Is biological age reversible?
The 12th Aging Research and Drug Discovery (ARDD) meeting brought speakers from all over the world, representing the comprehensive spectrum of the aging field, including academic research, industrial development, investment capital, and media coverage (Figure 1). A synopsis of the meeting’s proceedings is presented in Figure 2.
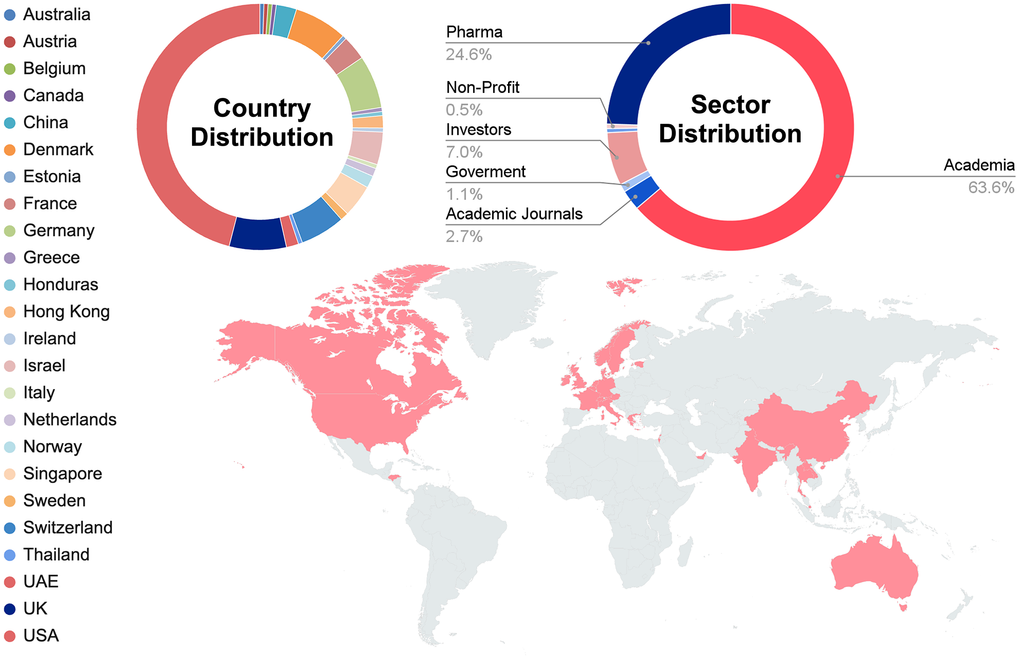
Figure 1. Statistics and Geography of ARDD 2025 speakers.
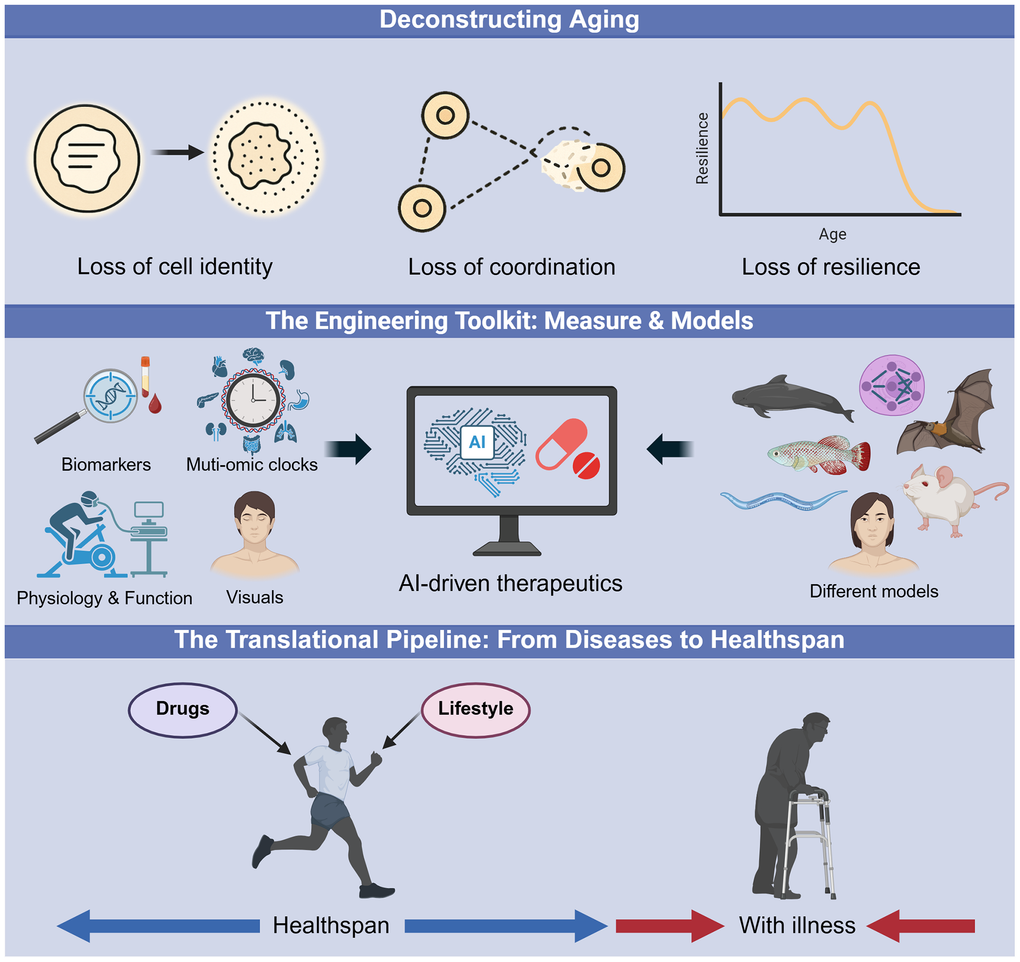
Figure 2. The roadmap to actionable longevity interventions. An overview of the conceptual frameworks presented at the 12th ARDD meeting. (Top) Deconstructing Aging: Moving beyond stochastic damage to identify modifiable drivers of aging, such as the loss of cellular identity, coordination, and systemic resilience. (Middle) The Engineering Toolkit: The integration of precise measurement tools, including organ-specific multi-omic clocks and biomarkers, with diverse preclinical models and AI platforms to accelerate therapeutic design. (Bottom) The Translational Pipeline: The strategic shift from reactive disease management to proactive healthspan extension, utilizing novel drugs and lifestyle interventions to maintain youthful function.
Among the many topics covered, a central question dominated the mechanistic sessions of the meeting: can we reverse biological age? Several presentations provided a nuanced “yes,” pointing to the epigenome as a master controller of cellular identity and function.
The reversible nature of epigenetic drift
The idea that aging is a reversible loss of youthful epigenetic information was brought into conversation. Jean-Marc Lemaitre (University of Montpellier, Montpellier, France) raised a key mechanistic question: is pluripotency necessary for rejuvenation? His work in a progeric mouse model demonstrated that a single, transient reprogramming event administered early in life extends healthy lifespan by 15%, preserving muscle integrity and preventing osteoarthritis, osteoporosis and lung fibrosis, suggesting the establishment of a persistent, youthful epigenetic state [1]. However, this intervention failed when applied late in life, suggesting that the system’s starting point is critical. Providing a unifying theory, Juan Carlos Izpisua Belmonte (Altos Labs, San Diego, CA, USA) framed aging as a progressive loss of cellular identity he termed “mesenchymal drift,” a process in which specialized cells shift toward more mesenchymal-like or hybrid states, driving tissue dysfunction and disease [2]. He argued that partial reprogramming may be particularly potent because it directly reverses this drift, a hypothesis that is increasingly well-supported. In a proof-of-concept study, his team rejuvenated aged kidneys ex vivo using reprogramming factors and successfully transplanted them into rats, bringing the vision of organ regeneration closer to clinical feasibility.
But does rejuvenation require treating every cell? In unpublished work, Thomas Rando (University of California Los Angeles, Los Angeles, CA, USA) revealed that local epigenetic reprogramming has profound systemic effects. By expressing Yamanaka factors only in the non-dividing muscle fibers of mice, his lab not only rejuvenated the muscle but also observed rejuvenation in distant organs like the spleen and kidney. This finding reframes muscle as an organ capable of communicating a pro-youthful state throughout the body, offering therapeutic potential where targeting one accessible tissue could yield systemic rejuvenation consistent with previous research [3].
Sharon Rosenzweig-Lipson (Life Biosciences, Boston, MA, USA) detailed their partial epigenetic reprogramming platform, using three Yamanaka factors (Oct4, Sox2, and KLF4, excluding c-Myc). She reported that recent Good Laboratory Practice (GLP) toxicology studies and results from a non-human primate nonarteritic anterior ischemic optic neuropathy (NAION) model showed that the therapy can reverse disease-associated DNA methylation patterns in retinal ganglion cells. Based on these findings, a first-in-human clinical trial for glaucoma and NAION is planned for the first quarter of 2026. Rosenzweig-Lipson also expanded the platform’s application by presenting new data from a metabolic dysfunction-associated steatohepatitis (MASH) mouse model, which demonstrated significant improvements in liver enzymes and steatosis without any corresponding body weight change, pointing to a direct, organ-specific effect.
Challenging conventional wisdom, Tuomas Tammela (Sloan Kettering Institute, New York, NY, USA) presented a surprising connection between aging and cancer. Contrary to expectations, his lab found that aged mice develop significantly fewer and smaller lung adenocarcinomas than their younger counterparts [4]. The protective mechanism was traced to an epigenetic program in aged alveolar stem cells, driven by DNA hypomethylation, which leads to high expression of the transcription factor NUPR1. This, in turn, creates a state of iron deficiency within the cells, impairing their ability to multiply and transform, thus acting as a potent barrier against tumor formation. This work reveals that some aspects of cellular aging can be protective, suggesting that therapies aimed at rejuvenating aged stem cells must be carefully considered in the context of cancer risk.
Genomic integrity and DNA damage response in aging
While the epigenome may be reversible, maintaining genomic integrity remains essential in the aging process. Jan Hoeijmakers (Erasmus Medical Center, Rotterdam, Netherlands) presented data showing that accumulating DNA damage causes a genome-wide “transcription stress,” where RNA polymerases stall on damaged DNA [5]. As DNA damage is random, this disproportionately silences long genes [6], a phenomenon that can explain up to 50% of the transcriptomic changes in aging organs, occurs in species ranging from worms to humans [7] and positions the unexpectedly long IGF-1 gene as a master DNA damage sensor.
Vanessa Sinatti (Aptah Biosciences, San Carlos, CA, USA) presented a platform targeting the U1 snRNP complex, a complex essential for safeguarding the transcriptional integrity of long genes found in the brain and retina. This complex becomes dysfunctional and aggregates in the cytoplasm in Alzheimer’s disease (AD), and potentially during aging, often appearing even before tau pathology [8]. Aptah Bio’s “RNA WiCo” technology acts as a scaffold to restore U1 function. In AD patient neurons, their compound restored electrical activity, and in aging mice it reduced amyloid-beta and phospho-tau. Sinatti also showed that the compound had superior anti-tumor efficacy over the standard of care in glioblastoma xenograft models.
Complementing this, Björn Schumacher (University of Cologne, Cologne, Germany) emphasized the genome’s unique, irreplaceable role in aging [9]. His lab developed a precise transcriptomic aging clock in C. elegans capable of predicting a young worm’s lifespan [10]. Single-cell analyses revealed that neuron types within the same animal age at different rates, with environmentally exposed neurons aging fastest. These conserved neuronal aging signatures enabled an in silico screen of FDA-approved drugs, identifying novel neuroprotective candidates [11].
What dictates the rate of this deterioration? Zane Koch (UC San Diego, La Jolla, CA, USA) discussed a potential master regulator: the DREAM complex, a transcriptional repressor of DNA repair pathways [12]. Across 93 species, from the short-lived to the long-lived, he and his colleagues found a significant negative correlation: species with lower DREAM activity, like humans, live longer [13]. Complementing these findings, Anna Murray (University of Exeter, Exeter, UK), showed that the rate of reproductive aging is also substantially influenced by DNA repair. Her analysis of over 200,000 women found that nearly two-thirds of the 290 genetic signals linked to the timing of menopause are in DNA damage response genes [14]. This work supports the view that ovarian aging is primarily driven by declining genomic maintenance.
Further challenging old dogmas, Ben Shenhar (Weizmann Institute of Science, Rehovot, Israel) argued that the heritability of human lifespan has been greatly underestimated. By correcting for extrinsic mortality (like accidents or infections) from historical data, his group suggests that the heritability of our intrinsic lifespan is around 50%, placing it on par with other major human traits [15]. A key source of this heritable, age-related damage may lie in the “dark genome,” as proposed by Andrei Seluanov (University of Rochester, Rochester, NY, USA). He demonstrated that the age-related activation of transposable elements triggers chronic inflammation via the cGAS-STING pathway [16]. Mice engineered to repress these transposable elements were leaner, less inflamed, and dramatically less frail in old age, prompting Seluanov to propose that existing HIV drugs that inhibit reverse transcriptase could be repurposed to achieve a similar anti-aging effect in humans [17].
Inflammaging and metabolic dysfunction
Chronic, low-grade inflammation, or “inflammaging”, was consistently identified as a key driver of aging. Miriam Merad (Precision Immunology Institute, New York, NY, USA) argued that macrophages are central mediators [18], showing that with age, protective tissue-resident macrophages are replaced by damaging, short-lived ones. Her lab found that administering spermidine, hypothesized to induce autophagy and limit monocyte-derived macrophage infiltration, restores the protective macrophage pool in aged mice, improving their immune responses. At the protein level, Andrea Heinz (University of Copenhagen, Copenhagen, Denmark) described elastin as a “non-renewable resource” synthesized only in early life. Its lifelong degradation not only causes loss of tissue function but releases inflammatory fragments called “elastokines,” creating a self-amplifying loop of damage [19, 20].
Neuroinflammation and brain resilience
Neuroinflammation creates localized pockets of pathology. Using high-resolution biology, Marissa Schafer (Mayo Clinic, Rochester, MN, USA) used spatial molecular imaging to map the aging brain. Her team discovered that specific clusters of senescent, “disease-associated” microglia accumulate in the white matter of aged female mice, forming a self-reinforcing network of neuroinflammation with other senescent-like cells [21]. This aligns with growing evidence that senescent cells are far more heterogeneous than previously recognized, encompassing both pathological forms, such as the chronic, slowly accumulating senescence associated with aging tissues, and physiological forms serving beneficial roles. Tomaz Rozmaric (Ludwig Boltzmann Institute for Traumatology, Vienna, Austria) illustrated this distinction, showing that skin injury triggers a rapid senescence response within just 15 minutes, driven by translation of pre-existing p21 mRNA [22]. Rather than contradicting the slow accumulation seen in aging, this finding suggests that the kinetics of senescence are context- and cell type-specific, with acute, wound-induced senescence being an essential component of normal tissue repair.
Andy P. Tsai (Stanford University, Stanford, CA, USA) explored the unique resilience of the cerebellum, a brain region that shows early signs of molecular aging but is largely spared from Alzheimer’s pathology. He found that cerebellar microglia adopt a more neuroprotective state with age compared to those in the hippocampus. Spatial transcriptomics revealed a novel “neuronal-associated microglia” state, which appears when microglia are in direct contact with granule cells, suggesting this interaction shapes their protective function [23].
Lida Katsimpardi (INSERM; University of Montpellier, Montpellier, France) provided compelling evidence for the rejuvenating power of systemic factors on the brain, showing that young blood exposure can rejuvenate aged mice by increasing neural stem cells, remodeling vasculature, and improving cognitive function [24]. She identified the circulating factor GDF11 as a key player that improves memory when administered by stimulating neuronal activity, autophagy, and reducing hippocampal senescence [25]. Her team is also investigating how conditions like depression accelerate brain aging, noting that senolytics may reverse these effects [26]. They are additionally using organ-on-a-chip models to examine how the blood–brain barrier ages.
Metabolic collapse and failure of quality control
In the brain, neuroinflammation manifests as metabolic collapse. Nobel laureate Morten Meldal (University of Copenhagen, Copenhagen, Denmark) presented a chemist’s approach to AD, designing “beta-bodies” to physically cap growing amyloid-beta (Aβ) fibrils. His lab also found that once Aβ peptides aggregate, they can catalyze their own further processing, turning the aggregated state itself into a toxic driver [27]. In the context of Parkinson’s disease, Michela Deleidi (Institute Imagine, Paris, France) used advanced human midbrain organoids to show that alpha-synuclein pathology causes a specific energy collapse in the most vulnerable neurons by disrupting mitochondrial NAD+ synthesis [28]. Riekelt Houtkooper (University of Amsterdam, Amsterdam, The Netherlands) provided a more nuanced view of this process, showing that while NAD+ levels drop significantly in the skeletal muscle of older adults [29], they remain stable in whole blood, questioning the use of blood NAD+ as a reliable biomarker, and calling for more clinical studies to strengthen the evidence for a role of NAD+ in human aging [30].
Ana Maria Cuervo (Albert Einstein College of Medicine, New York, NY, USA) detailed her work on chaperone-mediated autophagy (CMA), a selective lysosomal protein degradation pathway that declines with age. Using a novel fluorescent reporter mouse, her lab mapped CMA activity system-wide and showed overall CMA decline with age more pronounced in males [31]. They also found profound sex differences in what proteins are degraded by CMA in neurons [32]. Her team also developed a “CMA score” for humans, finding that it is low in patients with AD. Her lab has also developed small-molecule CMA activators [33] that successfully clear tau pathology and improve cognition in animal models of dementia [32].
Evandro Fang (University of Oslo, Oslo, Norway) argued that a decline in mitophagy, the recycling of damaged mitochondria, is a key driver of AD, leading to the buildup of A-beta and tau [34]. His lab found that the autophagy protein ULK1 is progressively reduced in AD patients. He demonstrated that NAD+ precursors and Urolithin A can restore mitophagy and improve memory in AD animal models, partly by reactivating autophagy genes via an NAD+-Sirt1-REST pathway. This work is now moving into clinical trials, including a Phase 2 trial for Urolithin A in AD patients [35].
This metabolic vulnerability is exacerbated by a mechanism highlighted by Luigi Ferrucci (National Institute on Aging, Baltimore, MD, USA): a systemic state of chronic cellular hypoxia driven by vascular and mitochondrial decline. He demonstrated that age-related vascular decline starves tissues of oxygen, accelerating epigenetic aging in mice, one such example of how the failure of one system can drive aging across the entire body [36]. Finally, Anne Brunet (Stanford University, Stanford, CA, USA) showcased her lab’s use of the turquoise killifish and longitudinal multi-omics to move beyond static snapshots. Her work maps the dynamic, interwoven trajectories of an individual’s lifespan, asking a critical new question: can we identify the tipping points where systemic decline becomes irreversible? [37].
Can we predict the future? The quest for high-resolution clocks and biomarkers
To intervene in aging, we must first learn to measure it with precision. The conference showcased an explosion of new tools moving beyond chronological age to quantify biological age with unprecedented resolution, asking not just “how old are you?” but “how fast are you aging,” and “in which tissues?”.
A major leap forward is the development of organ-specific clocks. Tony Wyss-Coray (Stanford University, Stanford, CA, USA) demonstrated that by using plasma proteomics, his team can calculate an “age gap” for individual organs. These gaps are highly predictive: an accelerated “brain age” forecasts AD risk 15 years later, while a signature of synaptic proteins in the blood outperforms traditional biomarkers for predicting cognitive decline [38]. Similarly, Vadim Gladyshev (Harvard Medical School, Boston, MA, USA) developed proteomic clocks that predict organ-specific diseases and mortality, and reported the discovery that menopause triggers an abrupt, multi-organ acceleration of biological age [39]. Diving deeper into metabolism, Peter Mullen (University of Southern California, Los Angeles, CA, USA) profiled 12 different organs in mice to reveal that each tissue has a unique metabolic aging trajectory, allowing for the creation of clocks that identify key metabolic drivers of age in specific locations [40].
Epigenetic clocks are becoming more sophisticated. Steve Horvath (Altos Labs, San Diego, CA, USA) highlighted a new algorithm that can computationally dissect bulk tissue data to estimate the epigenetic age of specific cell types. When applied to the brains of patients with AD, it revealed that the age acceleration signal was coming specifically from neurons, a finding previously obscured in bulk tissue analyses [41]. In a novel visual approach, Alexey Terskikh (University of Western Australia, Perth, WA, Australia) introduced a method to capture the “epigenetic face” of a single cell [42]. Instead of sequencing, his lab uses 3D imaging of histone modifications, creating a visual biomarker that can track aging trajectories in immune cells as well as predict hearing loss by mapping an epigenetic gradient in the cochlea. Adding another layer of resolution, Christoph Kuppe (RWTH Aachen University, Aachen, Germany) used spatial multi-omics to map the aging human heart, identifying a previously unknown population of polyploid cardiomyocytes that appears in diseased hearts and drives harmful tissue remodeling [43].
Translating clocks into actionable clinical biomarkers
Translating these clocks to the clinic is the next frontier. Ryan Smith (TruDiagnostic, Lexington, KY, USA) presented landmark data from human trials showing that statins can slow the DunedinPACE clock, while GLP-1 agonists like semaglutide can significantly reverse GrimAge in HIV patients [2, 44]. However, he also issued a critical warning of a “reproducibility crisis” due to technical inconsistencies, introducing a new unified microarray to standardize the field. Looking beyond the lab, researchers are developing more accessible biomarkers. Morten Scheibye-Knudsen (University of Copenhagen, Copenhagen, Denmark) demonstrated that computer vision is a powerful tool to measure effects of aging, for instance histological images can predict future cancer risk [45] and a simple facial photograph is a more powerful predictor of mortality upon hospital admission than any standard clinical test. Meanwhile, Jing-Dong Jackie Han’s lab (Peking University, Beijing, China) is using AI to develop novel clocks, including a single-cell morphological clock called “Morpho-Age,” which they used to screen thousands of chemicals for rejuvenation potential. Alexander Tyshkovskiy (Harvard Medical School, Boston, MA, USA) presented a suite of universal, interpretable transcriptomic aging clocks trained across multiple mammalian species [46]. His novel “mortality clocks” successfully track both aging and the effects of lifespan-modulating interventions, even at the single-cell level.
The quest for reliable biomarkers of aging is rapidly advancing, moving from broad systemic measures to more specific and actionable diagnostics. Nathan LeBrasseur (Mayo Clinic, Rochester, MN, USA) presented his work on developing a panel of circulating biomarkers of cellular senescence [47]. This panel, composed of proteins secreted by senescent cells, has proven to be a powerful predictor of a wide range of age-related health outcomes, including mobility disability, various chronic diseases, and mortality in older adults. His research has shown that these biomarkers are responsive to interventions like caloric restriction and exercise [48] and can help identify individuals who are most likely to benefit from senolytic therapies. LeBrasseur’s team is now leveraging advanced proteomics to discover new, organ-specific biomarkers of senescence, with the goal of creating more tailored diagnostics to assess the efficacy of interventions for specific conditions like idiopathic pulmonary fibrosis or heart failure.
Brian K. Kennedy (National University of Singapore, Singapore, Singapore) discussed the development and application of novel aging clocks. He presented data from a large community-based study that used a DNA methylation clock to assess the effects of various supplements on biological age. His findings suggest that certain interventions, such as time-released alpha-ketoglutarate, may have a significant impact on this biomarker [49]. Kennedy, in collaboration with Jan Gruber and Fong Sheng (National University of Singapore, Singapore, Singapore), also introduced a clinical chemistry-based clock that predicts mortality and functional decline, offering a more actionable tool for clinicians. This clock can identify the specific physiological systems that are driving an individual’s accelerated aging, allowing for more personalized and targeted interventions. He emphasized that improving near-term performance is a practical way to engage people in longevity interventions, since feeling better quickly can motivate sustained lifestyle changes that ultimately support long-term healthspan.
Fabrizio d'Adda di Fagagna (AIRC Institute of Molecular Oncology, Milan, Italy) demonstrated the therapeutic potential of targeting the DNA damage response at telomeres with antisense oligonucleotide designed to inhibit this pathway to reduce senescence, improve hematopoietic stem cell function, and fitness in various mouse models, including geriatric mice [50]. In addition, he offered a thought-provoking perspective on the relationship between different aging clocks. He presented data showing that while telomere shortening in mice leads to severe age-related pathologies, it may not impact some widely used DNA methylation-based aging clocks. This suggests that multiple, parallel aging processes may occur in the body without always intersecting, and that a multi-faceted approach targeting different aspects of aging may be necessary for comprehensive rejuvenation.
The cutting edge of skin diagnostics
On the cutting edge of skin aging diagnostics, Anastasia Georgievskaya (Haut.AI, Tallinn, Estonia) showcased the power of generative AI in creating photographic biomarkers of skin aging [51]. Her company has developed a suite of AI-powered tools that can analyze skin images to quantify a wide range of features, from wrinkles and pigmentation to redness and texture [52]. These tools can then be used to generate hyper-realistic simulations of how an individual’s skin will age under different conditions, such as with or without sun protection, or with changes in lifestyle. This technology is not only valuable for personalizing skincare recommendations but also for creating synthetic data to train more accurate and inclusive diagnostic models. By providing a visual representation of the aging process, Georgievskaya believes that AI can be a tool for educating consumers and motivating them to adopt healthier habits.
Cristiana Banila (Mitra Bio, London, UK) presented a non-invasive approach to tracking skin health through epigenetic biomarkers. Her company has developed a tape-stripping method to collect skin samples from the epidermis, from which they can extract high-quality DNA for methylation analysis [53]. Using these samples, they have built the largest known database of skin epigenetic data and have developed skin-specific aging clocks. These clocks can be used to assess the efficacy of various skin interventions, from topical treatments to laser therapies. Furthermore, Banila’s team is developing epigenetic biomarkers to predict development of specific skin phenotypes, such as wrinkles and hydration, which could provide more clinically relevant endpoints for dermatology trials.
Elias Bou Samra (L’Oréal Research and Innovation, Aulnay-sous-Bois, France) discussed the role of microRNAs as key epigenetic regulators in skin longevity. This research, conducted by Claire Marionnet’s team, identified specific microRNAs that are involved in skin regeneration and differentiation [54], as well as those that are dysregulated in response to sun exposure [55]. This research opens up new avenues for the development of targeted cosmetic and therapeutic interventions that can modulate microRNA expression to promote skin health and combat the signs of aging.
From discovery to design: The next generation of longevity interventions
With a deeper understanding of mechanisms and more precise biomarkers, the field is moving from repurposing old drugs to de novo design of novel therapeutics.
Targeting cellular senescence and inflammation
A key evolution is happening in the world of senolytics. Rather than simply killing senescent cells, researchers are developing more nuanced “senomorphics” that modulate their harmful secretions. Marco Demaria (European Institute for the Biology of Ageing, Groningen, The Netherlands) discovered that detrimental senescent cells rely on the purinergic receptor P2X1 to amplify their own inflammatory signaling [56]. An inhibitor of P2X1 acted as a potent senomorphic, quieting the inflammatory senescence-associated secretory phenotype (SASP) and improving healthspan in multiple animal models without killing senescent cells.
Further, Kotb Abdelmohsen (National Institute on Aging, Baltimore, MD, USA) in collaboration with Yuta Lee (Accelerated Biosciences, Philadelphia, PA, USA), presented findings on the use of secretomes from human trophoblast stem cells (hTSCs) as a potent senomorphic therapy [57]. These embryonic stem cells, ethically sourced from pre-placental tissue of ectopic pregnancies, release a cocktail of factors that can systemically suppress SASP. In preclinical studies, the administration of hTSC-derived secretome or isolated extracellular vesicles to senescent cells reduced the expression of key SASP factors like IL-6 and IL-8, and in aged mice, it lowered circulating levels of pro-aging factors such as GDF-15 and CXCL1. This work suggests that the hTSC secretome may work by restoring DNA repair mechanisms in senescent cells.
Laura Niedernhofer (University of Minnesota, Minneapolis, MN, USA) detailed a high-throughput screening platform to identify novel senolytics. Her team has uncovered several new classes of compounds with senolytic activity, including a novel lipid, epigenetic regulators like LSD1 and BET inhibitors, and a drug targeting mitochondrial function [58]. These discoveries have shown promise in preclinical models, reducing senescence markers, improving the epigenetic landscape, and enhancing physiological function in aged animals. This work underscores the interconnectedness of the hallmarks of aging, as targeting pathways like mitochondrial dysfunction or epigenetic alterations can effectively reduce the burden of senescent cells.
In a similar vein, In Hwa Jang (University of Minnesota, Minneapolis, MN, USA) presented her work on GDF3, a cytokine that promotes inflammation in the adipose tissue of aged mice. Her research revealed that GDF3, primarily secreted by inflammatory adipose tissue macrophages, acts through the SMAD2/3 signaling pathway to alter chromatin accessibility, pushing macrophages towards a more inflammatory state. Notably, genetic deletion or pharmaceutical inhibition of the GDF3-SMAD2/3 axis protected aged mice from endotoxemia-induced inflammation and mortality, a benefit not seen in young mice [59]. This age-specific effect highlights a promising new target for mitigating chronic inflammation in older individuals. From a different angle, Christiane Volbracht (H. Lundbeck A/S, Valby, Denmark) presented a focused knockdown screen to find new targets for tauopathies, identifying the deubiquitinating enzymes ubiquitin-specific proteases USP7 and USP10 as key regulators whose inhibition promotes the clearance of toxic tau aggregates [60].
Generative AI in drug discovery and protein engineering
The power of generative AI is rapidly accelerating drug discovery. In a significant achievement, Fedor Galkin (Insilico Medicine, Abu Dhabi, United Arab Emirates) showcased how their AI platform identified a novel target [61] for Idiopathic Pulmonary Fibrosis (IPF), designed a drug for it [62, 63], and advanced it through a successful Phase 2 trial [64]. The drug, rentosertib, improved lung function in patients and Insilico Medicine is currently inspecting its geroprotective properties with proteomic aging clocks. Based on preliminary results, rentosertib’s trial provides a strong proof-of-concept for developing therapies that treat a specific disease while targeting fundamental aging processes. In an even more futuristic approach, Joe Betts-LaCroix (Retro Biosciences, Redwood City, CA, USA) detailed a collaboration with OpenAI, in which a generative AI model designed entirely new versions of the Yamanaka reprogramming factors. Notably, some of these AI-generated proteins significantly outperformed the originals in reprogramming efficiency, indicating a new application for AI as a creative partner in protein engineering.
Novel chemistries and advanced cellular therapeutics
Xiaodong Liu (Westlake University, Hangzhou, China) addressed a persistent challenge in cellular therapy: iPSCs (induced pluripotent stem cells) frequently retain residual epigenetic memory from their somatic cell of origin, compromising differentiation fidelity, functional maturity, and therapeutic consistency. His team developed a “Transient Naive Treatment” (TNT) strategy, demonstrating that a brief, defined 5-day exposure to naïve pluripotency conditions is sufficient to erase this residual epigenetic signature [65]. This TNT method produces iPSCs that are epigenetically and functionally superior, more equivalent to embryonic stem cells. The lab is now advancing this technology toward the clinic with an autologous iPSC-derived neuron therapy for Parkinson’s disease, which has completed pre-clinical safety and efficacy studies.
Restoring youthful metabolism remains a prime target. Eric Verdin (Buck Institute, Novato, CA, USA) argued against simple NAD+ supplementation, proposing instead to “plug the leak” of NAD+ consumption driven by the enzyme CD38. His lab identified the choroid plexus as a critical hotspot for age-related CD38 expression, linking it to blood-brain barrier dysfunction and neuroinflammation. To counter this, Verdin presented NTX748, a novel and highly potent CD38 inhibitor that restored NAD+ levels, enhanced neuronal plasticity (LTP), and reversed cognitive deficits in aged mice, with Phase 1 clinical trials planned for the near future [66].
Complementing this, Javier María Peralta Ramos (Weizmann Institute of Science, Rehovot, Israel) identified CD38 as an immunometabolic checkpoint on dysfunctional T-cells in individuals with familial AD that, when targeted in an AD mouse model, restored metabolic fitness, improved cognitive performance and reduced neuroinflammation [67]. The focus on female reproductive aging as a key intervention point was highlighted by Yousin Suh (Columbia University, New York, NY, USA), who is leading the VIBRANT clinical trial to test whether the mTOR inhibitor rapamycin can extend reproductive lifespan in women [68]. Broadening the intervention landscape, Edward Rudnic (Maxwell Biosciences, Austin, TX, USA) introduced CLAROMER®, synthetic molecules that mimic innate immune peptides to eliminate multi-drug-resistant infections without harming the gut microbiome, which he termed an “anti-aging bioreactor.”
Are we more than our genes? The dialogue between lifestyle and our internal clocks
The systemic benefits of physical activity were a major focus. Bente Klarlund Pedersen (University of Copenhagen, Copenhagen, Denmark), a pioneer in the field, described how contracting muscle acts as an endocrine organ, secreting hundreds of “myokines” that directly counteract the process of inflammaging [69]. Conversely, her colleague Charlotte Suetta (University of Copenhagen, Copenhagen, Denmark), whose talk “Lying Still, Falling Fast” detailed how physical inactivity, such as during hospitalization, can trigger a rapid acceleration of muscle loss and aging [70]. Lykke Sylow (University of Copenhagen, Copenhagen, Denmark) highlighted the emerging concern that popular GLP-1-based weight-loss drugs can cause significant muscle loss, underscoring the need for interventions like exercise to preserve muscle and metabolic health [71].
Diet is also a powerful lever in aging. The work of Dudley Lamming (University of Wisconsin-Madison, Madison, WI, USA) challenged the “calorie is a calorie” paradigm, showing that restricting a single amino acid, valine, conferred many of the same healthspan benefits as caloric restriction in mice of both sexes, and extended male but not female lifespan [72]. In large human cohorts, Marta Guasch-Ferré (University of Copenhagen, Copenhagen, Denmark) found that adherence to healthy, plant-based dietary patterns was associated with up to an 86% greater chance of healthy aging, while consumption of ultra-processed foods was linked to significantly lower odds [73].
New models and computational approaches
The development of new tools and models accelerates progress in aging research, enabling researchers to move beyond traditional methods to embrace biological complexity. Steven Austad (University of Alabama, Birmingham, AL, USA) advocated for using diverse animal models, including long-lived species and companion animals, over homogenous, short-lived lab mice to improve preclinical translation [74]. Emma Teeling (University College Dublin, Dublin, Ireland) shared her research on the longest-lived mammals for their size: bats. Her team found that long-lived bats defy the typical hallmarks of aging: their telomeres don’t shorten, they upregulate DNA repair with age, and they actively dampen inflammation [75]. Dario Valenzano (Leibniz Institute on Aging, Jena, Germany) uses the short-lived turquoise killifish to model brain aging. His work revealed that intracellular Amyloid beta (Aβ) drives cognitive decline in these fish, and knocking out the precursor protein rescued deficits, suggesting a target for preserving cognitive healthspan [76]. Adding another perspective, Kazuto Kawamura (Max Planck Institute for Biology of Ageing, Cologne, Germany) showed that in C. elegans, the biological age increase induced by starvation is dramatically reversed upon refeeding, a process regulated by the linker histone H1-0 [77]. This suggests that age is more fluid than previously thought.
Todd White (Thalion Initiative, Boston, MA, USA) argued that low preclinical drug success rates (1.2%) stem from insufficient fundamental biological understanding. He described Thalion as an entity funding large-scale projects to generate foundational knowledge. White noted that poor-quality, siloed data limits AI utility. To address this, one of Thalion’s projects, the “Biobank of Mammalian Species,” a $100 million effort, samples 200 mammalian species to provide deep multi-omics open-access data.
Technology for testing interventions is also evolving. Martin Borch Jensen (Gordian Biotechnology, San Francisco, CA, USA) showcased a platform that bypasses artificial lab models entirely by screening hundreds of gene therapies at once, directly in vivo in aged animals with naturally occurring diseases, such as osteoarthritic horses, and a developed disease-modifying osteoarthritis therapy [78]. This approach bridges the gap between discovery and real-world complexity. Rafael de Cabo (National Institute on Aging, Baltimore, MD, USA) presented the Study of Longitudinal Aging in Mice, a large-scale project designed to systematically test interventions in genetically diverse mouse populations to build a predictive framework for translation [79].
Computationally, the field is moving from data analysis to causal reasoning. Kennedy Schaal (Rejuve Bio, Orange, CA, USA) introduced a neuro-symbolic AI that reasons over a “knowledge metagraph” to generate testable, evidence-based hypotheses about aging. Going a step further, Michael Antonov (Deep Origin, Palo Alto, CA, USA) outlined a vision for building an integrated “virtual cell” that combines AI with physics-based simulations to create more robust models for drug discovery.
Maxim Kholin (Gero, Singapore) presented Peter Fedichev's gerophysics theory, which defines aging as resilience loss [80, 81], suggesting that interventions should halt the rate of loss rather than reverse damage to yield the greatest gains in lifespan extension. Paul Kohlhaas (Molecule AG, Basel, Switzerland) discussed Decentralized Science, proposing web3 technologies to create open markets for scientific intellectual property.
From the science of longevity to the medicine of tomorrow
A consensus emerged on strategies to bring anti-aging drugs to market. Sergey Jakimov (Longevity Vision Fund, London, UK) urged researchers to build companies around specific diseases with clear regulatory paths. This disease-first strategy was perfectly exemplified by Insilico Medicine’s drug for IPF [64]. As Alex Zhavoronkov (Insilico Medicine, Cambridge, MA, USA) and Fedor Galkin explained, by achieving a clear clinical endpoint in a devastating disease while also demonstrating a reversal of aging biomarkers, they have created a new template for the field.
Jeroen Aerssens (Rejuvenate Biomed, Diepenbeek, Belgium) provided an update on RJx-01 [82], a combination therapy for sarcopenia that not only improved muscle strength in a Phase 1b trial but also reversed molecular signatures of aging in biopsies, with a Phase 2 trial now underway. Furthering the therapeutic pipeline, Mourad Topors (Repair Biotechnologies, Syracuse, NY, USA) presented a novel strategy for atherosclerosis that targets the buildup of free cholesterol within the liver, a “cholesterol curse”, which hinders the body’s ability to regress atherosclerotic plaque [83] while Rusty Montgomery (BioAge Labs, Richmond, CA, USA) showcased the clinical development of novel NLRP3 inflammasome inhibitors to directly combat inflammaging [84].
Lotte Bjerre Knudsen (Novo Nordisk, Måløv, Denmark) provided a compelling perspective on how a single therapeutic innovation can transform an entire field. She described the journey of GLP-1 receptor agonists, originally developed for diabetes and obesity, but now also recognized for their profound cardiovascular, liver and renal protective effects, as a clear example of how disease-targeted drugs can evolve into systemic, healthspan-promoting therapies [85].
Nobel laureate Michael Levitt (Stanford University, Stanford, CA, USA) analyzed the COVID-19 pandemic as a stress test exposing societal vulnerabilities [86]. Addressing institutional bottlenecks, Niklas Anzinger (Infinita City, Prospera, Roatán, Honduras) cited Eroom’s Law and the regulatory path itself holds back research and clinical development. Niklas’ organization is working directly on alternative regulatory pathways that allow more real-world data collection like the Prospera jurisdiction in Honduras, and Montana’s “Right to Try” bill SB 535 that allows the general population to access Phase 1-cleared therapies.
Bridging preclinical findings to human application
Translating basic aging research into clinical application remains a central challenge. John Newman (Buck Institute, Novato, CA, USA) addressed the challenge of translating basic aging research into clinical practice, describing his efforts to build a translational geroscience program bridging this gap. He highlighted the value of early proof-of-concept trials to de-risk interventions and validate preclinical mechanisms, using his research on ketone bodies as an example [87]. Collin Ewald (Novartis, Cambridge, MA, USA) outlined the company’s strategy to develop regenerative pharmacological interventions targeting diseases of aging mechanisms to restore cell and tissue function in patients, guided by large-scale human data. He highlighted focus areas including mitochondrial health, directing cell fate and cellular rebalancing, DNA repair, extracellular matrix homeostasis, and exercise biology, with translational applications in indications such as neurodegenerative, cardiovascular, metabolic, and immune-related diseases [88]. Andrew Adams (Eli Lilly, Indianapolis, IN, USA) proposed shifting from “sick care” to “health care” via preventative medicines. He cited statins and GLP-1 agonists as examples, suggesting GLP-1 agonists could function as longevity drugs due to their effects on age-related conditions and all-cause mortality. He also emphasized Lilly’s commitment to early intervention in AD.
Finally, Weilan Wang (National University of Singapore, Singapore, Singapore) presented unpublished real-world data on gerotherapeutic drugs in geriatric rehabilitation inpatients. Metformin and ACE inhibitors were associated with significantly lower one-year mortality. Machine learning models suggested these effects may be causal, and drug combinations showed greater benefit than single drugs.
Jamie Justice (XPRIZE, Los Angeles, CA, USA) detailed the $101 million Healthspan competition, a “moonshot” demanding not just the slowing of aging, but a 10-year functional restoration across muscle, cognition, and immunity within a single year. With the top 40 teams now advancing, the competition is driving a wave of multimodal strategies that combine lifestyle interventions with therapeutics, ranging from rapamycin to novel senolytics, to meet these rigorous new clinical endpoints [89].
Commercial, investment, and collaborative strategies
Industry leaders agree on a “disease-first” strategy. Companies must target recognized illnesses like IDF or AD. Repurposing cheap, off-patent drugs like metformin fails mathematically. Phase 3 clinical trials cost hundreds of millions of dollars. Companies cannot recover this money without a patent monopoly. Therefore, the industry tests new, patented drugs for specific diseases. During these trials, researchers simultaneously measure aging biomarkers like epigenetic clocks. This secondary strategy generates the hard numbers of regulators demand. The goal is to force regulators to classify aging as a reimbursable medical condition. This mirrors how objective data transformed obesity from a lifestyle choice into a treated disease.
This regulatory path dictates how investors allocate money. A panel of top investors (Murali Venkatesan, Tom Zuber, Kitsu Egerton, and Daisy Cai) outlined the funding mechanics. Growth-stage capital funds safe, patented platforms. Investors buy portfolios of multiple drugs to avoid the binary risk of a single clinical failure. At the seed stage, investors judge founders entirely on execution speed. The primary barrier to market is insurance reimbursement, not science. Furthermore, medical diagnostics take 10 to 20 years to clear regulators. To survive this delay, capital flows into consumer-facing models. These models bypass doctors and insurers entirely. Founders must build a profitable business today to fund the ultimate goal of systemic aging intervention.
These strict financial rules force biotech startups to partner with massive pharmaceutical companies. Industry leaders (Lisa Melton, Avi Spier, Heinrich Jasper, Kristen Fortney, Aisyah Sjöholm, and Lars Hartenstein) defined this exchange. Large pharma companies use aging biology to find novel targets for existing diseases. This strategy limits their risk and guarantees reproducible data. Startups secure these lucrative partnerships by offering exclusive, long-term human datasets. Large companies need this human data to translate lab results into approved cures. Biological validity is no longer the bottleneck. The actual barrier is a missing “transportable narrative.” Founders lack a simple, compelling story to extract money from investors and governments. Current funding remains disproportionately low relative to the societal burden of aging-related diseases.
Concluding vision and future directions
The 12th ARDD meeting was not merely a showcase of answers; it also generated new, more specific questions. The presentations collectively outlined a future where medicine is proactive rather than reactive, focused on maintaining youthful function rather than managing decline.
The new frontiers are clear. Can we develop a unified theory that connects the deterioration of the genome, the drift of the epigenome, and the loss of systemic resilience? How do we translate the dynamic, multi-omic maps of aging into personalized, combinatorial interventions that can steer an individual’s aging trajectory? What are the ethical frameworks required for a world where we can not only predict disease decades in advance but actively intervene to prevent it?
The research shared in Copenhagen signals that we are moving from the era of understanding aging to the era of engineering longevity (Figure 2). The ultimate challenge is no longer just to add years to life, but to add life to years, and—in doing so—to reimagine the very nature of human potential. The field is now equipped with new models, computational tools, and a clearer understanding of aging’s molecular drivers, positioning it for significant translational progress in the coming decade.
Author Contributions
A.D., S.L., Y.E.Y., and D.B. conceptualized, drafted, and prepared the manuscript. All other authors presented their primary research, data, or industry perspectives at the 12th Aging Research and Drug Discovery (ARDD) meeting, contributed to the intellectual content of their respective summarized sections, and reviewed the final manuscript.
Conflicts of Interest
S.H. is the sole owner of patents and patent applications, directed at epigenetic biomarkers (including GrimAge), licensed to the non-profit Epigenetic Clock Development Foundation, for which he is a founder and paid consultant. S.H. is also a Principal Investigator at Altos Labs, Cambridge Institute of Science. C.Y.E. is a co-founder and shareholder of Avea Life AG and Lichi3 GmbH. D.W.L. has received funding from, and is a scientific advisory board member of, Aeovian. J.C.N. is a co-founder and shareholder in, and inventor on patents licensed to, BHB Therapeutics and Selah Therapeutics, companies that develop ketone-based products and therapies. M.B.J. is a co-founder and shareholder in Gordian Biotechnology. M.L. is supported by National Institutes of Health (NIH) R35GM122543. J.C.I.B. is an employee of Altos Labs. T.T. is a scientific advisor with equity interests in Lime Therapeutics. His spouse is an employee of and has equity in Recursion Pharmaceuticals. The T.T. laboratory receives funding from Ono Pharma related to targeting highly plastic cells in lung cancer. A.M. is a co-founder and shareholder of OvartiX. V.V.C.S. is an employee of Aptah Bio Inc. X.L. is a co-founder of iCamuno Biotherapeutics. T.W.-C. is a co-founder, shareholder and advisor of Teal Rise Inc. and Vero Biosciences Inc. J.A. is an employee of Rejuvenate Biomed NV. Y.L. is a founder of Accelerated Biosciences Corp. B.K.K. is a Board member and advisor to Ponce de Leon Health and a founder and Board member of LiveBeyond. F.d’A.d.F. is a co-founder and shareholder in TAG Therapeutics.
Funding
M.G.F. is supported by the Novo Nordisk Foundation Center for Basic Metabolic Research, which is an independent research center at the University of Copenhagen, partially funded by an unrestricted donation from the Novo Nordisk Foundation (NNF23SA0084103). M.J.S. is supported by the NIH (UG3/UH3 CA275669). J.H.J.H. acknowledges financial support from the European Research Council Advanced Grant Dam2Age, the Oncode Institute (Dutch Cancer Society), the National Institutes of Health (NIH)/National Institute on Aging (NIA) (P01 AG017242; DNA repair, mutations and cell aging), Dutch research organization ZonMW Memorabel (project ID 733050810), and the European Joint Research Project on Rare Diseases (RD20-113, acronym TC-NER). A.M.C. is supported by the National Institutes of Health (NIH) (AG021904, AG054108 and AG031782) and the generous support of the Hevolution Foundation, Freedom Together Foundation (former JPB) and the Rainwaters Foundation. T.A.R. is supported by the US National Institutes of Health (P01 AG36695) and the Milky Way Research Foundation. L.S. is supported by the Novo Nordisk Foundation (NNF24SA0094136, NNF24OC0088663, NNF16OC0023418 and NNF18OC0032082), Independent Research Fund Denmark (4004-00233B and 9039-00170B), the Carlsberg Foundation (CF21-0369), and the Lundbeck Foundation (R467-2024-475). N.K.L. is supported by the NIH (AG062413 and AG089150) and the Glenn Foundation for Medical Research. T.T. is supported by the Mark Foundation for Cancer Research and the Josie Robertson Scholarship at MSKCC. A.H. acknowledges funding from the LEO Foundation (LF15007). A.M. is funded by the Wellcome Trust (302536/Z/23/Z) and UKRI (MR/Y003780/1). V.V.C.S. acknowledges Vesper Biotechnologies, LP (Delaware, US) for providing venture capital that enabled the execution of the experiments, and Ananda Devices Inc. (Quebec, Canada) for co-financing part of the electrophysiology studies. X.L. is supported by the Westlake Education Foundation. F.d’A.d.F.’s laboratory is supported by: ERC advanced grant (TELORNAGING - 835103); ERC POC (TELOVACCINE - 101113229); AIRC-IG (30471); Telethon (GMR23T2007); Next Generation EU, in the context of the National Recovery and Resilience Plan, Investment PE8 Project Age-It and Investment CN3 National Center for Gene Therapy and Drugs based on RNA Technology. R.d.C. and L.F. are supported by the Intramural Research Program of the NIA, NIH.
References
- 1. Alle Q, Le Borgne E, Bensadoun P, Lemey C, Béchir N, Gabanou M, Estermann F, Bertrand-Gaday C, Pessemesse L, Toupet K, Desprat R, Vialaret J, Hirtz C, et al. A single short reprogramming early in life initiates and propagates an epigenetically related mechanism improving fitness and promoting an increased healthy lifespan. Aging Cell. 2022; 21:e13714. https://doi.org/10.1111/acel.13714 [PubMed]
- 2. Lu JY, Tu WB, Li R, Weng M, Sanketi BD, Yuan B, Reddy P, Rodriguez Esteban C, Izpisua Belmonte JC. Prevalent mesenchymal drift in aging and disease is reversed by partial reprogramming. Cell. 2025; 188:5895–911.e17. https://doi.org/10.1016/j.cell.2025.07.031 [PubMed]
- 3. Liu L, Kim S, Buckley MT, Reyes JM, Kang J, Tian L, Wang M, Lieu A, Mao M, Rodriguez-Mateo C, Ishak HD, Jeong M, Wu JC, et al. Exercise reprograms the inflammatory landscape of multiple stem cell compartments during mammalian aging. Cell Stem Cell. 2023; 30:689–705.e4. https://doi.org/10.1016/j.stem.2023.03.016 [PubMed]
- 4. Zhuang X, Wang Q, Joost S, Ferrena A, Humphreys DT, Li Z, Blum M, Krause K, Ding S, Landais Y, Zhan Y, Zhao Y, Chaligne R, et al. Ageing limits stemness and tumorigenesis by reprogramming iron homeostasis. Nature. 2025; 637:184–94. https://doi.org/10.1038/s41586-024-08285-0 [PubMed]
- 5. Gyenis A, Chang J, Demmers JJP, Bruens ST, Barnhoorn S, Brandt RMC, Baar MP, Raseta M, Derks KWJ, Hoeijmakers JHJ, Pothof J. Genome-wide RNA polymerase stalling shapes the transcriptome during aging. Nat Genet. 2023; 55:268–79. https://doi.org/10.1038/s41588-022-01279-6 [PubMed]
- 6. Vermeij WP, Dollé ME, Reiling E, Jaarsma D, Payan-Gomez C, Bombardieri CR, Wu H, Roks AJ, Botter SM, van der Eerden BC, Youssef SA, Kuiper RV, Nagarajah B, et al. Restricted diet delays accelerated ageing and genomic stress in DNA-repair-deficient mice. Nature. 2016; 537:427–31. https://doi.org/10.1038/nature19329 [PubMed]
- 7. Soheili-Nezhad S, Ibáñez-Solé O, Izeta A, Hoeijmakers JHJ, Stoeger T. Time is ticking faster for long genes in aging. Trends Genet. 2024; 40:299–312. https://doi.org/10.1016/j.tig.2024.01.009 [PubMed]
- 8. Leal CBQ, Zimmer CGM, Sinatti VVC, Soares ES, Poppe B, de Wiart AC, Chua XY, da Silva RV, Magdesian MH, Rafii MS, Buée L, Bottos RM. Effects of the therapeutic correction of U1 snRNP complex on Alzheimer's disease. Sci Rep. 2024; 14:30085. https://doi.org/10.1038/s41598-024-81687-2 [PubMed]
- 9. Schumacher B, Pothof J, Vijg J, Hoeijmakers JHJ. The central role of DNA damage in the ageing process. Nature. 2021; 592:695–703. https://doi.org/10.1038/s41586-021-03307-7 [PubMed]
- 10. Meyer DH, Schumacher B. Aging clocks based on accumulating stochastic variation. Nat Aging. 2024; 4:871–85. https://doi.org/10.1038/s43587-024-00619-x [PubMed]
- 11. Schumacher B, Gallrein C, Meyer D. Neuron-type specific aging-rate reveals age decelerating interventions preventing neurodegeneration. Res Sq. 2024. https://doi.org/10.21203/rs.3.rs-4360587/v1
- 12. Bujarrabal-Dueso A, Sendtner G, Meyer DH, Chatzinikolaou G, Stratigi K, Garinis GA, Schumacher B. The DREAM complex functions as conserved master regulator of somatic DNA-repair capacities. Nat Struct Mol Biol. 2023; 30:475–88. https://doi.org/10.1038/s41594-023-00942-8 [PubMed]
- 13. Koch Z, Nandi SP, Licon K, Bujarrabal-Dueso A, Meyer DH, Saeed S, Perampalam P, Dick FA, Schumacher B, Alexandrov LB, Ideker T. The DREAM complex links somatic mutation, lifespan, and disease. bioRxiv. 2025; 18:2025.09.15.676396. https://doi.org/10.1101/2025.09.15.676396 [PubMed]
- 14. Ruth KS, Day FR, Hussain J, Martínez-Marchal A, Aiken CE, Azad A, Thompson DJ, Knoblochova L, Abe H, Tarry-Adkins JL, Gonzalez JM, Fontanillas P, Claringbould A, et al, and Biobank-based Integrative Omics Study (BIOS) Consortium, and eQTLGen Consortium, and Biobank Japan Project, and China Kadoorie Biobank Collaborative Group, and kConFab Investigators, and LifeLines Cohort Study, and InterAct consortium, and 23andMe Research Team. Genetic insights into biological mechanisms governing human ovarian ageing. Nature. 2021; 596:393–7. https://doi.org/10.1038/s41586-021-03779-7 [PubMed]
- 15. Shenhar B, Pridham G, De Oliveira TL, Raz N, Yang Y, Deelen J, Hägg S, Alon U. Heritability of intrinsic human life span is about 50% when confounding factors are addressed. Science. 2026; 391:504–10. https://doi.org/10.1126/science.adz1187 [PubMed]
- 16. Simon M, Van Meter M, Ablaeva J, Ke Z, Gonzalez RS, Taguchi T, De Cecco M, Leonova KI, Kogan V, Helfand SL, Neretti N, Roichman A, Cohen HY, et al. LINE1 Derepression in Aged Wild-Type and SIRT6-Deficient Mice Drives Inflammation. Cell Metab. 2019; 29:871–85.e5. https://doi.org/10.1016/j.cmet.2019.02.014 [PubMed]
- 17. Brochard T, McIntyre RL, Houtkooper RH, Seluanov A, Gorbunova V, Janssens GE. Repurposing nucleoside reverse transcriptase inhibitors (NRTIs) to slow aging. Ageing Res Rev. 2023; 92:102132. https://doi.org/10.1016/j.arr.2023.102132 [PubMed]
- 18. Park MD, Yatim N, Zhang J, Cho BA, Yoo SK, Schaefer MM, Chowell D, Puleston DJ, Merad M. Restoring resident tissue macrophages to combat aging and cancer. Nat Aging. 2025; 5:1383–92. https://doi.org/10.1038/s43587-025-00898-y [PubMed]
- 19. Heinz A. Elastic fibers during aging and disease. Ageing Res Rev. 2021; 66:101255. https://doi.org/10.1016/j.arr.2021.101255 [PubMed]
- 20. Heinz A. Elastases and elastokines: elastin degradation and its significance in health and disease. Crit Rev Biochem Mol Biol. 2020; 55:252–73. https://doi.org/10.1080/10409238.2020.1768208 [PubMed]
- 21. Carver CM, Gomez PT, Rodriguez SL, Kachergus JM, Liu Y, Shi J, Tran T, Wang L, Melov S, Thompson EA, Schafer MJ. Senescent and disease-associated microglia are modifiable features of aged brain white matter. Res Sq. 2023; 30:rs.3.rs-3467812. https://doi.org/10.21203/rs.3.rs-3467812/v1 [PubMed]
- 22. Valdivieso K, Rozmaric T, Victorelli S, Jadhav V, Ring NAR, Schädl B, Dworak H, Klinaki E, Lushchak O, Löscher A, Pirius NE, Gutasi A, Ferguson J, et al. Transcription-Independent Induction of Rapid-Onset Senescence is Integral to Healing. Rochester, NY: Social Science Research Network. 2024. https://doi.org/10.2139/ssrn.4766717
- 23. Tsai AP, Henze DE, Ramirez Lopez E, Haberberger J, Dong C, Lu N, Atkins M, Costa EK, Farinas A, Oh HS, Moran-Losada P, Le Guen Y, Isakova A, et al. Spatial and single-cell transcriptomics reveal the reorganization of cerebellar microglia with aging. Cell Rep. 2025; 44:116624. https://doi.org/10.1016/j.celrep.2025.116624 [PubMed]
- 24. Katsimpardi L, Litterman NK, Schein PA, Miller CM, Loffredo FS, Wojtkiewicz GR, Chen JW, Lee RT, Wagers AJ, Rubin LL. Vascular and neurogenic rejuvenation of the aging mouse brain by young systemic factors. Science. 2014; 344:630–4. https://doi.org/10.1126/science.1251141 [PubMed]
- 25. Moigneu C, Abdellaoui S, Ramos-Brossier M, Pfaffenseller B, Wollenhaupt-Aguiar B, de Azevedo Cardoso T, Camus C, Chiche A, Kuperwasser N, Azevedo da Silva R, Pedrotti Moreira F, Li H, Oury F, et al. Systemic GDF11 attenuates depression-like phenotype in aged mice via stimulation of neuronal autophagy. Nat Aging. 2023; 3:213–28. https://doi.org/10.1038/s43587-022-00352-3 [PubMed]
- 26. Abdellaoui S, Katsimpardi L. Neural stem cell secretome: a secret key to unlocking the power of regeneration in the adult and aging brain. Aging Brain. 2025; 8:100144. https://doi.org/10.1016/j.nbas.2025.100144 [PubMed]
- 27. Zhang Y, Borch LA, Fischer NH, Meldal M. Hydrodynamic Control of Alzheimer Aβ Fibrillation with Glucosaminic Acid Containing Click-Cyclized β-Bodies. J Am Chem Soc. 2024; 146:2654–62. https://doi.org/10.1021/jacs.3c12118 [PubMed]
- 28. Schöndorf DC, Ivanyuk D, Baden P, Sanchez-Martinez A, De Cicco S, Yu C, Giunta I, Schwarz LK, Di Napoli G, Panagiotakopoulou V, Nestel S, Keatinge M, Pruszak J, et al. The NAD+ Precursor Nicotinamide Riboside Rescues Mitochondrial Defects and Neuronal Loss in iPSC and Fly Models of Parkinson's Disease. Cell Rep. 2018; 23:2976–88. https://doi.org/10.1016/j.celrep.2018.05.009 [PubMed]
- 29. Janssens GE, Grevendonk L, Perez RZ, Schomakers BV, de Vogel-van den Bosch J, Geurts JMW, van Weeghel M, Schrauwen P, Houtkooper RH, Hoeks J. Healthy aging and muscle function are positively associated with NAD+ abundance in humans. Nat Aging. 2022; 2:254–63. https://doi.org/10.1038/s43587-022-00174-3 [PubMed]
- 30. Vinten KT, Trętowicz MM, Coskun E, van Weeghel M, Cantó C, Zapata-Pérez R, Janssens GE, Houtkooper RH. NAD+ precursor supplementation in human ageing: clinical evidence and challenges. Nat Metab. 2025; 7:1974–90. https://doi.org/10.1038/s42255-025-01387-7 [PubMed]
- 31. Khawaja RR, Martín-Segura A, Santiago-Fernández O, Sereda R, Lindenau K, McCabe M, Macho-González A, Jafari M, Scrivo A, Gomez-Sintes R, Chavda B, Saez-Ibanez AR, Tasset I, et al. Sex-specific and cell-type-specific changes in chaperone-mediated autophagy across tissues during aging. Nat Aging. 2025; 5:691–708. https://doi.org/10.1038/s43587-024-00799-6 [PubMed]
- 32. Khawaja RR, Griego E, Lindenau K, Salek A, Gambardella J, Scrivo A, Monday HR, Bourdenx M, Madero-Pérez J, Khan ZN, Chavda B, Cutler R, Graff S, et al. Chaperone-mediated autophagy regulates neuronal activity by sex-specific remodelling of the synaptic proteome. Nat Cell Biol. 2025; 27:1688–707. https://doi.org/10.1038/s41556-025-01771-1 [PubMed]
- 33. Gomez-Sintes R, Xin Q, Jimenez-Loygorri JI, McCabe M, Diaz A, Garner TP, Cotto-Rios XM, Wu Y, Dong S, Reynolds CA, Patel B, de la Villa P, Macian F, et al. Targeting retinoic acid receptor alpha-corepressor interaction activates chaperone-mediated autophagy and protects against retinal degeneration. Nat Commun. 2022; 13:4220. https://doi.org/10.1038/s41467-022-31869-1 [PubMed]
- 34. Fang EF, Hou Y, Palikaras K, Adriaanse BA, Kerr JS, Yang B, Lautrup S, Hasan-Olive MM, Caponio D, Dan X, Rocktäschel P, Croteau DL, Akbari M, et al. Mitophagy inhibits amyloid-β and tau pathology and reverses cognitive deficits in models of Alzheimer's disease. Nat Neurosci. 2019; 22:401–12. https://doi.org/10.1038/s41593-018-0332-9 [PubMed]
- 35. Hou Y, Chu X, Park JH, Zhu Q, Hussain M, Li Z, Madsen HB, Yang B, Wei Y, Wang Y, Fang EF, Croteau DL, Bohr VA. Urolithin A improves Alzheimer's disease cognition and restores mitophagy and lysosomal functions. Alzheimers Dement. 2024; 20:4212–33. https://doi.org/10.1002/alz.13847 [PubMed]
- 36. Ferrucci L, Wilson DM
3rd , Donegà S, Gorospe M. The energy-splicing resilience axis hypothesis of aging. Nat Aging. 2022; 2:182–5. https://doi.org/10.1038/s43587-022-00189-w [PubMed] - 37. Costa EK, Chen J, Guldner IH, Mboning L, Schmahl N, Tsenter A, Wu MR, Moran-Losada P, Bouchard LS, Wang S, Singh PP, Pellegrini M, Brunet A, Wyss-Coray T. Multi-tissue transcriptomic aging atlas reveals predictive aging biomarkers in the killifish. bioRxiv. 2025; 1:2025.01.28.635350. https://doi.org/10.1101/2025.01.28.635350 [PubMed]
- 38. Oh HS, Le Guen Y, Rappoport N, Urey DY, Farinas A, Rutledge J, Channappa D, Wagner AD, Mormino E, Brunet A, Greicius MD, Wyss-Coray T. Plasma proteomics links brain and immune system aging with healthspan and longevity. Nat Med. 2025; 31:2703–11. https://doi.org/10.1038/s41591-025-03798-1 [PubMed]
- 39. Goeminne LJE, Vladimirova A, Eames A, Tyshkovskiy A, Argentieri MA, Ying K, Moqri M, Gladyshev VN. Plasma protein-based organ-specific aging and mortality models unveil diseases as accelerated aging of organismal systems. Cell Metab. 2025; 37:205–22.e6. https://doi.org/10.1016/j.cmet.2024.10.005 [PubMed]
- 40. Pilley SE, Awad D, Latumalea D, New C, Esparza E, Wang S, Shi X, Zhang L, Unfried M, Lee JH, Schmid E, Mohanty I, Blum JLE, et al. A metabolic atlas of mouse aging. Cell Metab. 2026; 38:399–418.e7. https://doi.org/10.1016/j.cmet.2025.10.016 [PubMed]
- 41. Tong H, Guo X, Jacques M, Luo Q, Eynon N, Teschendorff AE. Cell-type specific epigenetic clocks to quantify biological age at cell-type resolution. Aging (Albany NY). 2024; 16:13452–504. https://doi.org/10.18632/aging.206184 [PubMed]
- 42. Alvarez-Kuglen M, Rodriguez D, Qin H, Ninomiya K, Fiengo L, Farhy C, Hsu WM, Havas A, Feng GS, Roberts AJ, Anderson RM, Serrano M, Adams PD, et al. Imaging-based chromatin and epigenetic age, ImAge, quantitates aging and rejuvenation. Res Sq. 2023; 7:rs.3.rs-3479973. https://doi.org/10.21203/rs.3.rs-3479973/v1 [PubMed]
- 43. Kuppe C, Ramirez Flores RO, Li Z, Hayat S, Levinson RT, Liao X, Hannani MT, Tanevski J, Wünnemann F, Nagai JS, Halder M, Schumacher D, Menzel S, et al. Spatial multi-omic map of human myocardial infarction. Nature. 2022; 608:766–77. https://doi.org/10.1038/s41586-022-05060-x [PubMed]
- 44. Kiessling P, Joodaki M, Pikulska D, Scheidereit E, Cesaro G, Ruiz M, Kühn D, Peng K, Goni O, Foecking S, Samiei S, Liao X, Li K, et al. Polyploid cardiomyocytes define disease-specific transcriptional states in the mammalian heart. bioRxiv. 2026; 2026.01.31.701472. https://doi.org/10.64898/2026.01.31.701472
- 45. Heckenbach I, Powell M, Fuller S, Henry J, Rysdyk S, Cui J, Teklu AA, Verdin E, Benz C, Scheibye-Knudsen M. Deep learning assessment of senescence-associated nuclear morphologies in mammary tissue from healthy female donors to predict future risk of breast cancer: a retrospective cohort study. Lancet Digit Health. 2024; 6:e681–90. https://doi.org/10.1016/S2589-7500(24)00150-X [PubMed]
- 46. Tyshkovskiy A, Kholdina D, Ying K, Davitadze M, Molière A, Tongu Y, Kasahara T, Kats LM, Vladimirova A, Moldakozhayev A, Liu H, Zhang B, Khasanova U, et al. Transcriptomic Hallmarks of Mortality Reveal Universal and Specific Mechanisms of Aging, Chronic Disease, and Rejuvenation. bioRxiv. 2024; 2024.07.04.601982. https://doi.org/10.1101/2024.07.04.601982
- 47. St Sauver JL, Weston SA, Atkinson EJ, Mc Gree ME, Mielke MM, White TA, Heeren AA, Olson JE, Rocca WA, Palmer AK, Cummings SR, Fielding RA, Bielinski SJ, LeBrasseur NK. Biomarkers of cellular senescence and risk of death in humans. Aging Cell. 2023; 22:e14006. https://doi.org/10.1111/acel.14006 [PubMed]
- 48. Aversa Z, White TA, Heeren AA, Hulshizer CA, Saul D, Zhang X, Molina AJA, Redman LM, Martin CK, Racette SB, Huffman KM, Bhapkar M, Khosla S, et al. Calorie restriction reduces biomarkers of cellular senescence in humans. Aging Cell. 2024; 23:e14038. https://doi.org/10.1111/acel.14038 [PubMed]
- 49. Asadi Shahmirzadi A, Edgar D, Liao CY, Hsu YM, Lucanic M, Asadi Shahmirzadi A, Wiley CD, Gan G, Kim DE, Kasler HG, Kuehnemann C, Kaplowitz B, Bhaumik D, et al. Alpha-Ketoglutarate, an Endogenous Metabolite, Extends Lifespan and Compresses Morbidity in Aging Mice. Cell Metab. 2020; 32:447–56.e6. https://doi.org/10.1016/j.cmet.2020.08.004 [PubMed]
- 50. Aguado J, Sola-Carvajal A, Cancila V, Revêchon G, Ong PF, Jones-Weinert CW, Wallén Arzt E, Lattanzi G, Dreesen O, Tripodo C, Rossiello F, Eriksson M, d'Adda di Fagagna F. Inhibition of DNA damage response at telomeres improves the detrimental phenotypes of Hutchinson-Gilford Progeria Syndrome. Nat Commun. 2019; 10:4990. https://doi.org/10.1038/s41467-019-13018-3 [PubMed]
- 51. Bobrov E, Georgievskaya A, Kiselev K, Sevastopolsky A, Zhavoronkov A, Gurov S, Rudakov K, Del Pilar Bonilla Tobar M, Jaspers S, Clemann S. PhotoAgeClock: deep learning algorithms for development of non-invasive visual biomarkers of aging. Aging (Albany NY). 2018; 10:3249–59. https://doi.org/10.18632/aging.101629 [PubMed]
- 52. Georgievskaya A, Tlyachev T, Kiselev K, Hillebrand G, Chekanov K, Danko D, Golodyaev A, Majmudar G. Predicting human chronological age via AI analysis of dorsal hand versus facial images: A study in a cohort of Indian females. Exp Dermatol. 2024; 33:e15045. https://doi.org/10.1111/exd.15045 [PubMed]
- 53. Menendez Vazquez A, Katsanos D, Vasile M, Graham A, Dyster V, Kaveh S, Moqri M, Banila C. Epigenetic age predictors for non-invasive assessment of human skin. NPJ Aging. 2025; 12:11. https://doi.org/10.1038/s41514-025-00314-0 [PubMed]
- 54. Vieu DL, Golebiewski C, Gastaldi C, Foucher A, Mari B, Rezzonico R, Droit A, Dumont M, Bastien P, Bernerd F, Marionnet C. Identification of miR-141 as a Regulator of Epidermal Homeostasis. J Invest Dermatol. 2025; 145:1670–82.e15. https://doi.org/10.1016/j.jid.2024.10.613 [PubMed]
- 55. Srivastava A, Karlsson M, Marionnet C, Bernerd F, Gueniche A, Rawadi CEL, Ståhle M, Sonkoly E, Breton L, Pivarcsi A. Identification of chronological and photoageing-associated microRNAs in human skin. Sci Rep. 2018; 8:12990. https://doi.org/10.1038/s41598-018-31217-8 [PubMed]
- 56. Wang B, Han J, Elisseeff JH, Demaria M. The senescence-associated secretory phenotype and its physiological and pathological implications. Nat Rev Mol Cell Biol. 2024; 25:958–78. https://doi.org/10.1038/s41580-024-00727-x [PubMed]
- 57. Abdelmohsen K, Martindale JL, Rossi M, Shin CH, Pal A, Munk R, Salamini-Montemurri M, Baranzini M, Arends EM, Mustapic M, Lee Y, Lee JN, Popma SH, et al. The Secretome of Human Trophoblast Stem Cells Attenuates Senescence-Associated Traits. Aging Cell. 2026; 25:e70368. https://doi.org/10.1111/acel.70368 [PubMed]
- 58. Zhang L, Pitcher LE, Prahalad V, Niedernhofer LJ, Robbins PD. Recent advances in the discovery of senolytics. Mech Ageing Dev. 2021; 200:111587. https://doi.org/10.1016/j.mad.2021.111587 [PubMed]
- 59. Jang IH, Kruglov V, Cholensky SH, Smith DM, Carey A, Bai S, Nottoli T, Bernlohr DA, Camell CD. GDF3 promotes adipose tissue macrophage-mediated inflammation via altered chromatin accessibility during aging. bioRxiv. 2024; 25:2024.09.23.614375. https://doi.org/10.1101/2024.09.23.614375 [PubMed]
- 60. Volbracht C, Fog K. Deubiquitinating Enzymes Ubiquitin-Specific Proteases 7 and 10 Regulate TAU Aggregation. Int J Mol Sci. 2025; 26:11062. https://doi.org/10.3390/ijms262211062 [PubMed]
- 61. Pun FW, Leung GHD, Leung HW, Liu BHM, Long X, Ozerov IV, Wang J, Ren F, Aliper A, Izumchenko E, Moskalev A, de Magalhães JP, Zhavoronkov A. Hallmarks of aging-based dual-purpose disease and age-associated targets predicted using PandaOmics AI-powered discovery engine. Aging (Albany NY). 2022; 14:2475–506. https://doi.org/10.18632/aging.203960 [PubMed]
- 62. Ren F, Aliper A, Chen J, Zhao H, Rao S, Kuppe C, Ozerov IV, Zhang M, Witte K, Kruse C, Aladinskiy V, Ivanenkov Y, Polykovskiy D, et al. A small-molecule TNIK inhibitor targets fibrosis in preclinical and clinical models. Nat Biotechnol. 2025; 43:63–75. https://doi.org/10.1038/s41587-024-02143-0 [PubMed]
- 63. Aladinskiy V, Kruse C, Qin L, Babin E, Fan Y, Andreev G, Zhao H, Fu Y, Zhang M, Ivanenkov Y, Aliper A, Zhavoronkov A, Ren F. Discovery of Bis-imidazolecarboxamide Derivatives as Novel, Potent, and Selective TNIK Inhibitors for the Treatment of Idiopathic Pulmonary Fibrosis. J Med Chem. 2024; 67:19121–42. https://doi.org/10.1021/acs.jmedchem.4c01580 [PubMed]
- 64. Xu Z, Ren F, Wang P, Cao J, Tan C, Ma D, Zhao L, Dai J, Ding Y, Fang H, Li H, Liu H, Luo F, et al. A generative AI-discovered TNIK inhibitor for idiopathic pulmonary fibrosis: a randomized phase 2a trial. Nat Med. 2025; 31:2602–10. https://doi.org/10.1038/s41591-025-03743-2 [PubMed]
- 65. Buckberry S, Liu X, Poppe D, Tan JP, Sun G, Chen J, Nguyen TV, de Mendoza A, Pflueger J, Frazer T, Vargas-Landín DB, Paynter JM, Smits N, et al. Transient naive reprogramming corrects hiPS cells functionally and epigenetically. Nature. 2023; 620:863–72. https://doi.org/10.1038/s41586-023-06424-7 [PubMed]
- 66. Verdin E, Fang J, Riley R, Schneider K, Perrone R, Kumaar P, King C, Kauwe G, Roberts A, Hormazabal GV, Zhang Y, Millard E, Liu X, et al. CD38 Inhibition Ameliorates Age-Related CognitiveDecline via a Choroid Plexus-Cerebrospinal Fluid-Hippocampus Axis. Res Sq. 2026; 6:rs.3.rs-8330519. https://doi.org/10.21203/rs.3.rs-8330519/v1 [PubMed]
- 67. Peralta Ramos JM, Castellani G, Kviatcovsky D, Croese T, Tsitsou-Kampeli A, Burgaletto C, Abellanas MA, Cahalon L, Phoebeluc Colaiuta S, Salame TM, Kuperman Y, Savidor A, Itkin M, et al. Targeting CD38 immunometabolic checkpoint improves metabolic fitness and cognition in a mouse model of Alzheimer's disease. Nat Commun. 2025; 16:3736. https://doi.org/10.1038/s41467-025-58494-y [PubMed]
- 68. Jin C, Wang X, Yang J, Kim S, Hudgins AD, Gamliel A, Pei M, Contreras D, Devos M, Guo Q, Vijg J, Conti M, Hoeijmakers J, et al. Molecular and genetic insights into human ovarian aging from single-nuclei multi-omics analyses. Nat Aging. 2025; 5:275–90. https://doi.org/10.1038/s43587-024-00762-5 [PubMed]
- 69. Chow LS, Gerszten RE, Taylor JM, Pedersen BK, van Praag H, Trappe S, Febbraio MA, Galis ZS, Gao Y, Haus JM, Lanza IR, Lavie CJ, Lee CH, et al. Exerkines in health, resilience and disease. Nat Rev Endocrinol. 2022; 18:273–89. https://doi.org/10.1038/s41574-022-00641-2 [PubMed]
- 70. Hansen SK, Hansen P, Nygaard H, Grønbæk HD, Berry TW, Olsen CM, Aagaard P, Hvid LG, Agergaard J, Dela F, Suetta C. Five days of bed rest in young and old adults: Retainment of skeletal muscle mass with neuromuscular electrical stimulation. Physiol Rep. 2024; 12:e16166. https://doi.org/10.14814/phy2.16166 [PubMed]
- 71. Carlsson M, Frank E, Màrmol JM, Ali MS, Raun SH, Battey E, Andersen NR, Irazoki A, Lund C, Henríquez-Olguin C, Højfeldt MK, Blomquist P, Bromer FD, et al. Activin receptor type IIA/IIB blockade increases muscle mass and strength, but compromises glycemic control in mice. Mol Metab. 2025; 102:102261. https://doi.org/10.1016/j.molmet.2025.102261 [PubMed]
- 72. Calubag MF, Ademi I, Green CL, Jayarathne HSM, Manchanayake DNH, Le SM, Lialios P, Breuer LE, Yakar S, Babygirija R, Sonsalla MM, Grunow I, Yeh CY, et al. Lifelong restriction of dietary valine has sex-specific benefits for health and lifespan in mice. bioRxiv. 2025; 4:2025.08.31.673254. https://doi.org/10.1101/2025.08.31.673254 [PubMed]
- 73. Tessier AJ, Wang F, Korat AA, Eliassen AH, Chavarro J, Grodstein F, Li J, Liang L, Willett WC, Sun Q, Stampfer MJ, Hu FB, Guasch-Ferré M. Optimal dietary patterns for healthy aging. Nat Med. 2025; 31:1644–52. https://doi.org/10.1038/s41591-025-03570-5 [PubMed]
- 74. Austad S. Animal Models and the Emergence of Geroscience. Innov Aging. 2022; 6:340. https://doi.org/10.1093/geroni/igac059.1345
- 75. Foley NM, Hughes GM, Huang Z, Clarke M, Jebb D, Whelan CV, Petit EJ, Touzalin F, Farcy O, Jones G, Ransome RD, Kacprzyk J, O'Connell MJ, et al. Growing old, yet staying young: The role of telomeres in bats' exceptional longevity. Sci Adv. 2018; 4:eaao0926. https://doi.org/10.1126/sciadv.aao0926 [PubMed]
- 76. de Bakker DEM, Mihaljević M, Gharat K, Richter Y, Bagnoli S, van Bebber F, Adam L, Shamim-Schulze F, Ohlenschläger O, Bens M, Cirri E, Antebi A, Matić I, et al. Amyloid beta precursor protein contributes to brain aging and learning decline in short-lived turquoise killifish (Nothobranchius furzeri). bioRxiv. 2025; 2024.10.11.617841. https://doi.org/10.1101/2024.10.11.617841
- 77. Kawamura K, Diederich AR, Gerisch B, Ripa R, Latza C, Steiner JD, Fernandes S, Artoni F, Meyer DH, Sant D, Oehm S, Grundmann F, Müller RU, et al. Resilience and restoration from fasting-refeeding mediated by a nutrient-regulated linker histone. bioRxiv. 2025; 2025.04.14.648802. https://doi.org/10.1101/2025.04.14.648802
- 78. Borch Jensen M, Marblestone A. In vivo Pooled Screening: A Scalable Tool to Study the Complexity of Aging and Age-Related Disease. Front Aging. 2021; 2:714926. https://doi.org/10.3389/fragi.2021.714926 [PubMed]
- 79. Palliyaguru DL, Vieira Ligo Teixeira C, Duregon E, di Germanio C, Alfaras I, Mitchell SJ, Navas-Enamorado I, Shiroma EJ, Studenski S, Bernier M, Camandola S, Price NL, Ferrucci L, de Cabo R. Study of Longitudinal Aging in Mice: Presentation of Experimental Techniques. J Gerontol A Biol Sci Med Sci. 2021; 76:552–60. https://doi.org/10.1093/gerona/glaa285 [PubMed]
- 80. Pyrkov TV, Avchaciov K, Tarkhov AE, Menshikov LI, Gudkov AV, Fedichev PO. Longitudinal analysis of blood markers reveals progressive loss of resilience and predicts human lifespan limit. Nat Commun. 2021; 12:2765. https://doi.org/10.1038/s41467-021-23014-1 [PubMed]
- 81. Fedichev P, Gruber J. A minimal model explains aging regimes and guides intervention strategies. bioRxiv. 2025; 2025.08.25.671954. https://doi.org/10.1101/2025.08.25.671954
- 82. Tezze C, Amendolagine FI, Nogara L, Baraldo M, Ciciliot S, Arcidiacono D, Zaramella A, Masiero G, Ferrarese G, Realdon S, Blaauw B, Detienne G, Beliën AT, et al. A combination of metformin and galantamine exhibits synergistic benefits in the treatment of sarcopenia. JCI Insight. 2023; 8:e168787. https://doi.org/10.1172/jci.insight.168787 [PubMed]
- 83. Strough G, Warren I, Sahbani K, Inyang K, Li J, Hickey B, Plencner S, Honkanen R, Bhatt D, Khan B, Reason, Topors M. Abstract 4365849: Selective Degradation of Excess Free Cholesterol in the Liver by REP-0003 Regresses Atherosclerotic Plaque in Ldlr-/- Mice: A Novel Approach for HoFH. Circulation. 2025; 152:A4365849. https://doi.org/10.1161/circ.152.suppl_3.4365849
- 84. Torp J, Ferber D, Buthmann H, Hagelueken G, Deshpande A, Hartman G, Hughes RE, Salazar T, Tronnes S, Duisembekova A, Marleaux M, Hochheiser IV, Coll RC, et al. Inhibition of NLRP3 by a CNS-penetrating indazole scaffold. bioRxiv. 2025; 2025.07.01.662566. https://doi.org/10.1101/2025.07.01.662566
- 85. Knudsen LB, Lau J. The Discovery and Development of Liraglutide and Semaglutide. Front Endocrinol (Lausanne). 2019; 10:155. https://doi.org/10.3389/fendo.2019.00155 [PubMed]
- 86. Ioannidis JPA, Zonta F, Levitt M. Variability in excess deaths across countries with different vulnerability during 2020-2023. Proc Natl Acad Sci U S A. 2023; 120:e2309557120. https://doi.org/10.1073/pnas.2309557120 [PubMed]
- 87. Newman JC, Covarrubias AJ, Zhao M, Yu X, Gut P, Ng CP, Huang Y, Haldar S, Verdin E. Ketogenic Diet Reduces Midlife Mortality and Improves Memory in Aging Mice. Cell Metab. 2017; 26:547–57.e8. https://doi.org/10.1016/j.cmet.2017.08.004 [PubMed]
- 88. Statzer C, Park JYC, Ewald CY. Extracellular Matrix Dynamics as an Emerging yet Understudied Hallmark of Aging and Longevity. Aging Dis. 2023; 14:670–93. https://doi.org/10.14336/AD.2022.1116 [PubMed]
- 89. XPRIZE Healthspan Competition. XPRIZE Foundation. XPRIZE. https://www.xprize.org/competitions/healthspan.



