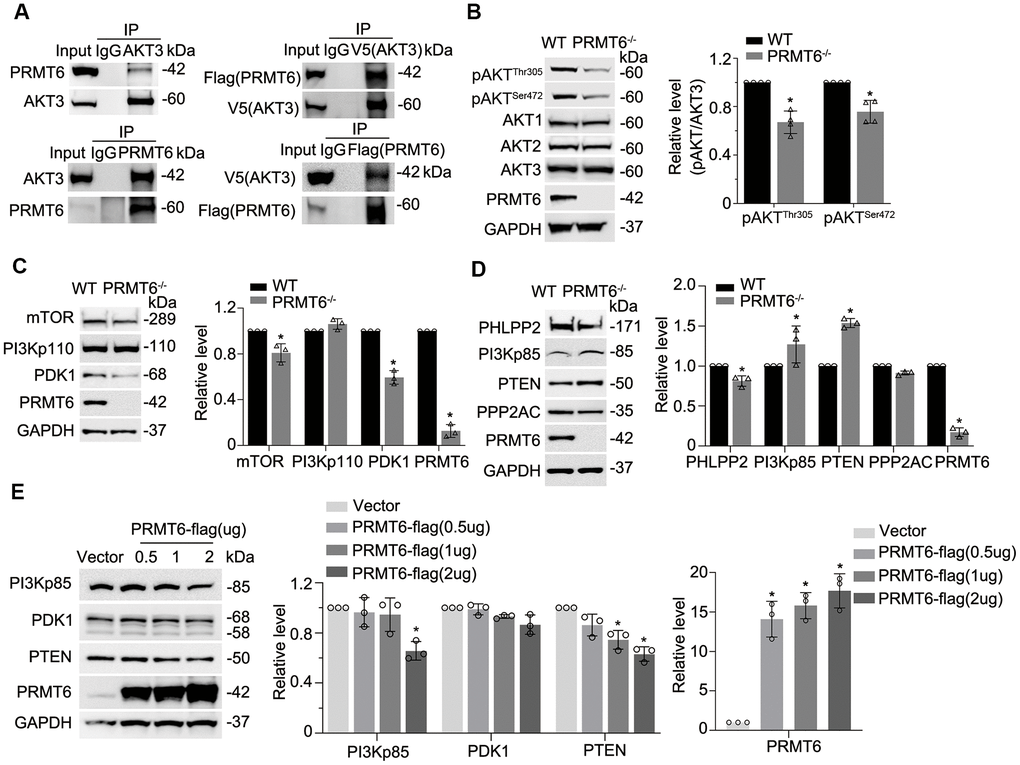
Figure 3.Genetic depletion of PRMT6 results in AKT dephosphorylation via impairment of PI3K-AKT signaling. (A) BEAS-2B cell lysates were immunoprecipitated with PRMT6 or AKT3 antibody, and the immunoprecipitates were analyzed with AKT3 and PRMT6 immunoblotting as indicated (left two panels). pcDNA3.1D-PRMT6-FLAG and pcDNA3.1D-AKT3-V5 plasmids were co-transfected into BEAS-2B cells. After 48h of transfection, cell lysates were immunoprecipitated with FLAG or V5 antibody, and the immunoprecipitants were analyzed with V5 and FLAG immunoblotting as indicated (right two panels). (B) PRMT6 CRISPR/Cas9 KO plasmid and HDR plasmid were applied to establish stable PRMT6 gene knockout BEAS-2B cell line. The knockout efficiency was determined by immunoblotting. The cell lysates of wild type (WT) and PRMT6 stable knockout BEAS-2B cell (PRMT6-/-) were applied for pAKTThr305, pAKTSer472, AKT isoforms 1, 2, 3 and PRMT6 immunoblotting (left panel). Plotted densitometry results of the pAKTThr305 and pAKTSer472 in WT and PRMT6 knockout group were presented (right panel). (C,D), Cell lysates of WT and PRMT6-/- BEAS-2B cells were collected and immunoblotted with indicated antibodies. At the right panel of each figure, the plotted densitometry results were presented. (E) pcDNA3.1D-His-V5 control plasmid (Vector) and pcDNA3.1D-PRMT6-V5 plasmids were delivered into BEAS-2B cells via electroporation. After 48h, the cell lysates were collected and immunoblotted with indicated antibodies. The plotted densitometry of PI3Kp85, PDK1, PTEN and PRMT6 were presented (middle and right panel). Values represent mean ± SD and “*” denotes p < 0.05. Results were representative of at least n=3 experiments.