Introduction
The aging population is increasing in a lot of countries, and age-related heart disease has become a major cause of cardiovascular mortality and morbidity in modern society [1, 2]. Cardiac aging is majorly characterized by increased diastolic dysfunction, accumulation of lipids, and oxidative stress [3–6]. Since the incidence of heart disease increases dramatically with age, it is important to understand the molecular mechanisms through which the heart becomes either more or less susceptible to stress. Sirtuin (SIRT1) and silent information regulator 2 (Sir2) proteins are classes of proteins that possess nicotinamide adenine dinucleotide (NAD+)-dependent deacetylase activity and ADP-ribosyltransferase activity, respectively, and they are evolutionarily conserved from bacteria to humans [7]. Sirt1 proteins participate in regulating cell aging, diabetes, obesity, and oxidative stress [8]. In mice hearts, SIRT1 overexpression relieves AngII-induced cardiomyocyte hypertrophy and apoptosis [9]. Up-regulation and activation of Sirt1 induced by phenylephrine are blocked by the inhibition or downregulation of AMPK, leading to improved cell survival under hypertrophic stress [10]. Besides, some studies examining the effects of resveratrol indicate that Sirt1 may have a beneficial role in failing hearts and that endogenous Sirt1 up-regulation is a protective mechanism in the early stage of heart failure [11, 12]. Sirt1 has also been reported to up-regulate Mn-SOD via hypoxia-inducible factor-2α and FoxO4, and several lines of evidence suggest that Sir2 plays a protective role against oxidative stress in cardiomyocytes and the heart. Cardiac-specific overexpression of Sirt1 induces an increase in the protein expression of catalase after exposure to paraquat. During cardiac I/R, overexpression of Sirt1 also upregulates Mn-SOD and Trx1, and attenuates oxidative stress [13–15]. Interestingly, lipid accumulation can evoke cardiac oxidative stress, inflammation and, eventually, cardiac dysfunction and heart failure [16]. Adipose FOXO expression protects against HFD-induced cardiac malfunction, and the expression of FOXO in myocardial cells autonomously protects the heart from the adverse effects of a high-fat-diet in Drosophila [17, 18]. Sirt1 has been found to deacetylate Foxo1 at K242, K245, and K262 and induce nuclear translocation, which may resist lipid accumulation in heart [19–21]. Therefore, the evidence suggests that Sirt1 may be a key gene involved in regulating heart aging. However, it remains unknown whether the cardiac Sir2 gene in flies can affect age-related heart diastolic dysfunction, lipid accumulation, and oxidative stress.
Exercise, as an inducible form of physiologic stress, represents a powerful tool in cardiac aging research. Exercise physiology has provided a wealth of knowledge into how age-related changes in cardiac structure and function translate to decreased exercise capacity, which is a strong determinant of heart failure prognosis [22]. Age-related lipid accumulation and myocardial fiber loss are important causes of pathologic myocardial hypertrophy, which can also induce lipid toxic injury of cardiomyocytes [3, 23]. Exercise training can speed up fat mobilization and promote lipid decomposition, which can effectively prevent the heart from excessive accumulation of lipids [24]. Besides, cardiac aging is characterized by concentric remodeling and decrements in diastolic functions, which are theorized to contribute to the increased risk of heart failure in older adults [6]. Since physical activity has been related to higher cardiac internal dimensions and improved diastolic function, age-associated cardiac remodeling might be an appropriate target for exercise therapy [25, 26]. Moreover, oxidative stress, defined as an excess production of reactive oxygen species (ROS) relative to antioxidant defense, has been shown to play an important role in the pathophysiology of cardiac remodeling and heart failure [27]. Meanwhile, treatment of diabetic rats with exercise significantly decreased the levels of MDA and increased the activity of SOD, GPx, and CAT compared with untreated diabetic rats; this indicated the protective effect of voluntary exercise against oxidative stress in the hearts of high-fat diet-induced type 2 diabetic rats [28, 29]. Finally, the expression of the pro-survival p-Akt protein decreased significantly with age and reduced cardiac performance. The IGF1R/PI3K/Akt survival pathway in the heart of young rats can indeed be increased through exercise training, and exercise training enhanced the SIRT longevity pathway instead of IGF1 survival signaling to improve cardiomyocyte survival [30]. In Drosophila, increasing evidence shows that endurance exercise or regular exercise also improves heart function and delay heart age-related phenotypes [31–36]. Therefore, the evidence indicates that exercise training can delay cardiac aging, but it remains unclear whether exercise training delaying heart aging is related to cardiac Sirt1-related pathways, and the effect of cardiac Sir2 gene differential expression combined with exercise training on heart aging is still unknown.
To explore the relationship between cardiac Sir2, exercise, and cardiac aging, cardiac Sir2 knockdown or overexpression was induced using the UAS/hand-Gal4 system in Drosophila. The flies underwent exercise training using a TreadWheel. The cardiac Sir2 expression level was tested by qRT-PCR. The heart period, systolic period, diastolic period, diastolic dysfunction index, fractional shortening, diastolic diameter, and systolic diameter were measured by an M-mode trace. Next, the cardiac triacylglycerol (TAG) and brummer (bmm) gene expression were measured by ELISA and qRT-PCR. The cardiac SOD activity level, MDA level, and Foxo gene expression level were detected by ELISA and qRT-PCR. Based on these indicators, we tried to understand the relationship between cardiac Sir2, exercise, and cardiac aging.
Results
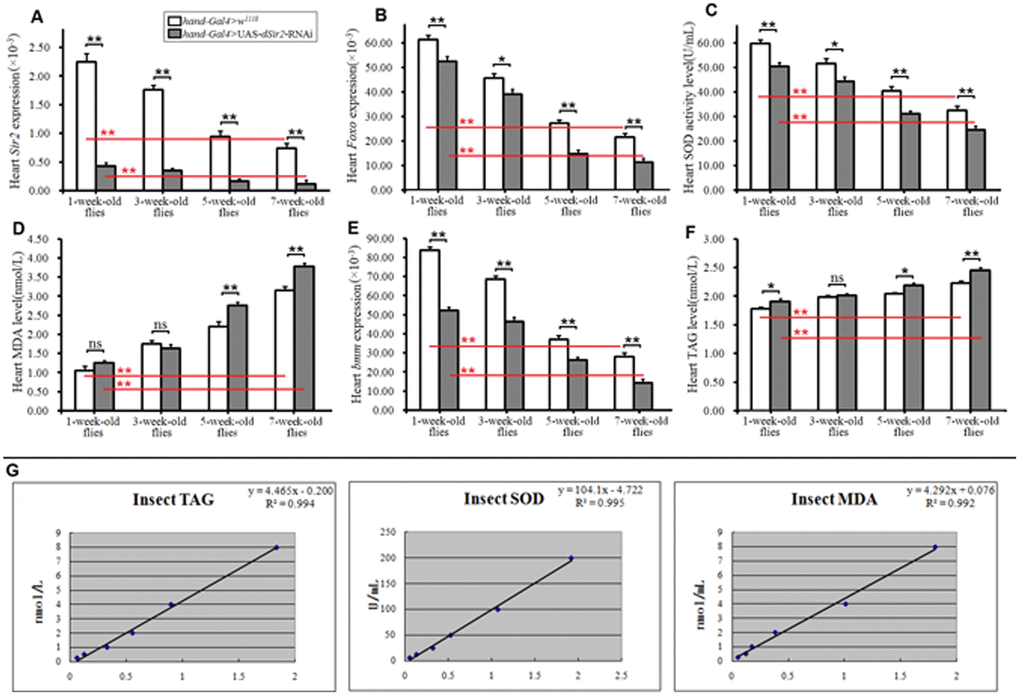
Cardiac dSir2 knockdown promotes heart aging in Drosophila
Cardiac dSir2 knockdown induced phenotypes similar to heart aging in young Drosophila
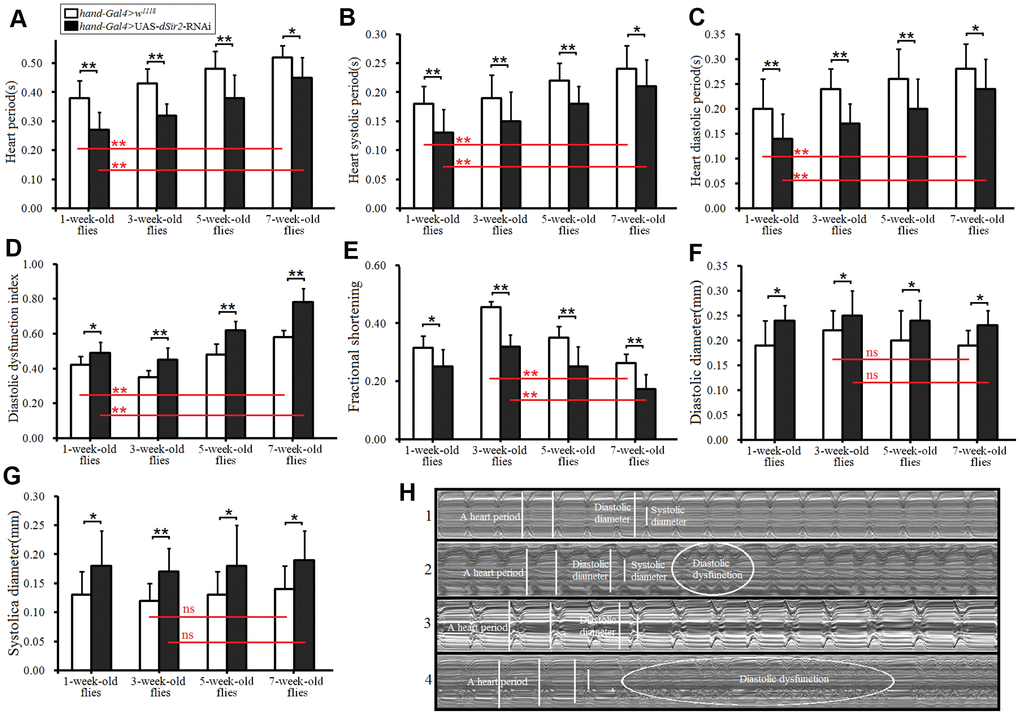
To further confirm whether cardiac dSir2 RNAi could induce age-related changes in the heart, the other cardiac dSir2 RNAi line was built by P{KK109370}VIE-260B and hand-Gla4 in 1-week-old flies. The results showed that the cardiac dSir2 expression of hand-Gal4>UAS-dSir2RNAi flies was significantly lower than that of hand-Gal4>w1118 flies (P<0.01) (a difference of about 5.6-fold; Figure 3A). This suggested that cardiac dSir2 knockdown was also successfully built by the UAS/hand-Gal4 system. Moreover, the results showed that cardiac dSir2 knockdown significantly reduced the SOD activity level and Foxo expression (P<0.01; Figure 3B and 3C), and notably increased MDA levels in young hearts (P<0.01; Figure 3D). In addition, cardiac dSir2 knockdown significantly increased cardiac TAG level (+19.1%) and reduced cardiac bmm expression (P<0.01; Figure 3E and 3F). Finally, cardiac dSir2 knockdown significantly reduced heart period, diastolic period, systolic period, and fractional shortening (P<0.05 or P<0.01; Figure 3G–3I, and 3K), and notably increased the diastolic dysfunction index and systolic diameter (P<0.01; Figure 3J, 3M and 3N). These results suggested that cardiac dSir2 knockdown could increase the incidence of oxidative damage, fat accumulation, and diastolic dysfunction in young hearts, which is similar to the phenotypes associated with aging hearts.
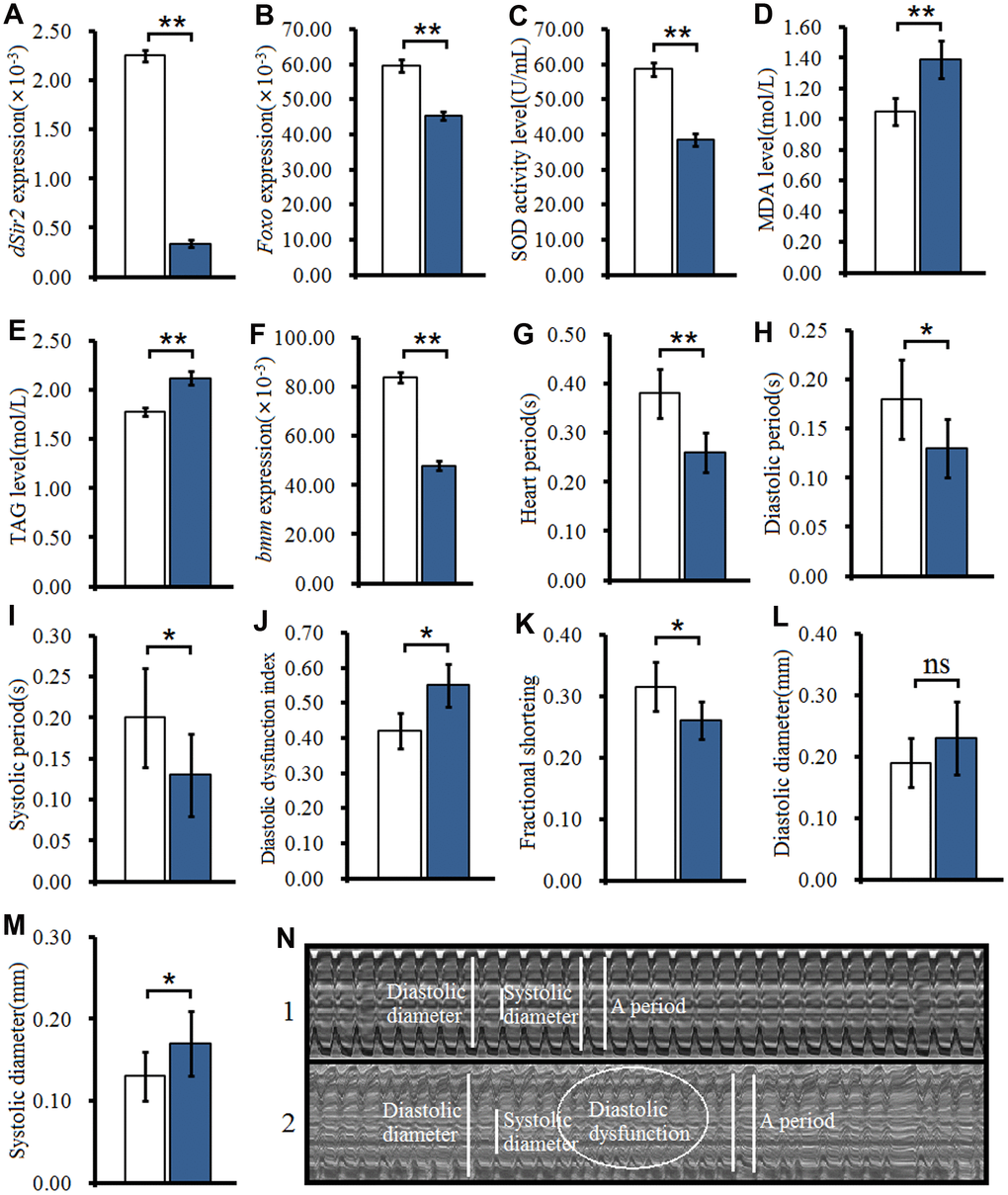
Figure 3. Effect of cardiac dSir2 knockdown on young hearts. (A) Cardiac dSir2 expression level. (B) Cardiac Foxo expression. (C) Cardiac SOD activity level. (D) Cardiac MDA level. (E) Cardiac TAG level. (F) Cardiac bmm expression. (G) Heart period. (H) Heart systolic period. (I) Heart diastolic period. (J) Heart diastolic dysfunction index. The diastolic dysfunction index is diastolic interval standard deviation/diastolic interval median). (K) Fractional shortening. (L) Diastolic diameter. (M) Systolic diameter. (N) Microscopic image of cardiac function from M-mode trace. 1: hand-Gal4>w11118 flies; 2: hand-Gal4>UAS-dSir2RNAi flies; 3: 7-week-old hand-Gal4>w11118 flies; 4: 7-week-old hand-Gal4>UAS-dSir2-RNAi flies. It can be observed from 1 and 2 that the cardiac dSir2 knockdown could reduce heart period and fractional shortening, and increase diastolic dysfunction. Independent-sample t tests were used to assess differences between the hand-Gal4>w11118 and hand-Gal4>UAS-dSir2RNAi flies to explore the effects of cardiac dSir2 knockdown on the heart. Data are represented as means ± SEM. *P<0.05; **P <0.01. Sample size was the same as in our previous experiments.
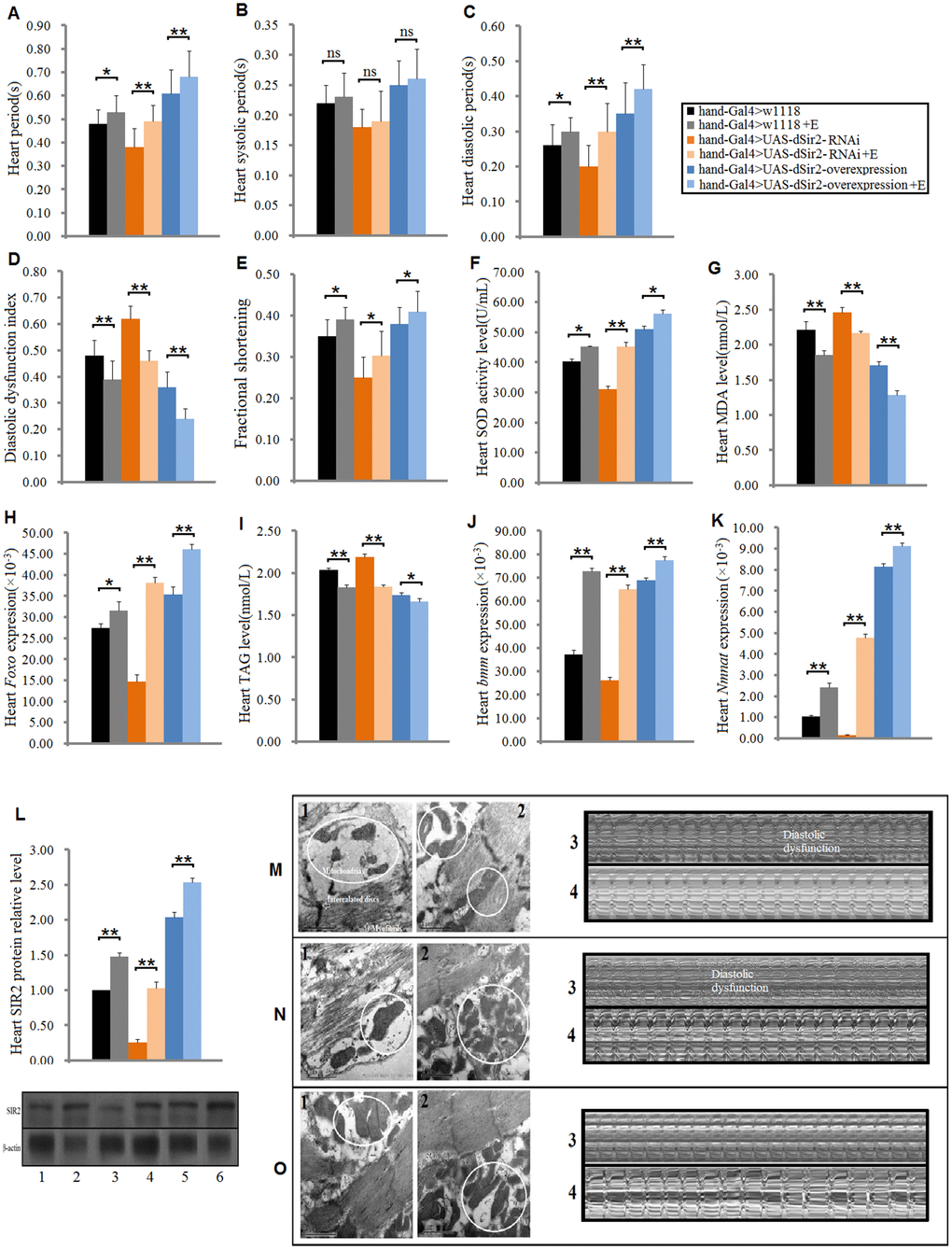
Cardiac dSir2 overexpression slows down heart aging in Drosophila
To further confirm whether cardiac dSir2 could regulate heart aging, the cardiac dSir2 overexpression line was built by UAS-dSir2 overexpression (w1118; P{EP}Sirt1EP2300 DnaJ-HEP2300/CyO) and hand-Gla4 in 7-week-old flies. Although our previous results indicated that cardiac dSir2 knockdown promoted heart aging and aging reduced cardiac dSir2 expression, it remains unknown whether cardiac dSir2 overexpression can affect heart aging. The results showed that the cardiac dSir2 expression of hand-Gal4>UAS-dSir2-overexpression flies was significant higher than that of hand-Gal4>w1118 flies (P<0.01) (a difference of about 5.01-fold) (Figure 4A), and the cardiac dSir2 expression of hand-Gal4>UAS-dSir2-overexpression flies was also significantly higher than that of UAS-dSir2-overexpression>w1118 flies (P<0.01) (a difference of about 6.6-fold) (Figure 4A). This suggested that the cardiac dSir2 overexpression was successfully induced by the UAS/hand-Gal4 system in 7-week-old Drosophila. Besides, the results showed that cardiac dSir2 overexpression significantly increased the SOD activity level and Foxo expression (P<0.01; Figure 4B and 4C), and notably decreased MDA levels in aging hearts (P<0.01; Figure 4D). Furthermore, cardiac dSir2 overexpression significantly decreased cardiac TAG level (P<0.01; by 17.6%; Figure 4E), and notably increased cardiac bmm expression (P<0.01; Figure 4F). Finally, cardiac dSir2 overexpression significantly increased heart period, diastolic period, and fractional shortening (P<0.01; Figure 3G, 3I, 3K, and 3N), and notably decreased the diastolic dysfunction index (P<0.01; Figure 3J and 3N). Although a dnaJ-homolog (dnaJ-H) gene partially overlaps with dSir2, increasing evidence indicates that moderately increased expression of dSir2 (2.5-fold, 3-fold, and 5-fold) from the native dSir2 locus can result in lifespan extension without an increase in dnaJ-H mRNA expression [52, 53]. The results showed that the heart dnaJ-H gene expression was not significantly changed by dSir2 overexpression (P>0.05; Figure 3O). Therefore, our results suggest that cardiac dSir2 overexpression could decrease the risk of oxidative damage, fat accumulation, and diastolic dysfunction in aging hearts, thus resisting the phenotypes associated with heart aging.
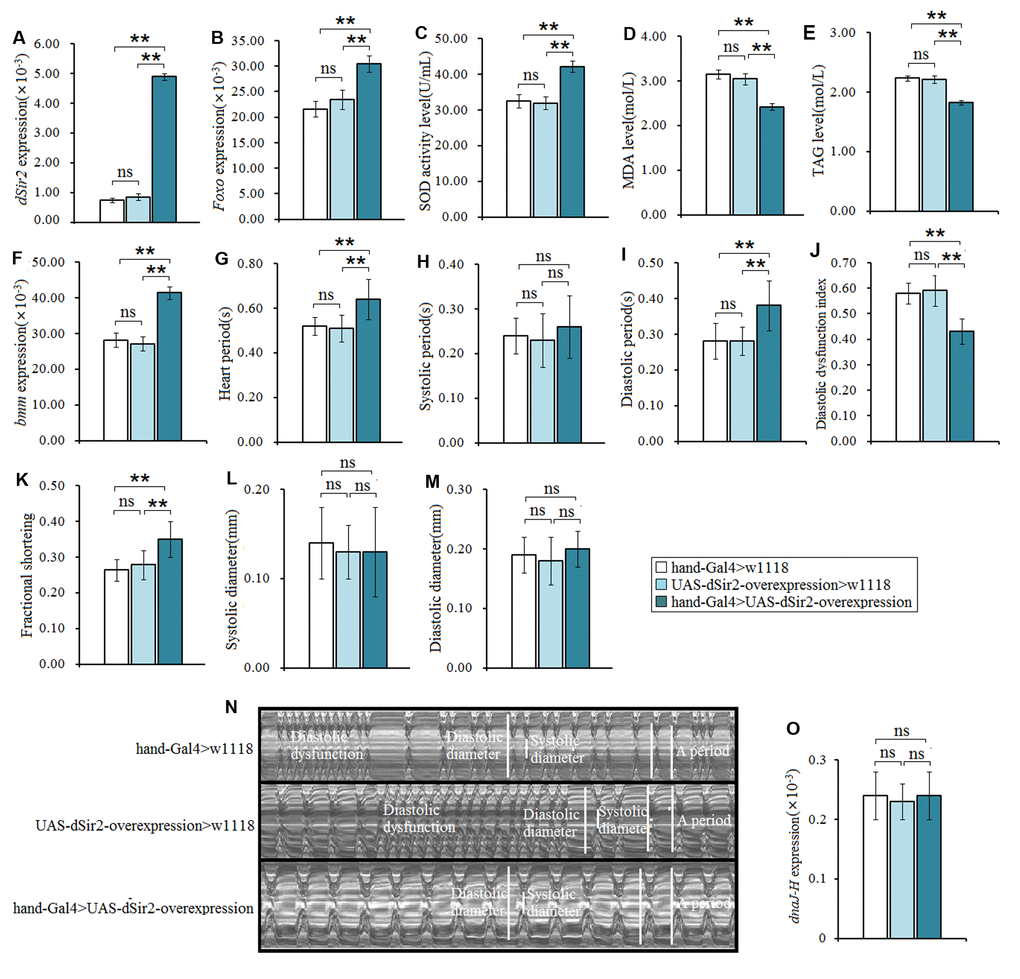
Figure 4. The influence of cardiac dSir2 overexpression on the heart in 7-weeek-old flies. (A) Cardiac dSir2 expression. (B) Cardiac Foxo expression. (C) Cardiac SOD activity level. (D) Cardiac MDA level. (E) Cardiac TAG level. (F) Cardiac bmm expression. (G) Heart period. (H) Heart systolic period. (I) Heart diastolic period. (J) Heart diastolic dysfunction index. The diastolic dysfunction index is diastolic interval standard deviation/diastolic interval median). (K) Fractional shortening. (L) Diastolic diameter. (M) Systolic diameter. (N) Microscopic image of cardiac function from M-mode trace. It can be observed that the cardiac dSir2 overexpression could increase heart period and fractional shortening, and decrease diastolic dysfunction. (O) Cardiac dnaJ-H expression level. One-way analysis of variance (ANOVA) with least significant difference (LSD) tests were used to identify differences among the hand-Gal4>w11118, UAS-dSir2-overexpression>w11118, and hand-Gal4>UAS-dSir2-overexpression flies to explore the effects of cardiac dSir2 overexpression on aging hearts. Data are represented as means ± SEM. *P<0.05; **P <0.01. The sample size was the same as in our previous experiments.
Effect of physical exercise and cardiac dSir2 differential expression on mobility and lifespan in Drosophila
To explore whether the cardiac Sir2 gene could affect the flies’ climbing ability and longevity, the climbing index and lifespan of the experimental flies were measured. As we all know, the heart is a very important organ to exercise in vertebrate animals since the brain and skeletal muscles get oxygen and nutrients by pumping blood through the heart. Especially during exercise or activity, if the brain and skeletal muscles do not get enough oxygen and nutrients, it will lead to reduced athletic ability and proneness to fatigue [57, 58]. Although the skeletal muscles and brain of flies get their oxygen without having to pump hemolymph through the heart, they need to get nutrition and energy from hemolymph [59, 60]. In this respect, there may be a relationship between heart function and climbing ability in fruit flies. Besides, exercise training has been shown to improve heart function and prevent some heart diseases [25, 36, 61]. Numerous studies have reported that age-related heart disease is the leading cause of death in the elderly [62–64]. These findings suggest that cardiac function is important for mobility and survival in older individuals. However, it remains unclear whether cardiac dSir2 different expression can affect the fruit fly's motor ability and longevity.
In this study, results showed that aging significantly reduced the climbing index in cardiac dSir2 differential-expression flies (P<0.01; Figure 6A and 6C). Cardiac dSir2 knockdown significantly reduced the climbing index in 5-week-old and 7-week-old flies (P<0.05), and also notably decreased the average lifespan of flies (P<0.05; Figure 6B, 6E, and 6F). On the contrary, cardiac dSir2 overexpression significantly increased the climbing index in 5-week-old flies (P<0.05) and notably increased the average lifespan of flies (P<0.05; Figure 6D, 6G, and 6H). For 1-week-old and 3-week-old flies, cardiac dSir2 knockdown and overexpression did not remarkably affect the climbing index (P>0.05; Figure 6A and 6C). The results also showed that in 1-week-old and 3-week-old and cardiac dSir2 differential-expression flies, the climbing index had no significant difference between the exercise group and no exercise group (P>0.05; Figure 7A). This suggested that before exercise training, the climbing index of the exercise group was almost the same as that of the no exercise group. After 2 weeks of exercise training, the climbing index was notably increased in cardiac dSir2 differential-expression and 5-week-old flies (P<0.05, P<0.01). Exercise training also notably increased the climbing index in cardiac dSir2 over- or normal-expression and 7-week-old flies (P<0.05), but this effect was not found in cardiac dSir2 knockdown and 7-week-old flies (P>0.05; Figure 7A). Moreover, exercise remarkably prolonged the average lifespan of cardiac dSir2 differential-expression flies (P<0.05, P<0.01; Figure 7B and 7C). Therefore, these results suggested that cardiac Sir2 overexpression and exercise in old flies could improve their mobility and longevity to a certain extent. Cardiac Sir2 knockdown reduced the mobility and survivability of flies, but these changes could be reversed by exercise training to a certain extent.
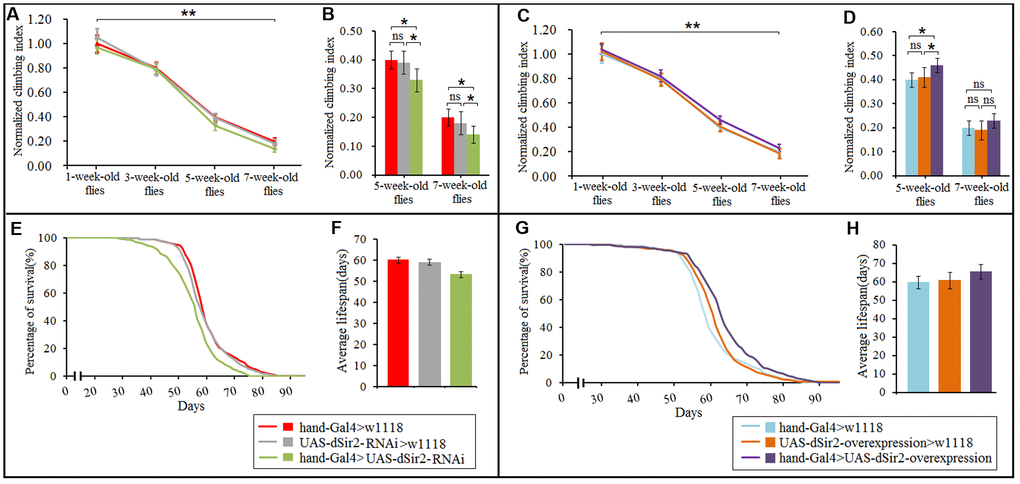
Figure 6. Effect of cardiac dSir2 differential-expression on the climbing index and average lifespan in Drosophila. (A) The climbing index change curves with aging of cardiac dSir2 knockdown flies. (B) The climbing index of cardiac dSir2 knockdown flies. The sample size was about 100 flies for each group. (C) The climbing index change curves with aging in cardiac dSir2 overexpression flies. (D) The climbing index of cardiac dSir2 overexpression flies. The sample size was about 100 flies for each group. (E) Fly population survival (%) curve of cardiac dSir2 knockdown flies. The leftmost curve represents the cardiac dSir2 knockdown group, of which flies had the shortest lifespan. (F) The average lifespan of cardiac dSir2 knockdown flies. The sample size was 200–220 flies for each group. (G) Fly population survival (%) curve of cardiac dSir2 overexpression flies. The leftmost curve represents the cardiac dSir2 overexpression group, of which flies had the longest lifespan. (H) The average lifespan of cardiac dSir2 overexpression flies. The sample size was 200–220 flies for each group. P-values for lifespan curves were calculated by the log-rank test. Data are represented as means ± SEM. *P<0.05; **P <0.01.
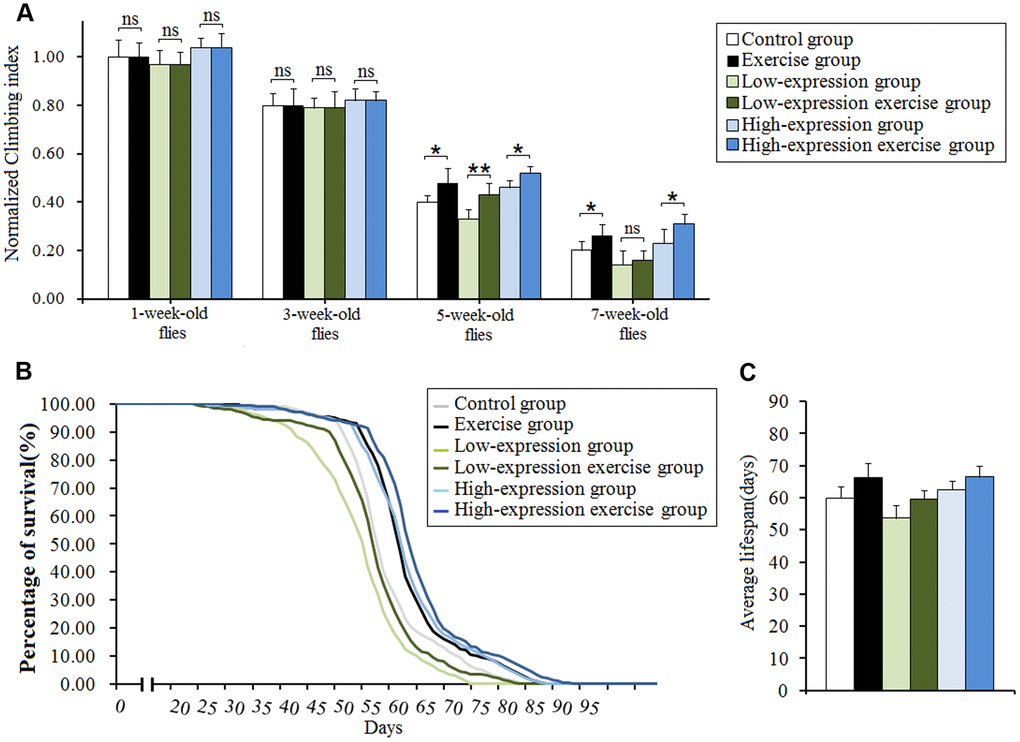
Figure 7. Effect of exercise and cardiac Sir2 on the climbing index and average lifespan in Drosophila. (A) The climbing index. The sample size was about 100 flies for each group. (B) The average lifespan. The sample size was 200–220 flies for each group. (C) Percents of survival curve. The rightmost curve represents the cardiac dSir2 overexpression combined with exercise group, of which flies had the longest lifespan. P-values for lifespan curves were calculated by the log-rank test. Data are represented as means ± SEM. *P<0.05; **P <0.01.
Discussion
In both mammals and fruit flies, the Sir2/Sirt1 gene is closely related to oxidative stress and lipid metabolism. In mammalian cells, the Sir2 homolog SIRT1 appears to control the cellular response to stress by regulating the FOXO family of Forkhead transcription factors, a family of proteins that function as sensors of the insulin signaling pathway and as regulators of organismal longevity. SIRT1 increased FOXO3's ability to induce cell cycle arrest and resistance to oxidative stress but inhibited FOXO3's ability to induce cell death [65]. In addition, hepatic SIRT1 plays an important role in hepatic fatty acid metabolism, and it has been shown that adenoviral knockdown of SIRT1 reduces expression of fatty acid β-oxidation genes in the liver of fasted mice [66]. SIRT1 can also regulate the brown remodeling of white adipose tissue in response to cold exposure by deacetylation of PPARγ, and SIRT1-dependent deacetylation of PPARγ is required to recruit the brown adipose tissue program coactivator Prdm16 to PPARγ, leading to selective induction of brown adipose tissue genes and repression of white adipose tissue genes [67]. However, it remains unclear whether the cardiac Sirt1 gene can regulate heart aging.
In Drosophila, overexpression of Sir2 enhances the survival of a number of model organisms undergoing calorie restriction, during which insulin receptor signaling is reduced, a condition that can enhance survivorship during starvation. Besides, defects in peripheral insulin sensitivity, but not in insulin secretion, can account for the reduced insulin signaling in sir2 mutants, and GAL4-driven expression of Sir2 in the fat body of wild-type flies is sufficient to reduce TAG levels, which is consistent with previous reports of SIRT1 overexpression in mice [68]. Moreover, overexpression of Sir2 in the adult fat body is sufficient to extend the lifespan of male and female Drosophila [53]. Importantly, increased Sir2 expression and reduced insulin receptor signaling results in an increase in the activity of the transcription factor FOXO [69]. In fly neuronal cells, increased Dyrk1a activates Sir2 to regulate the deacetylation of FOXO, which potentiates FOXO-induced sNPF/NPY expression and in turn promotes food intake. Recent studies report that modest Foxo overexpression is cardioprotective, ameliorating nonpathological functional decline with age in flies [70]. The expression of Foxo in myocardial cells autonomously protects the heart from the adverse effects of a high-fat diet [16]. Therefore, Sir2 is involved in the regulation of oxidative stress and lipid metabolism, possibly through the regulation of Foxo. However, it remains unclear whether Sir2 can upstream regulate Foxo in the heart of fruit flies.
In this study, we found that cardiac Sir2 overexpression increased cardiac Foxo expression in aging flies. The activation of FOXO transcription factors leads to the induction of a variety of genes encoding antioxidant proteins, including MnSOD [14]. Therefore, cardiac Sir2 overexpression increased heart SOD activity level and decreased MDA level via activating the Sir2/Foxo/SOD pathway in fly’s hearts; this may improve the antioxidant capacity of myocardial cells and reduce oxidative damage to myocardial cells. On the contrary, cardiac Sir2 knockdown or aging reduced heart SOD activity and increased heart MDA level via inhibiting the cardiac Sir2/Foxo/SOD pathway in Drosophila, which may weaken the antioxidant capacity of myocardial cells and increase oxidative damage to myocardial cells. We also found that cardiac Sir2 overexpression effectively reduced the level of cardiac TAG and increased cardiac bmm expression. Because it has been reported that there is no significant increase in heart-specific TAG accumulation in hearts of flies with high-fat diets when overexpressing either Foxo or bmm [16], cardiac Sir2 overexpression may decrease cardiac lipid accumulation via activating the cardiac Sir2/Foxo/bmm pathway in aging flies. Oppositely, cardiac Sir2 knockdown or aging caused excessive fat accumulation in the heart by suppressing the cardiac Sir2/Foxo/bmm pathway in flies. Furthermore, we found that cardiac Sir2 overexpression could reduce diastolic dysfunction. Since oxidative stress and excessive lipid accumulation are the main causes of cardiac diastolic dysfunction [71, 72], cardiac Sir2 overexpression may reduce the risk of diastolic dysfunction by activating the cardiac Sir2/Foxo/SOD pathway and Sir2/Foxo/bmm pathway. Inversely, cardiac Sir2 knockdown or aging increased the incidence of diastolic dysfunction by inhibiting the cardiac Sir2/Foxo/SOD pathway and Sir2/Foxo/bmm pathway in aging flies. Finally, we found that cardiac Sir2 overexpression could increase heart period and fractional shortening in aging flies, and this indicated that cardiac Sir2 overexpression enhanced heart contractility. Meanwhile, cardiac Sir2 knockdown reduced heart period and fractional shortening in aging flies. Therefore, for heart period, cardiac Sir2 knockdown seemed to resist aging, but this is not actually the case. In order to maintain normal metabolism and cardiac output, the heart may compensate for the reduced fractional shortening by reducing the cardiac period in cardiac Sir2 knockdown flies. The cardiac fractional shortening may be decreased by lipid accumulation and oxidative damage [71–74]. This suggests that cardiac Sir2 knockdown attenuated heart contractility. In aging flies, since their metabolic rate drops, the cardiac output may be maintained by decreasing the cardiac fractional shortening or increasing the cardiac period. Of course, aging may also reduce cardiac fractional shortening via increasing lipid accumulation and oxidative damage [3, 5, 6]. Therefore, we cardiac aging is majorly characterized by increased diastolic dysfunction, accumulation of lipid, oxidative stress, and reduced contractility in Drosophila. Cardiac Sir2 overexpression effectively delayed heart aging via activating the Sir2/Foxo/bmm pathway and Sir2/Foxo/SOD pathway, but cardiac Sir2 knockdown contributed to the aging process of the heart via inhibiting the Sir2/Foxo/bmm pathway and Sir2/ Foxo/SOD pathway.
Exercise training can prevent lipid accumulation in both aging mammals and fruit flies. Endogenous TAG represents an important source of fuel for endurance exercise. TAG oxidation increases progressively during exercise, and the specific rate is determined by energy requirements of working muscles, fatty acid delivery to muscle mitochondria, and the oxidation of other substrates. The catecholamine response to exercise increases lipolysis of adipose tissue TAGs, and, presumably, intramuscular TAGs. Moreover, increases in adipose tissue and muscle blood flow decreases fatty acid re-esterification and facilitates the delivery of released fatty acids to skeletal muscle [75]. Therefore, exercise training can accelerate the decomposition of free fatty acids to provide energy. Moreover, it can promote the decomposition of adipose tissue to provide energy, which can effectively prevent excessive accumulation of lipids in tissues and organs such as the liver and heart. In Drosophila, it has been reported that endurance exercise can effectively prevent excessive accumulation of lipids in the body and heart induced by a high-fat diet or aging, and this may be related to the benign regulation of Sir2 expression through exercise [35]. However, it remains unclear whether exercise training resistance to heart lipid accumulation is related to cardiac Sir2 gene activation. Besides, exercise training can improve cardiac function in both aging mammals and fruit flies. For example, swimming exercise upregulates antioxidant defense capacity and improves structural abnormalities of the senescent female rat heart, and it improves the contractile function of aged myocardium by mitigating the detrimental effects of oxidative stress [61]. Exercise training enhances the SIRT longevity pathway instead of IGF1 survival signaling to improve cardiomyocyte survival [30]. In Drosophila, exercise training can reduce age-related decline in mobility and cardiac performance [32]. Fatiguing exercise initiated later in life reduces the incidence of cardiac fibrillation in Drosophila [36]. Normal expression or overexpression of CG9940 had a positive influence on the adaptation of cardiac functions, mobility, and lifespan to exercise in aging Drosophila [34]. Therefore, since both the cardiac Sir2/Sirt1 gene and exercise training take part in regulating heart aging, it is important to understand the relationship between them.
In this study, for the first time, we found that in cardiac dSir2 overexpression, normal expression, knockdown, and 5-week-old flies, exercise training improved cardiac dSir2 expression and SIR2 protein levels. We also found that in the cardiac Sir2 differential-expression flies, exercise training increased cardiac SOD activity level and decreased MDA levels via activating the cardiac dSir2/Foxo/SOD pathway; these changes indicated that exercise training improved the antioxidant capacity of myocardial cells. In addition, exercise decreased cardiac lipid accumulation via activating cardiac the dSir2/Foxo/bmm pathway in cardiac Sir2 differential-expression and 5-week-old flies. Moreover, the risk of diastolic dysfunction was reduced by exercise training in cardiac Sir2 differential-expression and 5-week-old flies. Finally, we found that exercise training increased heart period and fractional shortening in cardiac Sir2 differential-expression and 5-week-old flies, and these changes indicated that exercise training enhanced heart contractility. These results suggested that the activation of the cardiac dSir2/Foxo/SOD pathway and cardiac dSir2/Foxo/bmm pathway may be two important molecular mechanisms through which exercise fights against heart aging.
Although it has been reported that exercise training can enhance the SIRT longevity pathway instead of IGF1 survival signaling to improve cardiomyocyte survival [30], the mechanism by which dSir2 expression is up-regulated in exercise training remains unclear. Increasing evidence has shown that exercise training increases muscle and blood NAD+ levels [54, 55], and the NAD+ has an upstream regulation function for Sir2/Sirt1 [7]. Therefore, we speculate that the dSir2 expression up-regulated by exercise training in the heart may be achieved by improving the cardiac NAD+ level. To test this hypothesis, we examined the expression of the Nmnat gene in the heart. Nicotinamide mononucleotide adenylyltransferease (NMNAT), a rate-limiting enzyme present in all organisms, reversibly catalyzes the important step in the biosynthesis of NAD from ATP and NMN, and overexpression of Nmnat has been shown to increase NAD+ levels in some tissues [56]. We found that both dSir2 overexpression and exercise training could improve Nmnat expression in the heart. This may be the result of the positive adaptive changes in the synthesis of related genes caused by the long-term high demand for NAD+ in the heart. However, this speculation needs to be confirmed by further experiments.
Age-related decline in heart function and heart disease are important causes of reduced physical activity and death in the elderly [22]. Previous findings showed that the cardiac dSir2 gene can affect heart aging in flies, but it remains unclear whether the cardiac dSir2 gene can affect their mobility and lifespan. Our results showed that cardiac dSir2 overexpression and exercise in old flies could improve their mobility and survivability to a certain extent. However, cardiac dSir2 knockdown reduced mobility and survivability in old flies. In young flies, cardiac dSir2 gene had no significant effect on climbing ability, and the reason for this may be that changes in heart function induced by cardiac Sir2 differential expression were not enough to cause hypoxia in tissues such as the brain [57, 58]. Cardiac Sir2 knockdown may contribute to age-related heart disease and lead to death in the elderly [62–64]. In addition, we found that exercise training improved the mobility in cardiac dSir2 differential-expression and old flies. Moreover, exercise training prolonged the average lifespan in cardiac dSir2 differential-expression flies. These findings suggest that exercise training could reverse the decline of mobility and survivability induced by cardiac dSir2 knockdown to a certain extent in old flies. This may be related to the reconstruction of the NAD/dSir2 pathway in the heart by exercise training. Besides, exercise training could improve the functioning of other organs and systems, and could prevent other age-related diseases, such as obesity, diabetes, and Parkinson's disease, which could thus extend lifespan in old flies [76, 77]. Therefore, this may be the reason why cardiac dSir2 overexpression combined with exercise training had the best benefit to lifespan and mobility.
In conclusion, we claim that cardiac Sir2 overexpression or knockdown can delay or promote heart aging by reducing or increasing age-related oxidative stress, lipid accumulation, diastolic dysfunction, and contractility debility. Activation of the cardiac Sir2/Foxo/SOD pathway and cardiac Sir2/Foxo/bmm pathway may be two important molecular mechanisms through which exercise fights against heart aging in Drosophila.
Materials and Methods
Fly stocks, diet, and husbandry
w1118 and hand-Gal4 flies were gifts from Xiu-shan Wu (Heart Development Center of Hunan Normal University). UAS-dSir2-overexpression (w1118; P{EP} Sirt1EP2300 DnaJ-HEP2300/CyO) flies were obtained from the Bloomington Stock Center. UAS-dSir2-RNAi (w1118; P{GD11580}v23201and P{KK109370}VIE-260B) lines were obtained from the Vienna Drosophila RNAi Center. To induce different expression of the dSir2 gene in the fly heart, male hand-Gal4 flies were crossed with female w1118 flies, UAS-dSir2-overexpression flies, and UAS-dSir2-RNAi flies [14]. All UAS and GAL4 insertions were backcrossed into the w1118 line at least 10 times to avoid excess phenotypes affecting the experimental results. Normal food contained 10% yeast, 10% sucrose, and 2% agar [16]. During the experimental time course, flies were housed in a 25°C incubator with 50% humidity and a 12-h light/dark cycle. Fresh food was provided every other day for the duration of the experiment. All flies were raised to the fourth weekend, and were then trained in their fifth week of life since we found that flies were more sensitive to exercise during this stage of life.
Exercise training device and protocols
When constructing the exercise device, the flies’ natural negative geotaxis behavior was taken to induce upward walking [78]. All exercise group flies started exercise from when they were 22 days old, and underwent a 2-week-long exercise program. Vials with the diet and housing 25 flies each were loaded horizontally into a steel tube that was rotated about its horizontal axis by an electric motor, with a gear regulating its shaft speed. Thus, with the accompanying rotating steel tube, each vial was rotated along its long axis, which made the flies climb (TreadWheel) [31, 33]. Most flies continued to respond by climbing throughout the exercise period. The few that failed to climb were actively walking at the inner wall of the vial [34, 36]. Flies were exercised in vials with a 2.8 cm inner diameter, rotated at 0.16 rev/s. Flies were exercised for 1.5 hours per day.
Semi-intact Drosophila preparation and image analysis
Flies were anesthetized with FlyNap for 2–3 min. The head, ventral thorax, and ventral abdominal cuticle were removed by special glass needles to expose the heart and abdomen. Dissections were done under oxygenated artificial hemolymph. These semi-intact preparations were allowed to equilibrate with oxygenation for 15–20 min before filming. Image analysis of heart contractions was performed using high-speed videos of the preparations. Videos were taken at 120–130 frames per second using a Hamamatsu (McBain Instruments, Chats worth, CA) EM-CCD digital camera on a Leica (McBain Instruments, Chatsworth, CA) DM LFSA microscope with a 10x immersion lens. To get a random sampling of heart function, a single 30-s recording was made for each fly. All images were acquired and contrast enhanced using Simple PCI imaging software (Compix, Sewickley, PA). The heart physiology of the flies was assessed using a semi-automated optical heartbeat analysis program that quantifies the heart period, systolic period, diastolic period, diastolic dysfunction index (diastolic intervals standard deviation/diastolic intervals median), fractional shortening, diastolic diameter, and systolic diameter [79]. The sample size was 30 flies for every group. Since the arrhythmia index calculates the heart period standard deviation normalized to the median heart period, we calculated the diastolic arrhythmicity index as the heart diastolic period standard deviation normalized to the median heart diastolic period.
ELISA assay
The SOD activity level, MDA level, and TAG level were measured by ELISA assay (Insect SOD activity, MDA, and TAG ELISA Kits, MLBIO). Fly hearts were homogenized in PBS (pH 7.2–7.4). Samples were rapidly frozen with liquid nitrogen and then maintained at 2°C–8°C after melting. Homogenize the samples with grinders, and centrifugation was conducted for 20 min at 2000–3000 rpm. Then we removed the supernatant. The specific steps are as follows: (1) Add standard: set standard wells, and test sample wells. Add standard 50 μl to the standard well. (2) Add sample: add 40μl of sample dilution to the testing sample well, then add 10 μl of the sample (the sample’s final dilution is 5-fold). Avoid touching the well wall as much as possible, and gently mix. (3) Add enzyme: add 100 μl of HRP-conjugate reagent to each well except for the blank well. Don’t add sample and HRP-Conjugate reagent to blank comparison wells, and other each step operation is same. (4) Incubate: after closing the plate with the closure plate membrane, incubate for 60 min at 37°C. (5) Washing: uncover the closure plate membrane, discard liquid, dry by swing, add washing buffer to every well, still for 30 s and then drain; repeat 5 times, and pat dry. (6) Color: add 50 μl of Chromogen Solution A and Chromogen Solution B to each well, evade the light preservation, and let sit for 15 min at 37°C. (7) Stop the reaction: add 50 μl of Stop Solution to each well to stop the reactions (the blue color will change to yellow). (8) Assay: take the blank well as zero, and read absorbance at 450 nm within 15 min of adding Stop Solution.
qRT-PCR
About 80 hearts from each group were homogenized in Trizol. First, 10 μg of the total RNA was purified by organic solvent extraction from the Trizol (TRIzol, Invitrogen). The purified RNA was treated with DNase I (RNase-free, Roche) and used to produce oligo dT-primed cDNAs (SuperScript II RT, Invitrogen), which were then used as templates for quantitative real-time PCR. The rp49 gene was used as an internal reference for normalizing the quantity of total RNAs. Real-time PCR was performed with SYBR green using an ABI7300 Real-time PCR Instrument (Applied Biosystems). Expression of the various genes was determined by the comparative CT method (ABI Prism 7700 Sequence Detection System User Bulletin #2, Applied Biosystems). Primer sequences of Sir2 were as follows: F: 5′-GCAGT GCCAGCCC AATAA-3′; R: 5′-AGCCGATCACGATC AGTAGA-3′. Primer sequences of bmm were as follows: F: F: 5′-ACTGCAC ATTTCGCTTACCC-3′; R: 5′-GAG AATCCGGGTATGAAGCA-3′. Primer sequences of Foxo were as follows: F: 5′-AACAACAGCAGCATC AGCAG-3′; R: 5′-CTGAACCCGAGCATTCAGAT-3′. Primer sequences of dnaJ-H were as follows: F: 5′-GCAAGATGGCACACGTAGCTG-3′; R: 5′-CCACTG TAGCAACACGTAATCACC-3′. Primer sequences of Rp49 were as follows: F: 5 -CTAAGCTG TCGCACAA ATGG-3′; R: 5′-AACTTCTTGAATCCGGTG GG-3′.
Western blots
Samples of 80 hearts were collected under the indicated conditions at 35 days of age, and homogenized in 100 μL of RIPA buffer containing 1X protease inhibitors (Roche cOmplete Mini EDTA-free protease inhibitor tablets). For dSir2 Western blots, the buffer also contained Calyculin A and okadaic acid. Equivalent amounts of protein were resolved by SDS-PAGE (10% acrylamide), transferred to PVDF membrane overnight at 4°C, and blocked with 5% BSA prior to immunoblotting. Western blots were probed with antibodies for dSir2 (1:50, Developmental Studies Hybridoma Bank #p4A10) and β-actin (1:1000, Cell Signaling #9441). The data shown in the figure5-k are representative of at least three biological replicates. Quantification was performed by measuring protein levels using Image J software. The values reported represent the experimental condition normalized to the control, unless otherwise specified.
Negative geotaxis assay
The climbing apparatus consisted of an 18-cm-long vial with an inner diameter of 2.8 cm, and flies were allowed to adapt to the vial for 10 min before assessing negative geotaxis. Sponges were placed in the ends of the tube to prevent escape while allowing air exchange. With a light box behind the vials, the rack was tapped down five times and on the fifth, a timed digital camera snapped a picture after 8 s. The extent of climbing could be analyzed visually or by imaging software. Five pictures of each group were taken and averaged to arrive at a fixed score for each vial. The total score for all the flies in a vial was tallied, and then divided by the number of flies in the vial to generate the “climbing index” for that trial. Each vial was subjected to 5 trials, and then the indices from the five trials were averaged [78].
Lifespan assays
Dead flies were recorded daily. Lifespan was estimated for each fly as the number of days alive from the day of hatching to the day of death. Mean and median lifespan and survival curves were used to characterize the lifespan. Sample sizes were 200 to 210 flies per group [80].
Statistical analyses
The 1-way analysis of variance (ANOVA) with least significant difference (LSD) tests was used to identify differences among the hand-Gal4>w11118, UAS-dSir2-overexpression>w11118, and hand-Gal4>UAS-dSir2-overexpression flies. Independent-sample t tests were used to assess differences between the hand-Gal4>w11118 and hand-Gal4>UAS-dSir2-RNAi flies. Analyses were performed using the Statistical Package for the Social Sciences (SPSS) version 16.0 for Windows (SPSS Inc., Chicago, USA), with statistical significance set at P<0.05. Data are represented as means ± SEM.
Acknowledgments
The authors thank Xiu-shan Wu (The Center for Heart Development, College of Life Science, Hunan normal University) for supporting Drosophila of w1118 and heart Shoot software technology. We also thank Karen Ocorr and Rolf Bodmer (American burnham medical institute of neurology and aging center) for supporting semi-automatic optical echocardiography analysis software. We thank LetPub (https://www.letpub.com) for its linguistic assistance during the preparation of this manuscript.
Conflicts of Interest
Authors have no conflicts of interest.
Funding
This work is supported by the National Natural Science Foundation of China (No. 31671243).
References
- 1. McCullough PA, Philbin EF, Spertus JA, Kaatz S, Sandberg KR, Weaver WD, and Resource Utilization Among Congestive Heart Failure (REACH) Study. Confirmation of a heart failure epidemic: findings from the Resource Utilization Among Congestive Heart Failure (REACH) study. J Am Coll Cardiol. 2002; 39:60–69. https://doi.org/10.1016/S0735-1097(01)01700-4 [PubMed]
- 2. Ouwerkerk W, Voors AA, Zwinderman AH. Factors influencing the predictive power of models for predicting mortality and/or heart failure hospitalization in patients with heart failure. JACC Heart Fail. 2014; 2:429–36. https://doi.org/10.1016/j.jchf.2014.04.006 [PubMed]
- 3. Drosatos K. Fatty old hearts: role of cardiac lipotoxicity in age-related cardiomyopathy. Pathobiol Aging Age Relat Dis. 2016; 6:32221. https://doi.org/10.3402/pba.v6.32221 [PubMed]
- 4. Klassen MP, Peters CJ, Zhou S, Williams HH, Jan LY, Jan YN. Age-dependent diastolic heart failure in an in vivo Drosophila model. eLife. 2017; 6:e20851. https://doi.org/10.7554/eLife.20851 [PubMed]
- 5. Obas V, Vasan RS. The aging heart. Clin Sci (Lond). 2018; 132:1367–82. https://doi.org/10.1042/CS20171156 [PubMed]
- 6. Pan B, Xu ZW, Xu Y, Liu LJ, Zhu J, Wang X, Nan C, Zhang Z, Shen W, Huang XP, Tian J. Diastolic dysfunction and cardiac troponin I decrease in aging hearts. Arch Biochem Biophys. 2016; 603:20–28. https://doi.org/10.1016/j.abb.2016.05.008 [PubMed]
- 7. Smith JS, Brachmann CB, Celic I, Kenna MA, Muhammad S, Starai VJ, Avalos JL, Escalante-Semerena JC, Grubmeyer C, Wolberger C, Boeke JD. A phylogenetically conserved NAD+-dependent protein deacetylase activity in the Sir2 protein family. Proc Natl Acad Sci USA. 2000; 97:6658–63. https://doi.org/10.1073/pnas.97.12.6658 [PubMed]
- 8. Frankel S, Rogina B. Sir2, caloric restriction and aging. Pathol Biol (Paris). 2006; 54:55–57. https://doi.org/10.1016/j.patbio.2005.04.003 [PubMed]
- 9. Shen T, Ding L, Ruan Y, Qin W, Lin Y, Xi C, Lu Y, Dou L, Zhu Y, Cao Y, Man Y, Bian Y, Wang S, et al. SIRT1 functions as an important regulator of estrogen-mediated cardiomyocyte protection in angiotensin II-induced heart hypertrophy. Oxid Med Cell Longev. 2014; 2014:713894. https://doi.org/10.1155/2014/713894 [PubMed]
- 10. Passariello CL, Zini M, Nassi PA, Pignatti C, Stefanelli C. Upregulation of SIRT1 deacetylase in phenylephrine-treated cardiomyoblasts. Biochem Biophys Res Commun. 2011; 407:512–16. https://doi.org/10.1016/j.bbrc.2011.03.049 [PubMed]
- 11. Gu XS, Wang ZB, Ye Z, Lei JP, Li L, Su DF, Zheng X. Resveratrol, an activator of SIRT1, upregulates AMPK and improves cardiac function in heart failure. Genet Mol Res. 2014; 13:323–35. https://doi.org/10.4238/2014.January.17.17 [PubMed]
- 12. Lu TM, Tsai JY, Chen YC, Huang CY, Hsu HL, Weng CF, Shih CC, Hsu CP. Downregulation of Sirt1 as aging change in advanced heart failure. J Biomed Sci. 2014; 21:57. https://doi.org/10.1186/1423-0127-21-57 [PubMed]
- 13. Alcendor RR, Gao S, Zhai P, Zablocki D, Holle E, Yu X, Tian B, Wagner T, Vatner SF, Sadoshima J. Sirt1 regulates aging and resistance to oxidative stress in the heart. Circ Res. 2007; 100:1512–21. https://doi.org/10.1161/01.RES.0000267723.65696.4a [PubMed]
- 14. Koh H, Kim H, Kim MJ, Park J, Lee HJ, Chung J. Silent information regulator 2 (Sir2) and Forkhead box O (FOXO) complement mitochondrial dysfunction and dopaminergic neuron loss in Drosophila PTEN-induced kinase 1 (PINK1) null mutant. J Biol Chem. 2012; 287:12750–58. https://doi.org/10.1074/jbc.M111.337907 [PubMed]
- 15. Ma J, Chen L, Song D, Zhang Y, Chen T, Niu P. SIRT1 attenuated oxidative stress induced by methyl tert-butyl ether in HT22 cells. Toxicol Res (Camb). 2017; 6:290–96. https://doi.org/10.1039/C7TX00016B [PubMed]
- 16. Birse RT, Choi J, Reardon K, Rodriguez J, Graham S, Diop S, Ocorr K, Bodmer R, Oldham S. High-fat-diet-induced obesity and heart dysfunction are regulated by the TOR pathway in Drosophila. Cell Metab. 2010; 12:533–44. https://doi.org/10.1016/j.cmet.2010.09.014 [PubMed]
- 17. Blice-Baum A, Kaushik G, Viswanathan M, Zambon A, Engler A, Bodmer R, Cammarato A. Overexpression of Foxo in the Heart Ameliorates Performance Decline through Enhanced UPS Processing in Aging Drosophila. Biophys J. 2015 (Suppl 1); 108:361a. https://doi.org/10.1016/j.bpj.2014.11.1979
- 18. Diop SB, Bodmer R. Drosophila as a model to study the genetic mechanisms of obesity-associated heart dysfunction. J Cell Mol Med. 2012; 16:966–71. https://doi.org/10.1111/j.1582-4934.2012.01522.x [PubMed]
- 19. Hori YS, Kuno A, Hosoda R, Horio Y. Regulation of FOXOs and p53 by SIRT1 modulators under oxidative stress. PLoS One. 2013; 8:e73875. https://doi.org/10.1371/journal.pone.0073875 [PubMed]
- 20. Lin CH, Lin CC, Ting WJ, Pai PY, Kuo CH, Ho TJ, Kuo WW, Chang CH, Huang CY, Lin WT. Resveratrol enhanced FOXO3 phosphorylation via synergetic activation of SIRT1 and PI3K/Akt signaling to improve the effects of exercise in elderly rat hearts. Age (Dordr). 2014; 36:9705. https://doi.org/10.1007/s11357-014-9705-5 [PubMed]
- 21. Zhang T, Berrocal JG, Frizzell KM, Gamble MJ, DuMond ME, Krishnakumar R, Yang T, Sauve AA, Kraus WL. Enzymes in the NAD+ salvage pathway regulate SIRT1 activity at target gene promoters. J Biol Chem. 2009; 284:20408–17. https://doi.org/10.1074/jbc.M109.016469 [PubMed]
- 22. Roh J, Rhee J, Chaudhari V, Rosenzweig A. The Role of Exercise in Cardiac Aging: From Physiology to Molecular Mechanisms. Circ Res. 2016; 118:279–95. https://doi.org/10.1161/CIRCRESAHA.115.305250 [PubMed]
- 23. Murawski U, Kriesten K, Egge H. Age-related changes of lipid fractions and total fatty acids in liver lipids and heart lipids of female and male rats aged 37-1200 days (liver) and 331-1200 days (heart). Comp Biochem Physiol B. 1990; 96:271–89. https://doi.org/10.1016/0305-0491(90)90375-4 [PubMed]
- 24. Zhang B. Effects of adopting exercise intervention for adolescents with simple obesity on heart rate, blood pressure and lipid metabolism. Basic Clin Pharmacol Toxicol. 2017; 121:7–7.
- 25. Nolte K, Schwarz S, Gelbrich G, Mensching S, Siegmund F, Wachter R, Hasenfuss G, Düngen HD, Herrmann-Lingen C, Halle M, Pieske B, Edelmann F. Effects of long-term endurance and resistance training on diastolic function, exercise capacity, and quality of life in asymptomatic diastolic dysfunction vs. heart failure with preserved ejection fraction. ESC Heart Fail. 2014; 1:59–74. https://doi.org/10.1002/ehf2.12007 [PubMed]
- 26. Sandri M, Kozarez I, Adams V, Mangner N, Höllriegel R, Erbs S, Linke A, Möbius-Winkler S, Thiery J, Kratzsch J, Teupser D, Mende M, Hambrecht R, et al. Age-related effects of exercise training on diastolic function in heart failure with reduced ejection fraction: the Leipzig Exercise Intervention in Chronic Heart Failure and Aging (LEICA) Diastolic Dysfunction Study. Eur Heart J. 2012; 33:1758–68. https://doi.org/10.1093/eurheartj/ehr469 [PubMed]
- 27. Byrne JA, Grieve DJ, Cave AC, Shah AM. Oxidative stress and heart failure. Arch Mal Coeur Vaiss. 2003; 96:214–21. [PubMed]
- 28. Ghorbanzadeh V, Mohammadi M, Mohaddes G, Dariushnejad H, Chodari L, Mohammadi S. Protective effect of crocin and voluntary exercise against oxidative stress in the heart of high-fat diet-induced type 2 diabetic rats. Physiol Int. 2016; 103:459–68. https://doi.org/10.1556/2060.103.2016.4.6 [PubMed]
- 29. Lund J, Hafstad AD, Boardman NT, Rossvoll L, Rolim NP, Ahmed MS, Florholmen G, Attramadal H, Wisløff U, Larsen TS, Aasum E. Exercise training promotes cardioprotection through oxygen-sparing action in high fat-fed mice. Am J Physiol Heart Circ Physiol. 2015; 308:H823–29. https://doi.org/10.1152/ajpheart.00734.2014 [PubMed]
- 30. Lai CH, Ho TJ, Kuo WW, Day CH, Pai PY, Chung LC, Liao PH, Lin FH, Wu ET, Huang CY. Exercise training enhanced SIRT1 longevity signaling replaces the IGF1 survival pathway to attenuate aging-induced rat heart apoptosis. Age (Dordr). 2014; 36:9706. https://doi.org/10.1007/s11357-014-9706-4 [PubMed]
- 31. Lowman KE, Wyatt BJ, Cunneely OP, Reed LK. The TreadWheel: Interval Training Protocol for Gently Induced Exercise in Drosophila melanogaster. J Vis Exp. 2018; 136:57788. https://doi.org/10.3791/57788 [PubMed]
- 32. Piazza N, Gosangi B, Devilla S, Arking R, Wessells R. Exercise-training in young Drosophila melanogaster reduces age-related decline in mobility and cardiac performance. PLoS One. 2009; 4:e5886. https://doi.org/10.1371/journal.pone.0005886 [PubMed]
- 33. Sujkowski A, Wessells R. Using Drosophila to Understand Biochemical and Behavioral Responses to Exercise. Exerc Sport Sci Rev. 2018; 46:112–20. https://doi.org/10.1249/JES.0000000000000139 [PubMed]
- 34. Wen DT, Zheng L, Ni L, Wang H, Feng Y, Zhang M. The expression of CG9940 affects the adaptation of cardiac function, mobility, and lifespan to exercise in aging Drosophila. Exp Gerontol. 2016; 83:6–14. https://doi.org/10.1016/j.exger.2016.07.006 [PubMed]
- 35. Wen DT, Zheng L, Yang F, Li HZ, Hou WQ. Endurance exercise prevents high-fat-diet induced heart and mobility premature aging and dsir2 expression decline in aging Drosophila. Oncotarget. 2017; 9:7298–311. https://doi.org/10.18632/oncotarget.23292 [PubMed]
- 36. Zheng L, Feng Y, Wen DT, Wang H, Wu XS. Fatiguing exercise initiated later in life reduces incidence of fibrillation and improves sleep quality in Drosophila. Age (Dordr). 2015; 37:9816. https://doi.org/10.1007/s11357-015-9816-7 [PubMed]
- 37. Busson D, Pret AM. GAL4/UAS targeted gene expression for studying Drosophila Hedgehog signaling. Methods Mol Biol. 2007; 397:161–201. https://doi.org/10.1007/978-1-59745-516-9_13 [PubMed]
- 38. Ou H, Lei T. A novel strategy for conditional gene knockout based on ΦC31 integrase and Gal4/UAS system in Drosophila. IUBMB Life. 2013; 65:144–48. https://doi.org/10.1002/iub.1119 [PubMed]
- 39. Yin Y, Han W, Cao Y. Association between activities of SOD, MDA and Na+-K+-ATPase in peripheral blood of patients with acute myocardial infarction and the complication of varying degrees of arrhythmia. Hellenic J Cardiol. 2018. [Epub ahead of print]. https://doi.org/10.1016/j.hjc.2018.04.003 [PubMed]
- 40. Jing L, Wang Y, Zhao XM, Zhao B, Han JJ, Qin SC, Sun XJ. Cardioprotective Effect of Hydrogen-rich Saline on Isoproterenol-induced Myocardial Infarction in Rats. Heart Lung Circ. 2015; 24:602–10. https://doi.org/10.1016/j.hlc.2014.11.018 [PubMed]
- 41. Zhao YB, Wang YZ, Yue YH, Zhao WC, Feng GX. Variation of plasma levels of endothelin, calcitonin gene-related peptide, nitric oxide, and malondialdehyde in acute myocardial ischemia reperfusion injury in a rabbit model. Genet Mol Res. 2015; 14:5577–84. https://doi.org/10.4238/2015.May.25.9 [PubMed]
- 42. Cao P, Shen D, Zhong Y, Yuan GX. Effect of Catalpol on MDA, SOD and GSH-Px of Vascular Endothelial Cell in Aging Rats Induced by D-galactose. J Am Geriatr Soc. 2018; 66:S490–490.
- 43. Brown KA, Didion SP, Andresen JJ, Faraci FM. Mn-SOD deficient mice exhibit increased oxidative stress and vascular dysfunction with aging. FASEB J. 2005; 19:A201–201.
- 44. Duryee MJ, Klassen LW, Schaffert CS, Tuma DJ, Hunter CD, Garvin RP, Anderson DR, Thiele GM. Malondialdehyde-acetaldehyde adduct is the dominant epitope after MDA modification of proteins in atherosclerosis. Free Radic Biol Med. 2010; 49:1480–86. https://doi.org/10.1016/j.freeradbiomed.2010.08.001 [PubMed]
- 45. Chen H, Xu Y, Wang J, Zhao W, Ruan H. Baicalin ameliorates isoproterenol-induced acute myocardial infarction through iNOS, inflammation and oxidative stress in rat. Int J Clin Exp Pathol. 2015; 8:10139–47. [PubMed]
- 46. Cui X, Gong J, Han H, He L, Teng Y, Tetley T, Sinharay R, Chung KF, Islam T, Gilliland F, Grady S, Garshick E, Li Z, Zhang JJ. Relationship between free and total malondialdehyde, a well-established marker of oxidative stress, in various types of human biospecimens. J Thorac Dis. 2018; 10:3088–3097. https://doi.org/10.21037/jtd.2018.05.92 [PubMed]
- 47. Liu N, Chen J, Gao D, Li W, Zheng D. Astaxanthin attenuates contrast agent-induced acute kidney injury in vitro and in vivo via the regulation of SIRT1/FOXO3a expression. Int Urol Nephrol. 2018; 50:1171–80. https://doi.org/10.1007/s11255-018-1788-y [PubMed]
- 48. Klotz LO. Redox regulation of FOXO transcription factors - role of glutathione. Free Radic Biol Med. 2017; 108:S5–5. https://doi.org/10.1016/j.freeradbiomed.2017.04.045
- 49. Pamplona R, Portero-Otín M, Riba D, Ledo F, Gredilla R, Herrero A, Barja G. Heart fatty acid unsaturation and lipid peroxidation, and aging rate, are lower in the canary and the parakeet than in the mouse. Aging (Milano). 1999; 11:44–49. https://doi.org/10.1007/BF03399636 [PubMed]
- 50. Men TT, Thanh DN, Yamaguchi M, Suzuki T, Hattori G, Arii M, Huy NT, Kamei K. A Drosophila Model for Screening Antiobesity Agents. Biomed Res Int. 2016; 2016:6293163. https://doi.org/10.1155/2016/6293163 [PubMed]
- 51. Czuriga D, Paulus WJ, Czuriga I, Édes I, Papp Z, Borbély A. Cellular mechanisms for diastolic dysfunction in the human heart. Curr Pharm Biotechnol. 2012; 13:2532–38. https://doi.org/10.2174/1389201011208062532 [PubMed]
- 52. Burnett C, Valentini S, Cabreiro F, Goss M, Somogyvári M, Piper MD, Hoddinott M, Sutphin GL, Leko V, McElwee JJ, Vazquez-Manrique RP, Orfila AM, Ackerman D, et al. Absence of effects of Sir2 overexpression on lifespan in C. elegans and Drosophila. Nature. 2011; 477:482–85. https://doi.org/10.1038/nature10296 [PubMed]
- 53. Hoffmann J, Romey R, Fink C, Yong L, Roeder T. Overexpression of Sir2 in the adult fat body is sufficient to extend lifespan of male and female Drosophila. Aging (Albany NY). 2013; 5:315–27. https://doi.org/10.18632/aging.100553 [PubMed]
- 54. Fukuwatari T, Shibata K, Ishihara K, Fushiki T, Sugimoto E. Elevation of blood NAD level after moderate exercise in young women and mice. J Nutr Sci Vitaminol (Tokyo). 2001; 47:177–79. https://doi.org/10.3177/jnsv.47.177 [PubMed]
- 55. White AT, Schenk S. NAD(+)/NADH and skeletal muscle mitochondrial adaptations to exercise. Am J Physiol Endocrinol Metab. 2012; 303:E308–21. https://doi.org/10.1152/ajpendo.00054.2012 [PubMed]
- 56. Jayaram HN, Kusumanchi P, Yalowitz JA. NMNAT expression and its relation to NAD metabolism. Curr Med Chem. 2011; 18:1962–72. https://doi.org/10.2174/092986711795590138 [PubMed]
- 57. Naumenko SE, Belavin AS, Kim SF. [Blood oxygen transport function during ftorotan application in patients with ischemic heart disease and low cardiac output]. Anesteziol Reanimatol. 2003; 18:50–53. [PubMed]
- 58. Shlyk SV, Terent’ev VP, Mikashinovich ZI. [The oxygen-transport function of the blood and cell metabolism in patients with heart failure of different origins]. Ter Arkh. 1999; 71:78–80. [PubMed]
- 59. Lee G, Park JH. Hemolymph sugar homeostasis and starvation-induced hyperactivity affected by genetic manipulations of the adipokinetic hormone-encoding gene in Drosophila melanogaster. Genetics. 2004; 167:311–23. https://doi.org/10.1534/genetics.167.1.311 [PubMed]
- 60. Terashima J, Takaki K, Sakurai S, Bownes M. Nutritional status affects 20-hydroxyecdysone concentration and progression of oogenesis in Drosophila melanogaster. J Endocrinol. 2005; 187:69–79. https://doi.org/10.1677/joe.1.06220 [PubMed]
- 61. Ozturk N, Olgar Y, Er H, Kucuk M, Ozdemir S. Swimming exercise reverses aging-related contractile abnormalities of female heart by improving structural alterations. Cardiol J. 2017; 24:85–93. https://doi.org/10.5603/CJ.a2016.0069 [PubMed]
- 62. Agarwal R, Norton JM, Konty K, Zimmerman R, Glover M, Lekiachvili A, McGruder H, Malarcher A, Casper M, Mensah GA, Thorpe L. Overreporting of deaths from coronary heart disease in New York City hospitals, 2003. Prev Chronic Dis. 2010; 7:A47. [PubMed]
- 63. Arsenos P, Gatzoulis K, Manis G, Gialernios T, Dilaveris P, Tsiachris D, Archontakis S, Kartsagoulis E, Mytas D, Stefanadis C. Decreased scale-specific heart rate variability after multiresolution wavelet analysis predicts sudden cardiac death in heart failure patients. Int J Cardiol. 2012; 154:358–60. https://doi.org/10.1016/j.ijcard.2011.11.007 [PubMed]
- 64. Gonzalez-Loyola F, Abellana R, Verdú-Rotellar JM, Bustamante Rangel A, Clua-Espuny JL, Muñoz MA. Mortality in heart failure with atrial fibrillation: role of digoxin and diuretics. Eur J Clin Invest. 2018; 48:e13014. https://doi.org/10.1111/eci.13014 [PubMed]
- 65. Brunet A, Sweeney LB, Sturgill JF, Chua KF, Greer PL, Lin Y, Tran H, Ross SE, Mostoslavsky R, Cohen HY, Hu LS, Cheng HL, Jedrychowski MP, et al. Stress-dependent regulation of FOXO transcription factors by the SIRT1 deacetylase. Science. 2004; 303:2011–15. https://doi.org/10.1126/science.1094637 [PubMed]
- 66. Ye X, Li M, Hou T, Gao T, Zhu WG, Yang Y. Sirtuins in glucose and lipid metabolism. Oncotarget. 2017; 8:1845–59. https://doi.org/10.18632/oncotarget.12157 [PubMed]
- 67. Qiang L, Wang L, Kon N, Zhao W, Lee S, Zhang Y, Rosenbaum M, Zhao Y, Gu W, Farmer SR, Accili D. Brown remodeling of white adipose tissue by SirT1-dependent deacetylation of Pparγ. Cell. 2012; 150:620–32. https://doi.org/10.1016/j.cell.2012.06.027 [PubMed]
- 68. Palu RA, Thummel CS. Sir2 Acts through Hepatocyte Nuclear Factor 4 to maintain insulin Signaling and Metabolic Homeostasis in Drosophila. PLoS Genet. 2016; 12:e1005978. https://doi.org/10.1371/journal.pgen.1005978 [PubMed]
- 69. Slade JD, Staveley BE. Extended longevity and survivorship during amino-acid starvation in a Drosophila Sir2 mutant heterozygote. Genome. 2016; 59:311–18. https://doi.org/10.1139/gen-2015-0213 [PubMed]
- 70. Blice-Baum AC, Zambon AC, Kaushik G, Viswanathan MC, Engler AJ, Bodmer R, Cammarato A. Modest overexpression of FOXO maintains cardiac proteostasis and ameliorates age-associated functional decline. Aging Cell. 2017; 16:93–103. https://doi.org/10.1111/acel.12543 [PubMed]
- 71. Abdurrachim D, Ciapaite J, Wessels B, Nabben M, Luiken JJ, Nicolay K, Prompers JJ. Cardiac diastolic dysfunction in high-fat diet fed mice is associated with lipotoxicity without impairment of cardiac energetics in vivo. Biochim Biophys Acta. 2014; 1842:1525–37. https://doi.org/10.1016/j.bbalip.2014.07.016 [PubMed]
- 72. Choi YS, de Mattos AB, Shao D, Li T, Nabben M, Kim M, Wang W, Tian R, Kolwicz SC
Jr . Preservation of myocardial fatty acid oxidation prevents diastolic dysfunction in mice subjected to angiotensin II infusion. J Mol Cell Cardiol. 2016; 100:64–71. https://doi.org/10.1016/j.yjmcc.2016.09.001 [PubMed] - 73. Li L, Zhao L, Yi-Ming W, Yu YS, Xia CY, Duan JL, Su DF. Sirt1 hyperexpression in SHR heart related to left ventricular hypertrophy. Can J Physiol Pharmacol. 2009; 87:56–62. https://doi.org/10.1139/Y08-099 [PubMed]
- 74. Pillai JB, Gupta M, Rajamohan SB, Lang R, Raman J, Gupta MP. Poly(ADP-ribose) polymerase-1-deficient mice are protected from angiotensin II-induced cardiac hypertrophy. Am J Physiol Heart Circ Physiol. 2006; 291:H1545–53. https://doi.org/10.1152/ajpheart.01124.2005 [PubMed]
- 75. Horowitz JF, Klein S. Lipid metabolism during endurance exercise. Am J Clin Nutr. 2000 (2 Suppl); 72:558S–63S. https://doi.org/10.1093/ajcn/72.2.558S [PubMed]
- 76. Intlekofer KA, Cotman CW. Exercise counteracts declining hippocampal function in aging and Alzheimer’s disease. Neurobiol Dis. 2013; 57:47–55. https://doi.org/10.1016/j.nbd.2012.06.011 [PubMed]
- 77. Touati S, Meziri F, He Y, Montezano A, Rhian RT, Pascal L. Exercise reverses endothelial dysfunction, oxidative stress and inflammation in rats with high-fat diet-induced obesity. Fundam Clin Pharmacol. 2010; 24:37–37.
- 78. Tinkerhess MJ, Ginzberg S, Piazza N, Wessells RJ. Endurance Training Protocol and Longitudinal Performance Assays for Drosophila melanogaster. J Vis Exp. 2012; 61:3786. https://doi.org/10.3791/3786 [PubMed]
- 79. Fink M, Callol-Massot C, Chu A, Ruiz-Lozano P, Izpisua Belmonte JC, Giles W, Bodmer R, Ocorr K. A new method for detection and quantification of heartbeat parameters in Drosophila, zebrafish, and embryonic mouse hearts. Biotechniques. 2009; 46:101–13. https://doi.org/10.2144/000113078 [PubMed]
- 80. He Y, Jasper H. Studying aging in Drosophila. Methods. 2014; 68:129–33. https://doi.org/10.1016/j.ymeth.2014.04.008 [PubMed]