Introduction
Aging involves a progressive decline of physiological functions in various organs, influenced by several factors, including genetic factors and environmental factors [1–3]. As the aged population has been growing rapidly around the world, the therapeutic and preventive approaches to decelerate senescence are of great concern. Among the features of aging, the decline in immune function has been widely examined, because it results in chronic low grade inflammation, which is a major risk factor for the incidence and prevalence of age-related diseases, including infectious diseases, tumors, and neurodegenerative diseases [4–8].
The retina, one of the neural tissues, is also affected by chronic low grade inflammation. Age-related retinal neurodegenerative diseases, such as age-related macular degeneration (AMD), are major causes of blindness in the elderly [9–12]. The disease is caused by age-related retinal cell loss, including retinal ganglion cell (RGC) death [13] and photoreceptor cell death [14,15], at least partly due to chronic inflammation [16–19]. Several therapeutic pharmacological agents for suppression of retinal diseases have been reported [20,21]. However, human eyes are exposed to daily chronic stress, such as photo-oxidative stress, and as a result, safe and long-term approaches based on diet to mitigate retinal chronic inflammation are especially attractive.
Age-related immune dysfunctions leading to chronic inflammation have been previously reported. Thymic involution and disruption of homeostatic T cell proliferation, including decreased numbers of naïve T cells, accumulation of memory T cells, and increased numbers of regulatory T cells (Tregs), have been studied [22–24], and altered numbers of B cells with aging, reduced antibody production, and age-related dysfunction of other innate immune cells have also been reported [25–30]. Although some food materials or constituents, for example, prebiotics and probiotics, can improve age-related immune defects [31–34], their mechanism remains poorly understood. Recent studies suggested that the gut microbiota composition may be associated with age-related immune dysfunctions [35–37]. Disruption of gut microbiota composition has been also implicated in retinal diseases, including AMD, through a gut-retina axis [38]. Therefore, preventive dietary approaches involving alterations of gut microbiota composition for improving age-related retinal chronic inflammation should be studied.
Lactic acid bacteria are widely consumed as probiotics and paraprobiotics to enhance gut barrier function and improve immune systems. Studies have also demonstrated functional roles of several lactic acid bacterial strains in humans, including for the prevention of diarrhea, allergies, and metabolic disorders [39]. However, the long-term effects of lactic acid bacteria on age-related chronic inflammation remain unclear. We previously reported that Lactobacillus paracasei KW3110 activated macrophages and suppressed excessive inflammation in mice and humans [40–43]. In this study, we demonstrated the suppressive effects of the long-term intake of L. paracasei KW3110 on age-related alterations of gut microbiota composition and expansion of inflammatory CD4-positive T cells in the lamina propria of the small intestine (SI-LP). Furthermore, we also revealed the protective effects of the long-term intake of L. paracasei KW3110 on age-related retinal cell loss. We proposed that the long-term intake of L. paracasei KW3110 contributed to the prevention of chronic inflammation and age-related retinal cell loss in physiologically aged mice.
RESULTS
Intake of L. paracasei KW3110 affected bacterial flora in aged mice
The gut microbiota plays a critical role in the immune system, and aging has been reported to alter gut bacterial flora composition [35]. Previous studies have reported that some prebiotics and probiotics can alter gut bacterial flora composition and improve immune defects [44,45]. Therefore, to investigate whether intake of L. paracasei KW3110 affected the gut microbiota composition in aged mice, 16-month-old mice were fed a diet with or without L. paracasei KW3110 for 6 months. We analyzed bacterial 16S ribosomal RNA gene sequences in the feces. The microbiota composition at the phylum level revealed that the Firmicutes/Bacteroidetes ratio was lower in aged mice fed a control diet than in young mice fed a control diet (Fig. 1A, B). This result is consistent with a previous report [46]. However, in aged mice fed a diet containing L. paracasei KW3110 for 6 months, the Firmicutes/Bacteroidetes ratio was decreased compared with that in age-matched control mice (Fig. 1B).
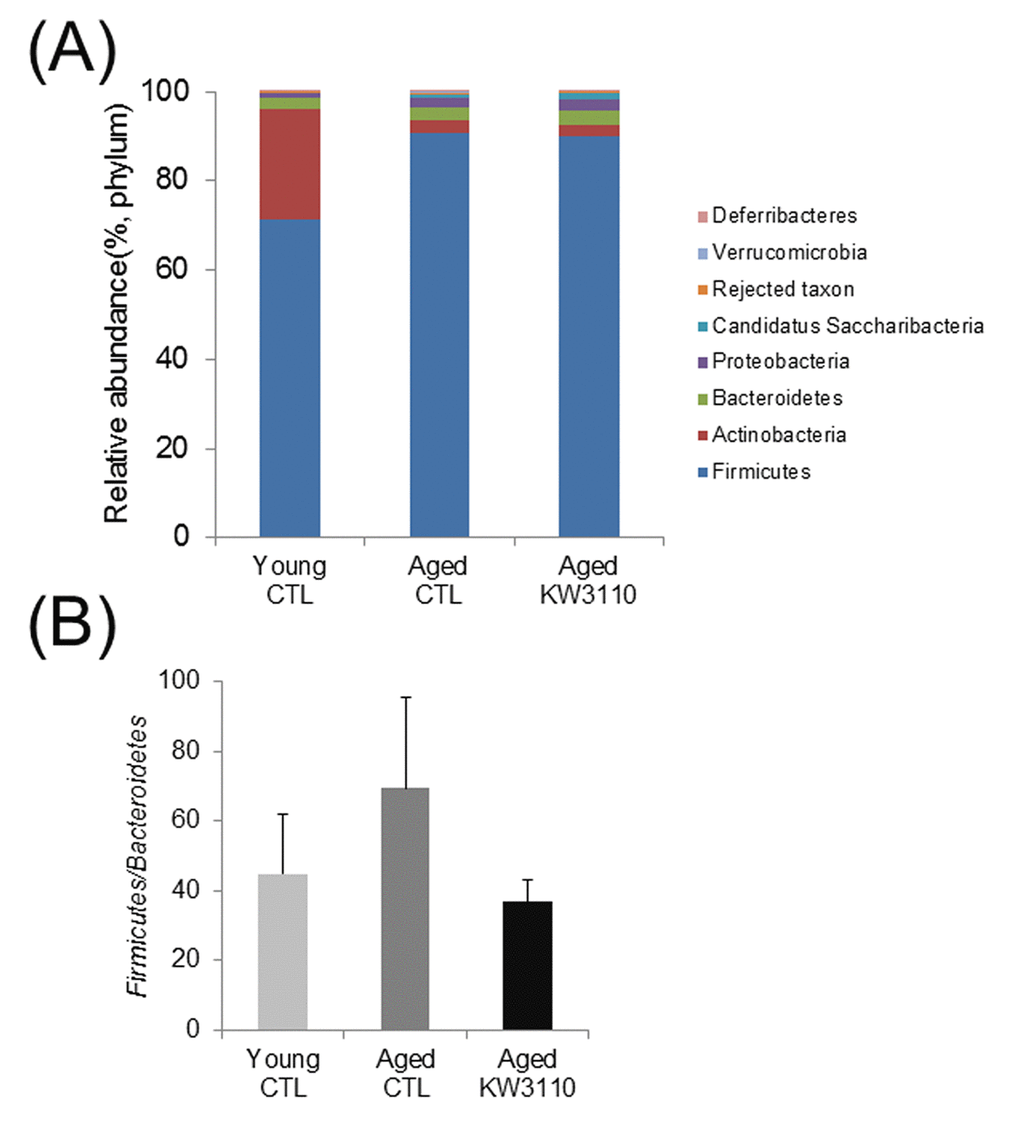
Figure 1AB. The intake of Lactobacillus paracasei KW3110 in aged mice affected the gut microbial composition. Feces were collected and subjected to flora analysis in young (3-months-old) and aged mice (22-months-old). (A) Distribution of gut microbiota (% of total 16S rDNA) at the phylum level. (B) Comparison of the Firmicutes to Bacteroidetes ratio. Values are presented as the means ± SEM of relative abundance of each phylum.
At the bacterial family level, the bacterial ratios in the feces were altered in aged mice as compared with young control mice (Fig. 1C). In the aged mice groups, the intake of L.paracasei KW3110 affected some bacterial abundances. For example, the mean relative abundances of Peptostreptococcaceae (p = 0.011) and Bifidobacteriaceae (p = 0.038) were significantly higher in aged mice fed a diet containing L. paracasei KW3110 for 6 months than in age-matched mice fed a control diet (Fig. 1D). In contrast, the mean relative abundance of Streptococcaceae was significantly lower (p = 0.0079) in aged mice fed a diet containing L. paracasei KW3110 than in age-matched mice fed a control diet (Fig. 1D).
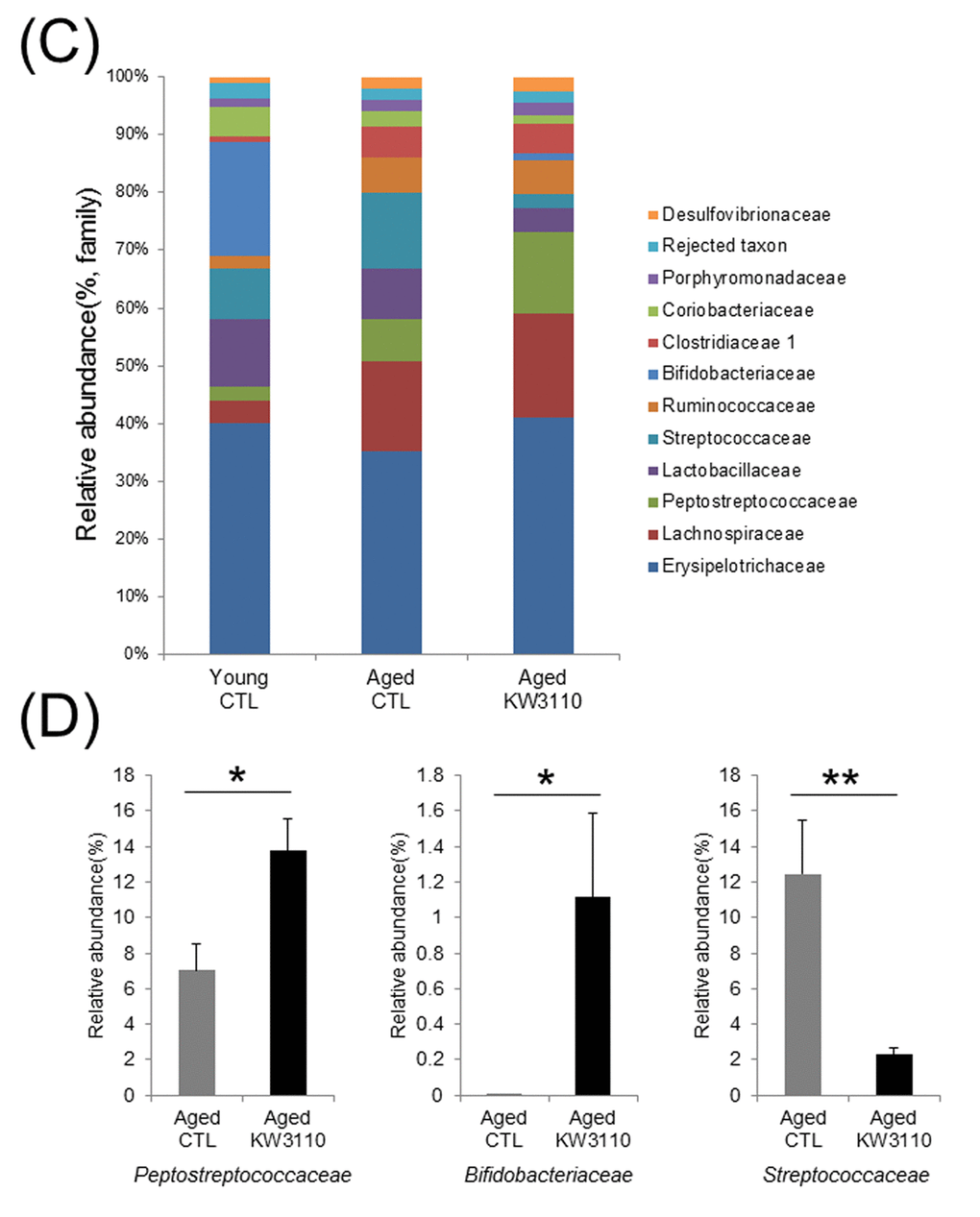
Figure 1CD. The intake of Lactobacillus paracasei KW3110 in aged mice affected the gut microbial composition. (C) Distribution of gut microbiota (% of total 16S rDNA) at the family level. Families with proportions less than 1% are not listed. (D) Comparisons of relative abundances of Peptostreptococcaceae (left panel), Bifidobacteriaceae (middle panel), and Streptococcaceae (right panel) families. Values are presented as the means ± SEM. Significance was assumed if the p value was < 0.05. *p < 0.05, **p < 0.01.CTL = control diet; KW3110 = Lactobacillus paracasei KW3110 diet
The intake of L. paracasei KW3110 affected the lymphocyte subpopulation of SI-LP in aged mice
We have previously shown that orally-provided L. paracasei KW3110 interacted with immune cells in the small intestine [43]. In addition, intake of L. paracasei KW3110 altered gut bacterial flora composition in aged mice (Fig. 1). Thus, to examine the effects of L. paracasei KW3110 on the immune system in the small intestine with aging, 11-month-old mice were fed a diet with or without L. paracasei KW3110 for 6 months. The ratio of CD3ε- and CD4-double positive T cells to live cells and the ratio of interferon-γ (IFN-γ)-producing CD4-positive T cells to CD4-positive T cells, known as indicators of age-related inflammation in SI-LP cells, in aged mice fed a control diet, was higher than that in control young mice (Fig. 2B, D). The expression of programmed cell death protein 1 (PD-1), known as an indicator of immune senescence in CD4-positive T cells, in aged mice fed a control diet, was also higher than that in control young mice (Fig. 2E). However, the intake of L. paracasei KW3110 for 6 months in aged mice significantly decreased the ratio of CD3ε- and CD4-double-positive T cells to live cells in SI-LP (Fig. 2A, B), the ratio of IFN-γ-producing CD4-positive T cells to CD4-positive T cells (Fig. 2C, D), and the expression of PD-1 in CD4-positive T cells (Figure 2E). In contrast, the ratio of CD4- and Foxp3-positive cells, known as regulatory T cells, in SI-LP was not changed in aged mice fed a diet either with or without L. paracasei KW3110 (Supplementary Fig. 1).
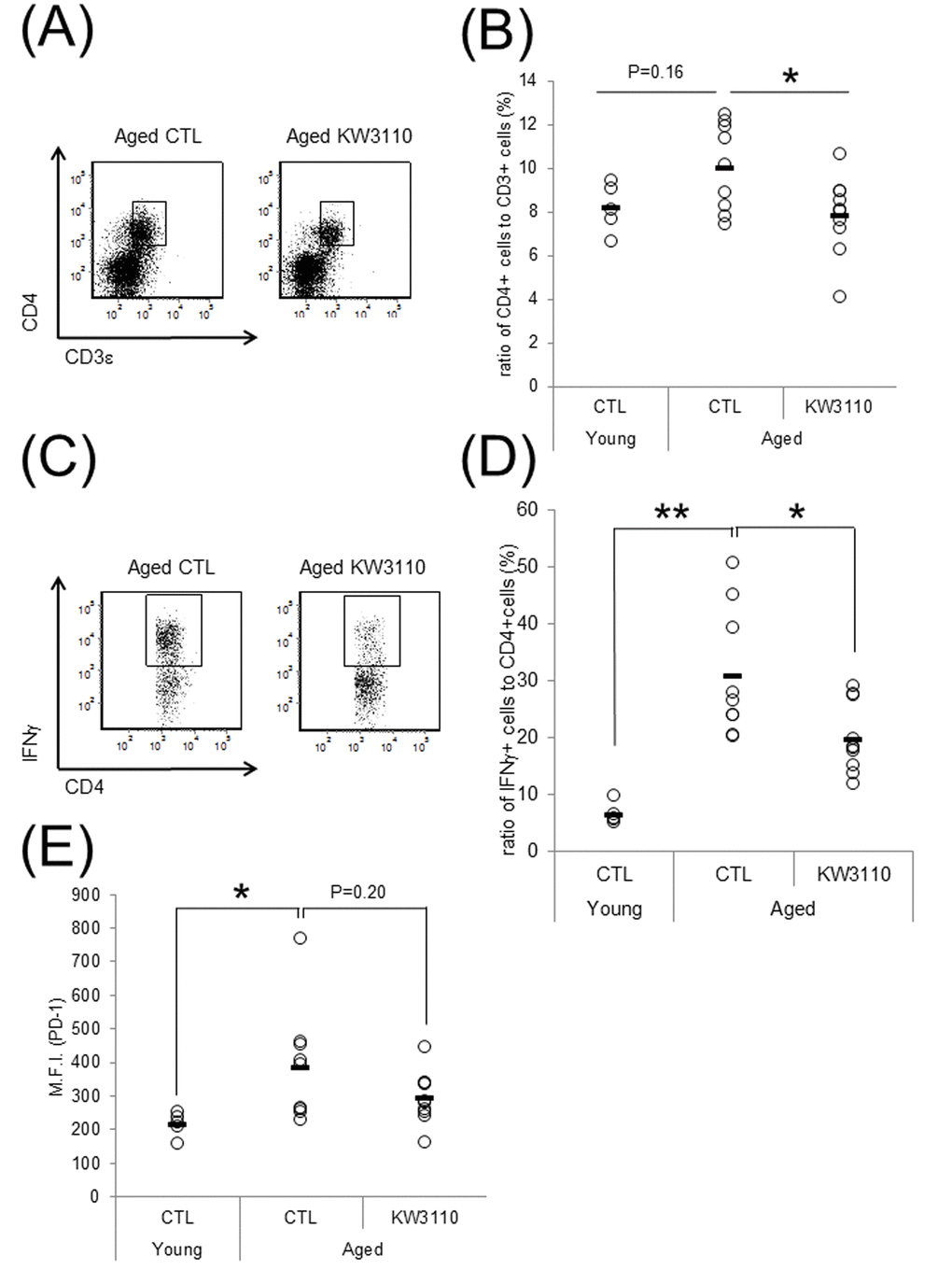
Figure 2. Intake of Lactobacillus paracasei KW3110 suppressed the inflammatory CD4-positive T cell expansion in the lamina propia of the small intestine (SI-LP). (A and B) To detect inflammatory cytokine-producing cells, SI-LP cells from young mice (3-months-old) and aged mice (17-months-old) were cultured under stimulation with Leukocyte Activation Cocktail plus BD GolgiPlug, and analyzed by flow cytometry. (A) Representative data of CD4-positive cells from aged mice fed a diet with (KW3110) or without (CTL) L. paracasei KW3110. (B) The ratio of CD3ε- and CD4-positive to live cells. (C) Representative data of CD4- and interferon gamma (IFN-γ)-positive cells from aged mice fed a diet with or without L. paracasei KW3110. (D) The ratio of CD4- and IFN-γ-positive cells to CD4-positive cells. (E) The expressions of programmed cell death protein 1 (PD-1) in CD3ε- and CD4-positive cells were analyzed by flow cytometry. M.F.I. indicates mean fluorescence intensity.
Intake of L. paracasei KW3110 decreased the levels of proinflammatory cytokines and chemokines in serum
The age-related inflammatory phenotypes in various tissues are associated with the serum levels of proinflammatory cytokines, which are produced from inflammatory immune cells. Therefore, we evaluated serum levels of proinflammatory cytokines and chemokines in aged mice. As shown in Fig. 3, the serum levels of proinflammatory cytokines and chemokines in aged mice were higher than those in control young mice (Fig. 3). Interestingly, serum levels of some cytokines and chemokines, interleukin-17 (IL-17), keratinocyte chemoattractant (KC), and interleukin-13 (IL-13), were significantly lower in aged mice fed a diet containing L. paracasei KW3110 from 16 months of age to 22 months of age for 6 months, than in age-matched mice fed a control diet (Fig. 3). The concentrations of the other proinflammatory cytokines were also lower in aged mice fed a diet containing L. paracasei KW3110 for 6 months. These changes of proinflammatory cytokine levels could be observed in aged mice fed a diet containing L. paracasei KW3110 only for 2 months (Supplementary Fig. 2).
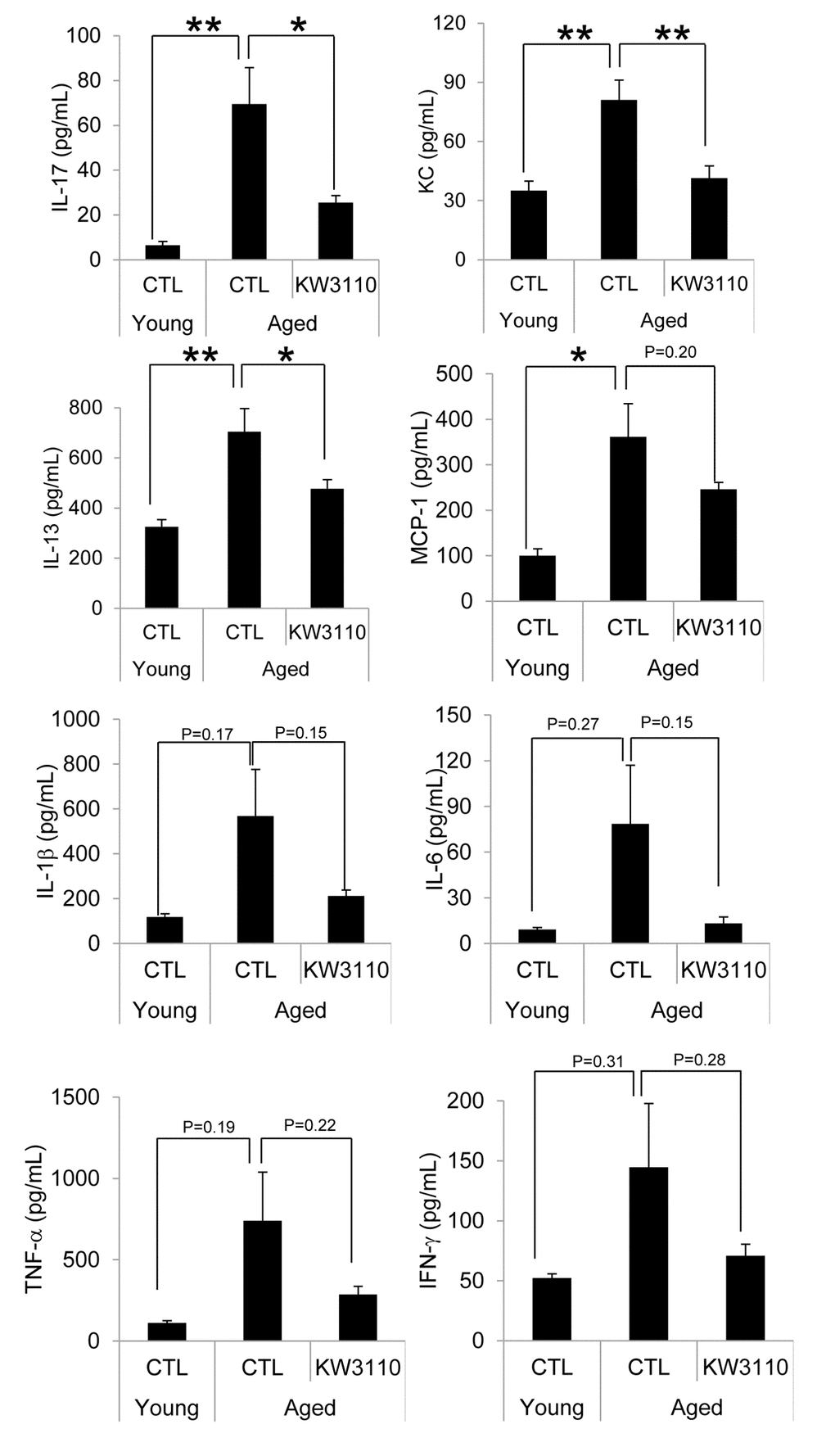
Figure 3. The levels of proinflammatory cytokines in the serum were mitigated in aged mice fed a diet containing Lactobacillus paracasei KW3110 as compared with age-matched control mice. Serum was collected and subjected to multiplex analyses to determine levels of cytokines (IL-1β, IL-6, IL-13, IL-17, IFN-γ, TNF-α, KC, and MCP1) in young (3-months-old) and aged mice (22-months-old). Values are presented as the means ± SEM. Significance was assumed if the p value was < 0.05. *p < 0.05; **p < 0.01. CTL = control diet; KW3110 = Lactobacillus paracasei KW3110 diet; IL = interleukin; IFN = interferon; TNF = tumor necrosis factor; KC = keratinocyte chemoattractant; MCP1 = monocyte chemoattractant protein 1.
Intake of L. paracasei KW3110 mitigated retinal inflammation
As the lower serum levels of proinflammatory cytokines in aged mice were seemingly related to the suppression of age-related inflammatory phenotypes in peripheral tissues, we investigated whether intake of L. paracasei KW3110 also mitigated age-related retinal inflammation. Intake of L. paracasei KW3110 from 11–17 months of age for 6 months, in aged mice, significantly decreased the expression of IFN-γ and interleukin-6 (IL-6) in CD11b-positive and F4/80-positive retinal immune cells and macrophage (Fig. 4A-C) as compared with in age-matched mice fed a control diet. The expression of tumor necrosis factor-α (TNF-α) in CD11b-positive and F4/80-positive retinal macrophage was also lower than in age-matched mice fed a control diet (Fig. 4D).
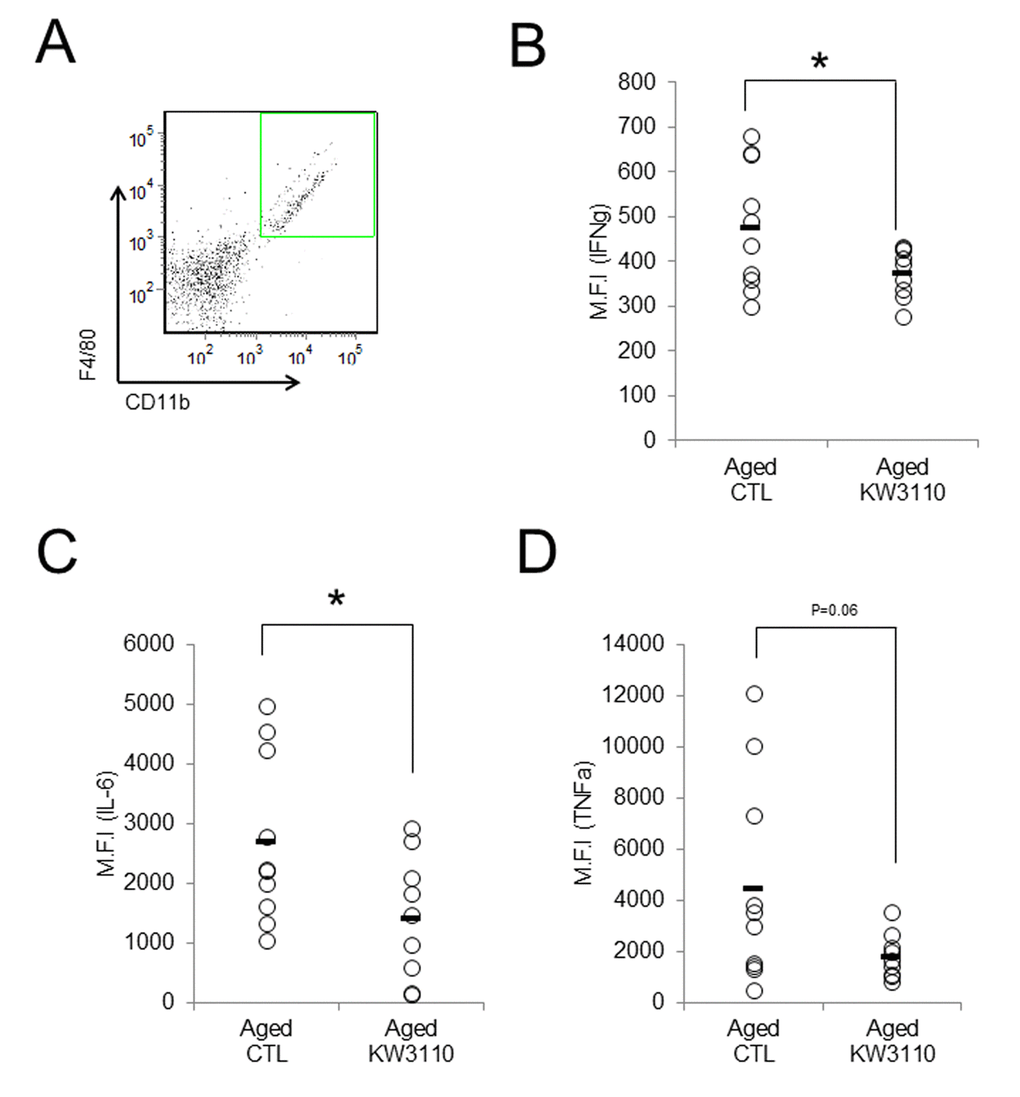
Figure 4. Intake of Lactobacillus paracasei KW3110 mitigated retinal inflammation. Intake of L. paracasei KW3110 in aged mice (17-months-old) suppressed the expression of inflammatory cytokines in retinal macrophage of aged mice. F4/80 and CD11b-positive macrophage in retina were gated as shown in (A), and median fluorescent intensity of intracellular IL-6, IFN-γ, and TNF-α were analyzed by flow cytometry (B-D). Significance was assumed if the p value was < 0.05; *p<0.05. All abbreviations are defined in the Figure 3 legend.
Discussion
Defective immune functions with aging are key triggers of age-related chronic inflammatory diseases, including infectious diseases, tumors, diabetes, and neurodegenerative diseases [8]. With rapid increases in the aging population, the prevention of age-related immunological dysfunctions and chronic inflammation are necessary to extend the healthy lifespan. In the present study, we demonstrated that long-term intake of heat-killed L. paracasei KW3110 in aged mice significantly enhanced the population of beneficial gut bacteria, of the Bifidobacterium family, and slowed the age-related immune dysfunctions, expansion of the inflammatory IFN-γ-producing CD4-positive T cells in SI-LP, and lowered the serum levels of proinflammatory cytokines. We also found that intake of L. paracasei KW3110 mitigated retinal inflammation and age-related retinal cell death, probably associated with the prevention of age-related retinal diseases.
Age-related alterations of gut microbiota composition have been reported to cause immune senescence and intestinal chronic inflammation [34,47]. Although some probiotics improve the intestinal environment and suppress inflammation, the effects of long-term ingestion of probiotics remain unclear. In the present study, we showed that intake of L. paracasei KW3110 improved the age-related changes of the gut microbiota composition (Fig. 1). The Firmicutes to Bacteroidetes bacterial ratio increased in aged mice fed a control diet as compared with aged mice fed a diet containing L. paracasei KW3110 for 6 months. The age-related increase of Firmicutes to Bacteroidetes ratio was consistent with a previous study [48]. An increased Firmicutes to Bacteroidetes ratio has been reported to be associated with intestinal inflammation in obese patients [49]. The long-term intake of L. paracasei KW3110 might mitigate intestinal inflammation and energy metabolic disorders by modulating the Firmicutes to Bacteroidetes ratio. The intake of L. paracasei KW3110 also increased the relative abundance of Bifidobacteriaceae families (Fig. 1D). Bifidobacterium is known as one of the most beneficial bacterial family, though the bacteria is not detected in the elderly [44,50,51]. In addition, the intake of Bifidobacterium has been reported to result in decreased levels of proinflammatory cytokines, such as TNF-α, in the elderly [44]. In contrast, intake of L. paracasei KW3110 decreased the relative abundance of Streptococcaceae (Fig. 1D). In a previous report, the Streptococcaceae bacteria stimulated the intestinal cells to induce CCL20 chemokine production [52] and inflammatory IFN-γ-producing CD4-positive T cells were attracted by the CCL20 chemokine [53]. In this study, the intake of L. paracasei KW3110 for 6 months in aged mice also significantly reduced Ccl20 gene expression in SI-LP as compared with that of age-matched control mice (Supplementary Fig. 3). These results suggested that the mitigation of age-related alterations in gut microbiota composition by the intake of L. paracasei KW3110 was important to suppress age-related intestinal chronic inflammation.
Indeed, the intake of L. paracasei KW3110 in aged mice significantly suppressed the age-related increase of inflammatory CD4-positive T cells, producing inflammatory cytokines (IFN-γ) at high levels in the SI-LP (Fig. 2). The expression of PD-1, one of the senescence markers, in CD4-positive T cells in SI-LP was also lower in aged mice fed a diet containing L. paracasei KW3110 than that of aged mice fed a control diet. These anti-inflammatory effects on intestinal immune cells might be due to mitigation of age-related decreases of Bifidobacterium. The modulatory effects on intestinal immune cell subpopulations might be associated with a direct interaction between L. paracasei KW3110 and intestinal immune cells. In a previous study, our group showed that L. paracasei KW3110 interacted with intestinal macrophages and suppressed excessive inflammation, including dermatitis, in mice and humans [40–42]. Because lactic acid bacteria, including L. paracasei KW3110, have some toll-like receptor (TLR) ligands, such as lipoteichoic acid, L. paracasei KW3110 might modulate intestinal immune cell activation by a TLR-dependent pathway. Further studies are required to determine the mechanism underlying the relationship between the intake of L. paracasei KW3110 and suppressive effects on age-related expansions of intestinal inflammatory immune cells.
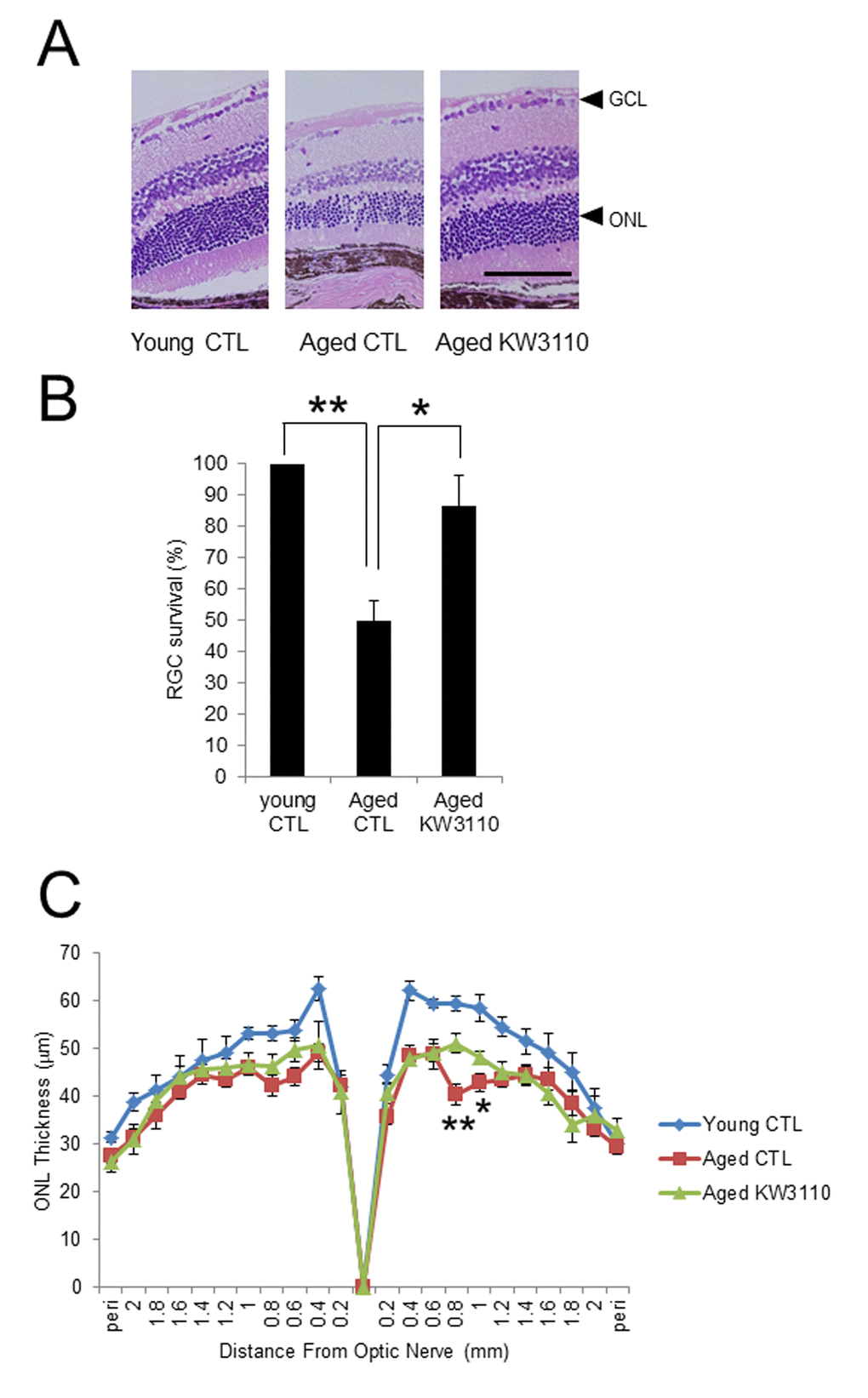
Proinflammatory cytokines produced by intestinal inflammatory immune cells are possibly transferred to other tissues, including the retina, through the blood. Age-related visual function declines and eye diseases, such as AMD, might be associated with retinal inflammation, because retinal chronic inflammation is toxic to retinal cells, including photoreceptor cells and RGCs [16–19]. In the present study, the intake of L. paracasei KW3110 mitigated the inflammation in the retinal macrophage in aged mice (Fig. 4). In addition, the anti-inflammatory effects of L. paracasei KW3110 resulted in inhibition of age-related retinal cell death (Fig. 5). In previous studies, RGCs have been reported to mediate behaviors associated with response to light information and genetic ablation of RGCs results in loss of light-evoked behaviors [54–57]. We have obtained preliminary data that intake of L. paracasei KW3110 in aged mice could preserve the light-evoked locomotor activities as compared with age-matched control mice (data not shown). Although further studies, including immunohistochemical analysis, are needed, L. paracasei KW3110 might suppress age-related retinal cell death. Immunological phenomena are mainly regulated by macrophage in the retina. Macrophage consists of at least two subgroups, classic inflammatory M1 macrophage or alternative anti-inflammatory M2 macrophage [58,59]. M1 macrophages produce inflammatory cytokines, such as TNFα and IL-6, whereas M2 macrophages are considered to be associated with anti-inflammatory responses, including tissue remodeling, through the production of neurotrophic factors and anti-inflammatory cytokines [60,61]. Recently, we found that the intake of L. paracasei KW3110 suppressed light-induced retinal inflammation (unpublished data). Although further studies are required to evaluate the effects of L. paracasei KW3110 on macrophage activation in the retina, the inhibitory effects of L. paracasei KW3110 on age-related retinal cell death might be accompanied at least in part, by the regulation of macrophage activities. In addition, blood-borne macrophages have been reported to enter the retina via the optic nerve and ciliary body in light exposure mice model [62]. Our flow cytometry analysis showed that CD11b and f4/80-positive retinal cells, the retinal macrophages, in aged mice increased more than in young mice (Supplementary Fig. 4). Taken together, proinflammatory macrophages might penetrate into the retina under the age-related retinal degenerative condition.
In the present study, the anti-inflammatory effects of L. paracasei KW3110 on immune cells were observed in aged mice fed each diet from 11–17 months of age for 6 months (Figs. 2 and 4). However, in aged mice fed a diet containing L. paracasei KW3110 from 16–22 months of age for 6 months, such anti-inflammatory effects on immune cells were mild and not significantly different as compared with that of age-match control mice (data not shown). These results suggested that 22 months of age was too old to evaluate the effects of L. paracasei KW3110 on intestinal immune cells.
Lactic acid bacteria are considered to be phagocytosed by intestinal M cells. In a previous report, M cells in aged mice (18 months of age) were not fully functional [63]. This may be because the effects of L. paracasei KW3110 on immune cells in aged mice of 22 months of age were milder than in aged mice of 17 months of age. In the present study, we showed that intake of L. paracasei KW3110 from 16–22 months of age for 6 months significantly suppressed the serum levels of proinflammatory cytokines, alteration of gut microbiota composition, and retinal cell loss (Figs. 1, 3, and 5). Because the anti-inflammatory effects on serum cytokine levels were observed in aged mice fed a diet containing L. paracasei KW3110 (Supplementary Fig. 2), it was suggested that these phenotypes were reflected by the accumulation of anti-inflammatory effects for several months. In other words, continuous preventive methods, like dietary supplementation, might be much more effective in the delay of chronic inflammation.
In conclusion, the intake of L. paracasei KW3110 mitigated chronic inflammation in the intestine and retina, and reduced age-related retinal cell death. Further studies are needed to evaluate the effects in age-related senescent changes of the retina.
Materials and Methods
Animals
The Mice (C57BL/6N, female) were purchased from Japan SLC (Hamamatsu, Japan). Young (1-month-old, n = 5), or aged (11-months-old, n = 10 in each group, or 16-months-old, n = 12 in each group) mice were acclimated until each aging study was started. Aged mice were divided by equal average weights into two groups. The control group mice were fed AIN93M (Oriental Yeast, Tokyo, Japan) and the Lactobacillus paracasei KW3110-fed mice (hereafter called the KW3110 group mice) were fed AIN93M containing 1 mg heat-killed L. paracasei KW3110/day/mouse for 6 months. The mice were housed in specific pathogen-free conditions under a 12-h light-dark photo cycle and had ad libitum access to water and the diet. The temperature in the room was kept at 25 ± 1°C and 60% ± 15% humidity.
All animal procedures and experiments were performed in accordance with the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research and institutional guidelines following approval by the Animal Care and Use Committee of National Center for Geriatrics and Gerontology (NCGG) (Obu, Japan).
SI-LP and retinal cell preparations and flow cytometry analyses
The SI-LP cells and retinal cells were prepared for flow cytometric analyses. The SI-LP cells were prepared as described previously [64]. Retinas were digested with 1 mg/mL collagenase II (Worthington, Lakewood, NJ, USA) for 40 minutes at 37°C in Hanks' Balanced Salt Solution (HBSS) buffer with 1.0% bovine serum albumin (BSA). The tissue digest was then filtered through a 70 m cell strainer, and washed with HBSS buffer with 1.0% BSA for 5 minutes at 1,300 rpm at 4°C. The supernatant was carefully removed and the digested tissue pellet was resuspended to form a single cell suspension. To investigate intracellular cytokine production, SI-LP and retinal cells were treated with a leukocyte activation cocktail with BD GolgiPlug (BD Biosciences, San Jose, CA, USA) for 4.5 hours and with a BD Cytofix/Cytoperm Fixation/Permeabilization kit (BD Biosciences), and then stained with the following antibodies to: CD3ε-BV421 (145-2C11) (Sony Biotechnology Inc., Tokyo, Japan), CD4-APC-Cy7 (GK1.5) (BioLegend, San Diego, CA, USA), IFN-γ-PE-Cy7 (XMG1.2) (eBiosciences, San Diego, CA, USA), TNF-α-BV421 (MP6-XT22) (BioLegend), IL-6-PE (MP5-20F3) (eBiosciences), IFN-γ-APC (XMG1.2) (eBiosciences), CD11b-APC-Cy7 (M1/70) (BD Biosciences), F4/80-PE-Cy7 (BM8) (BioLegend), programmed cell death protein 1 (PD-1)-BV421 (29F.1A12) (BioLegend), and 7-AAD (BD Pharmingen, San Jose, CA, USA). The 7-AAD− CD3ε- and CD4-positive cells were defined as CD4-positive T cells and 7-AAD−CD11b-, and the f4/80-positive cells were defined as retinal macrophage cells. To detect Tregs, cells were treated with a Foxp3 Staining Kit (BD Biosciences) and stained with the following antibodies to: CD3ε-FITC (145-2C11) (eBiosciences), Foxp3-PE-Cy7 (FJK-16s) (eBiosciences), CD4-APC-Cy7 (GK1.5) (BioLegend), and 7-AAD (BD Pharmingen). The 7-AAD− CD3ε-, CD4-, and Foxp3-positive cells were defined as Tregs.
Data were collected using a FACS Canto II flow cytometer (BD Biosciences) and analyzed by FCS Express software (De Novo Software, Los Angeles, CA, USA).
Analysis of serum cytokine concentrations
Blood samples were collected into heparin-coated tubes and centrifuged at 3,000 rpm for 5 min. Supernatants were collected and analyzed for serum cytokine concentrations using a Bio-Plex Pro mouse cytokine assay kit (Bio-Rad, Hercules, CA, USA).
Analyses of gut microbiota
Feces were collected from young mice and aged mice fed control or KW3110-containing diets and stored at -80°C until further analyses. DNA was extracted according to a previous report [65]. Pyrosequencing of 16S ribosomal RNA was performed by Technosruga Lab (Shizuoka, Japan). The 16S ribosomal RNA sequencing was performed using the MiSeqTMII system (Illumina, San Diego, CA, USA) according to a previously described method [65]. The V3-V4 regions of 16S ribosomal RNA genes were amplified by PCR from fecal genomic DNA using the following universal primers [66,67]: 341F, 5′-AATGATACGGCGACCACCGAGATCTACACTCTTTCCCTACACGACGCTCTTCCGATCTCCTACGGGAGGCAGCAGCCTACGGGAGGCAGCAG-3′, and 806R, 5′-CAAGCAGAAGACGGCATAGAGATNNNNNNGTGACTGGAGTTCAGACGTGTGCTCTTCCGATCTGGACTACHVGGGTWTCTAAT-3′. PCR products were purified using a MultiScreen PCR filter plate (Merck Millipore, Darmstadt, Germany). All amplicons were sequenced using the paired-end, 2 × 250 bp cycle run on the MiSeqTMII system with a MiSeq Reagent Kit. The sequences were evaluated and filtered for their length and quality. Paired-end sequencing was joined using the fastq-join program (http://code.google.com/p/ea-utils/). The low quality reads, quality value scores < 20 for more than 99% of the sequence, were removed. The nucleotide sequence dataset was deposited in DNA Data Bank of Japan (DDBJ) with the accession numbers SAMD00118680-SAMD00118706.
Analyses of the extracted sequence reads were carried out using the Ribosomal Database Project (RDP) Multiclassifier version 2.11 [68] and Basic Local Alignment Search Tool (BLAST) search using the Metagenome@KIN analysis software (World Fusion, Tokyo, Japan). Reads showing > 97% similarity were grouped in each taxonomic rank.
Measurements of outer nuclear layer thickness in the retina and retinal ganglion cell survival
Retinal sections, which included the optic nerve head to the most peripheral region of the retina, were fixed in neutral 10% formalin and stained with hematoxylin and eosin (HE), and the outer nuclear layer (ONL) thickness was measured in all areas and averaged at each point from the optic nerve head. The number of RGCs was counted within whole retinal sections.
Statistical analysis
All values are expressed as the mean ± SEM. Statistical differences between three groups (young mice group fed a control diet, aged mice group fed a control diet, and aged mice group fed a diet containing KW3110) were analyzed by one-way analysis of variance (ANOVA), followed by the Tukey-Kramer test with significance set at p < 0.05. Statistical differences between two groups (aged mice group fed a control diet versus aged mice group fed a diet containing KW3110) were determined using an unpaired, two-tailed Student's t-test with significance set at p < 0.05. All statistical analyses were performed by using the Ekuseru-Toukei 2012 software program (Social Survey Research Information, Tokyo, Japan).
Supplementary Materials
Acknowledgements
We thank T. Kirisako and M. Sugamata of Kirin Company, I. Wakaya, of Koiwai Dairy Products Company for valuable discussion. We are also indebted to T. Matsuda and M. Sugimoto for stimulating and helpful suggestions and advice as well as grateful to our colleagues, Y. Ohta, M. Kondo, A. Yamada, N. Matsui, M. Nagasaka and M. Okayama for excellent technical assistance.
Conflicts of Interest
The authors declare that they have no conflicts of interest with the content of this article.
Funding
This work was supported in part by the Research Funding for Longevity Sciences from National Center for Geriatrics and Gerontology (grant #29-26 to M.M.).
References
- 1. Flatt T. A new definition of aging? Front Genet. 2012; 3:148. https://doi.org/10.3389/fgene.2012.00148 [PubMed]
- 2. Rose MR, Flatt T, Graves JL, Greer LF, Martinez DE, Matos M, Mueller LD, Shmookler Reis RJ, Shahrestani P. What is Aging? Front Genet. 2012; 3:134. https://doi.org/10.3389/fgene.2012.00134 [PubMed]
- 3. Kennedy BK, Berger SL, Brunet A, Campisi J, Cuervo AM, Epel ES, Franceschi C, Lithgow GJ, Morimoto RI, Pessin JE, Rando TA, Richardson A, Schadt EE, et al. Geroscience: linking aging to chronic disease. Cell. 2014; 159:709–13. https://doi.org/10.1016/j.cell.2014.10.039 [PubMed]
- 4. Finkel T, Serrano M, Blasco MA. The common biology of cancer and ageing. Nature. 2007; 448:767–74. https://doi.org/10.1038/nature05985 [PubMed]
- 5. Gavazzi G, Krause KH. Ageing and infection. Lancet Infect Dis. 2002; 2:659–66. https://doi.org/10.1016/S1473-3099(02)00437-1 [PubMed]
- 6. Calçada D, Vianello D, Giampieri E, Sala C, Castellani G, de Graaf A, Kremer B, van Ommen B, Feskens E, Santoro A, Franceschi C, Bouwman J. The role of low-grade inflammation and metabolic flexibility in aging and nutritional modulation thereof: a systems biology approach. Mech Ageing Dev. 2014; 136-137:138–47. https://doi.org/10.1016/j.mad.2014.01.004 [PubMed]
- 7. Fulop T, Larbi A, Witkowski JM, Kotb R, Hirokawa K, Pawelec G. Immunosenescence and cancer. Crit Rev Oncog. 2013; 18:489–513. https://doi.org/10.1615/CritRevOncog.2013010597 [PubMed]
- 8. Deleidi M, Jäggle M, Rubino G. Immune aging, dysmetabolism, and inflammation in neurological diseases. Front Neurosci. 2015; 9:172. https://doi.org/10.3389/fnins.2015.00172 [PubMed]
- 9. Klein R, Cruickshanks KJ, Nash SD, Krantz EM, Nieto FJ, Huang GH, Pankow JS, Klein BE. The prevalence of age-related macular degeneration and associated risk factors. Arch Ophthalmol. 2010; 128:750–58. https://doi.org/10.1001/archophthalmol.2010.92 [PubMed]
- 10. Wong WL, Su X, Li X, Cheung CM, Klein R, Cheng CY, Wong TY. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob Health. 2014; 2:e106–16. https://doi.org/10.1016/S2214-109X(13)70145-1 [PubMed]
- 11. Pezzullo L, Streatfeild J, Simkiss P, Shickle D. The economic impact of sight loss and blindness in the UK adult population. BMC Health Serv Res. 2018; 18:63. https://doi.org/10.1186/s12913-018-2836-0 [PubMed]
- 12. Flaxman SR, Bourne RR, Resnikoff S, Ackland P, Braithwaite T, Cicinelli MV, Das A, Jonas JB, Keeffe J, Kempen JH, Leasher J, Limburg H, Naidoo K, et al, and Vision Loss Expert Group of the Global Burden of Disease Study. Global causes of blindness and distance vision impairment 1990-2020: a systematic review and meta-analysis. Lancet Glob Health. 2017; 5:e1221–34. https://doi.org/10.1016/S2214-109X(17)30393-5 [PubMed]
- 13. Weinreb RN, Aung T, Medeiros FA. The pathophysiology and treatment of glaucoma: a review. JAMA. 2014; 311:1901–11. https://doi.org/10.1001/jama.2014.3192 [PubMed]
- 14. Beatty S, Koh H, Phil M, Henson D, Boulton M. The role of oxidative stress in the pathogenesis of age-related macular degeneration. Surv Ophthalmol. 2000; 45:115–34. https://doi.org/10.1016/S0039-6257(00)00140-5 [PubMed]
- 15. Shahinfar S, Edward DP, Tso MO. A pathologic study of photoreceptor cell death in retinal photic injury. Curr Eye Res. 1991; 10:47–59. https://doi.org/10.3109/02713689109007610 [PubMed]
- 16. Kauppinen A, Paterno JJ, Blasiak J, Salminen A, Kaarniranta K. Inflammation and its role in age-related macular degeneration. Cell Mol Life Sci. 2016; 73:1765–86. https://doi.org/10.1007/s00018-016-2147-8 [PubMed]
- 17. Ambati J, Atkinson JP, Gelfand BD. Immunology of age-related macular degeneration. Nat Rev Immunol. 2013; 13:438–51. https://doi.org/10.1038/nri3459 [PubMed]
- 18. Parmeggiani F, Romano MR, Costagliola C, Semeraro F, Incorvaia C, D’Angelo S, Perri P, De Palma P, De Nadai K, Sebastiani A. Mechanism of inflammation in age-related macular degeneration. Mediators Inflamm. 2012; 2012:546786. https://doi.org/10.1155/2012/546786 [PubMed]
- 19. Scholl HP, Charbel Issa P, Walier M, Janzer S, Pollok-Kopp B, Börncke F, Fritsche LG, Chong NV, Fimmers R, Wienker T, Holz FG, Weber BH, Oppermann M. Systemic complement activation in age-related macular degeneration. PLoS One. 2008; 3:e2593. https://doi.org/10.1371/journal.pone.0002593 [PubMed]
- 20. Chen Y, Palczewska G, Mustafi D, Golczak M, Dong Z, Sawada O, Maeda T, Maeda A, Palczewski K. Systems pharmacology identifies drug targets for Stargardt disease-associated retinal degeneration. J Clin Invest. 2013; 123:5119–34. https://doi.org/10.1172/JCI69076 [PubMed]
- 21. Chen Y, Okano K, Maeda T, Chauhan V, Golczak M, Maeda A, Palczewski K. Mechanism of all-trans-retinal toxicity with implications for stargardt disease and age-related macular degeneration. J Biol Chem. 2012; 287:5059–69. https://doi.org/10.1074/jbc.M111.315432 [PubMed]
- 22. Goronzy JJ, Fang F, Cavanagh MM, Qi Q, Weyand CM. Naive T cell maintenance and function in human aging. J Immunol. 2015; 194:4073–80. https://doi.org/10.4049/jimmunol.1500046 [PubMed]
- 23. Maue AC, Yager EJ, Swain SL, Woodland DL, Blackman MA, Haynes L. T-cell immunosenescence: lessons learned from mouse models of aging. Trends Immunol. 2009; 30:301–05. https://doi.org/10.1016/j.it.2009.04.007 [PubMed]
- 24. Spadaro O, Goldberg EL, Camell CD, Youm YH, Kopchick JJ, Nguyen KY, Bartke A, Sun LY, Dixit VD. Growth Hormone Receptor Deficiency Protects against Age-Related NLRP3 Inflammasome Activation and Immune Senescence. Cell Reports. 2016; 14:1571–80. https://doi.org/10.1016/j.celrep.2016.01.044 [PubMed]
- 25. Frasca D, Blomberg BB. Effects of aging on B cell function. Curr Opin Immunol. 2009; 21:425–30. https://doi.org/10.1016/j.coi.2009.06.001 [PubMed]
- 26. Miller JP, Allman D. The decline in B lymphopoiesis in aged mice reflects loss of very early B-lineage precursors. J Immunol. 2003; 171:2326–30. https://doi.org/10.4049/jimmunol.171.5.2326 [PubMed]
- 27. Frasca D, Riley RL, Blomberg BB. Effect of age on the immunoglobulin class switch. Crit Rev Immunol. 2004; 24:297–320. https://doi.org/10.1615/CritRevImmunol.v24.i5.10 [PubMed]
- 28. Kovacs EJ, Palmer JL, Fortin CF, Fülöp T
Jr , Goldstein DR, Linton PJ. Aging and innate immunity in the mouse: impact of intrinsic and extrinsic factors. Trends Immunol. 2009; 30:319–24. https://doi.org/10.1016/j.it.2009.03.012 [PubMed] - 29. Shaw AC, Joshi S, Greenwood H, Panda A, Lord JM. Aging of the innate immune system. Curr Opin Immunol. 2010; 22:507–13. https://doi.org/10.1016/j.coi.2010.05.003 [PubMed]
- 30. Sakamoto A, Matsuda T, Kawaguchi K, Takaoka A, Maruyama M. Involvement of Zizimin2/3 in the age-related defect of peritoneal B-1a cells as a source of anti-bacterial IgM. Int Immunol. 2017; 29:431–38. https://doi.org/10.1093/intimm/dxx054 [PubMed]
- 31. Capri M, Monti D, Salvioli S, Lescai F, Pierini M, Altilia S, Sevini F, Valensin S, Ostan R, Bucci L, Franceschi C. Complexity of anti-immunosenescence strategies in humans. Artif Organs. 2006; 30:730–42. https://doi.org/10.1111/j.1525-1594.2006.00295.x [PubMed]
- 32. Adolfsson O, Huber BT, Meydani SN. Vitamin E-enhanced IL-2 production in old mice: naive but not memory T cells show increased cell division cycling and IL-2-producing capacity. J Immunol. 2001; 167:3809–17. https://doi.org/10.4049/jimmunol.167.7.3809 [PubMed]
- 33. Candore G, Balistreri CR, Colonna-Romano G, Grimaldi MP, Lio D, Listi’ F, Scola L, Vasto S, Caruso C. Immunosenescence and anti-immunosenescence therapies: the case of probiotics. Rejuvenation Res. 2008; 11:425–32. https://doi.org/10.1089/rej.2008.0662 [PubMed]
- 34. Cho SY, Kim J, Lee JH, Sim JH, Cho DH, Bae IH, Lee H, Seol MA, Shin HM, Kim TJ, Kim DY, Lee SH, Shin SS, et al. Modulation of gut microbiota and delayed immunosenescence as a result of syringaresinol consumption in middle-aged mice. Sci Rep. 2016; 6:39026. https://doi.org/10.1038/srep39026 [PubMed]
- 35. Biagi E, Candela M, Turroni S, Garagnani P, Franceschi C, Brigidi P. Ageing and gut microbes: perspectives for health maintenance and longevity. Pharmacol Res. 2013; 69:11–20. https://doi.org/10.1016/j.phrs.2012.10.005 [PubMed]
- 36. Bartley JM, Zhou X, Kuchel GA, Weinstock GM, Haynes L. Impact of Age, Caloric Restriction, and Influenza Infection on Mouse Gut Microbiome: An Exploratory Study of the Role of Age-Related Microbiome Changes on Influenza Responses. Front Immunol. 2017; 8:1164. https://doi.org/10.3389/fimmu.2017.01164 [PubMed]
- 37. Shen X, Miao J, Wan Q, Wang S, Li M, Pu F, Wang G, Qian W, Yu Q, Marotta F, He F. Possible correlation between gut microbiota and immunity among healthy middle-aged and elderly people in southwest China. Gut Pathog. 2018; 10:4. https://doi.org/10.1186/s13099-018-0231-3 [PubMed]
- 38. Rowan S, Jiang S, Korem T, Szymanski J, Chang ML, Szelog J, Cassalman C, Dasuri K, McGuire C, Nagai R, Du XL, Brownlee M, Rabbani N, et al. Involvement of a gut-retina axis in protection against dietary glycemia-induced age-related macular degeneration. Proc Natl Acad Sci USA. 2017; 114:E4472–81. https://doi.org/10.1073/pnas.1702302114 [PubMed]
- 39. Fontana L, Bermudez-Brito M, Plaza-Diaz J, Muñoz-Quezada S, Gil A. Sources, isolation, characterisation and evaluation of probiotics. Br J Nutr. 2013 (Suppl 2); 109:S35–50. https://doi.org/10.1017/S0007114512004011 [PubMed]
- 40. Ichikawa S, Fujii R, Fujiwara D, Komiyama Y, Kaisho T, Sakaguchi M, Konishi Y. MyD88 but not TLR2, 4 or 9 is essential for IL-12 induction by lactic acid bacteria. Biosci Biotechnol Biochem. 2007; 71:3026–32. https://doi.org/10.1271/bbb.70414 [PubMed]
- 41. Fujiwara D, Inoue S, Wakabayashi H, Fujii T. The anti-allergic effects of lactic acid bacteria are strain dependent and mediated by effects on both Th1/Th2 cytokine expression and balance. Int Arch Allergy Immunol. 2004; 135:205–15. https://doi.org/10.1159/000081305 [PubMed]
- 42. Wakabayashi H, Nariai C, Takemura F, Nakao W, Fujiwara D. Dietary supplementation with lactic acid bacteria attenuates the development of atopic-dermatitis-like skin lesions in NC/Nga mice in a strain-dependent manner. Int Arch Allergy Immunol. 2008; 145:141–51. https://doi.org/10.1159/000108139 [PubMed]
- 43. Ichikawa S, Miyake M, Fujii R, Konishi Y. Orally administered Lactobacillus paracasei KW3110 induces in vivo IL-12 production. Biosci Biotechnol Biochem. 2009; 73:1561–65. https://doi.org/10.1271/bbb.90058 [PubMed]
- 44. Ouwehand AC, Bergsma N, Parhiala R, Lahtinen S, Gueimonde M, Finne-Soveri H, Strandberg T, Pitkälä K, Salminen S. Bifidobacterium microbiota and parameters of immune function in elderly subjects. FEMS Immunol Med Microbiol. 2008; 53:18–25. https://doi.org/10.1111/j.1574-695X.2008.00392.x [PubMed]
- 45. Ibrahim F, Ruvio S, Granlund L, Salminen S, Viitanen M, Ouwehand AC. Probiotics and immunosenescence: cheese as a carrier. FEMS Immunol Med Microbiol. 2010; 59:53–59. https://doi.org/10.1111/j.1574-695X.2010.00658.x [PubMed]
- 46. Martinet KZ, Bloquet S, Bourgeois C. Ageing combines CD4 T cell lymphopenia in secondary lymphoid organs and T cell accumulation in gut associated lymphoid tissue. Immun Ageing. 2014; 11:8. https://doi.org/10.1186/1742-4933-11-8 [PubMed]
- 47. Fransen F, van Beek AA, Borghuis T, Aidy SE, Hugenholtz F, van der Gaast-de Jongh C, Savelkoul HF, De Jonge MI, Boekschoten MV, Smidt H, Faas MM, de Vos P. Aged Gut Microbiota Contributes to Systemical Inflammaging after Transfer to Germ-Free Mice. Front Immunol. 2017; 8:1385. https://doi.org/10.3389/fimmu.2017.01385 [PubMed]
- 48. Hoffman JD, Parikh I, Green SJ, Chlipala G, Mohney RP, Keaton M, Bauer B, Hartz AM, Lin AL. Age Drives Distortion of Brain Metabolic, Vascular and Cognitive Functions, and the Gut Microbiome. Front Aging Neurosci. 2017; 9:298. https://doi.org/10.3389/fnagi.2017.00298 [PubMed]
- 49. Verdam FJ, Fuentes S, de Jonge C, Zoetendal EG, Erbil R, Greve JW, Buurman WA, de Vos WM, Rensen SS. Human intestinal microbiota composition is associated with local and systemic inflammation in obesity. Obesity (Silver Spring). 2013; 21:E607–15. https://doi.org/10.1002/oby.20466 [PubMed]
- 50. Biagi E, Nylund L, Candela M, Ostan R, Bucci L, Pini E, Nikkïla J, Monti D, Satokari R, Franceschi C, Brigidi P, De Vos W. Through ageing, and beyond: gut microbiota and inflammatory status in seniors and centenarians. PLoS One. 2010; 5:e10667. https://doi.org/10.1371/journal.pone.0010667 [PubMed]
- 51. Drago L, Toscano M, Rodighiero V, De Vecchi E, Mogna G. Cultivable and pyrosequenced fecal microflora in centenarians and young subjects. J Clin Gastroenterol. 2012 (Suppl ); 46:S81–84. https://doi.org/10.1097/MCG.0b013e3182693982 [PubMed]
- 52. Veckman V, Miettinen M, Matikainen S, Lande R, Giacomini E, Coccia EM, Julkunen I. Lactobacilli and streptococci induce inflammatory chemokine production in human macrophages that stimulates Th1 cell chemotaxis. J Leukoc Biol. 2003; 74:395–402. https://doi.org/10.1189/jlb.0402212 [PubMed]
- 53. Mony JT, Khorooshi R, Owens T. Chemokine receptor expression by inflammatory T cells in EAE. Front Cell Neurosci. 2014; 8:187. https://doi.org/10.3389/fncel.2014.00187 [PubMed]
- 54. Hattar S, Liao HW, Takao M, Berson DM, Yau KW. Melanopsin-containing retinal ganglion cells: architecture, projections, and intrinsic photosensitivity. Science. 2002; 295:1065–70. https://doi.org/10.1126/science.1069609 [PubMed]
- 55. Berson DM, Dunn FA, Takao M. Phototransduction by retinal ganglion cells that set the circadian clock. Science. 2002; 295:1070–73. https://doi.org/10.1126/science.1067262 [PubMed]
- 56. Güler AD, Ecker JL, Lall GS, Haq S, Altimus CM, Liao HW, Barnard AR, Cahill H, Badea TC, Zhao H, Hankins MW, Berson DM, Lucas RJ, et al. Melanopsin cells are the principal conduits for rod-cone input to non-image-forming vision. Nature. 2008; 453:102–05. https://doi.org/10.1038/nature06829 [PubMed]
- 57. Mure LS, Hatori M, Zhu Q, Demas J, Kim IM, Nayak SK, Panda S. Melanopsin-Encoded Response Properties of Intrinsically Photosensitive Retinal Ganglion Cells. Neuron. 2016; 90:1016–27. https://doi.org/10.1016/j.neuron.2016.04.016 [PubMed]
- 58. Martinez FO, Helming L, Gordon S. Alternative activation of macrophages: an immunologic functional perspective. Annu Rev Immunol. 2009; 27:451–83. https://doi.org/10.1146/annurev.immunol.021908.132532 [PubMed]
- 59. Biswas SK, Mantovani A. Macrophage plasticity and interaction with lymphocyte subsets: cancer as a paradigm. Nat Immunol. 2010; 11:889–96. https://doi.org/10.1038/ni.1937 [PubMed]
- 60. Sica A, Mantovani A. Macrophage plasticity and polarization: in vivo veritas. J Clin Invest. 2012; 122:787–95. https://doi.org/10.1172/JCI59643 [PubMed]
- 61. Satoh T, Kidoya H, Naito H, Yamamoto M, Takemura N, Nakagawa K, Yoshioka Y, Morii E, Takakura N, Takeuchi O, Akira S. Critical role of Trib1 in differentiation of tissue-resident M2-like macrophages. Nature. 2013; 495:524–28. https://doi.org/10.1038/nature11930 [PubMed]
- 62. Joly S, Francke M, Ulbricht E, Beck S, Seeliger M, Hirrlinger P, Hirrlinger J, Lang KS, Zinkernagel M, Odermatt B, Samardzija M, Reichenbach A, Grimm C, Remé CE. Cooperative phagocytes: resident microglia and bone marrow immigrants remove dead photoreceptors in retinal lesions. Am J Pathol. 2009; 174:2310–23. https://doi.org/10.2353/ajpath.2009.090023 [PubMed]
- 63. Kobayashi A, Donaldson DS, Erridge C, Kanaya T, Williams IR, Ohno H, Mahajan A, Mabbott NA. The functional maturation of M cells is dramatically reduced in the Peyer’s patches of aged mice. Mucosal Immunol. 2013; 6:1027–37. https://doi.org/10.1038/mi.2012.141 [PubMed]
- 64. Sanos SL, Diefenbach A. Isolation of NK cells and NK-like cells from the intestinal lamina propria. Methods Mol Biol. 2010; 612:505–17. https://doi.org/10.1007/978-1-60761-362-6_32 [PubMed]
- 65. Takahashi S, Tomita J, Nishioka K, Hisada T, Nishijima M. Development of a prokaryotic universal primer for simultaneous analysis of Bacteria and Archaea using next-generation sequencing. PLoS One. 2014; 9:e105592. https://doi.org/10.1371/journal.pone.0105592 [PubMed]
- 66. Muyzer G, de Waal EC, Uitterlinden AG. Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol. 1993; 59:695–700. [PubMed]
- 67. Caporaso JG, Lauber CL, Walters WA, Berg-Lyons D, Lozupone CA, Turnbaugh PJ, Fierer N, Knight R. Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc Natl Acad Sci USA. 2011 (Suppl 1); 108:4516–22. https://doi.org/10.1073/pnas.1000080107 [PubMed]
- 68. Wang Q, Garrity GM, Tiedje JM, Cole JR. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol. 2007; 73:5261–67. https://doi.org/10.1128/AEM.00062-07 [PubMed]