Introduction
Lung adenocarcinoma (LUAD), the most common form of lung cancer, is the leading cause of cancer-related deaths worldwide [1]. Despite significant advancements in cancer treatment, the prognosis for LUAD patients remains poor due to the heterogeneity and complexity of the disease [2]. LUAD is characterized by complex genomic alterations, which contribute to disease initiation, progression, and metastasis [3]. Therefore, there is an urgent need for further research into the cellular and physiopathologic mechanisms underlying lung adenocarcinoma to develop more effective treatments.
ANGPTL4 is a secreted protein that has been implicated in multiple physiopathologic processes, including lipid metabolism, and cancer progression [4]. Through its roles in these processes, ANGPTL4 has garnered significant attention as a potential therapeutic target [5]. Specifically, ANGPTL4 has been found to play a role in the regulation of vascular permeability and angiogenesis, which has implications for the treatment of various vascular disorders, such as diabetic retinopathy and cancer [6]. Furthermore, ANGPTL4 has been shown to have profound effects on lipid metabolism, affecting both uptake and storage, and has been associated with obesity and metabolic disorders [7]. Most recently, ANGPTL4 has been implicated in cancer cell migration, invasion, and metastasis [8]. As such, ANGPTL4 represents an exciting therapeutic target in the fight against cancer [9]. Nevertheless, the specific functions of ANGPTL4 and the exact regulatory mechanisms affecting its expression in LUAD are yet to be fully understood.
MicroRNAs (miRNAs), a class of small non-coding RNAs, is involved in post-transcriptional regulation by suppressing target gene expression [10]. Among them, miR-133a-3p is involved in various aspects of cancer development and progression [11–13]. Dysregulation of miR-133a-3p has been found to promote cell proliferation, migration, invasion, and angiogenesis in several types of cancer, including lung cancer [14, 15]. Furthermore, miR-133a-3p has also been identified as an important diagnostic and prognostic biomarker in lung cancer, with its expression levels correlating with the clinical stage and outcome of the disease [16].
In this study, we found that ANGPTL4 is ectopic expressed in LUAD and that its upregulation is correlated with an unfavorable prognosis. Silencing ANGPTL4 inhibited the viability of LUAD cells. Moreover, miR-133a-3p negatively regulated ANGPTL4 through an interaction with its 3′-UTR. According to our findings, miR-133a-3p/ANGPTL4 might be utilized as a potential therapy in LUAD.
Methods
Bioinformatic analysis
The expression analysis for 522 cases LUAD patient samples and 59 cases paracancerous tissue samples were based on TCGA database LUAD dataset (https://portal.gdc.cancer.gov/) [17]. GSEA and GO analysis was based on LinkedOmics database (http://www.linkedomics.org/login.php) [18]. miRNA analysis was based on TargetScan database (https://www.targetscan.org/vert_80/) [19]. The overall survival of 522 cases of LUAD patient samples were also based on TCGA database LUAD dataset (https://portal.gdc.cancer.gov/) [17].
Cell culture and transfection
The LUAD cell lines (A549 and H1975) were obtained from the American Type Culture Collection (USA) and cultured in DMEM with 10% fetal bovine serum (FBS), 100 IU/mL penicillin, and 10 μg/mL streptomycin. We purchased ANGPTL4 shRNA, negative control vectors, ANGPTL4 OE, miR-133a-3p mimics, and miR-133a-3p inhibitors from HonorGene (Changsha, China) and used Lipofectamine 3000 (Thermo Fisher Scientific, USA) for transfecting the cells.
qRT-PCR
For specific methods, please refer to our previous publication [20]. The primer sequences were as follows: miR-133a-3p F: 5′-CTTTAACCATTCTAGCTTTTCCAGGTA-3′ R: 5′-GACTTCGGCTGTGGACAAGATTAG-3′ U6 F: 5′-CTCGCTTCGGCAGCACACA-3′ R: 5′-AACGCTTCACGAATTTGCGT-3′ ANGPTL4 F: 5′-TCCTGGACCACAAGCACCTAGAC-3′ R: 3′-CGGTTGAAGTCCACTGAGCCATC-5′ β-actin F: 5′-GGGACCTGACTGACTACCTC-3′ F: 5′-TCATACTCCTGCTTGCTGAT-3′.
Western blot
For specific methods, please refer to our previous publication [20]. The primary antibodies are ANGPTL4 (ab115798, Abcam, UK) and FASN (ab128870, Abcam).
IHC staining
For specific methods of IHC staining, please refer to our previous study [20]. The primary antibody for IHC were as follows: PCNA (ab265609, Abcam), E-cadherin (ab227639, Abcam), N-cadherin (ab76011, Abcam) and ANGPTL4 (ab196746, Abcam).
Proliferation analysis
For the MTT assay, 5000 cells were cultured in 96-well plates. After incubation, 20 μl of MTT solution (5 mg/ml, Sigma–Aldrich, USA) was added to each well, and the plates were further incubated for 4 hours at 37°C with 5% CO2. Next, 150 μl of DMSO was used to dissolve the precipitates.
For the colony formation assay, cells in the logarithmic growth phase were digested with 0.25% trypsin, suspended in DMEM culture medium containing 10% FBS, and the cell suspension was serially diluted. Then, 50 cells from each group were seeded into dishes containing 2 mL of preheated culture medium and incubated at 37°C with 5% CO2 for 14 days. The cells were gently rotated to disperse them evenly during incubation.
For the EdU assay, the kit instructions provided by RiboBio (Guangzhou, China) were followed.
Migration and invasion analysis
For the wound healing study, 5 × 105 cells were placed in 6-well plates, and the surface was scratched using a pipette tip. After washing the cells with DMEM, they were cultured at 37°C for 24 hours. Photographs were taken at 0 and 24 hours to evaluate the healing process.
For the Transwell assay, 2.5 × 105 cells were seeded in a 24-well Transwell chamber (Costar, USA) with Matrigel for the invasion assay. The cells were cultured in DMEM with a 10% FBS concentration for 16 hours at 37°C with 5% CO2. The invading cells on the filter’s lower surface were stained with crystal violet.
Dual-luciferase reporter gene
In A549 cells, luciferase assays were performed. Genes from wild-type ANGPTL4 and mutant controls were integrated into the psiCheck2 Luciferase vector (Promega Corporation, USA). We purchased the psiCHECK2 vector (cat. no. C8021) from Promega. We synthesized the miR-133a-3p mimics, negative control samples (NCs), and miR-133a-3p inhibitor and NC inhibitor at Guangzhou RiboBio Co., Ltd. (Guangzhou, China), which were transfected with Lipofectamine® 2000 (Invitrogen; Thermo Fisher Scientific). Firefly and Renilla luciferase activities were measured 48 h after transfection using the dual-luciferase assay kit (Promega Corporation). In the next step, cells were collected and lysed. On a Luminometer TD20/20 detector (E5311; Promega Corporation), luciferase activity was measured using the Dual Luciferase Reporter Assay System Kit (Promega Corporation). Finally, the relative activity of Firefly/Renilla luciferase was measured.
Xenograft model assay
The Vector and ANGPTL4 KD A549 cells were subcutaneously injected into adults with athymic BALB/c nude mice (4 weeks old). Each ten days, tumor volumes (cm3) were measured. The volume = width2 × length × 0.5. As a next step, the xenografts were measured at 50 days. In the last step, the xenografts were dehydrated and sectioned in order to be stained.
Statistical analysis
The statistical analyses were conducted using R language (Version 3.6). All statistical tests were two-tailed, and a significance level of P < 0.05 was deemed statistically significant.
Data availability statement
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found in the Article/Supplementary Materials.
Results
The expression and potential function of ANGPTL4 in LUAD
Firstly, we utilized the TCGA database LUAD dataset to confirm the expression of ANGPTL4 in LUAD patients, which indicated that ANGPTL4 level was obviously increased in LUAD samples (Figure 1A). Moreover, ANGPTL4 expression was also obviously and positively correlated with poor prognosis in LUAD patients (Figure 1B). LinkedOmics (http://www.linkedomics.org/login.php) was used to access the correlated gene dataset for ANGPTL4 mRNA in LUAD patients (Figure 1C, 1D). GO enrichment assay showed that these gene were enriched in biological regulation, metabolic process, and response to stimulus for BP term, membrane, nucleus, membrane-enclosed lumen for CC term, protein binding, ion binding, and nucleic acid binding for MF term (Figure 1E). GSEA assay indicated that these genes were enriched in ribosome, proteasome, oxidative phosphorylation, ether lipid metabolism and fatty acid degradation (Figure 1F). We used CCLE database to further confirm the ANGPTL4 in multiple LUAD cell lines (Figure 1G). Based on this result, we selected A549 and H1975 as subjects for further studies.
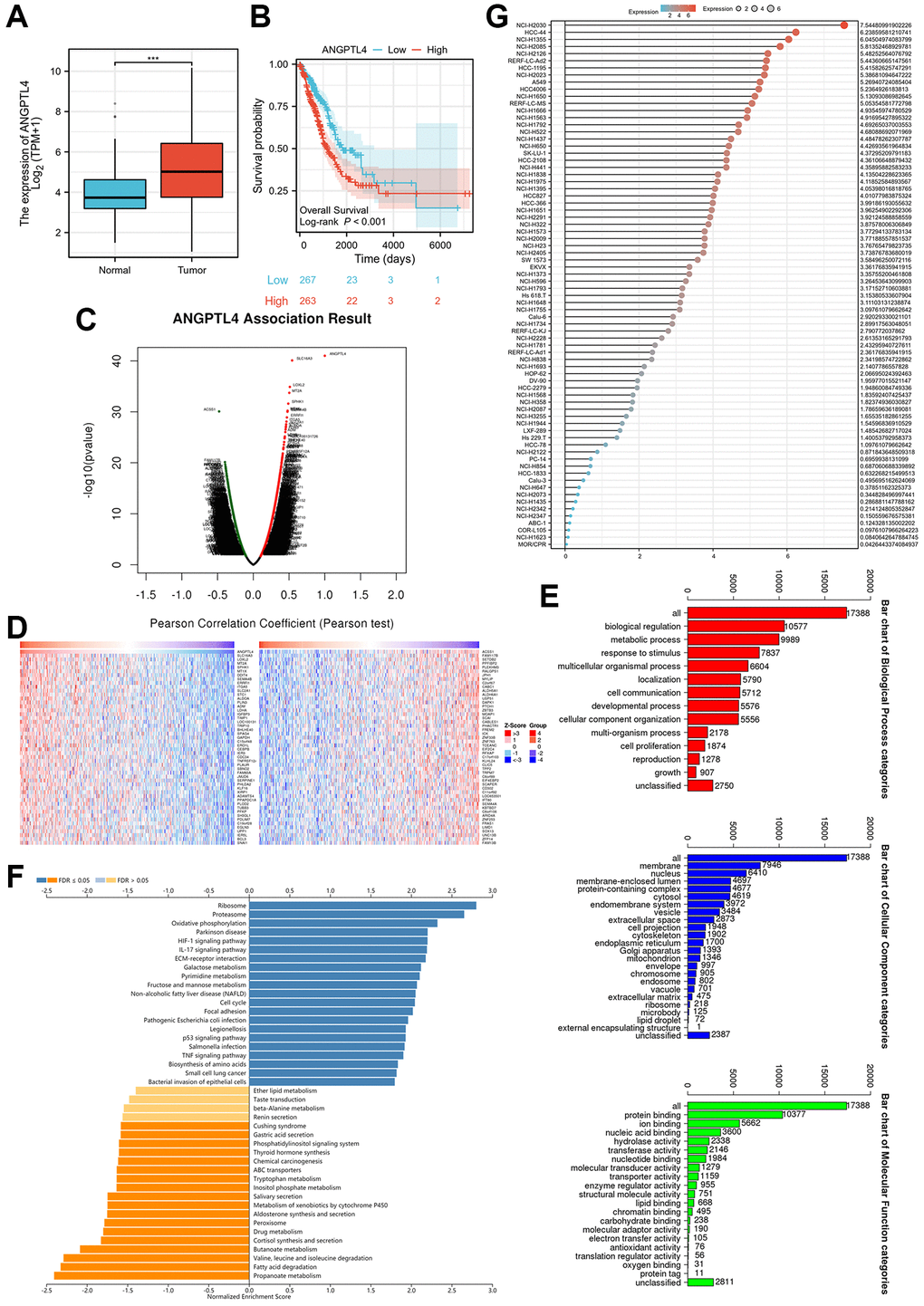
Figure 1. The expression and potential function of ANGPTL4 in LUAD. (A) The expression of ANAGPTL4 in LUAD patients based on TCGA database. (B) The overall survival of ANGPTL4 for LUAD patients. (C) The correlated gene of ANGPTL4 in LUAD patients by heat map. (D) The correlated gene of ANGPTL4 in LUAD patients by volcanic plot. (E) The GO enrichment analysis for these gene. (F) The GSEA assay for these gene. (G) The expression of ANGPTL4 in LUAD cell lines based on CCLE database. ***P < 0.001 indicates statistical significance compared with the control.
ANGPTL4 knockdown impeded proliferation, lipids accumulation and invasion in LUAD cell
Next, we constructed ANGPTL4 knockdown A549 and H1975 cell (Figure 2A). Oil red O staining showed that ANGPTL4 knockdown could significantly downregulate lipids production in A549 and H1975 cell (Figure 2B). MTT analysis showed that ANGPTL4 knockdown could significantly repress LUAD cell viability (Figure 2C). The clone formation assay showed that the ANGPTL4 inhibition could obviously impeded the clone formation ability in A549 and H1975 cell (Figure 2D). EdU assay showed that the DNA replication level was remarkedly decreased after ANGPTL4 knockdown in LUAD cell (Figure 2E). Wound healing analysis showed that ANGPTL4 inhibition could significantly downregulate the migration ability in A549 and H1975 cell (Figure 2F). Transwell invasion assay showed that ANGPTL4 knockdown could obviously decrease the invasion ability for LUAD cell (Figure 2G).
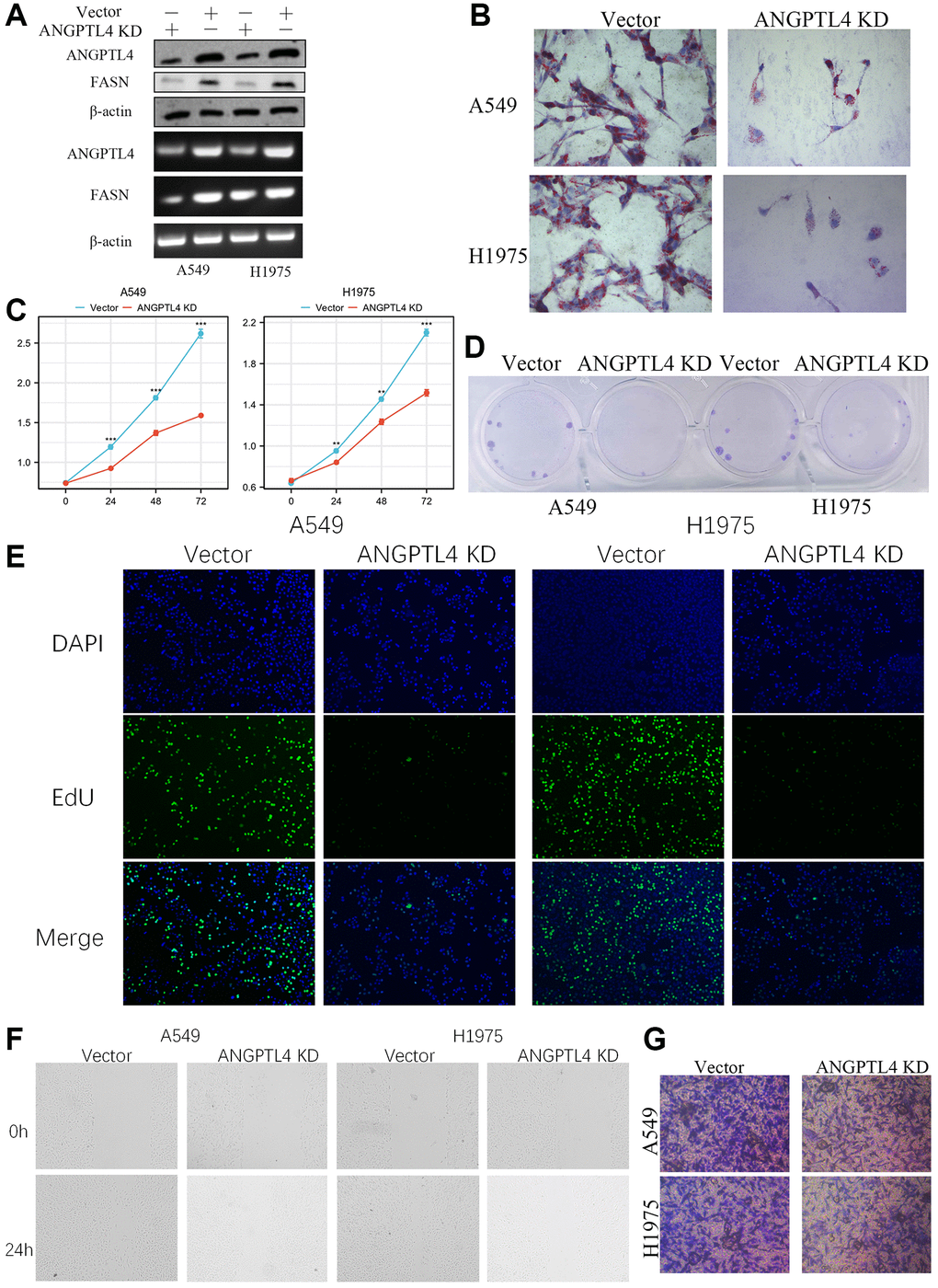
Figure 2. The effect of ANGPTL4 on LUAD cell proliferation, lipids production, migration and invasion. (A) The expression of ANGPTL4 and FASN in ANGPTL4 KD A549 and H1975 cell by WB and PCR. (B) The effect of ANGPTL4 KD on lipids metabolism by oil red O. MTT analysis (C), Clone formation assay (D), and EdU staining (E) were performed to confirm the effect of ANGPTL4 KD on cell proliferation. Wound healing assay (F) and Transwell invasion assay (G) were performed to confirm the effect of ANGPTL4 KD on cell migration and invasion. **P < 0.01, ***P < 0.001 indicates statistical significance compared with the control.
Silencing ANGPTL4 inhibits LUAD growth in vivo
For confirming the function of ANGPTL4 in vivo, we used stable ANGPTL4 knockdown or vector A549 cell to inject into nude mice for constructing xenograft model. We found silencing ANGPTL4 significantly attenuated the LUAD growth, with smaller size and less weight of xenografts in ANGPTL4 knockdown group (Figure 3A–3C). IHC staining suggested that N-cadherin and PCNA expression was obviously decreased in ANGPTL4 KD group, but the expression of E-cadherin was obviously increased in ANGPTL4 KD group (Figure 3D).
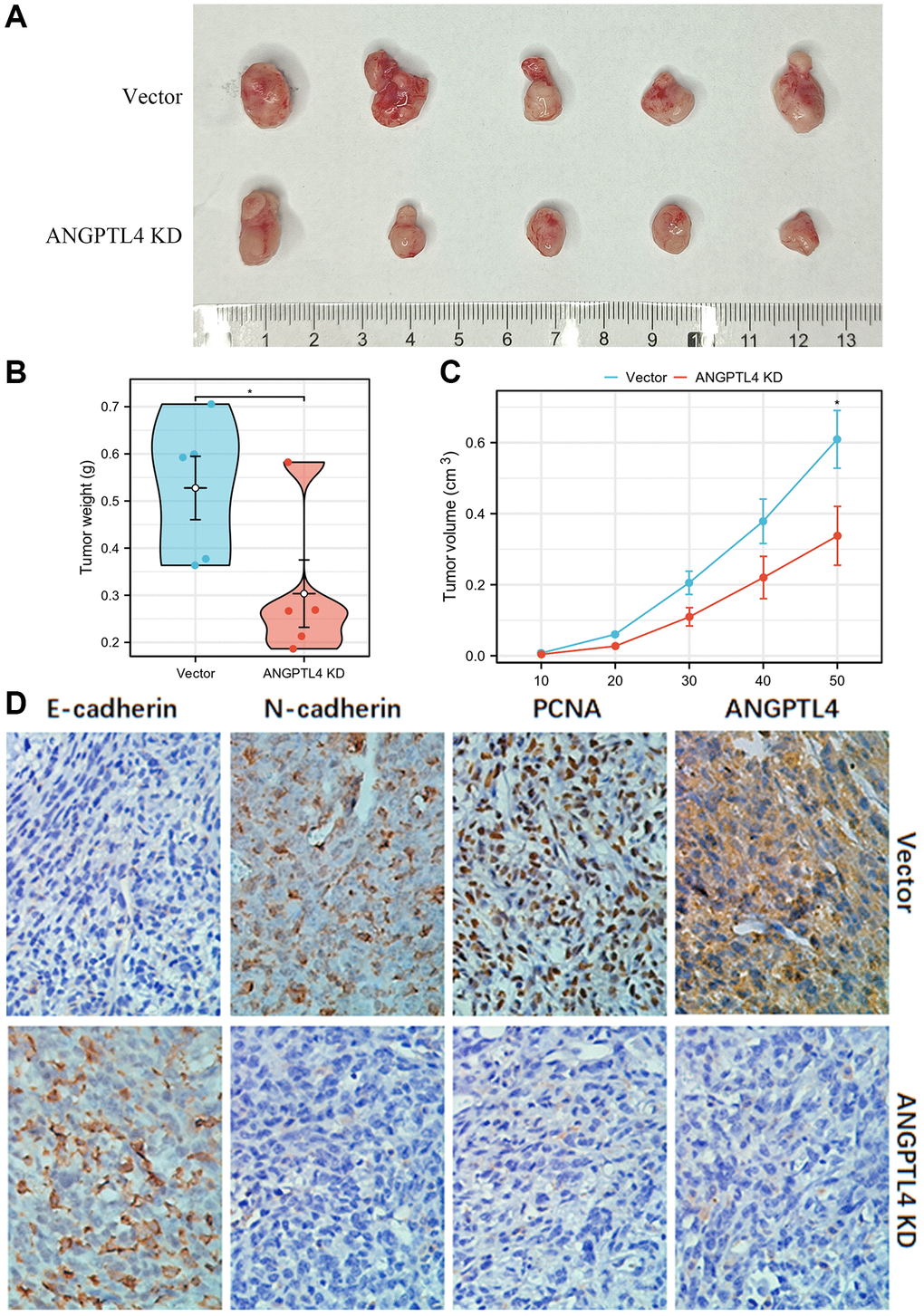
Figure 3. The effect of ANGPTL4 on LUAD cell growth in vivo. (A) The effects of ANGPTL4 shRNA lentivirus plasmid transfection or empty vector lentivirus plasmid transfection on A549 growth in vivo. The tumor weight (B) and volume (C) of the xenografts in ANGPTL4 KD group and vector group. (D) IHC staining for E-cadherin, N-cadherin, PCNA and ANGPTL4 in these xenografts for confirming the effects of ANGPTL4 on LUAD EMT cascade and proliferation. *P < 0.05 indicates statistical significance compared with the control.
ANGPTL4 is negatively mediated by miR-133a-3p
To elucidate how ANGPTL4 high expression is in LUAD, we used TargetScan database (https://www.targetscan.org/vert_80/) to seek for the miRNAs targeting ANGPTL4. We found miR-133a-3p has complementary sequences to binding against 3′-UTR of ANGPTL4 (Supplementary Figure 1). There is evidence that miR-133a-3p maybe a TSG in multiple cancer types, including LUAD [13], oesophageal cancer [12], prostate cancer [21], and gastric cancer [22]. We found the level of miR-133a-3p was significantly downregulated in LUAD samples based on TCGA database LUAD dataset (Figure 4A). Moreover, miR-133a-3p was negatively associated with poor prognosis (Figure 4B). The miR-133a-3p was significantly and negatively correlated with ANGPTL4 expression based on starBase database in LUAD patients (Figure 4C). We used luciferase reporter assay to confirm the directly regulation between ANGPTL4 and miR-133a-3p in A549 cell. The luciferase activity was significantly repressed in ANGPTL4 WT plus miR-133a-3p group compared to ANGPTL4 MUT plus miR-133a-3p group (Figure 4D). The qRT-PCR showed that miR-133a-3p mimics could decrease ANGPTL4 mRNA, and miR-133a-3p inhibitor could increase ANGPTL4 mRNA in A549 and H1975 cell (Figure 4E, 4F).
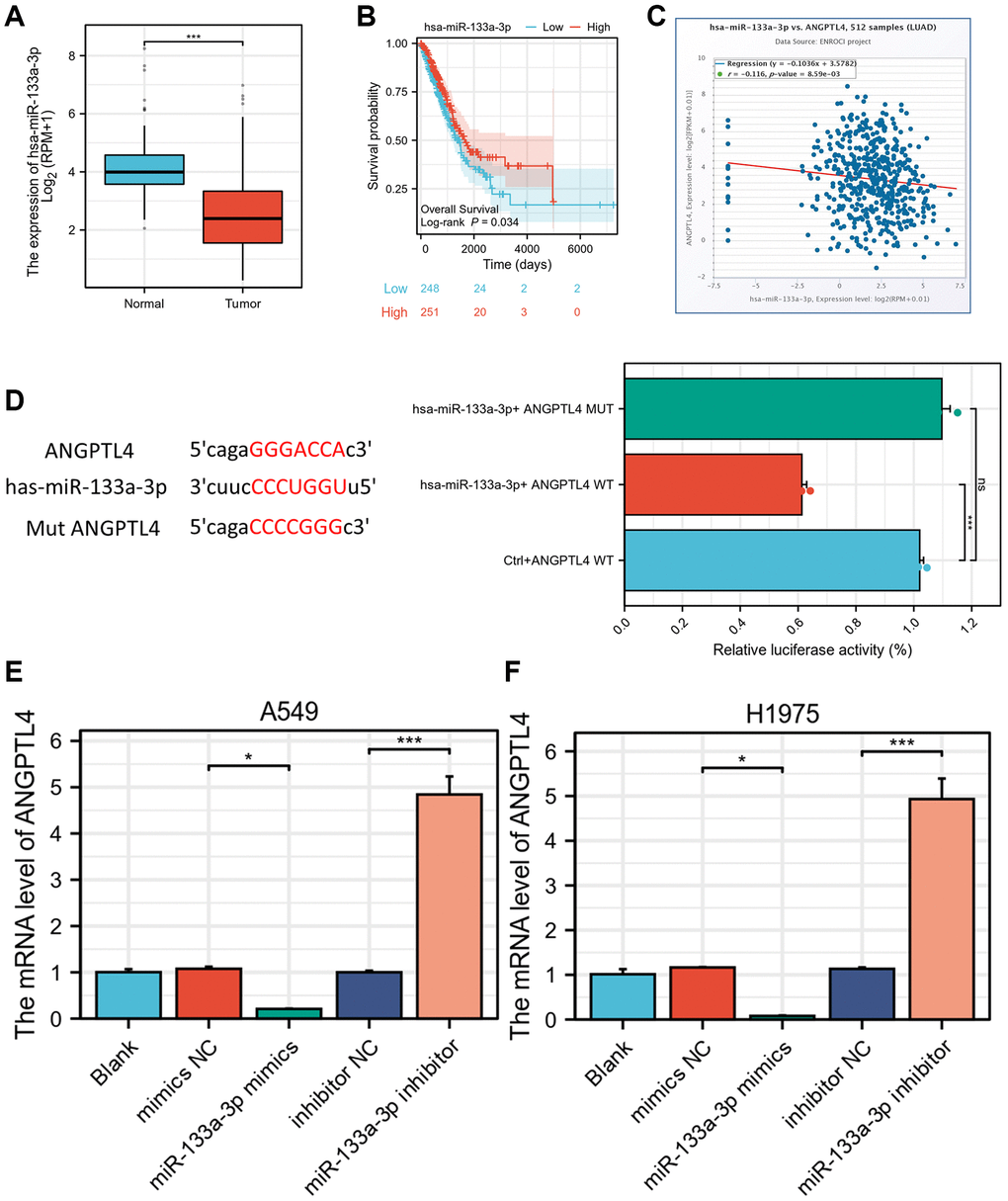
Figure 4. ANGPTL4 is a direct target of miR-133a-3p. (A) The expression of miR-133a-3p in LUAD patients based on TCGA database. (B) The overall survival between high or low level of miR-133a-3p LUAD patients. (C) The correlation analysis between ANGPTL4 and miR-133a-3p in LUAD patients. (D) Luciferase reporter analysis for WT or MUT 3′-UTR of ANGPTL4 in A549 cell. The qRT-PCR analysis for the effect of miR-133a-3p on ANGPTL4 expression in A549 cell (E) and H1975 cell (F). *P < 0.05 indicates statistical significance compared with the control. *P < 0.05 and ***P < 0.001 indicates statistical significance compared with the control.
The miR-133a-3p impeded the proliferation and invasion by targeting ANGPTL4 in LUAD cell
For further confirming miR-133a-3p/ANGPTL4 axis in LUAD development and progression, A549 and H1975 were transfected with vector, vector plus miR-133a-3p mimics, ANGPTL4, ANGPTL4 plus miR-133a-3p mimics (Figure 5A). MTT analysis showed that miR-133a-3p mimics could significantly repress cell proliferation ability, but ANGPTL4 overexpression could amplify cell proliferation ability (Figure 5B). Oil red O showed that ANGPTL4 could significantly increase lipids level, which could be reversed by miR-133a-3p (Figure 5C). EdU analysis showed that the DNA replication level was also upregulation in ANGPTL4 overexpression group, downregulation in miR-133a-3p mimics group and rescued in ANGPTL4 plus miR-133a-3p mimics group in A549 and H1975 cell (Figure 5D). The migration ability was significantly increased in ANGPTL4 group, but decreased in miR-133a-3p group, and rescued in ANGPTL4 plus miR-133a-3p mimics group in A549 and H1975 cell by wound healing assay (Figure 5E). Transwell invasion assay found that the invasion ability of A549 and H1975 cell was significantly decreased by miR-133a-3p and rescued by ANGPTL4 overexpression (Figure 5F).
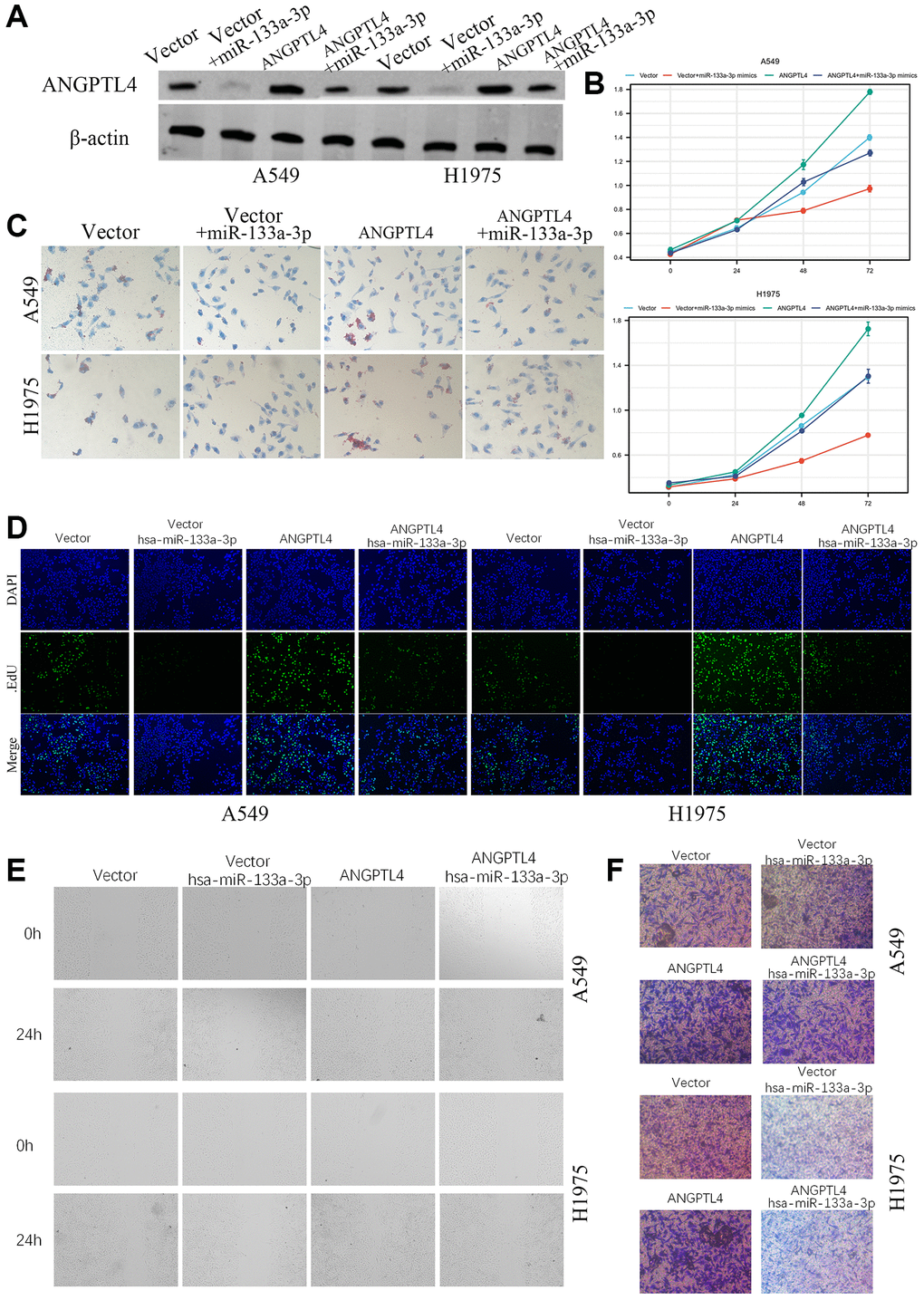
Figure 5. The role of miR-133a-3p/ANGPTL4 axis in LUAD cell proliferation and invasion. (A) The ANGPTL4 protein expression was confirmed by Western blot. (B) The effect of miR-133a-3p/ANGPTL4 axis on LUAD cell proliferation by MTT. (C) The effect of ANGPTL4 KD on lipids metabolism by oil red O. (D) LUAD proliferation ability for miR-133a-3p/ANGPTL4 axis was showed by EdU analysis. The effect of miR-133a-3p/ANGPTL4 axis on LUAD cell migration by wound healing (E) and invasion by Transwell analysis (F).
Discussion
ANGPTL4 play a key role in a variety of physiological and pathological nonmetabolic and metabolic conditions, such as angiogenesis, tumorigenesis, glucose homeostasis, lipid metabolism, energy homeostasis, immune infiltration, inflammation, and redox regulation [8]. There is evidence that ANGPTL4 is ectopically expressed in certain malignant tumor tissues, including ovarian cancer [23], cervical cancer [24], colorectal cancer [25], pancreatic cancer [26] and papillary thyroid cancer [27]. In this study, we found ANGPTL4 was significantly upregulated in LUAD patients, and correlated with poor prognosis.
Moreover, our results indicated that ANGPTL4 knockdown inhibited LUAD cell proliferation, lipids production and invasion in vitro, and attenuated LUAD growth in vivo. Previous study indicated that ANGPTL4 could bind to LPL and inhibit its biological activity to decrease lipids metabolism [28]. The mRNA level of LPL was significantly decreased in LUAD patients [29]. Moreover, LPL might be an excellent candidate target for cancer prevention or therapy in multiple cancer types [30]. We also found ANGPTL4 knockdown could significantly reduce the proliferation, migration and invasion ability in LUAD cell. Other study found that ANGPTL4 could regulate glutamine metabolism and fatty acid oxidation in lung cancer cells [31]. Shen et al. found that oleic acid could promoted colon cancer metastasis by driving ANGPTL4 [32]. Fang et al. indicated that he expression of ANGPTL4 regulated pyroptosis and apoptosis in LUAD cells by driving NLRP3/ASC/Caspase 8 pathway, leading to gefitinib resistance in those cells [33]. Zhu et al. found that ANGPTL4 could promote epithelial-mesenchymal transition cascade via ERK Pathway in lung cancer [34]. These results both indicated that ANGPTL4 might be a key role in LUAD carcinogenesis, which was still a need for further investigation.
We further found miR-133a-3p could remarkedly repressed ANGPTL4 mRNA expression via binding to its 3′-UTR, and the levels of miR-133a-3p was significantly decreased in LUAD patients. Li et al. found that miR-133a-3p could attenuate gefitinib resistance by targeting SPAG5 in LUAD [13]. Xu and his colleagues found that miR-133a-3p could suppress the LUAD malignant behavior via targeting ERBB2 [14]. Pan and his colleagues found that miR-133a-3p could interact with PURB to mediate MAPK and PI3K/Akt pathway in LUAD cell [35]. In this study, we found miR-133a-3p was correlated with favourable prognosis for LUAD patients, and negatively correlated with the ANGPTL4 mRNA expression to impede LUAD cell proliferation, migration and invasion. Taken together, these results both indicated that miR-133a-3p has multiple downstream targets involved in LUAD progression due to its complex regulation mechanism.
This study has several limitations that should be addressed in the future. Firstly, the mechanism and signaling pathway through which ANGPTL4 regulates lung cancer cell proliferation and invasion remain unknown. Secondly, it needs to be clarified whether miR-133a-3p regulates ANGPTL4 exclusively through negative regulation or whether it also controls other miRNAs. Furthermore, the potential value of ANGPTL4 as a prognosis biomarker should be further investigated.
In conclusion, we found ANGPTL4 was significantly increased in LUAD, and miR-133a-3p could directly repress ANGPTL4 expression and impeded LUAD proliferation, migration and invasion (Figure 6). Therefore, miR-133a-3p/ANGPTL4 axis might be a potential biomarker and therapeutic target for LUAD patients.
Figure 6. The potential mechanisms of miR-133a-3p/ANGPTL4 axis in LUAD.
Supplementary Materials
Author Contributions
QH Hu, and S Chen analyzed the data. YK Li and T Hu used online tools. JP Hu, C Wang and F Yang selected the analyzed results. X Yang, F Zhou and ZD Liu wrote the paper. W Xu and J Zhang revised the manuscript, designed the experiment. All authors contributed to the article and approved the submitted version.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical Statement
Animal experiments were approved by the Medical Ethics Committee of The First People’s Hospital of Changde City (protocol number CDTFPHEC2022083-01) that was conducted according to the ARRIVE guidelines. And all methods were carried out in accordance with relevant guidelines and regulations.
Funding
No funding was provided for this study.
References
- 1. Bade BC, Dela Cruz CS. Lung Cancer 2020: Epidemiology, Etiology, and Prevention. Clin Chest Med. 2020; 41:1–24. https://doi.org/10.1016/j.ccm.2019.10.001 [PubMed]
- 2. de Sousa VML, Carvalho L. Heterogeneity in Lung Cancer. Pathobiology. 2018; 85:96–107. https://doi.org/10.1159/000487440 [PubMed]
- 3. Rodriguez-Canales J, Parra-Cuentas E, Wistuba II. Diagnosis and Molecular Classification of Lung Cancer. Cancer Treat Res. 2016; 170:25–46. https://doi.org/10.1007/978-3-319-40389-2_2 [PubMed]
- 4. Aryal B, Price NL, Suarez Y, Fernández-Hernando C. ANGPTL4 in Metabolic and Cardiovascular Disease. Trends Mol Med. 2019; 25:723–34. https://doi.org/10.1016/j.molmed.2019.05.010 [PubMed]
- 5. Ploug M. ANGPTL4: a new mode in the regulation of intravascular lipolysis. Curr Opin Lipidol. 2022; 33:112–9. https://doi.org/10.1097/MOL.0000000000000800 [PubMed]
- 6. Fernández-Hernando C, Suárez Y. ANGPTL4: a multifunctional protein involved in metabolism and vascular homeostasis. Curr Opin Hematol. 2020; 27:206–13. https://doi.org/10.1097/MOH.0000000000000580 [PubMed]
- 7. Basu D, Goldberg IJ. Regulation of lipoprotein lipase-mediated lipolysis of triglycerides. Curr Opin Lipidol. 2020; 31:154–60. https://doi.org/10.1097/MOL.0000000000000676 [PubMed]
- 8. La Paglia L, Listì A, Caruso S, Amodeo V, Passiglia F, Bazan V, Fanale D. Potential Role of ANGPTL4 in the Cross Talk between Metabolism and Cancer through PPAR Signaling Pathway. PPAR Res. 2017; 2017:8187235. https://doi.org/10.1155/2017/8187235 [PubMed]
- 9. Zhu P, Goh YY, Chin HF, Kersten S, Tan NS. Angiopoietin-like 4: a decade of research. Biosci Rep. 2012; 32:211–9. https://doi.org/10.1042/BSR20110102 [PubMed]
- 10. Saliminejad K, Khorram Khorshid HR, Soleymani Fard S, Ghaffari SH. An overview of microRNAs: Biology, functions, therapeutics, and analysis methods. J Cell Physiol. 2019; 234:5451–65. https://doi.org/10.1002/jcp.27486 [PubMed]
- 11. Yu X, Wang D, Wang X, Sun S, Zhang Y, Wang S, Miao R, Xu X, Qu X. CXCL12/CXCR4 promotes inflammation-driven colorectal cancer progression through activation of RhoA signaling by sponging miR-133a-3p. J Exp Clin Cancer Res. 2019; 38:32. https://doi.org/10.1186/s13046-018-1014-x [PubMed]
- 12. Wang X, Zhu L, Lin X, Huang Y, Lin Z. MiR-133a-3p inhibits the malignant progression of oesophageal cancer by targeting CDCA8. J Biochem. 2022; 170:689–98. https://doi.org/10.1093/jb/mvab071 [PubMed]
- 13. Li Q, Wang Y, He J. MiR-133a-3p attenuates resistance of non-small cell lung cancer cells to gefitinib by targeting SPAG5. J Clin Lab Anal. 2021; 35:e23853. https://doi.org/10.1002/jcla.23853 [PubMed]
- 14. Xu Y, Zhang L, Xia L, Zhu X. MicroRNA-133a-3p suppresses malignant behavior of non-small cell lung cancer cells by negatively regulating ERBB2. Oncol Lett. 2021; 21:457. https://doi.org/10.3892/ol.2021.12718 [PubMed]
- 15. Ahmed S, Kurusamy S, David ELS, Khan K, Kalyanakrishnan K, Ian-Gobo M, Kola TM, Wilkinson RN, Kannappan V, Wang W, Gómez MJ, Redondo JM, Cotton J, Armesilla AL. Aberrant expression of miR-133a in endothelial cells inhibits angiogenesis by reducing pro-angiogenic but increasing anti-angiogenic gene expression. Sci Rep. 2022; 12:14730. https://doi.org/10.1038/s41598-022-19172-x [PubMed]
- 16. Yang ZQ, Wu CA, Cheng YX. Prognostic Value of microRNA-133a Expression and Its Clinicopathologic Significance in Non-Small Cell Lung Cancer: A Comprehensive Study Based on Meta-Analysis and the TCGA Database. Oncol Res Treat. 2018; 41:762–8. https://doi.org/10.1159/000492343 [PubMed]
- 17. Tomczak K, Czerwińska P, Wiznerowicz M. The Cancer Genome Atlas (TCGA): an immeasurable source of knowledge. Contemp Oncol (Pozn). 2015; 19:A68–77. https://doi.org/10.5114/wo.2014.47136 [PubMed]
- 18. Vasaikar SV, Straub P, Wang J, Zhang B. LinkedOmics: analyzing multi-omics data within and across 32 cancer types. Nucleic Acids Res. 2018; 46:D956–63. https://doi.org/10.1093/nar/gkx1090 [PubMed]
- 19. McGeary SE, Lin KS, Shi CY, Pham TM, Bisaria N, Kelley GM, Bartel DP. The biochemical basis of microRNA targeting efficacy. Science. 2019; 366:eaav1741. https://doi.org/10.1126/science.aav1741 [PubMed]
- 20. Li YK, Zeng T, Guan Y, Liu J, Liao NC, Wang MJ, Chen KX, Luo XY, Chen CY, Quan FF, Wang J, Zhang QF, Zou J. Validation of ESM1 Related to Ovarian Cancer and the Biological Function and Prognostic Significance. Int J Biol Sci. 2023; 19:258–80. https://doi.org/10.7150/ijbs.66839 [PubMed]
- 21. Tang Y, Pan J, Huang S, Peng X, Zou X, Luo Y, Ren D, Zhang X, Li R, He P, Wa Q. Downregulation of miR-133a-3p promotes prostate cancer bone metastasis via activating PI3K/AKT signaling. J Exp Clin Cancer Res. 2018; 37:160. https://doi.org/10.1186/s13046-018-0813-4 [PubMed]
- 22. Zhang X, Li Z, Xuan Z, Xu P, Wang W, Chen Z, Wang S, Sun G, Xu J, Xu Z. Novel role of miR-133a-3p in repressing gastric cancer growth and metastasis via blocking autophagy-mediated glutaminolysis. J Exp Clin Cancer Res. 2018; 37:320. https://doi.org/10.1186/s13046-018-0993-y [PubMed]
- 23. Bajwa P, Kordylewicz K, Bilecz A, Lastra RR, Wroblewski K, Rinkevich Y, Lengyel E, Kenny HA. Cancer-associated mesothelial cell-derived ANGPTL4 and STC1 promote the early steps of ovarian cancer metastasis. JCI Insight. 2023; 8:e163019. https://doi.org/10.1172/jci.insight.163019 [PubMed]
- 24. Nie D, Zheng Q, Liu L, Mao X, Li Z. Up-regulated of Angiopoietin-Like Protein 4 Predicts Poor Prognosis in Cervical Cancer. J Cancer. 2019; 10:1896–901. https://doi.org/10.7150/jca.29916 [PubMed]
- 25. Zheng X, Liu R, Zhou C, Yu H, Luo W, Zhu J, Liu J, Zhang Z, Xie N, Peng X, Xu X, Cheng L, Yuan Q, et al. ANGPTL4-Mediated Promotion of Glycolysis Facilitates the Colonization of Fusobacterium nucleatum in Colorectal Cancer. Cancer Res. 2021; 81:6157–70. https://doi.org/10.1158/0008-5472.CAN-21-2273 [PubMed]
- 26. Hui B, Ji H, Xu Y, Wang J, Ma Z, Zhang C, Wang K, Zhou Y. RREB1-induced upregulation of the lncRNA AGAP2-AS1 regulates the proliferation and migration of pancreatic cancer partly through suppressing ANKRD1 and ANGPTL4. Cell Death Dis. 2019; 10:207. https://doi.org/10.1038/s41419-019-1384-9 [PubMed]
- 27. Yang L, Wang Y, Sun R, Zhang Y, Fu Y, Zheng Z, Ji Z, Zhao D. ANGPTL4 Promotes the Proliferation of Papillary Thyroid Cancer via AKT Pathway. Onco Targets Ther. 2020; 13:2299–309. https://doi.org/10.2147/OTT.S237751 [PubMed]
- 28. Dijk W, Kersten S. Regulation of lipoprotein lipase by Angptl4. Trends Endocrinol Metab. 2014; 25:146–55. https://doi.org/10.1016/j.tem.2013.12.005 [PubMed]
- 29. Lu J, Li J, Ji C, Yu W, Xu Z, Huang S. Expression of lipoprotein lipase associated with lung adenocarcinoma tissues. Mol Biol Rep. 2008; 35:59–63. https://doi.org/10.1007/s11033-006-9053-3 [PubMed]
- 30. Takasu S, Mutoh M, Takahashi M, Nakagama H. Lipoprotein lipase as a candidate target for cancer prevention/therapy. Biochem Res Int. 2012; 2012:398697. https://doi.org/10.1155/2012/398697 [PubMed]
- 31. Xiao S, Nai-Dong W, Jin-Xiang Y, Long T, Xiu-Rong L, Hong G, Jie-Cheng Y, Fei Z. ANGPTL4 regulate glutamine metabolism and fatty acid oxidation in nonsmall cell lung cancer cells. J Cell Mol Med. 2022; 26:1876–85. https://doi.org/10.1111/jcmm.16879 [PubMed]
- 32. Shen CJ, Chang KY, Lin BW, Lin WT, Su CM, Tsai JP, Liao YH, Hung LY, Chang WC, Chen BK. Oleic acid-induced NOX4 is dependent on ANGPTL4 expression to promote human colorectal cancer metastasis. Theranostics. 2020; 10:7083–99. https://doi.org/10.7150/thno.44744 [PubMed]
- 33. Fang Y, Li X, Cheng H, Zhang L, Hao J. ANGPTL4 Regulates Lung Adenocarcinoma Pyroptosis and Apoptosis via NLRP3\ASC\Caspase 8 Signaling Pathway to Promote Resistance to Gefitinib. J Oncol. 2022; 2022:3623570. https://doi.org/10.1155/2022/3623570 [PubMed]
- 34. Zhu X, Guo X, Wu S, Wei L. ANGPTL4 Correlates with NSCLC Progression and Regulates Epithelial-Mesenchymal Transition via ERK Pathway. Lung. 2016; 194:637–46. https://doi.org/10.1007/s00408-016-9895-y [PubMed]
- 35. Pan J, Cai X, Zheng X, Zhu X, Feng J, Wang X. Luteolin inhibits viability, migration, angiogenesis and invasion of non-small cell lung cancer vascular endothelial cells via miR-133a-3p/purine rich element binding protein B-mediated MAPK and PI3K/Akt signaling pathways. Tissue Cell. 2022; 75:101740. https://doi.org/10.1016/j.tice.2022.101740 [PubMed]



