SK-Hep1 cells transfected with siRNAs and was increased in Huh7 cells overexpressing LINC01116 (Figure 8I). We also examined the expression of the EMT markers vimentin, E-cadherin, and N-cadherin by western blotting and immunofluorescence analysis. LINC01116 knockdown in SK-Hep1 cells decreased the levels of vimentin and N-cadherin and increased that of E-cadherin, with LINC01116 overexpression have the opposite effects in Huh7 cells (Figure 9A, 9B).
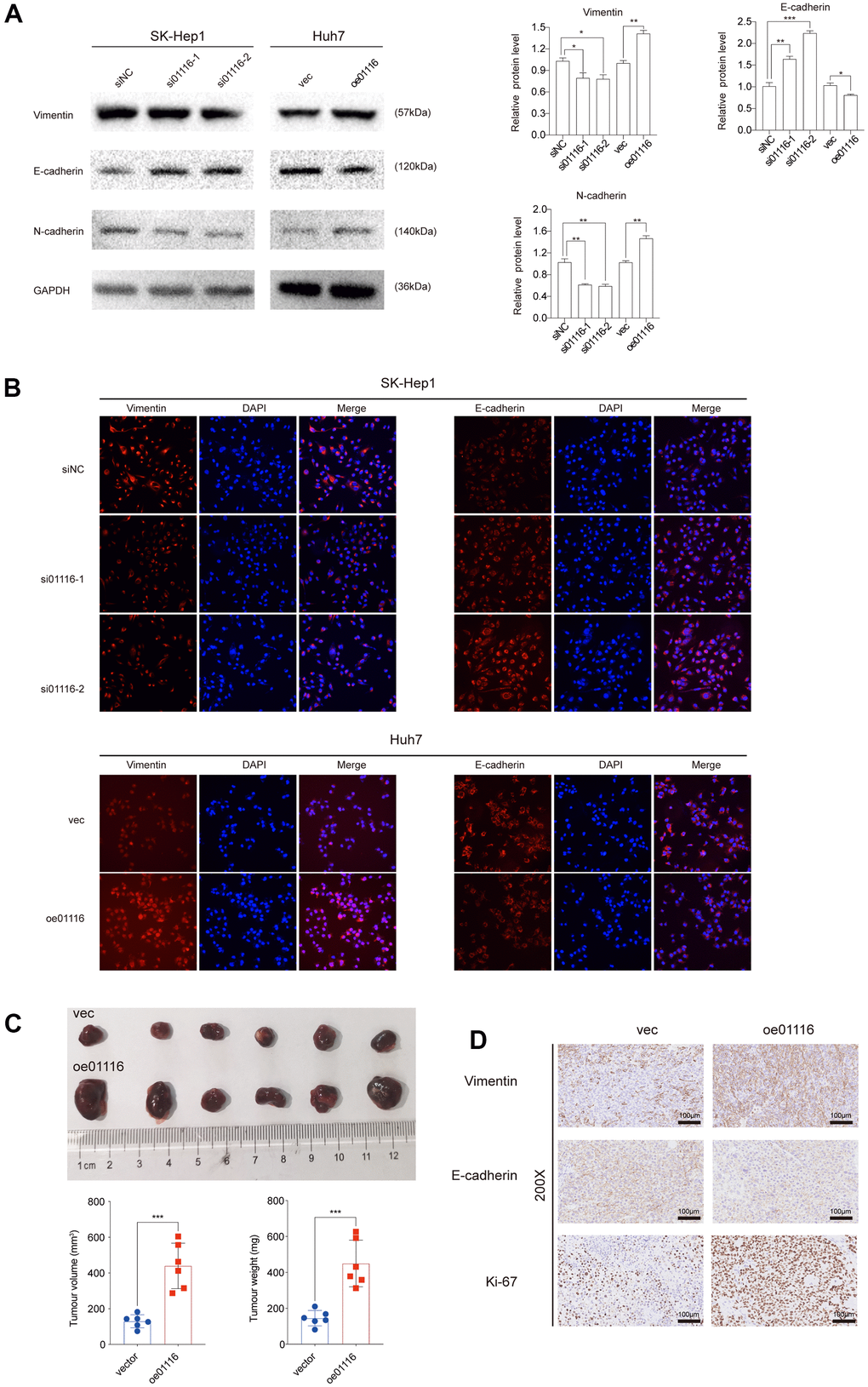
Figure 9. (A) EMT markers were examined via western blot analysis. (B) Immunofluorescence assay was used to detect EMT markers. (C) Image of subcutaneous tumor tissues. The volume and weight of tumors were measured. (D) Ki67, vimentin and E-cadherin were observed in subcutaneous tumor tissues by IHC. **P < 0.01 and ***P < 0.001.
To verify the above results in vivo we used a mouse xenograft model. Compared to the control group, Huh7 cells overexpressing LINC01116 formed larger tumors (Figure 9C); moreover, these tumors showed upregulation of Ki67 and vimentin and downregulation of E-cadherin by immunohistochemistry (Figure 9D). Collectively, these results indicate that LINC01116 promotes EMT and metastasis in HCC.
LINC01116 is associated with immune cell infiltration and expression of immune checkpoint targets
As described above, LINC01116 expression was found to be significantly correlated with the infiltration of 6 immune cell types based on the CIBERSORT algorithm (Figure 6A). We further examined the relationship between LINC01116 and TIICs using TIMER. LINC01116 expression level was positively and significantly correlated with B cell, CD4+ T cell, CD8+ T cell, macrophage, neutrophil, and dendritic cell infiltration (Figure 10A). To evaluate the influence of LINC01116 on ICB therapy in HCC, we performed a correlation analysis between LINC01116 and immune checkpoint targets (CTLA4, TIM-3, PD-L1, PD-L2, PD-1, IDO1, GITR, HDAC2, B7-H3, and VISTA) and found positive correlations with the levels of all 10 targets (Figure 10B). Thus, LINC01116 plays a critical role in immune regulation in HCC and is a potential target for ICB therapy.
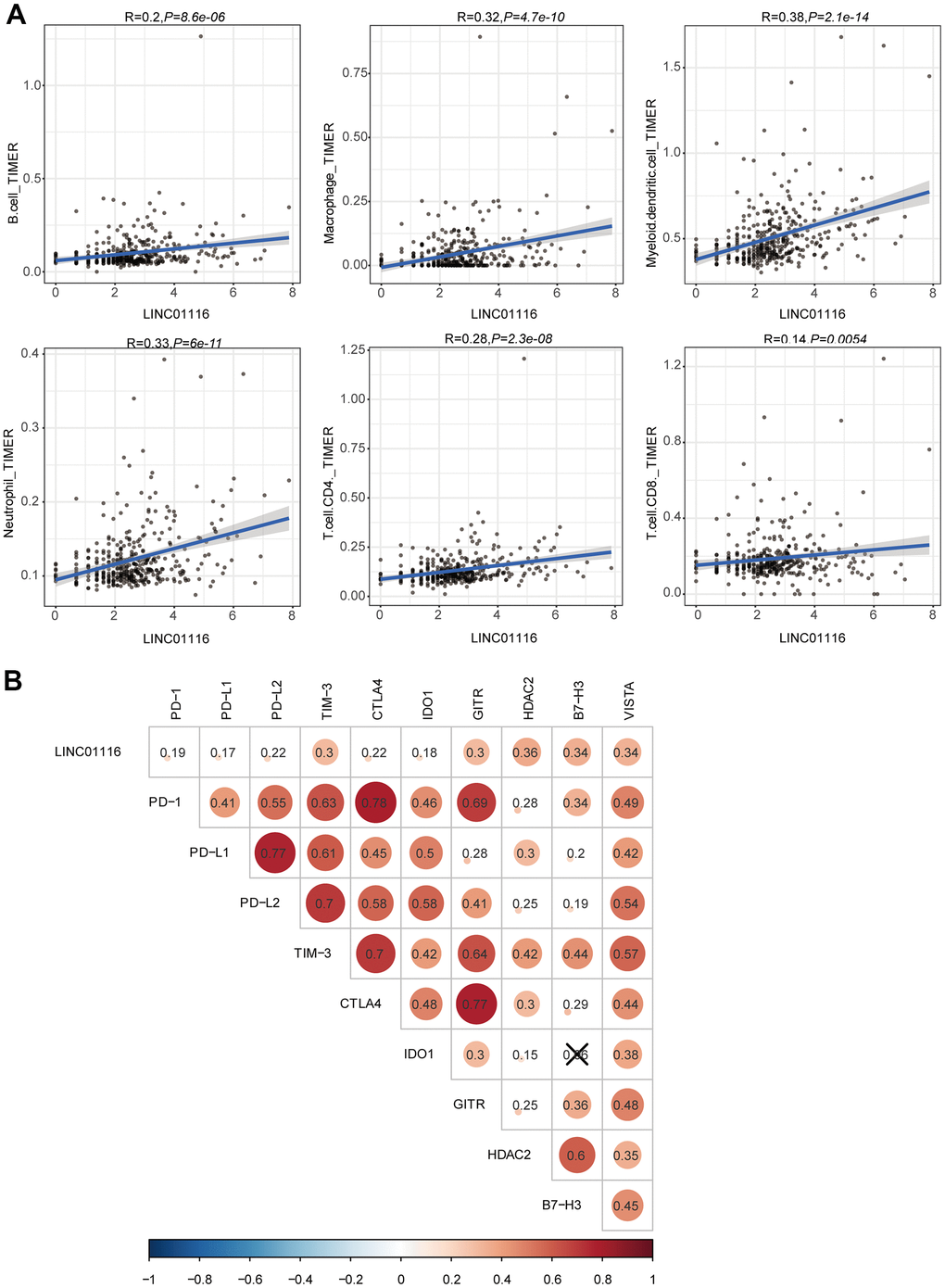
Figure 10. (A) Correlation analyses of LINC01116 with immune cell infiltration based on TIMER database. (B) Correlation analyses of LINC01116 with immune checkpoint targets.
Discussion
EMT plays a critical role in cancer pathogenesis and metastasis [17]. Some lncRNAs are known to interact with and modulate the expression and functions of master regulators of EMT [18, 19]. Therefore, identifying EMT-related lncRNAs and elucidating their mechanisms of action in HCC can reveal novel therapeutic targets for precision medicine and thereby improve the prognosis of patients.
In the present study, EMT-related lncRNAs were screened and uni- and multivariate Cox and LASSO analyses were carried out to establish an EMT-related lncRNA signature. The prognostic value of the signature in HCC was evaluated by Kaplan–Meier, multivariate Cox, and ROC curve analyses. Patients with a high risk score were found to have more malignant HCC and significantly worse prognosis. The AUCs indicated that the signature could effectively predict the OS of HCC patients at 1, 3, and 5 years in different risk groups. Thus, the EMT-related lncRNA signature can guide risk stratification and clinical decision-making in HCC management.
EMT plays important roles in immune regulation and treatment response in cancer [20]. For example, tumor-associated macrophages and myeloid-derived suppressor cells in the tumor microenvironment induce EMT to facilitate immune evasion [21, 22]. EMT creates an immunosuppressive tumor microenvironment that contributes to low therapeutic response and resistance to anti–PD-L1 therapy [23]. Furthermore, some lncRNAs were shown to be involved in both immune evasion and EMT [24, 25]. Thus, EMT-related lncRNAs can potentially modulate the behavior of infiltrating immune cells and response to immunotherapy. This was supported by the fact that the EMT-related lncRNA signature established in our study was significantly associated with the degree of infiltration of multiple immune cell types. Although ICB therapy has achieved good results in various solid tumors, its efficacy in HCC is low [26, 27]. Enhancing the precision of ICB therapy in HCC by identifying more specific drug targets and biomarkers for predicting treatment response can improve clinical outcomes. The correlation analysis in the present work showed that the EMT-related lncRNA signature was significantly associated with all 10 immune checkpoint targets examined; moreover, the risk score of the signature increased with the expression levels of these targets. These results suggest that the EMT-related lncRNA signature can predict the responsiveness of HCC patients to ICB treatment.
Three hub lncRNAs (AC009005.1, AP002478.1, and LINC01116) in the EMT-related lncRNA signature are known to contribute to the malignant phenotype of various cancers, while AC025176.1 and AC011352.3 have not been previously described. AC009005.1 is an autophagy-related lncRNA that has been used to construct a prognostic model for HCC; it was shown to be more highly expressed in tumors than in normal liver tissue, which was associated with shorter OS of HCC patients [28]. AP002478.1 has also been incorporated into several prognostic models for HCC based on its upregulation in HCC tissues and association with poor outcome. Additionally, AP002478.1 was shown to be overexpressed in 4 HCC cell lines compared to the normal hepatocyte cell line LO2, suggesting an involvement in the development of HCC [29, 30]. LINC01116 has been linked to various cancers. LINC01116 activated interleukin (IL)-1β expression in glioma by regulating the transcriptional regulator DEAD-box helicase (DDX)5, thereby promoting neutrophil recruitment and glioma cell proliferation [31]. LINC01116 also modulated gefitinib resistance in non-small cell lung cancer (NSCLC) cells by regulating interferon-induced protein (IFI)44 expression, suggesting a potential mechanism for drug resistance in NSCLC [32]. Through competitive binding of the miRNA miR-31-5p, LINC01116 increased the expression of the proangiogenic factor vascular endothelial growth factor (VEGF) to promote glioma tumorigenesis [33].
All 5 hub lncRNAs in the EMT-related lncRNA signature were highly expressed in HCC tissues of the Tongji Hospital cohort, which is consistent with the TCGA data. Given that it had the highest expression in HCC specimens compared to nontumor tissue in the Tongji Hospital cohort and has not been previously characterized in HCC, LINC01116 was selected for more detailed study. We found that LINC01116 enhanced proliferation, cell cycle progression, and migration in HCC cells in vitro, indicating that it can promote EMT and metastasis in HCC. The results of in vivo experiments confirmed that overexpression of LINC01116 increased tumor cell proliferation and induced the expression of EMT-related factors. We also found that LINC01116 was related not only to immune cell infiltration but to the expression of 10 immune checkpoint targets. Taken together, our findings indicate that LINC01116 functions as an oncogene in HCC that participates in both EMT and immune regulation.
Conclusions
In the present study we established an EMT-related lncRNA signature that can be used to predict the prognosis of HCC patients. We provided evidence that the signature is directly related to a high degree of malignancy in HCC and is closely associated with immune cell infiltration and multiple immune checkpoint targets. LINC01116, a hub gene in the signature, was identified as an immune-related oncogene in HCC and potential target for HCC immunotherapy. These findings can help to improve the management of HCC patients to achieve better clinical outcomes.
Supplementary Materials
Author Contributions
Haisu Tao: Conceptualization, Methodology, Software, Writing - review and editing. Yuxin Zhang: Investigation, Formal analysis. Tong Yuan: Investigation, Software. Jiang Li: Formal analysis. Junjie Liu: Formal analysis. Yixiao Xiong: Validation. Jinghan Zhu: Visualization. Ping Wang: Validation. Huifang Liang: Supervision. Erlei Zhang: Conceptualization, Writing - review and editing, Funding acquisition. Zhiyong Huang: Supervision.
Acknowledgments
We would like to thank The Analysis and Testing Center of Institute of Hydrobiology, Chinese Academy of Sciences for Guangxin Wang.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
This project was supported by the Science and Technology Planning Project of Hubei Province (No.2021BCA115) and the National Natural Science Foundation of China (No. 81902839).
Editorial Note
This corresponding author has a verified history of publications using a personal email address for correspondence
References
- 1. Kulik L, El-Serag HB. Epidemiology and Management of Hepatocellular Carcinoma. Gastroenterology. 2019; 156:477–91.e1. https://doi.org/10.1053/j.gastro.2018.08.065 [PubMed]
- 2. Pinto Marques H, Gomes da Silva S, De Martin E, Agopian VG, Martins PN. Emerging biomarkers in HCC patients: Current status. Int J Surg. 2020; 82:70–6. https://doi.org/10.1016/j.ijsu.2020.04.043 [PubMed]
- 3. Ruf B, Heinrich B, Greten TF. Immunobiology and immunotherapy of HCC: spotlight on innate and innate-like immune cells. Cell Mol Immunol. 2021; 18:112–27. https://doi.org/10.1038/s41423-020-00572-w [PubMed]
- 4. Wang X, Xing Z, Xu H, Yang H, Xing T. Development and validation of epithelial mesenchymal transition-related prognostic model for hepatocellular carcinoma. Aging (Albany NY). 2021; 13:13822–45. https://doi.org/10.18632/aging.202976 [PubMed]
- 5. Bakir B, Chiarella AM, Pitarresi JR, Rustgi AK. EMT, MET, Plasticity, and Tumor Metastasis. Trends Cell Biol. 2020; 30:764–76. https://doi.org/10.1016/j.tcb.2020.07.003 [PubMed]
- 6. Hasegawa T, Yashiro M, Nishii T, Matsuoka J, Fuyuhiro Y, Morisaki T, Fukuoka T, Shimizu K, Shimizu T, Miwa A, Hirakawa K. Cancer-associated fibroblasts might sustain the stemness of scirrhous gastric cancer cells via transforming growth factor-β signaling. Int J Cancer. 2014; 134:1785–95. https://doi.org/10.1002/ijc.28520 [PubMed]
- 7. Giannelli G, Koudelkova P, Dituri F, Mikulits W. Role of epithelial to mesenchymal transition in hepatocellular carcinoma. J Hepatol. 2016; 65:798–808. https://doi.org/10.1016/j.jhep.2016.05.007 [PubMed]
- 8. Wang RY, Chen L, Chen HY, Hu L, Li L, Sun HY, Jiang F, Zhao J, Liu GM, Tang J, Chen CY, Yang YC, Chang YX, et al. MUC15 inhibits dimerization of EGFR and PI3K-AKT signaling and is associated with aggressive hepatocellular carcinomas in patients. Gastroenterology. 2013; 145:1436–48.e1. https://doi.org/10.1053/j.gastro.2013.08.009 [PubMed]
- 9. Battistelli C, Cicchini C, Santangelo L, Tramontano A, Grassi L, Gonzalez FJ, de Nonno V, Grassi G, Amicone L, Tripodi M. The Snail repressor recruits EZH2 to specific genomic sites through the enrollment of the lncRNA HOTAIR in epithelial-to-mesenchymal transition. Oncogene. 2017; 36:942–55. https://doi.org/10.1038/onc.2016.260 [PubMed]
- 10. Hirata H, Hinoda Y, Shahryari V, Deng G, Nakajima K, Tabatabai ZL, Ishii N, Dahiya R. Long Noncoding RNA MALAT1 Promotes Aggressive Renal Cell Carcinoma through Ezh2 and Interacts with miR-205. Cancer Res. 2015; 75:1322–31. https://doi.org/10.1158/0008-5472.CAN-14-2931 [PubMed]
- 11. Liang WC, Fu WM, Wong CW, Wang Y, Wang WM, Hu GX, Zhang L, Xiao LJ, Wan DC, Zhang JF, Waye MM. The lncRNA H19 promotes epithelial to mesenchymal transition by functioning as miRNA sponges in colorectal cancer. Oncotarget. 2015; 6:22513–25. https://doi.org/10.18632/oncotarget.4154 [PubMed]
- 12. Lei Y, Yan W, Lin Z, Liu J, Tian D, Han P. Comprehensive analysis of partial epithelial mesenchymal transition-related genes in hepatocellular carcinoma. J Cell Mol Med. 2021; 25:448–62. https://doi.org/10.1111/jcmm.16099 [PubMed]
- 13. Qin S, Xu L, Yi M, Yu S, Wu K, Luo S. Novel immune checkpoint targets: moving beyond PD-1 and CTLA-4. Mol Cancer. 2019; 18:155. https://doi.org/10.1186/s12943-019-1091-2 [PubMed]
- 14. Ceeraz S, Nowak EC, Noelle RJ. B7 family checkpoint regulators in immune regulation and disease. Trends Immunol. 2013; 34:556–63. https://doi.org/10.1016/j.it.2013.07.003 [PubMed]
- 15. Le Mercier I, Chen W, Lines JL, Day M, Li J, Sergent P, Noelle RJ, Wang L. VISTA Regulates the Development of Protective Antitumor Immunity. Cancer Res. 2014; 74:1933–44. https://doi.org/10.1158/0008-5472.CAN-13-1506 [PubMed]
- 16. Zhang L, Tao H, Li J, Zhang E, Liang H, Zhang B. Comprehensive analysis of the competing endogenous circRNA-lncRNA-miRNA-mRNA network and identification of a novel potential biomarker for hepatocellular carcinoma. Aging (Albany NY). 2021; 13:15990–6008. https://doi.org/10.18632/aging.203056 [PubMed]
- 17. Singh M, Yelle N, Venugopal C, Singh SK. EMT: Mechanisms and therapeutic implications. Pharmacol Ther. 2018; 182:80–94. https://doi.org/10.1016/j.pharmthera.2017.08.009 [PubMed]
- 18. Peng PH, Chieh-Yu Lai J, Hsu KW, Wu KJ. Hypoxia-induced lncRNA RP11-390F4.3 promotes epithelial-mesenchymal transition (EMT) and metastasis through upregulating EMT regulators. Cancer Lett. 2020; 483:35–45. https://doi.org/10.1016/j.canlet.2020.04.014 [PubMed]
- 19. Gong J, Wang Y, Shu C. LncRNA CHRF promotes cell invasion and migration via EMT in gastric cancer. Eur Rev Med Pharmacol Sci. 2020; 24:1168–76. https://doi.org/10.26355/eurrev_202002_20168 [PubMed]
- 20. Terry S, Savagner P, Ortiz-Cuaran S, Mahjoubi L, Saintigny P, Thiery JP, Chouaib S. New insights into the role of EMT in tumor immune escape. Mol Oncol. 2017; 11:824–46. https://doi.org/10.1002/1878-0261.12093 [PubMed]
- 21. Mosser DM, Edwards JP. Exploring the full spectrum of macrophage activation. Nat Rev Immunol. 2008; 8:958–69. https://doi.org/10.1038/nri2448 [PubMed]
- 22. Ouzounova M, Lee E, Piranlioglu R, El Andaloussi A, Kolhe R, Demirci MF, Marasco D, Asm I, Chadli A, Hassan KA, Thangaraju M, Zhou G, Arbab AS, et al. Monocytic and granulocytic myeloid derived suppressor cells differentially regulate spatiotemporal tumour plasticity during metastatic cascade. Nat Commun. 2017; 8:14979. https://doi.org/10.1038/ncomms14979 [PubMed]
- 23. Jiang Y, Zhan H. Communication between EMT and PD-L1 signaling: New insights into tumor immune evasion. Cancer Lett. 2020; 468:72–81. https://doi.org/10.1016/j.canlet.2019.10.013 [PubMed]
- 24. Ye Y, Xu Y, Lai Y, He W, Li Y, Wang R, Luo X, Chen R, Chen T. Long non-coding RNA cox-2 prevents immune evasion and metastasis of hepatocellular carcinoma by altering M1/M2 macrophage polarization. J Cell Biochem. 2018; 119:2951–63. https://doi.org/10.1002/jcb.26509 [PubMed]
- 25. Zhao L, Liu Y, Zhang J, Liu Y, Qi Q. LncRNA SNHG14/miR-5590-3p/ZEB1 positive feedback loop promoted diffuse large B cell lymphoma progression and immune evasion through regulating PD-1/PD-L1 checkpoint. Cell Death Dis. 2019; 10:731. https://doi.org/10.1038/s41419-019-1886-5 [PubMed]
- 26. Zhu AX, Finn RS, Edeline J, Cattan S, Ogasawara S, Palmer D, Verslype C, Zagonel V, Fartoux L, Vogel A, Sarker D, Verset G, Chan SL, et al, and KEYNOTE-224 investigators. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): a non-randomised, open-label phase 2 trial. Lancet Oncol. 2018; 19:940–52. https://doi.org/10.1016/S1470-2045(18)30351-6 [PubMed]
- 27. El-Khoueiry AB, Sangro B, Yau T, Crocenzi TS, Kudo M, Hsu C, Kim TY, Choo SP, Trojan J, Welling TH Rd, Meyer T, Kang YK, Yeo W, et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet. 2017; 389:2492–502. https://doi.org/10.1016/S0140-6736(17)31046-2 [PubMed]
- 28. Wu H, Liu T, Qi J, Qin C, Zhu Q. Four Autophagy-Related lncRNAs Predict the Prognosis of HCC through Coexpression and ceRNA Mechanism. Biomed Res Int. 2020; 2020:3801748. https://doi.org/10.1155/2020/3801748 [PubMed]
- 29. Liao X, Wang X, Huang K, Han C, Deng J, Yu T, Yang C, Huang R, Liu X, Yu L, Zhu G, Su H, Qin W, et al. Integrated analysis of competing endogenous RNA network revealing potential prognostic biomarkers of hepatocellular carcinoma. J Cancer. 2019; 10:3267–83. https://doi.org/10.7150/jca.29986 [PubMed]
- 30. Long J, Bai Y, Yang X, Lin J, Yang X, Wang D, He L, Zheng Y, Zhao H. Construction and comprehensive analysis of a ceRNA network to reveal potential prognostic biomarkers for hepatocellular carcinoma. Cancer Cell Int. 2019; 19:90. https://doi.org/10.1186/s12935-019-0817-y [PubMed]
- 31. Wang T, Cao L, Dong X, Wu F, De W, Huang L, Wan Q. LINC01116 promotes tumor proliferation and neutrophil recruitment via DDX5-mediated regulation of IL-1β in glioma cell. Cell Death Dis. 2020; 11:302. https://doi.org/10.1038/s41419-020-2506-0 [PubMed]
- 32. Wang H, Lu B, Ren S, Wu F, Wang X, Yan C, Wang Z. Long Noncoding RNA LINC01116 Contributes to Gefitinib Resistance in Non-small Cell Lung Cancer through Regulating IFI44. Mol Ther Nucleic Acids. 2020; 19:218–27. https://doi.org/10.1016/j.omtn.2019.10.039 [PubMed]
- 33. Ye J, Zhu J, Chen H, Qian J, Zhang L, Wan Z, Chen F, Sun S, Li W, Luo C. A novel lncRNA-LINC01116 regulates tumorigenesis of glioma by targeting VEGFA. Int J Cancer. 2020; 146:248–61. https://doi.org/10.1002/ijc.32483 [PubMed]