Introduction
Ovarian cancer (OC) is one of the main causes of female death. 70% of patients are in the advanced stage (clinical stage III ~ IV), and peritoneal metastasis is the characteristic metastasis symptom of patients with advanced OC. Although the incidence rate is not high, the mortality rate is 2.9%, which is the top ten of female malignant tumor mortality. For OC, the current standard treatment is not optimistic [1]. One reason is that most patients are diagnosed only in the late stage of the disease. Standard treatment for OC usually includes surgery and platinum chemotherapy. Despite available treatments, the recurrence rate of OC is still high, and about 75% of advanced patients are incurable [2, 3]. Genomics research shows that there is no regulated gene mutation, and many clinical trials have failed to bring lasting clinical benefits [4]. Molecular typing of OC through molecular expression differences, exploring the microenvironment characteristics of different molecular subtypes of OC, and formulating accurate treatment may be a feasible strategy to improve the dilemma of OC treatment.
Since the discovery of lin-4 and let-7, the founding members of the microRNA (miRNA) family, hundreds of miRNAs have been found in viruses, plants, and animals by molecular cloning and bioinformatics methods [5]. There are more than 1000 known human miRNAs, which control more than 50% of mammalian protein-coding genes. MiRNAs can be overexpressed or inhibited in different diseases. It is a promising therapeutic research field to inhibit or replace microRNAs [6]. Although microRNA has only 20 nucleotides, it plays a key role in biological function by targeting plenty of mRNAs [7]. For example, one study found that miRNA-424-5p regulated ferroptosis by targeting Acyl-CoA Synthetase Long-Chain Family Member 4 in OC cells and indicated a potential therapeutic target for OC [8]. Wu et al. found that the downregulated microRNA-1301-3p inhabited lung carcinoma cell proliferation and migration and has a strong negative correlation with Polymerase I and transcript release factor [9]. Another study also indicated the expression level of miR-200-b control PD-L1 expression in lung carcinoma cells. Furthermore, it may function as a potential biomarker for PD-L1 expression in lung cancer patients [10]. These studies indicated that microRNAs may be related to the progression of cancer, and their mechanisms may take part in the pathogenesis of tumors via controlling cancer-associated genes.
In this article, DEGs and DEMs were selected by researching 1 OC mRNA microarray dataset and 1 miRNA dataset. We aimed to study the relationship between miR-142-3p, FAM83D (we identified), and the development of OC.
Results
Identification of DEGs
We used GEO2R to analysis the DEMs and DEGs from the GSE81873 and GSE27651. Based on the cut-off criteria, 56 DEMs such as hsa-microRNA-142-3p, hsa-microRNA-429, hsa-microRNA-199a-3p, hsa-microRNA-484, hsa-microRNA-139-5p, hsa-microRNA-483-5p, hsa-microRNA-423-5p, hsa-microRNA-342-3p, hsa-microRNA-885-5p, and 63 DEGs were selected (Figure 1).
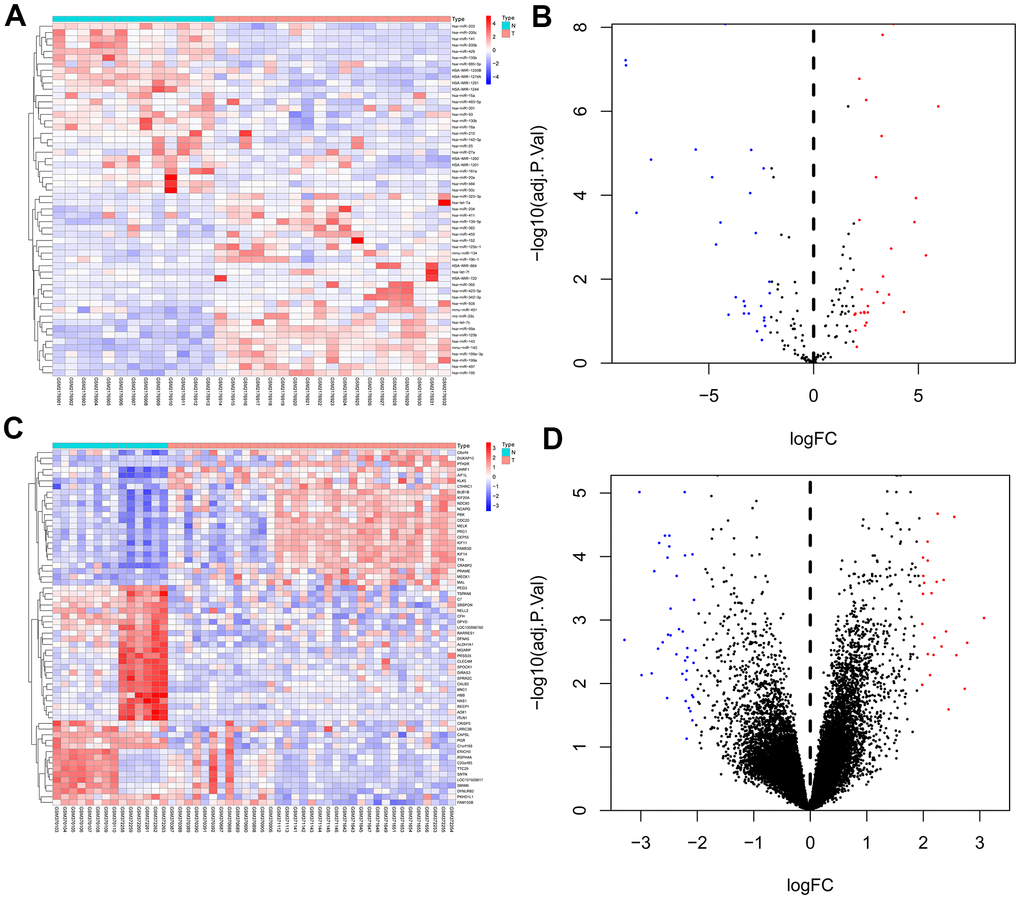
Figure 1. Heat map and volcano map of differentially expressed genes of GSE81873 and GSE27651. (A). Heat map of DEGs in GSE81873. (B). Volcano map of DEGs in GSE81873. (C). Heat map of DEGs in GSE27651. (D). Volcano map of DEGs in GSE27651. Red dots represent up-regulated genes and blue or green dots represent down-regulated genes.
Gene ontology enrichment analysis
By utilizing FunRich and R software, we performed TF enrichment analysis (Figure 2A).
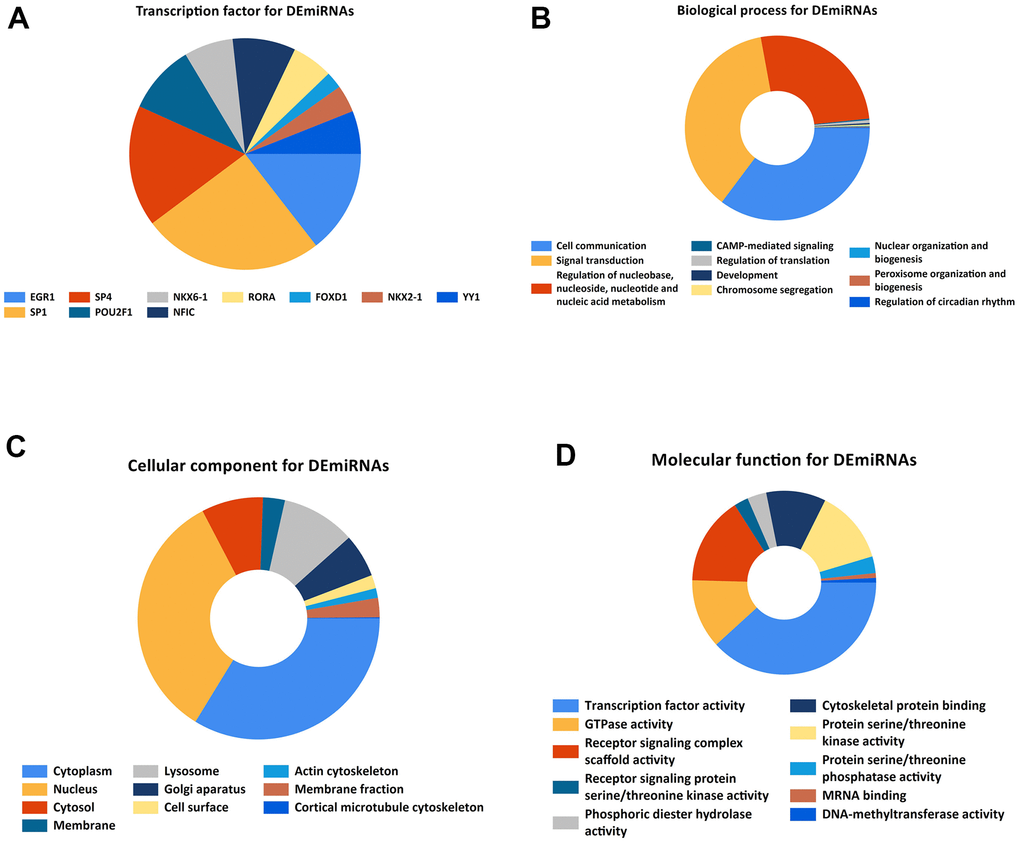
Figure 2. Gene ontology enrichment. (A). Identification of the potential transcription factors of DEMs by FunRich software. (B) biological process, (C) cellular component, and (D) molecular function enrichment analysis of the DEMs.
KEGG and GO function enrichment analyses were performed here. As for GO function analysis, three GO were selected: molecular function (MF), cellular component (CC), and biological process 156 (BP). Expression analysis showed that DEGs had the most uniquely enriched terms for Cell communication, Signal transduction, Regulation of nucleobase, nucleotide and nucleic acid metabolism, Cytoplasm, Nucleus, Cytosol, Membrane, Transcription factor activity, GTPase activity, Receptor signaling complex scaffold activity, Receptor signaling protein serine/threonine kinase activity, and CAMP-mediated signaling (Figure 2B–2D). Besides, DEMs were mainly enriched in 6 pathways: MAPK signaling pathway, PI3K-Akt signaling pathway, Wnt signaling pathway, TGF-beta signaling pathway, Pathways in cancer, and Bacterial invasion of epithelial cells (Figure 3A).
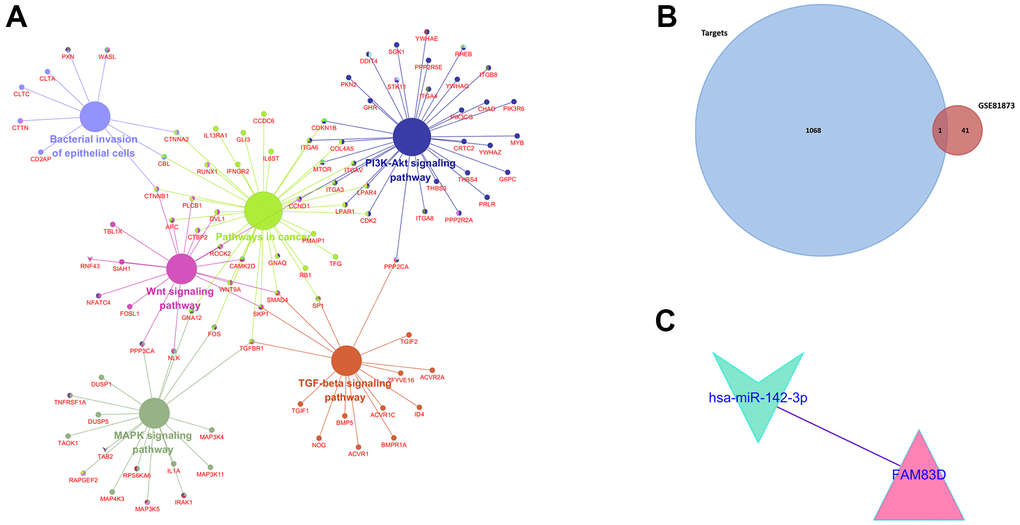
Figure 3. (A). KEGG pathway enrichment analysis of potential target mRNAs. (B). Venn Diagram of GSE81873 and GSE27651. (C). Identified target mRNAs and miRNA-mRNA regulatory network.
miRNA-mRNA regulatory network
According to FunRich software, 1069 potential target mRNAs were obtained and only 1 of them showed different expression levels in GSE27651 (FAM83D). Based on the association between them, 1 essential microRNA-mRNA pair (microRNA-142-3p and FAM83D) was selected for the next research (Figure 3B, 3C).
genes expression and their associations with OC overall survival
KM Plot was used to analyze the overall survival of patients with OC. According to uploading the miRNA and target gene we identified, we downloaded survival curves. The pictures showed that microRNA-124-3p and FAM83D were associated with the overall survival of patients with OC (Figure 4).
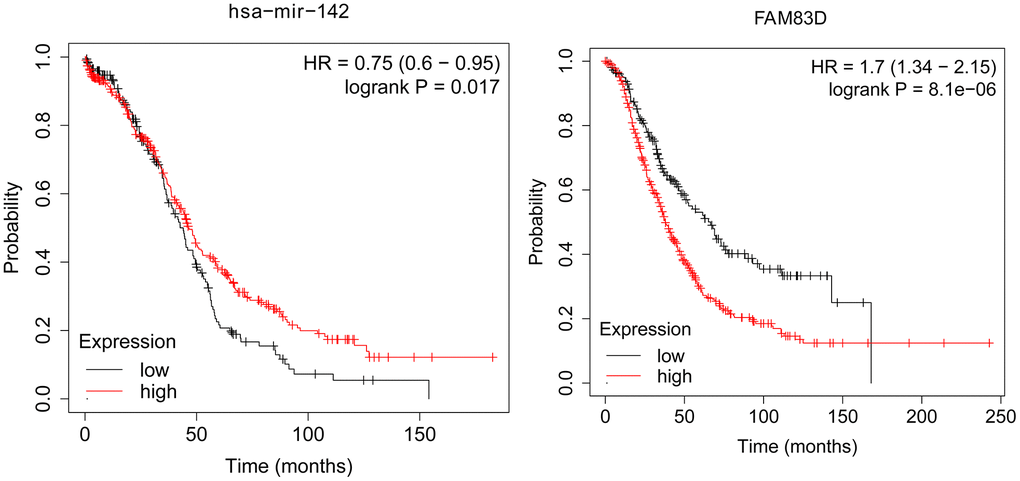
Figure 4. The association between the expression level of selected genes and overall survival of OC patients.
Relationship between clinical characteristics and genes expression level of OC
Clinical and gene expression information of OC patients were downloaded in the TCGA database, such as patients’ age, race, FIGO stage, and primary therapy outcome (Tables 1, 2). FAM83D and miR-142-3p expression of patients with Stage III and Stage IV was higher than that of patients with Stage I and Stage II (P<0.05) according to the FIGO stage. Furthermore, associations between clinical characteristics and overall survival in OC were analyzed. According to medium FAM83D and miR-142-3p expression, patients were divided into two groups. Our results indicated that FAM83D mRNA and miR-142-3p expression level (P<0.05), FIGO stage (P<0.05), Primary therapy outcome (P<0.05), and tumor stage (P<0.05) was related to the OS of patients with OC (Tables 3, 4).
Table 1. Relationship between the expression level of FAM83D and clinical characteristics in OC.
| Characteristic | Low expression of FAM83D | High expression of FAM83D | p |
| n | 189 | 190 | |
| FIGO stage, n (%) | 0.007 | ||
| Stage I | 0 (0%) | 1 (0.3%) | |
| Stage II | 53 (13.9%) | 10 (2.7%) | |
| Stage III | 100 (26.3%) | 155 (41.2%) | |
| Stage IV | 35 (9.3%) | 22 (5.9%) | |
| Primary therapy outcome, n (%) | 0.484 | ||
| PD | 12 (3.9%) | 15 (4.9%) | |
| SD | 14 (4.5%) | 8 (2.6%) | |
| PR | 22 (7.1%) | 21 (6.8%) | |
| CR | 102 (33.1%) | 114 (37%) | |
| Race, n (%) | 0.840 | ||
| Asian | 5 (1.4%) | 7 (1.9%) | |
| Black or African American | 13 (3.6%) | 12 (3.3%) | |
| White | 161 (44.1%) | 167 (45.8%) | |
| Age, n (%) | 0.383 | ||
| <=60 | 99 (26.1%) | 109 (28.8%) | |
| >60 | 90 (23.7%) | 81 (21.4%) | |
| Age, median (IQR) | 60 (52, 68) | 58 (50, 68) | 0.390 |
Table 2. Relationship between the expression level of hsa-miR-142-3p and clinical characteristics in OC.
| Characteristic | Low expression of hsa-miR-142-3p | High expression of hsa-miR-142-3p | p |
| n | 248 | 248 | |
| FIGO stage, n (%) | 0.009 | ||
| Stage I | 0 (0%) | 1 (0.2%) | |
| Stage II | 13 (2.6%) | 76 (15.3%) | |
| Stage III | 199 (40.4%) | 123 (24.8%) | |
| Stage IV | 35 (7.1%) | 45 (9.1%) | |
| Primary therapy outcome, n (%) | 0.210 | ||
| PD | 21 (5.1%) | 15 (3.7%) | |
| SD | 16 (3.9%) | 9 (2.2%) | |
| PR | 24 (5.9%) | 32 (7.8%) | |
| CR | 139 (34.1%) | 152 (37.3%) | |
| Race, n (%) | 0.472 | ||
| Asian | 10 (2.1%) | 6 (1.3%) | |
| Black or African American | 14 (2.9%) | 18 (3.8%) | |
| White | 215 (44.9%) | 216 (45.1%) | |
| Age, n (%) | 0.786 | ||
| <=60 | 139 (28%) | 135 (27.2%) | |
| >60 | 109 (22%) | 113 (22.8%) | |
| Age, median (IQR) | 58.5 (51, 69) | 59 (51.75, 68.25) | 0.873 |
Table 3. Relationship between overall survival and the expression level of FAM83D researched by univariate and multivariate Cox regression.
| Characteristics | Total(N) | Univariate analysis | Multivariate analysis | |||
| Hazard ratio (95% CI) | P value | Hazard ratio (95% CI) | P value | |||
| FIGO stage | 374 | |||||
| Stage I & Stage II | 24 | Reference | ||||
| Stage III | 293 | 2.045 (0.905-4.621) | 0.085 | 1.850 (0.670-5.108) | 0.235 | |
| Stage IV | 57 | 2.495 (1.057-5.889) | 0.037 | 2.563 (1.541-4.517) | 0.041 | |
| Primary therapy outcome | 307 | |||||
| PD | 27 | Reference | ||||
| SD | 22 | 0.441 (0.217-0.895) | 0.023 | 0.463 (0.222-0.967) | 0.040 | |
| PR | 42 | 0.652 (0.384-1.107) | 0.113 | 0.637 (0.360-1.126) | 0.121 | |
| CR | 216 | 0.152 (0.093-0.247) | <0.001 | 0.203 (0.120-0.344) | <0.001 | |
| Race | 364 | |||||
| Asian & Black or African American | 37 | Reference | ||||
| White | 327 | 0.637 (0.405-1.004) | 0.052 | 0.738 (0.434-1.255) | 0.262 | |
| FAM83D | 377 | 1.645 (1.916-3.192) | 0.011 | |||
| Tumor status | 336 | |||||
| Tumor free | 72 | Reference | ||||
| With tumor | 264 | 9.576 (4.476-20.486) | <0.001 | 9.616 (3.875-23.866) | <0.001 | |
Table 4. Relationship between overall survival and the expression level of hsa-miR-142-3p researched by univariate and multivariate Cox regression.
| Characteristics | Total(N) | Univariate analysis | Multivariate analysis | |||
| Hazard ratio (95% CI) | P value | Hazard ratio (95% CI) | P value | |||
| FIGO stage | 490 | |||||
| Stage I & Stage II | 30 | Reference | ||||
| Stage III | 380 | 2.179 (1.076-4.411) | 0.031 | 1.513 (1.664-3.446) | 0.004 | |
| Stage IV | 80 | 2.785 (1.331-5.829) | 0.007 | 1.333 (1.563-3.155) | 0.013 | |
| Primary therapy outcome | 407 | |||||
| PD | 36 | Reference | ||||
| SD | 25 | 0.453 (0.241-0.854) | 0.014 | 0.452 (0.237-0.862) | 0.016 | |
| PR | 55 | 0.668 (0.426-1.047) | 0.079 | 0.635 (0.400-1.010) | 0.055 | |
| CR | 291 | 0.168 (0.112-0.251) | <0.001 | 0.211 (0.139-0.319) | <0.001 | |
| Race | 478 | |||||
| Asian & Black or African American | 48 | Reference | ||||
| White | 430 | 0.764 (0.513-1.138) | 0.186 | |||
| hsa-miR-342-3p | 494 | 0.835 (0.738-0.946) | 0.005 | 1.910 (1.783-2.458) | 0.019 | |
| Tumor status | 442 | |||||
| Tumor free | 95 | Reference | ||||
| With tumor | 347 | 8.796 (4.784-16.170) | <0.001 | 8.362 (4.057-17.236) | <0.001 | |
Validation of the expression with qRT-PCR and IHC
To deeply evaluate the expression of miR-142-3p and FAM83D, 20 pairs of normal ovarian tissues (NOT) and OC tissues (OCT) were enrolled as a validation cohort. The qRT-PCR approach was utilized to verify the differential expression levels from the patient’s tissues. The same as the microarray data, hsa-miR-142-3p was significantly downregulated (Figure 5A) and FAM83D was overexpressed (Figure 5B) between 20 pairs of NOTs and OCTs which indicated that hsa-miR-142-3p and FAM83D could be the candidate biomarkers for OC. Besides, this expression level of genes was detected by IHC. The staining intensity of FAM83D was divided into 0, 1, 2, or 3, corresponding to colorless, light yellow, light brown, and squid ink. In addition, the percentage score is defined as: 0% to 5%, 0; 6% - 25%, 1 point; 26-50%, 2 points; 50-75%, 3 points; 76% to 100%, 4 points. The final histochemical score was calculated by multiplying the intensity score by the percentage score. The final staining scores were negative (0), low (6), and strong (≥ 6). Statistically, the overall expression of FAM83D was much higher in OC tissues than in the adjacent noncancerous tissues (P < 0.001) (Figure 5C–5F).
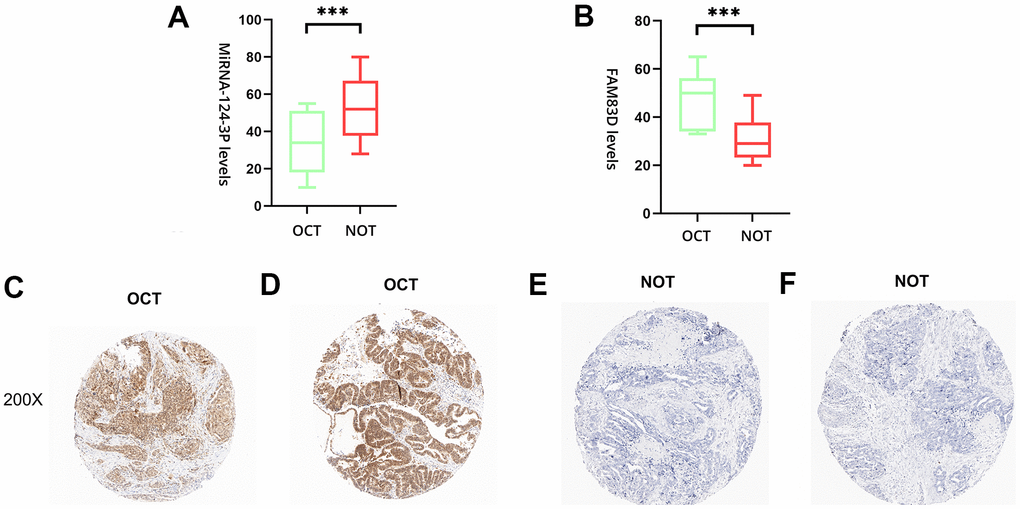
Figure 5. The expression level of hsa-miR-142-3p and FAM83D in normal ovarian tissues (NOTs) and OC tissues (OCTs). (A). Validation of hsa-miR-142-3p ((***, p < 0.001). (B). Validation of FAM83D (***, p < 0.001). (C, D). The expression level of FAM83D in human OCTs. (E, F). The expression level of FAM83D in human NOTs (scale bar: 200×).
Confirmation of the association between miR-142-3p and FAM83D
The Dual-Luciferase Reporter Assay System was utilized to evaluate the association between FAM83D and microRNA-142-3p. The FAM83D-WT activity of wild-type luciferase was 36.40%, and the FAM83D-MUT activity of mutant luciferase was 72.80%. These outcomes indicated that microRNA-142-3p reduced the luciferase activity of the wild-type FAM83D reporter gene (Figure 6).
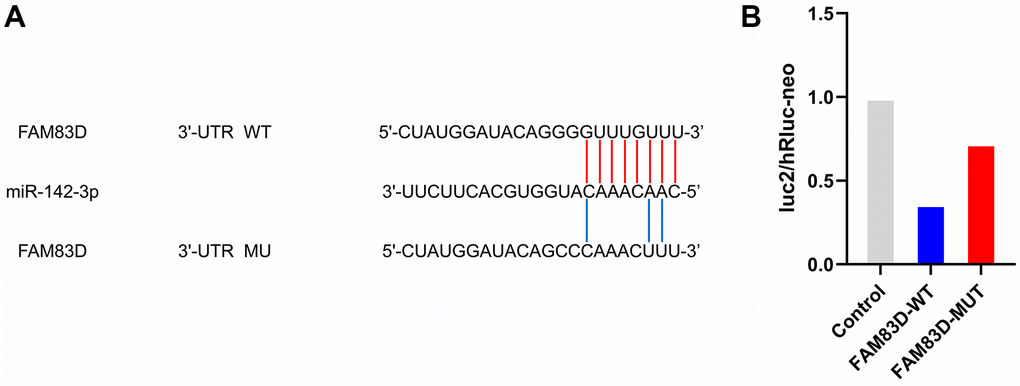
Figure 6. FAM83D is a direct target of miR-142-3p in OC cells. (A) Putative binding sites in FAM83D 3’UTR for miR-142-3p were predicted by bioinformatics analysis (microRNA.org). (B). Validation of luc2/hRluc-neo between miR-142-3p and FAM83D.
Discussion
In 2020, there were 19.29 million new cancer cases worldwide, including 10.06 million males and 9.23 million females; In 2020, there were 9.96 million cancer deaths worldwide, including 5.53 million males and 4.43 million females [11, 12]. In this research, GSE81873 and GSE27651 were obtained in the GEO database. 56 DEMs and 63 DEGs were identified. For deeply understanding the process of the 56 DEMs in OC, FunRich and R software were utilized for further study. GO and KEGG analysis indicated that these DEMs were primarily associated with the Cytoplasm, Nucleus, and Transcription factor activity. Previous studies have shown that Lysosomes and Nucleus may function as a vital role in plenty of human diseases, such as tumors, obesity, and infection [13–15]. Furthermore, KEGG analysis showed that DEGs were mainly enriched in 6 pathways such as MAPK signaling pathway, PI3K-Akt signaling pathway, Wnt signaling pathway, TGF-beta signaling pathway, Pathways in cancer, and Bacterial invasion of epithelial cells, which were indicated to influence migration and proliferation [16]. The MAPK signaling pathway is a common signaling pathway closely associated with carcinoma, which will not be discussed here. Phosphatidylinositol-4,5-diphosphate-3-kinase (PI3K) is activated by many genes. In PI3K / Akt signal transduction pathway, phospholipid dependent kinase promotes the binding of protein kinase B (Akt) to the cell membrane. The phosphorylation of threonine and serine promotes the transfer of Akt from the cytoplasm to the nucleus and further mediates the biological effects of enzymes, including cell proliferation, inhibition of apoptosis, cell migration, vesicle transport, and cell carcinogenesis [17]. Besides, it can influence the epithelial-mesenchymal transition in plenty of methods to affect tumor aggressiveness [18]. As for OC, a previous study reported that it took an important part in OC tumorigenesis, proliferation and progression, and pre-clinical and clinical experience with several PI3K/AKT/mTOR pathway inhibitors [19]. Neurotrophins are produced by target tissues innervated by developing neurons, so these factors act on the end of axons and produce signals that must be transmitted back to the cell body [20]. It can activate the PI3K/Akt signal transduction pathway and other oncogenic signaling pathways [21]. For example, it has been confirmed that in the early stage of cancer, the increase of nerve density is parallel to the increase of neurotrophin level, but it remains to be clarified which cells in TME are the source of neurotrophin and the nature of the stimulants that initiate the production of neurotrophin [22]. Besides, a previous study reported that Neurotrophin Receptor TrkB (NTRK2) and Wnt β-Estradiol and MAPK signaling pathways are closely related to the worse prognosis of neuroblastoma [23]. Besides, the changes in neurotrophin signals are related to neurodegenerative diseases and mental diseases [24].
The regulated network was performed according to Cytoscape. 56 miRNAs (hsa-microRNA-142-3p, hsa-microRNA-429, hsa-microRNA-199a-3p, and hsa-microRNA-484) were selected for further research. After that, 1069 target genes were achieved and 1 of them showed a different expression level in GSE27651 (FAM83D). MicroRNA-142-3p encoding human chromosome 17q22 is a new tumor suppressor factor, which is observed in many tumors including breast carcinoma, hepatocellular carcinoma, bladder carcinoma, and ovarian carcinoma [25–30]. The downregulation of microRNA-142-3p is related to tumorigenesis via regulation, cell migration, cell apoptosis, and invasion through various signaling pathways. Besides, microRNA-142-3p has been found as a carcinogenic microRNA in human T-cell acute lymphoblastic leukemia by acting on glucocorticoid receptors and cyclic adenosine monophosphate/protein kinase A pathway [31]. Upregulated microRNA-142-3p also monitors the characteristics of breast carcinoma stem cells, at least in section by sensitizing the WNT signaling pathway and microRNA-150 expression [32]. A previous study reported that upregulated microRNA-142-3p inhibited colorectal cancer cell migration and invasion, indicating that microRNA-142-3p may act as an oncogene during colorectal cancer tumorigenesis [33]. As for OC, another study concluded that microRNA-142-3p suppressed the proliferation and chemoresistance of OC cells by targeting SIRT1. This indicates that microRNA-142-3p may be a therapeutic target for the cure of OC [27].
FAM83D (family with sequence similarity 83, member D) is a mitosis-related gene located on chromosome 20q11 [34]. Previous studies have indicated that FAM83D may be amplified and upregulated in a variety of tumors, including hepatocellular tumors [35], ovarian carcinoma [36], colorectal carcinoma [37], and lung adenocarcinoma [38]. In addition, researches have demonstrated that FAM83D may act as a carcinogenic role by suppressing the invasion and proliferation of hepatocellular carcinoma and inhibiting the cell cycle of lung adenocarcinoma by suppressing FBXW7 in breast carcinoma [39–41]. These outcomes indicated that FAM83D may be widely taken part in process of tumors. Besides, FAM83D can advance epithelial-mesenchymal transition and metastasis of non-small cell lung carcinoma cells through the AKT/mTOR signal pathway, also improve the sensitivity of NSCLC cells to cisplatin [42]. Furthermore, FAM83D is highly expressed in invasive epithelial ovarian cancer and is related to tumor stage and grade [43]. Last but not least, FAM83D can also promote ovarian carcinoma cell invasion and proliferation, while suppressing autophagy through the PI3K/AKT/mTOR signaling pathway [44]. Therefore, we selected and tested whether microRNA-142-3p and FAM83D were differently expressed between NOTs and OCT.s qPCR results indicated that microRNA-142-3p and FAM83D were differentially expressed in NOTs and OCTs. Besides, IHC results indicated that FAM83D had a significant difference in OCTs compared with NOTs.
Many studies have shown that the abnormal expression of miRNAs is caused by gene aberrations (including genetic and epigenetic changes) of many cancer types, and plays a role in the occurrence and development of cancer through the imbalance of target gene expression. Therefore, many miRNAs and their target genes are closely related to the pathogenesis of tumors, including cell proliferation, cell survival, and cell invasion. Our study indicated that plenty of DEGs and DEMs were taken part in the process of OC by some pathways and had prognostic value. Therefore, suppression of FAM83D and upregulated miR-142-3p may have latent remedy worth in OC patients.
Conclusions
Our study indicated some reasons for the procession of OC. Plenty of DEMs and DEGs were selected between OC tissues and normal ovarian tissues. Besides, miR-142-3p and FAM83D were selected as latent biomarkers of OC. qPCR and IHC results indicated that microRNA-142-3p and FAM83D were differentially expressed in OC tissues. Besides, luciferase reporter assays verified that FAM83D was a direct target of miR-142-3p in OC cells. However, we need more cell experiments to prove it.
Materials and Methods
Microarray data
The RNA-seq data of OC samples and corresponding normal ovarian tissues were retrieved from the GEO dataset (https://www.ncbi.nlm.nih.gov/geo/). The datasets of GSE81873 and GSE27651 were downloaded and divided into two groups.
Differently expressed miRNAs research
GEO2R is software for differential analysis of expression microarray based on the GEO database.
Limma R package was used to identify DEGs in the construction cohort. The screening standards of DEGs for functional enrichment analysis were |log2FC|> 1 and FDR<0.05.
Gene ontology and pathway enrichment analysis
Transcription factors (TF), Kyoto Encyclopedia of Genes and Genomes (KEGG), and Gene Ontology (GO) enrichment analyses of the DEGs were performed by using R clusterProfiler package, including the package of "GOplot”, “ggplot2”, “stringi”, “colorspace” and “digest”. Then, the pathway and process enrichment analyses were carried out by using Cytoscape.
MicroRNA-mRNA regulatory network
At present, there are two generally recognized miRNA mechanisms: miRNA-mediated mRNA translation inhibition and miRNA-mediated mRNA-specific cleavage. In addition, researchers also found that miRNA may have other regulatory mechanisms, such as regulating the localization or stability of target mRNA, or acting on target molecules other than mRNAs, such as complementary binding with regulatory non-coding RNA or even miRNA, or competing with other RNAs to bind proteins to achieve its regulatory function. DEMs were uploaded to the FunRich software to achieve target mRNAs. Furthermore, GSE27651 was researched by utilizing R software. Based on the prediction conclusions of target mRNAs in FunRich software and the differentially expressed mRNAs of GSE27651, the microRNA-mRNA network was constructed.
The association between the expression level of identified genes and overall survival of patients with OC
Kaplan Meier plotter can assess the impact of 54K (mRNA, miRNA, protein) on the survival rate of 21 types of cancer (including breast cancer (n = 6234), ovarian cancer (n = 2190), lung cancer (n = 3452) and gastric cancer (n = 1440). The sources of the Kaplan Meier plotter database include GEO, EGA, and TCGA. The main purpose of the tool is the discovery and validation of survival biomarkers based on a meta-analysis. In this study, patients with OC were divided into two groups. By uploading the DEGs we identified, corresponding survival curves were obtained.
DEGs expression and clinical characteristics in the cancer genome atlas
The associated statistics offered by The Cancer Genome Atlas. The data of 1037 patients with ovarian cancer were downloaded in the TCGA database. The expression level of mRNAs, clinicopathological information, and general information of patients with OC were achieved.
Immunohistochemical staining
20 pairs of OC tissues were prepared by buffering with 10% formalin for 24 hours. The study was approved by the Ethics Committee of the Second Affiliated Hospital of Soochow University and informed consent was obtained from all patients. 2 consecutive 5-m sections were taken from each formalin-fixed paraffin-embedded block and mounted on the glass slide treated with aminoalkyl silane, dewaxing with xylene, passing through graded alcohol, and then continue rinsing in deionized water and phosphate-buffered saline. It was blocked by 3% non-immune horse serum. The sections were incubated with broad-spectrum anti-cytokeratin AE1 / 3 (Dako, Santa Barbara, CA, USA) in 1:50 dilution overnight at room temperature. After washing twice in buffer, an appropriate biotinylated secondary antibody was applied for 30 minutes. After two more washes in the buffer, appropriate biotinylated secondary antibodies were applied for 30 minutes. The sections were developed under the microscope in Tris HCl buffer (pH 7.4) and 0.03% hydrogen peroxide for 20 minutes. Application of light Mayer hematoxylin. The expressions of FAM83D and β - Catenin were detected by the Chi-square test or Fisher exact test. GraphPad Prism 9 software was utilized for statistical analysis. P-value < 0.05 was considered statistically significant.
Real-time quantitative polymerase chain reaction
Total RNA was extracted from OC tissues and normal tissues by utilizing TRIzol reagents (Invitrogen, Carlsbad, CA, USA) as per the manufacturer’s instructions. The total RNA was reverse-transcribed into cDNA by utilizing a PrimeScript™RT kit with gDNA Eraser (TaKaRa, China). We also constructed Quantitative Real-time PCR (qRT-PCR) by using SYBR Select Master Mix for CFX (Invitrogen) and the CFX Connect Real-Time PCR System (BioRad). The amplification conditions are 95° C for 15s, then 40 cycles, 95° C for 5S, 60° C for the 30s. Primer sequences of FAM83D were as follows: primer F, 5’- GCACTTCCCTTTGTTGTAGTC −3’, primer R, 5’-AGCACTTCCCTTAGGTTACTC −3’. Using glyceraldehyde-3-phosphate dehydrogenase (GAPDH) as endogenous control, the recorded data were analyzed and processed by 2−ΔΔCt.
Luciferase reporter assay
The relationship between miR-142-3p and FAM83D was verified by performing a luciferase reporter assay. To construct a luciferase ratio vector, we amplified the wild or mutant fragment FAM83D 3 ‘- UTR containing the hypothetical binding site miR-142-3p and subcloned it into luciferase pLUC vector (Wuhan, Ruibo, China). OC cells were co-transfected with pLUC-FAM83D 3 ‘- UTR wild (WT) or pLUC-FAM83D 3’ - UTR (MU) mutants and miR-142-3p or NC analogues using Lipofectamine 2000 according to the manufacturer’s instructions. After 48 hours of transfection, the relative activities of luciferase and double luciferase were detected by the analytical system (Promega, Madison, WI, USA). The luciferase activity of fireflies was normalized to Renilla luciferase activity.
Statistical methods
The data were expressed as mean ± standard error. All data analyses were performed using R software (version 3.6.6) and GraphPad Prism 9 software package (GraphPad Software, Inc., La Jolla, California, USA). The differences between the two addiction groups were compared by student’s t-test. All the experiments were made in triplicate.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Ethics Committee of the Soochow University.
Author Contributions
Conceptualization, Xiaofei Guo; Methodology, Guangyu Gao; Software, Wenyong Gu; Validation, Guangyu Gao; Formal Analysis, Guangyu Gao.; Investigation, Yufeng Lu; Resources, Guangyu Gao; Data Curation, Guangyu Gao; Writing – Original Draft Preparation, Guangyu Gao; Writing – Review and Editing, Guangyu Gao; Visualization, Guangyu Gao; Supervision, Zhigang Chen; Project Administration, Zhigang Chen; Funding Acquisition, Zhigang Chen.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This study was approved by Pre-Research Project of the National Natural Science Foundation of China (SDFEYGJ2014); Suzhou Science and Technology Development Project (SYSD2019108); Science and Technology Program of Suzhou City (SLJ2021012).
References
- 1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020; 70:7–30. https://doi.org/10.3322/caac.21590 [PubMed]
- 2. Lheureux S, Gourley C, Vergote I, Oza AM. Epithelial ovarian cancer. Lancet. 2019; 393:1240–53. https://doi.org/10.1016/S0140-6736(18)32552-2 [PubMed]
- 3. Wilson MK, Pujade-Lauraine E, Aoki D, Mirza MR, Lorusso D, Oza AM, du Bois A, Vergote I, Reuss A, Bacon M, Friedlander M, Gallardo-Rincon D, Joly F, et al, and participants of the Fifth Ovarian Cancer Consensus Conference. Fifth Ovarian Cancer Consensus Conference of the Gynecologic Cancer InterGroup: recurrent disease. Ann Oncol. 2017; 28:727–32. https://doi.org/10.1093/annonc/mdw663 [PubMed]
- 4. Kossaï M, Leary A, Scoazec JY, Genestie C. Ovarian Cancer: A Heterogeneous Disease. Pathobiology. 2018; 85:41–9. https://doi.org/10.1159/000479006 [PubMed]
- 5. Bushati N, Cohen SM. microRNA functions. Annu Rev Cell Dev Biol. 2007; 23:175–205. https://doi.org/10.1146/annurev.cellbio.23.090506.123406 [PubMed]
- 6. Simonson B, Das S. MicroRNA Therapeutics: the Next Magic Bullet? Mini Rev Med Chem. 2015; 15:467–74. https://doi.org/10.2174/1389557515666150324123208 [PubMed]
- 7. Krol J, Loedige I, Filipowicz W. The widespread regulation of microRNA biogenesis, function and decay. Nat Rev Genet. 2010; 11:597–610. https://doi.org/10.1038/nrg2843 [PubMed]
- 8. Tong J, Lu J, Yin Y, Wang Y, Zhang K. microRNA-195 Promotes Small Cell Lung Cancer Cell Apoptosis via Inhibiting Rap2C Protein-Dependent MAPK Signal Transduction. Technol Cancer Res Treat. 2020; 19:1533033820977546. https://doi.org/10.1177/1533033820977546 [PubMed]
- 9. Wu Y, Shen Q, Chen X, Wu Y, Niu Y, Lv F. miR-1301-3p promotes the proliferation and migration of lung cancer cells via direct repression of polymerase I and transcript release factor. Oncol Lett. 2020; 20:286. https://doi.org/10.3892/ol.2020.12149 [PubMed]
- 10. Katakura S, Kobayashi N, Hashimoto H, Kamimaki C, Tanaka K, Kubo S, Nakashima K, Teranishi S, Manabe S, Watanabe K, Horita N, Hara Y, Yamamoto M, et al. MicroRNA-200b is a potential biomarker of the expression of PD-L1 in patients with lung cancer. Thorac Cancer. 2020; 11:2975–82. https://doi.org/10.1111/1759-7714.13653 [PubMed]
- 11. Laconi E, Marongiu F, DeGregori J. Cancer as a disease of old age: changing mutational and microenvironmental landscapes. Br J Cancer. 2020; 122:943–52. https://doi.org/10.1038/s41416-019-0721-1 [PubMed]
- 12. Thong MS, van Noorden CJ, Steindorf K, Arndt V. Cancer-Related Fatigue: Causes and Current Treatment Options. Curr Treat Options Oncol. 2020; 21:17. https://doi.org/10.1007/s11864-020-0707-5 [PubMed]
- 13. Ballabio A. The awesome lysosome. EMBO Mol Med. 2016; 8:73–6. https://doi.org/10.15252/emmm.201505966 [PubMed]
- 14. Pucino V, Cucchi D, Mauro C. Lactate transporters as therapeutic targets in cancer and inflammatory diseases. Expert Opin Ther Targets. 2018; 22:735–43. https://doi.org/10.1080/14728222.2018.1511706 [PubMed]
- 15. Payen VL, Hsu MY, Rädecke KS, Wyart E, Vazeille T, Bouzin C, Porporato PE, Sonveaux P. Monocarboxylate Transporter MCT1 Promotes Tumor Metastasis Independently of Its Activity as a Lactate Transporter. Cancer Res. 2017; 77:5591–601. https://doi.org/10.1158/0008-5472.CAN-17-0764 [PubMed]
- 16. Galli L, Marcelli G. [Water-electrolyte balances in urological surgery]. Arch Ital Urol Nefrol. 1968; 41:199–217. [PubMed]
- 17. Xie Y, Shi X, Sheng K, Han G, Li W, Zhao Q, Jiang B, Feng J, Li J, Gu Y. PI3K/Akt signaling transduction pathway, erythropoiesis and glycolysis in hypoxia (Review). Mol Med Rep. 2019; 19:783–91. https://doi.org/10.3892/mmr.2018.9713 [PubMed]
- 18. Xu W, Yang Z, Lu N. A new role for the PI3K/Akt signaling pathway in the epithelial-mesenchymal transition. Cell Adh Migr. 2015; 9:317–24. https://doi.org/10.1080/19336918.2015.1016686 [PubMed]
- 19. Ediriweera MK, Tennekoon KH, Samarakoon SR. Role of the PI3K/AKT/mTOR signaling pathway in ovarian cancer: Biological and therapeutic significance. Semin Cancer Biol. 2019; 59:147–60. https://doi.org/10.1016/j.semcancer.2019.05.012 [PubMed]
- 20. Hamanaka D, Suzuki T, Kawanishi K, Sakamoto T, Yamazaki T, Odori T, Ishii Y, Torizuka K. Two cases of primary isolated chylopericardium diagnosed by oral administration of 131I-triolein. Radiat Med. 1983; 1:65–9. [PubMed]
- 21. Hinz E. [Contribution to the standardization of experimental cysticercosis of the mouse as a model experiment for testing of agents effective against cysticercus]. Z Tropenmed Parasitol. 1965; 16:322–31. [PubMed]
- 22. Dittrich M, Thomas AK, Stelma FF, Talla I, Niang M, Decam C, Sow S, Mbaye A, Gryseels B, Ehrich JH, Doehring E. Preliminary ultrasonographical observations of intestinal lesions in a community with heavy Schistosoma mansoni infection in Richard Toll, Senegal. Acta Trop. 1994; 58:331–6. https://doi.org/10.1016/0001-706x(94)90026-4 [PubMed]
- 23. Ruckes J, Stallkamp B. [On an unusually large neurinoma of the lung]. Zentralbl Allg Pathol. 1967; 110:306–13. [PubMed]
- 24. Watanabe M, Usui T, Shimizu M, Ito K, Okawara C. [Round table discussion; current status of the administrator of a nursing school and his problems]. Kango Kyoiku. 1968; 9:12–24. [PubMed]
- 25. Mansoori B, Mohammadi A, Gjerstorff MF, Shirjang S, Asadzadeh Z, Khaze V, Holmskov U, Kazemi T, Duijf PH, Baradaran B. miR-142-3p is a tumor suppressor that inhibits estrogen receptor expression in ER-positive breast cancer. J Cell Physiol. 2019; 234:16043–53. https://doi.org/10.1002/jcp.28263 [PubMed]
- 26. Hua S, Liu C, Liu L, Wu D. miR-142-3p inhibits aerobic glycolysis and cell proliferation in hepatocellular carcinoma via targeting LDHA. Biochem Biophys Res Commun. 2018; 496:947–54. https://doi.org/10.1016/j.bbrc.2018.01.112 [PubMed]
- 27. Gao J, Wu N, Liu X, Xia Y, Chen Y, Li S, Deng Z. MicroRNA-142-3p inhibits cell proliferation and chemoresistance in ovarian cancer via targeting sirtuin 1. Exp Ther Med. 2018; 15:5205–14. https://doi.org/10.3892/etm.2018.6107 [PubMed]
- 28. Godfrey JD, Morton JP, Wilczynska A, Sansom OJ, Bushell MD. MiR-142-3p is downregulated in aggressive p53 mutant mouse models of pancreatic ductal adenocarcinoma by hypermethylation of its locus. Cell Death Dis. 2018; 9:644. https://doi.org/10.1038/s41419-018-0628-4 [PubMed]
- 29. Lee YY, Yarmishyn AA, Wang ML, Chen HY, Chiou SH, Yang YP, Lin CF, Huang PI, Chen YW, Ma HI, Chen MT. MicroRNA-142-3p is involved in regulation of MGMT expression in glioblastoma cells. Cancer Manag Res. 2018; 10:775–85. https://doi.org/10.2147/CMAR.S157261 [PubMed]
- 30. Li WQ, Zhao WC, Xin J, Niu TL, Chao YF, Zhou P, Zheng MH, Xu B. MicroRNA-142-3p suppresses cell proliferation and migration in bladder cancer via Rac1. J Biol Regul Homeost Agents. 2020. [Epub ahead of print]. https://doi.org/10.23812/19-460-A [PubMed]
- 31. Lv M, Zhang X, Jia H, Li D, Zhang B, Zhang H, Hong M, Jiang T, Jiang Q, Lu J, Huang X, Huang B. An oncogenic role of miR-142-3p in human T-cell acute lymphoblastic leukemia (T-ALL) by targeting glucocorticoid receptor-α and cAMP/PKA pathways. Leukemia. 2012; 26:769–77. https://doi.org/10.1038/leu.2011.273 [PubMed]
- 32. Isobe T, Hisamori S, Hogan DJ, Zabala M, Hendrickson DG, Dalerba P, Cai S, Scheeren F, Kuo AH, Sikandar SS, Lam JS, Qian D, Dirbas FM, et al. miR-142 regulates the tumorigenicity of human breast cancer stem cells through the canonical WNT signaling pathway. Elife. 2014; 3:e01977. https://doi.org/10.7554/eLife.01977 [PubMed]
- 33. Gao X, Xu W, Lu T, Zhou J, Ge X, Hua D. MicroRNA-142-3p Promotes Cellular Invasion of Colorectal Cancer Cells by Activation of RAC1. Technol Cancer Res Treat. 2018; 17:1533033818790508. https://doi.org/10.1177/1533033818790508 [PubMed]
- 34. Deloukas P, Matthews LH, Ashurst J, Burton J, Gilbert JG, Jones M, Stavrides G, Almeida JP, Babbage AK, Bagguley CL, Bailey J, Barlow KF, Bates KN, et al. The DNA sequence and comparative analysis of human chromosome 20. Nature. 2001; 414:865–71. https://doi.org/10.1038/414865a [PubMed]
- 35. Ramakrishna M, Williams LH, Boyle SE, Bearfoot JL, Sridhar A, Speed TP, Gorringe KL, Campbell IG. Identification of candidate growth promoting genes in ovarian cancer through integrated copy number and expression analysis. PLoS One. 2010; 5:e9983. https://doi.org/10.1371/journal.pone.0009983 [PubMed]
- 36. van Dekken H, Paris PL, Albertson DG, Alers JC, Andaya A, Kowbel D, van der Kwast TH, Pinkel D, Schröder FH, Vissers KJ, Wildhagen MF, Collins C. Evaluation of genetic patterns in different tumor areas of intermediate-grade prostatic adenocarcinomas by high-resolution genomic array analysis. Genes Chromosomes Cancer. 2004; 39:249–56. https://doi.org/10.1002/gcc.20001 [PubMed]
- 37. Yan L, Yao J, Qiu J. miRNA-495 suppresses proliferation and migration of colorectal cancer cells by targeting FAM83D. Biomed Pharmacother. 2017; 96:974–81. https://doi.org/10.1016/j.biopha.2017.11.138 [PubMed]
- 38. Inamura K, Shimoji T, Ninomiya H, Hiramatsu M, Okui M, Satoh Y, Okumura S, Nakagawa K, Noda T, Fukayama M, Ishikawa Y. A metastatic signature in entire lung adenocarcinomas irrespective of morphological heterogeneity. Hum Pathol. 2007; 38:702–9. https://doi.org/10.1016/j.humpath.2006.11.019 [PubMed]
- 39. Wang Z, Liu Y, Zhang P, Zhang W, Wang W, Curr K, Wei G, Mao JH. FAM83D promotes cell proliferation and motility by downregulating tumor suppressor gene FBXW7. Oncotarget. 2013; 4:2476–86. https://doi.org/10.18632/oncotarget.1581 [PubMed]
- 40. Liao W, Liu W, Liu X, Yuan Q, Ou Y, Qi Y, Huang W, Wang Y, Huang J. Upregulation of FAM83D affects the proliferation and invasion of hepatocellular carcinoma. Oncotarget. 2015; 6:24132–47. https://doi.org/10.18632/oncotarget.4432 [PubMed]
- 41. Shi R, Sun J, Sun Q, Zhang Q, Xia W, Dong G, Wang A, Jiang F, Xu L. Upregulation of FAM83D promotes malignant phenotypes of lung adenocarcinoma by regulating cell cycle. Am J Cancer Res. 2016; 6:2587–98. [PubMed]
- 42. Yin C, Lin X, Wang Y, Liu X, Xiao Y, Liu J, Snijders AM, Wei G, Mao JH, Zhang P. FAM83D promotes epithelial-mesenchymal transition, invasion and cisplatin resistance through regulating the AKT/mTOR pathway in non-small-cell lung cancer. Cell Oncol (Dordr). 2020; 43:395–407. https://doi.org/10.1007/s13402-020-00494-9 [PubMed]
- 43. Zhang Q, Yu S, Lok SI, Wong AS, Jiao Y, Lee LT. FAM83D promotes ovarian cancer progression and its potential application in diagnosis of invasive ovarian cancer. J Cell Mol Med. 2019; 23:4569–81. https://doi.org/10.1111/jcmm.14360 [PubMed]
- 44. Zhu H, Diao S, Lim V, Hu L, Hu J. FAM83D inhibits autophagy and promotes proliferation and invasion of ovarian cancer cells via PI3K/AKT/mTOR pathway. Acta Biochim Biophys Sin (Shanghai). 2019; 51:509–16. https://doi.org/10.1093/abbs/gmz028 [PubMed]


