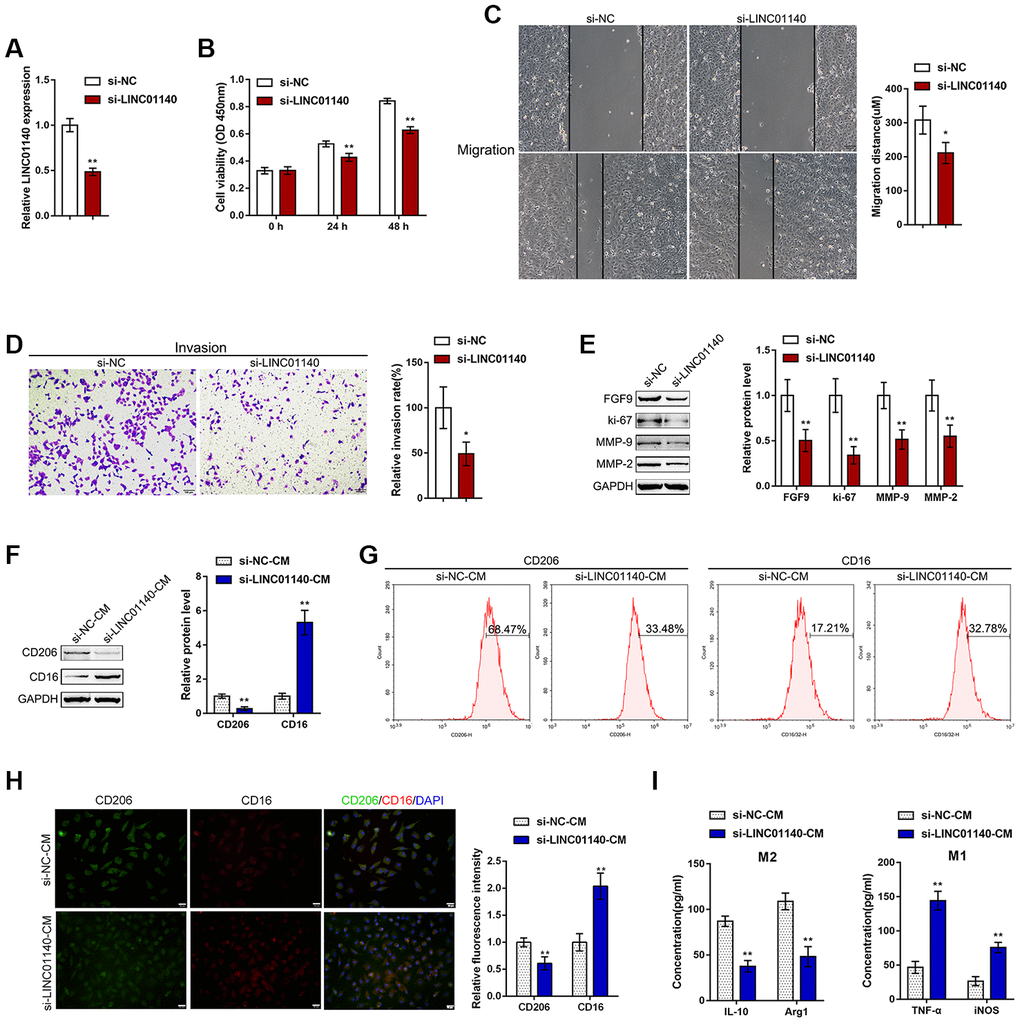
Figure 4.Effects of LINC01140 on bladder cancer cell aggressiveness and macrophage M2 polarization. (A) LINC01140 knockdown was generated in the T24 bladder cancer cell line by transfection with si-LINC01140. The transfection efficiency was validated by real-time PCR (P<0.01, student’s T test). Next, T24 cells were transfected with si-LINC01140 and examined for (B) cell viability by MTT assay, **P<0.01, one-way ANOVA test.; (C) migration capacity by wound healing assay, P<0.05, student’s T test; (D) invasive capacity by Transwell assay, P<0.05, student’s T test; and (E) protein levels of FGF9, ki-67, MMP-2, and MMP-9 by immunoblotting, P<0.01, student’s T test. T24 cells were transfected with si-LINC01140 or si-NC (negative control) and the culture medium (shown in the figures as conditioned medium, si-NC-CM and si-LINC01140-CM) was collected for macrophage incubation. Monocytes were treated with 50 ng/ml M-CSF to stimulate monocyte differentiation into M0 macrophages. M0 macrophages were divided into four groups: IL-4 (M2 polarization inducing) + si-NC-CM, IL-4 (M2 polarization inducing) + si-LINC01140-CM, LPS + IFNγ (M1 polarization inducing) + si-NC-CM, and LPS + IFNγ (M1 polarization inducing) + si-LINC01140-CM, and examined for (F) the protein levels of CD206 and CD16 by immunoblotting, P<0.01, student’s T test; (G) the percentage of CD206 and CD16-positive cells was determined by flow cytometry; (H) the inflorescence intensity of CD206 and CD16 were measured by IF staining, The inflorescence intensity is shown in the right panel. P<0.01, student’s T test. (I) The concentrations of IL-10, Arg1, iNOS, and TNF-α in the macrophage culture medium was determined by ELISA. **P<0.01, student’s T test.